1. Introduction
Tetrahymena rostrata (Kahl, 1926; Corliss 1952) (Ciliophora: Oligohymenophorea) has been shown to infect and kill species of land slugs and snails and has been proposed as a possible biological control agent of pest mollusks. This paper examines the infectivity of different life stages of T. rostrata to terrestrial mollusks with a view to advance these prospects.
Tetrahymena rostrata was first isolated from environmental samples, putrescent
Glyceria [
1], moss [
2] and leaf litter [
3]. Four developmental stages of
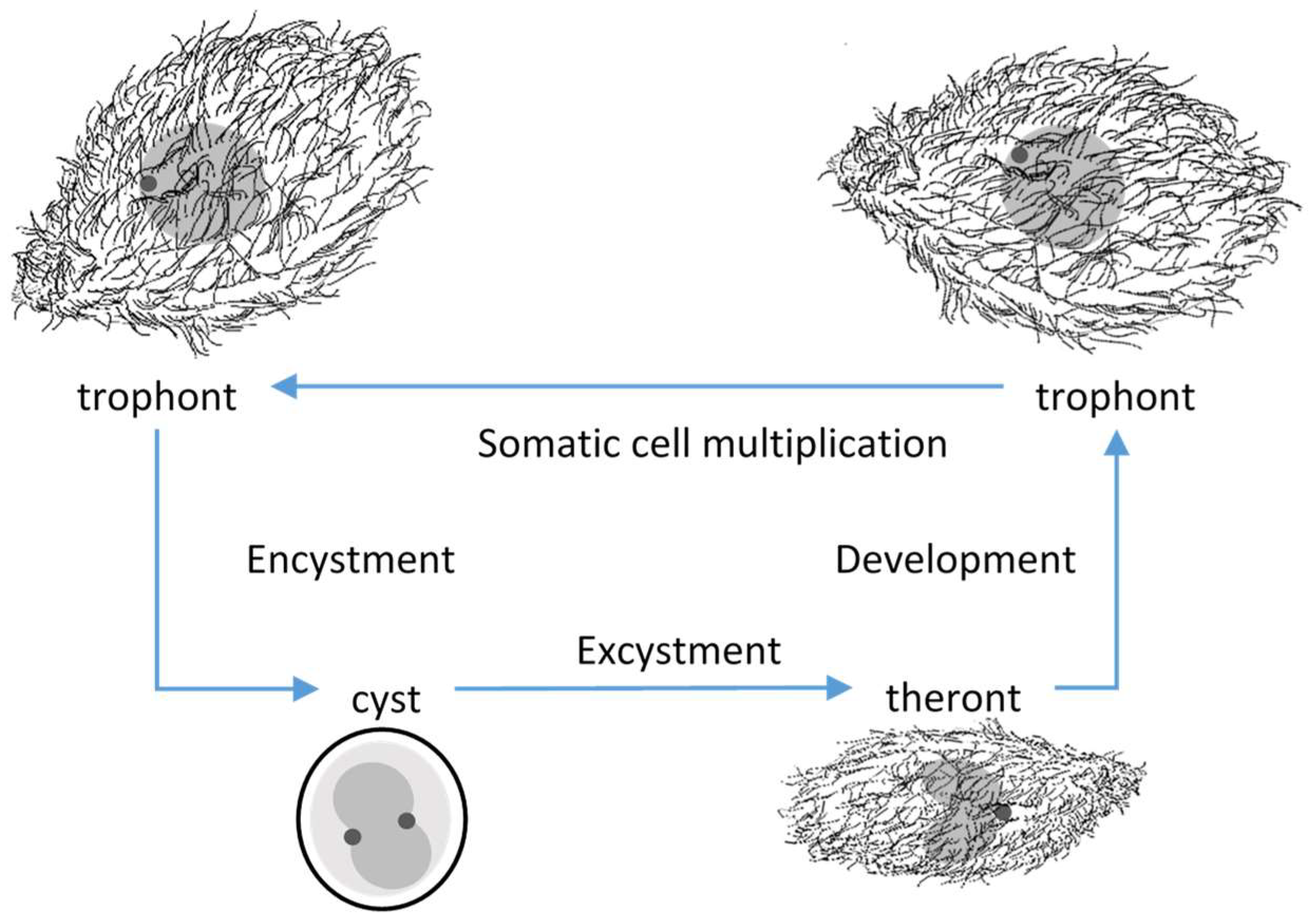
T. rostrata could be distinguished; trophonts, tomonts, tomites and theronts [
2,
3] (
Figure 1). The pyriform, free-swimming, feeding, trophonts occur when food sources are available. Trophonts are highly plastic and multiply by mitosis. In the absence of food, trophonts become fast-swimming tomites and secrete mucins, which form a soft capsule around tomonts, which develop into cysts in which meiosis occurs. Excystment occurs spontaneously or in response to the availability of nutrients. Theronts, which emerge from the cyst capsules, are attracted to proteinaceous materials and convert into trophonts after feeding [
3,
4].
