Microbial Nitrogen Cycling in Antarctic Soils
Abstract
1. Introduction
2. N Compound Speciation and Quantitation in Antarctic Soils
3. N-Cycling Taxa in Soils
3.1. Bacterial Nitrogen Cycling in Soils and Cryptic Niches
3.2. Archaea Are Drivers of Nitrification in Antarctic Soils and Niche Habitats
3.3. The Role of Fungi in Nitrogen Cycling
3.4. Viruses as Drivers of Nitrogen Cycling
4. N-Cycling Genes in Soils
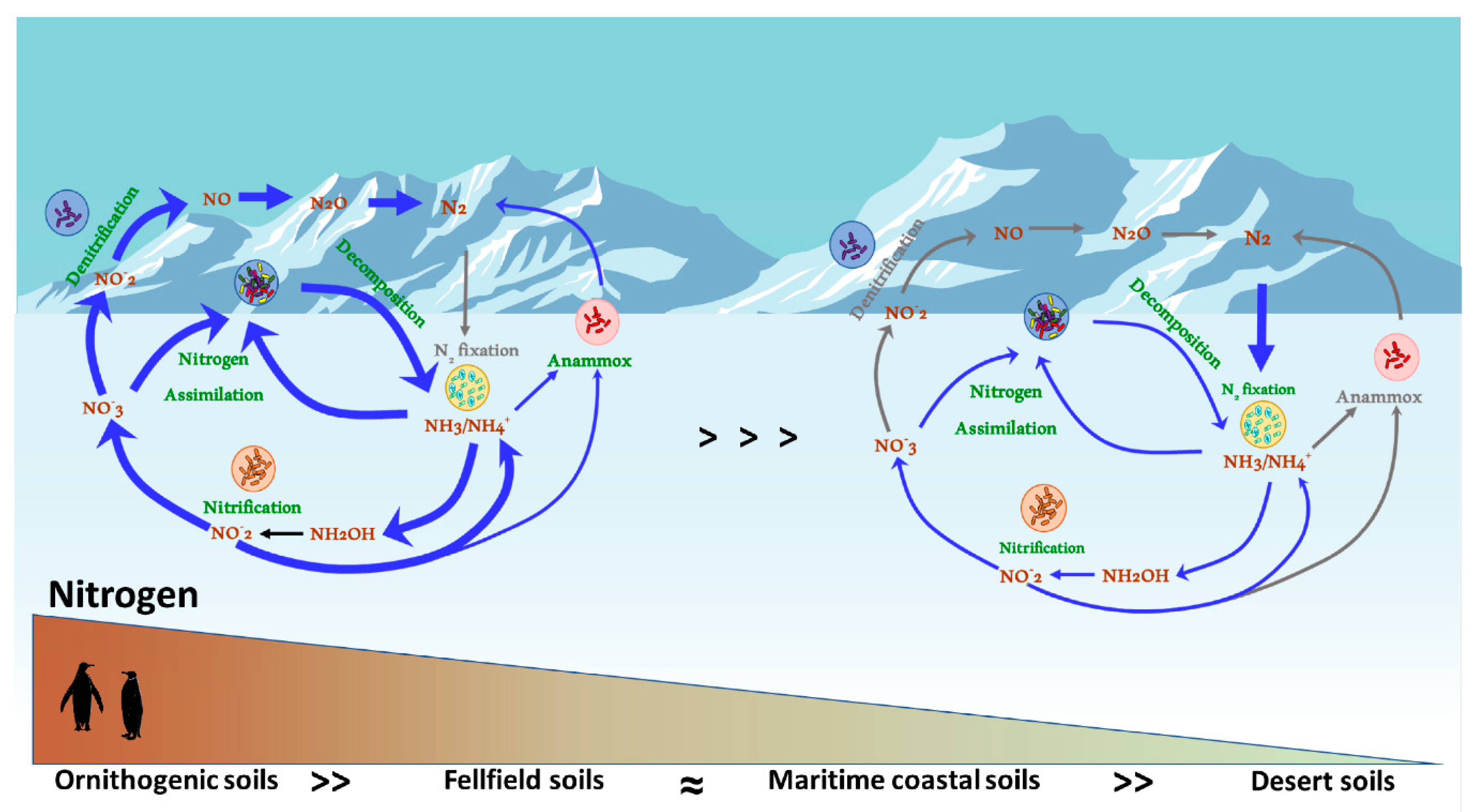
5. N-Cycling in Antarctic Soils: Rates, Processes and Ecosystem Services
6. N Supplementation Experiments
7. Gaps in Current Knowledge
- (1)
- In agreement with Guerra et al. [134], there is a need to establish a wider international effort for regular, long-term monitoring of different Antarctic environments in order to acquire data which can show temporal shifts in both soil nutrient content and in microbial community composition.
- (2)
- Given that much of the basic data on soil chemistry from terrestrial Antarctica are decades out of date, new datasets are desperately needed if we are to understand the effects of a changing Antarctic climate.
- (3)
- While there are now numerous published metagenomic studies available for various Antarctic regions and habitats, there is a distinct lack of transcriptomic, proteomic and metabolomic data, all of which are required for a deeper understanding of community function and nutrient cycling dynamics.
