1. Introduction
Antimicrobial resistance (AMR) is the ability of microorganisms to resist antimicrobial treatments, especially antibiotics. Infections due to AMR bacteria are a threat to modern health care and are responsible for an estimated 700,000 and 33,000 deaths/year globally and in Europe, respectively [
1]. Recently, the World Health Organization (WHO) published a list of pathogens for which urgent global action is needed [
2]. Extended spectrum β-lactamases (ESBL) producing and carbapenem-resistant
Enterobacteriaceae are among the priority one critical section of the WHO pathogen list. There has been a global rise in infections caused by multi-drug resistant clones of
Enterobacteriaceae, particularly
Klebsiella pneumoniae and
Escherichia coli [
3].
AMR can arise through various mechanisms, including mutations of chromosomal genes and the acquisition of antibiotic resistance genes (ARGs) from other strains in a process termed horizontal gene transfer (HGT). It is the sharing of genes through HGT that has largely contributed to the global dissemination of ARGs [
4]. The genomes of
E. coli and
K. pneumoniae are prone to a mutation in stress, depicting the genetic flexibility to upregulate their natural resistance and acquire foreign determinants through HGT due to mobile genetic elements. These elements, such as plasmids, transposons, integrons, and genomic islands, harbor ARGs [
5]. Several plasmids like IncF and IncI1 plasmids are known to carry resistance genes in
E. coli,
K. pneumoniae, and other
Enterobacteriaceae [
6]. Additionally, the ColE plasmids, which encode colicins, and have killing activity against different bacteria, are also important plasmids [
7]. Broad range resistance plasmids are known to be associated with pathogens; for example, a resistance plasmid from
Enterobacteriaceae can be transferred to a wide variety of Gram-negative organisms.
Whole-genome sequencing (WGS) is an effective method of tracking the onward transmission of bacteria or resistance plasmid transfer between bacteria. It has made it possible to determine and evaluate an organism’s whole DNA sequence at low costs in a short period of time. It allows for the identification of antimicrobial resistance and the early detection of outbreaks or their epidemiological investigation [
8]. Moreover, plasmid assembly and characterization following WGS is a difficult task. This happens because the plasmids tend to contain repeat sequences with sizes greater than sequences generated by sequencing platforms such as Illumina technology (San Diego, CA, USA) [
9]. Therefore, the need for in silico plasmid detection has emerged due to the difficulty of plasmid DNA purification if they are longer than 50 kbp [
10]. In addition, regarding the need for an efficient plasmid identification tool, ARG databases with comprehensive and accurate gene records are needed to assess AMR prevalence. Although several ARG databases exist, Comprehensive Antibiotic Resistance Database (CARD), and ResFinder are the most effective and have sustainable curation strategies [
11]. Recent studies have shown that the hybrid assemblies, which are a combination of Illumina and long-read sequencing (e.g., Oxford Nanopore Technology’s MinION) data, are better at identifying plasmids and ARGs [
12]. However, this requires advanced bioinformatic and machine learning methods for WGS data analysis [
13,
14,
15].
Globally, AMR is unevenly distributed. Recently, Klein et al. investigated the drug resistance index (DRI) for 41 countries [
16]. They have compared the reported data on antibiotics’ use and their resistance to the treatment of infections caused by microorganisms from the WHO priority list [
2]. Norway is among the countries with the lowest DRIs (third lowest), and has a DRI value around four-fold lower than that of the country with the highest DRI; India. However, there is an increasing trend in AMR cases in Norway. For example, the percentage of
E. coli with ESBL, causing septicemia, has increased ten-fold in the last ten years [
17]. There has also been a slightly increased prevalence of ESBLs for
E. coli (6.6% in 2017 and 6.5% in 2018) and
Klebsiella spp. (5.3% in 2017 and 6.6% in 2018) [
17].
This research has utilized different tools and databases to identify plasmids and predict plasmid-mediated ARGs in both E. coli and K. pneumoniae isolates. Our results indicate that plasmid identification and ARGs prediction are database/tool dependent. In this regard, a hybrid assembly can be considered an efficient way to identify plasmids and predict plasmid-mediated ARGs.
