Oral Microbiota and Salivary Levels of Oral Pathogens in Gastro-Intestinal Diseases: Current Knowledge and Exploratory Study
Abstract
1. Introduction
1.1. Oral Microbiota: An Overview
1.2. Oral Dysbiosis and Gastrointestinal Diseases
2. Materials and Methods
2.1. Patients
2.2. Saliva Analysis for Microbiota Evaluation
2.3. Statistical Analysis
3. Results
4. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- De Benedittis, M.; Petruzzi, M.; Favia, G.; Serpico, R. Oro-dental manifestations in Hallopeau-Siemens-type recessive dystrophic epidermolysis bullosa. Clin. Exp. Dermatol. 2004, 29, 128–132. [Google Scholar] [CrossRef]
- Della Vella, F.; Lauritano, D.; Lajolo, C.; Lucchese, A.; Di Stasio, D.; Contaldo, M.; Serpico, R.; Petruzzi, M. The Pseudolesions of the Oral Mucosa: Differential Diagnosis and Related Systemic Conditions. Appl. Sci. 2019, 9, 2412. [Google Scholar] [CrossRef]
- Della Vella, F.; Contaldo, M.; Fucile, R.; Panza, F.; Dibello, V.; Kalemaj, Z.; Ninivaggi, R.; Petruzzi, M.; Serpico, R. ORO-Dental Manifestations in West Syndrome. Curr. Top. Med. Chem. 2019, 19, 2824–2828. [Google Scholar] [CrossRef]
- Contaldo, M.; Luzzi, V.; Ierardo, G.; Raimondo, E.; Boccellino, M.; Ferati, K.; Bexheti-Ferati, A.; Inchingolo, F.; Di Domenico, M.; Serpico, R. Bisphosphonate-related osteonecrosis of the jaws and dental surgery procedures in children and young people with osteogenesis imperfecta: A systematic review. J. Stomatol. Oral Maxillofac. Surg. 2020, 121, 556–562. [Google Scholar] [CrossRef]
- Contaldo, M.; Itro, A.; Lajolo, C.; Gioco, G.; Inchingolo, F.; Serpico, R. Overview on Osteoporosis, Periodontitis and Oral Dysbiosis: The Emerging Role of Oral Microbiota. Appl. Sci. 2020, 10, 6000. [Google Scholar] [CrossRef]
- Adeyemo, T.A.; Adeyemo, W.L.; Adediran, A.; Akinbami, A.J.; Akanmu, A.S. Orofacial manifestations of hematological disorders: Anemia and hemostatic disorders. Indian J. Dent. Res. 2011, 22, 454–461. [Google Scholar] [CrossRef]
- Chiang, C.P.; Chang, J.Y.; Wang, Y.P.; Wu, Y.H.; Wu, Y.C.; Sun, A. Atrophic glossitis: Etiology, serum autoantibodies, anemia, hematinic deficiencies, hyperhomocysteinemia, and management. J. Formos. Med. Assoc. 2020, 119, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Pastore, L.; Lo Muzio, L.; Serpico, R. Atrophic glossitis leading to the diagnosis of celiac disease. N. Engl. J. Med. 2007, 356, 2547. [Google Scholar] [CrossRef] [PubMed]
- Pastore, L.; Carroccio, A.; Compilato, D.; Panzarella, V.; Serpico, R.; Lo Muzio, L. Oral manifestations of celiac disease. J. Clin. Gastroenterol. 2008, 42, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Onitake, T.; Ueno, Y.; Tanaka, S.; Hanaoka, R.; Yoshioka, K.; Hatakeyama, T.; Oka, S.; Yoshida, S.; Hiyama, T.; Ito, M.; et al. Cheilitis granulomatosa as an early manifestation of Crohn’s disease. Clin. J. Gastroenterol. 2009, 2, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Umar, S. Intestinal Stem Cells. Curr. Gastroenterol. Rep. 2010, 12, 340–348. [Google Scholar] [CrossRef]
- Behrens, M. Chapter: Extraoral Taste Receptors. In Reference Module in Neuroscience and Biobehavioral Psychology; Elsevier Inc.: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Troll, J.V.; Hamilton, M.K.; Abel, M.L.; Ganz, J.; Bates, J.M.; Stephens, W.Z.; Melancon, E.; Van Der Vaart, M.; Meijer, A.H.; Distel, M.; et al. Microbiota promote secretory cell determination in the intestinal epithelium by modulating host Notch signaling. Development 2018, 145, dev155317. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; McKinley, E.T.; von Moltke, J.; Coey, R.J.; Lau, K.S. Interpreting heterogeneity in intestinal tuft cell structure and function. J. Clin. Investig. 2018, 128, 1711–1719. [Google Scholar] [CrossRef] [PubMed]
- Peck, B.C.E.; Shanahan, M.T.; Singh, A.P.; Sethupathy, P. Gut Microbial Influences on the Mammalian Intestinal Stem Cell Niche. Stem Cells Int. 2017, 2017, 1–17. [Google Scholar] [CrossRef]
- Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M.C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Corral, G.H.; et al. Microbiome definition re-visited: Old concepts and new challenges. Microbiome 2020, 8, 103. [Google Scholar] [CrossRef] [PubMed]
- Baquero, F.; Nombela, C. The microbiome as a human organ. Clin. Microbiol. Infect. 2012, 18, 2–4. [Google Scholar] [CrossRef]
- Dewhirst, F.E.; Chen, T.; Izard, J.; Paster, B.J.; Tanner, A.C.; Yu, W.H.; Lakshmanan, A.; Wade, W.G. The human oral microbiome. J. Bacteriol. 2010, 192, 5002–5017. [Google Scholar] [CrossRef] [PubMed]
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Pannone, G.; Sanguedolce, F.; De Maria, S.; Farina, E.; Lo Muzio, L.; Serpico, R.; Emanuelli, M.; Rubini, C.; De Rosa, G.; Staibano, S.; et al. Cyclooxygenase isozymes in oral squamous cell carcinoma: A real-time RT-PCR study with clinic pathological correlations. Int. J. Immunopathol. Pharmacol. 2007, 20, 317–324. [Google Scholar] [CrossRef]
- Sala-Comorera, L.; Caudet-Segarra, L.; Galofré, B.; Lucena, F.; Blanch, A.R.; García-Aljaro, C. Unravelling the composition of tap and mineral water microbiota: Divergences between next-generation sequencing techniques and culture-based methods. Int. J. Food Microbiol. 2020, 334, 108850. [Google Scholar] [CrossRef]
- Appanna, V.D. The Human Microbiome: The Origin. In Human Microbes—The Power Within; Springer: Singapore, 2018; pp. 1–36. [Google Scholar]
- Perera, M.; Al-Hebshi, N.N.; Speicher, D.J.; Perera, I.; Johnson, N.W. Emerging role of bacteria in oral carcinogenesis: A review with special reference to perio-pathogenic bacteria. J. Oral Microbiol. 2016, 8, 32762. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Jurevic, R.J.; Mukherjee, P.K.; Cui, F.; Sikaroodi, M.; Naqvi, A.; Gillevet, P.M. Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathog. 2010, 6, e1000713. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Bhatia, S.; Sodhi, A.S.; Batra, N. Oral microbiome and health. AIMS Microbiol. 2018, 4, 42–66. [Google Scholar] [CrossRef] [PubMed]
- Petruzzi, M.; Della Vella, F.; Cassandro, A.; Mosca, A.; Di Comite, M.; Contaldo, M.; Grassi, F.R.; Lauritano, D. Dorsal tongue porphyrin autofluorescence and Candida saprophytism: A prospective observational study. PLoS ONE 2019, 14, e0223072. [Google Scholar] [CrossRef]
- Milillo, L.; Lo Muzio, L.; Carlino, P.; Serpico, R.; Coccia, E.; Scully, C. Candida-related denture stomatitis: A pilot study of the efficacy of an amorolfine antifungal varnish. Int. J. Prosthodont. 2005, 18, 55–59. [Google Scholar] [PubMed]
- Sardaro, N.; Della Vella, F.; Incalza, M.A.; Di Stasio, D.; Lucchese, A.; Contaldo, M.; Laudadio, C.; Petruzzi, M. Oxidative Stress and Oral Mucosal Diseases: An Overview. In Vivo 2019, 33, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, I.; Fusco, A.; Grimaldi, E.; Perillo, L.; Coretti, L.; Di Domenico, M.; Cozza, V.; Contaldo, M.; Serpico, R.; Guida, A.; et al. Assessment of host defence mechanisms induced by Candida species. Int. J. Immunopathol. Pharmacol. 2013, 26, 663–672. [Google Scholar] [CrossRef]
- Contaldo, M.; Romano, A.; Mascitti, M.; Fiori, F.; Della Vella, F.; Serpico, R.; Santarelli, A. Association between denture stomatitis, candida species and diabetic status. J. Biol. Regul. Homeost. Agents 2019, 33, 35–41. [Google Scholar]
- Di Domenico, M.; Pinto, F.; Quagliuolo, L.; Contaldo, M.; Settembre, G.; Romano, A.; Coppola, M.; Ferati, K.; Bexheti-Ferati, A.; Sciarra, A.; et al. The Role of Oxidative Stress and Hormones in Controlling Obesity. Front. Endocrinol. 2019, 1310, 540. [Google Scholar] [CrossRef]
- Pannone, G.; Santoro, A.; Carinci, F.; Bufo, P.; Papagerakis, S.M.; Rubini, C.; Campisi, G.; Giovannelli, L.; Contaldo, M.; Serpico, R.; et al. Double demonstration of oncogenic high risk human papilloma virus DNA and HPV-E7 protein in oral cancers. Int. J. Immunopathol. Pharmacol. 2011, 24, 95–101. [Google Scholar] [CrossRef]
