Effects of Calcium Soaps from Palm, Canola and Safflower Oils on Dry Matter Intake, Nutrient Digestibility, Milk Production, and Milk Composition in Dairy Goats
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Experimental Design and Diets
2.2. Calcium Soap Elaboration
2.3. Chemical Composition of Diets and Milk
2.4. In Vitro Gas Production
2.5. Calculations
2.6. Statistical Analysis
3. Results and Discussion
3.1. Ingredients and Chemical Composition of Dietary Treatments
3.2. Nutrient Intake and Digestibility
3.3. Nitrogen Balance
3.4. Milk Production and Milk Composition
3.5. In Vitro Gas Production
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanz Sampelayo, M.R.; Chilliard, Y.; Schmidely, P.; Boza, J. Influence of type of diet on the fat constituents of goat and sheep milk. Small Rumin. Res. 2007, 68, 42–63. [Google Scholar] [CrossRef]
- Abubakr, A.; Alimon, A.R.; Yaakub, H.; Abdullah, N.; Ivan, M. Effect of feeding palm oil by-products based diets on muscle fatty acid composition in goats. PLoS ONE 2015, 10, e0119756. [Google Scholar] [CrossRef] [PubMed]
- Karami, M.; Ponnampalam, E.N.; Hopkins, D.L. The effect of palm oil or canola oil on feedlot performance, plasma and tissue fatty acid profile and meat quality in goats. Meat Sci. 2013, 94, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Adeyemi, K.D.; Sazili, A.Q.; Ebrahimi, M.; Samsudin, A.A.; Alimon, A.R.; Karim, R.; Karsani, S.A.; Sabow, A.B. Effects of blend of canola oil and palm oil on nutrient intake and digestibility, growth performance, rumen fermentation and fatty acids in goats. Anim. Sci. J. 2016, 87, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Z.; Yan, C.G.; Lee, H.G.; Choi, C.W.; Song, M.K. Influence of dietary plant oils on mammary lipogenic enzymes and the conjugated linoleic acid content of plasma and milk fat of lactating goats. Anim. Feed Sci. Technol. 2012, 174, 26–35. [Google Scholar] [CrossRef]
- Jenkins, T.C.; Bridges, W.C., Jr. Protection of fatty acids against ruminal biohydrogenation in cattle. Eur. J. Lipid Sci. Technol. 2007, 109, 778–789. [Google Scholar] [CrossRef]
- Silva, M.M.C.D.; Rodrigues, M.T.; Branco, R.H.; Rodrigues, C.A.F.; Sarmento, J.L.R.; Queiroz, A.C.D.; Silva, S.P.D. Suplementação de lipídios em dietas para cabras em lactação: Consumo e eficiência de utilização de nutrientes. Rev. Bras. Zootec. 2007, 36, 257–267. [Google Scholar] [CrossRef]