4. Discussion
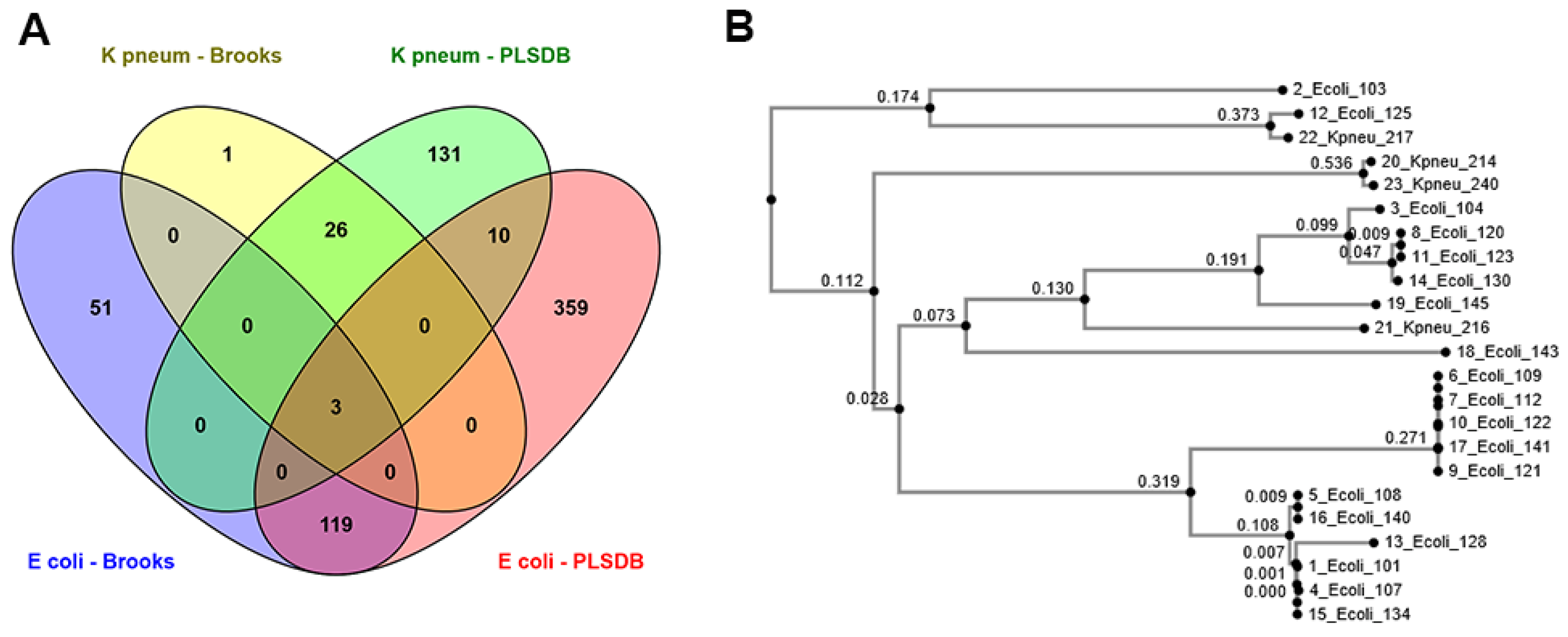
In the current research, the applicability of three different plasmid databases and two antibiotics resistance gene databases were assessed using E. coli and K. pneumoniae assemblies taken from Norwegian patients.
