Embryonic Thermal Manipulation Affects the Antioxidant Response to Post-Hatch Thermal Exposure in Broiler Chickens
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
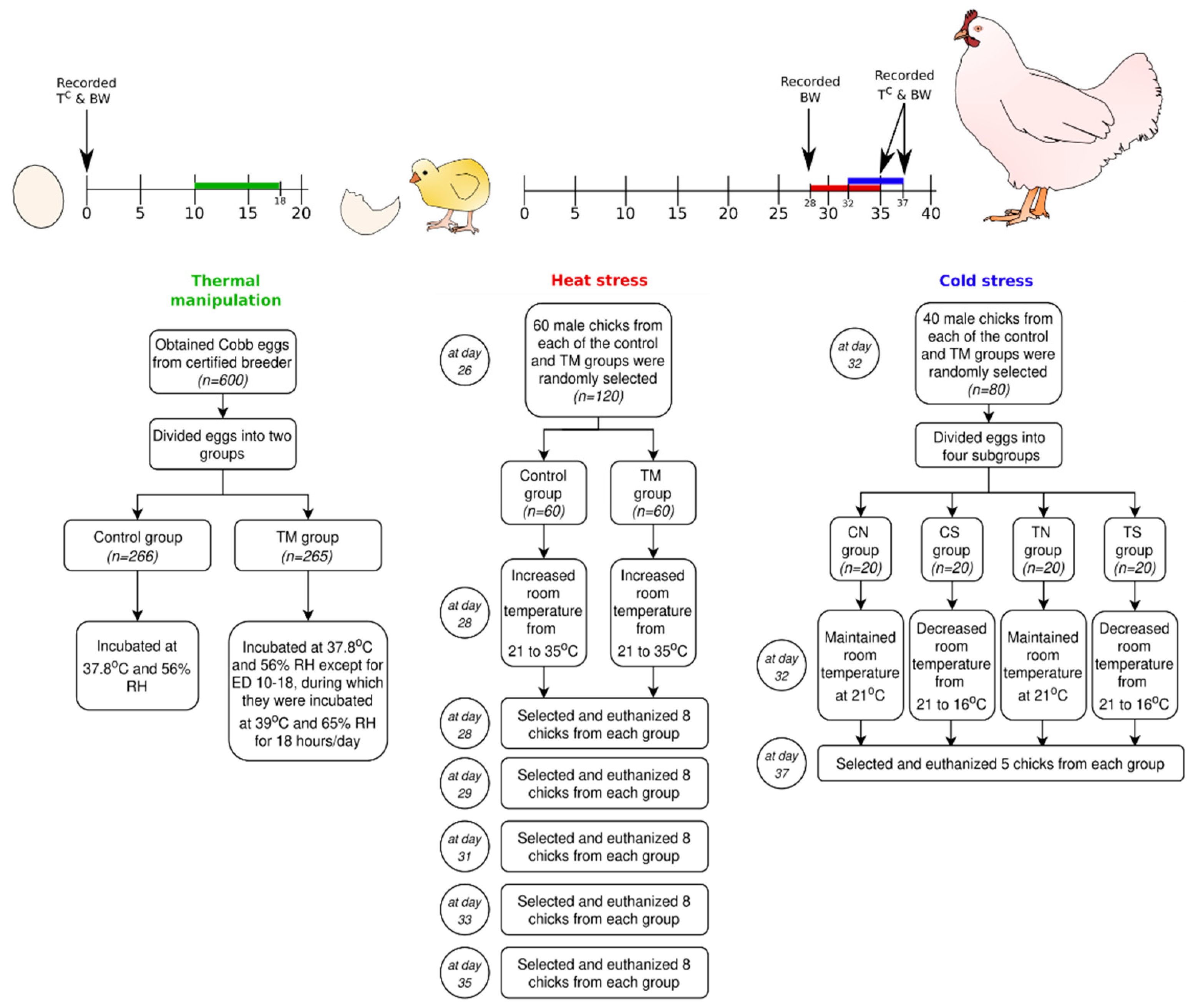
2.1. Egg Procurement and Incubation
2.2. Hatchery Management
2.3. Experiment 1: Post-Hatch Heat Exposure
2.4. Experiment 2: Post-Hatch Cold Exposure
2.5. cDNA Synthesis
2.6. Primer Design and Relative mRNA Quantitation Analysis by Real-Time RT-PCR
2.7. Statistical Analysis
3. Results
3.1. Effect of Thermal Manipulation (TM) on Hatchability and Physiological Parameters of Broiler Chicks
3.2. Effect of Post-Hatch Heat Stress on Physiological Parameters of Thermally Manipulated Broilers
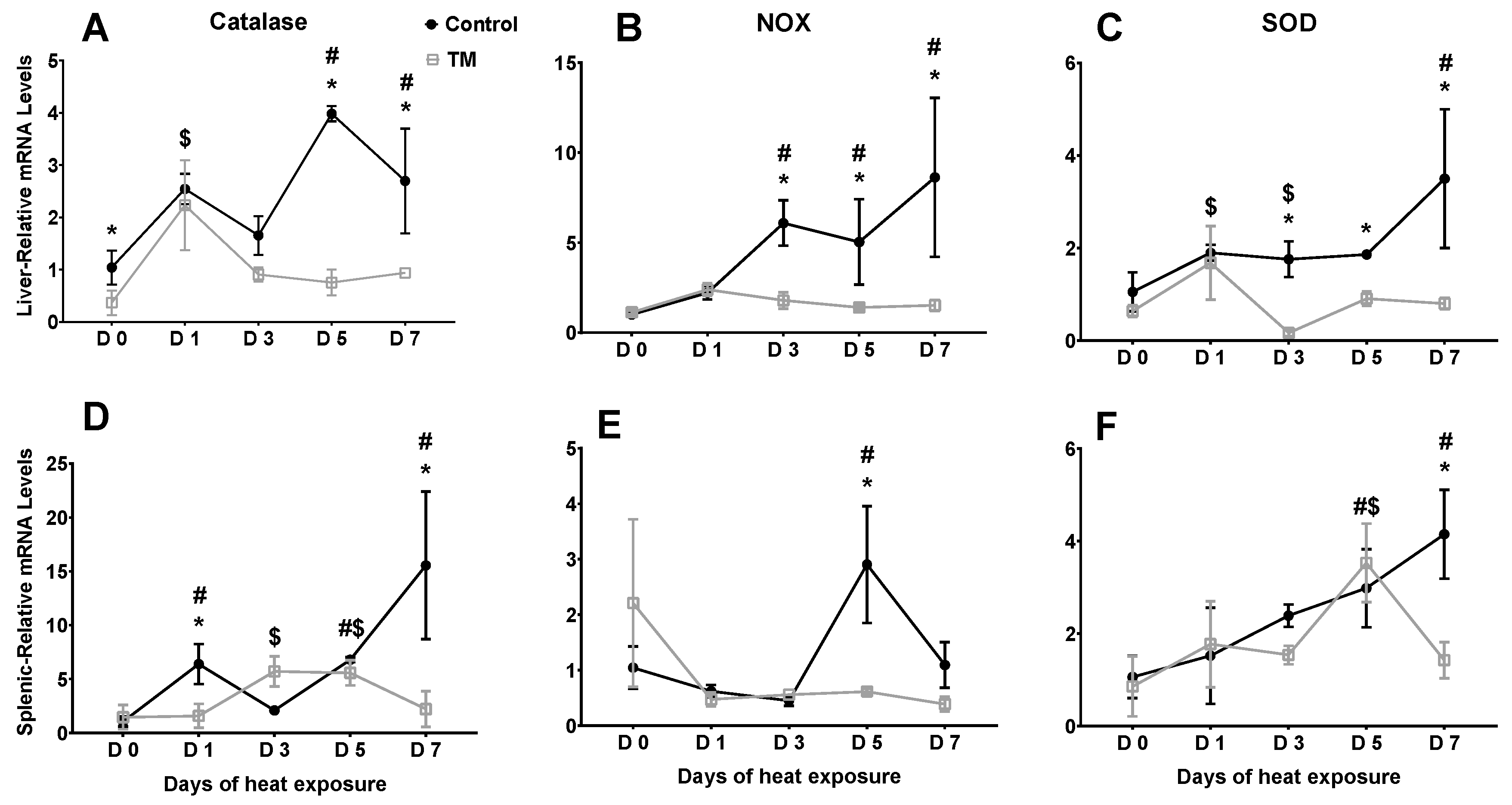
3.3. Effect of Post-Hatch Heat Stress on Antioxidant Enzyme mRNA Levels in Thermally Manipulated Broilers
3.4. Effect of Post-Hatch Cold Stress on Physiological Parameters of Thermally Manipulated Broilers
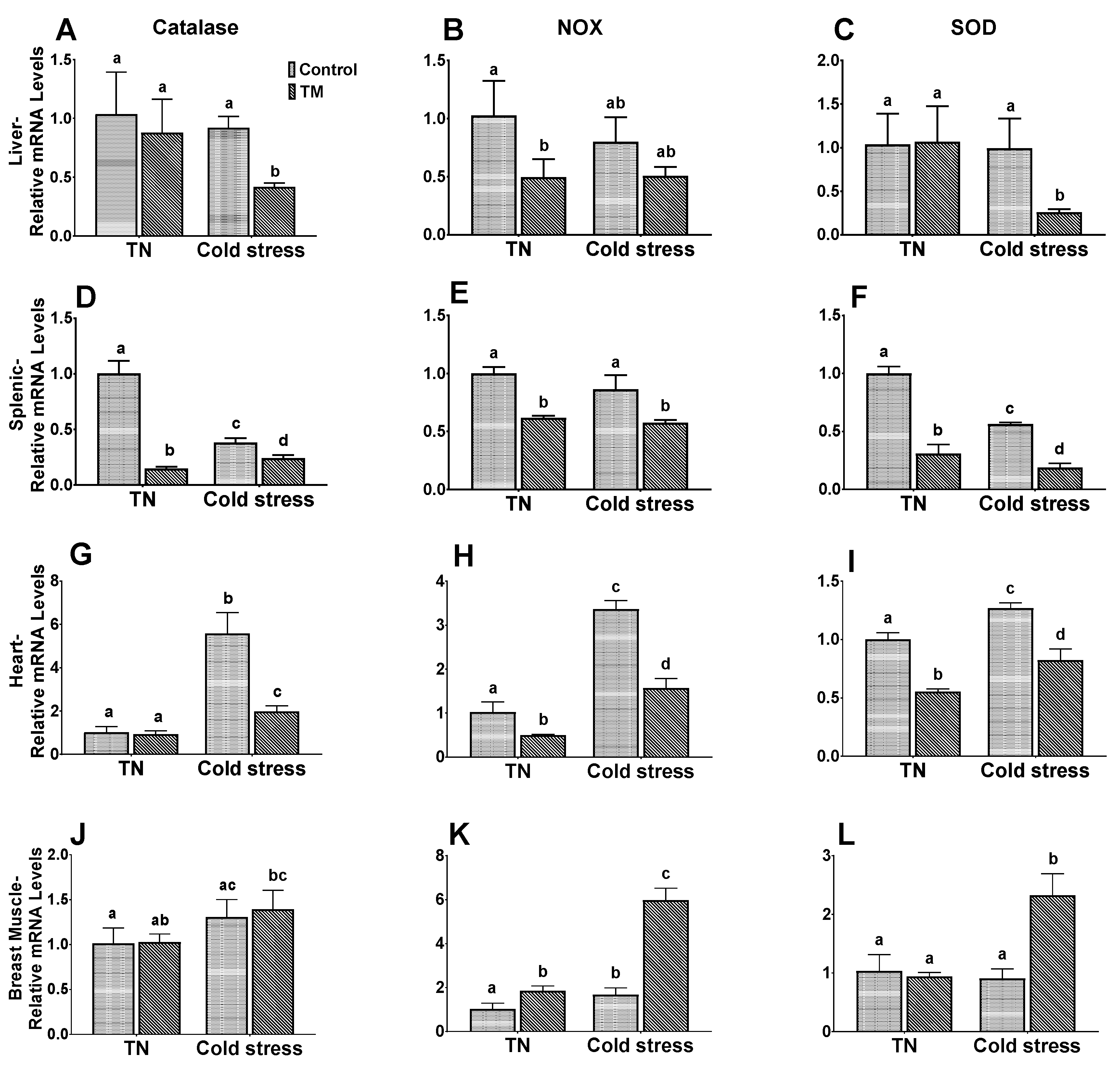
3.5. Effect of Post-Hatch Cold stress on mRNA Levels of Antioxidant Enzymes in Thermally Manipulated Broilers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McKay, J.C.; Barton, N.F.; Koerhuis, A.N.M.; McAdam, J. The challenge of genetic change in the broiler chicken. BSAP Occas. Publ. 2000, 27, 1–7. [Google Scholar] [CrossRef]
- Bennett, C.E.; Thomas, R.; Williams, M.; Zalasiewicz, J.; Edgeworth, M.; Miller, H.; Coles, B.; Foster, A.; Burton, E.J.; Marume, U. The broiler chicken as a signal of a human reconfigured biosphere. R. Soc. Open Sci. 2018, 5, 180325. [Google Scholar] [CrossRef] [PubMed]
- Tallentire, C.W.; Leinonen, I.; Kyriazakis, I. Breeding for efficiency in the broiler chicken: A review. Agron. Sustain. Dev. 2016, 36, 66. [Google Scholar] [CrossRef]
- Zuidhof, M.J.; Schneider, B.L.; Carney, V.L.; Korver, D.R.; Robinson, F.E. Growth, efficiency, and yield of commercial broilers from 1957, 1978, and 2005. Poult. Sci. 2014, 93, 2970–2982. [Google Scholar] [CrossRef] [PubMed]
- Zaboli, G.; Huang, X.; Feng, X.; Ahn, D.U. How can heat stress affect chicken meat quality? A review. Poult. Sci. 2019, 98, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Tickle, P.G.; Hutchinson, J.R.; Codd, J.R. Energy allocation and behaviour in the growing broiler chicken. Sci. Rep. 2018, 8, 4562. [Google Scholar] [CrossRef]
