Hot in Cold: Microbial Life in the Hottest Springs in Permafrost
Abstract
1. Introduction
2. Materials and Methods
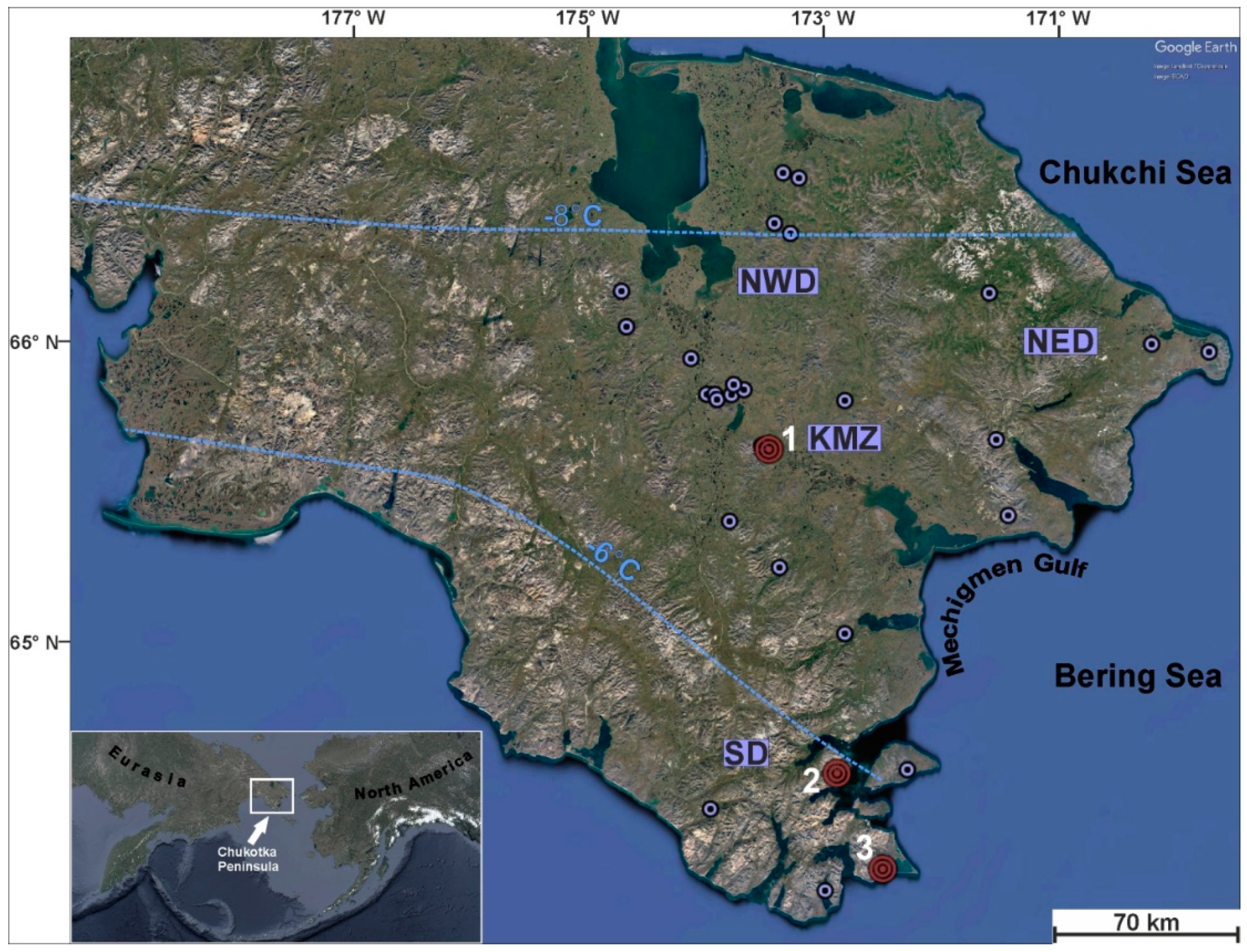
2.1. Characteristics of the Sampling Sites
2.2. Water Chemistry Analyses and Mineralogy of the Sampling Sites
2.3. High-Throughput 16S rRNA Gene Amplicon Sequencing
2.4. Data Analysis
3. Results
3.1. Mineralogy of the Sampling Sites
