Strategies for Feeding Unweaned Dairy Beef Cattle to Improve Their Health
Abstract
:Simple Summary
Abstract
1. Introduction
2. The Dairy Beef Calves
3. Nutrition of Unweaned Calves’ Previous to the Arrival to the Rearing Farm
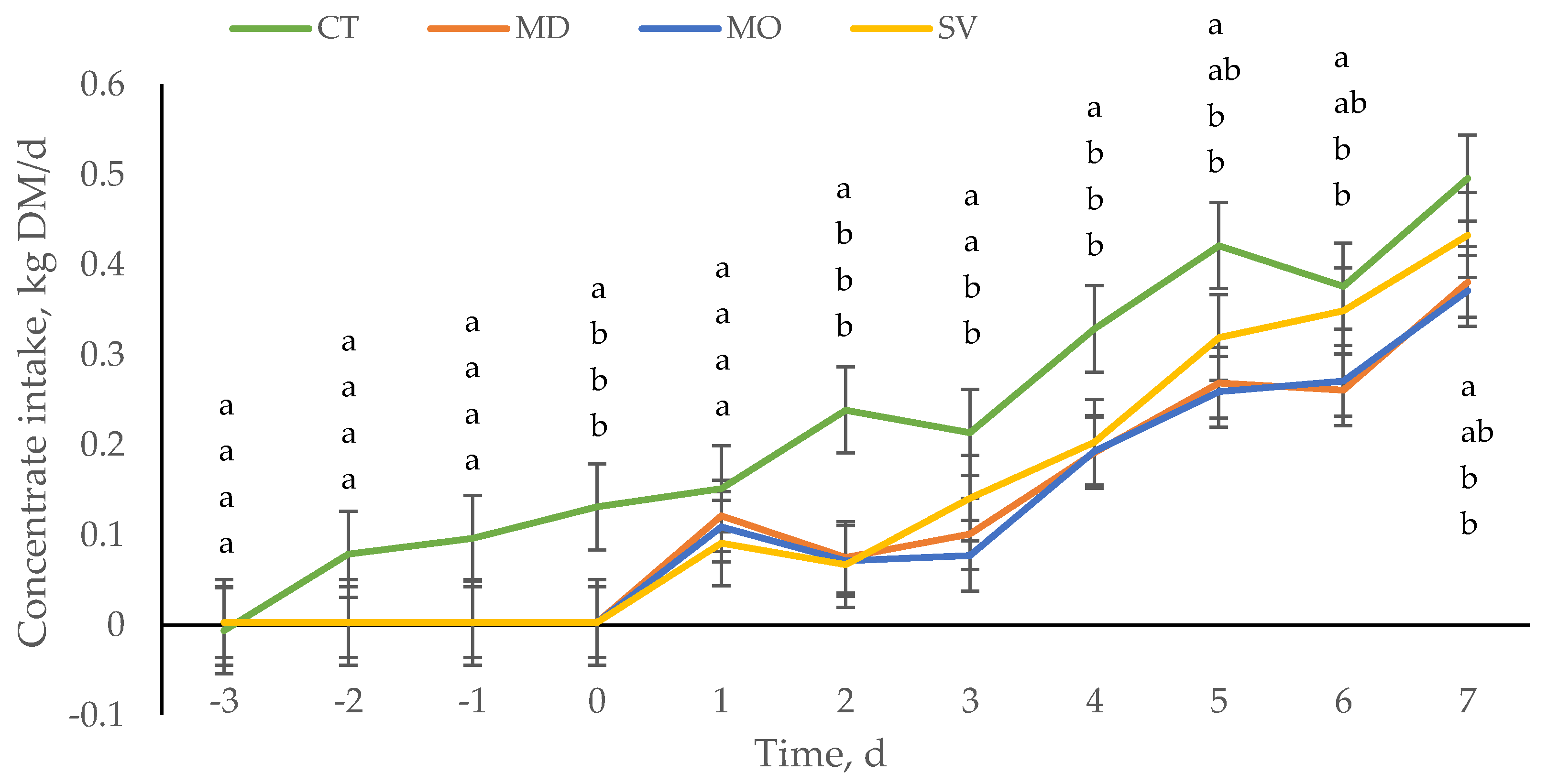
3.1. Nutrition before They Are Collected and Arrive to the Auction Market or Concentration Center
3.2. Nutrition during the Transition Period: From the Farm where the Animal Is Born and Collected to the Final Rearing Farm (Transport—Auction Market or Concentration Center—Transport)
4. Consequences of Previous Nutrition on Gut Health and Immune Status at Arrival to the Rearing Farm
5. Considerations to Advance in Future Nutritional Strategies
5.1. Considerations for Research to Prevent through Nutritional Strategies BRD
5.2. Considerations for Research to Recover Animal Gut and Immune System Functionality through Nutritional Strategies to Prevent BRD at Arrival to the Rearing Farm
5.3. Other Important Considerations for Research Related with the Nutritional Strategies to Prevent BRD
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, R.A. Impact of disease on feedlot performance: A review. J. Anim. Sci. 1998, 76, 272–274. [Google Scholar] [CrossRef] [PubMed]
- Nagaraja, T.G.; Titgemeyer, E.C. Ruminal acidosis in beef cattle: The current microbiological and nutritional outlook. J. Dairy Sci. 2007, 90, E17–E38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González, L.A.; Manteca, X.; Calsamiglia, S.; Schwartzkopf-Genswein, K.S.; Ferret, A. Ruminal acidosis in feedlot cattle: Interplay between feed ingredients, rumen function and feeding behavior (a review). Anim. Feed Sci. Technol. 2012, 172, 66–79. [Google Scholar] [CrossRef]
- Aschenbach, J.R.; Zebeli, Q.; Patra, A.K.; Greco, G.; Amasheh, S.; Penner, G.B. Symposium review: The importance of the ruminal epithelial barrier for a healthy and productive cow. J. Dairy Sci. 2019, 102, 1866–1882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duff, G.C.; Galyean, M.L. Board-invited review: Recent advances in management of highly stressed, newly received feedlot cattle. J. Anim. Sci. 2007, 85, 823–840. [Google Scholar] [CrossRef]
- Early, B.; Buckham Sporer, K.; Gupta, S. Invited review: Relationship between cattle transport, immunity, and respiratory disease. Animal 2017, 11, 486–492. [Google Scholar] [CrossRef]
- Edwards, T.A. Control Methods for Bovine Respiratory Disease for Feedlot Cattle. Vet. Clin. N. Am. Food Anim. Pract. 2010, 26, 273–284. [Google Scholar] [CrossRef]
- Blakebrough-Hall, C.; McNeniman, J.P.; González, L.A. An evaluation of the economic effects of bovine respiratory disease on animal performance, carcass traits, and economic outputs in feedlot cattle defined using four BRD diagnosis methods. J. Anim. Sci. 2020, 98, skaa005. [Google Scholar] [CrossRef]
- ASAS Editorial Infographic. Preview: Economic effects of bovine respiratory disease. J. Anim. Sci. 2020, 98, skaa042. [Google Scholar] [CrossRef]
- Pardon, B. Morbidity, Mortality and Drug Use in White Veal Calves with Emphasis on Respiratory Disease. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2012. [Google Scholar]
