A Comprehensive Review of Detection Methods for SARS-CoV-2
Abstract
1. Introduction
2. Serological Approaches in the Detection of SARS-CoV2
3. Molecular Approaches for Detection of SARS-CoV-2 Infection
3.1. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
3.2. Isothermal Nucleic Acid Amplification
3.3. Nucleic Acid Hybridization Using Microarray
3.4. Amplicon-Based Metagenomic Sequencing
4. Point-of-Care Detection of COVID-19
5. The Role of Smartphone in the Detection and Surveillance of COVID-19
6. Wastewater-Based Epidemiology
7. Nanotechnology-Based Approaches in the Detection of COVID-19
8. Future Perspectives
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khalilov, R.; Hosainzadegan, M.; Eftekhari, A.; Nasibova, A.; Hasanzadeh, A.; Vahedi, P. Overview of the environmental distribution, resistance, mortality, and genetic diversity of new coronavirus (COVID-19). Adv. Biol. Earth Sci. 2020, 5, 7–12. [Google Scholar]
- Rong, X.; Yang, L.; Chu, H.; Fan, M. Effect of delay in diagnosis on transmission of COVID-19. Math. Biosci. Eng. 2020, 17, 2725–2740. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, S.; Fathi, N.; Memar, M.Y.; Hosseiniyan Khatibi, S.M.; Khalilov, R.; Negahdari, R.; Zununi Vahed, S.; Maleki Dizaj, S. Anti-microbial activity of curcumin nanoformulations: New trends and future perspectives. Phytother. Res. 2020, 34, 1926–1946. [Google Scholar] [CrossRef] [PubMed]
- Filipić, A.; Gutierrez-Aguirre, I.; Primc, G.; Mozetič, M.; Dobnik, D. Cold plasma, a new hope in the field of virus inactivation. Trends Biotechnol. 2020, 38, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Waggoner, J.J.; Stittleburg, V.; Pond, R.; Saklawi, Y.; Sahoo, M.K.; Babiker, A.; Hussaini, L.; Kraft, C.S.; Pinsky, B.A.; Anderson, E.J.; et al. Triplex real-time RT-PCR for severe acute respiratory syndrome coronavirus 2. Emerg. Infect. Dis. 2020, 26, 1633. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Odiwuor, N.; Xiong, J.; Sun, L.; Nyaruaba, R.O.; Wei, H.; Tanner, N.A. Rapid molecular detection of SARS-CoV-2 (COVID-19) virus RNA using colorimetric LAMP. 2020. MedRxiv 2020. [Google Scholar] [CrossRef]
- Khalilov, R.; Hosainzadegan, M.; Eftekhari, A. Necessity of different countries to deal with similar phenomena of COVID-19 coronavirus. Adv. Biol. Earth Sci. 2020, 5, 5–6. [Google Scholar]
- Li, Z.; Yi, Y.; Luo, X.; Xiong, N.; Liu, Y.; Li, S.; Sun, R.; Wang, Y.; Hu, B.; Chen, W.; et al. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J. Med. Virol. 2020, 92, 1518–1524. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, D.; Yang, P.; Poon, L.L.; Wang, Q. Viral load of SARS-CoV-2 in clinical samples. Lancet Infect. Dis. 2020, 20, 411–412. [Google Scholar] [CrossRef]
- Miller, S.; Chiu, C.Y.; Rodino, K.G.; Miller, M.B. Point-Counterpoint: Should We Be Performing Metagenomic Next-Generation Sequencing for Infectious Disease Diagnosis in the Clinical Laboratory? J. Clin. Clin. Microbiol. 2019, 58, 58. [Google Scholar] [CrossRef]
- Sidiq, Z.; Hanif, M.; Kumar Dwivedi, K.; Chopra, K. Benefits and limitations of serological assays in COVID-19 infection. Indian J. Tuberc. 2020, 67, S163–S166. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, M.; Shen, C.; Wang, F.; Yuan, J.; Li, J.; Zhang, M.; Wang, Z.; Xing, L.; Wei, J. Laboratory diagnosis and monitoring the viral shedding of 2019-nCoV infections. MedRxiv 2020, 1, 100061. [Google Scholar]
- Padoan, A.; Bonfante, F.; Zuin, S.; Cosma, C.; Basso, D.; Plebani, M. Clinical performances of an ELISA for SARS-CoV-2 antibody assay and correlation with neutralization activity. Clin. Chim. Actt. J. Clin. Chem. 2020, 510, 654–655. [Google Scholar] [CrossRef] [PubMed]
- Research Use Only 2019-Novel Coronavirus (2019-nCoV) Real-time RT-PCR Primers and Probes. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/rt-pcr-panel-primer-probes.html (accessed on 20 December 2020).