We identified a total number of 490 and 52 exclusive putative plasmids using PLSDB and Brooks databases, respectively. Observed differences might be explained by the content of databases, as the method used for developing the databases and the date of last revision (October 2018 for Brooks and November 2020 for PLSDB) as well as their file size (11,677 and 13,789 entries in Brooks and PLSDB, respectively) are different. Although a BLASTn search against Brooks and PLSDB databases resulted in a higher number of putative plasmids than PlasmidFinder, the method has its disadvantages. For instance, using the BLASTn search, we have detected multiple hits with similar lengths, alignment coverage, and percentage identity for the same assigned contig. Therefore, assigning the putative plasmids as
PTRUE was challenging. Similar challenges following BLAST+ have been previously described for the FindPlasmid package [
29]. Using PlasmidFinder, researchers can directly upload raw files from sequencing platforms. Therefore, de novo assembly is not required, and PlasmidFinder can perform de novo assembly automatically, though the assembly results are not presented by the tool. On the other hand, manual de novo assembly is required in advance to BLASTn search when using other databases such as Brooks and PLSDB. However, one of the disadvantages of using PlasmidFinder is that it currently only covers
Enterobacteriaceae and a few Gram-positive bacterial species.
It is clinically relevant to perform downstream analyses such as the prediction of plasmid associated ARGs following plasmid identification. In this study, ResFinder performed better than CARD to predict plasmid associated antibacterial resistance genes (AMR
Plasmid-PlasmidFinder, AMR
Plasmid-Brooks, and AMR
Plasmid-PLSDB) for both plasmid and hybrid assembled data. In a study comparing the performance of resistance gene databases, both CARD and ResFinder performed equally when submitting a single gene sequence, but CARD performed slightly better for assembled data [
30]. Although CARD only accepts FASTA assembly files up to 20 Mb, but in addition to acquired gene information, it contains chromosomal mutation data too. However, ResFinder takes raw files, and assembly is not required. Furthermore, in ResFinder, users can choose between acquired genes or chromosomal mutations. One of the ResFinder advantages is flagging the hit with the true circular term, which indicates whether the hit is plasmid associated or not. Therefore, current data suggest using PlasmidFinder and its associated ResFinder online tools as the first choice to predict plasmid associated ARGs.