- Timsit, E.; Bareille, N.; Seegers, H.; Lehebel, A.; Assié, S. Visually undetected fever episodes in newly received beef bulls at a fattening operation: Occurance, duration, and impact on performance. J. Anim. Sci. 2011, 89, 4272–4280. [Google Scholar] [CrossRef] [Green Version]
- Edwards, T.A. Respiratory diseases of feedlot cattle in Central USA. Bov. Pract. 1996, 30, 5–7. [Google Scholar]
- Tejero, C.; Pujols, J.; Devant, M.; Marti, S. Evolution and risk factors of pulmonary lesions evaluated with thoracic ultrasonography in pre-weaned beef calves at arrival at the rearing facilities. J. Anim. Sci. 2019, 97, 208–209. [Google Scholar] [CrossRef]
- Baptiste, K.E.; Kysvsgaard, N.C. Do antimicrobial mass medications work? A systemic review and meta-analysis of randomized clinical trials investigating antimicrobial prophylaxis or metaphlyaxis against naturally occurring bovine respiratory disease. Pathog. Dis. 2017, 75, ftx083. [Google Scholar] [CrossRef] [PubMed]
- David, V.; Bleaugrand, F.; Gay, E.; Bastien, J.; Ducrot, C. Évolution de l’usage des antibiotiques en filières bovines: État d’avancement et perspectives. INRAE Prod. Anim. 2019, 32, 291–304. [Google Scholar] [CrossRef]
- Cameron, A.; McAllister, T. Antimicrobial usage and resistance in beef production. J. Anim. Sci. Biotech. 2016, 7, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Migura-Garcia, L.; Marti, S.; Aris, A.; Perez de Rozas, A.; Tejero, C.; Torra, J.; Pujols, J.; Devant, M. Antimicrobial resistance profiles of Pasteurella multocida, Mannheimia haemolytica and Moraxella spp. of bovine origin pre- and post-treatment with antimicrobials in calves. J. Anim. Sci. 2019, 97 (Suppl. 3), 212. [Google Scholar] [CrossRef]
- Budden, K.F.; Gellatly, S.L.; Wood, D.L.A.; Cooper, M.A.; Morrison, M.; Hugenholtz, P.; Hansbro, P.M. Emerging pathogenic links between microbiota and the gut-lung axis. Nat. Rev. Microbiol. 2017, 15, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Enaud, R.; Prevel, R.; Ciarlo, E.; Beaufils, F.; Wieërs, G.; Guery, B.; Delhaes, L. The gut-lung axis in health and respiratory diseases: A place for inter-organ and inter-kingdom crosstalks. Front. Cell. Infect. Microbiol. 2020, 10, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Engen, N.K.; Coetzee, J.F. Effects of transportation on cattle health and production: A review. Anim. Health Res. Rev. 2018, 19, 142–154. [Google Scholar] [CrossRef]
- Wilson, B.K.; Richards, C.J.; Step, D.L.; Krehbiel, C.R. Best management practices for newly weaned calves for improved health and well-being. J. Anim. Sci. 2017, 95, 2170–2182. [Google Scholar] [CrossRef]
- Eurostat. Agriculture, Forestry and Fishery Statistics; European Comission, European Union: Brussels, Belgium, 2019; ISBN 978-92-76-13194-6. [Google Scholar]
- EU Trade and Transport EU Trade and Transport of Live Animals. Available online: https://www.europarl.europa.eu/RegData/etudes/ATAG/2020/646170/EPRS_ATA(2020)646170_EN.pdf (accessed on 3 September 2020).
- Britt, J.H.; Cushman, R.A.; Dechow, C.D.; Dobson, H.; Humblot, P.; Huthens, M.F.; Jones, G.A.; Ruegg, P.S.; Sheldon, I.M.; Stevenson, J.S. Invited review: Learning from the future- a vision for dairy farms and cows in 2067. J. Dairy Sci. 2017, 101, 3722–3741. [Google Scholar] [CrossRef] [Green Version]
