Microenvironmental Conditions Drive the Differential Cyanobacterial Community Composition of Biocrusts from the Sahara Desert
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Site Descriptions
2.2. Biocrust Sampling
2.3. Characterization of Physicochemical Soil Properties
2.4. Strain Isolation and Culture Conditions
2.5. Morphological Characterization
2.6. DNA Isolation and Amplification of the 16S rRNA Gene of the Isolated Strains
2.7. Phylogenetic Analyses of 16S rRNA Gene Sequences of Isolated Strains
2.8. Analyses of Cyanobacterial Community Composition by Amplicon Metagenomics
2.9. Cyanobacterial Survival Bioassays
3. Results
3.1. Physicochemical Characteristics of Soils
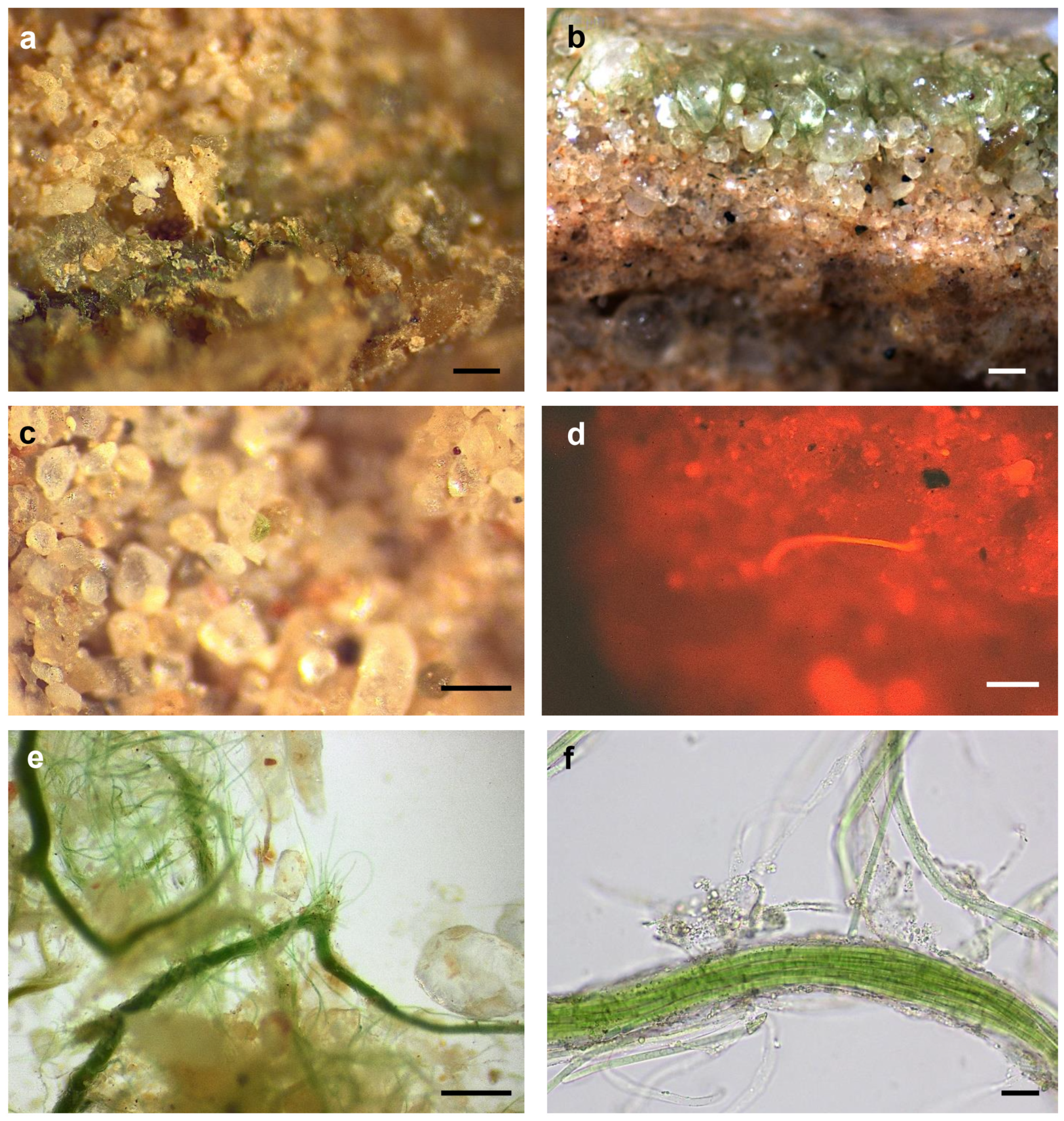
3.2. Biocrust Characteristics
3.3. Morphological and Molecular (16S rRNA Gene) Analysis of Cultures: Polyphasic Identification of Isolated Strains
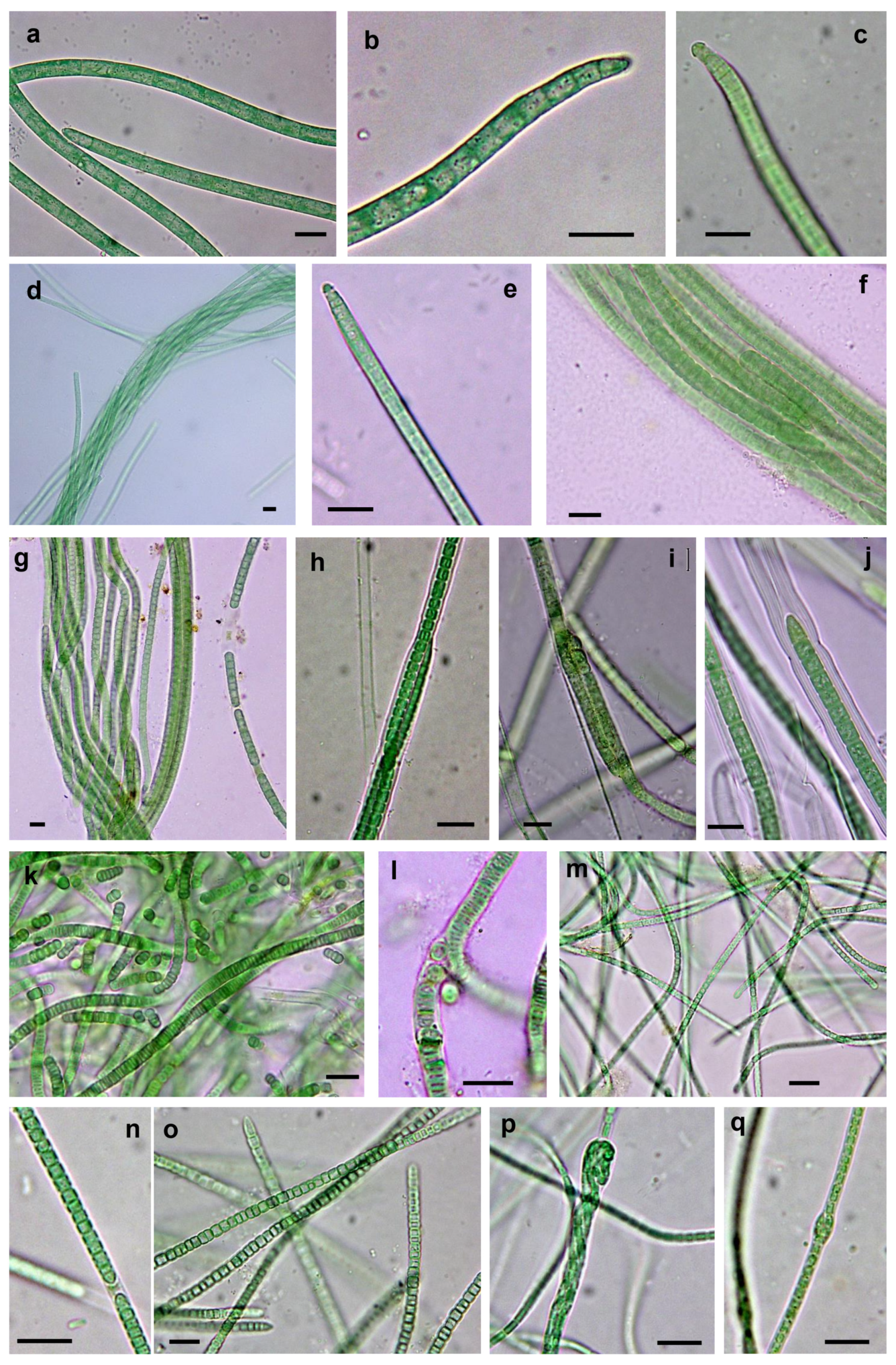
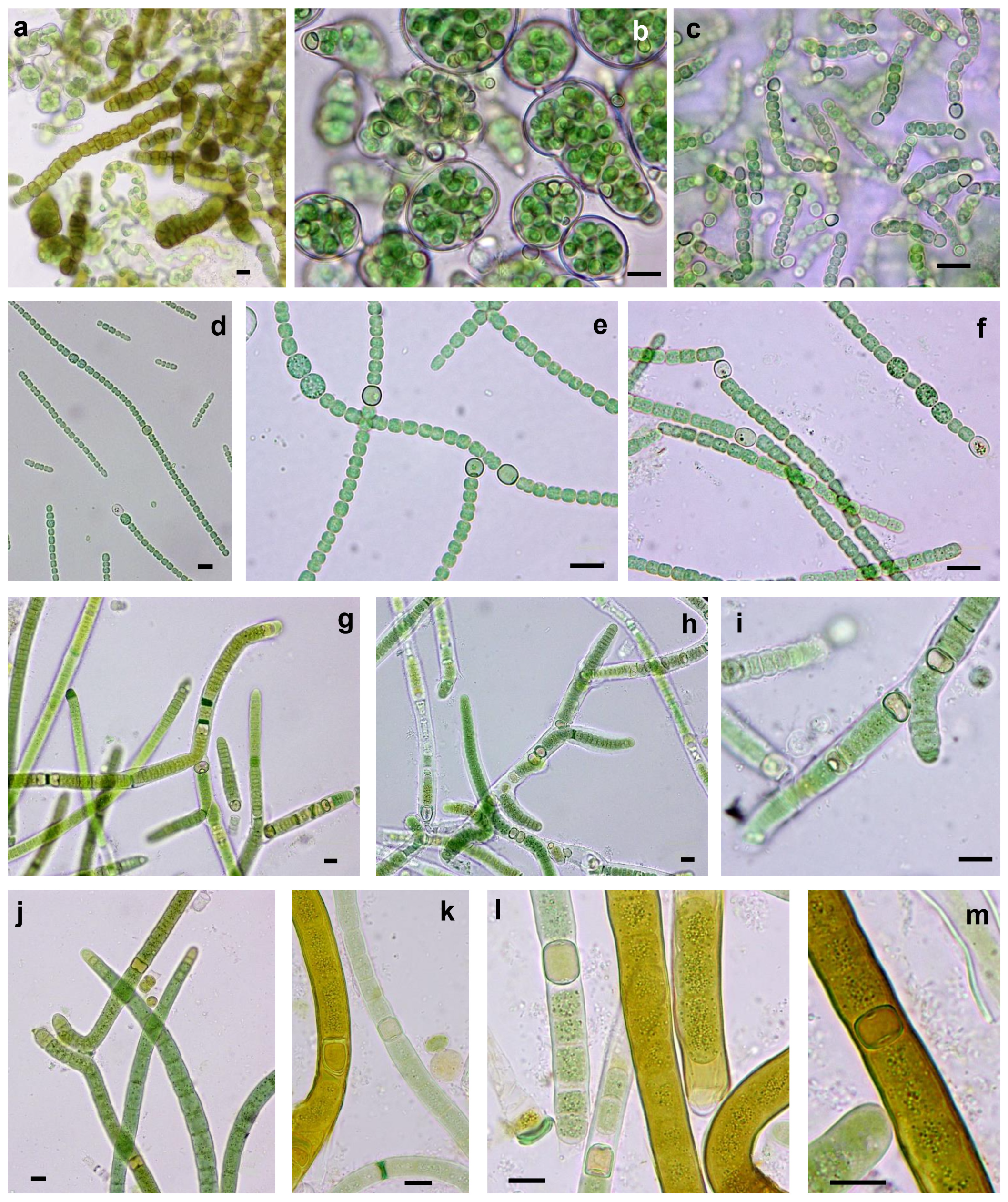
3.3.1. Non-Heterocystous Strains
3.3.2. Heterocystous Strains
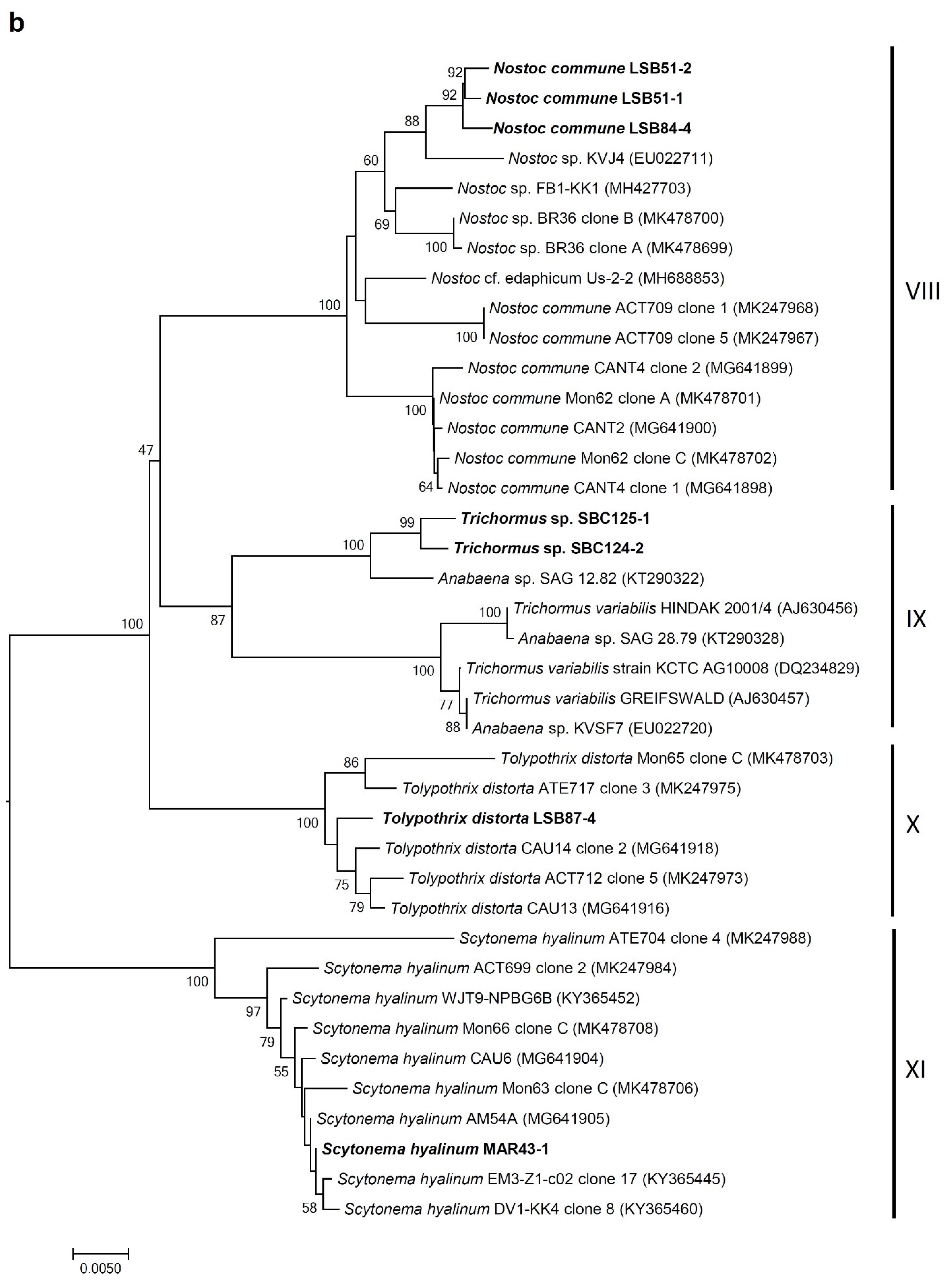
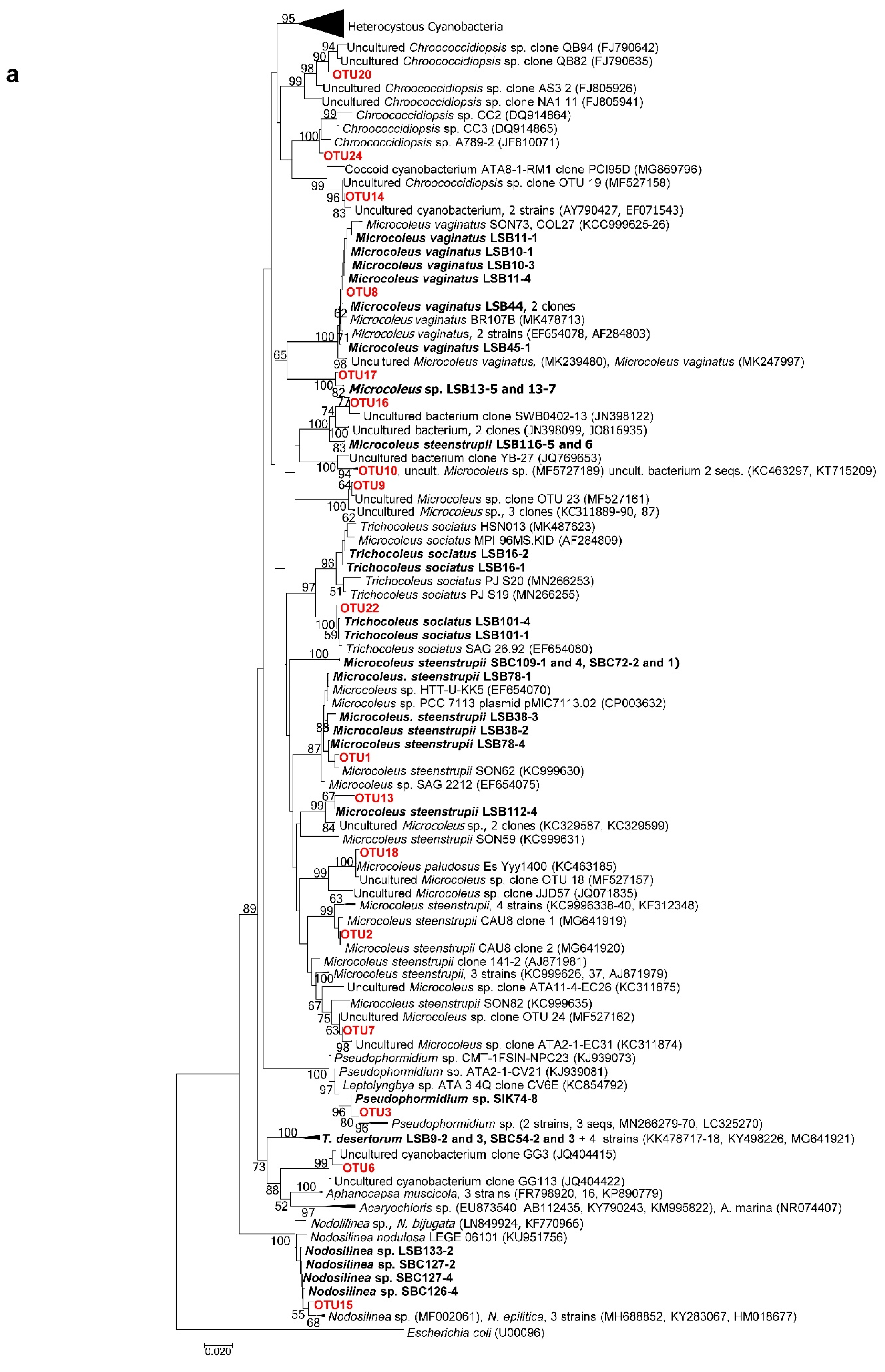
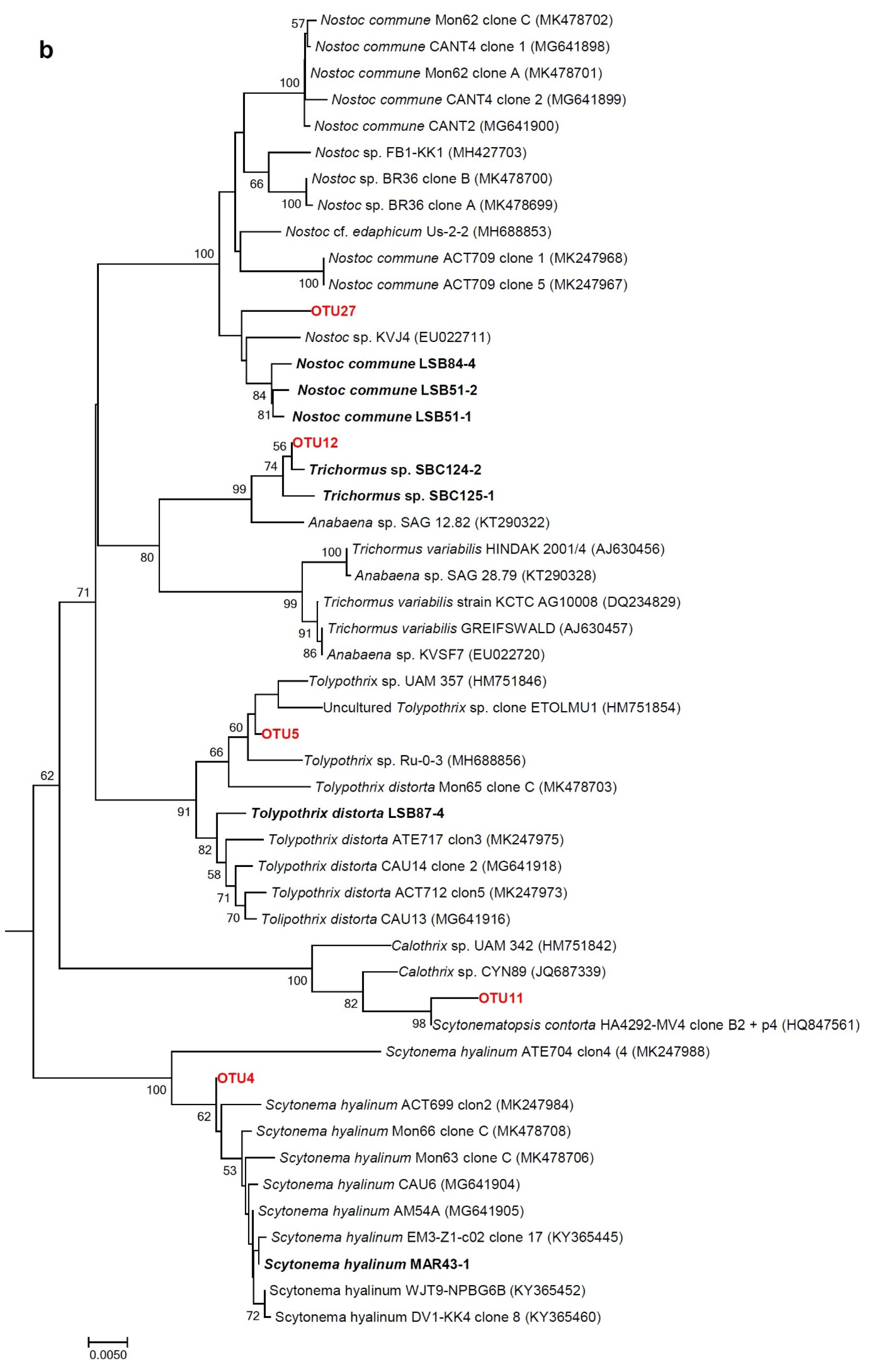
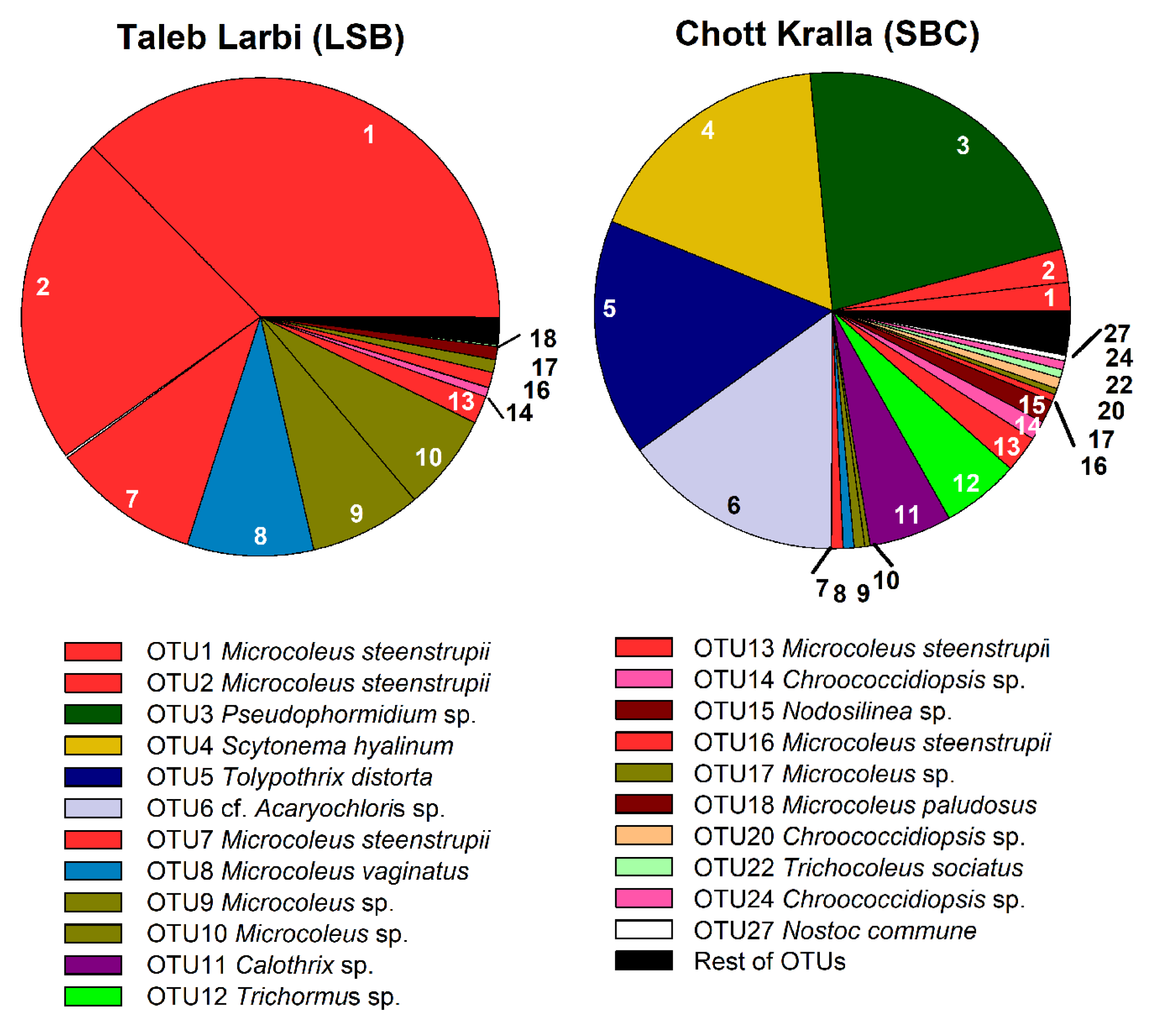
3.4. Molecular Analysis of Cyanobacterial Community Composition
3.5. Resistance of Strains to Extreme Heat and Desiccation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pointing, S.B.; Belnap, J. Microbial colonization and controls in dryland systems. Nat. Rev. Genet. 2012, 10, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Barrow, C. World Atlas of Desertification (United Nations Environment Programme); Middleton, N., Thomas, D.S.G., Eds.; Edward Arnold: London, UK, 1992. [Google Scholar]
- Rajeev, L.; Da Rocha, U.N.; Klitgord, N.; Luning, E.G.; Fortney, J.L.; Axen, S.D.; Shih, P.M.; Bouskill, N.J.; Bowen, B.P.; Kerfeld, C.A.; et al. Dynamic cyanobacterial response to hydration and dehydration in a desert biological soil crust. ISME J. 2013, 7, 2178–2191. [Google Scholar] [CrossRef]
- Lan, S.; Wu, L.; Zhang, D.; Hu, C. Effects of light and temperature on open cultivation of desert cyanobacterium Microcoleus vaginatus. Bioresour. Technol. 2015, 182, 144–150. [Google Scholar] [CrossRef]
- Castenholz, R.; Garcia-Pichel, F. Cyanobacterial Responses to UV Radiation. Ecology of Cyanobacteria II: Their Diversity in Space and Time; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Belnap, J.; Lange, O.L. Biological Soil Crusts: Structure, Function, and Management; Springer: Berlin, Germany, 2001; Volume 150, pp. 1–503. [Google Scholar]
- Hamdi-Aïssa, B.; Mehda, S.; Oustani, M.; Cammas, C.; Montoroi, J.P.; Hadj-Mahammed, M. Soil microstructures and microfabrics related to biological soil crust in the North African drylands (high plateau and Sahara desert), Algeria. In Proceedings of the 4th International Workshop on Biological Soil Crusts, Moreton Bay Research Station, The University of Queensland, Dunwich, QLD, Australia, 25 – 30 August 2019; p. 40. [Google Scholar]
- Rossi, F.; Li, H.; Liu, Y.; De Philippis, R. Cyanobacterial inoculation (cyanobacterisation): Perspectives for the development of a standardized multifunctional technology for soil fertilization and desertification reversal. Earth Sci. Rev. 2017, 171, 28–43. [Google Scholar] [CrossRef]