- Castillo, G.D.V.; Blanc, S.L.; Sotomayor, C.E.; Azcurra, A.I. Study of virulence factor of Candida species in oral lesions and its association with potentially malignant and malignant lesions. Arch. Oral Biol. 2018, 91, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Segata, N.; Haake, S.K.; Mannon, P.; Lemon, K.P.; Waldron, L.; Gevers, D.; Huttenhower, C.; Izard, J. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 2012, 13, R42. [Google Scholar] [CrossRef] [PubMed]
- Wade, W.G.; Prosdocimi, E.M. Profiling of Oral Bacterial Communities. J. Dent. Res. 2020, 99, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Teles, R.; Teles, F.; Frias-Lopez, J.; Paster, B.; Haffajee, A. Lessons learned and unlearned in periodontal microbiology. Periodontol. 2000 2013, 62, 95–162. [Google Scholar] [CrossRef] [PubMed]
- Contaldo, M.; Lucchese, A.; Lajolo, C.; Rupe, C.; Di Stasio, D.; Romano, A.; Petruzzi, M.; Serpico, R. The Oral Microbiota Changes in Orthodontic Patients and Effects on Oral Health: An Overview. J. Clin. Med. 2021, 10, 780. [Google Scholar] [CrossRef] [PubMed]
- Holmstrup, P.; Damgaard, C.; Olsen, I.; Klinge, B.; Flyvbjerg, A.; Nielsen, C.H.; Hansen, P.R. Comorbidity of periodontal disease: Two sides of the same coin? An introduction for the clinician. J. Oral Microbiol. 2017, 9, 1332710. [Google Scholar] [CrossRef]
- Rydén, L.; Buhlin, K.; Ekstrand, E.; de Faire, U.; Gustafsson, A.; Holmer, J.; Kjellström, B.; Lindahl, B.; Norhammar, A.; Nygren, A.; et al. Periodontitis Increases the Risk of a First Myocardial Infarction: A Report from the PAROKRANK Study. Circulation 2016, 133, 576–583. [Google Scholar]
- Carinci, F.; Martinelli, M.; Contaldo, M.; Santoro, R.; Pezzetti, F.; Lauritano, D.; Candotto, V.; Mucchi, D.; Palmieri, A.; Tagliabue, A.; et al. Focus on periodontal disease and development of endocarditis. J. Biol. Regul. Homeost. Agents 2018, 32, 143–147. [Google Scholar]
- Potempa, J.; Mydel, P.; Koziel, J. The case for periodontitis in the pathogenesis of rheumatoid arthritis. Nat. Rev. Rheumatol. 2007, 13, 606–620. [Google Scholar] [CrossRef]
- Pritchard, A.B.; Crean, S.; Olsen, I.; Singhrao, S.K. Periodontitis, Microbiomes and their Role in Alzheimer’s Disease. Front. Aging Neurosci. 2017, 9, 336. [Google Scholar] [CrossRef]
- Singhrao, S.K.; Olsen, I. Assessing the role of Porphyromonas gingivalis in periodontitis to determine a causative relationship with Alzheimer’s disease. J. Oral Microbiol. 2019, 11, 1563405. [Google Scholar] [CrossRef] [PubMed]
- Kayaçetin, S.; Güreşçi, S. What is gastritis? What is gastropathy? How is it classified? Turk. J. Gastroenterol. 2014, 25, 233–247. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.J.; Lin, J.C.; Tu, S.P. Etiology and Prevention of Gastric Cancer. Gastrointest. Tumors 2016, 3, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Cui, H.; Yang, M.; Du, S.; Li, J.; Li, Y.; Liu, L.; Zhang, X.; Li, S. Tongue coating microbiome as a potential biomarker for gastritis including precancerous cascade. Protein Cell 2019, 10, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Flynn, K.J.; Baxter, N.T.; Schloss, P.D. Metabolic and Community Synergy of Oral Bacteria in Colorectal Cancer. mSphere 2016, 1, e00102–e00116. [Google Scholar] [CrossRef]
- Abed, J.; Maalouf, N.; Manson, A.L.; Earl, A.M.; Parhi, L.; Emgård, J.E.M.; Klutstein, M.; Tayeb, S.; Almogy, G.; Atlan, K.A.; et al. Colon Cancer-Associated Fusobacterium nucleatum May Originate from the Oral Cavity and Reach Colon Tumors via the Circulatory System. Front. Cell Infect. Microbiol. 2020, 10, 400. [Google Scholar] [CrossRef] [PubMed]
- Flemer, B.; Warren, R.D.; Barrett, M.P.; Cisek, K.; Das, A.; Jeffery, I.B.; Hurley, E.; O’Riordain, M.; Shanahan, F.; O’Toole, P.W. The oral microbiota in colorectal cancer is distinctive and predictive. Gut 2018, 67, 1454–1463. [Google Scholar] [CrossRef]