- Sanz Sampelayo, M.R.; Pérez, L.; Martín Alonso, J.J.; Gil Extremera, F.; Boza, J. Effects of concentrates with different contents of protected fat rich in PUFAs on the performance of lactating Granadina goats: 1. Feed intake, nutrient digestibility, N and energy utilisation for milk production. Small Rumin. Res. 2002, 43, 133–139. [Google Scholar] [CrossRef]
- Casals, R.; Caja, G.; Such, X.; Torre, C.; Calsamiglia, S. Effects of calcium soaps and rumen undegradable protein on the milk production and composition of dairy ewes. J. Dairy Res. 1999, 66, 177–191. [Google Scholar] [CrossRef]
- Abdelhakim, H.H.; Abdelhafeez, H.M.; Tawfeek, S.S.; Hegazy, M.A. Effect of calcium soap of palm oil fatty acids on milk composition, ewe and lamb performance in a crossbred sheep. J. Vet. Med. Res. 2016, 23, 46–60. [Google Scholar] [CrossRef]
- Manso, T.; Castro, T.; Mantecón, A.R.; Jimeno, V. Effects of palm oil and calcium soaps of palm oil fatty acids in fattening diets on digestibility, performance and chemical body composition of lambs. Anim. Feed Sci. Technol. 2006, 127, 175–186. [Google Scholar] [CrossRef]
- Chouinard, P.Y.; Corneau, L.; Butler, W.R.; Bauman, D.E.; Chilliard, Y.; Drackley, J.K. Effect of Dietary Lipid Source on Conjugated Linoleic Acid Concentrations in Milk Fat1,2. J. Dairy Sci. 2001, 84, 680–690. [Google Scholar] [CrossRef]
- Castro, T.; Manso, T.; Jimeno, V.; Del Alamo, M.; Mantecón, A.R. Effects of dietary sources of vegetable fats on performance of dairy ewes and conjugated linoleic acid (CLA) in milk. Small Rumin. Res. 2009, 84, 47–53. [Google Scholar] [CrossRef]
- Savoini, G.; Omodei Zorini, F.; Farina, G.; Agazzi, A.; Cattaneo, D.; Invernizzi, G. Effects of Fat Supplementation in Dairy Goats on Lipid Metabolism and Health Status. Animals 2019, 9, 917. [Google Scholar] [CrossRef]
- Vargas-Bello-Pérez, E.; Vera, R.R.; Aguilar, C.; Lira, R.; Peña, I.; Fernández, J. Feeding olive cake to ewes improves fatty acid profile of milk and cheese. Anim. Feed Sci. Technol. 2013, 184, 94–99. [Google Scholar] [CrossRef]
- Vargas-Bello-Pérez, E.; Vera, R.R.; Aguilar, C.; Lira, R.; Peña, I.; Valenzuela, A.; Cerda, H. Effect of dietary inclusion of lampante olive oil on milk and cheese fatty acid profiles of ewes. Grasas Aceites 2013, 64, 295–303. [Google Scholar] [CrossRef]
- Vargas-Bello-Pérez, E.; Zhao, W.; Bionaz, M.; Luo, J.; Loor, J.J. Nutrigenomic Effect of Saturated and Unsaturated Long Chain Fatty Acids on Lipid-Related Genes in Goat Mammary Epithelial Cells: What Is the Role of PPARγ? Vet. Sci. 2019, 6, 54. [Google Scholar] [CrossRef]