- Zhang, Z.L.; Hou, Y.L.; Li, D.T.; Li, F.Z. Diagnostic efficacy of anti-SARS-CoV-2 IgG/IgM test for COVID-19: A meta-analysis. J. Med Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.K.; Pan, Y.; Cheng, S.M.; Hui, K.P.; Krishnan, P.; Liu, Y.; Ng, D.Y.M.; Wan, C.K.C.; Yang, P.; Wang, Q. Molecular diagnosis of a novel coronavirus (2019-nCoV) causing an outbreak of pneumonia. Clin. Chem. 2020, 66, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Yan, F. Patients with RT-PCR-confirmed COVID-19 and normal chest CT. Radiology 2020, 295, E3. [Google Scholar] [CrossRef]
- Whiting, P.; Singatullina, N.; Rosser, J.J.B.E. Computed tomography of the chest: I. Basic principles. Anaesth. Crit. Care Pain. 2015, 15, 299–304. [Google Scholar] [CrossRef]
- Lee, E.Y.; Ng, M.-Y.; Khong, P.-L. COVID-19 pneumonia: What has CT taught us? Lancet Infect. Dis. 2020, 20, 384–385. [Google Scholar] [CrossRef]
- Bernheim, A.; Mei, X.; Huang, M.; Yang, Y.; Fayad, Z.A.; Zhang, N.; Diao, K.; Lin, B.; Zhu, X.; Li, K.; et al. Chest CT findings in coronavirus disease-19 (COVID-19): Relationship to duration of infection. Radiology 2020, 200463. [Google Scholar] [CrossRef]
- Pan, F.; Ye, T.; Sun, P.; Gui, S.; Liang, B.; Li, L.; Zheng, D.; Wang, J.; Hesketh, R.L.; Yang, L.; et al. Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. Radiology 2020, 200370. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Mitsudomi, T. Management of ground-glass opacities: Should all pulmonary lesions with ground-glass opacity be surgically resected? Transl. Lung Cancer Res. 2013, 2, 354. [Google Scholar] [PubMed]
- Fang, Y.; Zhang, H.; Xie, J.; Lin, M.; Ying, L.; Pang, P.; Ji, W. Sensitivity of chest CT for COVID-19: Comparison to RT-PCR. Radiology 2020, 296, 200432. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Zhong, Z.; Zhao, W.; Zheng, C.; Wang, F.; Liu, J. Chest CT for typical 2019-nCoV pneumonia: Relationship to negative RT-PCR testing. Radiology 2020, 296, 200343. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, T.; de la Guardia, M.; Baradaran, B. Lateral flow assays towards point-of-care cancer detection: A review of current progress and future trends. Trac Trends Anal. Chem. 2020, 125, 115842. [Google Scholar] [CrossRef]
- Bustin, S.A. AZ Quantitative PCR; International University Line La Jolla: San Diego, CA, USA, 2004. [Google Scholar]
- Craw, P.; Balachandran, W. Isothermal nucleic acid amplification technologies for point-of-care diagnostics: A critical review. Lab. Chip 2012, 12, 2469–2486. [Google Scholar] [CrossRef]
- Lamb, L.; Bartolone, S.; Ward, E.; Chancellor, M.J.D. Rapid Detection of Novel Coronavirus (COVID-19) by Reverse Transcription-Loop-Mediated Isothermal Amplification. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3539654 (accessed on 20 December 2020).
- Mizumoto, K.; Kagaya, K.; Zarebski, A.; Chowell, G.J.E. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance 2020, 25, 2000180. [Google Scholar] [CrossRef]
- Bendavid, E.; Mulaney, B.; Sood, N.; Shah, S.; Ling, E.; Bromley-Dulfano, R. COVID-19 Antibody Seroprevalence in Santa Clara County, California. MedRxiv 2020. [Google Scholar] [CrossRef]
- Gallo, G.; La Torre, M.; Pietroletti, R.; Bianco, F.; Altomare, D.; Pucciarelli, S.; Gagliardi, G.; Perinotti, R. Italian society of colorectal surgery recommendations for good clinical practice in colorectal surgery during the novel coronavirus pandemic. Tech. Coloproctol. 2020, 24, 501–505. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Furuchi, M.; Kamimoto, A.; Honda, K.; Matsumura, H.; Kobayashi, R. Saliva-based PCR tests for SARS-CoV-2 detection. J. Oral Sci. 2020, 62, 350–351. [Google Scholar] [CrossRef]
- Smithgall, M.C.; Dowlatshahi, M.; Spitalnik, S.L.; Hod, E.A.; Rai, A.J. Types of Assays for SARS-CoV-2 Testing: A Review. Lab. Med. 2020, 51, e59–e65. [Google Scholar] [CrossRef]
- Van Guilder, H.D.; Vrana, K.E.; Freeman, W.M. Twenty-five years of quantitative PCR for gene expression analysis. Biotechniques 2008, 44, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.L.; Medrano, J.F. Real-time PCR for mRNA quantitation. Biotechniques 2005, 39, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, E.R.; Navas, J.; Pacheco, L.G. The COVID-19 Diagnostic Technology Landscape: Efficient Data Sharing Drives Diagnostic Development. Front. Public Health 2020, 8, 309. [Google Scholar] [CrossRef] [PubMed]
- Carter, L.J.; Garner, L.V.; Smoot, J.W.; Li, Y.; Zhou, Q.; Saveson, C.J.; Sasso, J.M.; Gregg, A.C.; Soares, D.J.; Beskid, T.R.; et al. Assay techniques and test development for COVID-19 diagnosis. ACS Publ. 2020, 6, 591–605. [Google Scholar]