- Liu, Y.; Baba, Y.; Ishimoto, T.; Iwatsuki, M.; Hiyoshi, Y.; Miyamoto, Y.; Yoshida, N.; Wu, R.; Baba, H. Progress in characterizing the linkage between Fusobacterium nucleatum and gastrointestinal cancer. J. Gastroenterol. 2019, 54, 33–41. [Google Scholar] [CrossRef]
- López, R.L.; Burgos, M.J.G.; Gálvez, A.; Pulido, R.P. The human gastrointestinal tract and oral microbiota in inflammatory bowel disease: A state of the science review. APMIS 2017, 125, 3–10. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Li, H.; Ni, C.; Du, Z.; Yan, F. Human oral microbiota and its modulation for oral health. Biomed. Pharmacother. 2018, 99, 883–893. [Google Scholar] [CrossRef]
- Szkaradkiewicz, A.K.; Karpiński, T.M. Microbiology of chronic periodontitis. J. Biol. Earth Sci. 2013, 3, M14–M20. [Google Scholar]
- Karpiński, T.M. Role of Oral Microbiota in Cancer Development. Microorganisms 2019, 7, 20. [Google Scholar] [CrossRef]
- Whitmore, S.E.; Lamont, R.J. Oral bacteria and cancer. PLoS Pathog. 2014, 10, e1003933. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Jermanus, C.; Barbetta, B.; Choi, C.; Verbeke, P.; Ojcius, D.M.; Yilmaz, Ö. Porphyromonas gingivalis infection sequesters pro-apoptotic Bad through Akt in primary gingival epithelial cells. Mol. Oral Microbiol. 2010, 25, 89–101. [Google Scholar] [CrossRef]
- Nakhjiri, S.F.; Park, Y.; Yilmaz, O.; Chung, W.O.; Watanabe, K.; El-Sabaeny, A.; Park, K.; Lamont, R.J. Inhibition of epithelial cell apoptosis by Porphyromonas gingivalis. FEMS Microbiol. Lett. 2001, 200, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Landskron, G.; De la Fuente, M.; Thuwajit, P.; Thuwajit, C.; Hermoso, M.A. Chronic inflammation and cytokines in the tumor microenvironment. J. Immunol. Res. 2014, 2014, 149185. [Google Scholar] [CrossRef] [PubMed]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef]
- Abranches, J.; Zeng, L.; Kajfasz, J.K.; Palmer, S.R.; Chakraborty, B.; Wen, Z.T.; Richards, V.P.; Brady, L.J.; Lemos, J.A. Biology of oral streptococci. Microbiol. Spectr. 2018, 6, 6. [Google Scholar] [CrossRef]
- Brauncajs, M.; Sakowska, D.; Krzemiński, Z. Production of hydrogen peroxide by lactobacilli colonising the human oral cavity. Med. Dośw. Mikrobiol. 2001, 53, 331–336. [Google Scholar]
- Hussain, S.P.; Hofseth, L.J.; Harris, C.C. Radical causes of cancer. Nat. Rev. Cancer 2003, 3, 276–285. [Google Scholar] [CrossRef]
- Piao, J.Y.; Lee, H.G.; Kim, S.J.; Kim, D.H.; Han, H.J.; Ngo, H.K.; Park, S.A.; Woo, J.H.; Lee, J.S.; Na, H.K.; et al. Helicobacter pylori activates IL-6-STAT3 signaling in human gastric cancer cells: Potential roles for reactive oxygen species. Helicobacter 2016, 21, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Attene-Ramos, M.S.; Wagner, E.D.; Plewa, M.J.; Gaskins, H.R. Evidence that hydrogen sulfide is a genotoxic agent. Mol. Cancer Res. 2006, 4, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Hellmich, M.R.; Szabo, C. Hydrogen sulfide and cancer. Handb. Exp. Pharmacol. 2015, 230, 233–241. [Google Scholar] [PubMed]
- Singh, S.B.; Lin, H.C. Hydrogen sulfide in physiology and diseases of the digestive tract. Microorganisms 2015, 3, 866–889. [Google Scholar] [CrossRef]
- Downes, J.; Wade, W.G. Peptostreptococcus stomatis sp. nov., isolated from the human oral cavity. Int. J. Syst. Evol. Microb. 2006, 56, 751–754. [Google Scholar] [CrossRef]
- Lunt, S.J.; Chaudary, N.; Hill, R.P. The tumor microenvironment and metastatic disease. Clin. Exp. Metastasis 2009, 26, 19–34. [Google Scholar] [CrossRef]
- Mazzio, E.; Smith, B.; Soliman, K. Evaluation of endogenous acidic metabolic products associated with carbohydrate metabolism in tumor cells. Cell Biol. Toxicol. 2010, 26, 177–188. [Google Scholar] [CrossRef]
- Pavlova, S.I.; Jin, L.; Gasparovich, S.R.; Tao, L. Multiple alcohol dehydrogenases but no functional acetaldehyde dehydrogenase causing excessive acetaldehyde production from ethanol by oral streptococci. Microbiology 2013, 159, 1437–1446. [Google Scholar] [CrossRef]