- (4)
- Investigations into Antarctic microbial community composition and function should be performed with an awareness of the uniqueness of the continent and its biota. A reliance on homology-based molecular screening methods may skew community structure and function data, given the possibility that Antarctic microorganisms may make use of pathways and genes which may be absent or considered inconsequential in other less “extreme” environments.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brooks, S.T.; Jabour, J.; van den Hoff, J.; Bergstrom, D.M. Our footprint on Antarctica competes with nature for rare ice-free land. Nat. Sustain. 2019, 2, 185–190. [Google Scholar] [CrossRef]
- Lee, C.K.; Laughlin, D.C.; Bottos, E.M.; Caruso, T.; Joy, K.; Barrett, J.E.; Hopkins, D.W.; Pointing, S.B.; McDonal, I.R.; Cowan, D.A.; et al. Biotic interactions are an unexpected yet critical control on the complexity of an abiotically driven polar ecosystem. Commun. Biol. 2019, 2, 62. [Google Scholar] [CrossRef]
- Bottos, E.M.; Laughlin, D.C.; Herbold, C.W.; Lee, C.K.; McDonald, I.R.; Cary, S.C. Abiotic factors influence patterns of bacterial diversity and community composition in the Dry Valleys of Antarctica. FEMS Microbiol. Ecol. 2020, 96, fiaa042. [Google Scholar] [CrossRef]
- Lee, C.K.; Barbier, B.A.; Bottos, E.M.; McDonald, I.R.; Cary, S.C. The inter-valley soil comparative survey: The ecology of dry valley edaphic microbial communities. ISME J. 2012, 6, 1046–1057. [Google Scholar] [CrossRef]
- Lepane, V.; Künnis-Beres, K.; Kaup, E.; Sharma, B. Dissolved organic matter, nutrients, and bacteria in Antarctic soil core from Schirmacher Oasis. J. Soils Sediments 2018, 18, 2715–2726. [Google Scholar] [CrossRef]
- Cowan, D.A. Microbiology of Antarctic Soils; Springer: Berlin/Heidelberg, Germany, 2014; 328p, ISBN 978-3-642-45212-3. [Google Scholar]
- Dennis, P.G.; Newsham, K.K.; Rushton, S.P.; O’Donnell, A.G.; Hopkins, D.W. Soil bacterial diversity is positively associated with air temperature in the maritime Antarctic. Sci. Rep. 2019, 9, 2686. [Google Scholar] [CrossRef]
- Walton, D.W.H.; Thomas, J. Cruise Report—Antarctic Circumnavigation Expedition (ACE) 20th December 2016—19th March 2017; OpenAIRE: Birmingham, UK, 2018; pp. 1–380. [Google Scholar] [CrossRef]
- Terauds, A.; Lee, J.R. Antarctic biogeography revisited: Updating the Antarctic Conservation Biogeographic Regions. Divers. Distribut. 2016, 22, 836–840. [Google Scholar] [CrossRef]
- Bokhorst, S.; Convey, P.; Aerts, R. Nitrogen inputs by marine vertebrates drive abundance and richness in Antarctic terrestrial ecosystems. Curr. Biol. 2019, 29, 1721–1729. [Google Scholar] [CrossRef]
- Winton, V.H.L.; Ming, A.; Caillon, N.; Hauge, L.; Jones, A.E.; Savarino, J.; Yang, X.; Frey, M.M. Deposition, recycling and archival of nitrate stable isotopes between the air-snow interface: Comparison beteen Dronning Maud Land and Dome C, Antarctica. Atmos. Chem. Phys. 2020, 20, 5861–5885. [Google Scholar] [CrossRef]
- Makhalanyane, T.P.; Valverde, A.; Velázquez, D.; Gunnigle, E.; Van Goethem, M.W.; Quesada, A.; Cowan, D.A. Ecology and biogeochemistry of cyanobacteria in soils, permafrost, aquatic and cryptic polar habitats. Biodivers. Conserv. 2015, 24, 819–840. [Google Scholar] [CrossRef]
- Selkirk, P.M. The nature and importance of the sub-Antarctic. In Papers and Proceedings of the Royal Society of Tasmania; The Royal Society of Tasmania: Hobart, Australia, 2007; Volume 1, pp. 1–6. [Google Scholar] [CrossRef]
- Convey, P. Antarctic ecosystems. In Encyclopedia of Biodiversity, 2nd ed.; Levin, S.A., Ed.; Elsevier: San Diego, CA, USA, 2013; pp. 179–188. [Google Scholar]
- Cameron, R.E. Cold desert characteristic and problems relevant to other arid land. In Arid Lands in Perspective; McGinnies, W.G., Goldman, B.G., Eds.; University of Arizona Press: Tucson, AZ, USA, 1969; pp. 167–205. [Google Scholar]
- Cameron, R.E.; Devaney, J.R. Antarctic soil algal crusts: A scanning electron and optical microscope study. Trans. Am. Microsc. Soc. 1970, 80, 264–273. [Google Scholar] [CrossRef]
- Cameron, R.E. Antarctic soil microbial and ecological investigations. In Research in the Antarctic; Quam, L.O., Porter, H.D., Eds.; Springer: Washington, DC, USA, 1971; pp. 137–189. [Google Scholar]
- Spider, T.W.; Cowling, J.C. Ornithogenic soils of the Cape Bird Adelie Penguin rookeries, Antarctica. Polar Biol. 1984, 2, 199–205. [Google Scholar] [CrossRef]
- Vishniac, H. The microbiology of Antarctic soils. In Antarctic Microbiology; Friedmann, E.I., Ed.; Wiley-Liss: New York, NY, USA, 1993; pp. 297–341. [Google Scholar]
- Cowan, D.A.; Ah Tow, L. Endangered Antarctic Environments. Annu. Rev. Microbiol. 2004, 58, 649–690. [Google Scholar] [CrossRef]
- Lachacz, A.; Kalisz, B.; Gielwanoska, I.; Olech, M.; Chwedorzewka, K.; Kellmann-Sopyla, W. Nutrient abundance and variability from soils in the coast of King George Island. Soil Sci. Plant Nutr. 2018, 2, 294–311. [Google Scholar] [CrossRef]
- Michalski, G.; Bockheim, J.G.; Kendall, C.; Thiemens, M. Isotopic composition of Antarctic Dry Valley nitrate: Implications for NOy sources and cycling in Antarctica. Geophys. Res. Lett. 2005, 32. [Google Scholar] [CrossRef]
