Parasitic Intestinal Protists of Zoonotic Relevance Detected in Pigs by Metabarcoding and Real-Time PCR
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. DNA Extraction
2.3. Detection and Differentiation of Parasitic Genera by Metabarcoding
2.4. Real-Time PCR for Cryptosporidium, Dientamoeba, Enterocytozoon, and Giardia
| Parasite (Target Gene) | Oligonucleotides (Primer/Probe) | Primer and Probe Sequences | PCR Product Size (bp) | Reference |
|---|---|---|---|---|
| Cryptosporidium spp. | Cryptosporidium CRY F3 | 5′-CTA CAC TGA TGC ATC CAT CRA GT-3′ | 78 | Present study |
| (18S) | Cryptosporidium CRY R3 | 5′-CCC ATC ACG ATG CAT AYT CAA AA-3′ | ||
| Cryptosporidium CRY P | VIC-TCC TGT TTC GAA GGA AAT GGG TAA TC-MGB | |||
| Dientamoeba fragilis | DF-124f | 5′-CAA CGG ATG TCT TGG CTC TTT A-3′ | 97 | [24] |
| (ITS1-5.8S-ITS2) | Df-221r | 5′-TGC ATT CAA AGA TCG AAC TTA TCA C-3′ | ||
| Probe Df-172 | 6-Fam CAA TTC TAG CCG CTT AT-MGBNFQ | |||
| Enterocytozoon bieneusi | EblTS-89F | 5′-TGT GTA GGC GTG AGA GTG TAT CTG-3′ | 103 | [25] |
| (ITS) | EblTS-191r | 5′-CAT CCA ACC ATC ACG TAC CAA TC-3′ | ||
| Probe EblTS-114rev T | FAM-CAC TGC ACC CAC ATC CCT CAC CCT T-BHQ-1 | |||
| Giardia duodenalis | Giardia-80F | 5′-GAC GGC TCA GGA CAA CGG TT-3′ | 62 | [28] |
| (18S) | Giardia-127R | 5′-TTG CCA GCG GTG TCC G-3′ | ||
| Giardia-105T | FAM-CCC GCG GCG GTC CCT GCT AG-BHQ-1 |
2.5. Analysis of DNA Sequence Read Outputs
3. Results
3.1. DNA Sequence Read Yield in the Metabarcoding Assay
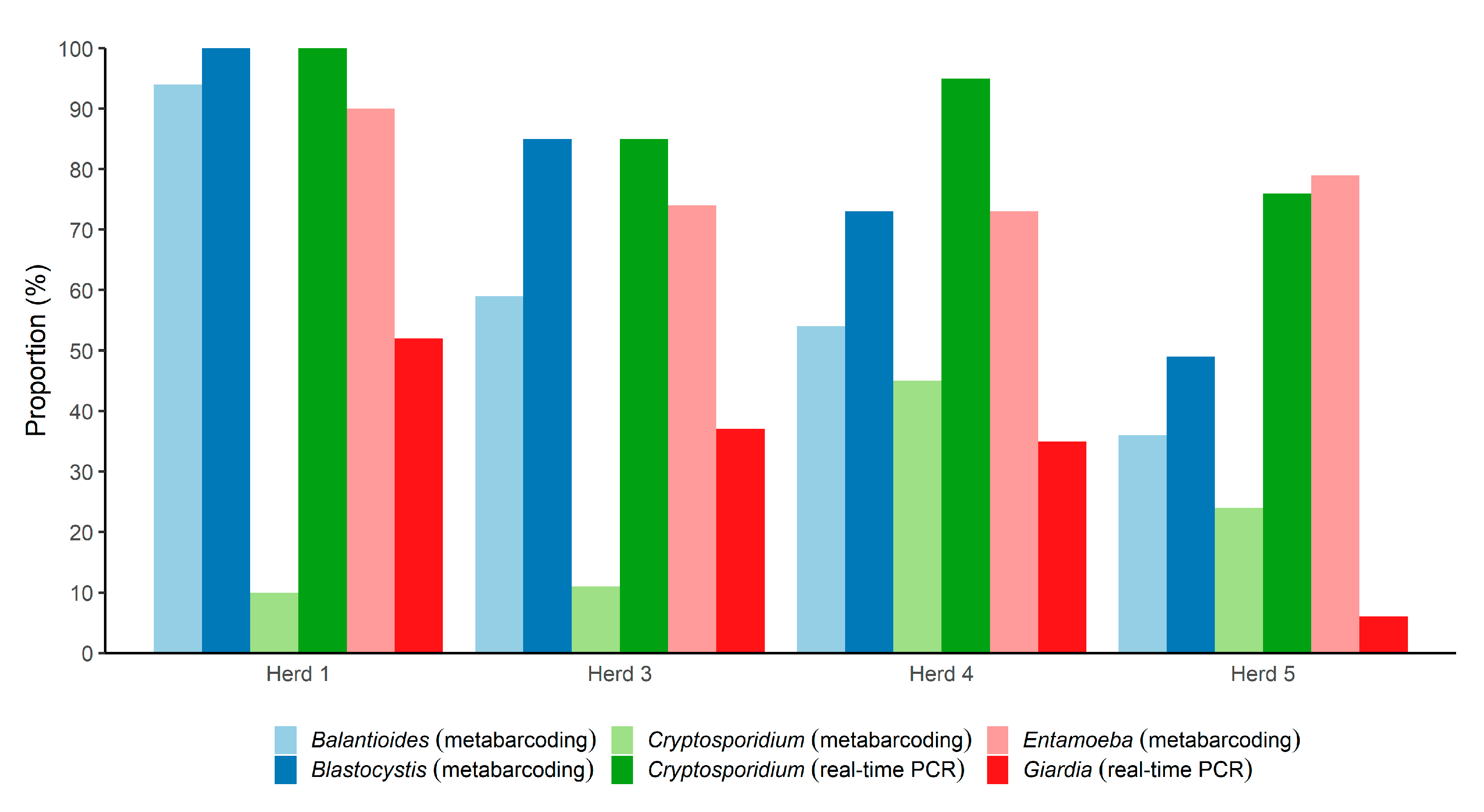
3.2. Parasites Detected by Metabarcoding and Real-Time PCR
3.3. Differentiation of Species and Subtypes Detected in the Samples by Metabarcoding
3.3.1. Cryptosporidium
3.3.2. Giardia
3.3.3. Entamoeba
3.3.4. Iodamoeba
3.3.5. Balantioides
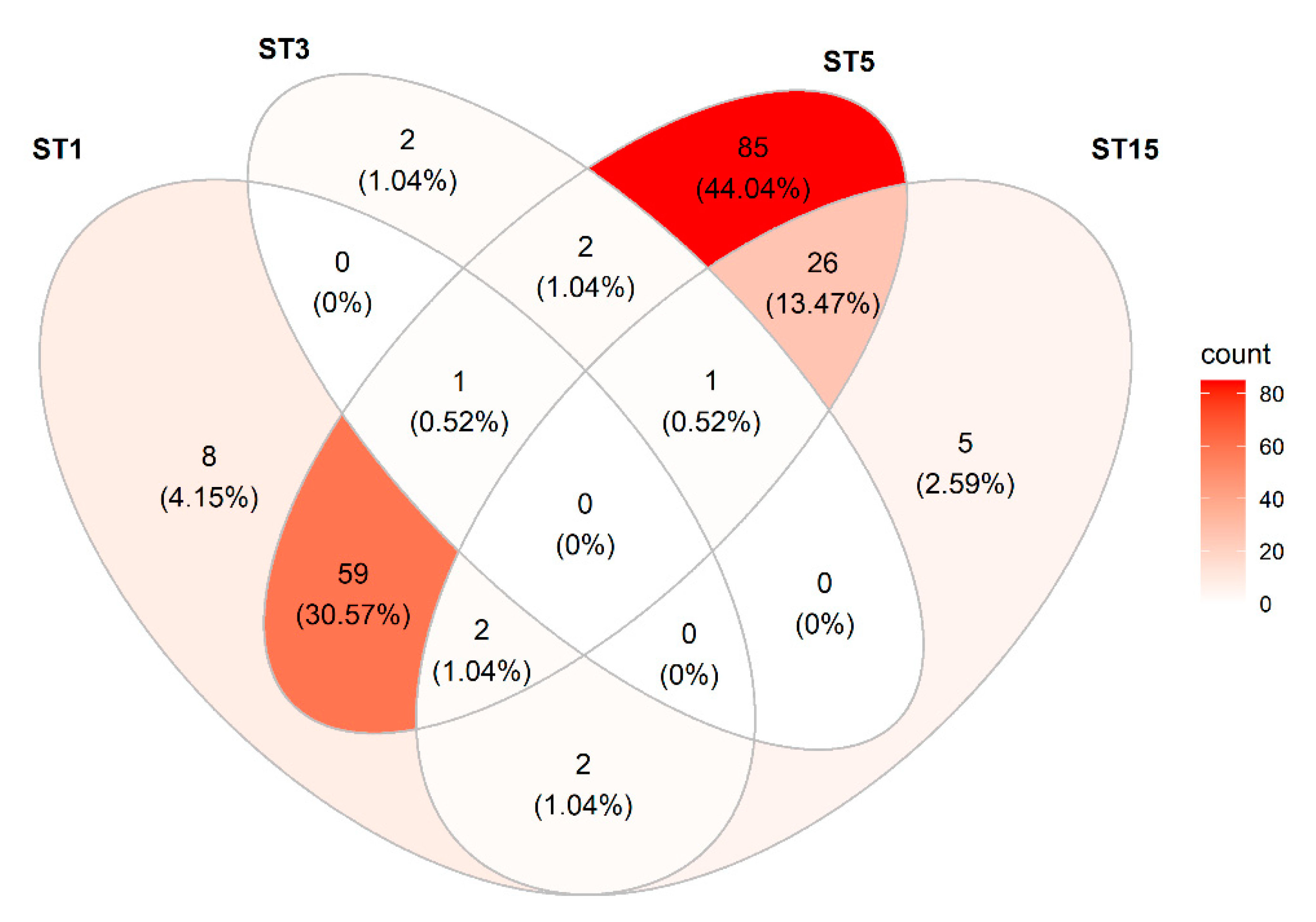
3.3.6. Blastocystis
3.4. Examples of Detected Polyparasitism
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robertson, L.J.; Lalle, M.; Paulsen, P. Why we need a European focus on foodborne parasites. Exp. Parasitol. 2020, 214, 107900. [Google Scholar] [CrossRef]
- Lhotská, Z.; Jirků, M.; Hložková, O.; Brožová, K.; Jirsová, D.; Stensvold, C.R.; Kolísko, M.; Jirků Pomajbíková, K. A Study on the Prevalence and Subtype Diversity of the Intestinal Protist Blastocystis sp. in a Gut-Healthy Human Population in the Czech Republic. Front. Cell Infect. Microbiol. 2020, 10, 544335. [Google Scholar] [CrossRef]
- Schuster, F.L.; Ramirez-Avila, L. Current world status of Balantidium coli. Clin. Microbiol. Rev. 2008, 21, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Pomajbikova, K.; Obornik, M.; Horak, A.; Petrzelkova, K.J.; Grim, J.N.; Levecke, B.; Todd, A.; Mulama, M.; Kiyang, J.; Modry, D. Novel insights into the genetic diversity of Balantidium and Balantidium-like cyst-forming ciliates. PLoS Negl. Trop. Dis. 2013, 7, e2140. [Google Scholar] [CrossRef] [PubMed]