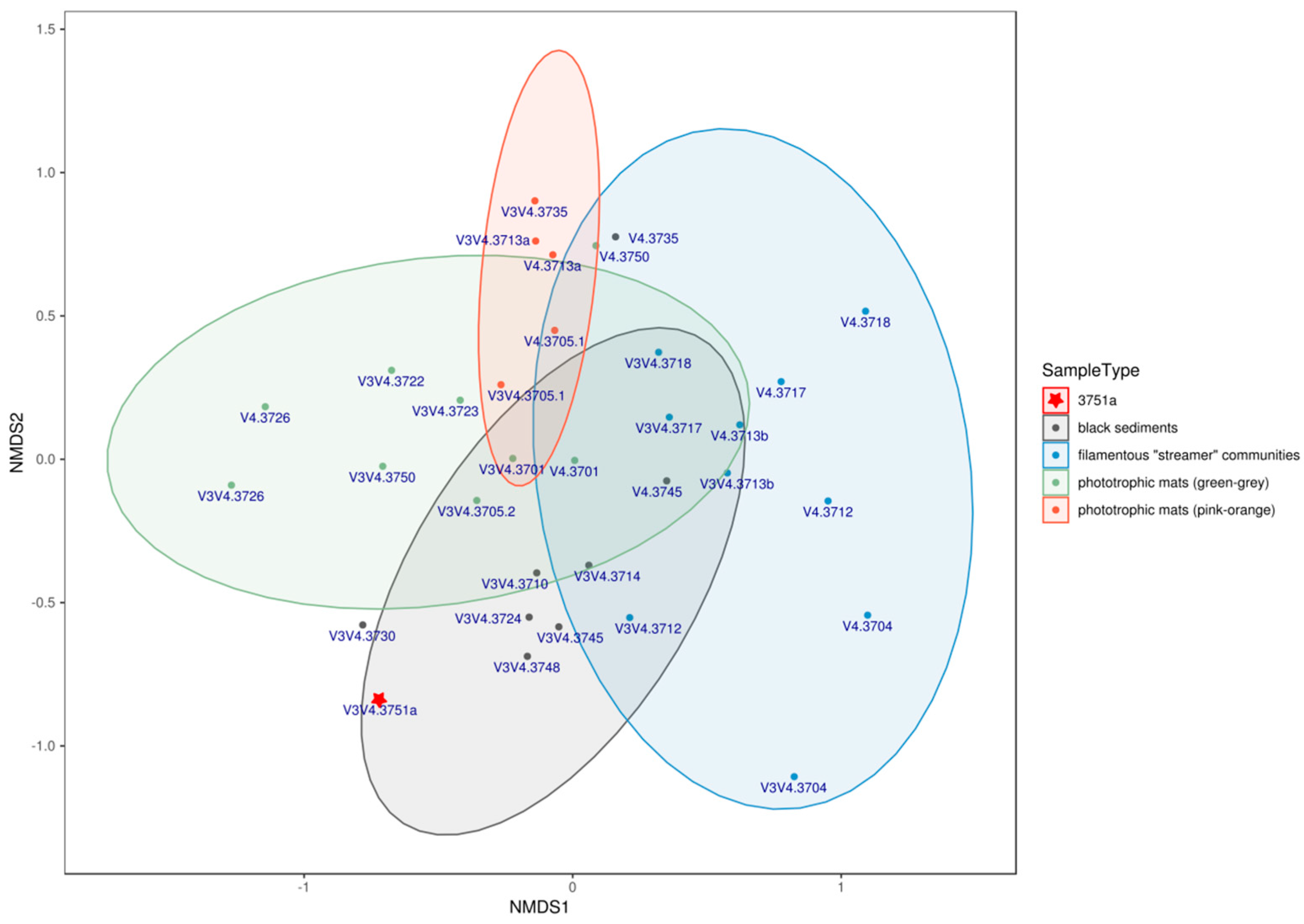
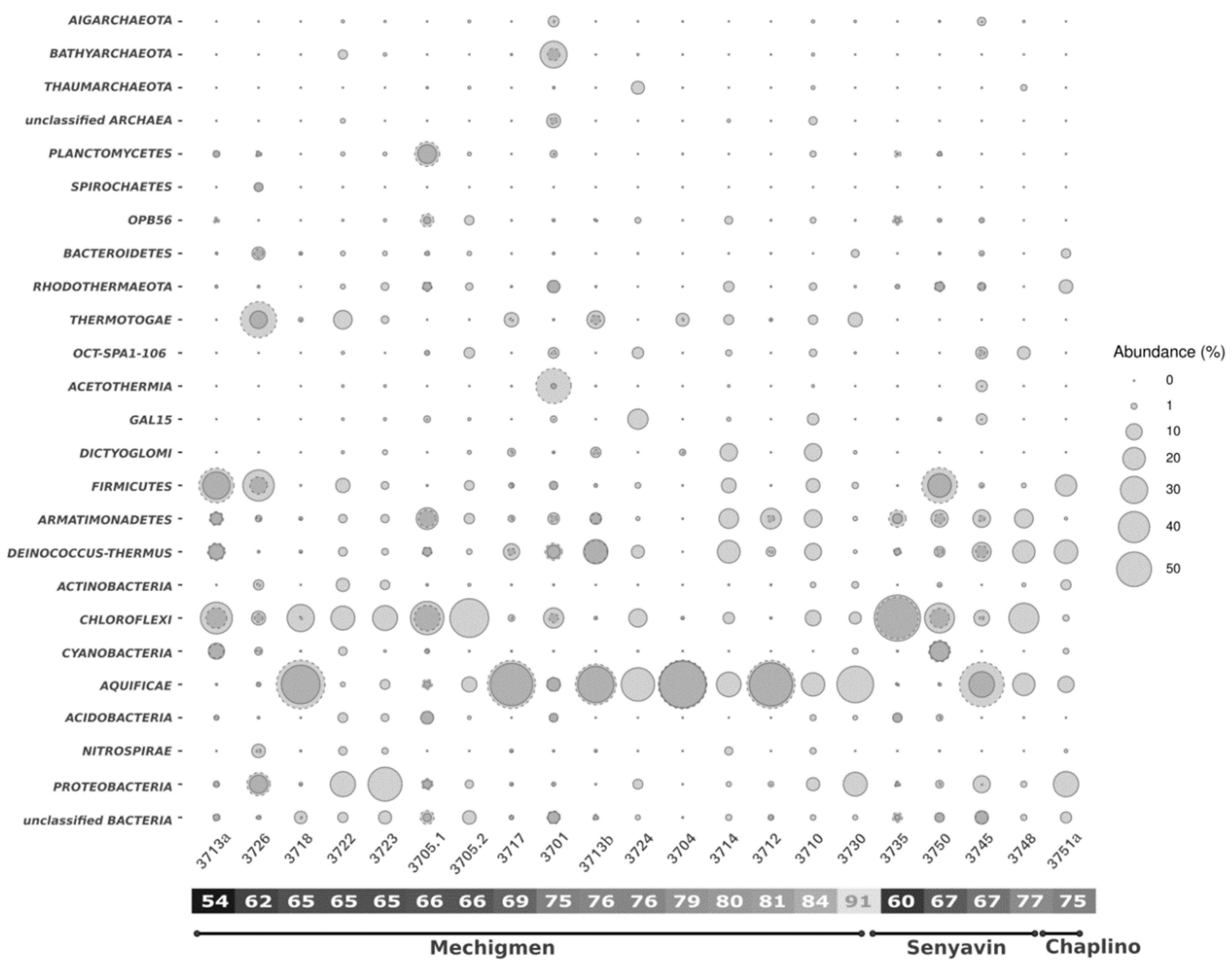
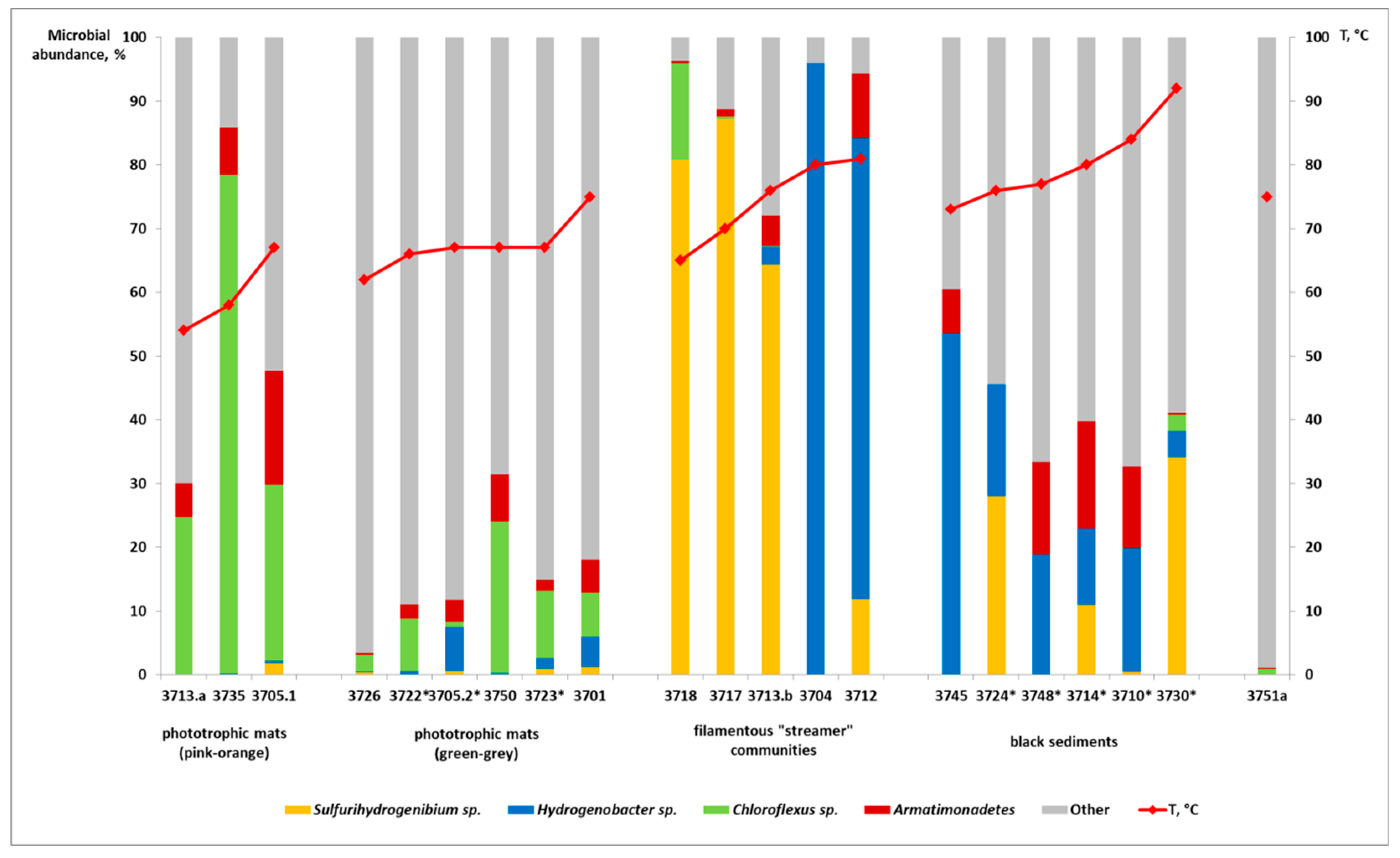
3.2. Analysis of Microbial Diversity Using High-Throughput Amplicon Profiling
4. Discussion
5. Conclusions
- Photosynthetic microbial communities are dominated by green bacteria of genus Chloroflexus, outcompeting thermophilic cyanobacteria due to a combination of physicochemical characteristics of CHS and sunlight insolation, characteristic of this polar region.
- In high-temperature CHS, neither sulfur compounds nor sulfur-metabolizing archaea are present, and the growth of dominating organisms—representatives of phylum Aquificae—is supposed to be supported by the oxidation of ferrous iron that is dissolved in the water or present in iron-containing minerals.