- Cordero, O.J.; Varela-Calviño, R. Oral hygiene might prevent cancer. Heliyon 2018, 4, e00879. [Google Scholar] [CrossRef]
- Salazar, C.R.; Sun, J.; Li, Y.; Francois, F.; Corby, P.; Perez-Perez, G.; Dasanayake, A.; Pei, Z.; Chen, Y. Association between selected oral pathogens and gastric precancerous lesions. PLoS ONE 2013, 8, e51604. [Google Scholar] [CrossRef]
- Coker, O.O.; Dai, Z.; Nie, Y.; Zhao, G.; Cao, L.; Nakatsu, G.; Wu, W.K.; Wong, S.H.; Chen, Z.; Sung, J.J.Y.; et al. Mucosal microbiome dysbiosis in gastric carcinogenesis. Gut 2018, 67, 1024–1032. [Google Scholar] [CrossRef]
- Yamamura, K.; Baba, Y.; Miyake, K.; Nakamura, K.; Shigaki, H.; Mima, K.; Kurashige, J.; Ishimoto, T.; Iwatsuki, M.; Sakamoto, Y.; et al. Fusobacterium nucleatum in gastroenterological cancer: Evaluation of measurement methods using quantitative polymerase chain reaction and a literature review. Oncol. Lett. 2017, 14, 6373–6378. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.Y.; Tung, S.Y.; Pan, H.Y.; Yen, C.W.; Xu, H.W.; Lin, Y.J.; Deng, Y.F.; Hsu, W.T.; Wu, C.S.; Li, C. Increased Abundance of Clostridium and Fusobacterium in Gastric Microbiota of Patients with Gastric Cancer in Taiwan. Sci. Rep. 2018, 8, 158. [Google Scholar] [CrossRef] [PubMed]
- Klimesova, K.; Zakostelska, Z.J.; Tlaskalova-Hogenova, H. Oral Bacterial and Fungal Microbiome Impacts Colorectal Carcinogenesis. Front. Microbiol. 2018, 9, 774. [Google Scholar] [CrossRef] [PubMed]
- Bandara, H.M.H.N.; Panduwawala, C.P.; Samaranayake, L.P. Biodiversity of the human oral mycobiome in health and disease. Oral Dis. 2019, 25, 363–371. [Google Scholar] [CrossRef]
- Fusco, A.; Savio, V.; Cammarota, M.; Alfano, A.; Schiraldi, C.; Donnarumma, G. Beta-Defensin-2 and Beta-Defensin-3 Reduce Intestinal Damage Caused by Salmonella typhimurium Modulating the Expression of Cytokines and Enhancing the Probiotic Activity of Enterococcus faecium. J. Immunol. Res. 2017, 2017, 6976935. [Google Scholar] [CrossRef]
- Martu, I.; Goriuc, A.; Martu, M.A.; Vata, I.; Baciu, R.; Mocanu, R.; Surdu, A.E.; Popa, C.; Luchian, I. Identification of Bacteria Involved in Periodontal Disease Using Molecular Biology Techniques. Rev. Chim. 2017, 68, 2407–2412. [Google Scholar] [CrossRef]
- Garlet, G.P. Destructive and protective roles of cytokines in periodontitis: A re-appraisal from host defense and tissue destruction viewpoints. J. Dent. Res. 2010, 89, 1349–1363. [Google Scholar] [CrossRef]
- Petersen, P.E.; Ogawa, H. The global burden of periodontal disease: Towards integration with chronic disease prevention and control. Periodontol. 2000 2012, 60, 15–39. [Google Scholar] [CrossRef]
- Kassebaum, N.J.; Smith, A.G.C.; Bernabe, E.; Fleming, T.D.; Reynolds, A.E.; Vos, T.; Murray, C.J.L.; Marcenes, W. GBD Oral Health Collaborators. Global, Regional, and National Prevalence, Incidence, and Disability-Adjusted Life Years for Oral Conditions for 195 Countries, 1990–2015: A Systematic Analysis for the Global Burden of Diseases, Injuries, and Risk Factors. J. Dent. Res. 2017, 96, 380–387. [Google Scholar] [CrossRef]
- Loos, B.G. Systemic effects of periodontitis. Ann. R. Australas. Coll. Dent. Surg. 2006, 18, 27–29. [Google Scholar] [CrossRef]
- Hajishengallis, G. Immunomicrobial pathogenesis of periodontitis: Keystones, pathobionts, and host response. Trends Immunol. 2014, 35, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.W. Bidirectional interrelationships between diabetes and periodontal diseases: An epidemiologic perspective. Ann Periodontol. 2001, 6, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.T.; Deng, A.P.; Li, C.; Xia, L.Y.; Niu, Y.M.; Leng, W.D. Periodontal disease and risk of head and neck cancer: A meta-analysis of observational studies. PLoS ONE 2013, 8, e79017. [Google Scholar] [CrossRef]