- Nyoni, N.M.B.; Grab, S.; Archer, E.R.M. Heat stress and chickens: Climate risk effects on rural poultry farming in low-income countries. Clim. Dev. 2019, 11, 83–90. [Google Scholar] [CrossRef]
- Lara, L.J.; Rostagno, M.H. Impact of heat stress on poultry production. Animals 2013, 3, 356–369. [Google Scholar] [CrossRef]
- Mishra, B.; Jha, R. Oxidative stress in the poultry gut: Potential challenges and interventions. Front. Vet. Sci. 2019, 6, 60. [Google Scholar] [CrossRef]
- Akbarian, A.; Michiels, J.; Degroote, J.; Majdeddin, M.; Golian, A.; De Smet, S. Association between heat stress and oxidative stress in poultry; mitochondrial dysfunction and dietary interventions with phytochemicals. J. Anim. Sci. Biotechnol. 2016, 7, 37. [Google Scholar] [CrossRef]
- Fellenberg, M.A.; Speisky, H. Antioxidants: Their effects on broiler oxidative stress and its meat oxidative stability. World’s Poult. Sci. J. 2006, 62, 53–70. [Google Scholar] [CrossRef]
- Betteridge, D.J. What is oxidative stress? In Metabolism: Clinical and Experimental; W.B. Saunders: Philadelphia, PA, USA, 2000; Volume 49, pp. 3–8. [Google Scholar]
- Al-Zghoul, M.B.; Sukker, H.; Ababneh, M.M. Effect of thermal manipulation of broilers embryos on the response to heat-induced oxidative stress. Poult. Sci. 2019, 98, 991–1001. [Google Scholar] [CrossRef]
- Nita, M.; Grzybowski, A. The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxid. Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, J.; Bae, S.J.; Kim, Y.; Park, B.J.; Choi, J.W.; Kwon, J.; Cha, G.H.; Yoo, H.J.; Jo, E.K.; et al. NADPH oxidase 4 is required for the generation of macrophage migration inhibitory factor and host defense against Toxoplasma gondii infection. Sci. Rep. 2017, 7, 6361. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Haigh, S.; Barman, S.; Fulton, D.J.R. From form to function: The role of Nox4 in the cardiovascular system. Front. Physiol. 2012, 3, 412. [Google Scholar] [CrossRef]
- Schröder, K.; Zhang, M.; Benkhoff, S.; Mieth, A.; Pliquett, R.; Kosowski, J.; Kruse, C.; Luedike, P.; Michaelis, U.R.; Weissmann, N.; et al. Nox4 Is a protective reactive oxygen species generating vascular NADPH oxidase. Circ. Res. 2012, 110, 1217–1225. [Google Scholar] [CrossRef]