- Barrett, J.E.; Virginia, R.A.; Wall, D.H.; Cary, S.C.; Adams, B.J.; Hacker, A.L.; Aislabie, J.M. Co-variation in soil biodiversity and biogeochemistry in northern and southern Victoria Land, Antarctica. Antarct. Sci. 2006, 18, 535–548. [Google Scholar] [CrossRef]
- Zhu, R.; Liu, Y.; Ma, E.; Sun, J.; Xu, H.; Sun, L. Nutrient compositions and potential greenhouse gas production in penguin guano, ornithogenic soils and seal colony soils in coastal Antarctica. Antarct. Sci. 2009, 21, 427–428. [Google Scholar] [CrossRef]
- Jung, J.; Yeom, J.; Kim, J.; Han, J.; Lim, H.S.; Park, H.; Hyun, S.; Park, W. Change in gene abundance in the nitrogen biogeochemical cycle with temperature and nitrogen addition in Antarctic soils. Res. Microbiol. 2011, 162, 1018–1026. [Google Scholar] [CrossRef]
- Cowan, D.A.; Makhalanyane, T.P.; Dennis, P.G.; Hopkins, D.W. Microbial ecology and biogeochemistry of continental antarctic soils. Front. Microbiol. 2014, 5, 154. [Google Scholar] [CrossRef]
- Parish, T.R. A numerical study of strong katabatic winds over Antarctica. Mon. Weather Rev. 1984, 112, 545–554. [Google Scholar] [CrossRef]
- Nylen, T.H.; Fountain, A.G. Climatolotogy of katabatic winds in the McMurdo Dry Valleys, Antarctica. J. Geophs. Res. 2004, 109, D03114. [Google Scholar] [CrossRef]
- Wood, S.; Rueckert, A.; Cowan, D.; Cary, S.C. Sources of edaphic cyanobacterial diversity in the Dry Valleys of Eastern Antarctica. ISME J. 2008, 2, 308–320. [Google Scholar] [CrossRef] [PubMed]
- Jungblut, A.D.; Hawes, I.; Mountfort, D.; Hitzfeld, B.; Dietrich, D.R.; Burns, B.P.; Neilan, B.A. Diversity within Cyanobacterial mat communities in variable salinity meltwater ponds of McMurdo Ice Shelf, Antarctica. Environ. Microbiol. 2005, 7, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.M.; Elster, J. Cyanobacteria in Antarctic lake Environments. In Algae and Cyanobacteria in Extreme Environments: Cellular Origin, Life in Extreme Habitats and Astrobiology; Seckbach, J., Ed.; Springer: Dordrecht, The Netherlands, 2007; Volume 11. [Google Scholar] [CrossRef]
- Hutchinson, G.E. Survey of existing knowledge of biogeochemistry. 3. of vertebrate excretion. Bull. Am. Mus. Natl. Hist. 1950, 96, 71–94. [Google Scholar]
- Tatur, A.; Myrcha, A. Ornithogenic soils on King George Island, South Shetland Island (Maritime Antarctic Zone). Pol. Polar Res. 1984, 5, 31–60. [Google Scholar]
- Chan, Y.; Van Nostrand, J.D.; Zhou, J.; Pointing, S.B.; Farrell, R.L. Functional ecology of an Antarctic Dry Valley. Proc. Natl. Acad. Sci. USA 2013, 110, 8990–8995. [Google Scholar] [CrossRef] [PubMed]
- Pointing, S.B.; Chan, Y.; Lacap, D.C.; Lau, M.C.Y.; Jurgens, J.A.; Farrell, R.L. Highly specialized microbial diversity in hyper-arid polar desert. Proc. Natl. Acad. Sci. USA 2009, 106, 19964–19969. [Google Scholar] [CrossRef] [PubMed]
- Cowan, D.A.; Sohm, J.A.; Makhalanyane, T.P.; Capone, D.G.; Green, T.G.A.; Cary, S.C.; Tuffin, I.M. Hypolithic communities: Important nitrogen sources in Antarctic desert soils. Environ. Microbiol. Rep. 2011, 3, 581–586. [Google Scholar] [CrossRef]
- Wei, S.T.S.; Lacap-Bugler, D.C.; Lau, M.C.Y.; Caruso, T.; Rao, S.; de los Rios, A.; Archer, S.K.; Chiu, J.M.Y.; Higgins, C.; Van Nostrand, J.D.; et al. Taxonomic and functional diversity of soil and hypolithic microbial communities in Miers Valley, McMurdo Dry Valleys, Antarctica. Front. Microbiol. 2016, 7, 1642. [Google Scholar] [CrossRef]
- Cary, S.C.; McDonald, I.R.; Barrett, J.E.; Cowan, D.A. On the rocks: The microbiology of Antarctic Dry Valley soils. Nat. Rev. Microbiol. 2010, 8, 129–138. [Google Scholar] [CrossRef]
- Komárek, J.; Genuário, D.B.; Fiore, M.F.; Elster, J. Heterocytous cyanobacteria of the Ulu Peninsula, James Ross Island, Antarctica. Polar Biol. 2015, 38, 475–492. [Google Scholar] [CrossRef]
- Van Goethem, M.W.; Cowan, D.A. Role of Cyanobacteria in the Ecology of Polar Environments. In The Ecological Role of Micro-Organisms in the Antarctic Environment; Springer Polar Sciences: Cham, Switzerland, 2019; pp. 3–23. ISBN 9783030027865. [Google Scholar]
- Hayashi, K.; Tanabe, Y.; Fujitake, N.; Kida, M.; Wang, Y.; Hayatsu, M.; Kudoh, S. Ammonia oxidation potentials and ammonia oxidizers of lichen–moss vegetated soils at two ice-free areas in east antarctica. Microbes Environ. 2020, 35, 2–6. [Google Scholar] [CrossRef]
- Magalhães, C.; Machado, A.; Frank-Fahle, B.; Lee, C.K.; Cary, C.S. The ecological dichotomy of ammonia-oxidizing archaea and bacteria in the hyper-arid soils of the Antarctic Dry Valleys. Front. Microbiol. 2014, 5, 515. [Google Scholar] [CrossRef]
- Fernández-Valiente, E.; Quesada, A.; Howard-Williams, C.; Hawes, I. N2-fixation in cyanobacterial mats from ponds on the McMurdo Ice Shelf, Antarctica. Microb. Ecol. 2001, 42, 338–349. [Google Scholar] [CrossRef]
- de Scally, S.Z.; Makhalanyane, T.P.; Frossard, A.; Hogg, I.D.; Cowan, D.A. Antarctic microbial communities are functionally redundant, adapted and resistant to short term temperature perturbations. Soil Biol. Biochem. 2016, 103, 160–170. [Google Scholar] [CrossRef]
