Loop-Mediated Isothermal Amplification Allows Rapid, Simple and Accurate Molecular Diagnosis of Human Cutaneous and Visceral Leishmaniasis Caused by Leishmania infantum When Compared to PCR
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Samples
2.2. Sample Preparation
- Samples from VL suspects: 200 μL of ethylene diamine tetra-acetic acid (EDTA)-treated peripheral blood, bone marrow.
- Samples from CL suspects: 200 μL skin biopsy (usually 5 mm diameter punch) macerated in NET10 buffer (10 mM NaCl, 10 mM EDTA, 10 mM Tris-HCl, pH 8.0).
2.3. Reference Test: Leishmania-Nested PCR (LnPCR)
2.4. Index Test-1: Loop-Mediated Isothermal Amplification (Loopamp)
2.5. Index Test-2: Real-Time Quantitative PCR (qPCR)
2.6. Background Information on Reference and Index Tests
2.7. Data Analysis
2.8. Ethics
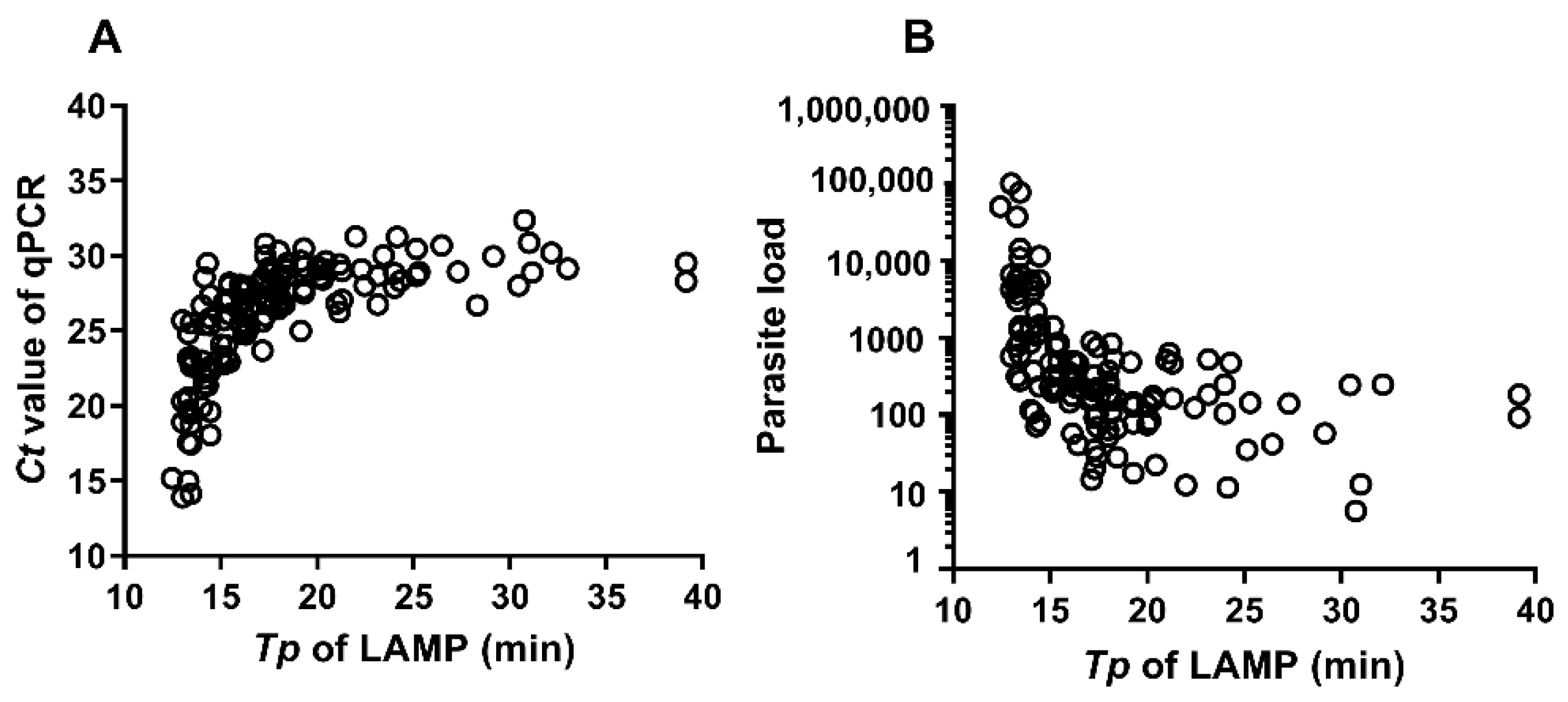
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Leishmaniasis Fact Sheet. Available online: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed on 24 February 2021).