- Kuroda, J.; Ago, T.; Matsushima, S.; Zhai, P.; Schneider, M.D.; Sadoshima, J. NADPH oxidase 4 (Nox4) is a major source of oxidative stress in the failing heart. Proc. Natl. Acad. Sci. USA 2010, 107, 15565–15570. [Google Scholar] [CrossRef]
- Nisimoto, Y.; Diebold, B.A.; Cosentino-Gomes, D.; Lambeth, J.D.; Lambeth, J.D. Nox4: A hydrogen peroxide-generating oxygen sensor. Biochemistry 2014, 53, 5111–5120. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Zhao, F.Q.; Zhang, Z.W.; Qu, J.P.; Yao, H.D.; Li, M.; Li, S.; Xu, S.W. Cold stress induces antioxidants and Hsps in chicken immune organs. Cell Stress Chaperones 2014, 19, 635–648. [Google Scholar] [CrossRef]
- Zhang, Z.W.; Lv, Z.H.; Li, J.L.; Li, S.; Xu, S.W.; Wang, X.L. Effects of cold stress on nitric oxide in duodenum of chicks. Poult. Sci. 2011, 90, 1555–1561. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Kidd, M.T. Antioxidant defence systems and oxidative stress in poultry biology: An update. Antioxidants 2019, 8, 235. [Google Scholar] [CrossRef] [PubMed]
- Baghbanzadeh, A.; Decuypere, E. Ascites syndrome in broilers: Physiological and nutritional perspectives. Avian Pathol. 2008, 37, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Tsiouris, V. Poultry management: A useful tool for the control of necrotic enteritis in poultry. Avian Pathol. 2016, 45, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Dadgar, S.; Crowe, T.G.; Classen, H.L.; Watts, J.M.; Shand, P.J. Broiler chicken thigh and breast muscle responses to cold stress during simulated transport before slaughter. Poult. Sci. 2012, 91, 1454–1464. [Google Scholar] [CrossRef] [PubMed]
- Olfati, A.; Mojtahedin, A.; Sadeghi, T.; Akbari, M.; Martínez-Pastor, F. Comparison of growth performance and immune responses of broiler chicks reared under heat stress, cold stress and thermoneutral conditions. Span. J. Agric. Res. 2018, 16. [Google Scholar] [CrossRef]
- Tsiouris, V.; Georgopoulou, I.; Batzios, C.; Pappaioannou, N.; Ducatelle, R.; Fortomaris, P. The effect of cold stress on the pathogenesis of necrotic enteritis in broiler chicks. Avian Pathol. 2015, 44, 430–435. [Google Scholar] [CrossRef]