- Cacciò, S.M.; Sannella, A.R.; Manuali, E.; Tosini, F.; Sensi, M.; Crotti, D.; Pozio, E. Pigs as natural hosts of Dientamoeba fragilis genotypes found in humans. Emerg. Infect. Dis. 2012, 18, 838–841. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Ijaz, M.; Ayyub, R.M.; Ghaffar, A.; Ghauri, H.N.; Aziz, M.U.; Ali, S.; Altaf, M.; Awais, M.; Naveed, M.; et al. Balantidium coli in domestic animals: An emerging protozoan pathogen of zoonotic significance. Acta Trop. 2020, 203, 105298. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.G.; Kaffashian, F.; Tawari, B.; Windsor, J.J.; Twigg-Flesner, A.; Davies-Morel, M.C.; Blessmann, J.; Ebert, F.; Peschel, B.; Le Van, A.; et al. New insights into the phylogeny of Entamoeba species provided by analysis of four new small-subunit rRNA genes. Int. J. Syst. Evol. Microbiol. 2006, 56, 2235–2239. [Google Scholar] [CrossRef]
- Dashti, A.; Rivero-Juarez, A.; Santin, M.; Lopez-Lopez, P.; Caballero-Gomez, J.; Frias-Casas, M.; Koster, P.C.; Bailo, B.; Calero-Bernal, R.; Briz, V.; et al. Enterocytozoon bieneusi (Microsporidia): Identification of novel genotypes and evidence of transmission between sympatric wild boars (Sus scrofa ferus) and Iberian pigs (Sus scrofa domesticus) in Southern Spain. Kransbound. Emerg. Dis. 2020, 67, 2869–2880. [Google Scholar] [CrossRef]
- Li, W.; Feng, Y.; Xiao, L. Diagnosis and molecular typing of Enterocytozoon bieneusi: The significant role of domestic animals in transmission of human microsporidiosis. Res. Vet. Sci. 2020, 133, 251–261. [Google Scholar] [CrossRef]
- Verweij, J.J.; Stensvold, C.R. Molecular Testing for Clinical Diagnosis and Epidemiological Investigations of Intestinal Parasitic Infections. Clin. Microbiol. Rev. 2014, 27, 371–418. [Google Scholar] [CrossRef]
- FAO. Epidemiology, diagnosis and control of helminth parasites of swine. In FAO Animal Health Manual; FAO: Rome, Italy, 1998. [Google Scholar]
- Krogsgaard, L.R.; Andersen, L.O.; Johannesen, T.B.; Engsbro, A.L.; Stensvold, C.R.; Nielsen, H.V.; Bytzer, P. Characteristics of the bacterial microbiome in association with common intestinal parasites in irritable bowel syndrome. Clin. Transl. Gastroenterol. 2018, 9, 161. [Google Scholar] [CrossRef]
- Ring, H.C.; Thorsen, J.; Saunte, D.M.; Lilje, B.; Bay, L.; Riis, P.T.; Larsen, N.; Andersen, L.O.; Nielsen, H.V.; Miller, I.M.; et al. The Follicular Skin Microbiome in Patients With Hidradenitis Suppurativa and Healthy Controls. JAMA Dermatol. 2017, 153, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Holmgaard, D.B.; Barnadas, C.; Mirbarati, S.H.; Andersen, L.O.; Nielsen, H.V.; Stensvold, C.R. Detection and identification of Acanthamoeba and other non-viral causes of infectious keratitis in corneal scrapings by real-time PCR and next-generation sequencing-based 16S-18S gene analysis. J. Clin. Microbiol. 2020, 59. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Lebbad, M.; Hansen, A.; Beser, J.; Belkessa, S.; O’Brien Andersen, L.; Clark, C.G. Differentiation of Blastocystis and parasitic archamoebids encountered in untreated wastewater samples by amplicon-based next-generation sequencing. Parasite Epidemiol. Control 2020, 9, e00131. [Google Scholar] [CrossRef] [PubMed]
- Wylezich, C.; Belka, A.; Hanke, D.; Beer, M.; Blome, S.; Höper, D. Metagenomics for broad and improved parasite detection: A proof-of-concept study using swine faecal samples. Int. J. Parasitol. 2019, 49, 769–777. [Google Scholar] [CrossRef]