T. rostrata can be free living and can be grown in an axenic culture. However, natural infections have been recorded in enchytraeid worms,
Enchytraeidae [
3],
Deroceras reticulatum (Müller 1744) slugs [
5,
6,
7] and some species of snail [
4,
7,
8]. Experimental infections of
D. reticulatum with
T. rostrata trophonts suggested that the ciliates enter the slug via a pouch under the mantle and then migrate primarily to the renal tissues and later to other organs, resulting in the death of the host [
5].
The life cycles of many other ciliates which undergo encystment, have been described but the role of the theronts has received relatively little attention. This is possibly due to the technical difficulty of separating theronts and trophonts in cultures. The best studied theronts are from the commercially important fish parasites,
Ichthyophthirius multifiliis (Fouquet, 1876) (Ciliophora; Oligohymenophorea) [
9,
10] and
Cryptocaryon irritans (Brown, 1951) (Ciliophora: Prostomatea) [
11,
12] and the bivalve parasite
Ophryoglena hemophaga (Molloy, Lynn and Giamberini, 2005) (Ciliophora: Ophryoglenidae) [
13], whose theronts are short-lived if they do not find a suitable host. In contrast, the free-living aquatic ciliates,
Holophrya teres (Ehrenberg, 1834) (Ciliophora: Prostomatea) and
Prorodon aklitolophon ((Hiller and Bardele, 1988) (Ciliophora: Prostomatea) form reproductive cysts, which release theronts that can last for several days [
14,
15]. However, if food does not become available, the theronts form a thin mucous coating and re-encyst. Successive rounds of spontaneous excystment and re-encystment result in gradual reduction in the size of the theronts until they are too small to feed. In the case of
P. aklitolophon, theronts survived under continual starvation for up to 8 weeks [
15].
The only published experimental infections with
T. rostrata were performed using
D. reticulatum, and those studies utilized trophonts derived from axenic cultures [
5,
16]. In this paper, we show that
T. rostrata theronts are more effective at establishing lethal infections of slugs than are trophonts, and that theronts can be long-lived in the absence of nutrients.
2. Materials and Methods
2.1. Tetrahymena rostrata
T. rostrata isolate TRAUS was originally isolated from the egg of a
D. reticulatum slug collected in Melbourne, Australia, in 2014 [
17]. Cultures of
T. rostrata were maintained at 20 °C in the dark in sterile PPYE medium (0.5%
w/v proteose peptone (Oxoid, LP0085), 0.5%
w/v yeast extract (Oxoid, LP0021) and 0.125%
w/v glucose), and subcultures were performed fortnightly. Cultures prepared for encystment were grown in sterile PP medium (1%
w/v proteose peptone (Oxoid, LP0085), and 0.125%
w/v glucose prior to nutrient starvation. The taxonomic systematics of Lynn, 2008 were followed when referring to ciliate species [
18].
2.2. Preparation of Theronts
The encystment buffer was prepared by mixing 50 g of medium grade composted pine bark (Australian Growing Solutions) in 1.2 L of MilliQ ultrapure water for 15 min. The mixture was sieved, and the flow-through was centrifuged at 300× g for 10 min to remove large solids. The decanted supernatant was sterilized by autoclaving at 121 °C for 20 min. N-(2-Hydroxyethyl)piperazine-N′-(2-ethanesulfonic acid) (HEPES) pH7 (NaOH) was added to a final concentration of 10mM prior to use.
T. rostrata cells from exponentially growing PP cultures were collected by centrifugation (800× g, 10 min) and washed with 10 mM HEPES pH 7 NaOH, and then resuspended in encystment buffer at a final density of 1–3 × 104 cells/mL. Cells were placed into 6-well plates (Greiner Bio-one Cat No. 657 185) in 3 mL aliquots or in T150 vented tissue culture flasks in 100 mL aliquots and incubated at 26 °C to allow cysts to form. After 1–3 days at 26 °C, the cyst cultures were transferred to 20 °C to excyst and release the theronts. Excystment started on day 5 and peaked at day 7, with 85 to 90% of the cysts being excysted. Theronts were stored in the encystment buffer at 20 °C until use.
Preparations of theronts, free from unexcysted cells, were used to infect slugs for histology. This involved excystment as above, followed by centrifugation at 300× g for 10 min to collect the cells and debris into a pellet, after which the tubes were left undisturbed for 2 h to allow the theronts to swim upwards and be harvested from the supernatant with a pipette. Trophonts used for the challenge experiments were derived from cysts and cultured in PPYE for 4 days to stimulate excystment and conversion to trophonts. Trophonts were washed free of media using 10 mM HEPES pH7 NaOH immediately before use in a slug challenge experiment. The numbers of trophonts and theronts in the cultures were determined, using a Fuchs–Rosenthal counting chamber with a Leica DMLS light microscopy, and adjusted to the desired concentration by harvesting the cells by centrifugation (800× g, 10 min) or dilution in 10 mM HEPES pH7 NaOH.