- Yalcin, S.; Siegel, P. Exposure to cold or heat during incubation on developmental stability of broiler embryos. Poult. Sci. 2003, 82, 1388–1392. [Google Scholar] [CrossRef]
- Yahav, S.; Rath, R.S.; Shinder, D. The effect of thermal manipulations during embryogenesis of broiler chicks (Gallus domesticus) on hatchability, body weight and thermoregulation after hatch. J. Therm. Biol. 2004, 29, 245–250. [Google Scholar] [CrossRef]
- Piestun, Y.; Shinder, D.; Ruzal, M.; Halevy, O.; Brake, J.; Yahav, S. Thermal manipulations during broiler embryogenesis: Effect on the acquisition of thermotolerance. Poult. Sci. 2008, 87, 1516–1525. [Google Scholar] [CrossRef]
- Piestun, Y.; Shinder, D.; Ruzal, M.; Halevy, O.; Yahav, S. The effect of thermal manipulations during the development of the thyroid and adrenal axes on in-hatch and post-hatch thermoregulation. J. Therm. Biol. 2008, 33, 413–418. [Google Scholar] [CrossRef]
- Al-Zghoul, M.B.; Saleh, K.M.; Ababneh, M.M.K. Effects of pre-hatch thermal manipulation and post-hatch acute heat stress on the mRNA expression of interleukin-6 and genes involved in its induction pathways in 2 broiler chicken breeds. Poult. Sci. 2019, 98, 1805–1819. [Google Scholar] [CrossRef] [PubMed]
- Al-Zghoul, M.B.; Alliftawi, A.R.S.; Saleh, K.M.M.; Jaradat, Z.W. Expression of digestive enzyme and intestinal transporter genes during chronic heat stress in the thermally manipulated broiler chicken. Poult. Sci. 2019, 98, 4113–4122. [Google Scholar] [CrossRef] [PubMed]
- Cooper-Mullin, C.; McWilliams, S.R. The role of the antioxidant system during intense endurance exercise: Lessons from migrating birds. J. Exp. Biol. 2016, 219, 3684–3695. [Google Scholar] [CrossRef] [PubMed]
- Saleh, K.M.M.; Al-Zghoul, M.B. Effect of acute heat stress on the mrna levels of cytokines in broiler chickens subjected to embryonic thermal manipulation. Animals 2019, 9, 499. [Google Scholar] [CrossRef] [PubMed]
- Al-Zghoul, M.B.; El-Bahr, S.M. Thermal manipulation of the broilers embryos: Expression of muscle markers genes and weights of body and internal organs during embryonic and post-hatch days. BMC Vet. Res. 2019, 15, 166. [Google Scholar] [CrossRef]