- Ramayo-Caldas, Y.; Prenafeta-Boldu, F.; Zingaretti, L.M.; Gonzalez-Rodriguez, O.; Dalmau, A.; Quintanilla, R.; Ballester, M. Gut eukaryotic communities in pigs: Diversity, composition and host genetics contribution. Anim. Microbiome 2020, 2, 18. [Google Scholar] [CrossRef]
- Græsbøll, K.; Damborg, P.; Mellerup, A.; Herrero-Fresno, A.; Larsen, I.; Holm, A.; Nielsen, J.P.; Christiansen, L.E.; Angen, Ø.; Ahmed, S.; et al. Effect of Tetracycline Dose and Treatment Mode on Selection of Resistant Coliform Bacteria in Nursery Pigs. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef]
- Græsbøll, K.; Larsen, I.; Clasen, J.; Birkegård, A.C.; Nielsen, J.P.; Christiansen, L.E.; Olsen, J.E.; Angen, Ø.; Folkesson, A. Effect of tetracycline treatment regimens on antibiotic resistance gene selection over time in nursery pigs. BMC Microbiol. 2019, 19, 269. [Google Scholar] [CrossRef]
- Larsen, I.; Hjulsager, C.K.; Holm, A.; Olsen, J.E.; Nielsen, S.S.; Nielsen, J.P. A randomised clinical trial on the efficacy of oxytetracycline dose through water medication of nursery pigs on diarrhoea, faecal shedding of Lawsonia intracellularis and average daily weight gain. Prev. Vet. Med. 2016, 123, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Larsen, I.; Nielsen, S.S.; Olsen, J.E.; Nielsen, J.P. The efficacy of oxytetracycline treatment at batch, pen and individual level on Lawsonia intracellularis infection in nursery pigs in a randomised clinical trial. Prev. Vet. Med. 2016, 124, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Fredensborg, B.L.; Fossdal í Kálvalíð, I.; Johannesen, T.B.; Stensvold, C.R.; Nielsen, H.V.; Kapel, C.M.O. Parasites modulate the gut-microbiome in insects: A proof-of-concept study. PLoS ONE 2020, 15, e0227561. [Google Scholar] [CrossRef]
- Thomas-Lopez, D.; Muller, L.; Vestergaard, L.S.; Christoffersen, M.; Andersen, A.M.; Jokelainen, P.; Agerholm, J.S.; Stensvold, C.R. Veterinary Students Have a Higher Risk of Contracting Cryptosporidiosis when Calves with High Fecal Cryptosporidium Loads Are Used for Fetotomy Exercises. Appl. Environ. Microbiol. 2020, 86. [Google Scholar] [CrossRef] [PubMed]
- Verweij, J.J.; Mulder, B.; Poell, B.; van Middelkoop, D.; Brienen, E.A.; van Lieshout, L. Real-time PCR for the detection of Dientamoeba fragilis in fecal samples. Mol. Cell. Probes 2007, 21, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Verweij, J.J.; Ten Hove, R.; Brienen, E.A.; van Lieshout, L. Multiplex detection of Enterocytozoon bieneusi and Encephalitozoon spp. in fecal samples using real-time PCR. Diagn. Microbiol. Infect. Dis. 2007, 57, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Escalante, L.; Yang, C.; Sulaiman, I.; Escalante, A.A.; Montali, R.J.; Fayer, R.; Lal, A.A. Phylogenetic analysis of Cryptosporidium parasites based on the small-subunit rRNA gene locus. Appl. Environ. Microbiol. 1999, 65, 1578–1583. [Google Scholar] [CrossRef] [PubMed]
- Buckholt, M.A.; Lee, J.H.; Tzipori, S. Prevalence of Enterocytozoon bieneusi in swine: An 18-month survey at a slaughterhouse in Massachusetts. Appl. Environ. Microbiol. 2002, 68, 2595–2599. [Google Scholar] [CrossRef]