- Uncultivated and Candidatus bacterial and archaeal lineages are rather abundant in some of the CHS, indicating these environments could serve for future metagenomic and cultivation studies of these lineages, their metabolism, and ecological function reconstruction.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brown, J.; Ferrians, O.J.; Heginbottom, J.A.; Melnikov, E.S. Circum-Arctic Map of Permafrost and Ground-Ice Conditions; Circum-Pacific Map Series, Map CP-45; US Geological Survey: Reston, VA, USA, 1997. [CrossRef]
- Polyak, B.G.; Dubinina, E.O.; Lavrushin, V.Y.; Cheshko, A.L. Isotopic composition of thermal waters in Chukotka. Lithol. Miner. Resour. 2008, 43, 429–453. [Google Scholar] [CrossRef]
- Polyak, B.G.; Prasolov, E.M.; Lavrushin, V.Y.; Cheshko, A.L.; Kamenskii, I.L. He, Ar, C and N isotopes in thermal springs of the Chukotka Peninsula: Geochemical evidence of the recent rifting in the north-eastern Asia. Chem. Geol. 2013, 339, 127–140. [Google Scholar] [CrossRef]
- Faulds, J.E.; Coolbaugh, M.F.; Vice, G.S.; Edwards, M.L. Characterizing structural controls of geothermal fields in the northwestern Great Basin: A progress report. Geotherm. Resour. Counc. Trans. 2006, 30, 69–76. [Google Scholar]
- Erkan, K.; Holdman, G.; Blackwell, D.; Benoit, W. Thermal characteristics of the Chena hot springs Alaska geothermal system. In Proceedings of the Thirty-Second Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 22–24 January 2007; pp. 1–8. [Google Scholar]
- Costa, K.C.; Navarro, J.B.; Shock, E.L.; Zhang, C.L.; Soukup, D.; Hedlund, B.P. Microbiology and geochemistry of great boiling and mud hot springs in the United States Great Basin. Extremophiles 2009, 13, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, E.; Mykytczuk, N.C.S.; Pellerin, A.; Goordial, J.; Twine, S.M.; Wing, B.; Foote, S.J.; Fulton, K.; Whyte, L.G. Thiomicrorhabdus streamers and sulfur cycling in perennial hypersaline cold springs in the Canadian high Arctic. Environ. Microbiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Grasby, S.E.; Hutcheon, I.; Krouse, H.R. The influence of water-rock interaction on the chemistry of thermal springs in western Canada. Appl. Geochem. 2000, 15, 439–454. [Google Scholar] [CrossRef]
- Starke, V.; Kirshtein, J.; Fogel, M.L.; Steele, A. Microbial community composition and endolith colonization at an Arctic thermal spring are driven by calcite precipitation. Environ. Microbiol. Rep. 2013. [Google Scholar] [CrossRef]
- Roeselers, G.; Norris, T.B.; Castenholz, R.W.; Rysgaard, S.; Glud, R.N.; Kühl, M.; Muyzer, G. Diversity of phototrophic bacteria in microbial mats from Arctic hot springs (Greenland). Environ. Microbiol. 2007, 9, 26–38. [Google Scholar] [CrossRef]
- Flores, P.A.; Amenábar, M.J.; Blamey, J.M. Hot environments from antarctica: Source of thermophiles and hyperthermophiles, with potential biotechnological applications. In Thermophilic Microbes in Environmental and Industrial Biotechnology: Biotechnology of Thermophiles; Springer: Dordrecht, The Netherlands, 2013; pp. 99–118. ISBN 9789400758995. [Google Scholar]
- Flores, P.A.M.; Correa-Llantén, D.N.; Blamey, J.M. A thermophilic microorganism from Deception Island, Antarctica with a thermostable glutamate dehydrogenase activity. Biol. Res. 2018, 51, 55. [Google Scholar] [CrossRef]
- Sharp, C.E.; Brady, A.L.; Sharp, G.H.; Grasby, S.E.; Stott, M.B.; Dunfield, P.F. Humboldt’s spa: Microbial diversity is controlled by temperature in geothermal environments. ISME J. 2014, 8, 1166–1174. [Google Scholar] [CrossRef]
- Glotova, L.P. The History of Hydrogeological Investigations; Nedra: Moscow, Russia, 1972; pp. 11–27. (In Russian) [Google Scholar]
- Marmur, J. A procedure for the isolation of deoxyribonucleic acid from micro-organisms. J. Mol. Biol. 1961, 3, 208–218. [Google Scholar] [CrossRef]
- Fadrosh, D.W.; Ma, B.; Gajer, P.; Sengamalay, N.; Ott, S.; Brotman, R.M.; Ravel, J. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2014, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Tomita, J.; Nishioka, K.; Hisada, T.; Nishijima, M. Development of a Prokaryotic Universal Primer for Simultaneous Analysis of Bacteria and Archaea Using Next-Generation Sequencing. PLoS ONE 2014, 9, e105592. [Google Scholar] [CrossRef] [PubMed]
- Gohl, D.M.; Vangay, P.; Garbe, J.; MacLean, A.; Hauge, A.; Becker, A.; Gould, T.J.; Clayton, J.B.; Johnson, T.J.; Hunter, R.; et al. Systematic improvement of amplicon marker gene methods for increased accuracy in microbiome studies. Nat. Biotechnol. 2016, 34, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Hugerth, L.W.; Wefer, H.A.; Lundin, S.; Jakobsson, H.E.; Lindberg, M.; Rodin, S.; Engstrand, L.; Andersson, A.F. DegePrime, a Program for Degenerate Primer Design for Broad-Taxonomic-Range PCR in Microbial Ecology Studies. Appl. Environ. Microbiol. 2014, 80, 5116–5123. [Google Scholar] [CrossRef]