- Zaboli, G.-R.; Rahimi, S.; Shariatmadari, F.; Torshizi, M.A.K.; Baghbanzadeh, A.; Mehri, M. Thermal manipulation during Pre and Post-Hatch on thermotolerance of male broiler chickens exposed to chronic heat stress. Poult. Sci. 2017, 96, 478–485. [Google Scholar] [CrossRef]
- Mack, L.A.; Felver-Gant, J.N.; Dennis, R.L.; Cheng, H.W. Genetic variations alter production and behavioral responses following heat stress in 2 strains of laying hens. Poult. Sci. 2013, 92, 285–294. [Google Scholar] [CrossRef]
- Loyau, T.; Berri, C.; Bedrani, L.; Métayer-Coustard, S.; Praud, C.; Duclos, M.J.; Tesseraud, S.; Rideau, N.; Everaert, N.; Yahav, S.; et al. Thermal manipulation of the embryo modifies the physiology and body composition of broiler chickens reared in floor pens without affecting breast meat processing quality1. J. Anim. Sci. 2013, 91, 3674–3685. [Google Scholar] [CrossRef]
- Piestun, Y.; Druyan, S.; Brake, J.; Yahav, S. Thermal manipulations during broiler incubation alter performance of broilers to 70 days of age. Poult. Sci. 2013, 92, 1155–1163. [Google Scholar] [CrossRef]
- Yahav, S.; Collin, A.; Shinder, D.; Picard, M. Thermal manipulations during broiler chick embryogenesis: Effects of timing and temperature. Poult. Sci. 2004, 83, 1959–1963. [Google Scholar] [CrossRef] [PubMed]
- Piestun, Y.; Halevy, O.; Shinder, D.; Ruzal, M.; Druyan, S.; Yahav, S. Thermal manipulations during broiler embryogenesis improves post-hatch performance under hot conditions. J. Therm. Biol. 2011, 36, 469–474. [Google Scholar] [CrossRef]
- Altan, Ö.; Pabuçcuoǧlu, A.; Altan, A.; Konyalioǧlu, S.; Bayraktar, H. Effect of heat stress on oxidative stress, lipid peroxidation and some stress parameters in broilers. Br. Poult. Sci. 2003, 44, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Decuypere, E.; Buyse, J. Acute heat stress induces oxidative stress in broiler chickens. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2006, 144, 11–17. [Google Scholar] [CrossRef]
- Günal, M. The effects of early-age thermal manipulation and daily short-term fasting on performance and body temperatures in broiler exposed to heat stress. J. Anim. Physiol. Anim. Nutr. 2012, 97, 854–860. [Google Scholar] [CrossRef]