- Opio, C.; Gerber, P.; Mottet, A.; Falcucci, A.; Tempio, G.; MacLeod, M.; Vellinga, T.; Henderson, B.; Steinfeld, H. Greenhouse Gas Emissions from Ruminant Supply Chains—A Global Life Cycle Assessment; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013; ISBN 978-92-5-107945-4. [Google Scholar]
- Knowles, T.G.; Warriss, P.D.; Brown, S.N.; Edwards, J.E.; Watkins, P.E.; Phillips, A.J. Effects on calves less than one month old of feeding or not feeding them during road transport of up to 24 hours. Vet. Rec. 1997, 140, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Burciaga-Robles, L.O. Neonatal and young (<205 kg) feeding programs in calf-fed Holsteins. In Proceedings of the ADSA-ASAS Joint Annual Meeting 2015, Orlando, FL, USA, 14 July 2015. Abstract 516. [Google Scholar]
- Renaud, D.L.; Duffield, T.F.; LeBlanc, S.J.; Haley, D.B.; Kelton, D.F. Management practices for male calves on Canadian dairy farms. J. Dairy Sci. 2017, 100, 6862–6871. [Google Scholar] [CrossRef]
- Hulbert, L.E.; Moisá, S.J. Stress, immunity, and the management of calves. J. Dairy Sci. 2016, 99, 3199–3216. [Google Scholar] [CrossRef] [Green Version]
- Odle, J.; Zijlstra, R.T.; Donovan, S.M. Intestinal effects of milkborne growth factors in neonates of agricultural importance. J. Anim. Sci. 1996, 74, 2509–2522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blum, J.W. Nutritional physiology of neonatal calves. J. Anim. Physiol. Anim. Nutr. 2006, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Hill, C. Milk intelligence: Mining milk for bioactive substances associated with human health. Int. Dairy J. 2011, 21, 377–401. [Google Scholar] [CrossRef]
- Baumrucker, C.; Hadsell, D.L. Effects of dietary insulin-like growth factor-1 on growth and insulin-like growth factor receptors in neonatal calf intestine. J. Anim. Sci. 1994, 72, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Blättler, U.; Hammon, H.M.; Morel, C.; Philipona, C.; Rauprich, A.; Romé, V.; Huërou-Luron, I.L.; Guilloteau, P.; Blum, J.W. Feeding colostrum, its composition and feeding duration variably modify proliferation and morphology of the intestine and digestive enzyme activities of neonatal calves. J. Nutr. 2001, 131, 1256–1263. [Google Scholar] [CrossRef]
- Ballou, M.A.; Hanson, D.L.; Cobb, C.J.; Obeidat, B.S.; Sellers, M.D.; Pepper-Yowell, A.R.; Carroll, J.A.; Earleywine, T.J.; Lawhon, S.D. Plane of nutrition influences the performance, innate leukocyte responses, and resistance to an oral Salmonella enterica serotype Typhimurium challenge in Jersey calves. J. Dairy Sci. 2015, 98, 1972–1982. [Google Scholar] [CrossRef]
- Lorenz, I.; Earley, B.; Gilmore, J.; Hogan, I.; Kennedy, E.; More, S.J. Calf health from birth to weaning. III. Housing and management of calf pneumonia. Irish Vet. J. 2011, 64, 9–14. [Google Scholar] [CrossRef] [Green Version]
- Tao, S.; Dahl, G.E. Invited review: Heat stress effects during late gestation on dry cows and their calves. J. Dairy Sci. 2013, 96, 4079–4093. [Google Scholar] [CrossRef]
- Renaud, D.L.; Waalderbos, K.M.; Beavers, L.; Duffield, T.F.; Leslie, K.E.; Windeyer, M.C. Risk factors associated with failed transfer of passive immunity in male and female dairy calves: A 2008 retrospective cross-sectional study. J. Dairy Sci. 2020, 103, 3521–3528. [Google Scholar] [CrossRef]
