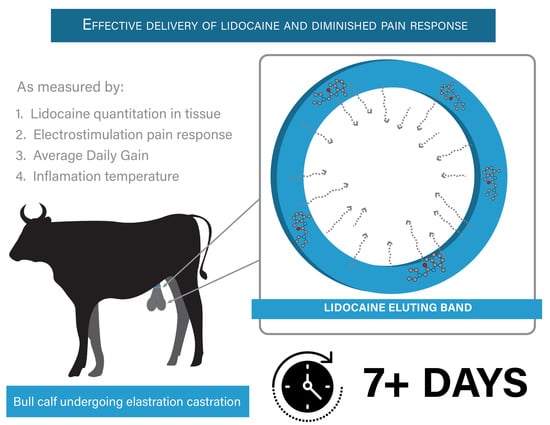
Development and Field Validation of Lidocaine-Loaded Castration Bands for Bovine Pain Mitigation
Abstract
Simple Summary
Abstract
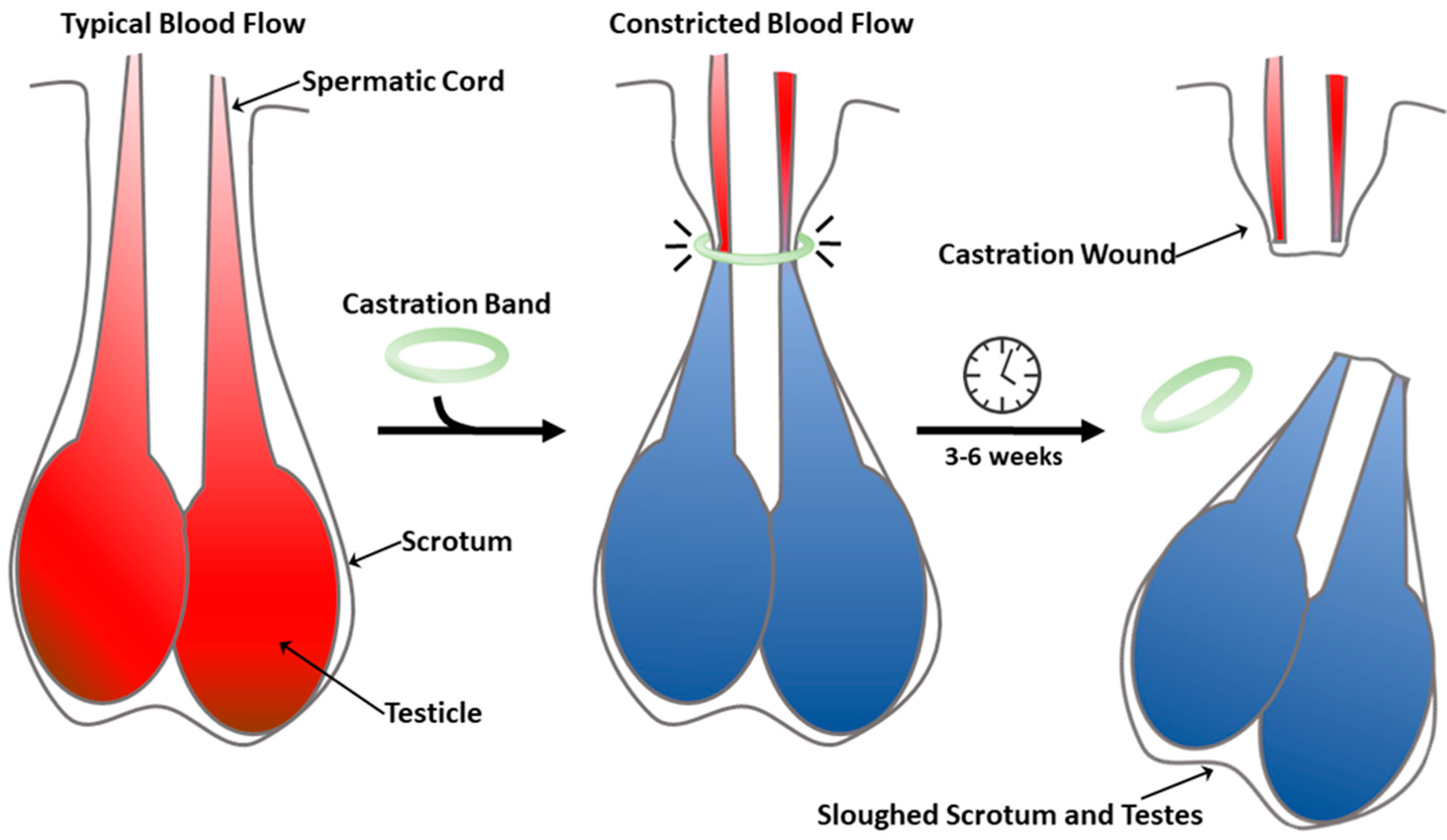
1. Introduction
2. Materials and Methods
