Microbiological Sulfide Removal—From Microorganism Isolation to Treatment of Industrial Effluent
Abstract
1. Introduction
2. Materials and Methods
2.1. Microbial Consortium for Sulfide Oxidation
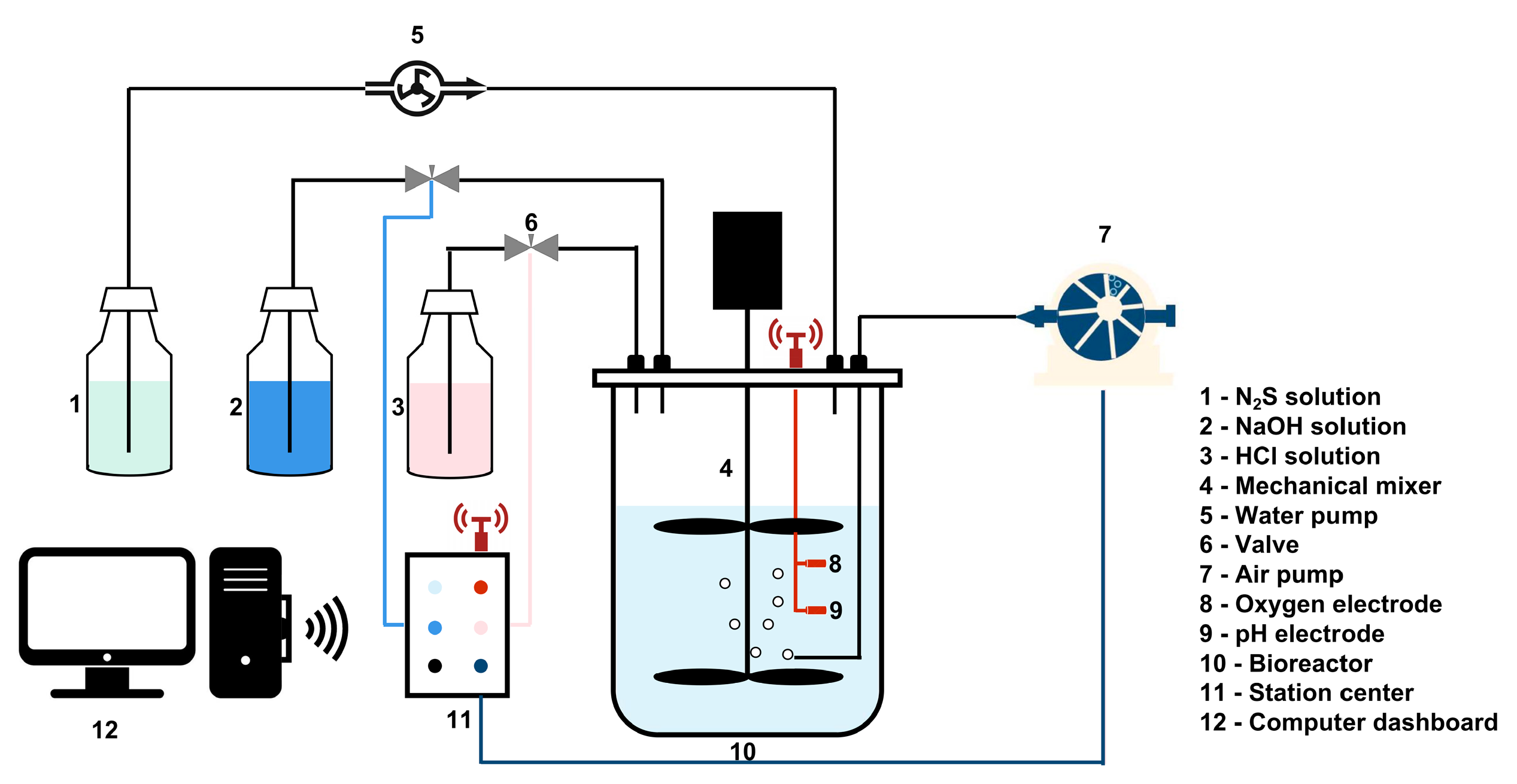
2.2. Experimental Set-Up
2.2.1. Na2S Bio-Oxidation and Sulfur Production
2.2.2. Two-Stage Bio-Oxidation of Industrial Effluent
2.3. Analytical Methods
2.3.1. Chemical Analysis
2.3.2. Solid Phase Analysis
2.3.3. Biomass Estimation
2.4. Bioinformatic Analysis and Data Realization
2.4.1. Extraction of DNA and PCR Amplification
2.4.2. 16S rDNA Sequencing
3. Results and Discussion
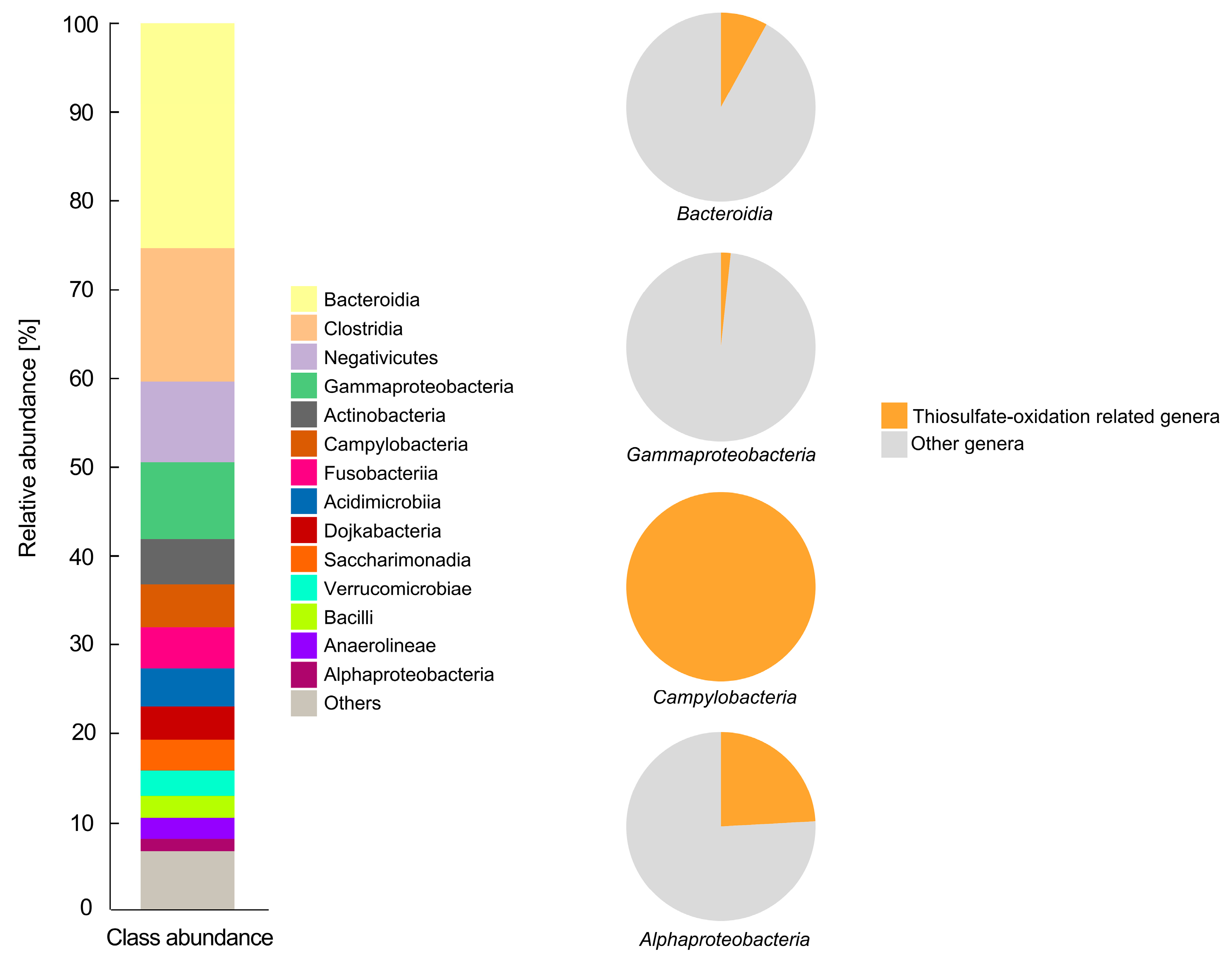
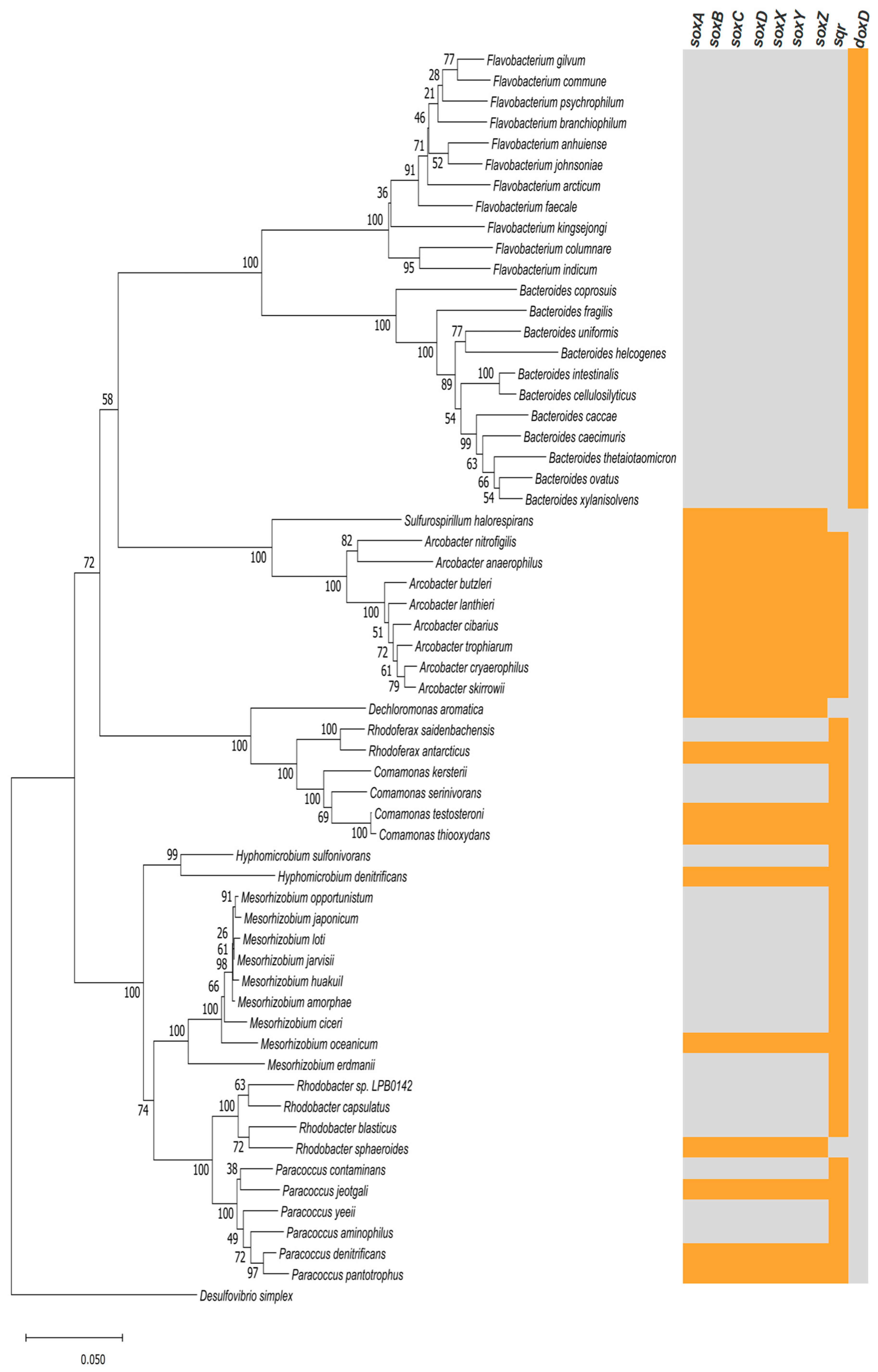