- Sutter, F.; Rauch, E.; Ehard, M.; Sargent, R.; Weber, C.; Heuwieser, W.; Borchardt, S. Evaluation of different analytical methods to assess failure of passive transfer in neonatal calves. J. Dairy Sci. 2020, 103, 5387–5397. [Google Scholar] [CrossRef] [PubMed]
- Kampen, A.H.; Olsen, I.; Tollersrud, T.; Storset, A.K.; Lund, A. Lymphocyte subpopulations and neutrophil function in calves during the first 6 months of life. Vet. Immunol. Immunopathol. 2006, 113, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Palczynski, L.J.; Bleach, E.C.L.; Brennan, M.L.; Robinson, P.A. Appropiate dairy calf feeding from birth to weaning: “It’s an investment for the future”. Animals 2020, 10, 116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kahn, M.A.; Bach, A.; Weary, D.M.; von Keyserlingk, M.A.G. Invited review: Transitioning from milk to solid feed in dairy heifers. J. Dairy Sci. 2016, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kertz, A.F.; Hill, T.M.; Quigley, J.D., III; Heindrichs, A.J.; Linn, J.G.; Drackley, J.K. A 100-Year review: Calf nutrition and management. J. Dairy Sci. 2017, 100, 10151–10172. [Google Scholar] [CrossRef] [PubMed]
- Meale, S.J.; Chaucheyras-Durand, F.; Berends, H.; Guan, L.L.; Steele, M. From pre- to postweaning: Transformation of the young calf’s gastrointestinal tract. J. Dairy Sci. 2017, 100, 5984–5995. [Google Scholar] [CrossRef]
- Heinrichs, A.J.; Jones, C.M.; Erickson, P.S.; Chester-Jones, H.; Anderson, J.L. Symposium review: Colostrum management and calf nutrition for profitable and sustainable dairy farms. J. Dairy Sci. 2020, 103, 5694–5699. [Google Scholar] [CrossRef]
- Marcato, F.; van den Brand, H.; Kemp, B.; Engel, B.; Wolthuis-Fillerup, M.; van Reenen, K. Effects of pretransport diet, transport duration, and type of vehicle on physiological status of young veal calves. J. Dairy Sci. 2020, 103, 3505–3520. [Google Scholar] [CrossRef] [Green Version]
- Marquou, S.; Blouin, L.; Djakite, H.; Laplante, R.; Buczinski, S. Health parameters and their association with price in young calves sold at auction market for veal operations in Québec, Canada. J. Dairy Sci. 2019, 102, 6454–6465. [Google Scholar] [CrossRef] [Green Version]
- Winder, C.B.; Kelton, D.F.; Duffield, T.F. Mortality risk factors for calves entering a multi-location white veal farm in Ontario, Canada. J. Dairy Sci. 2016, 99, 10174–10181. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.J.; Stojkov, J.; Renaud, D.L.; Fraser, D. Short-communication: Condition of male dairy calves at auction markets. J. Dairy Sci. 2020, 103, 8530–8534. [Google Scholar] [CrossRef] [PubMed]
- Bernardini, D.; Gerardi, G.; Peli, A.; Nanni Costa, L.; Amadori, M.; Segato, S. The effects of different enviromental conditions on thermoregulation and clinical and hematological variables in long-distance road-transported calves. J. Anim. Sci. 2012, 90, 1183–1191. [Google Scholar] [CrossRef]
- Verdú, M.; Solé, A.; Devant, M. The influence of age at arrival in the design of milk-feeding program on concentrate consumption and performance in milk-fed Holstein bull calves from auction markets. J. Anim. Sci. 2018, 96, 430. [Google Scholar] [CrossRef]