- Verweij, J.J.; Schinkel, J.; Laeijendecker, D.; van Rooyen, M.A.; van Lieshout, L.; Polderman, A.M. Real-time PCR for the detection of Giardia lamblia. Mol. Cell Probes 2003, 17, 223–225. [Google Scholar] [CrossRef]
- Morgan, U.M.; Monis, P.T.; Fayer, R.; Deplazes, P.; Thompson, R.C. Phylogenetic relationships among isolates of Cryptosporidium: Evidence for several new species. J. Parasitol. 1999, 85, 1126–1133. [Google Scholar] [CrossRef] [PubMed]
- Kváč, M.; Kestřánová, M.; Pinková, M.; Květoňová, D.; Kalinová, J.; Wagnerová, P.; Kotková, M.; Vítovec, J.; Ditrich, O.; McEvoy, J.; et al. Cryptosporidium scrofarum n. sp. (Apicomplexa: Cryptosporidiidae) in domestic pigs (Sus scrofa). Vet. Parasitol. 2013, 191, 218–227. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Lebbad, M.; Victory, E.L.; Verweij, J.J.; Tannich, E.; Alfellani, M.; Legarraga, P.; Clark, C.G. Increased sampling reveals novel lineages of Entamoeba: Consequences of genetic diversity and host specificity for taxonomy and molecular detection. Protist 2011, 162, 525–541. [Google Scholar] [CrossRef]
- Nilles-Bije, M.L.; Rivera, W.L. Ultrastructural and molecular characterization of Balantidium coli isolated in the Philippines. Parasitol. Res. 2010, 106, 387–394. [Google Scholar] [CrossRef]
- Stolzenbach, S.; Myhill, L.J.; Andersen, L.O.; Krych, L.; Mejer, H.; Williams, A.R.; Nejsum, P.; Stensvold, C.R.; Nielsen, D.S.; Thamsborg, S.M. Dietary Inulin and Trichuris suis Infection Promote Beneficial Bacteria Throughout the Porcine Gut. Front. Microbiol. 2020, 11, 312. [Google Scholar] [CrossRef]
- Stensvold, C.R.; O’Brien Andersen, L.; Jokelainen, P. Protocol for 18S rDNA-based amplicon sequencing for detection of relevant Foodborne Parasites. Zenodo 2021. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Winiecka-Krusnell, J.; Lier, T.; Lebbad, M. Evaluation of a PCR Method for Detection of Entamoeba polecki, with an Overview of Its Molecular Epidemiology. J. Clin. Microbiol. 2018, 56. [Google Scholar] [CrossRef]
- Xiao, L.; Feng, Y. Molecular epidemiologic tools for waterborne pathogens Cryptosporidium spp. and Giardia duodenalis. Food Waterborne Parasitol. 2017, 8–9, 14–32. [Google Scholar] [CrossRef]
- Kvac, M.; Kvetonova, D.; Sak, B.; Ditrich, O. Cryptosporidium pig genotype II in immunocompetent man. Emerg. Infect. Dis. 2009, 15, 982–983. [Google Scholar] [CrossRef]
- Wylezich, C.; Caccio, S.M.; Walochnik, J.; Beer, M.; Höper, D. Untargeted metagenomics shows a reliable performance for synchronous detection of parasites. Parasitol. Res. 2020, 119, 2623–2629. [Google Scholar] [CrossRef] [PubMed]
- Parfrey, L.W.; Walters, W.A.; Lauber, C.L.; Clemente, J.C.; Berg-Lyons, D.; Teiling, C.; Kodira, C.; Mohiuddin, M.; Brunelle, J.; Driscoll, M.; et al. Communities of microbial eukaryotes in the mammalian gut within the context of environmental eukaryotic diversity. Front. Microbiol. 2014, 5, 298. [Google Scholar] [CrossRef] [PubMed]
- Pettersson, E.; Ahola, H.; Frossling, J.; Wallgren, P.; Troell, K. Detection and molecular characterisation of Cryptosporidium spp. in Swedish pigs. Acta Vet. Scand. 2020, 62, 40. [Google Scholar] [CrossRef]