- Papale, M.; Conte, A.; Mikkonen, A.; Michaud, L.; La Ferla, R.; Azzaro, M.; Caruso, G.; Paranhos, R.; Cabral Anderson, S.; Maimone, G.; et al. Prokaryotic assemblages within permafrost active layer at Edmonson Point (Northern Victoria Land, Antarctica). Soil Biol. Biochem. 2018, 123, 165–179. [Google Scholar] [CrossRef]
- Garrido-Benavent, I.; Pérez-Ortega, S.; Durán, J.; Ascaso, C.; Pointing, S.B.; Rodríguez-Cielos, R.; Navarro, F.; de los Ríos, A. Differential Colonization and Succession of Microbial Communities in Rock and Soil Substrates on a Maritime Antarctic Glacier Forefield. Front. Microbiol. 2020, 11, 126. [Google Scholar] [CrossRef]
- Richter, I.; Herbold, C.W.; Lee, C.K.; McDonald, I.R.; Barrett, J.E.; Cary, S.C. Influence of soil properties on archaeal diversity and distribution in the McMurdo Dry Valleys, Antarctica. FEMS Microbiol. Ecol. 2014, 89, 347–359. [Google Scholar] [CrossRef]
- Han, J.; Jung, J.; Park, M.; Hyun, S.; Park, W. Short-term effect of elevated temperature on the abundance and diversity of bacterial and archaeal amoA genes in antarctic soils. J. Microbiol. Biotechnol. 2013, 23, 1187–1196. [Google Scholar] [CrossRef]
- Karaevskaya, E.S.; Demchenko, L.S.; Demidov, N.E.; Rivkina, E.M.; Bulat, S.A.; Gilichinsky, D.A. Archaeal diversity in permafrost deposits of Bunger Hills Oasis and King George Island (Antarctica) according to the 16S rRNA gene sequencing. Microbiology 2014, 83, 398–406. [Google Scholar] [CrossRef]
- Buzzini, P.; Branda, E.; Goretti, M.; Turchetti, B. Psychrophilic yeasts from worldwide glacial habitats: Diversity, adaptation strategies and biotechnological potential. FEMS Microbiol. Ecol. 2012, 82, 217–241. [Google Scholar] [CrossRef]
- Buzzini, P.; Margesin, R. Cold-adapted yeasts: A lesson from the cold and a challenge for the XXI century. In Cold-Adapted Yeasts; Springer: Berlin/Heidelberg, Germany, 2014; pp. 3–22. [Google Scholar]
- Vishniac, H.S. Yeast Biodiversity in the Antarctic. In Biodiversity and Ecophysiology of Yeasts; Springer: Berlin/Heidelberg, Germany, 2006; pp. 419–440. [Google Scholar] [CrossRef]
- Amarelle, V.; Carrasco, V.; Fabiano, E. The hidden life of Antarctic rocks. In The Ecological Role of Micro-Organisms in the Antarctic Environment; Springer: Cham, Switzerland, 2019; pp. 221–237. [Google Scholar] [CrossRef]
- Makhalanyane, T.P.; Valverde, A.; Lacap, D.C.; Pointing, S.B.; Tuffin, M.I.; Cowan, D.A. Evidence of species recruitment and development of hot desert hypolithic communities. Environ. Microbiol. Rep. 2013, 5, 219–224. [Google Scholar] [CrossRef]
- Van Dorst, J.; Benaud, N.; Ferrari, B. New Insights into the Microbial Diversity of Polar Desert Soils: A Biotechnological Perspective. In Microbial Ecology of Extreme Environments; Springer: Cham, Switzerland, 2017; pp. 169–183. ISBN 9783319516868. [Google Scholar]
- Ferrari, B.C.; Bissett, A.; Snape, I.; van Dorst, J.; Palmer, A.S.; Ji, M.; Siciliano, S.D.; Stark, J.S.; Winsley, T.; Brown, M.V. Geological connectivity drives microbial community structure and connectivity in polar, terrestrial ecosystems. Environ. Microbiol. 2016, 18, 1834–1849. [Google Scholar] [CrossRef]
- Ji, M.; Greening, C.; Vanwonterghem, I.; Carere, C.R.; Bay, S.K.; Steen, J.A.; Montgomery, K.; Lines, T.; Beardall, J.; Van Dorst, J.; et al. Atmospheric trace gases support primary production in Antarctic desert surface soil. Nature 2017, 552, 400–403. [Google Scholar] [CrossRef]
- Yergeau, E.; Hogues, H.; Whyte, L.G.; Greer, C.W. The functional potential of high Arctic permafrost revealed by metagenomic sequencing, qPCR and microarray analyses. ISME J. 2010, 4, 1206–1214. [Google Scholar] [CrossRef]
- Treusch, A.H.; Leininger, S.; Kietzin, A.; Schuster, S.C.; Klenk, H.P.; Schleper, C. Novel genes for nitrite reductase and Amo-related proteins indicate a role of uncultivated mesophilic crenarchaeota in nitrogen cycling. Environ. Microbiol. 2005, 7, 1985–1995. [Google Scholar] [CrossRef]
- Hatzenpichler, R.; Lebedeva, E.V.; Spieck, E.; Stoecker, K.; Richter, A.; Daims, H.; Wagner, M. A moderately thermophilic ammonia-oxidizing crenarchaeote from a hot spring. Proc. Natl. Acad. Sci. USA 2008, 105, 2134–2139. [Google Scholar] [CrossRef]
- Leininger, S.; Urich, T.; Schloter, M.; Schwark, L.; Qi, J.; Nicol, G.W.; Prosser, J.I.; Schuster, S.C.; Schleper, C. Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 2006, 442, 806–809. [Google Scholar] [CrossRef]
- Magalhães, C.; Stevens, M.I.; Cary, S.C.; Ball, B.A.; Storey, B.C.; Wall, D.H.; Türk, R.; Ruprecht, U. At Limits of Life: Multidisciplinary Insights Reveal Environmental Constraints on Biotic Diversity in Continental Antarctica. PLoS ONE 2012, 7, e44578. [Google Scholar] [CrossRef]
- Barnard, S.; Van Goethem, M.W.; de Scally, S.Z.; Cowan, D.A.; van Rensburg, P.J.; Claassens, S.; Makhalanyane, T.P. Increased temperatures alter viable microbial biomass, ammonia oxidizing bacteria and extracellular enzymatic activities in Antarctic soils. FEMS Microbiol. Ecol. 2020, 96, fiaa065. [Google Scholar] [CrossRef]
- Rao, S.; Chan, Y.; Lacap, D.C.; Hyde, K.D.; Pointing, S.B.; Farrell, R.L. Low-diversity fungal assemblage in an Antarctic Dry Valleys soil. Polar Biol. 2012, 35, 567–574. [Google Scholar] [CrossRef]