- Younes, N.; Al-Sadeq, D.W.; Al-Jighefee, H.; Younes, S.; Al-Jamal, O.; Daas, H.I.; Yassine, H.M.; Nasrallah, G.K. Challenges in Laboratory Diagnosis of the Novel Coronavirus SARS-CoV-2. Viruses 2020, 12, 582. [Google Scholar] [CrossRef]
- Russo, A.; Minichini, C.; Starace, M.; Astorri, R.; Calò, F.; Coppola, N. Current status of laboratory Diagnosis for COVID-19: A narrative review. Fectidrug Resist. 2020, 13, 2657. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, E63. [Google Scholar] [CrossRef]
- Hong, T.C.; Mai, Q.L.; Cuong, D.V.; Parida, M.; Minekawa, H.; Notomi, T.; Hasebe, F.; Morita, K. Development and evaluation of a novel loop-mediated isothermal amplification method for rapid detection of severe acute respiratory syndrome coronavirus. J. Clin. Microbiol. 2004, 42, 1956–1961. [Google Scholar]
- Vashist, S.K. In vitro diagnostic assays for COVID-19: Recent advances and Emerg. trends. Multidisciplinary Digital Publishing Institute. Diagnostics 2020, 10, 202. [Google Scholar] [CrossRef]
- Loeffelholz, M.J.; Tang, Y.-W. infections. Laboratory diagnosis of Emerg. human coronavirus infections–the state of the art. Emerg. Microbes Fections 2020, 9, 747–756. [Google Scholar] [CrossRef]
- Gootenberg, J.S.; Abudayyeh, O.O.; Lee, J.W.; Essletzbichler, P.; Dy, A.J.; Joung, J.; Verdine, V.; Donghia, N.; Daringer, N.M.; Freije, C.A.; et al. Nucleic acid detection with CRISPR-Cas13a/C2c2. Science 2017, 356, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.S.; Ma, E.; Harrington, L.B.; Da Costa, M.; Tian, X.; Palefsky, J.M.; Doudna, J.A. CRISPR-Cas12a target binding unleashes indiscriminate single-stranded DNase activity. Science 2018, 360, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-Y.; Cheng, Q.-X.; Wang, J.-M.; Li, X.-Y.; Zhang, Z.-L.; Gao, S.; Cao, R.-B.; Zhao, G.-P.; Wang, J. CRISPR-Cas12a-assisted nucleic acid detection. Cell Discov. 2018, 4, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.; Zeng, W.; Yang, M.; Chen, W.; Ren, L.; Ai, J.; Wu, J.; Liao, Y.; Gou, X.; Li, Y.; et al. Development and evaluation of a CRISPR-based diagnostic for 2019-novel coronavirus. MedRxiv 2020. [Google Scholar] [CrossRef]
- Chen, Q.; Li, J.; Deng, Z.; Xiong, W.; Wang, Q.; Hu, Y.Q. Comprehensive detection and identification of seven animal coronaviruses and human respiratory coronavirus 229E with a microarray hybridization assay. Tervirology 2010, 53, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Geng, P.; Wang, Q.; Cao, B.; Liu, B. Development of a single nucleotide polymorphism DNA microarray for the detection and genotyping of the SARS coronavirus. J. Microbiol. Biotechnol. 2014, 24, 1445–1454. [Google Scholar] [CrossRef]
- Hardick, J.; Metzgar, D.; Risen, L.; Myers, C.; Balansay, M.; Malcom, T.; Rothman, R.; Gaydos, C. Initial performance evaluation of a spotted array Mobile Analysis Platform (MAP) for the detection of influenza A/B, RSV, and MERS coronavirus. Diagn Microbiol. Infect Dis. 2018, 91, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, P.; Ranjbar, R.; Alavian, S.M. Nucleic acid-based approaches for detection of viral hepatitis. Jundishapur J. Microbiol. 2015, 8, e17449. [Google Scholar] [CrossRef]
- Moore, S.C.; Penrice-Randal, R.; Alruwaili, M.; Dong, X.; Pullan, S.T.; Carter, D.; Kevin, B.; Zhao, Q.; Sun, Y.; Hartley, C.; et al. Amplicon based MinION sequencing of SARS-CoV-2 and metagenomic characterisation of nasopharyngeal swabs from patients with COVID-19. MedRxiv 2020. [Google Scholar] [CrossRef]
- Stefanini, I.; Cavalieri, D. Metagenomic approaches to investigate the contribution of the vineyard environment to the quality of wine fermentation: Potentials and difficulties. Frontiers Microbiol. 2018, 9, 991. [Google Scholar] [CrossRef]
- Choi, J.R. Development of Point-of-Care Biosensors for COVID-19. Frontiers Chem. 2020, 8, 517. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Yan, M.; Li, H.; Liu, T.; Lin, C.; Huang, S.; Shen, C. Evaluation of Enzyme-Linked Immunoassay and Colloidal Gold-Immunochromatographic Assay Kit for Detection of Novel Coronavirus (SARS-Cov-2) Causing an Outbreak of Pneumonia (COVID-19). MedRxiv 2020. [Google Scholar] [CrossRef]
- Huang, P.; Wang, H.; Cao, Z.; Jin, H.; Chi, H.; Zhao, J.; Yu, B.; Yan, F.; Hu, X.; Wu, F.; et al. A Rapid and Specific Assay for the Detection of MERS-CoV. Front. Microbiol. 2018, 9, 1101. [Google Scholar] [CrossRef] [PubMed]
- Spengler, M.; Adler, M.; Niemeyer, C.M. Highly sensitive ligand-binding assays in pre-clinical and clinical applications: Immuno-PCR and other emerging techniques. Analyst 2015, 140, 6175–6194. [Google Scholar] [CrossRef]
- Foudeh, A.M.; Didar, T.F.; Veres, T.; Tabrizian, M. Microfluidic designs and techniques using lab-on-a-chip devices for pathogen detection for point-of-care diagnostics. Lab. Chip 2012, 12, 3249–3266. [Google Scholar] [CrossRef]