Wet mounts and fixed Giemsa stained cells were imaged with a Leica DMLS light microscopy and used to confirm cyst, theront and trophont forms. Viable counts were determined using most probable number method, using PPYE as the medium [
19].
2.3. Dose and Lethality Experiments
Laboratory-reared
D. reticulatum were maintained as previously described [
20]. Young slugs, ~0.8–1 cm long, were selected for all experiments. Each slug was housed in a 25 mL tube containing 3 g of moist potting soil (Plugger 111-Seedraising Mix). The tubes were placed in a humidified box and kept in a controlled environment at 17 °C with a 12 h photoperiod. There were 20 slugs per treatment. The slug’s grazing activity and mortality was assessed daily or weekly, depending on the experiment. Each slug was provided with Chinese cabbage for food, and the cabbage was replaced as required. For dose experiments, 20 replicates of individually housed
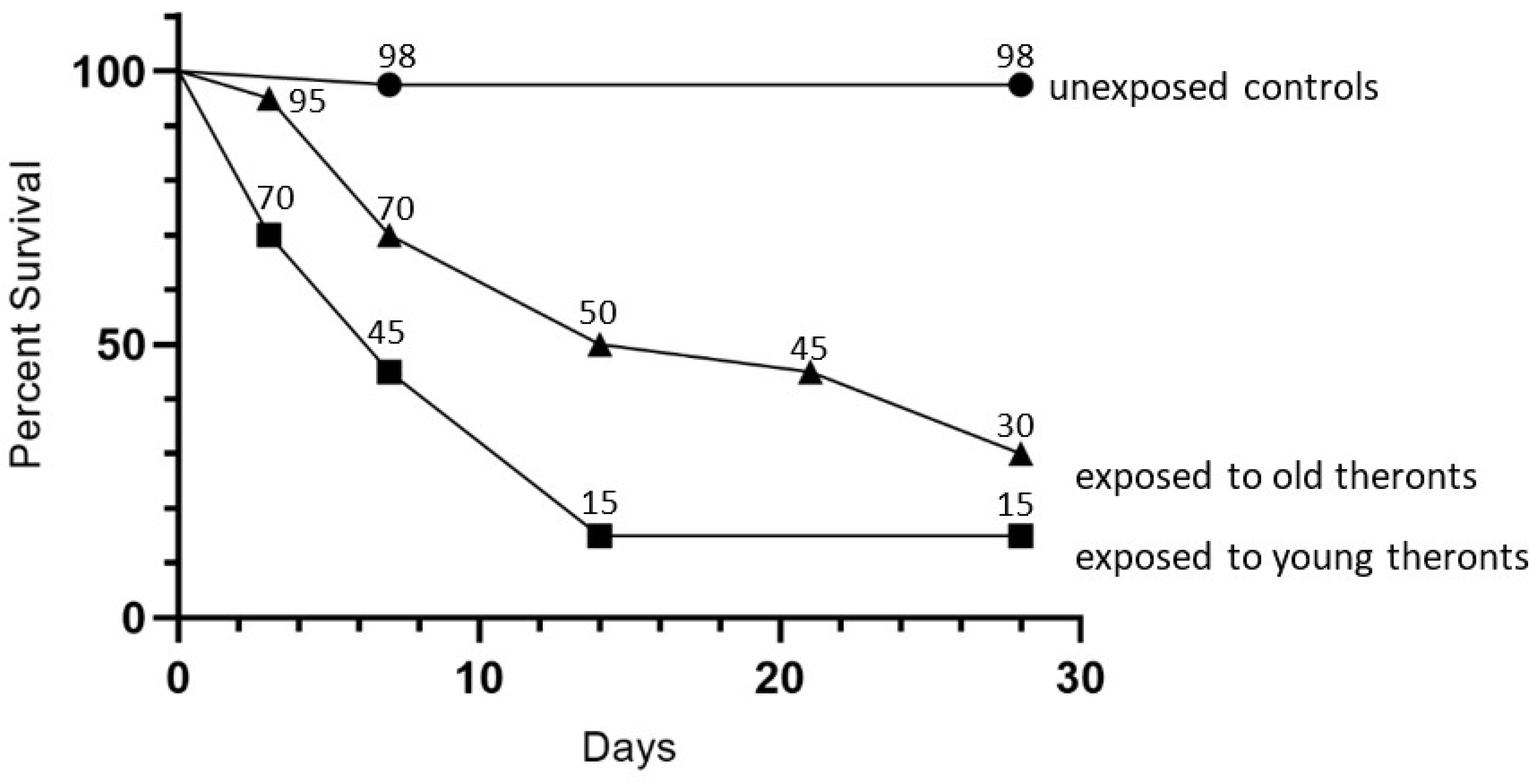
D. reticulatum slugs were exposed to 100,000, 30,000, 10,000, 3000, 1000 or zero trophonts or theronts. The testing of starved theronts was performed, using a dose of 10
5 theronts, which had all excysted over the preceding 7 days and were used immediately or kept in the encystment buffer at 20 °C until use. Kaplan–Meier plots and analyses were performed in GraphPad Prism 9.0.1.