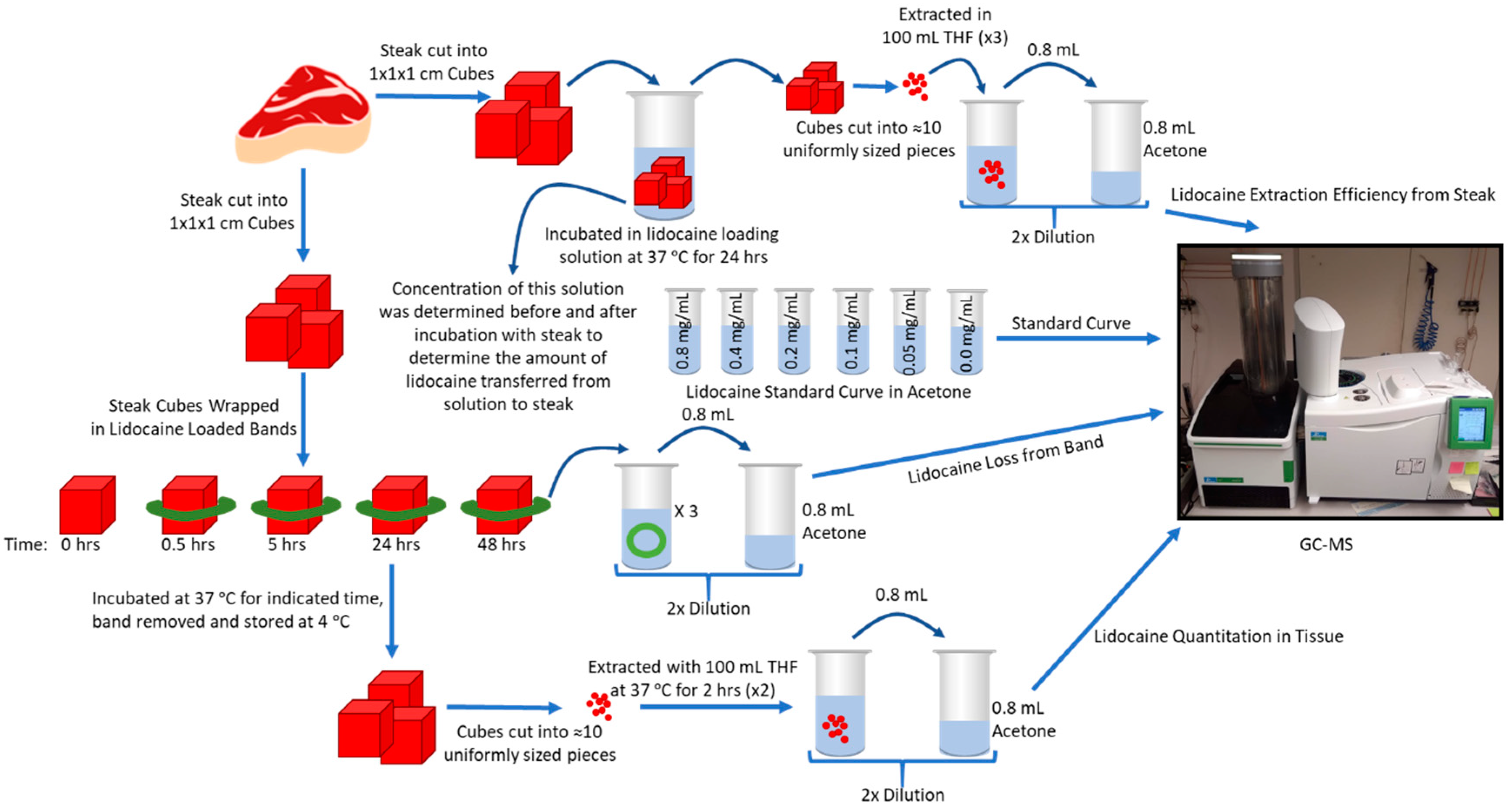
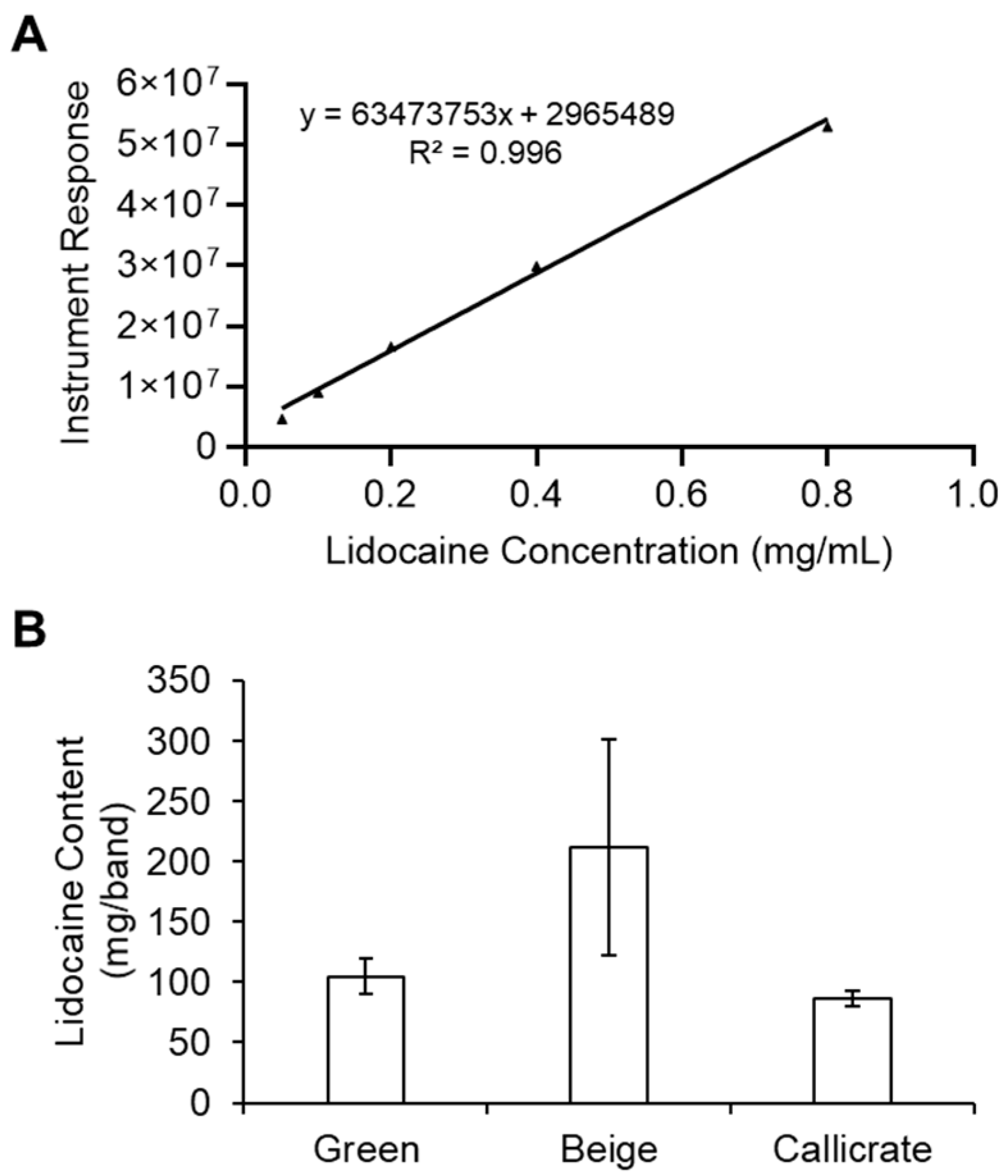
2.1. In Vitro Measurement of Initial Lidocaine Content in the Bands
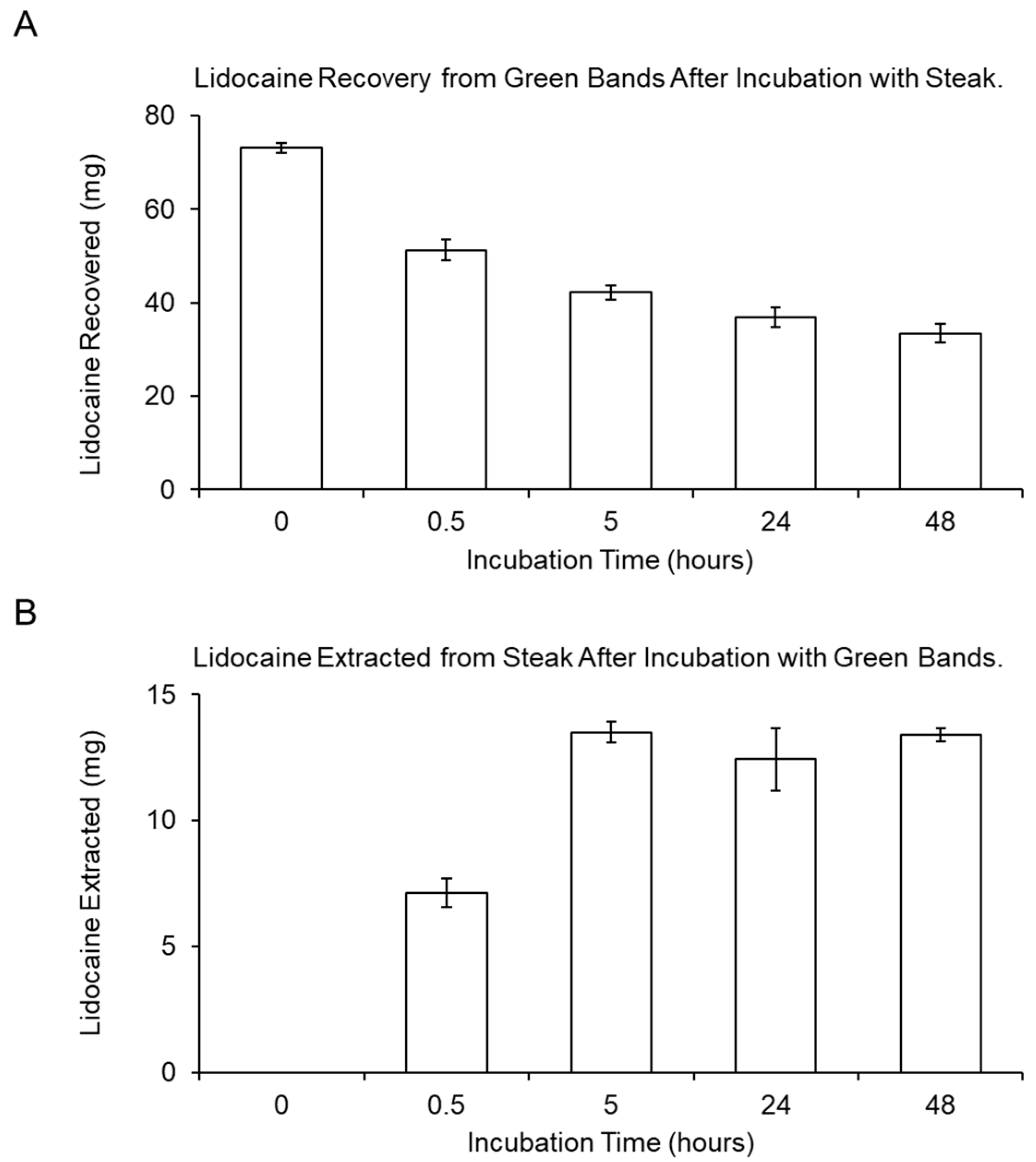
2.2. In Vitro Measurement of Lidocaine Transfer into Steak Tissue over Time
2.3. Field Study Overview