- WHO Expert Committee on the Control of the Leishmaniases & World Health Organization. Control of the Leishmaniases: Report of a Meeting of the WHO Expert Commitee on the Control of Leishmaniases, Geneva, 22–26 March 2010. World Health Organization. Available online: https://apps.who.int/iris/handle/10665/44412 (accessed on 14 March 2021).
- Kaye, P.M.; Cruz, I.; Picado, A.; Van Bocxlaer, K.; Croft, S.L. Leishmaniasis immunopathology—impact on design and use of vaccines, diagnostics and drugs. Semin. Immunopathol. 2020, 42, 247–264. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Leishmaniasis in High-Burden Countries: An Epidemiological Update Based on Data Reported in 2014. Wkly Epidemiol. Rec. 2016, 91, 287–296. Available online: https://www.who.int/wer/2016/wer9122.pdf?ua=1 (accessed on 14 March 2021).
- Verrest, L.; Kip, A.E.; Musa, A.; Schoone, G.J.; Schallig, H.D.F.H.; Mbui, J.; Khalil, E.A.G.; Younis, B.M.; Olobo, J.; Were, L.; et al. Blood parasite load as an early marker to predict treatment response in visceral leishmaniasis in Eastern Africa. Clin. Infect. Dis. 2021, ciab124. [Google Scholar] [CrossRef] [PubMed]
- De Vries, H.J.C.; Reedijk, S.H.; Schallig, H.D.F.H. Cutaneous Leishmaniasis: Recent Developments in Diagnosis and Management. Am. J. Clin. Dermatol. 2015, 16, 99–109. [Google Scholar] [CrossRef] [PubMed]
- De Ruiter, C.M.; Van Der Veer, C.; Leeflang, M.M.G.; Deborggraeve, S.; Lucas, C.J.; Adams, E.R. Molecular Tools for Diagnosis of Visceral Leishmaniasis: Systematic Review and Meta-Analysis of Diagnostic Test Accuracy. J. Clin. Microbiol. 2014, 52, 3147–3155. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchai, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, E63. [Google Scholar] [CrossRef]
- Adams, E.R.; Schoone, G.; Versteeg, I.; Gomez, M.A.; Diro, E.; Mori, Y.; Perlee, D.; Downing, T.; Saravia, N.; Assaye, A.; et al. Development and Evaluation of a Novel Loop-Mediated Isothermal Amplification Assay for Diagnosis of Cutaneous and Visceral Leishmaniasis. J. Clin. Microbiol. 2018, 56, e00386-18. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar, M.; Ali, S.S.; Boshara, S.A.; Albertini, A.; Monnerat, S.; Bessell, P.; Mori, Y.; Kubota, Y.; Ndung’U, J.M.; Cruz, I. Sensitive and less invasive confirmatory diagnosis of visceral leishmaniasis in Sudan using loop-mediated isothermal amplification (LAMP). PLoS Negl. Trop. Dis. 2018, 12, e0006264. [Google Scholar] [CrossRef]
- Vink, M.M.; Nahzat, S.M.; Rahimi, H.; Buhler, C.; Ahmadi, B.A.; Nader, M.; Zazai, F.R.; Yousufzai, A.S.; van Loenen, M.; Schallig, H.D.; et al. Evaluation of point-of-care tests for cutaneous leishmaniasis diagnosis in Kabul, Afghanistan. EBioMedicine 2018, 37, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Schallig, H.D.F.H.; Hu, R.V.P.; Kent, A.D.; Van Loenen, M.; Menting, S.; Picado, A.; Oosterling, Z.; Cruz, I. Evaluation of point of care tests for the diagnosis of cutaneous leishmaniasis in Suriname. BMC Infect. Dis. 2019, 19, 25. [Google Scholar] [CrossRef]