- de Souza, J.; Batistel, F.; Santos, F.A.P. Effect of sources of calcium salts of fatty acids on production, nutrient digestibility, energy balance, and carryover effects of early lactation grazing dairy cows. J. Dairy Sci. 2017, 100, 1072–1085. [Google Scholar] [CrossRef]
- NRC. Nutrient Requirements of Small Ruminants: Sheep, Goats, Cervids, and New World Camelids; National Academies Press: Washington, DC, USA, 2007; p. 362. [Google Scholar]
- AOAC. Association of Official Analytical Chemists. In Official Methods of Analysis, 18th ed.; AOAC: Gaithersburg, MD, USA, 2005; p. 432. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for Dietary Fiber, Neutral Detergent Fiber, and Nonstarch Polysaccharides in Relation to Animal Nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Jenkins, T.C.; Palmquist, D.L. Effect of fatty acids or calcium soaps on rumen and total nutrient digestibility of dairy rations. J. Dairy Sci. 1984, 67, 978–986. [Google Scholar] [CrossRef]
- Romero-Davila, A.; Herrera-Corredor, C.; Calderón-Aranda, J.A.; Buendía-Rodriguez, G.; Pescador Salas, N.; González-Ronquillo, M. Uso de jabones de calcio como alternativa de suplementación en rumiantes en épocas se sequia en zonas áridas y semiáridas. In SUSTENTABILIDAD AGROPECUARIA, Experiencias de Investigación Para el Desarrollo Agropecuario, Forestal y Rural; Brunett Perez, L., William, G.D., Gutierrez Castillo, A., Jaimes Arriaga, E., Eds.; Colofón: Ciudad de Mexico, Mexico; Universidad Autónoma del Estado de Mexico: Estado de Mexico, Mexico, 2017; pp. 155–168. ISBN 978607858301. [Google Scholar]
- AOAC. Association of Official Analytical Chemists. In Official Methods of Analysis Review 3, 18th ed.; AOAC: Gaithersburg, MD, USA, 2010. [Google Scholar]
- Theodorou, M.K.; Williams, B.A.; Dhanoa, M.S.; McAllan, A.B.; France, J. A simple gas production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Anim. Feed Sci. Technol. 1994, 48, 185–197. [Google Scholar] [CrossRef]
- Menke, K.H.; Steingass, H. Estimation of energetic feed value obtained from chemical analyses and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–55. [Google Scholar]
- SAS. Statistical Analysis Systems Institute. User’s Guide Version Eight; SAS Institute Inc.: Cary, NC, USA, 2002. [Google Scholar]
- Krishnamoorthy, U.; Soller, H.; Steingass, H.; Menke, K.H. A comparative study on rumen fermentation of energy supplements in vitro. J. Anim. Physiol. Anim. Nutr. 1991, 65, 28–35. [Google Scholar] [CrossRef]
- Tyrrell, H.F.; Reid, J.T. Prediction of the Energy Value of Cow’s Milk1, 2. J. Dairy Sci. 1965, 48, 1215–1223. [Google Scholar] [CrossRef]