3.1. Isolation and Identification of Microbial S[2−] Oxidation Consortium
3.1.1. Structure of the Microbial Community
3.1.2. Sulfur Metabolic Pathways Prediction
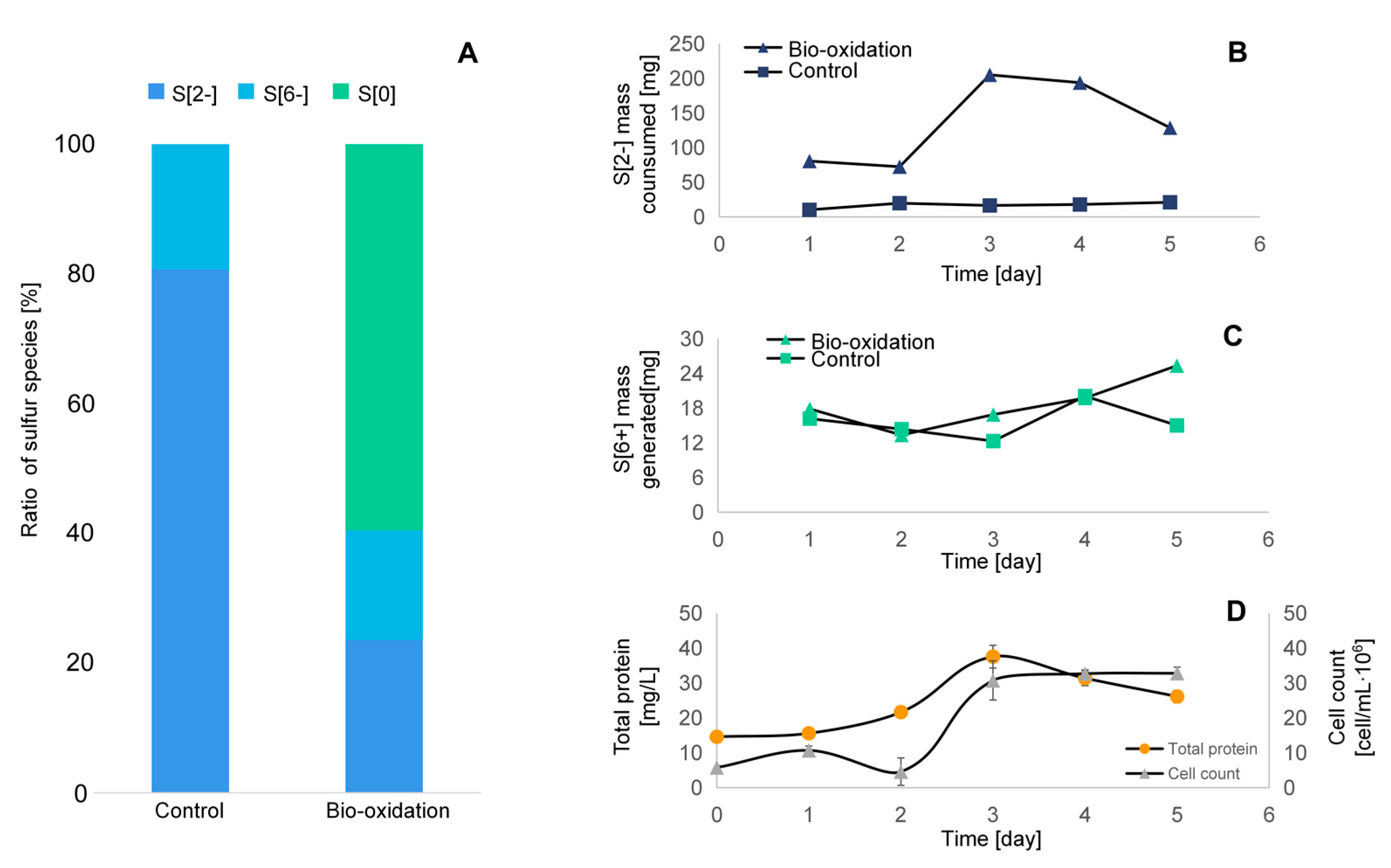
3.2. S[2−] Oxidation to Elemental Sulfur—Verification on the Synthetic Media
3.2.1. Total Sulfur Mass Balance
3.2.2. Sulfur Species as a Function of Time
3.2.3. Growth Curve of SOB during Sulfide Bio-Oxidation
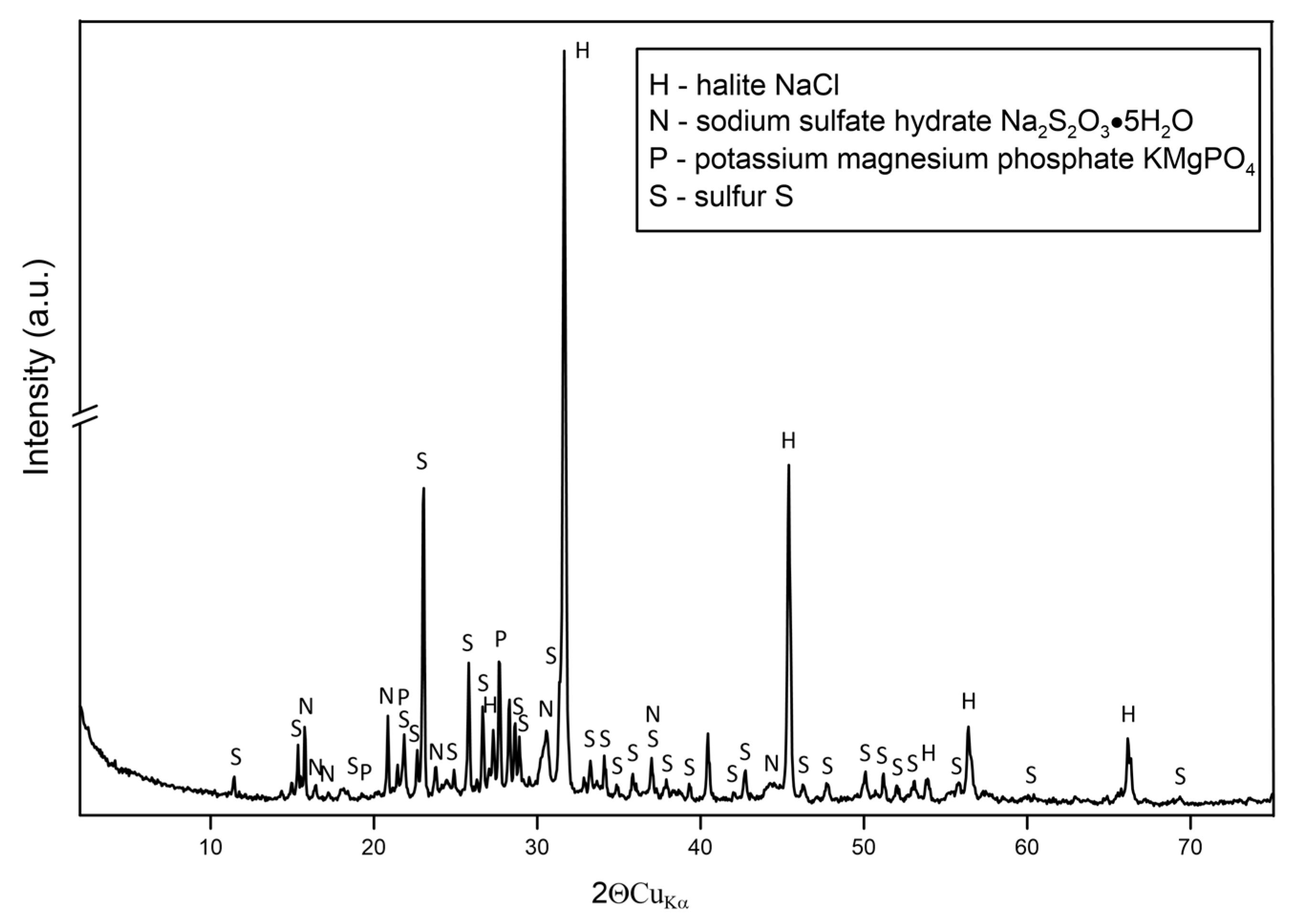
3.2.4. XRD Analysis of Sediments
3.3. Bioaugmented Treatment of the S[2−] Containing Industrial Effluent
3.3.1. Characterization of the Industrial Effluent
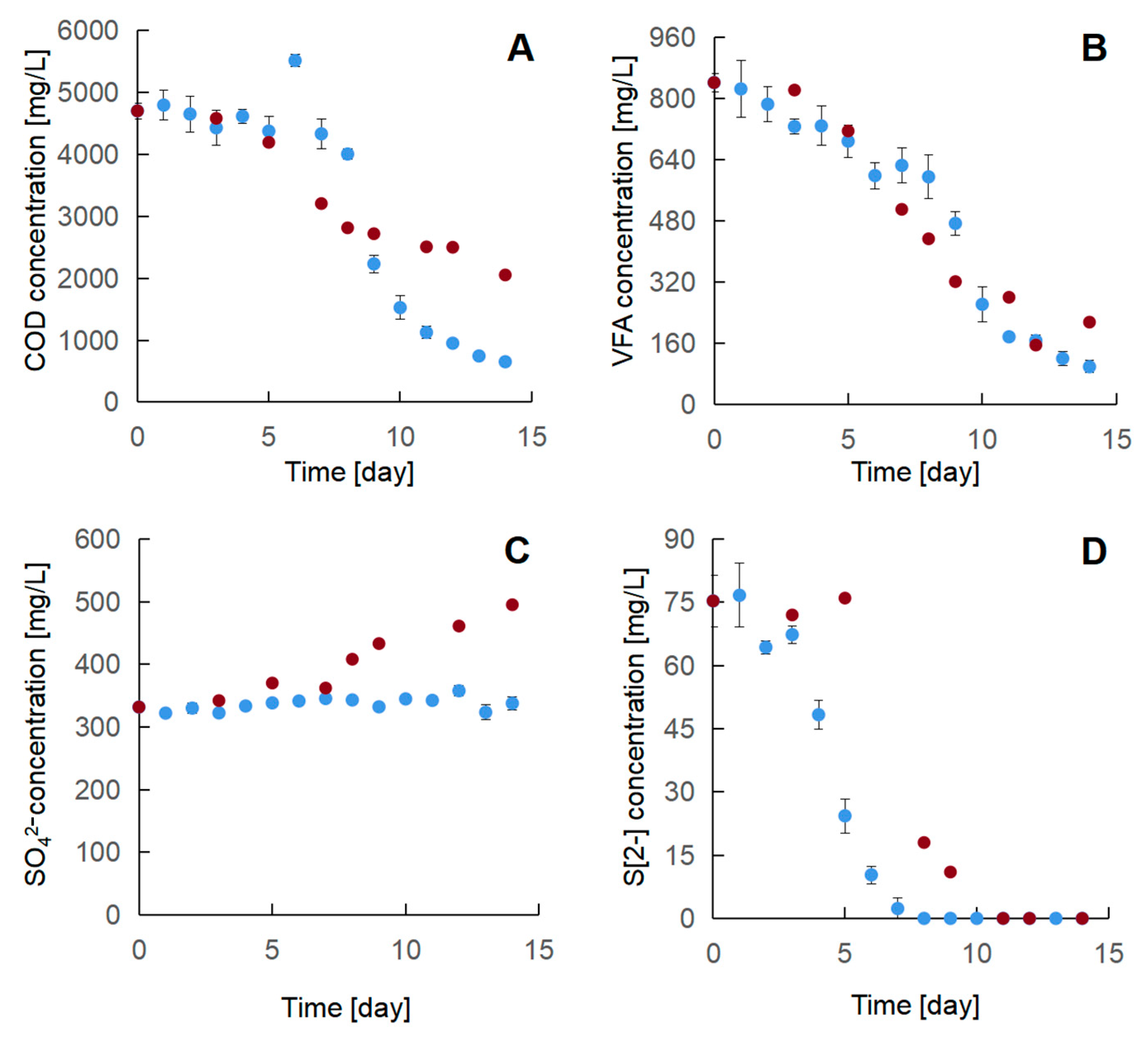
3.3.2. Two-Stage Oxidation of the Industrial Effluent