- Zeng, X.T.; Tu, M.L.; Liu, D.Y.; Zheng, D.; Zhang, J.; Leng, W. Periodontal disease and risk of chronic obstructive pulmonary disease: A meta-analysis of observational studies. PLoS ONE 2012, 7, e46508. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Liu, R.; Li, G.; Deng, M.; Liu, L.; Zeng, X.T.; Nie, X. History of periodontitis as a risk factor for long-term survival of dental implants: A meta-analysis. Int. J. Oral Maxillofac. Implants 2014, 29, 1271–1280. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.T.; Avila-Ortiz, G.; Allareddy, V.; Johnson, G.K.; Elangovan, S. The association between periodontitis and coronary heart disease: A quality assessment of systematic reviews. J. Am. Dent. Assoc. 2013, 144, 371–379. [Google Scholar] [CrossRef]
- Yu, H.C.; Chen, T.P.; Wei, C.Y.; Chang, Y.C. Association between Peptic Ulcer Disease and Periodontitis: A Nationwide Population-Based Case-Control Study in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 912. [Google Scholar] [CrossRef] [PubMed]
- Brito, F.; de Barros, F.C.; Zaltman, C.; Pugas-Carvalho, A.T.; de Vasconcellos-Carneiro, A.J.; Guimarães-Fischer, R.; Gustafsson, A.; de Silva-Figueredo, C.M. Prevalence of periodontitis and DMFT index in patients with Crohn’s disease and ulcerative colitis. J. Clin. Periodontol. 2008, 35, 555–560. [Google Scholar] [CrossRef]
- Habashneh, R.A.; Khader, Y.S.; Alhumouz, M.K.; Jadallah, K.; Aylouni, Y. The association between inflammatory bowel disease and periodontitis among Jordanians: A case-control study. J. Periodontal. Res. 2012, 47, 293–298. [Google Scholar] [CrossRef]
- Wei, X.; Zhao, H.Q.; Ma, C.; Zhang, A.B.; Feng, H.; Zhang, D.; Liu, C. The association between chronic periodontitis and oral Helicobacter pylori: A meta-analysis. PLoS ONE 2019, 14, e0225247. [Google Scholar] [CrossRef] [PubMed]
- Boylan, M.R.; Khalili, H.; Huang, E.S.; Michaud, D.S.; Izard, J.; Joshipura, K.J.; Chan, A.T. A prospective study of periodontal disease and risk of gastric and duodenal ulcer in male health professionals. Clin. Transl. Gastroenterol. 2014, 5, e49. [Google Scholar] [CrossRef] [PubMed]
- Byun, S.H.; Min, C.; Hong, S.J.; Choi, H.G.; Koh, D.H. Analysis of the Relation between Periodontitis and Chronic Gastritis/Peptic Ulcer: A Cross-Sectional Study Using KoGES HEXA Data. Int. J. Environ. Res. Public Health 2020, 17, 4387. [Google Scholar] [CrossRef] [PubMed]
- Umeda, M.; Kobayashi, H.; Takeuchi, Y.; Hayashi, J.; Morotome-Hayashi, Y.; Yano, K.; Aoki, A.; Ohkusa, T.; Ishikawa, I. High prevalence of Helicobacter pylori detected by PCR in the oral cavities of periodontitis patients. J. Periodontol. 2003, 74, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Franzosa, E.A.; Morgan, X.C.; Segata, N.; Waldron, L.; Reyes, J.; Earl, A.M.; Giannoukos, G.; Boylan, M.R.; Ciulla, D.; Gevers, D.; et al. Relating the metatranscriptome and metagenome of the human gut. Proc. Natl. Acad. Sci. USA 2014, 111, E2329–E2338. [Google Scholar] [CrossRef]
- Andersen, R.N.; Ganeshkumar, N.; Kolenbrander, P.E. Helicobacter pylori adheres selectively to Fusobacterium spp. Oral Microbiol. Immunol. 1998, 13, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.S.; Kamath, K.P.; Anil, S. Role of dental plaque, saliva and periodontal disease in Helicobacter pylori infection. World J. Gastroenterol. 2014, 20, 5639–5653. [Google Scholar] [CrossRef] [PubMed]
- Teoman, I.; Ozmeriç, N.; Ozcan, G.; Alaaddinoğlu, E.; Dumlu, S.; Akyön, Y.; Baloş, K. Comparison of different methods to detect Helicobacter pylori in the dental plaque of dyspeptic patients. Clin. Oral Investig. 2007, 11, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Chua, E.G.; Chong, J.Y.; Lamichhane, B.; Webberley, K.M.; Marshall, B.J.; Wise, M.J.; Tay, C.J. Gastric Helicobacter pylori infection perturbs human oral microbiota. PeerJ 2019, 7, e6336. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Gao, X.; Guo, J.; Yu, D.; Xiao, Y.; Wang, H.; Li, Y. Helicobacter pylori infection alters gastric and tongue coating microbial communities. Helicobacter 2019, 24, e12567. [Google Scholar] [CrossRef]