- Vero, S.; Garmendia, G.; Martínez-Silveira, A.; Cavello, I.; Wisniewski, M. Yeast Activities Involved in Carbon and Nitrogen Cycles in Antarctica. In The Ecological Role of Micro-organisms in the Antarctic Environment; Springer: Cham, Switzerland, 2019; pp. 45–64. [Google Scholar]
- Kurtzman, C.; Fell, J.W.; Boekhout, T. The Yeasts: A Taxonomic Study; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Anesio, A.M.; Bellas, C.M. Are low temperature habitats hot spots of microbial evolution driven by viruses? Trends Microbiol. 2011, 19, 52–57. [Google Scholar] [CrossRef]
- Wei, S.T.S.; Higgins, C.M.; Adriaenssens, E.M.; Cowan, D.A.; Pointing, S.B. Genetic signatures indicate widespread antibiotic resistance and phage infection in microbial communities of the McMurdo Dry Valleys, East Antarctica. Polar Biol. 2015, 38, 919–925. [Google Scholar] [CrossRef]
- Zablocki, O.; van Zyl, L.; Adriaenssens, E.M.; Rubagotti, E.; Tuffin, M.; Cary, S.C.; Cowan, D. High-level diversity of tailed phages, eukaryote-associated viruses, and virophage-like elements in the metaviromes of antarctic soils. Appl. Environ. Microbiol. 2014, 80, 6879–6887. [Google Scholar] [CrossRef]
- Adriaenssens, E.M.; Kramer, R.; Van Goethem, M.W.; Makhalanyane, T.P.; Hogg, I.; Cowan, D.A. Environmental drivers of viral community composition in Antarctic soils identified by viromics. Microbiome 2017, 5, 83. [Google Scholar] [CrossRef]
- Dávila-Ramos, S.; Castelán-Sánchez, H.G.; Martínez-ávila, L.; Sánchez-Carbente, M.D.R.; Peralta, R.; Hernández-Mendoza, A.; Dobson, A.D.W.; Gonzalez, R.A.; Pastor, N.; Batista-García, R.A. A review on viral metagenomics in extreme environments. Front. Microbiol. 2019, 10, 2403. [Google Scholar] [CrossRef]
- Hutchins, P.R.; Miller, S.R. Genomics of variation in nitrogen fixation activity in a population of the thermophilic cyanobacterium Mastigocladus laminosus. ISME J. 2017, 11, 78–86. [Google Scholar] [CrossRef]
- Coyne, K.J.; Parker, A.E.; Lee, C.K.; Sohm, J.A.; Kalmbach, A.; Gunderson, T.; León-Zayas, R.; Capone, D.G.; Carpenter, E.J.; Cary, S.C. The distribution and relative ecological roles of autotrophic and heterotrophic diazotrophs in the McMurdo Dry Valleys, Antarctica. FEMS Microbiol. Ecol. 2020, 96, fiaa010. [Google Scholar] [CrossRef]
- Yergeau, E.; Kang, S.; He, Z.; Zhou, J.; Kowalchuk, G.A. Functional microarray analysis of nitrogen and carbon cycling genes across an Antarctic latitudinal transect. ISME J. 2007, 1, 163–179. [Google Scholar] [CrossRef]
- Niederberger, T.; Sohm, J.; Tirindelli, J.; Gunderson, T.; Capone, D.; Carpenter, E.; Cary, S. Diverse and highly active diazotrophic assemblages inhabit ephermally wetted soils of the Antarctic Dry Valleys. FEMS Microbiol. Ecol. 2012, 82, 376–390. [Google Scholar] [CrossRef]
- Crane, S.L.; van Dorst, J.; Hose, G.C.; King, C.K.; Ferrari, B.C. Microfluidic qPCR enables high throughput quantification of microbial functional genes but requires strict curation of primers. Front. Environ. Sci. 2018, 6, 145. [Google Scholar] [CrossRef]
- Asuming-Brempong, S. Microarray Technology and Its Applicability in Soil Science—A Short Review. Open J. Soil Sci. 2012, 2, 333–340. [Google Scholar] [CrossRef]
- Levy-Booth, D.J.; Prescott, C.E.; Grayston, S.J. Microbial functional genes involved in nitrogen fixation, nitrification and denitrification in forest ecosystems. Soil Biol. Biochem. 2014, 75, 11–25. [Google Scholar] [CrossRef]
- Bellenger, J.P.; Darnajoux, R.; Zhang, X.; Kraepiel, A.M.L. Biological nitrogen fixation by alternative nitrogenases in terrestrial ecosystems: A review. Biogeochemistry 2020, 149, 53–73. [Google Scholar] [CrossRef]
- Rubio, L.M.; Ludden, P.W. Maturation of nitrogenase: A biochemical puzzle. J. Bacteriol. 2005, 187, 405–414. [Google Scholar] [CrossRef]
- Dos Santos, P.C.; Fang, Z.; Mason, S.W.; Setubal, J.C.; Dixon, R. Distribution of nitrogen fixation and nitrogenase-like sequences amongst microbial genomes. BMC Genom. 2012, 13, 162. [Google Scholar] [CrossRef]
- Seefeldt, L.C.; Hoffman, B.M.; Dean, D.R. Mechanism of Mo-dependent nitrogenase. Annu. Rev. Biochem. 2009, 78, 701–722. [Google Scholar] [CrossRef]
- Hoffman, B.M.; Lukoyanov, D.; Dean, D.R.; Seefeldt, L.C. Nitrogenase: A draft mechanism. Acc. Chem. Res. 2013, 46, 587–595. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Godfrey, L.V. Electrons, life and the evolution of Earth’s oxygen cycle. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 2705–2716. [Google Scholar] [CrossRef]
- Godfrey, L.V.; Falkowski, P.G. The cycling and redox state of nitrogen in the Archaean ocean. Nat. Geosci. 2009, 2, 725–729. [Google Scholar] [CrossRef]
- Zehr, J.P.; Jenkins, B.D.; Short, S.M.; Steward, G.F. Nitrogenase gene diversity and microbial community structure: A cross-system comparison. Environ. Microbiol. 2003, 5, 539–554. [Google Scholar] [CrossRef] [PubMed]
- Latysheva, N.; Junker, V.L.; Palmer, W.J.; Codd, G.A.; Barker, D. The evolution of nitrogen fixation in cyanobacteria. Bioinformatics 2012, 28, 603–606. [Google Scholar] [CrossRef]