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xu, Y.-N.; Chen, Y. Advances in heavy metal removal by sulfate-reducing bacteria. Water Sci. Technol. 2020, 81, 1797–1827. [Google Scholar] [CrossRef]
- Feng, D.; Aldrich, C.; Tan, H. Treatment of acid mine water by use of heavy metal precipitation and ion exchange. Miner. Eng. 2000, 13, 623–642. [Google Scholar] [CrossRef]
- Iakovleva, E.; Sillanpää, M. The use of low-cost adsorbents for wastewater purification in mining industries. Environ. Sci. Pollut. Res. 2013, 20, 7878–7899. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.A.; Matthews, Z.N.; Morgan, G.; Bullen, C.; Stanley, P. Sono-electrochemistry (Electrolysis with assisted Power Ultrasound) Treatment Trials of discharges from Cwm Rheidol–Ystumtuen mines, Ceredigion, Mid Wales, UK. In Proceedings of the IMWA 2019 Conference—Mine Water: Technological and Ecological Challenges, Perm, Russia, 15–19 July 2019. [Google Scholar]
- Ayangbenro, A.S.; Olanrewaju, O.S.; Babalola, O.O.; Ayangbenro, A. Sulfate-Reducing Bacteria as an Effective Tool for Sustainable Acid Mine Bioremediation. Front. Microbiol. 2018, 9, 1986. [Google Scholar] [CrossRef]
- Vinçon-Laugier, A.; Cravo-Laureau, C.; Mitteau, I.; Grossi, V. Temperature-Dependent Alkyl Glycerol Ether Lipid Composition of Mesophilic and Thermophilic Sulfate-Reducing Bacteria. Front. Microbiol. 2017, 8, 1532. [Google Scholar] [CrossRef]
- Oliveira, F.R.; Surendra, K.; Jaisi, D.P.; Lu, H.; Unal-Tosun, G.; Sung, S.; Khanal, S.K. Alleviating sulfide toxicity using biochar during anaerobic treatment of sulfate-laden wastewater. Bioresour. Technol. 2020, 301, 122711. [Google Scholar] [CrossRef]
- Kiran, M.G.; Pakshirajan, K.; Das, G. Heavy metal removal from multicomponent system by sulfate reducing bacteria: Mechanism and cell surface characterization. J. Hazard. Mater. 2017, 324, 62–70. [Google Scholar] [CrossRef] [PubMed]
- UMass Extension (n.d.). Hydrogen Sulfide and Sulfate in Private Drinking Water Wells. Available online: https://ag.umass.edu/sites/ag.umass.edu/files/fact-sheets/pdf/hydrogensulfide.pdf (accessed on 23 January 2021).