In the current study, the AMR
Plasmid gene profiles differed between
E. coli strains carrying plasmids of the same type. Similar results have been reported for
Salmonella entrica isolates in Ghana [
31]. This further highlights the mobility of genetic elements between plasmids, resulting in acquiring or losing the ability for antimicrobial resistance. IncF plasmids are known carriers of a broad spectrum of antibiotic resistance genes in
E. coli [
32,
33,
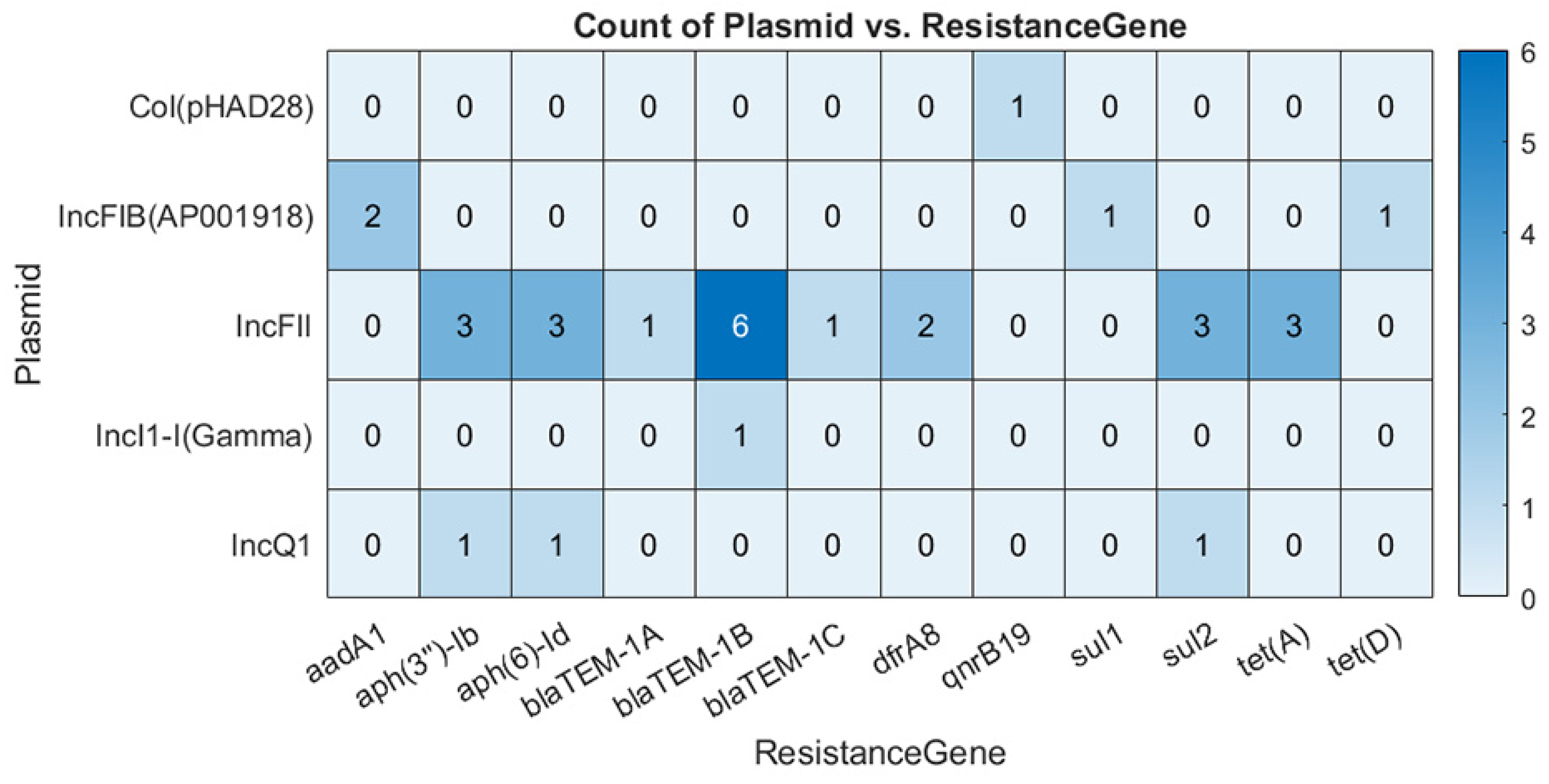
34]. In line with this, IncFII plasmids were strongly associated with various resistance genes in our study. These plasmids carried TEM-1B,
aph, sul, tetA, and
dfr genes conferring resistance to penicillins, aminoglycosides, sulfonamides, tetracyclines, and trimethoprim [
35]. IncFIB were the most prevalent plasmids in our dataset, and they exhibited a low association with antibiotic resistance genes. As such,
aad (aminoglycoside),
sul (sulfonamides), and
tet (tetracycline) were located on IncFIB plasmid contig in two cases, highlighting the low, albeit growing, antibiotic resistance in Norway [
16]. However, phylogenetic analysis of IncFIB plasmids revealed that plasmid sequences were shared between
E. coli and
K. pneumoniae, probably indicating its ability for inter-species transfer, which raises a concern over rising antibiotic resistance in Norway [
17]. Additionally, in this work, we documented the co-existence of blaCTX−M genes with other genes corresponding to resistance against sulfonamide, aminoglycoside, trimethoprim, and tetracycline. This agrees with previous reports indicating that plasmids harboring blaCTX−M genes frequently also carry other genes encoding resistance to other antimicrobials [
36,
37,
38].
The high sequence error rate in Oxford Nanopore Technologies and incongruity between short/fragmented reads from MiSeq Illumina platform and large repetitive regions in plasmids often results in the inaccurate prediction of plasmid-mediated ARGs. To overcome this issue, hybrid assembly has been suggested [
12,
39]. In the present research, we found that the prediction of ARG
Plasmids following hybrid assembly was more consistent across different databases. Having a less fragmented assembly where the circular plasmids are apparent makes the prediction of ARG
plasmid more accurate. The current conclusion regarding the applicability of hybrid assembly for plasmid-mediated ARGs detection previously has been made [
40]. Therefore, future work implanting hybrid assembly to identify ARGs in bacteria is worth investigating.