- Liu, Y.; Cockell, C.S.; Wang, G.; Hu, C.; Chen, L.; De Philippis, R. Control of Lunar and Martian Dust—Experimental Insights from Artificial and Natural Cyanobacterial and Algal Crusts in the Desert of Inner Mongolia, China. Astrobiology 2008, 8, 75–86. [Google Scholar] [CrossRef]
- Wang, W.; Liu, Y.; Li, D.; Hu, C.; Rao, B. Feasibility of cyanobacterial inoculation for biological soil crusts formation in desert area. Soil Biol. Biochem. 2009, 41, 926–929. [Google Scholar] [CrossRef]
- Reynolds, J.F.; Smith, D.M.S.; Lambin, E.F.; Turner, B.L.; Mortimore, M.J.; Batterbury, S.P.J.; Downing, T.E.; Dowlatabadi, H.; Fernández, R.J.; Herrick, J.E.; et al. Global Desertification: Building a Science for Dryland Development. Science 2007, 316, 847–851. [Google Scholar] [CrossRef]
- Safriel, U.N. Status of Desertification in the Mediterranean Region. In Building Knowledge for Geohazard Assessment and Management in the Caucasus and other Orogenic Regions; Springer: Berlin, Germany, 2009; pp. 33–73. [Google Scholar]
- Büdel, B.; Darienko, T.; Deutschewitz, K.; Dojani, S.; Friedl, T.; Mohr, K.I.; Salisch, M.; Reisser, W.; Weber, B. Southern African Biological Soil Crusts are Ubiquitous and Highly Diverse in Drylands, Being Restricted by Rainfall Frequency. Microb. Ecol. 2008, 57, 229–247. [Google Scholar] [CrossRef]
- Lalley, J.; Viles, H.; Henschel, J.; Lalley, V. Lichen-dominated soil crusts as arthropod habitat in warm deserts. J. Arid. Environ. 2006, 67, 579–593. [Google Scholar] [CrossRef]
- Zedda, L.; Gröngröft, A.; Schultz, M.; Petersen, A.; Mills, A.; Rambold, G. Distribution patterns of soil lichens across the principal biomes of southern Africa. J. Arid. Environ. 2011, 75, 215–220. [Google Scholar] [CrossRef]
- Guerra, C.A.; Heintz-Buschart, A.; Sikorski, J.; Chatzinotas, A.; Guerrero-Ramírez, N.; Cesarz, S.; Beaumelle, L.; Rillig, M.C.; Maestre, F.T.; Delgado-Baquerizo, M.; et al. Blind spots in global soil biodiversity and ecosystem function research. Nat. Commun. 2020, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Briere, P.R. Playa, playa lake, sabkha: Proposed definitions for old terms. J. Arid. Environ. 2000, 45, 1–7. [Google Scholar] [CrossRef]
- Hamdi-Aissa, B.; Vallès, V.; Aventurier, A.; Ribolzi, O. Soils and Brine Geochemistry and Mineralogy of Hyperarid Desert Playa, Ouargla Basin, Algerian Sahara. Arid. Land Res. Manag. 2004, 18, 103–126. [Google Scholar] [CrossRef]
- Khan, M.A.; Böer, B.; Kust, G.S.; Barth, H.-J. Sabkha Ecosystems: Volume II: West and Central Asia; Springer: Berlin, Germany, 2008; Volume 42. [Google Scholar]
- Patil, K.R.; Haider, P.; Pope, P.B.; Turnbaugh, P.J.; Morrison, M.; Scheffer, T.; McHardy, A.C. Taxonomic metagenome se-quence assignment with structured output models. Nat. Methods 2011, 8, 191–192. [Google Scholar] [CrossRef] [PubMed]
- Weber, B.; Belnap, J.; Büdel, B. Synthesis on Biological Soil Crust Research. In Mediterranean-Type Ecosystems; Springer: Berlin, Germany, 2016; Volume 226, pp. 527–534. [Google Scholar]
- Becerra-Absalón, I.; Muñoz-Martín, M.Á.; Montejano, G.; Mateo, P. Differences in the Cyanobacterial Community Composition of Biocrusts From the Drylands of Central Mexico. Are There Endemic Species? Front. Microbiol. 2019, 10, 937. [Google Scholar] [CrossRef]
- Muñoz-Martín, M.Á.; Becerra-Absalón, I.; Perona, E.; Fernández-Valbuena, L.; Garcia-Pichel, F.; Mateo, P. Cyanobacterial biocrust diversity in Mediterranean ecosystems along a latitudinal and climatic gradient. New Phytol. 2019, 221, 123–141. [Google Scholar] [CrossRef]