- Zhang, S.; Albornoz, R.I.; Aschenbach, J.R.; Barreda, D.R.; Penner, G.B. Short-term feed restriction impairs the absorptive function of the reticulo-rumen and total tract barrier function in beef cattle. J. Anim. Sci. 2013, 91, 1685–1695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marti, S.; Velarde, A.; Quintana, B.; Moles, X.; Sole, A.; Devant, M. Evaluation of different types of journeys on welfare indicators of un-weaned calves transported by road in Europe. J. Anim. Sci. 2019, 97, 10–11. [Google Scholar] [CrossRef]
- Pisoni, L.; Devant, M.; Marti, S. Simulation of feed restriction and anorexia suffered in auction market and transportation and its effect on performance and gut permeability in unweaned Angus-Holstein calves. J. Dairy Sci. 2020, under preparation—unpublished. [Google Scholar]
- Kvidera, S.K.; Horst, E.A.; Sanz Fernandez, M.V.; Abuajamieh, M.; Ganesan, S.; Gorden, P.J.; Green, H.B.; Schoenberg, K.M.; Trout, W.E.; Keating, A.F.; et al. Characterizing effects of feed restriction and glucagon-like peptide 2 administration on biomarkers of inflammation and intestinal morphology. J. Dairy Sci. 2017, 100, 9402–9417. [Google Scholar] [CrossRef] [Green Version]
- Keogh, K.; Waters, S.M.; Cormican, P.; Kelly, A.K.; Kenny, D.A. Effect of dietary restriction and subsequent re-alimentation on the transcriptional profile of bovine jejunal epithelium. PLoS ONE 2018, 13, e0194445. [Google Scholar] [CrossRef]
- Lambert, G.P. Stress-induced gastrointestinal barrier dysfunction and its inflammatory effects. J. Anim. Sci. 2009, 87, E101–E108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wood, K.M.; Palmer, S.I.; Steele, M.A.; Metcalf, J.A.; Penner, G.B. The influence of age and weaning on permeability of the gastrointestinal tract in Holstein bull calves. J. Dairy Sci. 2015, 98, 7226–7237. [Google Scholar] [CrossRef]
- Bischoff, S.; Barbara, G.; Buurman, W.; Ockhiizen, T.; Schulze, J.D.; Serino, M.; Tilg, H.; Watson, A.; Wells, J.M. Intestinal permeability-a new target for disease prevention and therapy. BMC Gastroenterol. 2014, 14, 189. [Google Scholar] [CrossRef] [Green Version]
- Fragkos, K.; Forbes, A. Citrulline as a marker of intestinal function and absorption in clinical settings: A systemic review and meta-analysis. United Eur. Gastroenterol. J. 2018, 6, 181–191. [Google Scholar] [CrossRef]
- Robles, J.A.; Marti, S.; Pisoni, L.; Bassols, A.M.; Devant, M. Serum citrulline concentration of unweaned calves transported by road as a potential biomarker of gut functionality. J. Anim. Sci. 2020, in press. [Google Scholar]
- Kvidera, S.K.; Horst, E.A.; Abuajamieh, M.; Mayorga, E.J.; Sanz Fernandez, M.V.; Baumgard, L.H. Technical note: A procedure to estimate glucose requirements of an activated immune system in steers. J. Anim. Sci. 2016, 94, 4591–4599. [Google Scholar] [CrossRef]
- Bouvier-Muller, J.; Allain, C.; Tabouret, G.; Enjalbert, F.; Portes, D.; Noirot, C.; Rupp, R.; Fourcras, G. Whole blood transcriptome analysis reveals potential competition in metabolic pathways between negative energy balance and response to inflammatory challenge. Sci. Rep. 2017, 7, 2379. [Google Scholar] [CrossRef] [Green Version]