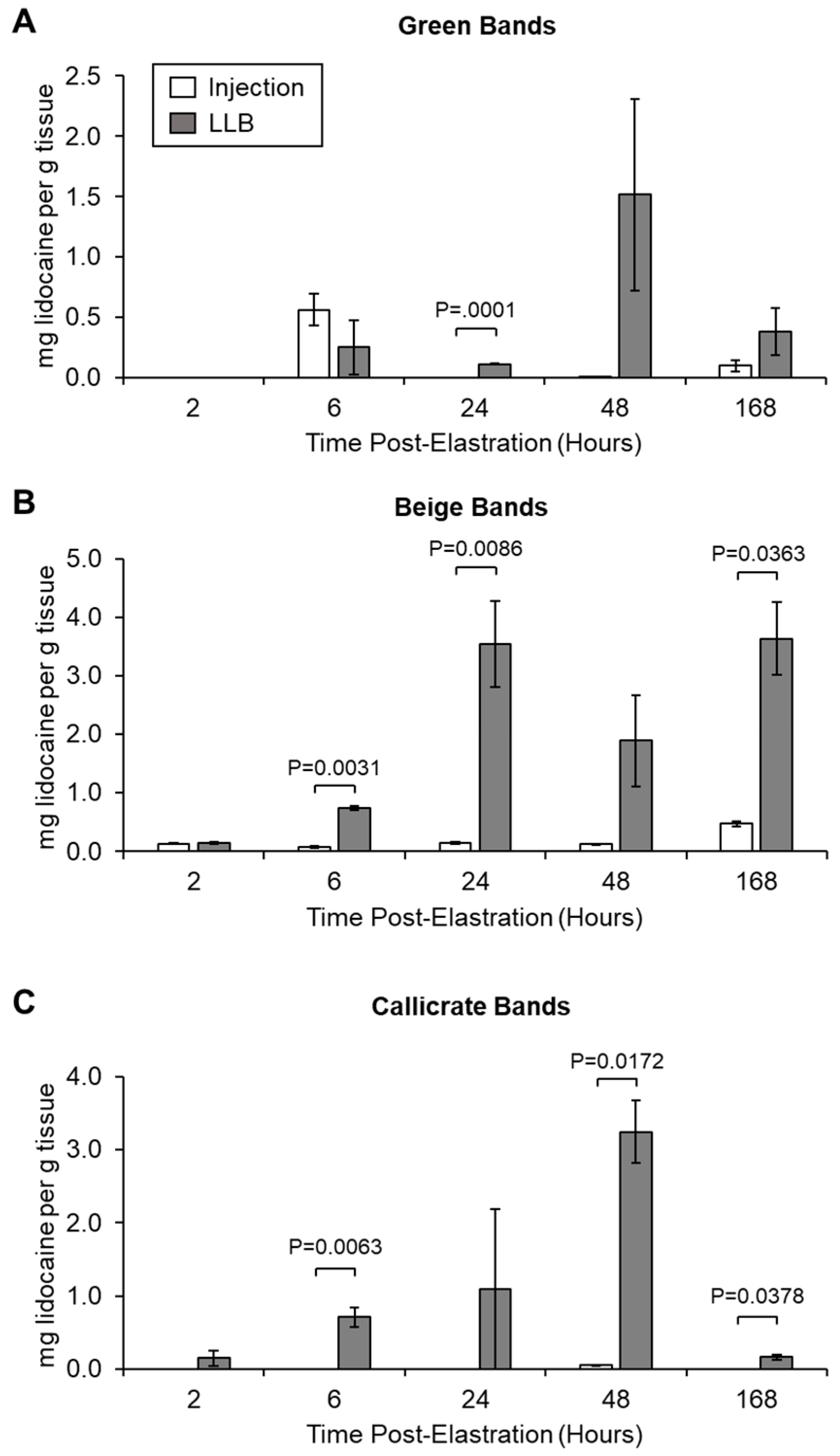
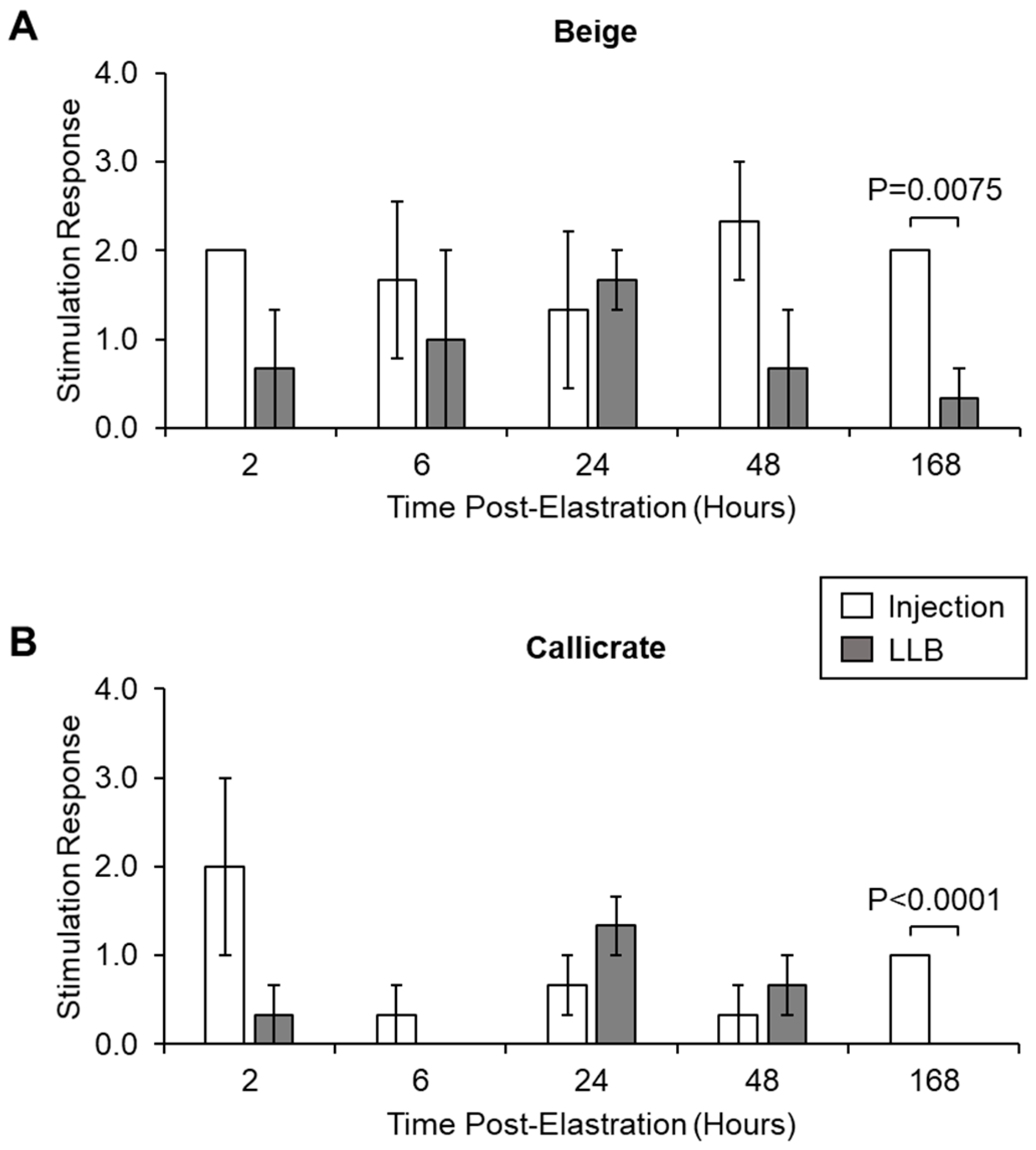
2.4. Measuring Lidocaine Release Kinetics and Pain-Associated Behaviour In Vivo
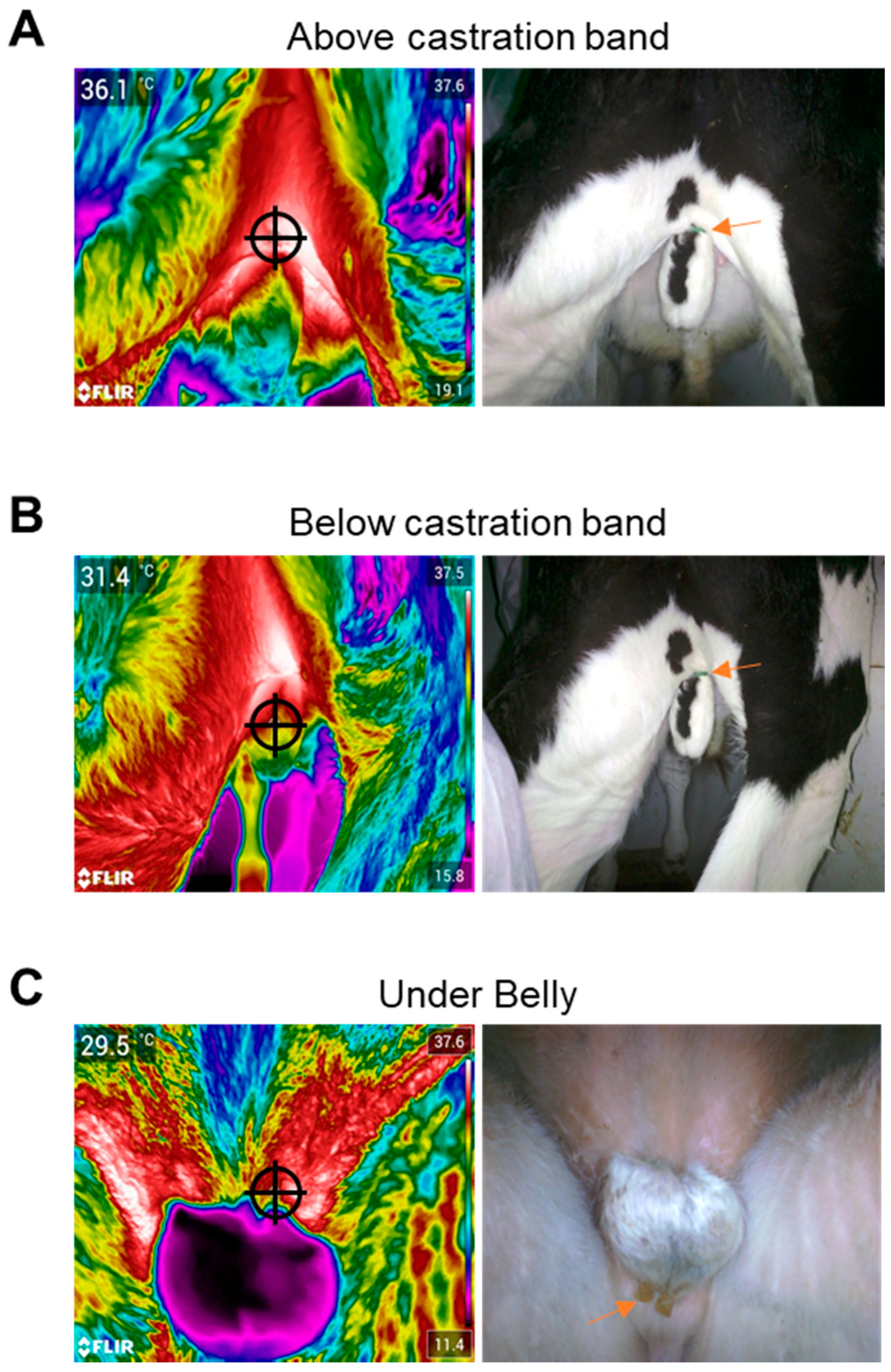
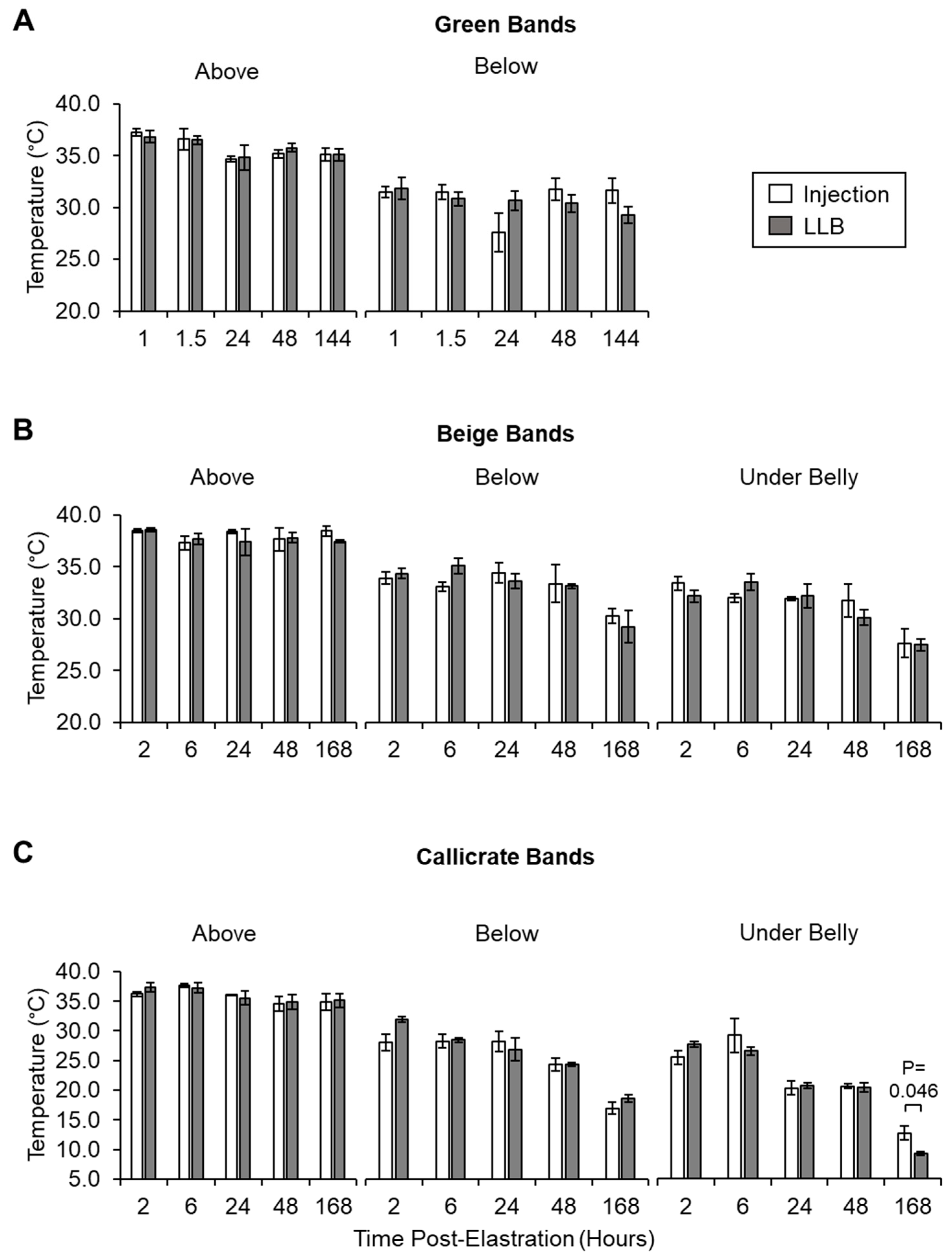
2.4.1. Infrared Imaging of Scrotal Temperature
2.4.2. Electrostimulation
2.5. Statistical Analyses
3. Results
3.1. Transfer Kinetics for Lidocaine-Loaded Castration Bands (LLBs) In Vitro
3.2. LLBs Deliver Therapeutic Quantities of Lidocaine into Scrotal Tissue In Vivo
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Coetzee, J.F.; Nutsch, A.L.; Barbur, L.A.; Bradburn, R.M. A survey of castration methods and associated livestock management practices performed by bovine veterinarians in the United States. BMC Vet. Res. 2010, 6, 12. [Google Scholar] [CrossRef] [PubMed]
- AVMA. Literature Review on the Welfare Implications of Castration of Cattle. Am. Vet. Med. Assoc. 2014. Available online: https://www.avma.org/sites/default/files/resources/castration-cattle-bgnd.pdf (accessed on 8 December 2020).