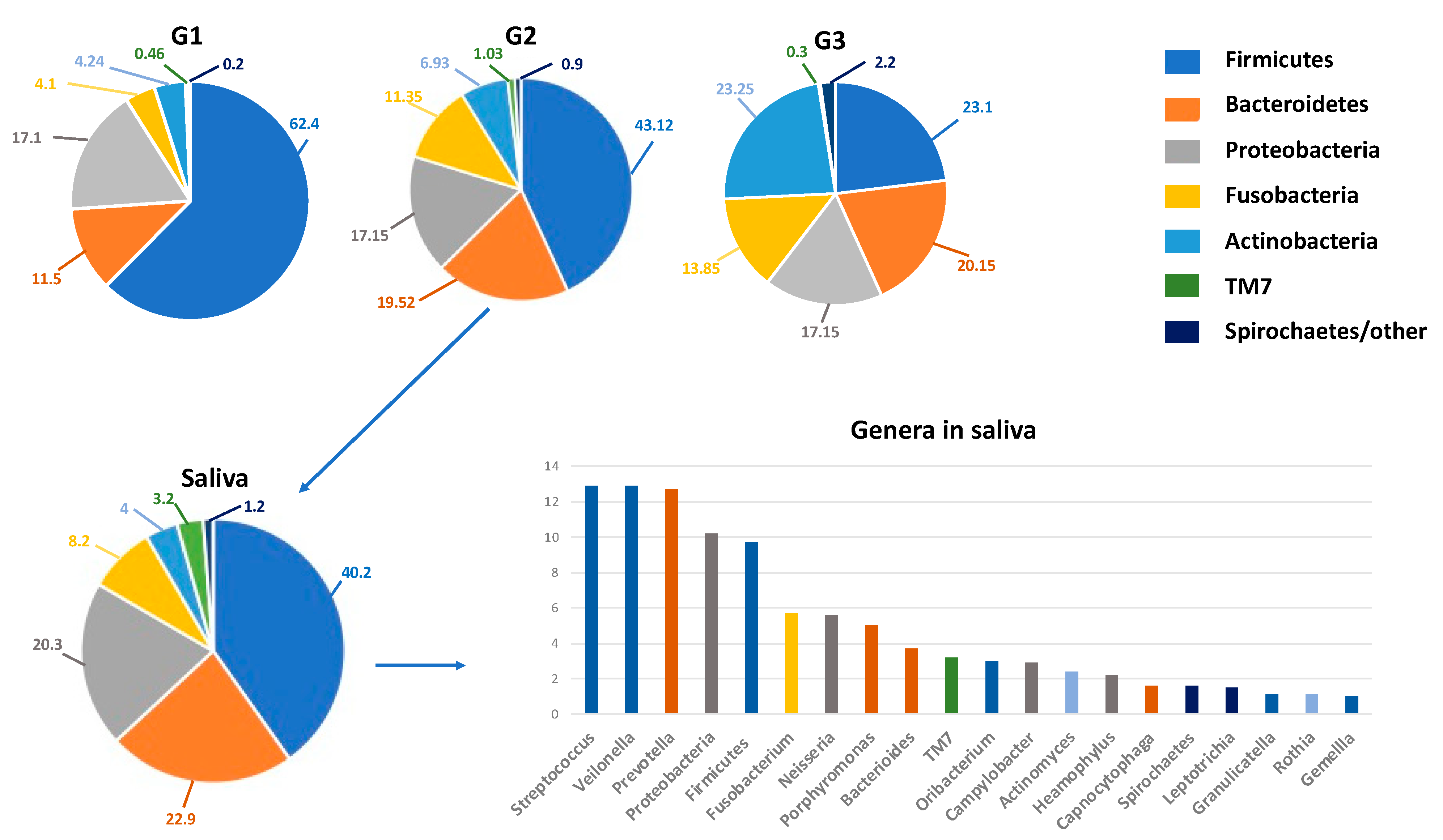
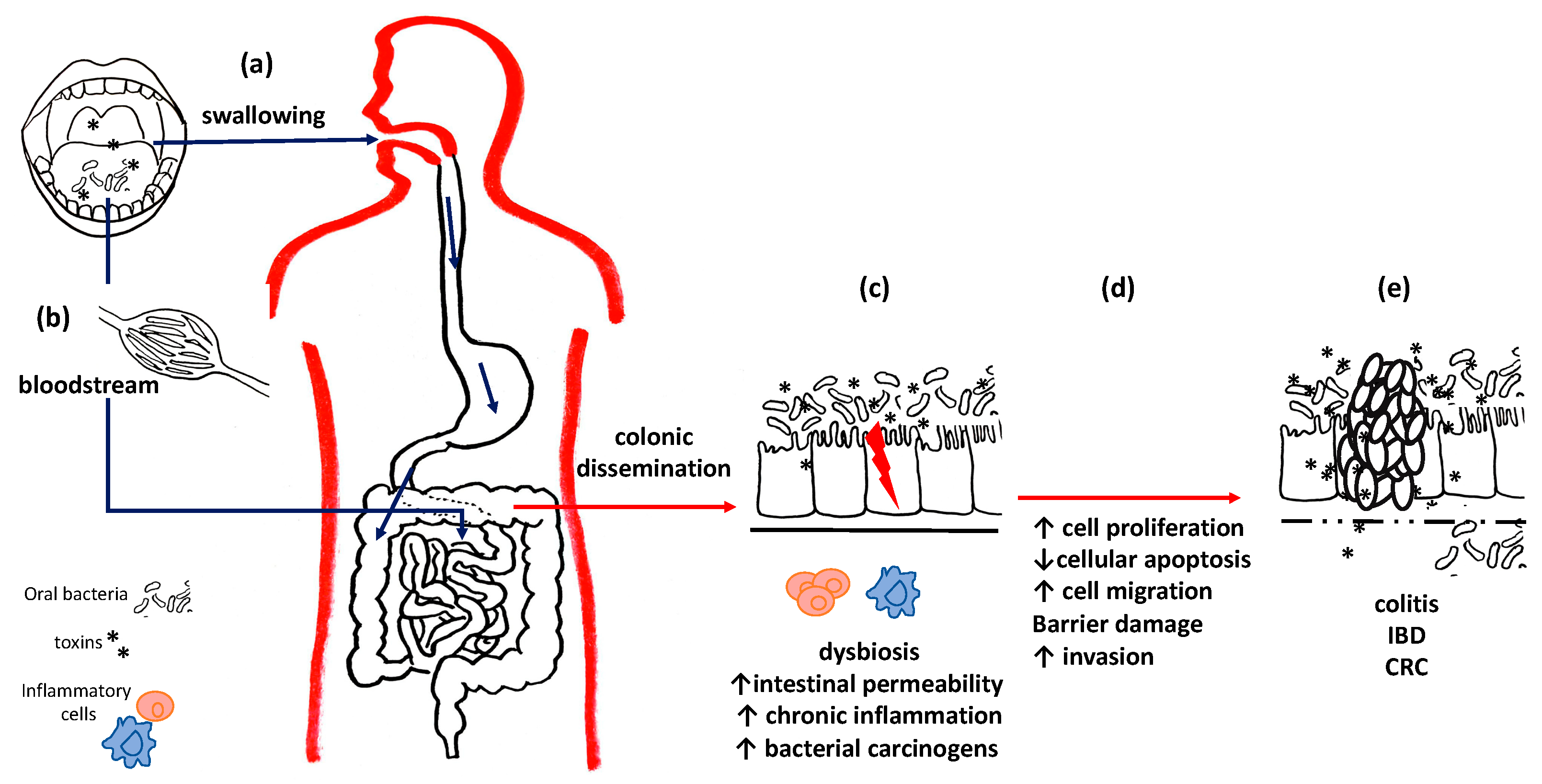
| G-i Diseases | Oral Bacteria/Fungi | Main Findings | Ref. |
|---|---|---|---|
| Gastritis | Streptococci, Fusobacteria | significantly higher in gastritis vs. healthy controls | [46] |
| Veillonella parvula, Corynebacterium matruchotii, Kingella oralis, Atopobium rimae, Aggregatibacter aphrophilus, Streptococcus sanguinis, Acinetobacter lwoffii, Prevotella amnii, Prevotella bivia, Cardiobacterium hominis and Oribacterium sinus | decreased in gastritis patients vs. healthy control | [46] | |
| Streptococcus infantis, Treponema vincentii, Leptotrichia unclassified, Campylobacter rectus, Campylobacter showae, Capnocytophaga gingivalis, Leptotrichia buccalis, Campylobacter concisus, Selenomonas flueggei and Leptotrichia hofstadii | increased in gastritis patients vs. healthy control (mainly Campylobacter spp.) | [46] | |
| Gastric Precancerous lesions | Campylobacter concisus | positively associated with the precancerous cascade of gastritis | [46] |
| Porphyromonas gingivalis, Treponema denticola | increased in dental plaque of subjects with gastric precancerous lesions | [72] | |
| Actinobacillus actinomycetemcomitans, Treponema denticola | significantly associated with gastric precancerous lesions | [72] | |
| Tannerella forsythia | significantly inversely associated with gastric precancerous lesions | [72] | |
| Oesophageal and Gastric Cancers | Tannerella forsythia, Porphyromonas gingivalis | associated with higher risk of oesophageal cancers | [52] |
| Streptococcus anginosus | higher in oesophageal cancer tissues than in oral cancer tissues | [52] | |
| Fusobacterium nucleatum | higher in oesophageal cancer tissues than matched normal mucosa; significantly associated with tumor stage and cancer-specific survival | [52] | |
| Neisseria spp., Candida glabrata | potential role in alcohol-related carcinogenesis | [52] | |
| Parvimonas micra, Peptostreptococcus stomatis, Prevotella intermedia, Fusobacterium nucleatum, Prevotella oris, Gemella and Catonella morbi, Streptococcus anginosus, Dialister pneumosintes, Slackia exigua | significantly increased in gastric cancer compared with precancerous stages | [73] | |
| Inflammatory bowel diseases (IBDs) | Bacteroidetes | significantly increased in IBDs | [51] |
| Proteobacteria and Actinobacteria | increased in IBDs | [51] | |
| Campylobacter concisus | increases the mucosal permeability by affecting the tight junctions in IBDs | [51] | |
| Fusobacterium nucleatum | overrepresented in IBDs | [51] | |
| Candida albicans | isolated from the intestine more frequently in IBD patients | [51] | |
| CRC | Haemophilus spp., Prevotella spp., Alloprevotella Lachnoanaerobaculum, Neisseria and Streptococcus spp. | less abundant in CRC than healthy controls | [49] |
| Fusobacterium nucleatum, Parvimonas micra, Peptostreptococcus stomatis, Dialister pneumosintes | tumor-associated bacteria | [49] | |
| Peptostreptococcus, Parvimonas, Fusobacterium | more abundant in CRC than in healthy controls | [49] | |
| Fusobacterium nucleatum | induces inflammatory response and promotes CRC development | [52] | |
| Treponema denticola, Prevotella intermedia | increases the CRC risk | [52] | |
| Porphyromonas gingivalis | causes inflammation and promotes tissue degenerative processes | [52] | |
| Fusobacterium nucleatum | associated with CRC regional lymph node metastases | [55] | |
| Fusobacterium nucleatum, Selenomonas, Prevotella, Parvimonas micra, Peptostreptococcus stomatis | increased in CRC; induces colon cancer growth and progression | [71] | |
| Lachnospiraceae | can protect against CRC | [71] | |
| Fusobacterium nucleatum | sustains both the biofilm and the CRC tumorigenesis | [47] |
| Gene | Primers Sequence | Conditions | ProductSize (bp) |
|---|---|---|---|
| Fusobacterium nucleatum | 5′-AGAGTTTGATCCTGGCTCAG-3′ 5′-GTCATCGTGCACACAGAATTGCTG-3′ | 5″ at 95 °C, 16″ at 55 °C, 8″ at 72 °C for 40 cycles | 407 |