- Lacap-Bugler, D.C.; Lee, K.K.; Archer, S.; Gillman, L.N.; Lau, M.; Leuzinger, S.; Lee, C.K.; Maki, T.; McKay, C.P.; Perrott, J.K.; et al. Global Diversity of Desert Hypolithic Cyanobacteria. Front. Microbiol. 2017, 8, 867. [Google Scholar] [CrossRef] [PubMed]
- Voytek, M.A.; Priscu, J.C.; Ward, B.B. The distribution and relative abundance of ammonia-oxidizing bacteria in lakes of the McMurdo Dry Valley, Antarctica. Hydrobiologia 1999, 401, 113–130. [Google Scholar] [CrossRef]
- Ayton, J.; Aislabie, J.; Barker, G.M.; Saul, D.; Turner, S. Crenarchaeota affiliated with group 1.1 b are prevalent in coastal mineral soils of the Ross Sea region of Antarctica. Environ. Microbiol. 2010, 12, 689–703. [Google Scholar] [CrossRef] [PubMed]
- Howard-Williams, C.; Hawes, I. Ecological processes in Antarctic inland waters: Interactions between physical processes and the nitrogen cycle. Antarct. Sci. 2007, 19, 205–217. [Google Scholar] [CrossRef]
- Tolar, B.B.; Ross, M.J.; Wallsgrove, N.J.; Liu, Q.; Aluwihare, L.I.; Popp, B.N. Contribution of ammonia oxidation to chemoautotrophy in Antarctic coastal waters. ISME J. 2016, 10, 2605–2619. [Google Scholar] [CrossRef]
- Monteiro, M.; Baptista, M.S.; Séneca, J.; Torgo, L.; Lee, C.K.; Cary, S.C.; Magalhães, C. Understanding the Response of Nitrifying Communities to Disturbance in the McMurdo Dry Valleys, Antarctica. Microorganisms 2020, 8, 404. [Google Scholar] [CrossRef]
- Stephen, J.R.; McCaig, A.E.; Smith, Z.; Prosser, J.I.; Embley, T.M. Molecular diversity of soil and marine 16S rRNA gene sequences related to b-subgroup ammonia-oxidizing bacteria. Appl. Environ. Microbiol. 1996, 62, 4147–4154. [Google Scholar] [CrossRef]
- Stephen, J.R.; Kowalchuk, G.A.; Bruns, M.A.V.; McCaig, A.E.; Phillips, C.J.; Embley, T.M.; Prosser, J.I. Analysis of β-subgroup Proteobacterial ammonia oxidizer populations in soil by denaturating gradient gel electrophoresis analysis and hierarchical phylogenetic probing. Appl. Environ. Microbiol. 1998, 64, 2958–2965. [Google Scholar] [CrossRef]
- McCaig, A.E.; Phillips, C.J.; Stephen, J.R.; Kowalchuk, G.A.; Harvey, S.M.; Herbert, R.A.; Embley, T.M.; Prosser, J.I. Nitrogen cycling and community structure of proteobacterial beta-subgroup ammonia-oxidizing bacteria within polluted marine fish farm sediments. Appl. Environ. Microbiol. 1999, 65, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, C.; Machado, A.; Bordalo, A.A. Temporal variability of relative abundance of ammonia oxidizing bacteria vs. archaea in the sandy at of the Douro River estuary, Portugal. Aquat. Microb. Ecol. 2009, 56, 13–23. [Google Scholar] [CrossRef]
- Philippot, L.; Hojberg, O. Dissimilatory nitrate reductases in bacteria. Biochim. Biophys. Acta. 1999, 1577, 1–23. [Google Scholar] [CrossRef]
- Braker, G.; Zhou, J.; Wu, L.; Devol, A.H.; Tiedje, J.M. Nitrite reductase genes (nirK and nirS) as functional markers to investigate diversityof denitrifying bacteria in Pacific Northwest marine sediment communities. Appl. Environ. Microbiol. 2000, 66, 2096–2104. [Google Scholar] [CrossRef]
- Prieme, A.; Braker, G.; Tiedje, J.M. Diversity of nitrite reductase (nirK and nirS) gene fragments in forested upland and wetland soils. Appl. Environ. Microbiol. 2002, 68, 1893–1900. [Google Scholar] [CrossRef]
- Zumft, W.G. Cell biology and molecular basis of denitrification. Microbiol. Mol. Biol. 1997, 61, 533–616. [Google Scholar] [CrossRef]
- Pérez, C.A.; Aravena, J.C.; Ivanovich, C.; McCulloh, R. Effects of penguin guano and moisture on nitrogen biological fixation in maritime Antarctic soils. Polar Biol. 2017, 40, 437–448. [Google Scholar] [CrossRef]
- Chan, Y.; Lacap, D.C.; Lau, M.C.Y.; Ha, K.Y.; Warren-Rhodes, K.A.; Cockell, C.S.; Cowan, D.A.; McKay, C.P.; Pointing, S.B. Hypolithic microbial communities: Between a rock and a hard place. Environ. Microbiol. 2012, 14, 2272–2282. [Google Scholar] [CrossRef]
- Davey, A.; Marchant, H.J. Seasonal variation in nitrogen fixation by Nostoc commune Vaucher at the Vestfold Hills, Antarctica. Phycologia 1983, 22, 377–385. [Google Scholar] [CrossRef]
- Nakatsubo, T.; Ino, Y. Nitrogen cycling in an Antarctic ecosystem 2. Estimation of the amount of nitrogen fixation in a moss community on East Ongul Island. Ecol. Res. 1987, 2, 31–40. [Google Scholar] [CrossRef]
- Line, M.A. Nitrogen fixation in the sub-Antarctic Macquarie Island. Polar Biol. 1991, 11, 601–606. [Google Scholar] [CrossRef]
- Vincent, W. Cyanobacterial Dominance in the Polar Regions. In The Ecology of Cyanobacteria; Whitton, B.A., Potts, M., Eds.; Kluwer: Dordrecht, The Netherlands, 2007; pp. 321–340. [Google Scholar]
- Hopkins, D.; Sparrow, A.; Elberling, B.; Gregorich, E.; Novis, P.; Greenfield, L.; Tilston, E. Carbon, nitrogen and temperature controls on microbial activity in soils from an Antarctic Dry Valley. Soil Biol. Biochem. 2006, 38, 3130–3140. [Google Scholar] [CrossRef]
- Strauss, S.L.; Garcia-Pichel, F.; Day, T.A. Soil microbial carbon and nitrogen transformations at a glacial foreland on Anvers Island, Antarctic Peninsula. Polar Biol. 2012, 35, 1459–1471. [Google Scholar] [CrossRef]