- Blümmel, M.; Steingass, H.; Becker, K. The relationship between in vitro gas production, in vitro microbial biomass yield and 15N incorporation and its implications for the prediction of voluntary feed intake of roughages. Br. J. Nutr. 1997, 77, 911–921. [Google Scholar] [CrossRef]
- Getachew, G.; Makkar, H.P.S.; Becker, K. Tropical browses: Contents of phenolic compounds, in vitro gas production and stoichiometric relationship between short chain fatty acid and in vitro gas production. J. Agric. Sci. 2003, 139, 341–352. [Google Scholar] [CrossRef]
- Bouattour, M.A.; Casals, R.; Albanell, E.; Such, X.; Caja, G. Feeding Soybean Oil to Dairy Goats Increases Conjugated Linoleic Acid in Milk. J. Dairy Sci. 2008, 91, 2399–2407. [Google Scholar] [CrossRef]
- Bernard, L.; Leroux, C.; Faulconnier, Y.; Durand, D.; Shingfield, K.J.; Chilliard, Y. Effect of sunflower-seed oil or linseed oil on milk fatty acid secretion and lipogenic gene expression in goats fed hay-based diets. J. Dairy Res. 2009, 76, 241–248. [Google Scholar] [CrossRef]
- Kumar, R.; Sivaiah, K.; Reddy, Y.R.; Ekambram, B.; Reddy, T.J.; Reddy, G.V.N. Effect of supplementation of dietary protected lipids on intake and nutrient utilization in Deccani lambs. Trop. Anim. Health Prod. 2006, 38, 151–158. [Google Scholar] [CrossRef]
- Kumar, S.; Reddy, Y.R.; Kumari, N.N.; Sridhar, K.; Rao, D.S. In vitro, in situ and in vivo evaluation of straw based diets supplemented with bypass fat as concentrated energy source in Murrah buffaloes. Buffalo Bull. 2017, 36, 357–368. [Google Scholar]
- Maia, M.d.O.; Susin, I.; Ferreira, E.M.; Nolli, C.P.; Gentil, R.S.; Pires, A.V.; Mourão, G.B. Intake, nutrient apparent digestibility and ruminal constituents of sheep fed diets with canola, sunflower or castor oils. Rev. Bras. Zootec. 2012, 41, 2350–2356. [Google Scholar]
- Nur Atikah, I.; Alimon, A.R.; Yaakub, H.; Abdullah, N.; Jahromi, M.F.; Ivan, M.; Samsudin, A.A. Profiling of rumen fermentation, microbial population and digestibility in goats fed with dietary oils containing different fatty acids. BMC Vet. Res. 2018, 14, 344. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Bello-Pérez, E.; Márquez-Hernández, R.I.; Hernández-Castellano, L.E. Bioactive peptides from milk: Animal determinants and their implications in human health. J. Dairy Res. 2019, 86, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Hristov, A.N.; Heyler, K.S.; Cassidy, T.W.; Long, M.; Corl, B.A.; Karnati, S.K.R. Effects of dietary protein concentration and coconut oil supplementation on nitrogen utilization and production in dairy cows. J. Dairy Sci. 2011, 94, 5544–5557. [Google Scholar] [CrossRef] [PubMed]