- Loyau, T.; Métayer-Coustard, S.; Berri, C.; Crochet, S.; Cailleau-Audouin, E.; Sannier, M.; Chartrin, P.; Praud, C.; Hennequet-Antier, C.; Rideau, N.; et al. Thermal manipulation during embryogenesis has long-term effects on muscle and liver metabolism in fast-growing chickens. PLoS ONE 2014, 9, e105339. [Google Scholar] [CrossRef]
- Collin, A.; Berri, C.; Tesseraud, S.; Rodon, F.E.R.; Skiba-Cassy, S.; Crochet, S.; Duclos, M.J.; Rideau, N.; Tona, K.; Buyse, J.; et al. Effects of thermal manipulation during early and late embryogenesis on thermotolerance and breast muscle characteristics in broiler chickens. Poult. Sci. 2007, 86, 795–800. [Google Scholar] [CrossRef]
- Zamocky, M.; Furtmüller, P.G.; Obinger, C. Evolution of catalases from bacteria to humans. Antioxid. Redox Signal. 2008, 10, 1527–1547. [Google Scholar] [CrossRef] [PubMed]
- Chelikani, P.; Fita, I.; Loewen, P.C. Diversity of structures and properties among catalases. Cell. Mol. Life Sci. 2004, 61, 192–208. [Google Scholar] [CrossRef]
- Del Vesco, A.P.; Khatlab, A.S.; Goes, E.S.R.; Utsunomiya, K.S.; Vieira, J.S.; Oliveira Neto, A.R.; Gasparino, E. Age-related oxidative stress and antioxidant capacity in heat-stressed broilers. Animal 2017, 11, 1783–1790. [Google Scholar] [CrossRef]
- Wei, H.; Zhang, R.; Su, Y.; Bi, Y.; Li, X.; Zhang, X.; Li, J.; Bao, J. Effects of acute cold stress after long-term cold stimulation on antioxidant status, heat shock proteins, inflammation and immune cytokines in broiler heart. Front. Physiol. 2018, 9, 1589. [Google Scholar] [CrossRef] [PubMed]
- Jeong, B.Y.; Lee, H.Y.; Park, C.G.; Kang, J.; Yu, S.-L.; Choi, D.; Han, S.-Y.; Park, M.H.; Cho, S.; Lee, S.Y.; et al. Oxidative stress caused by activation of NADPH oxidase 4 promotes contrast-induced acute kidney injury. PLoS ONE 2018, 13, e0191034. [Google Scholar] [CrossRef] [PubMed]
- Vendrov, A.E.; Vendrov, K.C.; Smith, A.; Yuan, J.; Sumida, A.; Robidoux, J.; Runge, M.S.; Madamanchi, N.R. NOX4 NADPH oxidase-dependent mitochondrial oxidative stress in aging-associated cardiovascular disease. Antioxid. Redox Signal. 2015, 23, 1389–1409. [Google Scholar] [CrossRef] [PubMed]