- Pregel, P.; Bollo, E.; Cannizzo, F.T.; Biolatti, B.; Contato, E.; Biolatti, P.G. Antioxidant capacity as reliable marker of stress in dairy calves transported by road. Vet. Rec. 2005, 156, 53–54. [Google Scholar] [CrossRef] [PubMed]
- Urban-Chmiel, R.; Wernicki, A.; Puchalski, A.; Dec, M. The effect of α-tocopherol and ascorbic acid supplementation on selected immunological parameters in bovine leukocytes in vitro. Acta Vet. Brno 2009, 78, 589–594. [Google Scholar] [CrossRef]
- Gutterridge, J.M.C.; Halliwell, B. Mini-review: Oxidative stress, redox stress or redox success? Biochem. Biophys. Res. Commun. 2018, 502, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Stefanon, B.; Gabai, G.; Gelain, M.E.; Bonsembiante, F. Oxidative stress and nutraceuticals in the current modulation of the immune function: Current knowledge in animal of veterinary interest. Antioxidants 2019, 8, 28. [Google Scholar] [CrossRef] [Green Version]
- Carroll, J.A.; Forsberg, N.E. Influence of stress and nutrition on cattle immunity. Vet. Clin. Food. Anim. 2007, 23, 105–149. [Google Scholar] [CrossRef]
- Hutcheson, D.P.; Andy Cole, N. Management of transit-stress syndrome in cattle: Nutritional and environmental effects. J. Anim. Sci. 1986, 62, 555–560. [Google Scholar] [CrossRef]
- Pisoni, L.; Devant, M.; Marti, S. Simulation of feed restriction and anorexia during auction market and transportation and its effects on body weight recovery and concentrate intake on unweaned Angus-Holstein calves. J. Anim. Sci. 2020, in press; Issue Supplement. [Google Scholar]
- Steinhardt, M.; Thielscher, H.H. The effect of haemoglobin content of blood on the reactionsof suckler calves exposed to shout haul road transport and temporary separation form herd mates. Tieraertztl Umschau 2005, 60, 356. [Google Scholar]
- Joerling, J.; Doll, K. Monitoring of iron deficiency in calves by determination of serum ferritin in comparison with serum iron: A preliminary study. Open Vet. J. 2019, 9, 177–184. [Google Scholar] [CrossRef] [Green Version]
- Bami, M.H.; Mohri, M.; Seifi, H.A.; Tabatabaee, A.A.A. Effects of parenteral supply of iron and copper on hematology, weight gain, and health in neonatal dairy calves. Vet. Res. Commun. 2008, 32, 553–561. [Google Scholar] [CrossRef]
- EFSA Panel on Animal Health and Welfare (AHAW). Scientific Opinion on the welfare of cattle kept for beef production and the welfare in intensive calf farming systems. EFSA J. 2012, 10, 2669. [Google Scholar] [CrossRef] [PubMed]
- Oppenheimer, S.J. Iron and its relation to immunity and infectious disease. J. Nutr. 2001, 131, 616S–635S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Celi, P.; Cowieson, A.J.; Fru-Nji, F.; Steinert, R.E.; Kluenter, A.-M.; Verlhac, V. Gastrointestinal functionality in animal nutrition and health: New opportunities for sustainable animal production. Anim. Feed Sci. Technol. 2017, 234, 88–100. [Google Scholar] [CrossRef]
- Smith, G.W. Treatment of calf diarrhea: Oral fluid therapy. Vet. Clin. Food Anim. 2008, 25, 55–72. [Google Scholar] [CrossRef]