- Safety Data Sheet According to Regulation (EC) No. 1907/2006 Version 6.2. Available online: https://www.sigmaaldrich.com/MSDS/MSDS/DisplayMSDSPage.do?country=PL&language=pl&productNumber=295442&brand=ALDRICH&PageToGoToURL=http%3A%2F%2Fwww.sigmaaldrich.com%2Fcatalog%2Fproduct%2Faldrich%2F295442%3Flang%3Dpl (accessed on 23 January 2021).
- Díaz, I.; Lopes, A.C.; Pérez, S.I.; Fdz-Polanco, M. Determination of the optimal rate for the microaerobic treatment of several H2S concentrations in biogas from sludge digesters. Water Sci. Technol. 2011, 64, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Ramos, I.; Pérez, R.; Fdz-Polanco, M. Microaerobic desulphurisation unit: A new biological system for the removal of H2S from biogas. Bioresour. Technol. 2013, 142, 633–640. [Google Scholar] [CrossRef]
- Sumner, M.E.; Noble, A.D. Soil acidification: The world story. In Handbook of Soil Acidity; Marcel Dekker: New York, NY, USA, 2003; pp. 1–28. [Google Scholar]
- Haruna, S.Y.; Faruq, U.Z.; Zubairu, A.Y.; Liman, M.G.; Riskuwa, M.L. Comparative studies on reduction of sulphur content of heavy crude oil using KMnO4+ H2O2/CH3COOH and KMnO4+ H2O2/HCOOH via oxidative desulphurization (ODS). Am. J. Appl. Chem. 2018, 6, 15–24. [Google Scholar] [CrossRef]
- Lin, S.; Mackey, H.R.; Hao, T.; Guo, G.; van Loosdrecht, M.C.; Chen, G. Biological sulfur oxidation in wastewater treatment: A review of emerging opportunities. Water Res. 2018, 143, 399–415. [Google Scholar] [CrossRef]
- Alcántara, S.; Velasco, A.; Muñoz, A.; Cid, J.; Revah, S.; Razo-Flores, E. Hydrogen sulfide oxidation by a microbial consortium in a recirculation reactor system: Sulfur formation under oxygen limitation and removal of phenols. Environ. Sci. Technol. 2004, 38, 918–923. [Google Scholar] [CrossRef]
- Krishnakumar, B.; Majumdar, S.; Manilal, V.; Haridas, A. Treatment of sulphide containing wastewater with sulphur recovery in a novel reverse fluidized loop reactor (RFLR). Water Res. 2005, 39, 639–647. [Google Scholar] [CrossRef]
- Krayzelova, L.; Bartacek, J.; Díaz, I.; Jeison, D.; Volcke, E.I.P.; Jenicek, P. Microaeration for hydrogen sulfide removal during anaerobic treatment: A review. Rev. Environ. Sci. Bio/Technology 2015, 14, 703–725. [Google Scholar] [CrossRef]
- THIOPAQ. Bio-Desulphurization System. 2020. Available online: https://www.slb.com/well-production/processing-and-separation/gas-treatment/thiopaq-biodesulfurization-system (accessed on 17 January 2021).
- Rewerski, B.; Ruszkowski, D.; Chajduk, E.; Sklodowska, A. Environmental aspects of post mining uranium wastes deposited in Radoniow, Poland. Physicochem. Probl. Miner. Process. 2014, 50, 71–77. [Google Scholar] [CrossRef]
- Vishniac, W.; Santer, M. The thiobacilli. Bacteriol. Rev. 1957, 21, 195. [Google Scholar] [CrossRef] [PubMed]
- Kuenen, J.G.; Robertson, L.A. The use of natural bacterial populations for the treatment of sulphur-containing wastewater. Microorg. Combat Pollut. 1992, 115–130. [Google Scholar]
- Karpmoi. A Vector-Graphics Illustration for the Entry Oxidation State. 2016. Available online: https://en.wikipedia.org/wiki/File:16oxstate.svg (accessed on 15 January 2021).
- Yang, Z.; Uhrynowski, W.; Jakusz, G.; Retka, J.; Karczewska-Golec, J.; Debiec-Andrzejewska, K.; Rogulski, Z.; Drewniak, L. Biochemical treatment of leachates from hydrometallurgical recycling of spent alkaline batteries. Hydrometallurgy 2020, 191, 105223. [Google Scholar] [CrossRef]
- Bartlett, J.K.; Skoog, D.A. Colorimetric Determination of Elemental Sulfur in Hydrocarbons. Anal. Chem. 1954, 26, 1008–1011. [Google Scholar] [CrossRef]
- Marciniak, H.; Diduszko, R.; Kozak, M. XRAYAN. Program do Rentgenowskiej Analizy Fazowej, Wersja. 2006, Volume 4. Available online: http://212.85.108.199/grafika/xrayan.pdf (accessed on 15 January 2021).