- Kikusato, M.; Yoshida, H.; Furukawa, K.; Toyomizu, M. Effect of heat stress-induced production of mitochondrial reactive oxygen species on NADPH oxidase and heme oxygenase-1 mRNA levels in avian muscle cells. J. Therm. Biol. 2015, 52, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Velarde, M.C.; Flynn, J.M.; Day, N.U.; Melov, S.; Campisi, J. Mitochondrial oxidative stress caused by Sod2 deficiency promotes cellular senescence and aging phenotypes in the skin. Aging Albany. NY 2012, 4, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, S.; Lasagna, E.; Sarti, F.M.; Marelli, S.P.; Cozzi, M.C.; Bernardini, G.; Terova, G. Expression profile of six stress-related genes and productive performances of fast and slow growing broiler strains reared under heat stress conditions. Meta Gene 2015, 6, 17–25. [Google Scholar] [CrossRef]
- Breschi, A.; Djebali, S.; Gillis, J.; Pervouchine, D.D.; Dobin, A.; Davis, C.A.; Gingeras, T.R.; Guigó, R. Gene-specific patterns of expression variation across organs and species. Genome Biol. 2016, 17, 151. [Google Scholar] [CrossRef]
- Scheuermann, G.N.; Bilgili, S.F.; Hess, J.B.; Mulvaney, D.R. Breast muscle development in commercial broiler chickens. Poult. Sci. 2003, 82, 1648–1658. [Google Scholar] [CrossRef]
- Kuttappan, V.A.; Hargis, B.M.; Owens, C.M. White striping and woody breast myopathies in the modern poultry industry: A review. Poult. Sci. 2016, 95, 2724–2733. [Google Scholar] [CrossRef]
- Huang, X.; Ahn, D.U. The incidence of muscle abnormalities in broiler breast meat—A review. Korean J. Food Sci. Anim. Resour. 2018, 38, 835–850. [Google Scholar] [CrossRef]
- Kawasaki, T.; Iwasaki, T.; Yamada, M.; Yoshida, T.; Watanabe, T. Rapid growth rate results in remarkably hardened breast in broilers during the middle stage of rearing: A biochemical and histopathological study. PLoS ONE 2018, 13, e0193307. [Google Scholar] [CrossRef] [PubMed]
- Zahoor, I.; De Koning, D.J.; Hocking, P.M. Transcriptional profile of breast muscle in heat stressed layers is similar to that of broiler chickens at control temperature. Genet. Sel. Evol. 2017, 49, 69. [Google Scholar] [CrossRef] [PubMed]
| Gene | Forward (5′ to 3′) | Reverse (5′ to 3′) |
|---|---|---|
| NOX4 | CCAGACCAACTTAGAGGAACAC | TCTGGGAAAGGCTCAGTAGTA |
| SOD2 | CTGACCTGCCTTACGACTATG | CGCCTCTTTGTATTTCTCCTCT |
| Catalase | GAAGCAGAGAGGTTCCCATTTA | CATACGCCATCTGTTCTACCTC |
| 28S rRNA | CCTGAATCCCGAGGTTAACTATT | GAGGTGCGGCTTATCATCTATC |