- Langkjaer, R.B.; Vigre, H.; Enemark, H.L.; Maddox-Hyttel, C. Molecular and phylogenetic characterization of Cryptosporidium and Giardia from pigs and cattle in Denmark. Parasitology 2007, 134, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Petersen, H.H.; Jianmin, W.; Katakam, K.K.; Mejer, H.; Thamsborg, S.M.; Dalsgaard, A.; Olsen, A.; Enemark, H.L. Cryptosporidium and Giardia in Danish organic pig farms: Seasonal and age-related variation in prevalence, infection intensity and species/genotypes. Vet. Parasitol. 2015, 214, 29–39. [Google Scholar] [CrossRef]
- Sak, B.; Kvác, M.; Hanzlíková, D.; Cama, V. First report of Enterocytozoon bieneusi infection on a pig farm in the Czech Republic. Vet. Parasitol. 2008, 153, 220–224. [Google Scholar] [CrossRef]
- Decraene, V.; Lebbad, M.; Botero-Kleiven, S.; Gustavsson, A.M.; Löfdahl, M. First reported foodborne outbreak associated with microsporidia, Sweden, October 2009. Epidemiol. Infect. 2012, 140, 519–527. [Google Scholar] [CrossRef]
- Li, W.; Feng, Y.; Santin, M. Host Specificity of Enterocytozoon bieneusi and Public Health Implications. Trends Parasitol. 2019, 35, 436–451. [Google Scholar] [CrossRef]
- Jokelainen, P.; Hebbelstrup Jensen, B.; Andreassen, B.U.; Petersen, A.M.; Röser, D.; Krogfelt, K.A.; Nielsen, H.V.; Stensvold, C.R. Dientamoeba fragilis—A Commensal in Children in Danish Day Care Centers. J. Clin. Microbiol. 2017, 55, 1707–1713. [Google Scholar] [CrossRef]
- Roser, D.; Simonsen, J.; Nielsen, H.V.; Stensvold, C.R.; Molbak, K. Dientamoeba fragilis in Denmark: Epidemiological experience derived from four years of routine real-time PCR. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Crotti, D.; Sensi, M.; Crotti, S.; Grelloni, V.; Manuali, E. Dientamoeba fragilis in swine population: A preliminary investigation. Vet. Parasitol. 2007, 145, 349–351. [Google Scholar] [CrossRef] [PubMed]
- Krogsgaard, L.R.; Engsbro, A.L.; Stensvold, C.R.; Nielsen, H.V.; Bytzer, P. The prevalence of intestinal parasites is not greater among individuals with irritable bowel syndrome: A population-based case-control study. Clin. Gastroenterol. Hepatol. 2015, 13, 507–513.e502. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.O.; Bonde, I.; Nielsen, H.B.; Stensvold, C.R. A retrospective metagenomics approach to studying Blastocystis. FEMS Microbiol. Ecol. 2015, 91. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Alfellani, M.A.; Nørskov-Lauritsen, S.; Prip, K.; Victory, E.L.; Maddox, C.; Nielsen, H.V.; Clark, C.G. Subtype distribution of Blastocystis isolates from synanthropic and zoo animals and identification of a new subtype. Int. J. Parasitol. 2009, 39, 473–479. [Google Scholar] [CrossRef]
- Navarro, C.; Domínguez-Márquez, M.V.; Garijo-Toledo, M.M.; Vega-García, S.; Fernández-Barredo, S.; Pérez-Gracia, M.T.; García, A.; Borrás, R.; Gómez-Muñoz, M.T. High prevalence of Blastocystis sp. in pigs reared under intensive growing systems: Frequency of ribotypes and associated risk factors. Vet. Parasitol. 2008, 153, 347–358. [Google Scholar] [CrossRef]
- Thathaisong, U.; Worapong, J.; Mungthin, M.; Tan-Ariya, P.; Viputtigul, K.; Sudatis, A.; Noonai, A.; Leelayoova, S. Blastocystis isolates from a pig and a horse are closely related to Blastocystis hominis. J. Clin. Microbiol. 2003, 41, 967–975. [Google Scholar] [CrossRef]