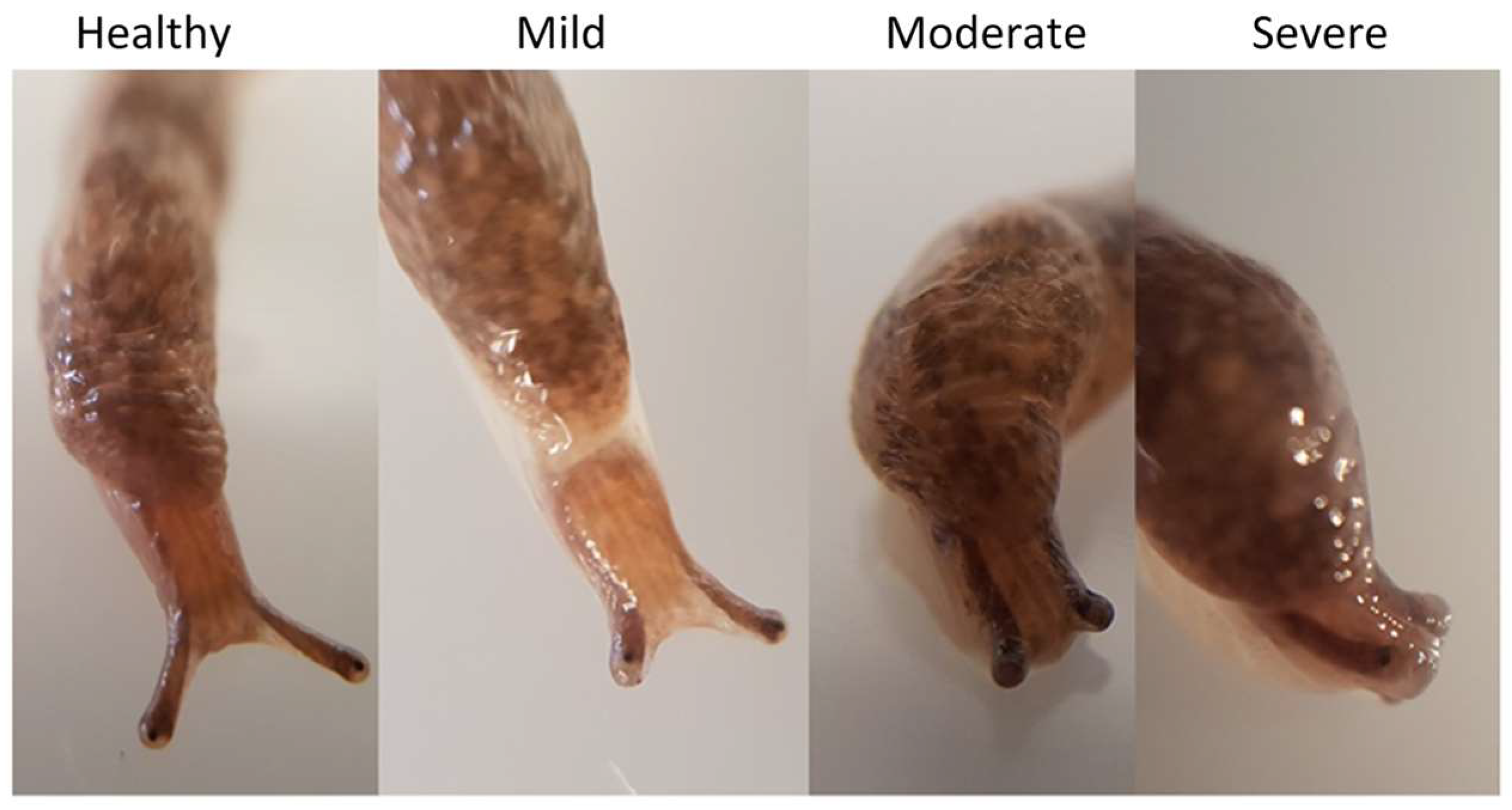
To examine behavioral changes and histology in experimental infections of D. reticulatum, slugs ~1 cm long, were placed individually in 5 cm diameter lidded plastic pots lined with damp filter paper. Half a circular piece of cabbage 2.3 cm in diameter was added to each container. Treated slugs received 8.4 × 103 theronts applied to the filter paper and food. Control slugs were exposed to a mock inoculum of the same volume of buffered solution of encystment buffer. The slugs were monitored daily for early signs of infection, such as a swollen mantle, inability to extend the superior tentacles and reduced locomotion. Reduced extension of the superior tentacles commonly occurred in the days preceding death and was an indication that a slug had a lethal infection. These criteria were used to select moribund slugs for histology to determine the site of infection. D. reticulatum slugs were exposed to T. rostrata theronts as above and, after 7 days of exposure, 10 slugs from the control group and 26 moribund slugs from the theront-exposed group were selected and euthanized by submersion in soda water, fixed in neutral buffered formalin for 48 h and then transferred to 70% ethanol. The fixed samples were embedded in paraffin wax and sectioned transversely in sections 5 µm thick, which were then were mounted on glass slides and stained with haematoxylin and eosin (H & E). The slides were examined, using a Leica DMLS light microscope. Statistical analysis was performed using GraphPad Prism version 9.0.1.