- Wilms, J.N.; Leal, L.N.; Martín-Tereso, J. Short communication: Hypernatremia in diarrheic calves associated with oral electrolyte administration in water and milk replacer in absence of access to water. J. Dairy Sci. 2020, 103, 5495–5500. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.M.; Bateman, H.G.; Aldrich, J.M.; Quigley, J.D.; Schlotterbeck, R.L. Evaluation of ad libitum acidified milk replacer programs for dairy calves. J. Dairy Sci. 2013, 96, 3153–3162. [Google Scholar] [CrossRef] [PubMed]
- Tood, C.G.; Leslie, K.E.; Millman, S.T.; Bielmann, V.; Anderson, N.G.; Sargeant, J.M.; DeVries, T.J. Clinical trial on the effects of a free-access acidified milk replacer feeding program on the health and growth of dairy replacement heifers and veal calves. J. Dairy Sci. 2017, 100, 713–725. [Google Scholar] [CrossRef] [Green Version]
- Tood, C.G.; Millman, S.T.; Leslie, K.E.; Anderson, N.G.; Sargeant, J.M.; DeVries, T.J. Effects milk replacer acidification and a free-access acidified feeding on feeding, oral, and lying behavior of dairy calves. J. Dairy Sci. 2018, 101, 8236–8247. [Google Scholar] [CrossRef] [PubMed]
- Hellekant, G.; Hård af Segerstad, C.; Roberts, T.W. Sweet taste in the calf: III. Behavioral responses to sweeteners. Physiol. Behav. 1994, 56, 555–562. [Google Scholar] [CrossRef]
- Habold, C.; Foltzer-Jourdainne, C.; LeMaho, Y.; Lignot, J.H.; Oudart, H. Intestinal gluconeogenesis and glucose transport according to body fuel availability in rats. J. Physiol. 2005, 566, 575–586. [Google Scholar] [CrossRef]
- Mithieux, G.; Bady, I.; Gautier, A.; Croset, M.; Rajas, F.; Zitoun, C. Induction of control genes in intestinal gluconeogenesis is sequential during fasting and maximal in diabetes. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E370–E375. [Google Scholar] [CrossRef] [Green Version]
- Debnam, E.S.; Levin, R.J. Effects of fasting and semistarvation on the kinetics of active and passive sugar absorption across the small intestine in vivo. J Physiol. 1975, 252, 681–700. [Google Scholar] [CrossRef] [Green Version]
- Das, S.; Yadav, R.K.; Nagchoudhuri, J. Effect of fasting on the intestinal absorption of d-glucose and d-xylose in rats in vivo. Indian J. Physiol. Pharmacol. 2001, 45, 451–456. [Google Scholar]
- Blickslager, A.T.; Moeser, A.J.; Gookin, J.L.; Jones, S.L.; Odle, J. Restoration of barrier function in injured intestinal mucosa. Physiol. Rev. 2007, 87, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Huen, S.C.; Luan, H.H.; Yu, S.; Zhang, C.; Gallezot, J.D.; Booth, C.J.; Medzhitov, R. Opposite effects of fasting metabolism on tissue tolerance in bacterial and viral inflammation. Cell 2016, 166, 1512–1525.e12. [Google Scholar] [CrossRef] [Green Version]
- Fahey, M.J.; Fisher, A.J.; Steele, M.A.; Greenwood, S.L. Characterization of the colostrum and transition milk proteomes from primiparous and multiparous Holstein dairy cows. J. Dairy Sci. 2020, 103, 1993–2005. [Google Scholar] [CrossRef] [Green Version]
- Pyo, J.; Hare, K.; Pletts, S.; Inabu, Y.; Haines, D.; Sugino, T.; Guan, L.L.; Steele, M.A. Feeding colostrum or 1:1 colostrum mixture for 3 days postnatal increases small intestinal development and minimally influences plasma glucagon-like peptide-2 and serum insulin-like growth factor-1 concentrations in Holstein bull calves. J. Dairy Sci. 2020, 103, 4236–4251. [Google Scholar] [CrossRef]