- Roncero-Ramos, B.; Muñoz-Martín, M.; Cantón, Y.; Chamizo, S.; Rodríguez-Caballero, E.; Mateo, P. Land degradation effects on composition of pioneering soil communities: An alternative successional sequence for dryland cyanobacterial biocrusts. Soil Biol. Biochem. 2020, 146, 107824. [Google Scholar] [CrossRef]
- Le Houerou, H.N. Restoration and Rehabilitation of Arid and Semiarid Mediterranean Ecosystems in North Africa and West Asia: A Review. Arid. Soil Res. Rehabil. 2000, 14, 3–14. [Google Scholar] [CrossRef]
- ONM, Office National de la Météorologie. Synthèse de Données Climatiques. In Les Données Météorologiques d’El Oued; Météo-Algérie: El Oued, Algeria, 2019. [Google Scholar]
- IUSS-Working-Group-WRB. World Reference Base for Soil Resources 2014, Update 2015: International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015; p. 192. [Google Scholar]
- Pétard, J. Les Méthodes D’analyse: Tome 1. Analyse de Sols; ORSTOM: Nouméa, France, 1993. [Google Scholar]
- Visconti, F.; De Paz, J.M.; Rubio, J.L. What information does the electrical conductivity of soil water extracts of 1 to 5 ratio (w/v) provide for soil salinity assessment of agricultural irrigated lands? Geoderma 2010, 154, 387–397. [Google Scholar] [CrossRef]
- PN-ISO-14235. Soil Quality. Determination of Organic Carbon by Sulfochromic Oxidation; ISO: Geneva, Switzerland, 2003. [Google Scholar]
- Pansu, M.; Gautheyrou, J. Handbook of Soil Analysis: Mineralogical, Organic and Inorganic Methods; Springer: Berlin, Germany, 2007. [Google Scholar]
- Porta, J.; Lopez-Acevedo, M.; Rodriguez, R. Tecnicas y Experimentos en Edafología; COIAC: Barcelona, Spain, 1986. [Google Scholar]
- Loeppert, R.; Suarez, G. Carbonates and Gypsum. In Methods of Soil Analysis Part 3, Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America: Madison, WI, USA; American Society of Agronomy: Madison, WI, USA, 1996; pp. 437–474. [Google Scholar]
- Rippka, R.; Stanier, R.Y.; Deruelles, J.; Herdman, M.; Waterbury, J.B. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Garcia-Pichel, F.; Loza, V.; Marusenko, Y.; Mateo, P.; Potrafka, R.M. Temperature Drives the Continental-Scale Distribution of Key Microbes in Topsoil Communities. Science 2013, 340, 1574–1577. [Google Scholar] [CrossRef]
- Perona, E.; Aboal, M.; Bonilla, I.; Mateo, P. Cyanobacterial diversity in a Spanish river determined by means of isolation of cultures. Morphological variability of isolates in relation to natural populations. Algol. Stud. 2003, 109, 475–486. [Google Scholar] [CrossRef]
- Komárek, J. 19/3: Cyanoprokaryota; Teil 3: Heterocystous Genera; Springer: Berlin, Germany, 2013. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota 1. Teil: Chroococcales. In Sußwasserflora Von mitteleuropa 19/1; Gustav Fischer: Jena, Germany, 1999; p. 548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprocaryota 2. Teil: Oscillatoriales. In Sußwasserflora von Mitteleuropa 19/2; Elsevier: Heidelberg, Germany, 2005; p. 759. [Google Scholar]
- Loza, V.; Perona, E.; Mateo, P. Molecular Fingerprinting of Cyanobacteria from River Biofilms as a Water Quality Monitoring Tool. Appl. Environ. Microbiol. 2013, 79, 1459–1472. [Google Scholar] [CrossRef] [PubMed]
- Wilmotte, A.; Van Der Auwera, G.; De Wachter, R. Structure of the 16 S ribosomal RNA of the thermophilic cyanobacterium chlorogloeopsis HTF (‘mastigocladus laminosus HTF’) strain PCC7518, and phylogenetic analysis. FEBS Lett. 1993, 317, 96–100. [Google Scholar] [CrossRef]