- Laksanasopin, T.; Guo, T.W.; Nayak, S.; Sridhara, A.A.; Xie, S.; Olowookere, O.O.; Cadinu, P.; Meng, F.; Chee, N.H.; Kim, J.; et al. A smartphone dongle for diagnosis of infectious diseases at the point of care. Sci. Transl. Med. 2015, 7, 273re1. [Google Scholar] [CrossRef]
- Afsahi, S.; Lerner, M.B.; Goldstein, J.M.; Lee, J.; Tang, X.; Bagarozzi, D.A., Jr.; Pan, D.; Locascio, L.; Walker, A.; Barron, F.; et al. Novel graphene-based biosensor for early detection of Zika virus infection. Biosens Bioelectron. 2018, 100, 85–88. [Google Scholar] [CrossRef]
- Parrilla, M.; Cánovas, R.; Jeerapan, I.; Andrade, F.J.; Wang, J. A textile-based stretchable multi-ion potentiometric sensor. Adv. Healthc. Mater. 2016, 5, 996–1001. [Google Scholar] [CrossRef]
- Hussein, H.A.; Hassan, R.Y.; Chino, M.; Febbraio, F. Point-of-Care Diagnostics of COVID-19: From Current Work to Future Perspectives. Sensors 2020, 20, 4289. [Google Scholar] [CrossRef]
- Luppa, P.B.; Müller, C.; Schlichtiger, A.; Schlebusch, H. Point-of-care testing (POCT): Current techniques and future perspectives. TrAC Trends Anal. Chem. 2011, 30, 887–898. [Google Scholar] [CrossRef]
- Gates, B. Responding to Covid-19—a once-in-a-century pandemic? Mass Med. Soc. 2020, 382, 1677–1679. [Google Scholar] [CrossRef] [PubMed]
- Smith RDJSs, medicine. Responding to global infectious disease outbreaks: Lessons from SARS on the role of risk perception, communication and management. Soc. Sci. Med. 2006, 63, 3113–3123. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.S.; Thomas, M.R.; Budd, J.; Mashamba-Thompson, T.P.; Herbst, K.; Pillay, D.; Peeling, R.W.; Johnson, A.M.; McKendry, R.A.; Stevens, M.M. Taking connected mobile-health diagnostics of infectious diseases to the field. Nature 2019, 566, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.; Blumenfeld, N.R.; Laksanasopin, T.; Sia, S. Point-of-care diagnostics: Recent developments in a connected age. Anal. Chem. 2017, 89, 102–123. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Dickens, B.L.; Chen, M.; Cook, A.R.; Clapham, H.E. Estimating number of global importations of COVID-19 from Wuhan, risk of transmission outside mainland China and COVID-19 introduction index between countries outside mainland China. MedRxiv 2020. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, H.; Li, F.; Zhang, B.; Liu, Q.; Li, X.; Luo, L. Transmission and epidemiological characteristics of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) infected Pneumonia (COVID-19): Preliminary evidence obtained in comparison with 2003-SARS. MedRxiv 2020. [Google Scholar] [CrossRef]
- Tuite, A.; Bogoch, I.; Sherbo, R.; Watts, A.; Fisman, D.; Khan, K. Estimation of COVID-2019 burden and potential for international dissemination of infection from Iran. MedRxiv 2020. [Google Scholar] [CrossRef]
- Danquah, L.O.; Hasham, N.; MacFarlane, M.; Conteh, F.E.; Momoh, F.; Tedesco, A.A.; Jambai, A.; Ross, D.A.; Weiss, H.A. Use of a mobile application for Ebola contact tracing and monitoring in northern Sierra Leone: A proof-of-concept study. BMC Infect Dis. 2019, 19, 810. [Google Scholar] [CrossRef]
- Brangel, P.; Sobarzo, A.; Parolo, C.; Miller, B.S.; Howes, P.D.; Gelkop, S.; Lutwama, J.J.; Dye, J.M.; McKendry, R.A.; Lobel, L.; et al. A Serological Point-of-Care Test for the Detection of IgG Antibodies against Ebola Virus in Human Survivors. ACS Nano 2018, 12, 63–73. [Google Scholar] [CrossRef]
- Iribarren, S.J.; Schnall, R.; Stone, P.W.; Carballo-Diéguez, A. Smartphone applications to support tuberculosis prevention and treatment: Review and evaluation. JMIR mHealth uHealth 2016, 4, e25. [Google Scholar] [CrossRef]
- Skowronski, D.M.; Petric, M.; Daly, P.; Parker, R.A.; Bryce, E.; Doyle, P.W.; Noble, M.A.; Roscoe, D.L.; Tomblin, J.; Yang, T.C.; et al. Coordinated response to SARS, Vancouver, Canada. Emerg Infect Dis. 2006, 12, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Skowronski, D.M.; Astell, C.; Brunham, R.C.; Low, D.E.; Petric, M.; Roper, R.L.; Talbot, P.J.; Tam, T.; Babiuk, L. Severe acute respiratory syndrome (SARS): A year in review. Annu. Rev. Med. 2005, 56, 357–381. [Google Scholar] [CrossRef] [PubMed]
- Blozik, E.; Grandchamp, C.; von Overbeck, J. Influenza surveillance using data from a telemedicine centre. Int. J. Public Health 2012, 57, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Interim Guidance for Implementing Home Care of People not Requiring Hospitalization for 2019 Novel Coronavirus (2019-nCoV). Updated 12 February 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html (accessed on 20 December 2020).