- Merkel, A.Y.; Tarnovetskii, I.Y.; Podosokorskaya, O.A.; Toshchakov, S.V. Analysis of 16S rRNA Primer Systems for Profiling of Thermophilic Microbial Communities. Microbiology 2019, 88, 671–680. [Google Scholar] [CrossRef]
- Renaud, G.; Stenzel, U.; Maricic, T.; Wiebe, V.; Kelso, J. deML: Robust demultiplexing of Illumina sequences using a likelihood-based approach. Bioinformatics 2015, 31, 770–772. [Google Scholar] [CrossRef]
- Kozich, J.J.; Westcott, S.L.; Baxter, N.T.; Highlander, S.K.; Schloss, P.D. Development of a Dual-Index Sequencing Strategy and Curation Pipeline for Analyzing Amplicon Sequence Data on the MiSeq Illumina Sequencing Platform. Appl. Environ. Microbiol. 2013, 79, 5112–5120. [Google Scholar] [CrossRef]
- Westcott, S.L.; Schloss, P.D. OptiClust, an Improved Method for Assigning Amplicon-Based Sequence Data to Operational Taxonomic Units. mSphere 2017, 2. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naïve Bayesian Classifier for Rapid Assignment of rRNA Sequences into the New Bacterial Taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- McMurdie, P.J.; Holmes, S. phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [PubMed]
- Valero-Mora, P.M. ggplot2: Elegant Graphics for Data Analysis. J. Stat. Softw. 2010, 35. [Google Scholar] [CrossRef]
- Tindall, B.J.; Rosselló-Móra, R.; Busse, H.J.; Ludwig, W.; Kämpfer, P. Notes on the characterization of prokaryote strains for taxonomic purposes. Int. J. Syst. Evol. Microbiol. 2010, 60, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Albores, F.; Ortiz-Suárez, L.E.; Villalpando-Canchola, E.; Martínez-Porchas, M. Size-variable zone in V3 region of 16S rRNA. RNA Biol. 2017, 14, 1514–1521. [Google Scholar] [CrossRef] [PubMed]
- Baskov EA, S.S. Alkalic (and Slightly Acidic) Hydrotherms; Nedra: Leningrad, Russia, 1989; pp. 76–79. (In Russian) [Google Scholar]
- Chan, C.S.; Chan, K.-G.; Ee, R.; Hong, K.-W.; Urbieta, M.S.; Donati, E.R.; Shamsir, M.S.; Goh, K.M. Effects of Physiochemical Factors on Prokaryotic Biodiversity in Malaysian Circumneutral Hot Springs. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Castenholz, R.W.; Pierson, B.K. Ecology of Thermophilic Anoxygenic Phototrophs. In Anoxygenic Photosynthetic Bacteria; Springer: Dordrecht, The Netherlands, 2006; pp. 87–103. [Google Scholar]
- van der Meer, M.T.J.; Schouten, S.; Bateson, M.M.; Nübel, U.; Wieland, A.; Kühl, M.; de Leeuw, J.W.; Sinninghe Damsté, J.S.; Ward, D.M. Diel Variations in Carbon Metabolism by Green Nonsulfur-Like Bacteria in Alkaline Siliceous Hot Spring Microbial Mats from Yellowstone National Park. Appl. Environ. Microbiol. 2005, 71, 3978–3986. [Google Scholar] [CrossRef] [PubMed]
- Finke, N.; Hoehler, T.M.; Polerecky, L.; Buehring, B.; Thamdrup, B. Competition for inorganic carbon between oxygenic and anoxygenic phototrophs in a hypersaline microbial mat, Guerrero Negro, Mexico. Environ. Microbiol. 2013, 15, 1532–1550. [Google Scholar] [CrossRef]
- Pierson, B.K.; Castenholz, R.W. The Family Chloroflexaceae. In The Prokaryotes; Springer: New York, NY, USA, 1992; pp. 3754–3774. [Google Scholar]
- Tang, K.-H.; Barry, K.; Chertkov, O.; Dalin, E.; Han, C.S.; Hauser, L.J.; Honchak, B.M.; Karbach, L.E.; Land, M.L.; Lapidus, A.; et al. Complete genome sequence of the filamentous anoxygenic phototrophic bacterium Chloroflexus aurantiacus. BMC Genom. 2011, 12, 334. [Google Scholar] [CrossRef]
- Kanno, N.; Haruta, S.; Hanada, S. Sulfide-dependent Photoautotrophy in the Filamentous Anoxygenic Phototrophic Bacterium, Chloroflexus aggregans. Microbes Environ. 2019, 34, 304–309. [Google Scholar] [CrossRef]
- Kavokin, A.; Malpuech, G. Thin Films and Nanostructures: Cavity Polaritons; Academic Press: Cambridge, MA, USA, 2003; ISBN 0125330324. [Google Scholar]
- Hanada, S.; Hiraishi, A.; Shimada, K.; Matsuura, K. Chloroflexus aggregans sp. nov., a filamentous phototrophic bacterium which forms dense cell aggregates by active gliding movement. Int. J. Syst. Bacteriol. 1995, 45, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Jungblut, A.D.; Vincent, W.F. Cyanobacteria in polar and alpine ecosystems. In Psychrophiles: From Biodiversity to Biotechnology, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 181–206. ISBN 9783319570570. [Google Scholar]
- Sanfilippo, J.E.; Garczarek, L.; Partensky, F.; Kehoe, D.M. Chromatic Acclimation in Cyanobacteria: A Diverse and Widespread Process for Optimizing Photosynthesis. Annu. Rev. Microbiol. 2019, 73, 407–433. [Google Scholar] [CrossRef] [PubMed]
- Skirnisdottir, S.; Hreggvidsson, G.O.; Hjörleifsdottir, S.; Marteinsson, V.T.; Petursdottir, S.K.; Holst, O.; Kristjansson, J.K. Influence of sulfide and temperature on species composition and community structure of hot spring microbial mats. Appl. Environ. Microbiol. 2000, 66, 2835–2841. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.K.; Peacock, J.P.; Dodsworth, J.A.; Williams, A.J.; Thompson, D.B.; Dong, H.; Wu, G.; Hedlund, B.P. Sediment microbial communities in Great Boiling Spring are controlled by temperature and distinct from water communities. ISME J. 2013, 7, 718–729. [Google Scholar] [CrossRef]