- Coetzee, J.F. A review of pain assessment techniques and pharmacological approaches to pain relief after bovine castration: Practical implications for cattle production within the United States. Appl. Anim. Behav. Sci. 2011, 135, 192–213. [Google Scholar] [CrossRef]
- Fajt, V.R.; Wagner, S.A.; Norby, B. Analgesic drug administration and attitudes about analgesia in cattle among bovine practitioners in the United States. J. Am. Vet. Med. Assoc. 2011, 238, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Melendez, D.M.; Marti, S.; Pajor, E.A.; Sidhu, P.K.; Gellatly, D.; Moya, D.; Janzen, E.D.; Coetzee, J.F.; Schwartzkopf-Genswein, K.S. Effect of meloxicam and lidocaine administered alone or in combination on indicators of pain and distress during and after knife castration in weaned beef calves. PLoS ONE 2018, 13, e0207289. [Google Scholar] [CrossRef] [PubMed]
- Olson, M.E.; Ralston, B.; Burwash, L.; Matheson-Bird, H.; Allan, N.D. Efficacy of oral meloxicam suspension for prevention of pain and inflammation following band and surgical castration in calves. BMC Vet. Res. 2016, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- NFACC. Code of Practice for the Care and Handling of Beef Cattle; National Farm Animal Care Council: Lacombe, AB, Canada, 2013; Available online: https://www.nfacc.ca/pdfs/codes/beef_code_of_practice.pdf (accessed on 8 December 2020).
- Anderson, N. Castration of Calves; Ontario Ministry of Agriculture, Food and Rural Affairs: Guelph, ON, Canada, 2015. Available online: http://www.omafra.gov.on.ca/english/livestock/beef/facts/07-029.htm (accessed on 8 December 2020).
- Carter, B.; Mathis, C.P.; Löest, C.; Wenzel, J. Castrating Beef Calves: Age and Method; New Mexico State University: College of Agricultural, Consumer, and Environmental Sciences: Las Cruces, NM, USA, 2011; Available online: https://aces.nmsu.edu/pubs/_b/B227.pdf (accessed on 8 December 2020).
- Molony, V.; Kent, J.E.; Robertson, I.S. Assessment of acute and chronic pain after different methods of castration of calves. Appl. Anim. Behav. Sci. 1995, 46, 33–48. [Google Scholar] [CrossRef]
- Golzari, S.E.; Soleimanpour, H.; Mahmoodpoor, A.; Safari, S.; Ala, A. Lidocaine and pain management in the emergency department: A review article. Anesth. Pain Med. 2014, 4, e15444. [Google Scholar] [CrossRef] [PubMed]
- Stafford, K.J.; Mellor, D.J.; Todd, S.E.; Bruce, R.A.; Ward, R.N. Effects of local anaesthesia or local anaesthesia plus a non-steroidal anti-inflammatory drug on the acute cortisol response of calves to five different methods of castration. Res. Vet. Sci. 2002, 73, 61–70. [Google Scholar] [CrossRef]
- Tennessen, T.; Connor, L.; Passillé, A.M.D.; Duncan, I.; Feddes, J.; Keaney, M.; Kochhar, H.; MacDonald, S.; Rushen, J.; Silversides, F.; et al. CCAC Guidelines on: The Care and Use of Farm Animals in Research, Teaching and Testing; Canadian Council on Animal Care: Ottawa, ON, Canada, 2009; Available online: https://www.ccac.ca/Documents/Standards/Guidelines/Farm_Animals.pdf (accessed on 8 December 2020).
- Fierheller, E.E.; Caulkett, N.A.; Haley, D.B.; Florence, D.; Doepel, L. Onset, duration and efficacy of four methods of local anesthesia of the horn bud in calves. Vet. Anaesth. Analg. 2012, 39, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Alsaaod, M.; Schaefer, A.L.; Büscher, W.; Steiner, A. The Role of Infrared Thermography as a Non-Invasive Tool for the Detection of Lameness in Cattle. Sensors 2015, 15, 14513–14525. [Google Scholar] [CrossRef] [PubMed]
- Purohit, R.C.; Hudson, R.S.; Riddell, M.G.; Carson, R.L.; Wolfe, D.F.; Walker, D.F. Thermography of the bovine scrotum. Am. J. Vet. Res. 1985, 46, 2388–2392. [Google Scholar] [PubMed]
- Padula, C.; Colombo, G.; Nicoli, S.; Catellani, P.L.; Massimo, G.; Santi, P. Bioadhesive film for the transdermal delivery of lidocaine: In vitro and in vivo behavior. J. Control. Release 2003, 88, 277–285. [Google Scholar] [CrossRef]
- Tanaka, E.; Yoshida, K.; Kawaai, H.; Yamazaki, S. Lidocaine Concentration in Oral Tissue by the Addition of Epinephrine. Anesth. Prog. 2016, 63, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Musk, G.C.; Jacobsen, S.; Hyndman, T.H.; Lehmann, H.S.; Tuke, S.J.; Collins, T.; Gleerup, K.B.; Johnson, C.B.; Laurence, M. Objective Measures for the Assessment of Post-Operative Pain in Bos indicus Bull Calves Following Castration. Animals 2017, 7, 76. [Google Scholar] [CrossRef] [PubMed]