- Xiang, Y.T.; Yang, Y.; Li, W.; Zhang, L.; Zhang, Q.; Cheung, T.; Ng, C.H. Timely mental health care for the 2019 novel coronavirus outbreak is urgently needed. Lancet Psychiatry 2020, 7, 228–229. [Google Scholar] [CrossRef]
- Liu, S.; Yang, L.; Zhang, C.; Xiang, Y.T.; Liu, Z.; Hu, S.; Zhang, B. Online mental health services in China during the COVID-19 outbreak. Lancet Psychiatry 2020, 7, e17–e18. [Google Scholar] [CrossRef]
- Karimuribo, E.D.; Mutagahywa, E.; Sindato, C.; Mboera, L.; Mwabukusi, M.; Kariuki Njenga, M.; Teesdale, S.; Olsen, J.; Rweyemamu, M. A Smartphone App (AfyaData) for Innovative One Health Disease Surveillance from Community to National Levels in Africa: Intervention in Disease Surveillance. JMIR Public Health Surveill. 2017, 3, e94. [Google Scholar] [CrossRef]
- Alqahtani, A.S.; BinDhim, N.F.; Tashani, M.; Willaby, H.W.; Wiley, K.E.; Heywood, A.E.; Booy, R.; Rashid, H. Pilot use of a novel smartphone application to track traveller health behaviour and collect infectious disease data during a mass gathering: Hajj pilgrimage 2014. J. Epidemiol. Glob Health 2016, 6, 147–155. [Google Scholar] [CrossRef]
- Malekjahani, A.; Sindhwani, S.; Syed, A.M. Chan WCJAocr. Engineering Steps for Mobile Point-of-Care Diagnostic Devices. Acc. Chem. Res. 2019, 52, 2406–2414. [Google Scholar] [CrossRef]
- D’Ambrosio, M.V.; Bakalar, M.; Bennuru, S.; Reber, C.; Skandarajah, A.; Nilsson, L.; Switz, N.; Kamgno, J.; Pion, S.; Boussinesq, M.; et al. Point-of-care quantification of blood-borne filarial parasites with a mobile phone microscope. Sci. Transl. Med. 2015, 7, 286re4. [Google Scholar] [CrossRef]
- Kanazawa, T.; Nakagami, G.; Goto, T.; Noguchi, H.; Oe, M.; Miyagaki, T.; Hayashi, A.; Sasaki, S.; Sanada, H. Use of smartphone attached mobile thermography assessing subclinical inflammation: A pilot study. J. Wound Care 2016, 25, 177–182. [Google Scholar] [CrossRef]
- Mudanyali, O.; Dimitrov, S.; Sikora, U.; Padmanabhan, S.; Navruz, I.; Ozcan, A. Integrated rapid-diagnostic-test reader platform on a cellphone. Lab. Chip 2012, 12, 2678–2686. [Google Scholar] [CrossRef] [PubMed]
- Mao, K.; Zhang, H.; Yang, Z. Can a paper-based device trace COVID-19 sources with wastewater-based epidemiology? Environ. Sci. Technol. 2020, 54, 3733–3735. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Kasprzyk-Hordern, B.; Frost, C.G.; Estrela, P.; Thomas, K.V. Community sewage sensors for monitoring public health. Environ. Sci. Technol. 2015, 49, 5845–5846. [Google Scholar] [CrossRef] [PubMed]