- Lepere, C.; Wilmotte, A.; Meyer, B. Molecular Diversity of Microcystis Strains (Cyanophyceae, Chroococcales) Based on 16S rDNA Sequences. Syst. Geogr. Plants 2000, 70, 275. [Google Scholar] [CrossRef]
- Mateo, P.; Perona, E.; Berrendero, E.; Leganés, F.; Martín, M.; Golubić, S. Life cycle as a stable trait in the evaluation of diver-sity of Nostoc from biofilms in rivers. FEMS Microbiol. Ecol. 2011, 76, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. In Nucleic Acids Symposium Series; Information Retrieval Ltd.: London, UK, 1999; Volume 41, pp. 95–98, c1979–c2000. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Tajima, F.; Nei, M. Estimation of evolutionary distance between nucleotide sequences. Mol. Biol. Evol. 1984, 1, 269–285. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence Limits on Phylogenies: An Approach Using the Bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl. Environ. Microbiol. 1997, 63, 3327–3332. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME Allows Analysis of High-Throughput Community Sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Smith, H.; Silva, A.G.; Belnap, J.; Garcia-Pichel, F. Differential responses of dinitrogen fixation, diazotrophic cyano-bacteria and ammonia oxidation reveal a potential warming-induced imbalance of the N-cycle in biological soil crusts. PLoS ONE 2016, 11, e0164932. [Google Scholar] [CrossRef] [PubMed]
- Baize, D. Guide des Analyses Courantes en Pédologie; INRA: Paris, France, 2000; 257p. [Google Scholar]
- Barzanji, A.F. Gypsiferous Soils of Iraq; Ghent University: Ghent, Belgium, 1973. [Google Scholar]
- Aubert, G. Méthodes D’analyses des Sols; Centre régional de documentation pédagogique de Marseille: Marseille, France, 1978. [Google Scholar]
- Garcia-Pichel, F.; Pringault, O. Cyanobacteria track water in desert soils. Nat. Cell Biol. 2001, 413, 380–381. [Google Scholar] [CrossRef] [PubMed]
- Machado-De-Lima, N.M.; Fernandes, V.M.C.; Roush, D.; Ayuso, S.V.; Rigonato, J.; Garcia-Pichel, F.; Branco, L.H.Z. The Compositionally Distinct Cyanobacterial Biocrusts From Brazilian Savanna and Their Environmental Drivers of Community Diversity. Front. Microbiol. 2019, 10, 2798. [Google Scholar] [CrossRef] [PubMed]
- Whitton, B.A. Ecology of Cyanobacteria II: Their Diversity in Space and Time; Springer: Berlin, Germany, 2012. [Google Scholar]
- Loza, V.; Perona, E.; Mateo, P. Specific responses to nitrogen and phosphorus enrichment in cyanobacteria: Factors influencing changes in species dominance along eutrophic gradients. Water Res. 2014, 48, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Jung, P.; Briegel-Williams, L.; Schermer, M.; Büdel, B. Strong in combination: Polyphasic approach enhances arguments for cold-assigned cyanobacterial endemism. Microbiology 2019, 8, e00729. [Google Scholar] [CrossRef]
- Sepehr, A.; Hassanzadeh, M.; Rodriguez-Caballero, E. The protective role of cyanobacteria on soil stability in two Aridisols in northeastern Iran. Geoderma Reg. 2019, 16, e00201. [Google Scholar] [CrossRef]
- Oren, A. Salts and Brines. In Ecology of Cyanobacteria II; Springer: Berlin, Germany, 2012; pp. 401–426. [Google Scholar]
- Chen, L.; Li, D.; Liu, Y. Salt tolerance of Microcoleus vaginatus Gom., a cyanobacterium isolated from desert algal crust, was enhanced by exogenous carbohydrates. J. Arid. Environ. 2003, 55, 645–656. [Google Scholar] [CrossRef]