- Tan, T.C.; Tan, P.C.; Sharma, R.; Sugnaseelan, S.; Suresh, K.G. Genetic diversity of caprine Blastocystis from Peninsular Malaysia. Parasitol. Res. 2013, 112, 85–89. [Google Scholar] [CrossRef]
- Rauff-Adedotun, A.A.; Mohd Zain, S.N.; Farah Haziqah, M.T. Current status of Blastocystis sp. in animals from Southeast Asia: A review. Parasitol. Res. 2020, 119, 3559–3570. [Google Scholar] [CrossRef]
- Danisova, O.; Valencakova, A. First detection of Blastocystis sp. in pigs in Slovakia and in Europe. Parasitol. Int. 2020, 81, 102235. [Google Scholar] [CrossRef]
- Jimenez, P.A.; Jaimes, J.E.; Ramirez, J.D. A summary of Blastocystis subtypes in North and South America. Parasit. Vectors 2019, 12, 376. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Alfellani, M.; Clark, C.G. Levels of genetic diversity vary dramatically between Blastocystis subtypes. Infect. Genet. Evol. 2012, 12, 263–273. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Taner-Mulla, D.; Jacob, A.S.; Imeede, C.A.; Yoshikawa, H.; Stensvold, C.R.; Clark, C.G. Genetic diversity of Blastocystis in livestock and zoo animals. Protist 2013, 164, 497–509. [Google Scholar] [CrossRef]
- Russini, V.; Di Filippo, M.M.; Fanelli, R.; Polidori, M.; Berrilli, F.; Di Cave, D.; Novelletto, A.; Calderini, P. Characterization of prevalence and genetic subtypes of Blastocystis sp. in wild and domestic Suidae of central Italy aided by amplicon NGS. Vet. Parasitol. Reg. Stud. Rep. 2020, 22, 100472. [Google Scholar] [CrossRef] [PubMed]
- Burrows, R.B. Morphological differentiation of Entamoeba hartmanni and E. polecki from E. histolytica. Am. J. Trop. Med. Hyg. 1959, 8, 583–589. [Google Scholar] [CrossRef]
- Burrows, R.B. Microscopic Diagnosis of the Parasites of Man; Yale University Press: New Haven, CT, USA, 1965; pp. 118–119. [Google Scholar]
- Li, W.C.; Geng, J.Z.; Chen, C.; Qian, L.; Zhang, T.; Liu, J.L.; Luo, J.X.; Gu, Y.F. First report on the occurance of intestinal Entamoeba spp. in pigs in China. Acta Trop. 2018, 185, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Verweij, J.J.; Polderman, A.M.; Clark, C.G. Genetic Variation among Human Isolates of Uninucleated Cyst-Producing Entamoeba Species. J. Clin. Microbiol. 2001, 39, 1644–1646. [Google Scholar] [CrossRef]
- Ji, T.; Cao, H.X.; Wu, R.; Cui, L.L.; Su, G.M.; Niu, C.; Zhang, N.; Wang, S.K.; Zhou, D.H. Prevalence and Genetic Identification of Three Entamoeba Species in Pigs in Southeastern China. Biomed Res. Int. 2019, 2824017. [Google Scholar] [CrossRef] [PubMed]
- Wardhana, A.H.; Sawitri, D.H.; Ekawasti, F.; Martindah, E.; Apritadewi, D.; Shibahara, T.; Kusumoto, M.; Tokoro, M.; Sasai, K.; Matsubayashi, M. Occurrence and genetic identifications of porcine Entamoeba, E. suis and E. polecki, at Tangerang in West Java, Indonesia. Parasitol. Res. 2020, 119, 2983–2990. [Google Scholar] [CrossRef]