- Holshue, M.L.; DeBolt, C.; Lindquist, S.; Lofy, K.H.; Wiesman, J.; Bruce, H.; Spitters, C.; Ericson, K.; Wilkerson, S.; Tural, A.; et al. Washington State 2019-nCoV Case Investigation Team. First Case of 2019 Novel Coronavirus in the United States. N. Engl. J. Med. 2020, 382, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Magro, L.; Escadafal, C.; Garneret, P.; Jacquelin, B.; Kwasiborski, A.; Manuguerra, J.C.; Monti, F.; Sakuntabhai, A.; Vanhomwegen, J.; Lafaye, P.; et al. Paper microfluidics for nucleic acid amplification testing (NAAT) of infectious diseases. Lab. Chip 2017, 17, 2347–2371. [Google Scholar] [CrossRef] [PubMed]
- Reboud, J.; Xu, G.; Garrett, A.; Adriko, M.; Yang, Z.; Tukahebwa, E.M.; Rowell, C.; Cooper, J.M. Paper-based microfluidics for DNA diagnostics of malaria in low resource underserved rural communities. Proc. Natl. Acad. Sci. USA 2019, 116, 4834–4842. [Google Scholar] [CrossRef]
- Wigginton, K.R.; Boehm, A.B. Environmental engineers and scientists have important roles to play in stemming outbreaks and pandemics caused by enveloped viruses. Environ. Sci. Technol. 2020, 54, 3736–3739. [Google Scholar] [CrossRef]
- Ghernaout, D.; Elboughdiri, N. Environmental Engineering for Stopping Viruses Pandemics. Open Access Libr. J. 2020, 7, 1–17. [Google Scholar]
- Ghernaout, D.; Elboughdiri, N. Electrochemical Technology for Wastewater Treatment: Dares and Trends. Open Access Libr. J. 2020, 7, 1–17. [Google Scholar] [CrossRef]
- Woolhouse, M.E.; Adair, K. The diversity of human RNA viruses. Future Virol. 2013, 8, 159–171. [Google Scholar] [CrossRef]
- Aquino de Carvalho, N.; Stachler, E.N.; Cimabue, N.; Bibby, K. Evaluation of Phi6 persistence and suitability as an enveloped virus surrogate. Environ. Sci. Technol. 2017, 51, 8692–8700. [Google Scholar] [CrossRef] [PubMed]
- Ghernaout, D.; Alghamdi, A.; Touahmia, M.; Aichouni, M.; Ait Messaoudene, N. Environmental, Engineering, C. Nanotechnology phenomena in the light of the solar energy. J. Energy Environ. Chem. Eng. 2018, 3, 1–8. [Google Scholar] [CrossRef]
- Larsen, D.A.; Wigginton, K.R. Tracking COVID-19 with wastewater. Nature Biotechnol. 2020, 38, 1151–1153. [Google Scholar] [CrossRef] [PubMed]
- Eftekhari, A.; Maleki Dizaj, S.; Sharifi, S.; Salatin, S.; Rahbar Saadat, Y.; Zununi Vahed, S.; Samiei, M.; Ardalan, M.; Rameshrad, M.; Ahmadian, E.; et al. The Use of Nanomaterials in Tissue Engineering for Cartilage Regeneration; Current Approaches and Future Perspectives. Int. J. Mol. Sci. 2020, 21, 536. [Google Scholar] [CrossRef] [PubMed]
- Whitworth, J. COVID-19: A fast evolving pandemic. Trans. R. Soc. Trop. Med. Hyg. 2020, 114, 241. [Google Scholar] [CrossRef] [PubMed]
- Moitra, P.; Alafeef, M.; Dighe, K.; Frieman, M.; Pan, D. Selective Naked-Eye Detection of SARS-CoV-2 Mediated by N Gene Targeted Antisense Oligonucleotide Capped Plasmonic Nanoparticles. ACS Nano 2020. [Google Scholar] [CrossRef]