- Lan, S.; Wu, L.; Zhang, D.; Hu, C.; Liu, Y. Effects of drought and salt stresses on man-made cyanobacterial crusts. Eur. J. Soil Biol. 2010, 46, 381–386. [Google Scholar] [CrossRef]
- Yoshimura, H.; Kotake, T.; Aohara, T.; Tsumuraya, Y.; Ikeuchi, M.; Ohmori, M. The role of extracellular polysaccharides produced by the terrestrial cyanobacterium Nostoc sp. strain HK-01 in NaCl tolerance. Environ. Boil. Fishes 2012, 24, 237–243. [Google Scholar] [CrossRef]
- Sommer, V.; Karsten, U.; Glaser, K. Halophilic Algal Communities in Biological Soil Crusts Isolated from Potash Tailings Pile Areas. Front. Ecol. Evol. 2020, 8, 8. [Google Scholar] [CrossRef]
- Sommer, V.; Mikhailyuk, T.; Glaser, K.; Karsten, U. Uncovering Unique Green Algae and Cyanobacteria Isolated from Biocrusts in Highly Saline Potash Tailing Pile Habitats, Using an Integrative Approach. Microorganisms 2020, 8, 1667. [Google Scholar] [CrossRef] [PubMed]
- Tsujimura, S.; Nakahara, H.; Kosaki, T.; Ishida, N.; Karbozova, E. Distribution of soil algae in salinized irrigation land in the arid region of Central Asia. Soil Sci. Plant Nutr. 1998, 44, 53–65. [Google Scholar] [CrossRef]
- Golubic, S. Halophily and Halotolerance in Cyanophytes. In Limits of Life; Springer: Berlin, Germany, 1980; pp. 69–83. [Google Scholar]
- Büdel, B.; Dulić, T.; Darienko, T.; Rybalka, N.; Friedl, T. Cyanobacteria and Algae of Biological Soil Crusts. In Mediterranean-Type Ecosystems; Springer: Berlin, Germany, 2016; pp. 55–80. [Google Scholar]
- Büdel, B. Cyanobacteria: Habitats and Species. In Plant Desiccation Tolerance; Lüttge, U., Beck, E., Bartels, D., Eds.; Springer: Berlin, Germany, 2011; pp. 11–21. [Google Scholar] [CrossRef]
- Coxson, D.S.; Kershaw, K.A. Rehydration response of nitrogenase activity and carbon fixation in terrestrial Nostoc commune from Stipa–Bouteloa grassland. Can. J. Bot. 1983, 61, 2658–2668. [Google Scholar] [CrossRef]
- Scherer, S.; Ernst, A.; Chen, T.-W. Rewetting of drought-resistant blue-green algae: Time course of water uptake and reappearance of respiration, photosynthesis, and nitrogen fixation. Oecologia 1984, 62, 418–423. [Google Scholar] [CrossRef]
- Shirkey, B.; McMaster, N.J.; Smith, S.C.; Wright, D.J.; Rodriguez, H.; Jaruga, P.; Birincioglu, M.; Helm, R.F.; Potts, M. Genomic DNA of Nostoc commune (Cyanobacteria) becomes covalently modified during long-term (decades) desiccation but is protected from oxidative damage and degradation. Nucleic Acids Res. 2003, 31, 2995–3005. [Google Scholar] [CrossRef]
- Oren, N.; Raanan, H.; Kedem, I.; Turjeman, A.; Bronstein, M.; Kaplan, A.; Murik, O. Desert cyanobacteria prepare in advance for dehydration and rewetting: The role of light and temperature sensing. Mol. Ecol. 2019, 28, 2305–2320. [Google Scholar] [CrossRef]
- Billi, D.; Potts, M. Life and death of dried prokaryotes. Res. Microbiol. 2002, 153, 7–12. [Google Scholar] [CrossRef]
- Feofilova, E.P. Deceleration of vital activity as a universal biochemical mechanism ensuring adaptation of microorganisms to stress factors: A review. Appl. Biochem. Microbiol. 2003, 39, 1–18. [Google Scholar] [CrossRef]
- Billi, D. Anhydrobiotic rock-inhabiting cyanobacteria: Potential for astrobiology and biotechnology. In Adaption of Microbial Life to Environmental Extremes: Novel Research Results and Application; Stan-Lotter, H., Fendrihan, S., Eds.; Springer: Vienna, Austria, 2012; pp. 119–132. [Google Scholar] [CrossRef]