- Schubnell, F.; von Ah, S.; Graage, R.; Sydler, T.; Sidler, X.; Hadorn, D.; Basso, W. Occurrence, clinical involvement and zoonotic potential of endoparasites infecting Swiss pigs. Parasitol. Int. 2016, 65, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Roesel, K.; Dohoo, I.; Baumann, M.; Dione, M.; Grace, D.; Clausen, P.H. Prevalence and risk factors for gastrointestinal parasites in small-scale pig enterprises in Central and Eastern Uganda. Parasitol. Res. 2017, 116, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Barbosa, A.; Ponce-Gordo, F.; Dib, L.V.; Antunes Uchoa, C.M.; Bastos, O.M.P.; Pissinatti, A.; Amendoeira, M.R.R. First molecular characterization of Balantioides coli (Malmsten, 1857) isolates maintained in vitro culture and from feces of captive animals, Rio de Janeiro, Brazil. Vet. Parasitol. Reg. Stud. Rep. 2017, 10, 102–113. [Google Scholar] [CrossRef]
- Li, Y.H.; Yao, Q.; Dong, H.P.; Wang, S.S.; Chen, R.R.; Song, J.K.; Yan, W.C.; Zhao, G.H. Molecular characterization of Balantioides coli in pigs from Shaanxi province, northwestern China. Parasitol. Res. 2020, 119, 3075–3081. [Google Scholar] [CrossRef]
- Ponce-Gordo, F.; Fonseca-Salamanca, F.; Martinez-Diaz, R.A. Genetic heterogeneity in internal transcribed spacer genes of Balantidium coli (Litostomatea, Ciliophora). Protist 2011, 162, 774–794. [Google Scholar] [CrossRef]
- Lopez Arias, L.; Guillemi, E.; Bordoni, N.; Farber, M.; Garbossa, G. Development of a PCR assay for identification of Neobalantidium coli () in Argentina. Vet. Parasitol. Reg. Stud. Rep. 2017, 10, 114–118. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Lebbad, M.; Clark, C.G. Last of the human protists: The phylogeny and genetic diversity of Iodamoeba. Mol. Biol Evol. 2012, 29, 39–42. [Google Scholar] [CrossRef][Green Version]
| Primer Pair | Small Subunit rDNA Target | Primer Sequences |
|---|---|---|
| 341F3/806R5 | 16S | 5′-ACTCCTAYGGGRBGCASCAG-3′ 5′-AGCGTGGACTACNNGGGTATCTAAT-3′ |
| G3F1/G3R1 | 18S | 5′-GCCAGCAGCCGCGGTAATTC-3′ 5′-ACATTCTTGGCAAATGCTTTCGCAG-3′ |
| G4F3/G4R3 | 18S | 5′-CAGCCGCGGTAATTCCAGCTC-3′ 5′-GGTGGTGCCCTTCCGTCAAT-3′ |
| G6F1/G6R1 | 18S | 5′-TGGAGGGCAAGTCTGGTGCC-3′ 5′-ACGGTATCTGATCGTCTTCGATCCC-3′ |
| Genus | Primer Set * | Sequence Reads per Sample, Range | Sequence Reads per Positive Sample, Median (IQR) | No. of Samples Positive/Tested (%) | No. of Samples Positive/Tested by Real-Time PCR |
|---|---|---|---|---|---|
| Balantioides | G4 | 0–41,925 | 508 (215.5–1404.5) | 148/259 (57%) | NA |
| Blastocystis | G6 | 0–19,318 | 681 (212.5–2439.5) | 193/259 (75%) | NA |
| Cryptosporidium | G3 | 0–4678 | 560 (169–1096) | 90/259 (35%) | 239/259 (92%) |
| Dientamoeba | NA | NA | NA | NA | 0/37 (0%) |
| Entamoeba | G6 | 0–37,557 | 602 (225–2361) | 195/259 (75%) | NA |
| Enterocytozoon | NA | NA | NA | NA | 34/37 (92%) |
| Giardia | NA | NA | NA | NA | 86/259 (33%) |
| Iodamoeba | G6 | NA | NA | 1/259 (<1%) | NA |
| Number of Reads | |
|---|---|
| TOTAL | 19,777,284 |
| Archaea | 262,016 |
| Prokaryotes | 11,406,189 |
| Eukaryotes | 8,109,079 |
| Blastocystis | 1,568,032 |
| Entamoeba | 396,129 |
| Balantioides | 265,409 |
| Cryptosporidium | 80,159 |
| Iodamoeba | 1273 |
| Enterocytozoon | 0 |
| Giardia | 0 |
| Dientamoeba | 0 |
| Fungal DNA | 2,239,641 |
| Host DNA | 1,357,652 |
| Plant DNA | 1,236,282 |
| Other (e.g., nematodes) | 964,502 |
| Genus | Species | Subtype | No. of Samples Positive (%) |
|---|---|---|---|
| Blastocystis | sp. | ST1 | 72 (28%) |