- Stewart, W.D.; Fitzgerald, G.P.; Burris, R.H. In situ studies on N2 fixation using the acetylene reduction technique. Proc. Nat. Acad. Sci. USA 1967, 58, 2071–2078. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.E.; Virginia, R.A.; Lyons, W.B.; McKnight, D.M.; Priscu, J.C.; Doran, P.T.; Fountain, A.G.; Wall, D.H.; Moorhead, D.L. Biogeochemical stoichiometry of Antarctic Dry Valley ecosystems. J. Geophys. Res. 2007, 112, G01010. [Google Scholar] [CrossRef]
- Barrett, J.; Virginia, R.; Hopkins, D.; Aislabie, J.; Bargagli, R.; Bockheim, J.; Campbell, I.; Lyons, W.; Moorhead, D.; Nkem, J.; et al. Terrestrial ecosystem processes of Victoria Land. Antarctica. Soil Biol. Biochem. 2006, 38, 3019–3034. [Google Scholar] [CrossRef]
- Kohler, T.J.; Stanish, L.F.; Liptzin, D.; Barrett, J.E.; McKnight, D.M. Catch and release: Hyporheic retention and mineralization of N-fixing Nostoc sustains downstream microbial mat biomass in two polar desert streams. Limnol. Oceanogr. 2018, 3, 357–364. [Google Scholar] [CrossRef]
- Dennis, P.G.; Newsham, K.K.; Rushton, S.P.; Ord, V.J.; O’Donnell, A.G.; Hopkins, D.W. Warming constrains bacterial community responses to nutrient inputs in a southern, but not northern, maritime Antarctic soil. Soil Biol. Biochem. 2013, 57, 248–255. [Google Scholar] [CrossRef]
- Van Horn, D.J.; Okie, J.G.; Buelow, H.N.; Gooseff, M.N.; Barrett, J.E.; Takacs-Vesbach, C.D. Soil microbial responses to increased moisture and organic resources along a salinity gradient in a polar desert. Appl. Environ. Microb. 2014, 80, 3034–3043. [Google Scholar] [CrossRef]
- Ball, B.A.; Adams, B.J.; Barrett, J.E.; Wall, D.H.; Virginia, R.A. Soil biological responses to C, N and P fertilization in a polar desert of Antarctica. Soil Biol. Biochem. 2018, 122, 7–18. [Google Scholar] [CrossRef]
- Hopkins, D.W.; Sparrow, A.D.; Shillam, L.L.; English, L.C.; Dennis, P.G.; Novis, P.; Elberling, B.; Gregorich, E.G.; Greenfield, L.G. Enzymatic activities and microbial communities in an Antarctic dry valley soil: Responses to C and N supplementation. Soil Biol. Biochem. 2008, 40, 2130–2136. [Google Scholar] [CrossRef]
- Sparrow, A.D.; Gregorich, E.G.; Hopkins, D.W.; Novis, P.; Elberling, B.; Greenfield, L.G. Resource limitations on soil microbial activity in an Antarctic dry valley. Soil Sci. Soc. Am. J. 2011, 75, 2188–2197. [Google Scholar] [CrossRef]
- Dennis, P.G.; Sparrow, A.D.; Gregorich, E.G.; Novis, P.M.; Elberling, B.; Greenfield, L.G.; Hopkins, D.W. Microbial responses to carbon and nitrogen supplementation in an Antarctic dry valley soil. Antarct. Sci. 2013, 25, 55–61. [Google Scholar] [CrossRef]
- Newsham, K.K.; Tripathi, B.M.; Dong, K.; Yamamoto, N.; Adams, J.M.; Hopkins, D.W. Bacterial community composition and diversity respond to nutrient amendment but not warming in a maritime Antarctic soil. Microb. Ecol. 2019, 78, 974–984. [Google Scholar] [CrossRef] [PubMed]
- Ball, B.A.; Virginia, R.A. Microbial biomass and respiration responses to nitrogen fertilization in a polar desert. Polar Biol. 2014, 3, 573–585. [Google Scholar] [CrossRef]
- Benhua, S.; Dennis, P.G.; Laudicina, V.A.; Ord, V.J.; Rushton, S.P.; O’Donnell, A.G.; Newsham, K.K.; Hopkins, D.W. Biogeochemical responses to nutrient, moisture and temperature manipulations of soil from Signy Island, South Orkney Islands in the Maritime Antarctic. Antarct. Sci. 2014, 26, 513–520. [Google Scholar] [CrossRef]
- Hopkins, D.W.; Dennis, P.G.; Rushton, S.P.; Newsham, K.K.; O’Donnell, T.G. Lean and keen: Microbial activity in soils from the Maritime Antarctic. Eur. J. Soil. Sci. 2020, 1–19. [Google Scholar] [CrossRef]
- Schwartz, E.; Van Horn, D.J.; Buelow, H.N.; Okie, J.G.; Gooseff, M.N.; Barrett, J.E.; Takacs-Vesbach, C.D. Characterization of growing bacterial populations in McMurdo Dry Valley soils through stable isotope probing with 18O-water. FEMS Microbiol. Ecol. 2014, 89, 415–425. [Google Scholar] [CrossRef]
- Buelow, H.N.; Winter, A.S.; Van Horn, D.J.; Barrett, J.E.; Gooseff, M.N.; Schwartz, E.; Takacs-Vesbach, C.D. Microbial community responses to increased water and organic matter in the arid soils of the mcmurdo dry valleys, Antarctica. Front. Microbiol. 2016, 7, 1040. [Google Scholar] [CrossRef]
- Cain, M.L.; Subler, S.; Evans, J.P.; Fortin, M.-J. Sampling spatial and temporal variation in soil nitrogen availability. Oecologia 1999, 118, 397–404. [Google Scholar] [CrossRef]
- Knops, J.M.H.; Tilman, D. Dynamics of soil nitrogen and carbon accumulation for 61 years after agricultural abandonment. Ecology 2000, 81, 88–98. [Google Scholar] [CrossRef]
- Niederberger, T.D.; Bottos, E.M.; Sohm, J.A.; Gunderson, T.; Parker, A.; Coyne, K.J.; Capone, D.G.; Carpenter, E.J.; Cary, S.C. Rapid Microbial Dynamics in Response to an Induced Wetting Event in Antarctic Dry Valley Soils. Front. Microbiol. 2019, 10, 621. [Google Scholar] [CrossRef] [PubMed]
- Doran, P.T.; Priscu, J.C.; Lyons, W.B.; Walsh, J.E.; Fountain, A.G.; McKnight, D.M.; Moorhead, D.L.; Virginia, R.A.; Wall, D.H.; Clow, G.D.; et al. Antarctic climate cooling and terrestrial ecosystem response. Nature 2002, 415, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.E.; Virginia, R.A.; Wall, D.H.; Doran, P.T.; Fountain, A.G.; Welch, K.A.; Lyons, W.B. Persistent effects of a discrete warming event on a polar desert ecosystem. Glob. Chang. Biol. 2008, 14, 2249–2261. [Google Scholar] [CrossRef]