- Ibarra-Meneses, A.V.; Cruz, I.; Chicharro, C.; Sánchez, C.; Biéler, S.; Broger, T.; Moreno, J.; Carrillo, E. Evaluation of fluorimetry and direct visualization to interpret results of a loop-mediated isothermal amplification kit to detect Leishmania DNA. Parasites Vectors 2018, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cruz, I.; Cañavate, C.; Rubio, J.; Morales, M.; Chicharro, C.; Laguna, F.; Jiménez-Mejías, M.; Sirera, G.; Videla, S.; Alvar, J. A nested polymerase chain reaction (Ln-PCR) for diagnosing and monitoring Leishmania infantum infection in patients co-infected with human immunodeficiency virus. Trans. R. Soc. Trop. Med. Hyg. 2002, 96, S185–S189. [Google Scholar] [CrossRef]
- Cruz, I.; Millet, A.; Carrillo, E.; Chenik, M.; Salotra, P.; Verma, S.; Veland, N.; Jara, M.; Adaui, V.; Castrillón, C.; et al. An approach for interlaboratory comparison of conventional and real-time PCR assays for diagnosis of human leishmaniasis. Exp. Parasitol. 2013, 134, 281–289. [Google Scholar] [CrossRef]
- Cruz, I.; Chicharro, C.; Nieto, J.; Bailo, B.; Canavate, C.; Figueras, M.A.-C.; Alvar, J. Comparison of New Diagnostic Tools for Management of Pediatric Mediterranean Visceral Leishmaniasis. J. Clin. Microbiol. 2006, 44, 2343–2347. [Google Scholar] [CrossRef][Green Version]
- Mirã, G.; Oliva, G.; Cruz, I.; Cañavate, C.; Mortarino, M.; Vischer, C.; Bianciardi, P. Multicentric, controlled clinical study to evaluate effectiveness and safety of miltefosine and allopurinol for canine leishmaniosis. Vet. Dermatol. 2009, 20, 397–404. [Google Scholar] [CrossRef]
- Cunha, J.; Carrillo, E.; Sánchez, C.; Cruz, I.; Moreno, J.; Cordeiro-Da-Silva, A. Characterization of the biology and infectivity of Leishmania infantum viscerotropic and dermotropic strains isolated from HIV+ and HIV- patients in the murine model of visceral leishmaniasis. Parasites Vectors 2013, 6, 122. [Google Scholar] [CrossRef]
- Cruz, I.; Acosta, L.; Gutiérrez, M.N.; Nieto, J.; Cañavate, C.; Deschutter, J.; Bornay-Llinares, F.J. A canine leishmaniasis pilot survey in an emerging focus of visceral leishmaniasis: Posadas (Misiones, Argentina). BMC Infect. Dis. 2010, 10, 342. [Google Scholar] [CrossRef]
- Salam, M.; Mondal, D.; Kabir, M.; Ekram, A.; Haque, R. PCR for diagnosis and assessment of cure in kala-azar patients in Bangladesh. Acta Trop. 2010, 113, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.S.; Ghosh, D.; Khan, G.M.; Islam, M.F.; Mondal, D.; Itoh, M.; Islam, N.; Haque, R. Survey of domestic cattle for anti-Leishmania antibodies and Leishmania DNA in a visceral leishmaniasis endemic area of Bangladesh. BMC Vet. Res. 2011, 7, 27. [Google Scholar] [CrossRef]
- Salam, M.A.; Khan, M.G.M.; Bhaskar, K.R.H.; Afrad, M.H.; Huda, M.M.; Mondal, D. Peripheral Blood Buffy Coat Smear: A Promising Tool for Diagnosis of Visceral Leishmaniasis. J. Clin. Microbiol. 2011, 50, 837–840. [Google Scholar] [CrossRef]
- Pérez, M.I.S.; Vidal, X.H.; Barbeito, G.N.; Silva, L.C.; Fariñas, H.; Vázquez, E.; Bacallao, J.; Mújica, O.J. El programa epidat: Usos y perspectivas. Revista Panamericana de Salud Pública 2010, 27, 80–82. [Google Scholar] [CrossRef] [PubMed]
- Ejov, M.; Dagne, D. Strategic Framework for Leishmaniasis Control in the WHO European Region 2014-2020. World Health Organ. 2014. Available online: https://www.euro.who.int/__data/assets/pdf_file/0017/245330/Strategic-framework-for-leishmaniasis-control-in-the-WHO-European-Region-20142020.pdf (accessed on 14 March 2021).