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Dziewit, L.; Pyzik, A.; Romaniuk, K.; Sobczak, A.; Szczesny, P.; Lipinski, L.; Bartosik, D.; Drewniak, L. Novel molecular markers for the detection of methanogens and phylogenetic analyses of methanogenic communities. Front. Microbiol. 2015, 6, 694. [Google Scholar] [CrossRef] [PubMed]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef]
- Kobayashi, T.; Li, Y.Y.; Kubota, K.; Harada, H.; Maeda, T.; Yu, H.Q. Characterization of sulfide-oxidizing microbial mats developed inside a full-scale anaerobic digester employing biological desulfurization. Appl. Microbiol. Biotechnol. 2012, 93, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Díaz, I.; Pérez, S.; Ferrero, E.; Fdz-Polanco, M. Effect of oxygen dosing point and mixing on the microaerobic removal of hydrogen sulphide in sludge digesters. Bioresour. Technol. 2011, 102, 3768–3775. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, C.G.; Rother, D.; Bardischewsky, F.; Quentmeier, A.; Fischer, J. Oxidation of Reduced Inorganic Sulfur Compounds by Bacteria: Emergence of a Common Mechanism? Appl. Environ. Microbiol. 2001, 67, 2873–2882. [Google Scholar] [CrossRef] [PubMed]
- Rother, D.; Henrich, H.-J.; Quentmeier, A.; Bardischewsky, F.; Friedrich, C.G. Novel Genes of the sox Gene Cluster, Mutagenesis of the Flavoprotein SoxF, and Evidence for a General Sulfur-Oxidizing System in Paracoccus pantotrophus GB17. J. Bacteriol. 2001, 183, 7253–7259. [Google Scholar] [CrossRef]
- Muyzer, G.; Stams, A.J. The ecology and biotechnology of sulphate-reducing bacteria. Nat. Rev. Microbiol. 2008, 6, 441–454. [Google Scholar] [CrossRef]
- Barton, L.L.; Fauque, G.D. Biochemistry, physiology and biotechnology of sulfate-reducing bacteria. Adv. Appl. Microbiol. 2009, 68, 41–98. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Lancaster, J.R. Chemical foundations of hydrogen sulfide biology. Nitric Oxide 2013, 35, 21–34. [Google Scholar] [CrossRef]
- Janssen, A.J.H.; Sleyster, R.; Van Der Kaa, C.; Jochemsen, A.; Bontsema, J.; Lettinga, G. Biological sulphide oxidation in a fed-batch reactor. Biotechnol. Bioeng. 1995, 47, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Kleinjan, W. Biologically Produced Sulphur Particles and Polysulphide Ions. Effects on a Biotechnological Process for the Removal of Hydrogen Sulphide from Gas Streams. Doctoral Dissertation, Wageningen University & Research, Wageningen, The Netherlands, 2005. [Google Scholar]
- Ramos, I.; Pérez, R.; Fdz-Polanco, M. The headspace of microaerobic reactors: Sulphide-oxidising population and the impact of cleaning on the efficiency of biogas desulphurisation. Bioresour. Technol. 2014, 158, 63–73. [Google Scholar] [CrossRef]
- Sublette, K.L. Aerobic oxidation of hydrogen sulfide by Thiobacillus denitrifican. Biotechnol. Bioeng. 1987, 29, 690–695. [Google Scholar] [CrossRef] [PubMed]
- De Zwart, J.; Sluis, J.; Kuenen, J.G. Competition for Dimethyl Sulfide and Hydrogen Sulfide by Methylophaga sulfidovorans and Thiobacillus thioparus T5 in Continuous Cultures. Appl. Environ. Microbiol. 1997, 63, 3318–3322. [Google Scholar] [CrossRef]
- Bosch, P.L.F.V.D.; Sorokin, D.Y.; Buisman, C.J.N.; Janssen, A.J.H. The Effect of pH on Thiosulfate Formation in a Biotechnological Process for the Removal of Hydrogen Sulfide from Gas Streams. Environ. Sci. Technol. 2008, 42, 2637–2642. [Google Scholar] [CrossRef] [PubMed]
- Fortuny, M.; Baeza, J.A.; Gamisans, X.; Casas, C.; Lafuente, J.; Deshusses, M.A.; Gabriel, D. Biological sweetening of energy gases mimics in biotrickling filters. Chemosphere 2008, 71, 10–17. [Google Scholar] [CrossRef]
- Lohwacharin, J.; Annachhatre, A.P. Biological sulfide oxidation in an airlift bioreactor. Bioresour. Technol. 2010, 101, 2114–2120. [Google Scholar] [CrossRef]
- Visser, J.M.; Stefess, G.C.; Robertson, L.A.; Kuenen, J.G. Thiobacillus sp. W5, the dominant autotroph oxidizing sulfide to sulfur in a reactor for aerobic treatment of sulfidic wastes. Antonie Leeuwenhoek 1997, 72, 127–134. [Google Scholar] [CrossRef]