4. Discussion
There are eight classes of ciliates and six species of
Tetrahymena that can form resting cysts [
18]. The triggers for ciliate excystment are highly variable [
21]. For
T. rostrata, triggers of excystment include media containing peptone and tryptone or milk, kidney extract, mollusk mucous and changes in temperature or salinity [
3,
4,
22]. Spontaneous excystment of reproductive cysts is frequent, indicating that metabolic triggers also exist [
4,
21,
22]. Our method of preparation of cysts results in highly efficient, synchronized encystment with almost 100% of cells encysting. Synchronized excystment results in almost pure populations of theronts, which can be further separated from unencysted or dead cells, if required. These methods enabled us to compare the performance of trophonts and theronts in slug infection experiments.
Previous studies have shown that
T. rostrata trophonts are capable of infecting and killing
D. reticulatum slugs [
5], and naturally-infected specimens of a variety of slugs and snails have been described. Clearly,
T. rostrata trophonts are infective. In edaphic environments, ciliates, such as
T. rostrata, would be expected to occur as trophonts in a growth-division cycle when moisture and nutrients are not limiting and in an encystment cycle when conditions are less favorable.
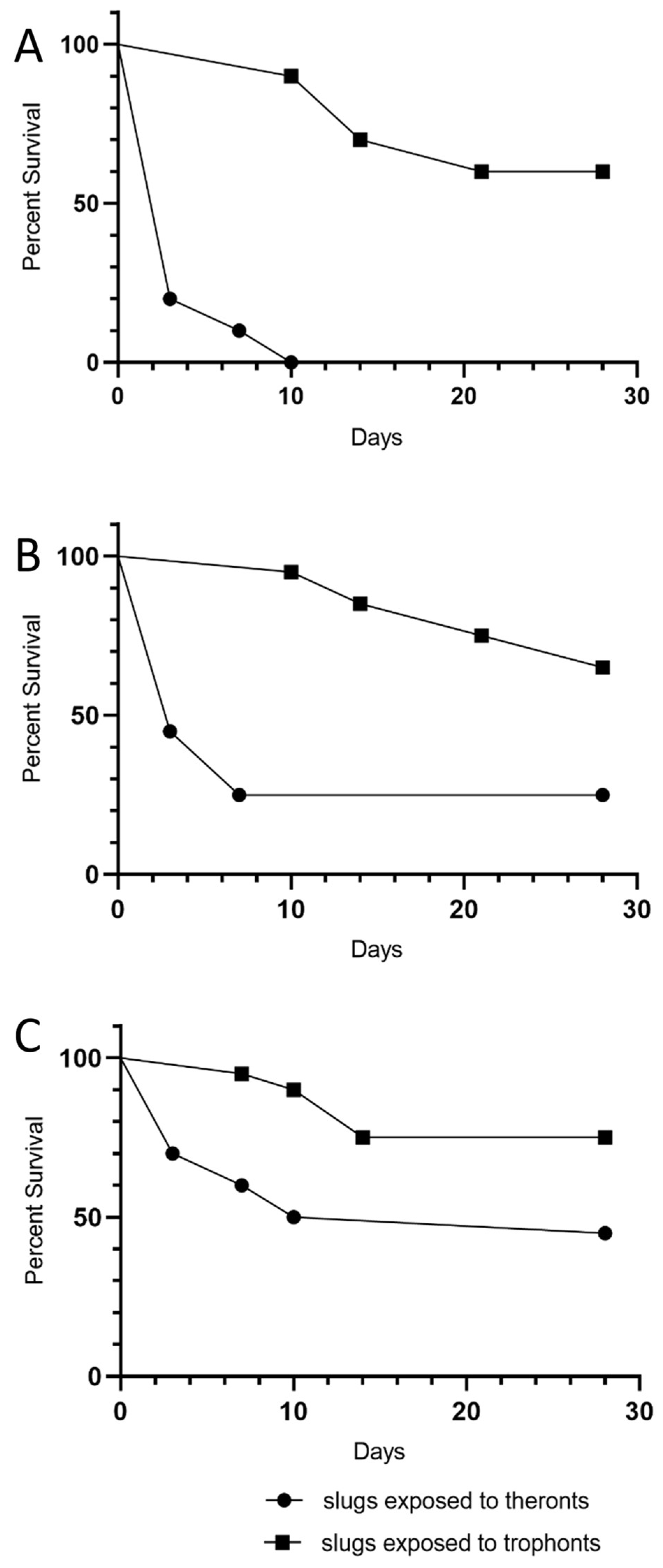
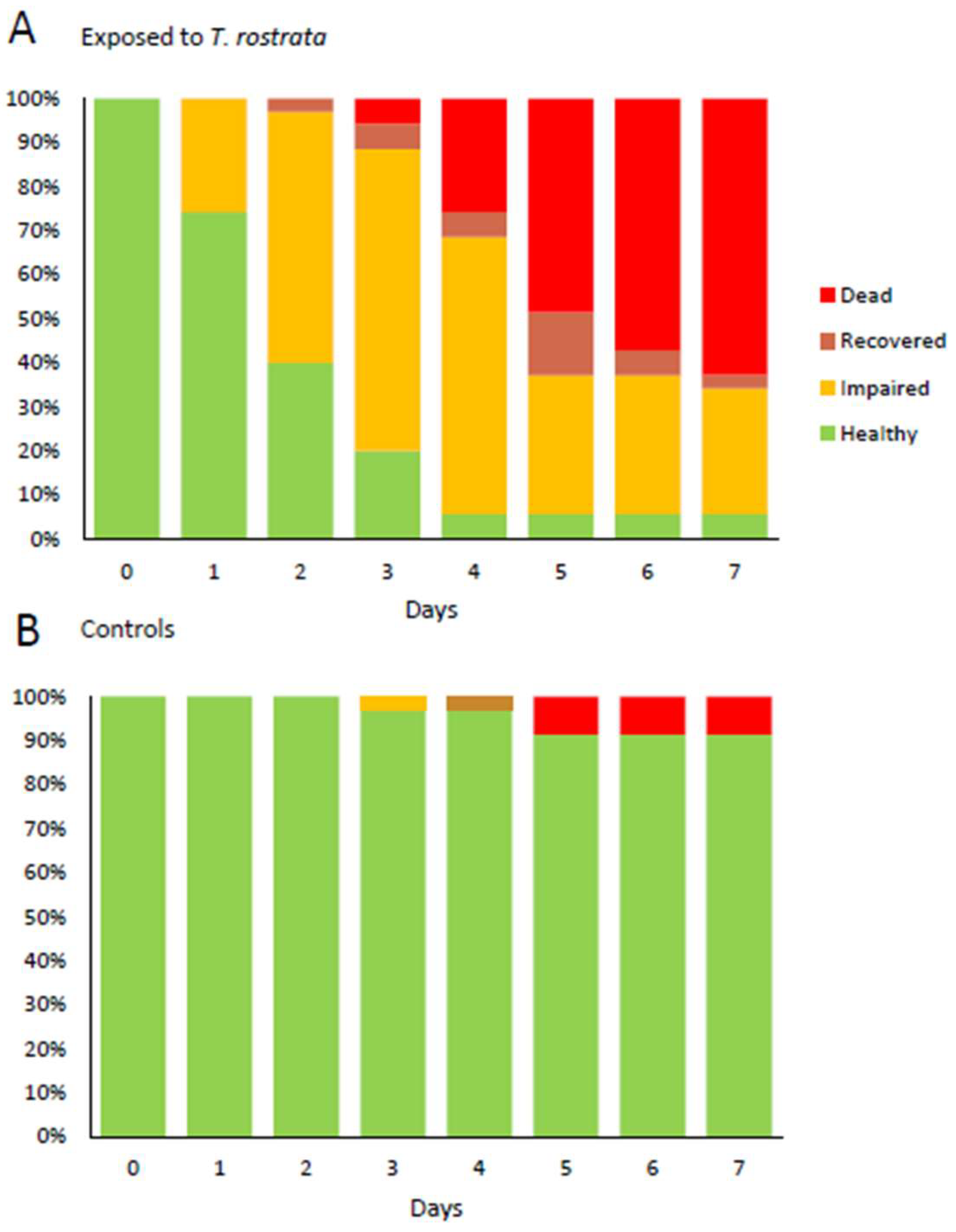
Our experiments showed that exposure to theronts was more likely to result in rapid slug death than exposure to the same dose of trophonts. Tests with trophonts may have resulted in the same eventual death rate if the experiment had gone long enough. Following infection and initial feeding, the theronts convert to trophonts, which cause tissue damage and ultimately kill the slugs. This may be an important consideration if
T. rostrata is to be used as a biopesticide for the control of pest slugs. We used young slugs in these studies and housed them individually. Slugs are known to exhibit huddling behavior when they are threatened with dehydration [
23]. It would be interesting to measure the efficacy of theronts against different sizes of slugs, housed singly and in groups.