- Nzelu, C.O.; Kato, H.; Peters, N.C. Loop-mediated isothermal amplification (LAMP): An advanced molecular point-of-care technique for the detection of Leishmania infection. PLoS Negl. Trop. Dis. 2019, 13, e0007698. [Google Scholar] [CrossRef] [PubMed]
- WHO Collaborating Centres Global Database [Internet]. Available online: https://apps.who.int/whocc/Detail.aspx?qEjNpHwf6G82WRkvO8QOgA (accessed on 14 March 2021).
| Reference Test | |||
|---|---|---|---|
| Condition | Sample Type | LnPCR Positive | LnPCR Negative |
| VL suspect | Peripheral blood | 38 | 25 |
| Bone marrow | 48 | 19 | |
| CL suspect | Skin biopsy | 56 | 44 |
| Tests | LnPCR-Positive (Cases) | LnPCR-Negative (Controls) | Sensitivity (%) (95% CI) | Specificity (%) (95% CI) | |
|---|---|---|---|---|---|
| VL (Peripheral Blood) | Loopamp-Positive | 37 | 1 | 97.4 (0.9–100) | 96 (86.3–100) |
| Loopamp-Negative | 1 | 24 | |||
| qPCR-Positive | 38 | 1 | 100 (98.7–100) | 96 (86.3–100) | |
| qPCR-Negative | 0 | 24 | |||
| VL (Bone Marrow) | Loopamp-Positive | 47 | 0 | 97.9 (92.8–100) | 100 (97.4–100) |
| Loopamp-Negative | 1 | 19 | |||
| qPCR-Positive | 47 | 0 | 97.9 (92.8–100) | 100 (97.4–100) | |
| qPCR-Negative | 1 | 19 | |||
| CL (Skin Biopsy) | Loopamp-Positive | 56 | 0 | 100 (99.1–100) | 100 (98.8–100) |
| Loopamp-Negative | 0 | 44 | |||
| qPCR-Positive | 55 | 0 | 98.2 (93.8–100) | 100 (98.8–100) | |
| qPCR-Negative | 1 | 44 |
| qPCR | LnPCR | |
|---|---|---|
| LnPCR | C: 98.4% (94.4–99.8) K: 0.96 (0.91–1) | |
| Loopamp | C: 97.9% (93.2–99.5) K: 0.95 (0.9–1) | C: 98.4% (4.4–99.8) K: 0.96 (0.91–1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibarra-Meneses, A.V.; Chicharro, C.; Sánchez, C.; García, E.; Ortega, S.; Ndung’u, J.M.; Moreno, J.; Cruz, I.; Carrillo, E. Loop-Mediated Isothermal Amplification Allows Rapid, Simple and Accurate Molecular Diagnosis of Human Cutaneous and Visceral Leishmaniasis Caused by Leishmania infantum When Compared to PCR. Microorganisms 2021, 9, 610. https://doi.org/10.3390/microorganisms9030610
Ibarra-Meneses AV, Chicharro C, Sánchez C, García E, Ortega S, Ndung’u JM, Moreno J, Cruz I, Carrillo E. Loop-Mediated Isothermal Amplification Allows Rapid, Simple and Accurate Molecular Diagnosis of Human Cutaneous and Visceral Leishmaniasis Caused by Leishmania infantum When Compared to PCR. Microorganisms. 2021; 9(3):610. https://doi.org/10.3390/microorganisms9030610
Chicago/Turabian StyleIbarra-Meneses, Ana Victoria, Carmen Chicharro, Carmen Sánchez, Emilia García, Sheila Ortega, Joseph Mathu Ndung’u, Javier Moreno, Israel Cruz, and Eugenia Carrillo. 2021. "Loop-Mediated Isothermal Amplification Allows Rapid, Simple and Accurate Molecular Diagnosis of Human Cutaneous and Visceral Leishmaniasis Caused by Leishmania infantum When Compared to PCR" Microorganisms 9, no. 3: 610. https://doi.org/10.3390/microorganisms9030610
APA StyleIbarra-Meneses, A. V., Chicharro, C., Sánchez, C., García, E., Ortega, S., Ndung’u, J. M., Moreno, J., Cruz, I., & Carrillo, E. (2021). Loop-Mediated Isothermal Amplification Allows Rapid, Simple and Accurate Molecular Diagnosis of Human Cutaneous and Visceral Leishmaniasis Caused by Leishmania infantum When Compared to PCR. Microorganisms, 9(3), 610. https://doi.org/10.3390/microorganisms9030610