- Mager, D.M.; Thomas, A. Extracellular polysaccharides from cyanobacterial soil crusts: A review of their role in dryland soil processes. J. Arid. Environ. 2011, 75, 91–97. [Google Scholar] [CrossRef]
- Potts, M. Mechanisms of desiccation tolerance in cyanobacteria. Eur. J. Phycol. 1999, 34, 319–328. [Google Scholar] [CrossRef]
- Garcia-Pichel, F.; Felde, V.J.M.N.L.; Drahorad, S.L.; Weber, B. Microstructure and Weathering Processes Within Biological Soil Crusts. In Mediterranean-Type Ecosystems; Springer: Berlin, Germany, 2016; Volume 226, pp. 237–255. [Google Scholar]
- Pentecost, A.; Whitton, B.A. Subaerial Cyanobacteria. In Ecology of Cyanobacteria II; Springer: Berlin, Germany, 2012; pp. 291–316. [Google Scholar]
- Gómez-Silva, B. Lithobiontic life: “Atacama rocks are well and alive. ” Antonie Leeuwenhoek 2018, 111, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Orellana, G.; Gómez-Silva, B.; Urrutia, M.; Galetović, A. UV-A Irradiation Increases Scytonemin Biosynthesis in Cyanobacteria Inhabiting Halites at Salar Grande, Atacama Desert. Microorganisms 2020, 8, 1690. [Google Scholar] [CrossRef] [PubMed]
- Fleming, E.D.; Castenholz, R.W. Effects of periodic desiccation on the synthesis of the UV-screening compound, scytonemin, in cyanobacteria. Environ. Microbiol. 2007, 9, 1448–1455. [Google Scholar] [CrossRef]
- Couradeau, E.; Karaoz, U.; Lim, H.C.; Da Rocha, U.N.; Northen, T.; Brodie, E.; Garcia-Pichel, F. Bacteria increase arid-land soil surface temperature through the production of sunscreens. Nat. Commun. 2016, 7, 10373. [Google Scholar] [CrossRef]
- Jung, P.; Schermer, M.; Briegel-Williams, L.; Baumann, K.; Leinweber, P.; Karsten, U.; Lehnert, L.; Achilles, S.; Bendix, J.; Büdel, B. Water availability shapes edaphic and lithic cyanobacterial communities in the Atacama Desert. J. Phycol. 2019, 55, 1306–1318. [Google Scholar] [CrossRef]
- Pietrasiak, N.; Osorio-Santos, K.; Shalygin, S.; Martin, M.P.; Johansen, J.R. When Is A Lineage A Species? A Case Study InMyxacorysgen. nov. (Synechococcales: Cyanobacteria) with the Description of Two New Species from The Americas. J. Phycol. 2019, 55, 976–996. [Google Scholar] [CrossRef]
- Samolov, E.; Baumann, K.; Büdel, B.; Jung, P.; Leinweber, P.; Mikhailyuk, T.; Karsten, U.; Glaser, K. Biodiversity of Algae and Cyanobacteria in Biological Soil Crusts Collected Along a Climatic Gradient in Chile Using an Integrative Approach. Microorganisms 2020, 8, 1047. [Google Scholar] [CrossRef]
- Gaysina, L.A.; Bohunická, M.; Hazuková, V.; Johansen, J.R. Biodiversity of Terrestrial Cyanobacteria of the South Ural Region. Cryptogamie, Algologie 2018, 39, 167–198. [Google Scholar] [CrossRef]
- Bahl, J.; Lau, M.C.Y.; Smith, G.J.D.; Vijaykrishna, D.; Cary, S.C.; Lacap, D.C.; Lee, C.K.; Papke, R.T.; Warren-Rhodes, K.A.; Wong, F.K.Y.; et al. Ancient origins determine global biogeography of hot and cold desert cyanobacteria. Nat. Commun. 2011, 2, 163. [Google Scholar] [CrossRef]
- Crits-Christoph, A.; Robinson, C.K.; Ema, B.; Eravel, J.; Ewierzchos, J.; Eascaso, C.; Eartieda, O.; Esouza-Egipsy, V.; Casero, M.C.; DiRuggiero, J. Phylogenetic and Functional Substrate Specificity for Endolithic Microbial Communities in Hyper-Arid Environments. Front. Microbiol. 2016, 7, 301. [Google Scholar] [CrossRef]