Soil ciliates are expected to occur largely as cysts in soil and to enter the vegetative growth phase periodically when conditions are favorable. The extensive studies of the late W. Foissner are testament to the persistence of cysts in soil (for example, [
24]). The longevity and infectivity of starved
T. rostrata theronts described here was unexpected. We are cautious about extrapolating from controlled laboratory conditions to environmental situations. However, it may be possible that, under some conditions, theronts might persist for prolonged periods until they encounter a suitable host mollusk or other nutrient source. In the slug exposure experiment, the trophonts and theronts may have several responses. They may remain in their developmental form and infect a slug or remain in the substrate. Alternatively, they may die or change their developmental form. We know at least some theronts developed into trophonts, as these are what we see in the histological sections. Trophonts could encyst and release theronts. It is most likely that all of these changes occur within the population depending on the microniches that cells encounter.
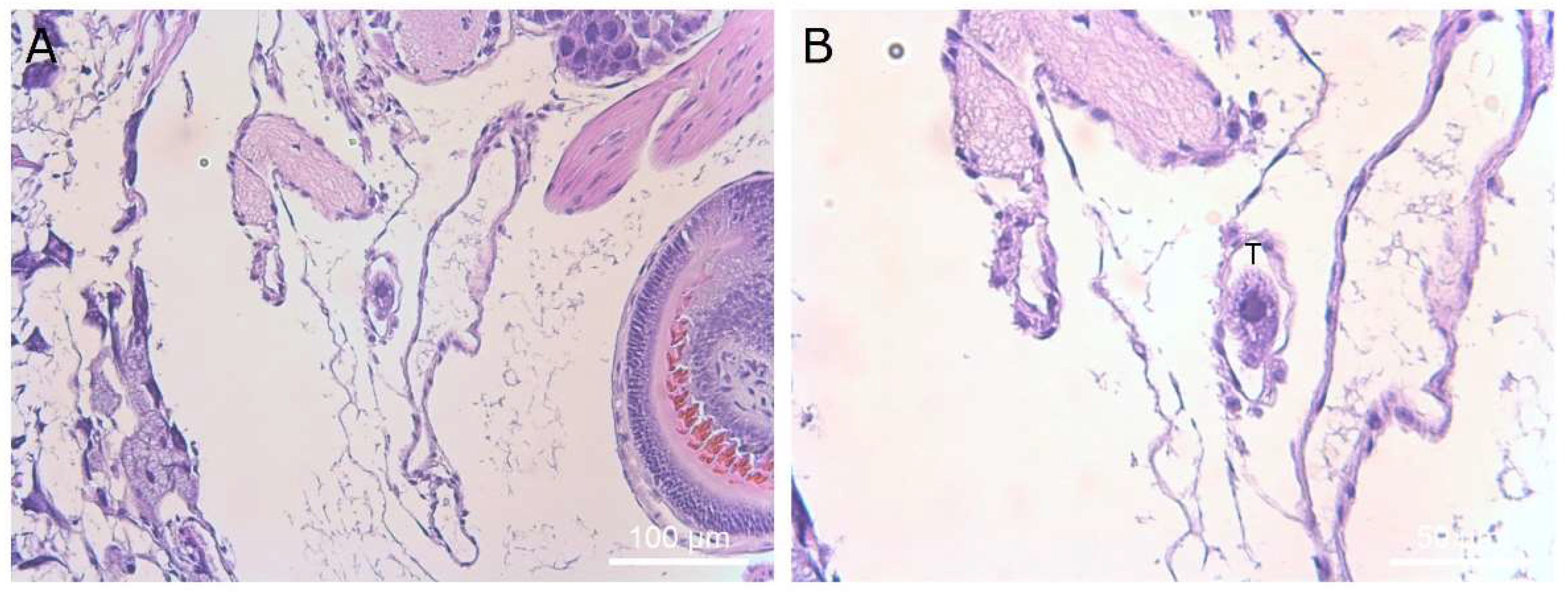
The occurrence of reduced mobility of the superior tentacles was a strong indicator of the subsequent death of slugs exposed to theronts. There was no histological evidence of damage to the superior tentacles, nor were any ciliates identified in the area. The ciliates were found predominantly in the kidney and pulmonary chamber. The ciliates found in the kidney appeared to be multiplying, as dividing cells were observed. It is possible that T. rostrata is attracted to metabolites concentrated in the kidney, such as urea or purines.
The presence of individual ciliates in places other than the kidney in these slugs suggests that they were mobile within the slugs. The histological sectioning also provides insight into the life stage of the ciliates inside the slugs. Theronts have a characteristic lobulated macronucleus, whereas the ciliates that were present in the sectioned slugs showed the characteristic form of trophonts with a single round macronucleus and associated micronucleus. This was not unexpected, as the transformation from theront to trophont in the presence of suitable food takes only a few hours, and the slugs were sectioned 7 days after they were exposed to theronts. We placed 6-week-old theronts onto autoclaved slug tissues and observed them immediately feeding, with the majority of theronts converting to trophonts within 4 h (data not shown).