- Sheoran, A.; Sheoran, V.; Choudhary, R. Bioremediation of acid-rock drainage by sulphate-reducing prokaryotes: A review. Miner. Eng. 2010, 23, 1073–1100. [Google Scholar] [CrossRef]
- Visser, J.M.; Robertson, L.A.; Van Verseveld, H.W.; Kuenen, J.G. Sulfur production by obligately chemolithoau-totrophic thiobacillus species. Appl. Environ. Microbiol. 1997, 63, 2300–2305. [Google Scholar] [CrossRef] [PubMed]
- Díaz, I.; Lopes, A.; Pérez, S.; Fdz-Polanco, M. Performance evaluation of oxygen, air and nitrate for the microaerobic removal of hydrogen sulphide in biogas from sludge digestion. Bioresour. Technol. 2010, 101, 7724–7730. [Google Scholar] [CrossRef]
- Rodríguez, E.; Lopes, A.; Fdz-Polanco, M.; Stams, A.J.M.; Garcia-Encina, P.A. Molecular analysis of the biomass of a fluidized bed reactor treating synthetic vinasse at anaerobic and micro-aerobic conditions. Appl. Microbiol. Biotechnol. 2011, 93, 2181–2191. [Google Scholar] [CrossRef]
- Hao, O.J.; Chen, J.M.; Huang, L.; Buglass, R.L. Sulfate-reducing bacteria. Crit. Rev. Environ. Sci. Technol. 1996, 26, 155–187. [Google Scholar] [CrossRef]
| KO Number | Gene | Encoding Protein | Abundance (%) |
|---|---|---|---|
| K02439 | glpE | thiosulfate sulfurtransferase | 0.31 |
| K03154 | thiS | sulfur carrier protein for thiamime biosynthesis | 4.01 |
| K03636 | moaD | molybdopterin synthase sulfur carrier subunit | 2.25 |
| K04091 | ssuD | alkanesulfonate monooxygenase | 1.50 |
| K10831 | tauB | taurine transport system ATP-binding protein | 0.30 |
| K15551 | tauA | taurine transport system substrate-binding protein | 0.30 |
| K15552 | tauC | taurine transport system permease protein | 0.37 |
| K15553 | ssuA | sulfonate transport system substrate-binding protein | 1.68 |
| K15554 | ssuC | sulfonate transport system permease protein | 1.14 |
| K15555 | ssuB | sulfonate transport system ATP-binding protein | 1.47 |
| K16937 | doxD | thiosulfate dehydrogenase (quinone) large subunit | 0.38 |
| K16950 | asrA | sulfite reductase subunit A | 0.4 |
| K16951 | asrB | sulfite reductase subunit B | 0.36 |
| K17218 | sqr | sulfide (quinone) oxidoreductase | 1.38 |
| K17222 | soxA | sulfur-oxidizing protein SoxA | 0.34 |
| K17223 | soxX | sulfur-oxidizing protein SoxX | 0.34 |
| K17224 | soxB | sulfur-oxidizing protein SoxB | 0.29 |
| K17225 | soxC | sulfane dehydrogenase subunit SoxC | 0.31 |
| K17226 | soxY | sulfur-oxidizing protein SoxY | 0.72 |
| K17227 | soxZ | sulfur-oxidizing protein SoxZ | 0.44 |
| Parameter | Measured Value |
|---|---|
| COD [mg/L] | 4703.3 ± 128.6 |
| VFA [mg/L] | 911.7 ± 12.6 |
| NH4-N [mg/L] | 180.0 ± 2.0 |
| T-N [mg/L] | 246.0 ± 3.6 |
| NO3−-N [mg/L] | 49.0 ± 1.7 |
| SO42− [mg/L] | 331.7 ± 1.5 |
| S[2−] [mg/L] | 75.3 ± 6.1 |
| pH | 7.2 ± 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Z.; Liu, Z.; Sklodowska, A.; Musialowski, M.; Bajda, T.; Yin, H.; Drewniak, L. Microbiological Sulfide Removal—From Microorganism Isolation to Treatment of Industrial Effluent. Microorganisms 2021, 9, 611. https://doi.org/10.3390/microorganisms9030611
Yang Z, Liu Z, Sklodowska A, Musialowski M, Bajda T, Yin H, Drewniak L. Microbiological Sulfide Removal—From Microorganism Isolation to Treatment of Industrial Effluent. Microorganisms. 2021; 9(3):611. https://doi.org/10.3390/microorganisms9030611
Chicago/Turabian StyleYang, Zhendong, Zhenghua Liu, Aleksandra Sklodowska, Marcin Musialowski, Tomasz Bajda, Huaqun Yin, and Lukasz Drewniak. 2021. "Microbiological Sulfide Removal—From Microorganism Isolation to Treatment of Industrial Effluent" Microorganisms 9, no. 3: 611. https://doi.org/10.3390/microorganisms9030611
APA StyleYang, Z., Liu, Z., Sklodowska, A., Musialowski, M., Bajda, T., Yin, H., & Drewniak, L. (2021). Microbiological Sulfide Removal—From Microorganism Isolation to Treatment of Industrial Effluent. Microorganisms, 9(3), 611. https://doi.org/10.3390/microorganisms9030611