- Mir, Z.; Goonewardene, L.A.; Okine, E.; Jaegar, S.; Scheer, H.D. Effect of feeding canola oil on constituents, conjugated linoleic acid (CLA) and long chain fatty acids in goats milk. Small Rumin. Res. 1999, 33, 137–143. [Google Scholar] [CrossRef]
- Chilliard, Y.; Ferlay, A.; Rouel, J.; Lamberet, G. A Review of Nutritional and Physiological Factors Affecting Goat Milk Lipid Synthesis and Lipolysis1. J. Dairy Sci. 2003, 86, 1751–1770. [Google Scholar] [CrossRef] [PubMed]
- Lock, A.L.; Rovai, M.; Gipson, T.A.; de Veth, M.J.; Bauman, D.E. A conjugated linoleic acid supplement containing trans-10, cis-12 conjugated linoleic acid reduces milk fat synthesis in lactating goats. J. Dairy Sci. 2008, 91, 3291–3299. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.D. Implication of Feeding Isoenergetic Diets Containing Animal Fat on Milk Composition of Alpine Does During Early Lactation1. J. Dairy Sci. 1993, 76, 1137–1147. [Google Scholar] [CrossRef]
- Palmquist, D.L. The Role of Dietary Fats in Efficiency of Ruminants. J. Nutr. 1994, 124, 1377S–1382S. [Google Scholar] [CrossRef]
- Storry, J.E.; Hall, A.J.; Johnson, V.W. The effects of increasing amounts of dietary tallow on milk-fat secretion in the cow. J. Dairy Res. 1973, 40, 293–299. [Google Scholar] [CrossRef]
- Palmquist, D.L.; Jenkins, T.C. Fat in Lactation Rations1, 2: Review. J. Dairy Sci. 1980, 63, 1–14. [Google Scholar] [CrossRef]
- McGuire, M.A.; Bauman, D.E. Milk biosynthesis and secretion. In Encyclopedia of Dairy Science; Roginsky, H., Fuquay, J.W., Fox, P.F., Eds.; Academic Press: New York, NY, USA, 2003; pp. 1828–1834. [Google Scholar]
- Christensen, R.A.; Drackley, J.K.; LaCount, D.W.; Clark, J.H. Infusion of four long-chain fatty acid mixtures into the abomasum of lactating dairy cows. J. Dairy Sci. 1994, 77, 1052–1069. [Google Scholar] [CrossRef]
- Luna, P.; Bach, A.; Juárez, M.; de la Fuente, M.A. Effect of a Diet Enriched in Whole Linseed and Sunflower Oil on Goat Milk Fatty Acid Composition and Conjugated Linoleic Acid Isomer Profile. J. Dairy Sci. 2008, 91, 20–28. [Google Scholar] [CrossRef]