- Merkel, A.Y.; Pimenov, N.V.; Rusanov, I.I.; Slobodkin, A.I.; Slobodkina, G.B.; Tarnovetckii, I.Y.; Frolov, E.N.; Dubin, A.V.; Perevalova, A.A.; Bonch-Osmolovskaya, E.A. Microbial diversity and autotrophic activity in Kamchatka hot springs. Extremophiles 2017, 21, 307–317. [Google Scholar] [CrossRef]
- Blank, C.E.; Cady, S.L.; Pace, N.R. Microbial Composition of Near-Boiling Silica-Depositing Thermal Springs throughout Yellowstone National Park. Appl. Environ. Microbiol. 2002, 68, 5123–5135. [Google Scholar] [CrossRef]
- Reysenbach, A.-L.; Banta, A.; Civello, S.; Daly, J.; Mitchel, K.; Ladonde, S.; Konhauserm, K.; Rodman, A.; Rusterholtz, K.; Takacs-Vesbach, C. Aquificales in Yellowstone National Park. In Geothermal Biology and Geochemistry in Yellowstone National Park; Montana State University Publications: Bozeman, MT, Canada, 2005; pp. 129–142. [Google Scholar]
- Hou, W.; Wang, S.; Dong, H.; Jiang, H.; Briggs, B.R.; Peacock, J.P.; Huang, Q.; Huang, L.; Wu, G.; Zhi, X.; et al. A Comprehensive Census of Microbial Diversity in Hot Springs of Tengchong, Yunnan Province China Using 16S rRNA Gene Pyrosequencing. PLoS ONE 2013, 8, e53350. [Google Scholar] [CrossRef]
- Huang, Q.; Jiang, H.; Briggs, B.R.; Wang, S.; Hou, W.; Li, G.; Wu, G.; Solis, R.; Arcilla, C.A.; Abrajano, T.; et al. Archaeal and bacterial diversity in acidic to circumneutral hot springs in the Philippines. FEMS Microbiol. Ecol. 2013, 85, 452–464. [Google Scholar] [CrossRef]
- Wang, S.; Hou, W.; Dong, H.; Jiang, H.; Huang, L.; Wu, G.; Zhang, C.; Song, Z.; Zhang, Y.; Ren, H.; et al. Control of Temperature on Microbial Community Structure in Hot Springs of the Tibetan Plateau. PLoS ONE 2013, 8, e62901. [Google Scholar] [CrossRef]
- Chernyh, N.A.; Mardanov, A.V.; Gumerov, V.M.; Miroshnichenko, M.L.; Lebedinsky, A.V.; Merkel, A.Y.; Crowe, D.; Pimenov, N.V.; Rusanov, I.I.; Ravin, N.V.; et al. Microbial life in Bourlyashchy, the hottest thermal pool of Uzon Caldera, Kamchatka. Extremophiles 2015, 19, 1157–1171. [Google Scholar] [CrossRef]
- Schubotz, F.; Hays, L.E.; Meyer-Dombard, D.R.; Gillespie, A.; Shock, E.L.; Summons, R.E. Stable isotope labeling confirms mixotrophic nature of streamer biofilm communities at alkaline hot springs. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Urschel, M.R.; Kubo, M.D.; Hoehler, T.M.; Peters, J.W.; Boyd, E.S. Carbon Source Preference in Chemosynthetic Hot Spring Communities. Appl. Environ. Microbiol. 2015, 81, 3834–3847. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hedlund, B.P.; Reysenbach, A.-L.; Huang, L.; Ong, J.C.; Liu, Z.; Dodsworth, J.A.; Ahmed, R.; Williams, A.J.; Briggs, B.R.; Liu, Y.; et al. Isolation of diverse members of the Aquificales from geothermal springs in Tengchong, China. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Inskeep, W. The YNP metagenome project: Environmental parameters responsible for microbial distribution in the Yellowstone geothermal ecosystem. Front. Microbiol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Colman, D.R.; Jay, Z.J.; Inskeep, W.P.; Jennings, R.D.; Maas, K.R.; Rusch, D.B.; Takacs-Vesbach, C.D. Novel, Deep-Branching Heterotrophic Bacterial Populations Recovered from Thermal Spring Metagenomes. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Jennings, R.D.; Moran, J.J.; Jay, Z.J.; Beam, J.P.; Whitmore, L.M.; Kozubal, M.A.; Kreuzer, H.W.; Inskeep, W.P. Integration of metagenomic and stable carbon isotope evidence reveals the extent and mechanisms of carbon dioxide fixation in high-temperature microbial communities. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Swingley, W.D.; Meyer-Dombard, D.R.; Shock, E.L.; Alsop, E.B.; Falenski, H.D.; Havig, J.R.; Raymond, J. Coordinating Environmental Genomics and Geochemistry Reveals Metabolic Transitions in a Hot Spring Ecosystem. PLoS ONE 2012, 7, e38108. [Google Scholar] [CrossRef]
- Rozanov, A.S.; Bryanskaya, A.V.; Ivanisenko, T.V.; Malup, T.K.; Peltek, S.E. Biodiversity of the microbial mat of the Garga hot spring. BMC Evol. Biol. 2017, 17, 254. [Google Scholar] [CrossRef]
- Dodsworth, J.A.; Ong, J.C.; Williams, A.J.; Dohnalkova, A.C.; Hedlund, B.P. Thermocrinis jamiesonii sp. nov., a thiosulfate-oxidizing, autotropic thermophile isolated from a geothermal spring. Int. J. Syst. Evol. Microbiol. 2015, 65, 4769–4775. [Google Scholar] [CrossRef]
- Aguiar, P.; Beveridge, T.J.; Reysenbach, A.-L. Sulfurihydrogenibium azorense, sp. nov., a thermophilic hydrogen-oxidizing microaerophile from terrestrial hot springs in the Azores. Int. J. Syst. Evol. Microbiol. 2004, 54, 33–39. [Google Scholar] [CrossRef]