- Bundy, J.G.; Davey, M.P.; Viant, M.R. Environmental metabolomics: A critical review and future perspectives. Metabolomics 2009, 5, 3–21. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J.; Rivas-Ubach, A. Ecological metabolomics: Overview of current developments and future challenges. Chemoecology 2011, 21, 191–225. [Google Scholar] [CrossRef]
- Coleine, C.; Gevi, F.; Fanelli, G.; Onofri, S.; Timperio, A.M.; Selbmann, L. Specific adaptations are selected in opposite sun exposed Antarctic cryptoendolithic communities as revealed by untargeted metabolomics. PLoS ONE 2020, 15, e0233805. [Google Scholar] [CrossRef]
- Guerra, C.A.; Heintz-Buschart, A.; Sikorski, J.; Chatzinotas, A.; Guerrero-Ramírez, N.; Cesarz, S.; Beaumelle, L.; Rillig, M.C.; Maestre, F.T.; Delgado-Baquerizo, M.; et al. Blind spots in global soil biodiversity and ecosystem function research. BioRxiv 2019, 20, 774356. [Google Scholar] [CrossRef]
| Inorganic N | Total Organic N | References | ||
|---|---|---|---|---|
| NO3−-N | NH4+-N | |||
| (µg g−1) | (µg g−1) | (ppm) | ||
| Ornithogenic Soils | ||||
| Marion Island | 0–130 | 101–664 | - | [18,19] |
| Signy Island | 90 | 1369 | ||
| S. Shetland Island | 18.8–35 | 140–400 | ||
| Ross Island/20–35 soil water | 0 | 6000 | ||
| Desert Soils | ||||
| Ross Desert | 0–120 | 6–40 | - | [15,16,17,18,19] |
| Ross Desert | 0–960 | 0–2.2 | ||
| Pensacola Mountains | <1–1250 | <1 | ||
| Pensacola Mountains | 0.7–6.4 | 0.3–1.1 | ||
| Victoria Valley | 2 | 1 | 0.003 | |
| Coalsack Bluff | 742 | 0.1 | 0.024 | |
| Barwick Valley | 1 | 0.8 | 0.004 | |
| Wheeler Valley | 7 | ND* | 0.024 | |
| Fellfield Soils | ||||
| Marion Island | 0 | 2 | - | [18,19] |
| Signy Island/10–20 soil water | 1.9–6 | 43,894 | ||
| Signy Island | 0.1 | 0.1 | ||
| Coastal Antarctica | 43,850 | 15–20 | ||
| Maritime Coastal Soils | ||||
| Penguin Rock, soils with penguin | 31.19 | 385.52 | ||
| Rakusa Point | 4.74 | 66.32 | ||
| Puchalski grave, tundra on slope | 1.62 | 23.6 | - | [21] |
| Jersak Hills, scree debris | 1.32 | 1.16 | ||
| Arctowski station, base soils | 5.25 | 60.43 | ||
| Phylum | Taxa | Role | References |
|---|---|---|---|
| Cyanobacteria | Nostoc commune | Nitrogen fixation | [38] |
| Dichothrix Nodularia Hydrocoryne Hydrocoryne | Nitrogen sequestration | [39,40] | |
| Nitrosospira Nitrosomonas | Ammonia oxidation | [41,42] | |
| Nostoc Anabeana | Nitrification | [12,43] | |
| Actinobacteria | Streptomyces Frankeniaceae | Nitrogen fixation | [44,45] |
| - | Denitrification | [25,37] | |
| Bacteroidetes | - | Denitrification | [34,36,44] |
| Proteobacteria | Burkholderiales | Nitrogen input | [46] |
| Deltaproteobacteria | Denitrification | [34,36,44] | |
| Chloroflexi | - | Denitrification | [44,45] |
| Thaumarchaeota | Crenarchaeota Marine Group 1.1b | Nitrogen fixation | [47] |
| Nitrososphaerales | Ammonia oxidizing | [25,41,42,48,49] | |
| Basidiomycota Ascomycota | Rhodotorula muscorum Rhodotorula mucilaginosa Cryptococcus aerius Cryptococcus albidus | Nitrogen mineralization, nitrogen assimilation and ammonification | [50,51] |
| Trichosporon cutaneum | Denitrification | [52] | |
| Candida sp. | Denitrification | [52] |
| Source | N2-Fixation/Nitrification Rates | Year | References |
|---|---|---|---|
| Moss-associated Nostoc in Vestfold Hills | 52–119 mg N m−2 yr−1 | 1983 | [104] |
| Moss community in East Ongul Island | 0–15 mg N m−2 day−1 | 1987 | [105] |
| Soil and grassland from Macquarie Island | 0–1372 nmol C2H2 reduction h−1 10 cm−1 | 1992 | [106] |
| Dry turf and wet moss carpets, Signy Island | 1–4.9 kg N ha−1 yr−1 | 2000 | [107] |
| Soil from Garwood Valley | 5–1000 ng NO2-N g−1 soil week−1 | 2006 | [108] |
| Hypoliths in the McMurdo Valley | 0.38 kg N yr−1 | 2011 | [36] |
| Soil samples in Anvers Island | 12–59 μmol N m−2 h−1 | 2012 | [109] |
| Soils in Miers Valley | 0–5.8 nmol N cm−3 h−1 | 2012 | [75] |
| Biological soil crusts in Ardley Island | 0–3 kg N ha−1 yr−1 | 2017 | [102] |
| Soil from Miers Valley and Beacon Valley | 0.025–0.07 pmol NH4+ oxidation g−1 day−1 | 2020 | [93] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz, M.; Bosch, J.; Coclet, C.; Johnson, J.; Lebre, P.; Salawu-Rotimi, A.; Vikram, S.; Makhalanyane, T.; Cowan, D. Microbial Nitrogen Cycling in Antarctic Soils. Microorganisms 2020, 8, 1442. https://doi.org/10.3390/microorganisms8091442
Ortiz M, Bosch J, Coclet C, Johnson J, Lebre P, Salawu-Rotimi A, Vikram S, Makhalanyane T, Cowan D. Microbial Nitrogen Cycling in Antarctic Soils. Microorganisms. 2020; 8(9):1442. https://doi.org/10.3390/microorganisms8091442
Chicago/Turabian StyleOrtiz, Max, Jason Bosch, Clément Coclet, Jenny Johnson, Pedro Lebre, Adeola Salawu-Rotimi, Surendra Vikram, Thulani Makhalanyane, and Don Cowan. 2020. "Microbial Nitrogen Cycling in Antarctic Soils" Microorganisms 8, no. 9: 1442. https://doi.org/10.3390/microorganisms8091442
APA StyleOrtiz, M., Bosch, J., Coclet, C., Johnson, J., Lebre, P., Salawu-Rotimi, A., Vikram, S., Makhalanyane, T., & Cowan, D. (2020). Microbial Nitrogen Cycling in Antarctic Soils. Microorganisms, 8(9), 1442. https://doi.org/10.3390/microorganisms8091442