- Ayeb, N.; Addis, M.; Fiori, M.; Khorchani, S.; Atigui, M.; Khorchani, T. Quality and fatty acid profile of the milk of indigenous goats subjected to different local diets in Tunisian arid lands. J. Anim. Physiol. Anim. Nutr. 2016, 100, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.X.; Susenbeth, A.; Südekum, K.H. In vitro gas production measurements to evaluate interactions between untreated and chemically treated rice straws, grass hay, and mulberry leaves. J. Anim. Sci. 2002, 80, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Kurade, M.B.; Saha, S.; Salama, E.S.; Patil, S.M.; Govindwar, S.P.; Jeon, B.H. Acetoclastic methanogenesis led by Methanosarcina in anaerobic co-digestion of fats, oil and grease for enhanced production of methane. Bioresour. Technol. 2019, 272, 351–359. [Google Scholar] [CrossRef]
- Vargas-Bello-Pérez, E.; Cancino-Padilla, N.; Romero, J.; Garnsworthy, P.C. Quantitative analysis of ruminal bacterial populations involved in lipid metabolism in dairy cows fed different vegetable oils. Animal 2016, 10, 1821–1828. [Google Scholar] [CrossRef]
- Szumacher-Strabel, M.; Cieślak, A.; Nowakowska, A. Effect of oils rich in linoleic acid on in vitro rumen fermentation parameters of sheep, goats and dairy cows. J. Anim. Feed Sci. 2009, 18, 440–452. [Google Scholar] [CrossRef]
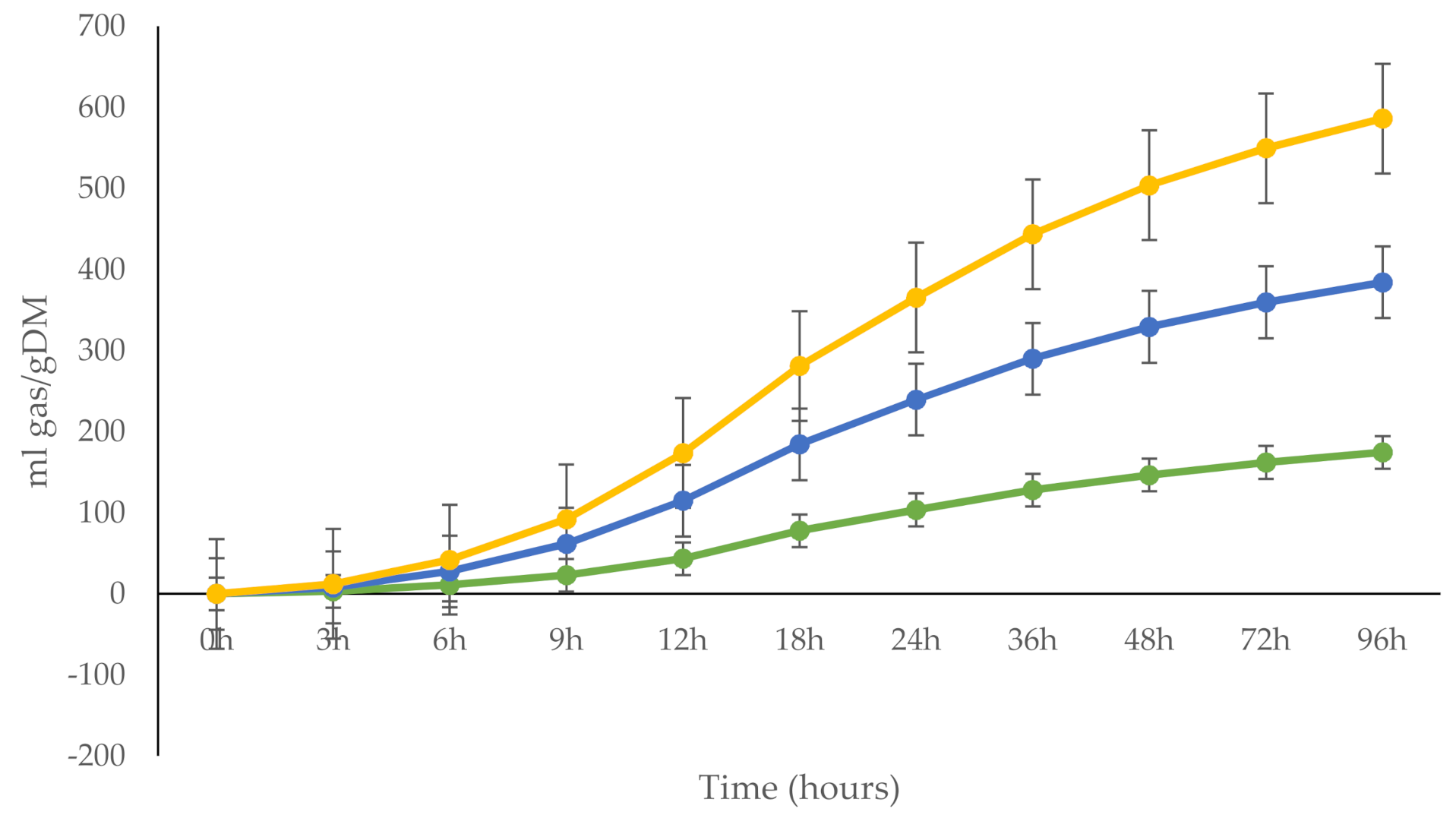
| Ingredient 1 | DM | CP | EE | NDF | ADF | Ca | P | ME 3 |
|---|---|---|---|---|---|---|---|---|
| Soybean meal | 900 | 440 | 17 | 161 | 80 | 3 | 7 | 12.9 |
| Barley hay | 880 | 120 | 20 | 650 | 370 | 1 | 4 | 8.8 |
| Corn grain | 900 | 80 | 41 | 145 | 26 | 1 | 3 | 14.5 |
| Corn silage | 387 | 68 | 30 | 550 | 300 | 2 | 2 | 10.9 |
| Calcium soaps of palm oil | 972 | 0 | 848 | 0 | 0 | 18 | 0 | 26.8 |
| Calcium soaps of canola oil | 969 | 0 | 840 | 0 | 0 | 18 | 0 | 25.8 |
| Calcium soaps of safflower oil | 970 | 0 | 845 | 0 | 0 | 18 | 0 | 26.4 |
| Vitamin and mineral premix 2 | 1000 | 0 | 0 | 0 | 0 | 4.5 | 0.14 | 0 |
| Parameters | SO | CO | PO |
|---|---|---|---|
| Ingredients, g/kg DM | |||
| Corn silage | 500 | 500 | 500 |
| Soybean meal | 102 | 102 | 102 |
| Barley hay | 105 | 105 | 105 |
| Corn grain | 250 | 250 | 250 |
| Calcium soaps of palm oil | 0 | 0 | 27 |
| Calcium soaps of canola oil | 0 | 27 | 0 |
| Calcium soaps of safflower oil | 27 | 0 | 0 |
| Vitamin and minerals premix 1 | 16 | 16 | 16 |
| Chemical composition, g/kg DM | |||
| Dry matter | 644 | 644 | 644 |
| Crude protein | 111 | 111 | 111 |
| Neutral detergent fiber | 395 | 395 | 395 |
| Acid detergent fiber | 203 | 203 | 203 |
| Ether extract | 40.6 | 40.7 | 40.9 |
| Ca | 2.1 | 2.2 | 2.2 |
| P | 2.9 | 2.9 | 2.9 |
| Metabolizable energy, MJ/kg DM 2 | 12.0 | 12.0 | 12.0 |
| Item | SO | CO | PO | SEM | p-Value |
|---|---|---|---|---|---|
| Initial live weight, LW, kg | 52.0 | 51.1 | 51.6 | 1.64 | 0.938 |