| Post-Hatch Day | Control | TM | |
|---|---|---|---|
| TC (°C) | 1 | 39.63 ± 0.24 a | 39.48 ± 0.23 b |
| 37 | 39.05 ± 0.24 a | 38.38 ± 0.22 b | |
| BW (g) | 1 | 44.2 ± 3.8 a | 42.7 ± 2.8 a |
| 37 | 2302.8 ± 79.7 a | 2440 ± 82.5 b |
| Normal Conditions | Heat Stress | |||
|---|---|---|---|---|
| (21 °C; RH 45%–52%) | (35 °C; RH 45%–52%) | |||
| Control | TM | Control | TM | |
| TC (°C) | 39.65 ± 0.28 a | 39.08 ± 0.21 b | 41.35 ± 0.24 c | 40.15 ± 0.26 d |
| BW (g) | ||||
| Day 0 (PD 28) | 1373.3 ± 51.3 a | 1675.7 ± 83.8 b | 1456.7 ± 82.8 a | 1704 ± 74.4 b |
| Day 7 (PD 35) | 1847.9 ± 108.1 a | 2108.8 ± 95.8 b | 1645 ± 40.6 c | 1930 ± 50.2 d |
| BW gain (g) | 474.6 ± 70.9 a | 433 ± 39.1 a | 188.3 ± 47.3 b | 226 ± 34.7 b |
| Normal Conditions | Cold Stress | |||
|---|---|---|---|---|
| (21 °C; RH 45%–52%) | (16 °C; RH 45%–52%) | |||
| Control (CN) | TM (TN) | Control (CS) | TM (TS) | |
| TC (°C) | 39.18 ± 0.35 a | 38.5 ± 0.34 b | 39.3 ± 0.2 a | 38.93 ± 0.32 ab |
| BW (g) | ||||
| Day 0 (PD 32) | 1717.9 ± 137.5 a | 1834 ± 112.8 a | 1720 ± 147.5 a | 1845.5 ± 119.9 a |
| Day 5 (PD 37) | 2244.3 ± 134.4 a | 2281 ± 101.9 a | 1993.1 ± 131.3 b | 2179 ± 134.8 a |
| BW gain (g) | 526.4 ± 41.1 a | 447 ± 42.2 b | 273.1 ± 23.9 c | 333.5 ± 53.2 d |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saleh, K.M.M.; Tarkhan, A.H.; Al-Zghoul, M.B. Embryonic Thermal Manipulation Affects the Antioxidant Response to Post-Hatch Thermal Exposure in Broiler Chickens. Animals 2020, 10, 126. https://doi.org/10.3390/ani10010126
Saleh KMM, Tarkhan AH, Al-Zghoul MB. Embryonic Thermal Manipulation Affects the Antioxidant Response to Post-Hatch Thermal Exposure in Broiler Chickens. Animals. 2020; 10(1):126. https://doi.org/10.3390/ani10010126
Chicago/Turabian StyleSaleh, Khaled M. M., Amneh H. Tarkhan, and Mohammad Borhan Al-Zghoul. 2020. "Embryonic Thermal Manipulation Affects the Antioxidant Response to Post-Hatch Thermal Exposure in Broiler Chickens" Animals 10, no. 1: 126. https://doi.org/10.3390/ani10010126
APA StyleSaleh, K. M. M., Tarkhan, A. H., & Al-Zghoul, M. B. (2020). Embryonic Thermal Manipulation Affects the Antioxidant Response to Post-Hatch Thermal Exposure in Broiler Chickens. Animals, 10(1), 126. https://doi.org/10.3390/ani10010126