- Mahmoudi, M. Emerging Biomolecular Testing to Assess Risk of Mortality from COVID-19 Infection. Mol. Pharm. 2020. [Google Scholar] [CrossRef]
- Zheng, T.; Pierre-Pierre, N.; Yan, X.; Huo, Q.; Almodovar, A.J.; Valerio, F.; Rivera-Ramirez, I.; Griffith, E.; Decker, D.D.; Chen, S.; et al. Gold nanoparticle-enabled blood test for early stage cancer detection and risk assessment. ACS Appl. Mater. Interfaces 2015, 7, 6819–6827. [Google Scholar] [CrossRef]
- Caracciolo, G.; Safavi-Sohi, R.; Malekzadeh, R.; Poustchi, H.; Vasighi, M.; Chiozzi, R.Z.; Capriotti, A.L.; Laganà, A.; Mohammad Hajipour, M.; Domenico, M.D.; et al. Disease-specific protein corona sensor arrays may have disease detection capacity. Nanoscale Horiz. 2019, 4, 1063–1076. [Google Scholar] [CrossRef]
- Hajipour, M.J.; Laurent, S.; Aghaie, A.; Rezaee, F.; Mahmoudi, M. Personalized protein coronas: A “key” factor at the nanobiointerface. Biomater. Sci. 2014, 2, 1210–1221. [Google Scholar] [CrossRef]
- Ashkarran, A.A.; Olfatbakhsh, T.; Ramezankhani, M.; Crist, R.C.; Berrettini, W.H.; Milani, A.S.; Pakpour, S.; Mahmoudi, M. Evolving Magnetically Levitated Plasma Proteins Detects Opioid Use Disorder as a Model Disease. Adv. Healthc. Mater. 2020, 9, e1901608. [Google Scholar] [CrossRef] [PubMed]
- Campos, E.V.R.; Pereira, A.E.S.; de Oliveira, J.L.; Carvalho, L.B.; Guilger-Casagrande, M.; de Lima, R.; Fraceto, L.F. How can nanotechnology help to combat COVID-19? Opportunities and urgent need. J. Nanobiotechnol. 2020, 18, 125. [Google Scholar] [CrossRef] [PubMed]
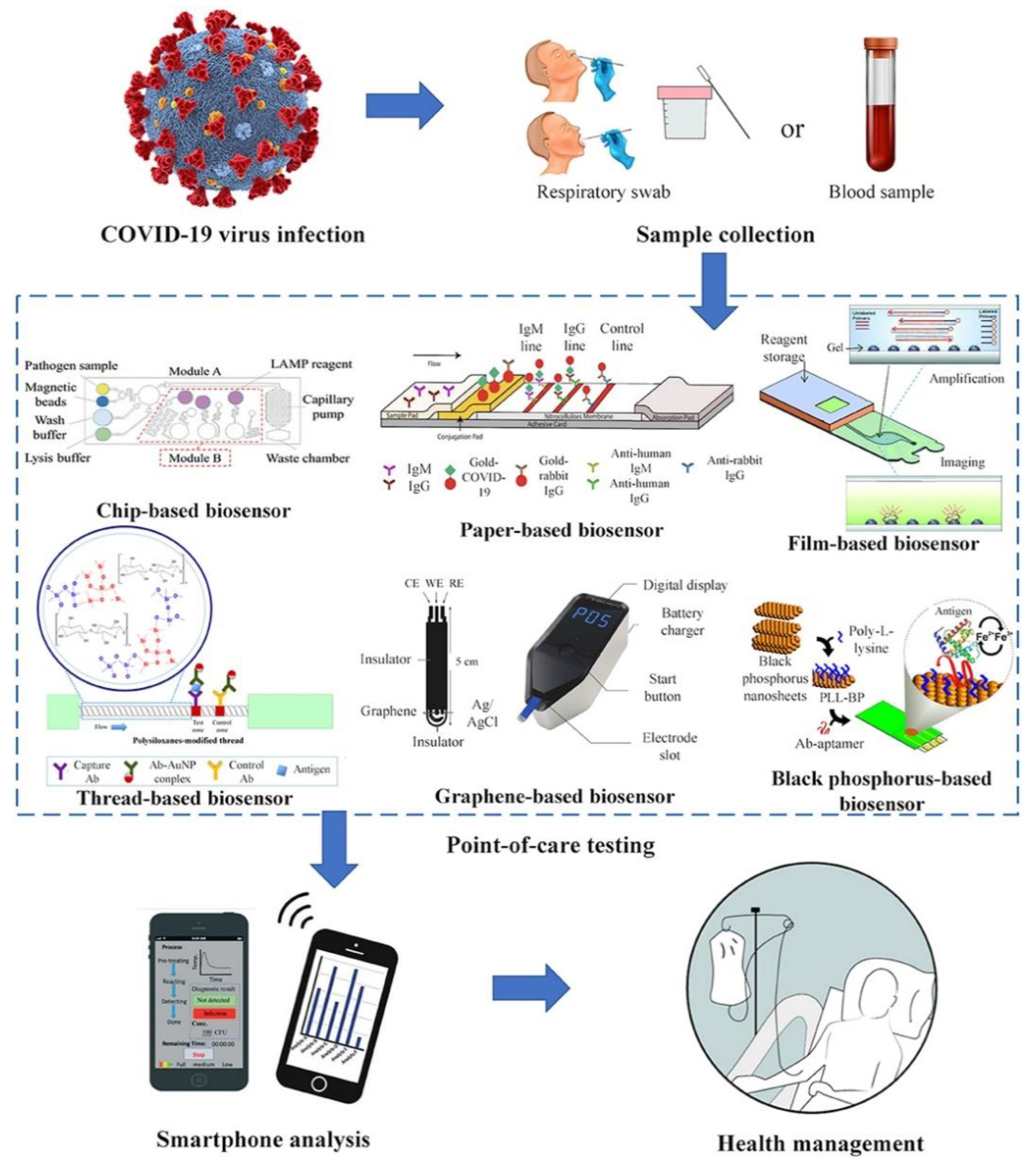
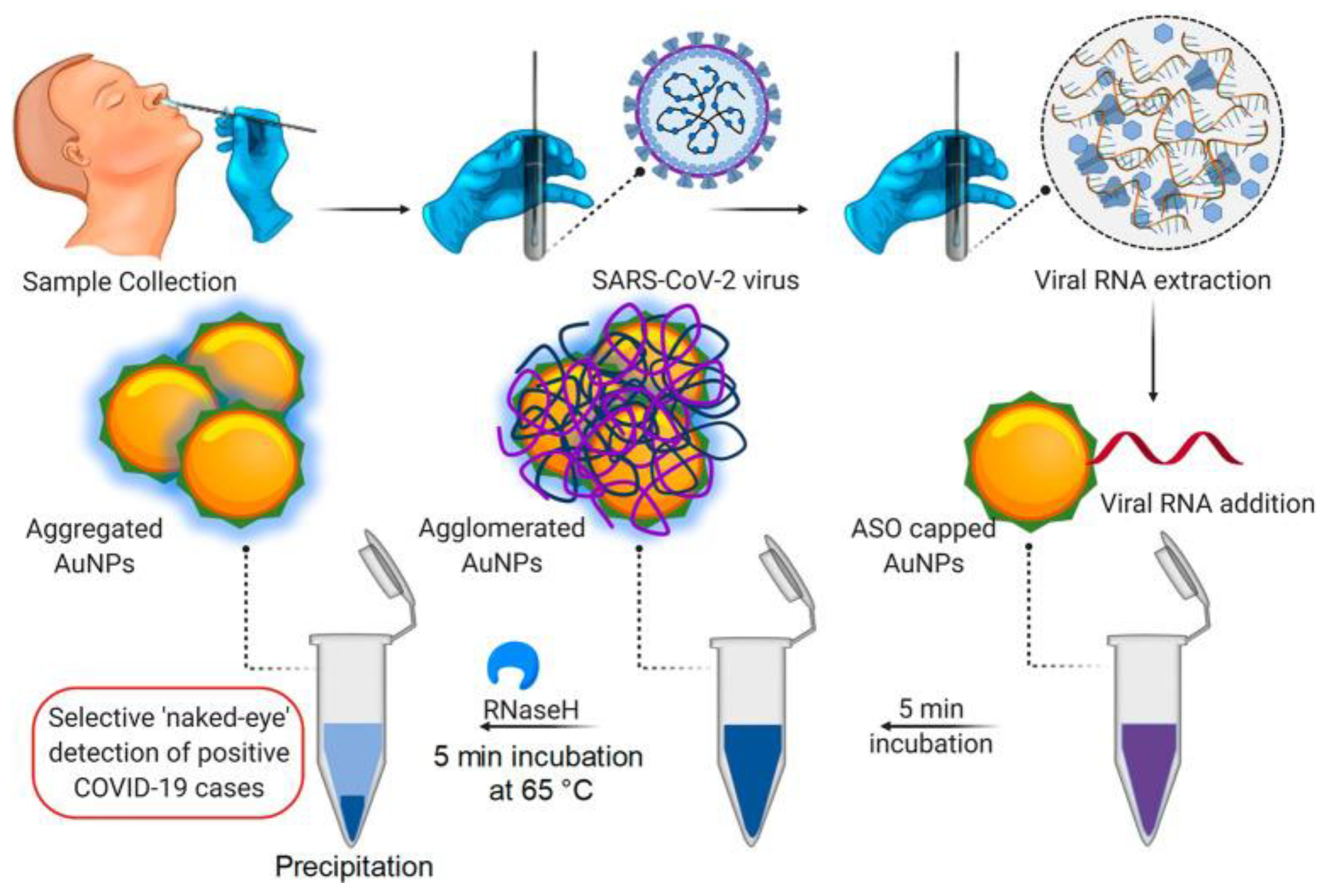
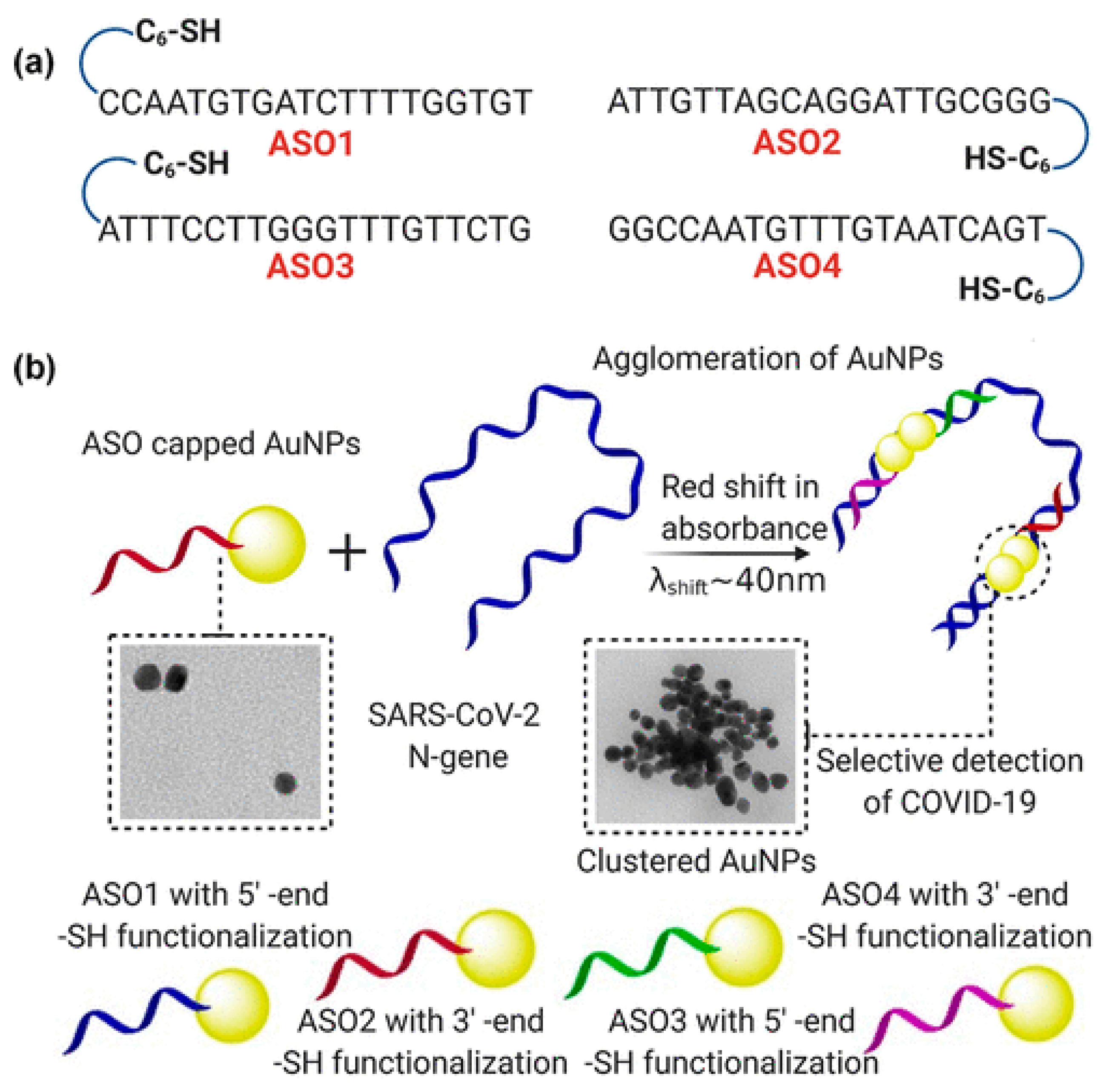
| Methodologies | Test | Principle | Sample | Advantage | Massive Used | Cost |
|---|---|---|---|---|---|---|
| Serological approaches | Enzyme-linked immunosorbent assay (ELISA) | Binding of antibody against COVID-19 with coated antigen in ELISA plates to forming complex and detect with labeled secondary antibody which produced color or fluorescence. | Blood serum or plasma |
| ✓ | Not very expensive |
| Chemiluminescence immunoassay (CLIA) | By chemical probes which could produce light emission via chemical reaction to label the antibody against COVID-19. | Blood serum or plasma |
| Expensive | ||
| COVID antigen assay | Detection of COVID-19 antigen with its specific antibody based on ELISA or CLIA | Blood serum or plasma |
| Variable costs | ||
| Molecular approaches | RT-PCR | Conversion of RNA of COVID-19 to cDNA via transcriptase enzyme followed by real-time PCR for amplification of cDNA | Upper respiratory specimens |
| ✓ | Expensive |
| RT-LAMP | Conversion of RNA of COVID-19 to cDNA via transcriptase enzyme and is performed at a temperature between 60 and 65 °C. | Upper respiratory specimens |