- De los Ríos, A.; Grube, M.; Sancho, L.G.; Ascaso, C. Ultrastructural and genetic characteristics of endolithic cyanobacterial biofilms colonizing Antarctic granite rocks. FEMS Microbiol. Ecol. 2007, 59, 386–395. [Google Scholar] [CrossRef]
- Khomutovska, N.; Jerzak, M.; Kostrzewska-Szlakowska, I.; Kwiatowski, J.; Suska-Malawska, M.; Syczewski, M.; Jasser, I. Life in Extreme Habitats: Diversity of Endolithic Microorganisms from Cold Desert Ecosystems of Eastern Pamir. Pol. J. Ecol. 2017, 65, 303–319. [Google Scholar] [CrossRef]
- Rego, A.; Raio, F.; Martins, T.P.; Ribeiro, H.; Sousa, A.G.G.; Séneca, J.; Baptista, M.S.; Lee, C.K.; Cary, S.C.; Ramos, V.; et al. Actinobacteria and Cyanobacteria Diversity in Terrestrial Antarctic Microenvironments Evaluated by Culture-Dependent and Independent Methods. Front. Microbiol. 2019, 10, 1018. [Google Scholar] [CrossRef]
- Billi, D. Desert cyanobacteria under space and planetary simulations: A tool for searching for life beyond Earth and supporting human space exploration. Int. J. Astrobiol. 2019, 18, 483–489. [Google Scholar] [CrossRef]
- Behrendt, L.; Larkum, A.W.; Norman, A.; Qvortrup, K.; Chen, M.; Ralph, P.; Sørensen, S.J.; Trampe, E.; Kühl, M. Endolithic chlorophyll d-containing phototrophs. ISME J. 2010, 5, 1072–1076. [Google Scholar] [CrossRef]
- Wierzchos, J.; Casero, M.C.; Artieda, O.; Ascaso, C. Endolithic microbial habitats as refuges for life in polyextreme environment of the Atacama Desert. Curr. Opin. Microbiol. 2018, 43, 124–131. [Google Scholar] [CrossRef]
- Giraldo-Silva, A.; Fernandes, V.M.C.; Bethany, J.; Garcia-Pichel, F. Niche Partitioning with Temperature among Heterocystous Cyanobacteria (Scytonema spp., Nostoc spp., and Tolypothrix spp.) from Biological Soil Crusts. Microorganisms 2020, 8, 396. [Google Scholar] [CrossRef]
- Kützing, F.T. Phycologia generalis: Oder Anatomie, Physiologie und Systemkunde der Tange; Brockhaus: Leipzig, Germany, 1843. [Google Scholar]
- Geitler, L. Cyanophyceae. Rabenhorst’s Kryptogamen-Flora von Deutschland, Österreich und der Schweiz; Koeltz Scientific Books: Königstein, Germany, 1932; Volume 14, pp. 1–1196. [Google Scholar]



| Soil Characteristic | Site | |
|---|---|---|
| Taleb Larbi (LSB) | Chott Kralla (SBC) | |
| pH | 7.44 | 7.51 |
| EC 1:5 (dS·m−1) | 1.76 | 3.28 |
| Salinity (%) | 1.99 | 3.71 |
| Total limestone (%) | 9.81 | 11.16 |
| Gypsum (%) | 7.82 | 13.76 |
| Organic matter (%) | 0.58 | 0.45 |
| Organic C (%) | 0.34 | 0.26 |
| Total N(%) | 0.042 | 0.019 |
| Taxon | Strain | Culture Collection N° | Sampling Site | Type of Cell | Breath (µm) | Length (µm) | |
|---|---|---|---|---|---|---|---|
| Microcoleus vaginatus | LSB45 * | UAM 886 | Taleb Larbi | Vegetative cells | 3.6 ± 0.3/2.9–4.1 | 4.4 ± 0.9/2.6–6.9 | |
| LSB44 * | UAM 887 | Taleb Larbi | Vegetative cells | 3.8 ± 0.3/3.2–4.5 | 3.9 ± 0.7/2.8–5.6 | ||
| LSB10 * | UAM 888 | Taleb Larbi | Vegetative cells | 5.1 ± 0.4/4.3–5.8 | 3.9 ± 0.5/2.6–5.9 | ||
| LSB11 * | UAM 889 | Taleb Larbi | Vegetative cells | 5.5 ± 0.5/4.7–6.8 | 4.4 ± 0.6/3.2–5.9 | ||
| Microcoleus sp. | LSB13 * | UAM 885 | Taleb Larbi | Vegetative cells | 4.8 ± 0.6/3.5–5.9 | 3 ± 0.6/2.1–4.6 | |
| Microcoleus steenstrpii | Cluster IVa | SBC109 | UAM 872 | Chott Kralla | Vegetative cells | 2.7 ± 0.2/2–3.7 | 3.9 ± 0.6/2.7–5.9 |
| SBC72 | UAM 873 | Chott Kralla | Vegetative cells | 3.6 ± 0.4/2.8–4.4 | 3.6 ± 0.4/2.7–4.6 | ||