- Krehbiel, C.R.; Zhang, G. Modulation of gut health in beef, dairy cattle vital to productivity. Feedstuffs 2017, 89, 3. [Google Scholar]
- Wilms, J.N.; Berends, H.; Martín-Tereso, J. Hypertonic milk replacers increase gastrointestinal permeability in healthy dairy calves. J. Dairy Sci. 2019, 102, 1237–1246. [Google Scholar] [CrossRef] [Green Version]
- Amado, L.; Berends, H.; Leal, L.N.; Wilms, J.; van Laar, H.; Gerrits, W.J.J.; Martín-Tereso, J. Effect of energy source in calf milk replacer on performance, digestibility, and gut permeability in rearing calves. J. Dairy Sci. 2019, 102, 3994–4001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urie, N.J.; Lombard, J.E.; Shivley, C.B.; Kopral, C.A.; Adams, A.E.; Earlyewine, T.J.; Olson, J.D.; Garry, F.B. Preweaned heifer management on US dairy operations. Part V. Factors associated with morbility and mortality in preweaned dairy heifer calves. J. Dairy Sci. 2018, 101, 9229–9244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisoni, L.; Relling, A.E. The effects of supplementing yeast fermentation products on gut permeability, hormone concentration, and growth in newborn dairy calves. Transl. Anim. Sci. 2020, 4, 809–821. [Google Scholar] [CrossRef]
- Alassane-Kpembi, I.; Pinton, P.; Oswald, I.P. Effects of mycotoxins on the intestine. Toxins 2019, 11, 159. [Google Scholar] [CrossRef] [Green Version]
- Deters, E.L.; Hansen, S.L. Invited review: Linking road transportation with oxidative stress in cattle and other species. Appl. Ani. Sci. 2020, 36, 183–200. [Google Scholar] [CrossRef]
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Juniper, D.T. Revisiting oxidative stress and use of organic selenium in dairy cow nutrition. Animals 2019, 9, 462. [Google Scholar] [CrossRef] [Green Version]
- Scott, M.A.; Woolums, A.R.; Swiderski, C.E.; Perkins, A.D.; Nanduri, B.; Smith, D.R.; Karisch, B.B.; Epperson, W.B.; Blanton, J.R., Jr. Whole blood transcriptomic analysis of beef cattle at arrival identifies potential predictive molecules and mechanisms that indicate animals that naturally resist bovine respiratory disease. PLoS ONE 2020, 15, e0227507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Renaud, D.L.; Overton, M.W.; Kelton, D.F.; LeBlanc, S.J.; Dhuyvetter, K.C.; Duffield, T.F. Effect of health status evaluated at arrival on growth in milk-fed veal calves: A prospective single cohort study. J. Dairy Sci. 2018, 101, 10383–10390. [Google Scholar] [CrossRef] [PubMed]
- Zabielski, R.; Onaga, T.; Mineo, H.; Kato, S. Periodic fluctuations in pancreatic secretion and duodenal motility investigated in neonatal calves. Exp. Physiol. 1993, 78, 675–684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devant, M.; Marti, S. Strategies for Feeding Unweaned Dairy Beef Cattle to Improve Their Health. Animals 2020, 10, 1908. https://doi.org/10.3390/ani10101908
Devant M, Marti S. Strategies for Feeding Unweaned Dairy Beef Cattle to Improve Their Health. Animals. 2020; 10(10):1908. https://doi.org/10.3390/ani10101908
Chicago/Turabian StyleDevant, Maria, and Sonia Marti. 2020. "Strategies for Feeding Unweaned Dairy Beef Cattle to Improve Their Health" Animals 10, no. 10: 1908. https://doi.org/10.3390/ani10101908