- Canozzi, M.E.A.; Mederos, A.; Manteca, X.; Turner, S.; McManus, C.; Zago, D.; Barcellos, J.O.J. A meta-analysis of cortisol concentration, vocalization, and average daily gain associated with castration in beef cattle. Res. Vet. Sci. 2017, 114, 430–443. [Google Scholar] [CrossRef] [PubMed]
- Van der Saag, D.; Lomax, S.; Windsor, P.A.; Taylor, C.; Thomson, P.; Hall, E.; White, P.J. Effects of topical anaesthetic and buccal meloxicam on average daily gain, behaviour and inflammation of unweaned beef calves following surgical castration. Animal 2018, 12, 2373–2381. [Google Scholar] [CrossRef] [PubMed]
- Winder, C.B.; LeBlanc, S.J.; Haley, D.B.; Lissemore, K.D.; Godkin, M.A.; Duffield, T.F. Clinical trial of local anesthetic protocols for acute pain associated with caustic paste disbudding in dairy calves. J. Dairy Sci. 2017, 100, 6429–6441. [Google Scholar] [CrossRef] [PubMed]
- Bomberg, H.; Wetjen, L.; Wagenpfeil, S.; Schöpe, J.; Kessler, P.; Wulf, H.; Wiesmann, T.; Standl, T.; Gottschalk, A.; Döffert, J.; et al. Risks and Benefits of Ultrasound, Nerve Stimulation, and Their Combination for Guiding Peripheral Nerve Blocks: A Retrospective Registry Analysis. Anesth. Analg. 2018, 127, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Warren, V.T.; Fisher, A.G.; Rivera, E.M.; Saha, P.T.; Turner, B.; Reside, G.; Phillips, C.; White, R.P., Jr. Buffered 1% Lidocaine with Epinephrine Is as Effective as Non-Buffered 2% Lidocaine With Epinephrine for Mandibular Nerve Block. J. Oral Maxillofac. Surg. 2017, 75, 1363–1366. [Google Scholar] [CrossRef] [PubMed]
| Instrument or Parameter | Details |
|---|---|
| GC–MS | PerkinElmer Clarus 680-AxION iQT |
| Column | Elite-5MS, 30 m × 0.25 mm, 0.25 µm |
| Carrier Gas | Nitrogen |
| Analysis Time | 15 min |
| Flow Rate | 1 mL/min |
| Injection Volume | 0.5 µL |
| Injection Temperature | 250 °C |
| Split Ratio | 20:1 |
| Oven Program | 60 °C → 300 °C over 15 min |
| Detection | MS Scan 50—1045 m/z |
| GC–MS | PerkinElmer Clarus 680-AxION iQT |
| Column | Elite-5MS, 30 m × 0.25 mm, 0.25 µm |
| Instrument or Parameter | Details |
|---|---|
| HPLC | Hewlett Packard 1100 Series |
| Column | Kinetex 2.6 µm C18 (100A, 150 × 4.6 mm) |
| Mobile Phase | 40:60 (Acetonitrile: 0.05 M Sodium Phosphate Buffer Solution), 0.05% Diethylamine |
| Analysis Time | 6 min |
| Flow Rate | 1 mL/min |
| Injection Volume | 10 µL |
| Column Temperature | 28 °C |
| Detector | Variable Wavelength Detector (VWD) |
| Wavelength | 210 nm |
| Bandwidth | 10 nm |
| Average Animal Size at Banding | Band Type | Treatment Group | ADG a (Kg/Day) | ± SD b |
|---|---|---|---|---|
| 225 ± 20 Kg | CallicrateTM | Injection (n = 14) | 0.43 | 0.52 |
| LLB (n = 15) | 0.80 | 0.67 | ||
| 56 ± 8 Kg | Beige | Injection (n = 21) | 0.93 | 0.33 |
| LLB (n = 20) | 0.99 | 0.23 | ||
| 53 ± 4 Kg | Green | Injection (n = 3) | 0.73 | 0.27 |
| LLB (n = 3) | 0.89 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saville, J.W.; Ross, J.A.; Trefz, T.; Schatz, C.; Matheson-Bird, H.; Ralston, B.; Granot, O.; Schmid, K.; Terry, R.; Allan, N.D.; et al. Development and Field Validation of Lidocaine-Loaded Castration Bands for Bovine Pain Mitigation. Animals 2020, 10, 2363. https://doi.org/10.3390/ani10122363
Saville JW, Ross JA, Trefz T, Schatz C, Matheson-Bird H, Ralston B, Granot O, Schmid K, Terry R, Allan ND, et al. Development and Field Validation of Lidocaine-Loaded Castration Bands for Bovine Pain Mitigation. Animals. 2020; 10(12):2363. https://doi.org/10.3390/ani10122363
Chicago/Turabian StyleSaville, James W., Joseph A. Ross, Tyler Trefz, Crystal Schatz, Heather Matheson-Bird, Brenda Ralston, Ori Granot, Karin Schmid, Richard Terry, Nicholas D. Allan, and et al. 2020. "Development and Field Validation of Lidocaine-Loaded Castration Bands for Bovine Pain Mitigation" Animals 10, no. 12: 2363. https://doi.org/10.3390/ani10122363
APA StyleSaville, J. W., Ross, J. A., Trefz, T., Schatz, C., Matheson-Bird, H., Ralston, B., Granot, O., Schmid, K., Terry, R., Allan, N. D., Wulff, J. E., & Olson, M. (2020). Development and Field Validation of Lidocaine-Loaded Castration Bands for Bovine Pain Mitigation. Animals, 10(12), 2363. https://doi.org/10.3390/ani10122363