| Final live weight, LW, kg | 52.6 | 51.3 | 54.6 | 1.76 | 0.453 |
| Average live weight, LW, kg | 52.3 | 51.2 | 53.1 | 1.37 | 0.635 |
| Average metabolic live weight, LW0.75 | 19.4 | 19.1 | 19.6 | 0.38 | 0.637 |
| Intake, g/d | |||||
| Dry matter | 1820 | 1968 | 2020 | 175 | 0.716 |
| Organic matter | 1667 | 1765 | 1810 | 157 | 0.812 |
| Neutral detergent fiber | 720 | 779 | 800 | 69.1 | 0.716 |
| Acid detergent fiber | 370 | 400 | 411 | 35.5 | 0.717 |
| Fat | 74 | 80 | 82 | 7.11 | 0.689 |
| Metabolizable energy, MJ/d 2 | 20 | 21 | 22 | 1.90 | 0.803 |
| Intake, g/kg LW0.75 | |||||
| Dry matter | 92.9 | 102 | 105 | 10.2 | 0.709 |
| Organic matter | 85.1 | 91.7 | 93.7 | 9.21 | 0.795 |
| Neutral detergent fiber | 36.8 | 40.5 | 93.4 | 4.05 | 0.709 |
| Acid detergent fiber | 18.9 | 20.8 | 21.3 | 2.08 | 0.709 |
| Fat | 3.8 | 4.2 | 4.3 | 0.42 | 0.686 |
| Metabolizable energy, MJ/d 2 | 1.02 | 1.10 | 1.13 | 0.11 | 0.795 |
| Digestibility, g/kg | |||||
| Dry matter | 552 b | 653 a | 588 b | 15.5 | 0.001 |
| Organic matter | 559 b | 663 a | 606 ab | 17.6 | 0.001 |
| Neutral detergent fiber | 460 b | 616 a | 510 b | 17.5 | 0.001 |
| Acid detergent fiber | 424 c | 658 a | 519 b | 16.9 | 0.001 |
| Item | SO | CO | PO | SEM | p-Value |
|---|---|---|---|---|---|
| g/d | |||||
| N intake | 32.46 | 35.09 | 36.04 | 3.11 | 0.716 |
| N excretion | |||||
| Urine | 11.79 | 12.87 | 13.84 | 2.61 | 0.860 |
| Feces | 21.18 | 12.52 | 16.40 | 3.30 | 0.257 |
| N Balance | −0.51 | 9.69 | 5.79 | 4.41 | 0.326 |
| g/kg LW0.75 | |||||
| N intake | 1.65 | 1.82 | 1.86 | 0.18 | 0.709 |
| N excretion | |||||
| Urine | 0.60 | 0.66 | 0.71 | 0.13 | 0.852 |
| Feces | 1.08 | 0.64 | 0.85 | 0.17 | 0.294 |
| N Balance | −0.03 | 0.51 | 0.30 | 0.23 | 0.323 |
| Item | SO | CO | PO | SEM | p-Value |
|---|---|---|---|---|---|
| Milk production, kg/d | 1.10 a | 0.95 b | 0.88 b | 0.09 | 0.009 |
| Fat-corrected milk 3.5% | 1.07 | 0.93 | 0.88 | 0.18 | 0.740 |
| Energy corrected milk | 1.08 | 0.99 | 0.86 | 0.17 | 0.667 |
| Feed efficiency | 0.36 | 0.30 | 0.24 | 0.05 | 0.169 |
| FE FCM | 0.36 | 0.30 | 0.25 | 0.05 | 0.265 |
| Milk production efficiency | 6.19 | 6.59 | 4.80 | 0.88 | 0.330 |
| Fat, g/d | 35.0 a | 32.3 b | 34.3 a | 0.56 | 0.002 |
| Protein, g/d | 37.7 a | 29.8 b | 34.6 a | 0.48 | 0.003 |
| Lactose, g/d | 45.1 | 39.9 | 37.0 | 3.82 | 0.076 |
| Total solids, g/d | 127 a | 111 b | 113 b | 0.85 | 0.005 |
| Non-fat solids, g/d | 100 a | 78.9 b | 78.9 b | 0.96 | 0.002 |
| Fat, g/kg | 33 b | 35 b | 41 a | 0.52 | 0.001 |