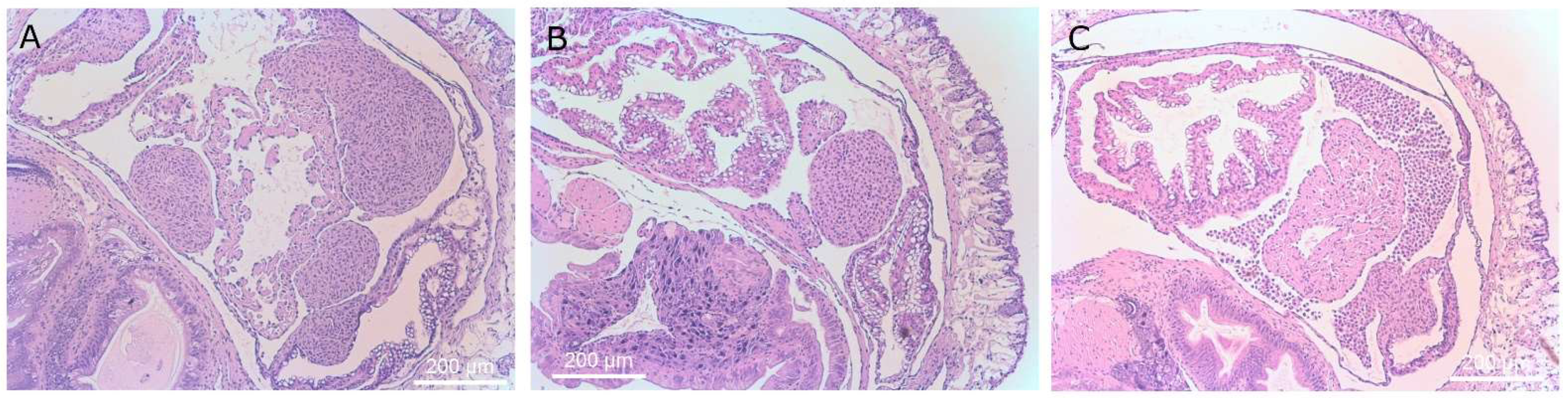
Trophonts were most abundant in the saccular portion of the kidneys and were associated with damage to the renal cells, leaving the basal cells intact. These finding reinforce those of Brooks [
5] who concluded that the mechanism for
T. rostrata in the destruction of the kidney was enzymatic, mechanical, or both [
5]. It is likely that slugs with renal tissue damage would have impaired ability maintain their water balance and the behavior observed, i.e., contracted postures with retracted tentacles, is consistent with slugs that are dehydrated [
23].
Ciliates were seen in the muscle, between the skin and muscle layers of the slug and in the interstitial space. The ciliates in the muscle appeared smaller than those in the kidney, possibly because of space constraints or because those niches are not so conducive to feeding and growth. Ciliates were also found individually adjacent to the developing gonads of some slugs. However, these slugs used were young, and the gonads were not fully developed. Infection of the albumen gland was reported in naturally infected D
. reticulatum and was associated with transovarial transmission [
5]. It is worth noting that the
T. rostrata isolate used in these studies was originally isolated from the albumen of a
D. reticulatum egg laid by an infected parent. It is surprising that
T. rostrata encyst inside
D. reticulatum eggs [
5]. This may be an adaptation, with the more infective theronts being released when eggs are ruptured. This would be an ideal adaptation for this species because the infective theronts would have ready access to neonates in the egg cluster. However, in our experience, it is rare to find infected eggs, even in infected laboratory- reared
D. reticulatum. Neither trophonts nor theronts are able to penetrate eggs that have already been laid.
Some ciliates appeared to be trapped within groups in abnormally proliferating renal cells, possibly hypertrophic fibroblasts and amoebocytes. These granulomata formed around individual and groups of ciliates. Three of the slugs exposed to theronts and examined though histology, displayed abnormal changes in the heart. The pericardial cavities of these three slugs were filled with cells and hypertrophic amoebocytes. These cellular masses appeared identical to those seen by Brooks [
5], who described them as being composed of hypertrophic amoebocytes surrounded by a layer of epicardial cells.
No attempt was made to determine the route of entry of the theronts into the slugs. It was suggested that they enter via the integumental pouch under the mantle. However, we did not observe any trophonts in the mantle cavity. We did detect ciliates in the pneumostome passage passing under the pulmonary chamber. The tissue walls are very thin in this area and would seem to be vulnerable to mechanical damage from the ciliates, allowing access to the pulmonary chamber. We did not observe any direct evidence of trophonts squeezing between epithelial cells, as occurs with
I. multifiliis theronts [
10]. The ingestion of ciliates remains a possibility as the route of infection, as ciliates were also found in the hepatopancreas (also known as the digestive gland). However, this is less likely, as there were very few
T. rostrata ciliates seen in this gland, possibly due to size limitations for particles passing into the gland. Once at the hepatopancreas, the ciliates would need to cause mechanical damage to rupture the gland wall. Due to the small number of ciliates seen in this organ and the surrounding tissues and arteries, this remains unlikely.
The method for preparation of 100% theront cultures and the behavioral signs of infection are useful tools for further studies on the infection process.