| sp. | ST3 | 6 (2%) | |
| sp. | ST5 | 176 (68%) | |
| sp. | ST15 * | 36 (14%) | |
| Entamoeba | hartmanni | NA | 8 (3%) |
| polecki | ST1 | 104 (40%) | |
| polecki | ST3 | 161 (62%) | |
| Iodamoeba | bütschlii | RL2 | 1 (0.4%) |
| Cryptosporidium | suis | NA | 53 (20%) |
| scrofarum | NA | 45 (17%) | |
| Balantioides | coli | NA | 148 (57%) |
| Subtype | Sequence Type | Number of Samples Positive | Examples of Closest Matches to Reference DNA Sequence Entry (Accession no.) in NCBI Database |
|---|---|---|---|
| ST1 | ST1a | 49 | AB107961 (pig, Japan) (99.76%) |
| ST1b | 55 | AB107962 (human, Japan) (100%) KU719525 (human, Iran) (100%) MK801400 (pig, Germany) (100%) | |
| ST3 | ST3a | 3 | AB070986 (human, Japan) (100%) MT330276 (human, Thailand) (100%) AM779042 (human, Turkey) (100%) MK801389 (pig, Germany) (100%) |
| ST3b | 5 | KM216257 (human, Thailand) (100%) LC414152 (human, Iran) (100%) MK418914 (macaque, China) (100%) | |
| ST5 | ST5a | 2 | AB107966 (cattle, Japan) (99.76%) AB070998 (pig, Japan) (99.76%) MK801415 (pig, Austria) (99.76%) |
| ST5b | 60 | AB107964 (pig, Japan) (100%) MH104976 (pig, Thailand) (100%) MK801372 (pig, Germany) (100%) | |
| ST5c | 8 | AB107966 (cattle, Japan) (100%) AB070998 (pig, Japan) (100%) MK801415 (pig, Austria) (100%) | |
| ST5d | 8 | MK375237 (human, Thailand) (99.76%) MK801375 (pig, Germany) (99.76%) MK375227 (pig, China) (99.76%) | |
| ST5e | 27 | KT819615 (pig, Thailand) (100%) MK375229 (pig, China) (110%) | |
| ST5f | 129 | MK375237 (pig, China) (100%) MK801375 (pig, Germany) (100%) | |
| ST15 | ST15* | NA * | MK801393 (pig, Germany) (99.34%) KC148211 (gibbon, zoo) (99.12%) KC148210 (camel, Egypt) (97.81%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stensvold, C.R.; Jirků-Pomajbíková, K.; Tams, K.W.; Jokelainen, P.; Berg, R.P.K.D.; Marving, E.; Petersen, R.F.; Andersen, L.O.; Angen, Ø.; Nielsen, H.V. Parasitic Intestinal Protists of Zoonotic Relevance Detected in Pigs by Metabarcoding and Real-Time PCR. Microorganisms 2021, 9, 1189. https://doi.org/10.3390/microorganisms9061189
Stensvold CR, Jirků-Pomajbíková K, Tams KW, Jokelainen P, Berg RPKD, Marving E, Petersen RF, Andersen LO, Angen Ø, Nielsen HV. Parasitic Intestinal Protists of Zoonotic Relevance Detected in Pigs by Metabarcoding and Real-Time PCR. Microorganisms. 2021; 9(6):1189. https://doi.org/10.3390/microorganisms9061189
Chicago/Turabian StyleStensvold, Christen Rune, Kateřina Jirků-Pomajbíková, Katrine Wegener Tams, Pikka Jokelainen, Rebecca P. K. D. Berg, Ellinor Marving, Randi Føns Petersen, Lee O’Brien Andersen, Øystein Angen, and Henrik Vedel Nielsen. 2021. "Parasitic Intestinal Protists of Zoonotic Relevance Detected in Pigs by Metabarcoding and Real-Time PCR" Microorganisms 9, no. 6: 1189. https://doi.org/10.3390/microorganisms9061189
APA StyleStensvold, C. R., Jirků-Pomajbíková, K., Tams, K. W., Jokelainen, P., Berg, R. P. K. D., Marving, E., Petersen, R. F., Andersen, L. O., Angen, Ø., & Nielsen, H. V. (2021). Parasitic Intestinal Protists of Zoonotic Relevance Detected in Pigs by Metabarcoding and Real-Time PCR. Microorganisms, 9(6), 1189. https://doi.org/10.3390/microorganisms9061189