| Protein, g/kg | 35 b | 33 b | 41 a | 0.57 | 0.003 |
| Lactose, g/kg | 41 | 42 | 42 | 0.85 | 0.365 |
| Total solids, g/kg | 120 b | 122 b | 136 a | 1.75 | 0.049 |
| Non-fat solids, g/kg | 95 a | 86 b | 94 a | 0.91 | 0.002 |
| Item | SO | CO | PO | SEM | p-Value |
|---|---|---|---|---|---|
| B | 206 d | 174 e | 201 d | 4.6 | <0.001 |
| C | 0.05 d | 0.04 e | 0.04 de | 0.001 | 0.001 |
| Lag time | 4.47 e | 5.23 d | 4.86 de | 0.188 | 0.030 |
| Gas production, mL gas/g DM | |||||
| 6 h | 16.6 a | 11.0 b | 14.3 ab | 1.42 | 0.033 |
| 12 h | 71.3 a | 43.5 b | 58.5 a | 3.56 | <0.001 |
| 24 h | 135 a | 103 b | 125 a | 4.59 | <0.001 |
| 48 h | 182 a | 146 b | 174 a | 4.97 | <0.001 |
| DMD 96 h | 74 | 72 | 77 | 5.6 | 0.782 |
| PF 96 h | 290 | 293 | 263 | 34.8 | 0.796 |
| GY 24 h | 33.9 a | 25.9 b | 31.4 a | 1.14 | <0.001 |
| SCFA | 0.75 a | 0.57 b | 0.69 a | 0.03 | <0.001 |
| MCP | 670 | 660 | 704 | 57.6 | 0.854 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vargas-Bello-Pérez, E.; Robles-Jimenez, L.E.; Ayala-Hernández, R.; Romero-Bernal, J.; Pescador-Salas, N.; Castelán-Ortega, O.A.; González-Ronquillo, M. Effects of Calcium Soaps from Palm, Canola and Safflower Oils on Dry Matter Intake, Nutrient Digestibility, Milk Production, and Milk Composition in Dairy Goats. Animals 2020, 10, 1728. https://doi.org/10.3390/ani10101728
Vargas-Bello-Pérez E, Robles-Jimenez LE, Ayala-Hernández R, Romero-Bernal J, Pescador-Salas N, Castelán-Ortega OA, González-Ronquillo M. Effects of Calcium Soaps from Palm, Canola and Safflower Oils on Dry Matter Intake, Nutrient Digestibility, Milk Production, and Milk Composition in Dairy Goats. Animals. 2020; 10(10):1728. https://doi.org/10.3390/ani10101728
Chicago/Turabian StyleVargas-Bello-Pérez, Einar, Lizbeth Esmeralda Robles-Jimenez, Rafael Ayala-Hernández, Jose Romero-Bernal, Nazario Pescador-Salas, Octavio Alonso Castelán-Ortega, and Manuel González-Ronquillo. 2020. "Effects of Calcium Soaps from Palm, Canola and Safflower Oils on Dry Matter Intake, Nutrient Digestibility, Milk Production, and Milk Composition in Dairy Goats" Animals 10, no. 10: 1728. https://doi.org/10.3390/ani10101728
APA StyleVargas-Bello-Pérez, E., Robles-Jimenez, L. E., Ayala-Hernández, R., Romero-Bernal, J., Pescador-Salas, N., Castelán-Ortega, O. A., & González-Ronquillo, M. (2020). Effects of Calcium Soaps from Palm, Canola and Safflower Oils on Dry Matter Intake, Nutrient Digestibility, Milk Production, and Milk Composition in Dairy Goats. Animals, 10(10), 1728. https://doi.org/10.3390/ani10101728