- Ward, L.M.; Idei, A.; Nakagawa, M.; Ueno, Y.; Fischer, W.W.; McGlynn, S.E. Geochemical and Metagenomic Characterization of Jinata Onsen, a Proterozoic-Analog Hot Spring, Reveals Novel Microbial Diversity including Iron-Tolerant Phototrophs and Thermophilic Lithotrophs. Microbes Environ. 2019, 34, 278–292. [Google Scholar] [CrossRef] [PubMed]
- Moses, C.O.; Herman, J.S. Pyrite oxidation at circumneutral pH. Geochim. Cosmochim. Acta 1991, 55, 471–482. [Google Scholar] [CrossRef]
- Bosch, J.; Meckenstock, R.U. Rates and potential mechanism of anaerobic nitrate-dependent microbial pyrite oxidation. Biochem. Soc. Trans. 2012, 40, 1280–1283. [Google Scholar] [CrossRef]
- Percak-Dennett, E.; He, S.; Converse, B.; Konishi, H.; Xu, H.; Corcoran, A.; Noguera, D.; Chan, C.; Bhattacharyya, A.; Borch, T.; et al. Microbial acceleration of aerobic pyrite oxidation at circumneutral pH. Geobiology 2017, 15, 690–703. [Google Scholar] [CrossRef] [PubMed]
- Shelobolina, E.; Xu, H.; Konishi, H.; Kukkadapu, R.; Wu, T.; Blöthe, M.; Roden, E. Microbial Lithotrophic Oxidation of Structural Fe(II) in Biotite. Appl. Environ. Microbiol. 2012, 78, 5746–5752. [Google Scholar] [CrossRef]
- Benzine, J.; Shelobolina, E.; Xiong, M.Y.; Kennedy, D.W.; McKinley, J.P.; Lin, X.; Roden, E.E. Fe-phyllosilicate redox cycling organisms from a redox transition zone in Hanford 300 Area sediments. Front. Microbiol. 2013, 4. [Google Scholar] [CrossRef]
- Kochetkova, T.V.; Zayulina, K.S.; Zhigarkov, V.S.; Minaev, N.V.; Chichkov, B.N.; Novikov, A.A.; Toshchakov, S.V.; Elcheninov, A.G.; Kublanov, I.V. Tepidiforma bonchosmolovskayae gen. nov., sp. nov., a moderately thermophilic Chloroflexi bacterium from a Chukotka hot spring (Arctic, Russia), representing a novel class, Tepidiformia, which includes the previously uncultivated lineage OLB14. Int. J. Syst. Evol. Microbiol. 2020, 70, 1192–1202. [Google Scholar] [CrossRef]
- Stott, M.B.; Crowe, M.A.; Mountain, B.W.; Smirnova, A.V.; Hou, S.; Alam, M.; Dunfield, P.F. Isolation of novel bacteria, including a candidate division, from geothermal soils in New Zealand. Environ. Microbiol. 2008, 10, 2030–2041. [Google Scholar] [CrossRef]
- Lee, K.C.-Y.; Dunfield, P.F.; Morgan, X.C.; Crowe, M.A.; Houghton, K.M.; Vyssotski, M.; Ryan, J.L.J.; Lagutin, K.; McDonald, I.R.; Stott, M.B. Chthonomonas calidirosea gen. nov., sp. nov., an aerobic, pigmented, thermophilic micro-organism of a novel bacterial class, Chthonomonadetes classis nov., of the newly described phylum Armatimonadetes originally designated candidate division OP10. Int. J. Syst. Evol. Microbiol. 2011, 61, 2482–2490. [Google Scholar] [CrossRef]
- Tamaki, H.; Tanaka, Y.; Matsuzawa, H.; Muramatsu, M.; Meng, X.-Y.; Hanada, S.; Mori, K.; Kamagata, Y. Armatimonas rosea gen. nov., sp. nov., of a novel bacterial phylum, Armatimonadetes phyl. nov., formally called the candidate phylum OP10. Int. J. Syst. Evol. Microbiol. 2011, 61, 1442–1447. [Google Scholar] [CrossRef]
- Im, W.-T.; Hu, Z.-Y.; Kim, K.-H.; Rhee, S.-K.; Meng, H.; Lee, S.-T.; Quan, Z.-X. Description of Fimbriimonas ginsengisoli gen. nov., sp. nov. within the Fimbriimonadia class nov., of the phylum Armatimonadetes. Antonie Leeuwenhoek 2012, 102, 307–317. [Google Scholar] [CrossRef]
- Li, J.; Kudo, C.; Tonouchi, A. Capsulimonas corticalis gen. nov., sp. nov., an aerobic capsulated bacterium, of a novel bacterial order, Capsulimonadales ord. nov., of the class Armatimonadia of the phylum Armatimonadetes. Int. J. Syst. Evol. Microbiol. 2019, 69, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Dunfield, P.F.; Tamas, I.; Lee, K.C.; Morgan, X.C.; McDonald, I.R.; Stott, M.B. Electing a candidate: A speculative history of the bacterial phylum OP10. Environ. Microbiol. 2012, 14, 3069–3080. [Google Scholar] [CrossRef]
- Lin, X.; Kennedy, D.; Fredrickson, J.; Bjornstad, B.; Konopka, A. Vertical stratification of subsurface microbial community composition across geological formations at the Hanford Site. Environ. Microbiol. 2012, 14, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Thiel, V.; Garcia Costas, A.M.; Fortney, N.W.; Martinez, J.N.; Tank, M.; Roden, E.E.; Boyd, E.S.; Ward, D.M.; Hanada, S.; Bryant, D.A. “Candidatus Thermonerobacter thiotrophicus”, A Non-phototrophic Member of the Bacteroidetes/Chlorobi With Dissimilatory Sulfur Metabolism in Hot Spring Mat Communities. Front. Microbiol. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Hedlund, B.P.; Murugapiran, S.K.; Alba, T.W.; Levy, A.; Dodsworth, J.A.; Goertz, G.B.; Ivanova, N.; Woyke, T. Uncultivated thermophiles: Current status and spotlight on ‘Aigarchaeota’. Curr. Opin. Microbiol. 2015, 25, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Hiras, J.; Wu, Y.-W.; Eichorst, S.A.; Simmons, B.A.; Singer, S.W. Refining the phylum Chlorobi by resolving the phylogeny and metabolic potential of the representative of a deeply branching, uncultivated lineage. ISME J. 2016, 10, 833–845. [Google Scholar] [CrossRef]