| Porphyromonas gingivalis | 5′-TGTAGATGACTGATGGTGAAAACC-3′ 5′-ACGTCATCCCCACCTTCCTC-3′ | 5″ at 95 °C, 5” at 52 °C, 4″ at 72 °C for 40 cycles | 198 |
| Candida albicans | 5′-TTTATCAACTTGTCACACCAGA-3′ 5′-GGTCAAAGTTTGAAGATATACGT-3′ | 10″ at 95 °C, 10″ at 58 °C, 15″ at 72 °C for 30 cycles | 354 |
| Id. Patient | Age (Years) | Sex | F.n. (ng/dL) | P.g. (pg/mL) | C.a. (pg/mL) | F.n. per Group (Mean ± SD) | P.g. per Group (Mean ± SD) | C.a. per Group * (Mean ± SD) | |
|---|---|---|---|---|---|---|---|---|---|
| CG Group | 2 | 49 | F | 0.10 | 0.03 | 156 | 1.10 ± 1.62 | 0.57 ± 1.17 | 39.36 ± 57.80 |
| 3 | 50 | F | 0.02 | 0.05 | 0 | ||||
| 6 | 68 | F | 2.40 | 0.04 | 0 | ||||
| 8 | 23 | F | 4.20 | 3.20 | 71.5 | ||||
| 10 | 47 | F | 0.00 | 0.15 | 12 | ||||
| 30 | 55 | F | 0.93 | 0.08 | 0 | ||||
| 44 | 46 | F | 0.08 | 0.44 | 36 | ||||
| Ex-CRC Group | 17 | 67 | M | 40.50 | 29.75 | 0 | 31.62 ± 34.40 | 7.78 ± 11.37 | 14.08 ± 15.76 |
| 22 | 58 | M | 3.85 | 1.4 | 0 | ||||
| 24 | 66 | F | 1.37 | 8.75 | 0 | ||||
| 27 | 63 | M | 95.00 | 0.01 | 32.5 | ||||
| 48 | 71 | F | 28.50 | 6.75 | 22.5 | ||||
| 50 | 62 | M | 20.50 | 0.000 | 29.50 | ||||
| CRC Group | 20 | 80 | M | 9.50 | 0.05 | 365.5 | 9.13 ± 6.03 | 2.88 ± 3.68 | 91.25 ± 182.50 |
| 29 | 63 | M | 1.50 | 0.03 | 0 | ||||
| 31 | 87 | F | 9.25 | 3.70 | 0 | ||||
| 39 | 63 | M | 16.25 | 7.75 | 0 | ||||
| Healthy control | 9 | 49 | F | 56.50 | 296.50 | 0 | 65.06 ± 14.92 | 110.19 ± 127.37 | 0 |
| 42 | 54 | F | 85.00 | 10.25 | 0 | ||||
| 45 | 51 | M | 67.50 | 78.00 | 0 | ||||
| 34 | 64 | F | 51.25 | 56.5 | 0 |
| CG Group | Ex-CRC Group | CRC Group | Healthy Group | |
|---|---|---|---|---|
| Sample size | 7 | 6 | 4 | 4 |
| Mean F.n. values | 1.10 | 31.62 | 9.13 | 65.06 |
| Standard Deviation | 1.62 | 34.40 | 6.03 | 14.92 |
| t-student test | CG vs. ex-CRC * p < 0.05 CG vs. CRC * p < 0.05 CG vs. Healthy * p < 0.05 Healthy vs. CRC * p < 0.05 Healthy vs. Ex-CRC n.s. (p = 0.11) CRC vs. ex-CRC n.s. (p = 0.24) | |||
| CG Group | Ex-CRC Group | CRC Group | Healthy Control | |
|---|---|---|---|---|
| Sample size | 7 | 6 | 4 | 4 |
| Mean P.g. values | 0.57 | 7.78 | 2.88 | 110.19 |
| Standard Deviation | 1.17 | 11.37 | 3.68 | 127.37 |
| t-student test | CG vs. ex-CRC n.s. (p = 0.12) CG vs. CRC n.s. (p = 0.15) CG vs. Healthy * p < 0.05 Healthy vs. CRC n.s. (p = 0.14) Healthy vs. Ex-CRC n.s. (p = 0.07) CRC vs. ex-CRC n.s. (p = 0.44) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contaldo, M.; Fusco, A.; Stiuso, P.; Lama, S.; Gravina, A.G.; Itro, A.; Federico, A.; Itro, A.; Dipalma, G.; Inchingolo, F.; et al. Oral Microbiota and Salivary Levels of Oral Pathogens in Gastro-Intestinal Diseases: Current Knowledge and Exploratory Study. Microorganisms 2021, 9, 1064. https://doi.org/10.3390/microorganisms9051064
Contaldo M, Fusco A, Stiuso P, Lama S, Gravina AG, Itro A, Federico A, Itro A, Dipalma G, Inchingolo F, et al. Oral Microbiota and Salivary Levels of Oral Pathogens in Gastro-Intestinal Diseases: Current Knowledge and Exploratory Study. Microorganisms. 2021; 9(5):1064. https://doi.org/10.3390/microorganisms9051064
Chicago/Turabian StyleContaldo, Maria, Alessandra Fusco, Paola Stiuso, Stefania Lama, Antonietta Gerarda Gravina, Annalisa Itro, Alessandro Federico, Angelo Itro, Gianna Dipalma, Francesco Inchingolo, and et al. 2021. "Oral Microbiota and Salivary Levels of Oral Pathogens in Gastro-Intestinal Diseases: Current Knowledge and Exploratory Study" Microorganisms 9, no. 5: 1064. https://doi.org/10.3390/microorganisms9051064
APA StyleContaldo, M., Fusco, A., Stiuso, P., Lama, S., Gravina, A. G., Itro, A., Federico, A., Itro, A., Dipalma, G., Inchingolo, F., Serpico, R., & Donnarumma, G. (2021). Oral Microbiota and Salivary Levels of Oral Pathogens in Gastro-Intestinal Diseases: Current Knowledge and Exploratory Study. Microorganisms, 9(5), 1064. https://doi.org/10.3390/microorganisms9051064