| Very cost effective | ||
| Nucleic Acid Hybridization Using Microarray | Conversion of RNA of COVID-19 to cDNA via transcriptase enzyme followed by adding it in wells containing fixed COVID-19-specific oligonucleotides then washing the hybridized virus cDNA for remains and emitting signal for positive samples. | Upper respiratory specimens |
| Expensive | ||
| Amplicon-Based Metagenomic Sequencing | Hypervariable regions of conserved genes or intergenic regions are amplified by PCR, evaluated by the next-generation sequencing (NGS), and the resulting sequences are compared against databases. | Upper respiratory specimens |
| Expensive | ||
| Point-of-Care detection of COVID-19 | Lateral flow assays | The antigens bind to the gold nanoparticle–antibody conjugates. The red and blue lines appear. The red lines are presented as gold nanoparticles exclusively, and blue lines as a clustered gold solution on account of plasmon band coupling. | blood or urine |
| Cheap | |
| Biosensors | Based on type of sensor, its principle is different. | Upper respiratory specimens or blood or urine |
| Expensive | ||
| Nanotechnology-based approaches | The application of nanoparticles in several methods which are mentioned above | Based on type of method, its principle is different. | Upper respiratory specimens or blood or urine |
| Expensive |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eftekhari, A.; Alipour, M.; Chodari, L.; Maleki Dizaj, S.; Ardalan, M.; Samiei, M.; Sharifi, S.; Zununi Vahed, S.; Huseynova, I.; Khalilov, R.; et al. A Comprehensive Review of Detection Methods for SARS-CoV-2. Microorganisms 2021, 9, 232. https://doi.org/10.3390/microorganisms9020232
Eftekhari A, Alipour M, Chodari L, Maleki Dizaj S, Ardalan M, Samiei M, Sharifi S, Zununi Vahed S, Huseynova I, Khalilov R, et al. A Comprehensive Review of Detection Methods for SARS-CoV-2. Microorganisms. 2021; 9(2):232. https://doi.org/10.3390/microorganisms9020232
Chicago/Turabian StyleEftekhari, Aziz, Mahdieh Alipour, Leila Chodari, Solmaz Maleki Dizaj, Mohammadreza Ardalan, Mohammad Samiei, Simin Sharifi, Sepideh Zununi Vahed, Irada Huseynova, Rovshan Khalilov, and et al. 2021. "A Comprehensive Review of Detection Methods for SARS-CoV-2" Microorganisms 9, no. 2: 232. https://doi.org/10.3390/microorganisms9020232
APA StyleEftekhari, A., Alipour, M., Chodari, L., Maleki Dizaj, S., Ardalan, M., Samiei, M., Sharifi, S., Zununi Vahed, S., Huseynova, I., Khalilov, R., Ahmadian, E., & Cucchiarini, M. (2021). A Comprehensive Review of Detection Methods for SARS-CoV-2. Microorganisms, 9(2), 232. https://doi.org/10.3390/microorganisms9020232