| Cluster IVb | LSB112 * | UAM 874 | Taleb Larbi | Vegetative cells | 5.3 ± 0.4/4.3–6.4 | 8 ± 1.3/5.1–11.2 | |
| Claster IVc | LSB116 * | UAM 877 | Taleb Larbi | Vegetative cells | 6.1 ± 1.1/4.2–8.6 | 5.6 ± 0.9/3.5–8.1 | |
| Cluster IVd | LSB78 * | UAM 875 | Taleb Larbi | Vegetative cells | 6.8 ± 0.5/5.3–7.7 | 4.6 ± 0.6/3.4–6.7 | |
| LSB38 * | UAM 876 | Taleb Larbi | Vegetative cells | 6 ± 0.9/4.4–8.4 | 6.3 ± 1.2/4–9.4 | ||
| Trichocoleus sociatus | LSB16 | UAM 883 | Taleb Larbi | Vegetative cells | 5.2 ± 0.2/4.6–5.8 | 3.3 ± 0.4/2.6–4.3 | |
| LSB101 | UAM 884 | Taleb Larbi | Vegetative cells | 4.5 ± 0.8/3.2–7.1 | 4.2 ± 0.7/3.2–6.1 | ||
| Trichocoleus desertorum | SBC54 | UAM 890 | Chott Kralla | Vegetative cells | 2.8 ± 0.2/2.4–3.3 | 2.8 ± 0.4/2–3.6 | |
| LSB90 | UAM 891 | Taleb Larbi | Vegetative cells | 3.9 ± 0.3/3.2–4.5 | 3 ± 0.5/2.2–4.5 | ||
| Nodosilinea sp. | SBC127 | UAM 892 | Chott Kralla | Vegetative cells | 2 ± 0.2/1.6–2.6 | 2.5 ± 0.3/1.7–3.3 | |
| SBC126 | UAM 893 | Chott Kralla | Vegetative cells | 2.1 ± 0.3/1.4–2.8 | 3.6 ± 0.5/2.4–5 | ||
| LSB133 | UAM 894 | Taleb Larbi | Vegetative cells | 2 ± 0.3/1.4–2.9 | 3.1 ± 0.6/2–4.3 | ||
| Nostoc commune | LSB51 | UAM 881 | Taleb Larbi | Vegetative cells | 4.4 ± 0.8/3.2–6.2 | 4.3 ± 0.9/2.9–6.8 | |
| Heterocysts | 3.9 ± 0.4/3.1–5.1 | 4.5 ± 0.8/3.2–6.6 | |||||
| LSB84 | UAM 882 | Taleb Larbi | Vegetative cells | 3.7 ± 0.3/3–4.5 | 3.4 ± 0.5/2.3–5.1 | ||
| Heterocysts | 3.8 ± 0.3/3–4.7 | 4.5 ± 0.5/3–5.8 | |||||
| Tolypothrix distorta | LSB87 | UAM 878 | Taleb Larbi | Vegetative cells | 9.7 ± 1/7.4–12.3 | 4.3 ± 0.7/2.6–6.2 | |
| Intercalary Heterocysts | 9.7 ± 0.7/8.3–11.6 | 9.9 ± 1.3/7–13.5 | |||||
| Terminal Heterocysts | 9.5 ± 1.1/7.6–12 | 9.7 ± 0.7/8.5–10.8 | |||||
| Trichormus sp. | SBC124 | UAM 879 | Chott Kralla | Vegetative cells | 4.5 ± 0.4/3.7–5.8 | 4.3 ± 0.6/3–5.8 | |
| Heterocysts | 6.2 ± 0.2/5.9–7 | 6.3 ± 0.2/5.8–6.6 | |||||
| Akinete | 8.3 ± 0.7/7–11 | 9.3 ± 1.3/5.7–12.3 | |||||
| SBC125 | UAM 880 | Chott Kralla | Vegetative cells | 3.5 ± 0.3/2.8–4.1 | 5.3 ± 0.6/3.9–6.9 | ||
| Heterocysts | 5.7 ± 0.4/4.9–6.5 | 6.5 ± 0.8/4.8–8.9 | |||||
| Akinete | 6.9 ± 0.6/5.4–8 | 9.8 ± 1.4/6.2–12.6 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mehda, S.; Muñoz-Martín, M.Á.; Oustani, M.; Hamdi-Aïssa, B.; Perona, E.; Mateo, P. Microenvironmental Conditions Drive the Differential Cyanobacterial Community Composition of Biocrusts from the Sahara Desert. Microorganisms 2021, 9, 487. https://doi.org/10.3390/microorganisms9030487
Mehda S, Muñoz-Martín MÁ, Oustani M, Hamdi-Aïssa B, Perona E, Mateo P. Microenvironmental Conditions Drive the Differential Cyanobacterial Community Composition of Biocrusts from the Sahara Desert. Microorganisms. 2021; 9(3):487. https://doi.org/10.3390/microorganisms9030487
Chicago/Turabian StyleMehda, Smail, Maria Ángeles Muñoz-Martín, Mabrouka Oustani, Baelhadj Hamdi-Aïssa, Elvira Perona, and Pilar Mateo. 2021. "Microenvironmental Conditions Drive the Differential Cyanobacterial Community Composition of Biocrusts from the Sahara Desert" Microorganisms 9, no. 3: 487. https://doi.org/10.3390/microorganisms9030487
APA StyleMehda, S., Muñoz-Martín, M. Á., Oustani, M., Hamdi-Aïssa, B., Perona, E., & Mateo, P. (2021). Microenvironmental Conditions Drive the Differential Cyanobacterial Community Composition of Biocrusts from the Sahara Desert. Microorganisms, 9(3), 487. https://doi.org/10.3390/microorganisms9030487