- He, Y.; Li, M.; Perumal, V.; Feng, X.; Fang, J.; Xie, J.; Sievert, S.M.; Wang, F. Genomic and enzymatic evidence for acetogenesis among multiple lineages of the archaeal phylum Bathyarchaeota widespread in marine sediments. Nat. Microbiol. 2016, 1, 16035. [Google Scholar] [CrossRef]
- Evans, P.N.; Parks, D.H.; Chadwick, G.L.; Robbins, S.J.; Orphan, V.J.; Golding, S.D.; Tyson, G.W. Methane metabolism in the archaeal phylum Bathyarchaeota revealed by genome-centric metagenomics. Science 2015, 350, 434–438. [Google Scholar] [CrossRef]
- Lazar, C.S.; Baker, B.J.; Seitz, K.; Hyde, A.S.; Dick, G.J.; Hinrichs, K.-U.; Teske, A.P. Genomic evidence for distinct carbon substrate preferences and ecological niches of Bathyarchaeota in estuarine sediments. Environ. Microbiol. 2016, 18, 1200–1211. [Google Scholar] [CrossRef]
- McKay, L.J.; Hatzenpichler, R.; Inskeep, W.P.; Fields, M.W. Occurrence and expression of novel methyl-coenzyme M reductase gene (mcrA) variants in hot spring sediments. Sci. Rep. 2017, 7, 7252. [Google Scholar] [CrossRef] [PubMed]
- Winkel, M.; Mitzscherling, J.; Overduin, P.P.; Horn, F.; Winterfeld, M.; Rijkers, R.; Grigoriev, M.N.; Knoblauch, C.; Mangelsdorf, K.; Wagner, D.; et al. Anaerobic methanotrophic communities thrive in deep submarine permafrost. Sci. Rep. 2018, 8, 1291. [Google Scholar] [CrossRef]
- Takami, H.; Arai, W.; Takemoto, K.; Uchiyama, I.; Taniguchi, T. Functional Classification of Uncultured “Candidatus Caldiarchaeum subterraneum” Using the Maple System. PLoS ONE 2015, 10, e0132994. [Google Scholar] [CrossRef] [PubMed]
- Rinke, C.; Schwientek, P.; Sczyrba, A.; Ivanova, N.N.; Anderson, I.J.; Cheng, J.-F.; Darling, A.; Malfatti, S.; Swan, B.K.; Gies, E.A.; et al. Insights into the phylogeny and coding potential of microbial dark matter. Nature 2013, 499, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Menzel, P.; Gudbergsdóttir, S.R.; Rike, A.G.; Lin, L.; Zhang, Q.; Contursi, P.; Moracci, M.; Kristjansson, J.K.; Bolduc, B.; Gavrilov, S.; et al. Comparative Metagenomics of Eight Geographically Remote Terrestrial Hot Springs. Microb. Ecol. 2015, 70, 411–424. [Google Scholar] [CrossRef]

| Sample | T, °C | pH | Eh, mV | Coordinates | Number of OTUs Detected | Number of OTUs with More than 1% Abundance | Number of Genera Detected | Number of Genera with More than 1% Abundance | Number of Phyla Detected | Number of Phyla with More than 1% Abundance | Chao1 | ACE | Shannon | Simpson | InvSimpson |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3713.a | 54 | 7.3 | −320 | N65.806362/W173.39634 | 764 | 10 | 65 | 8 | 16 | 7 | 3420.05 ± 402.09 | 3389.21 ± 34.21 | 2.47 | 0.85 | 6.69 |
| 3735 | 60 | 8.0 | +210 | N64.73667/W172.84869 | 376 | 6 | 35 | 6 | 18 | 4 | 1556.17 ± 240.7 | 2034.59 ± 24.84 | 1.16 | 0.36 | 1.57 |
| 3705.1 | 66 | 7.0 | ND | N65.806419/W173.39557 | 1843 | 13 | 148 | 12 | 29 | 11 | 7801.98 ± 571.02 | 8676.04 ± 58.47 | 2.88 | 0.88 | 8.65 |
| 3726 | 62 | 6.8 | −400 | N65.805696 W173.39655 | 1116 | 15 | 377 | 18 | 28 | 11 | 2398.71 ± 156.76 | 2217.86 ± 28.81 | 4.06 | 0.94 | 16.71 |
| 3722 | 65 | 6.7 | −100 | N65.805433/W173.39929 | 860 | 17 | 168 | 26 | 34 | 12 | 4058.07 ± 554.57 | 3321.55 ± 36.41 | 4.08 | 0.96 | 22.37 |
| 3705.2 | 66 | 7.0 | −140/−200 | N65.806419/W173.39557 | 1027 | 11 | 147 | 9 | 32 | 9 | 3688.38 ± 353.27 | 3478.97 ± 37.97 | 2.27 | 0.66 | 2.94 |
| 3750 | 67 | 8.0 | −300 | N64.736091/W172.84917 | 705 | 10 | 169 | 10 | 25 | 9 | 2568.96 ± 312.22 | 2122.26 ± 29.01 | 2.43 | 0.81 | 5.31 |
| 3723 | 65 | 6.7 | −70 | N65.80555/W173.39721 | 1090 | 17 | 199 | 17 | 36 | 12 | 3590.43 ± 307.57 | 3679.47 ± 40.28 | 2.97 | 0.81 | 5.39 |
| 3701 | 75 | 6.8 | −120/−20 | N65.806401/W173.39534 | 503 | 20 | 147 | 17 | 39 | 15 | 1710.71 ± 219.36 | 1790.2 ± 27.12 | 3.41 | 0.93 | 15.06 |
| 3718 | 65 | 6.8 | −326 | N65.805704/W173.40537 | 729 | 3 | 85 | 3 | 22 | 3 | 3149.37 ± 360.19 | 3513.99 ± 34.05 | 1.19 | 0.53 | 2.13 |
| 3717 | 69 | 6.8 | −326 | N65.805566/W173.40547 | 878 | 6 | 129 | 5 | 29 | 6 | 3188.73 ± 305.45 | 3526.48 ± 37.68 | 1.15 | 0.38 | 1.61 |
| 3713.b | 76 | 6.7 | −300 | N65.806362/W173.39634 | 473 | 8 | 42 | 5 | 20 | 5 | 1991.07 ± 279.21 | 2166.89 ± 27.29 | 1.87 | 0.68 | 3.15 |
| 3704 | 80 | 6.7 | −278 | N65.806329/W173.39563 | 90 | 3 | 28 | 3 | 14 | 3 | 275.77 ± 68.09 | 311.17 ± 4.61 | 0.4 | 0.15 | 1.18 |
| 3712 | 81 | 6.4 | −305 | N65.806335/W173.39644 | 1109 | 4 | 115 | 4 | 26 | 3 | 4077.9 ± 346.39 | 4632.53 ± 44.21 | 1.4 | 0.6 | 2.51 |
| 3745 | 72 | 8.1 | −250/−50 | N64.73656/W172.8518 | 649 | 15 | 56 | 13 | 17 | 12 | 4526.68 ± 741.86 | 3924.76 ± 25.81 | 2.89 | 0.89 | 8.87 |
| 3724 | 76 | 6.7 | −96 | N65.805774/W173.397 | 446 | 12 | 77 | 11 | 25 | 9 | 1375.96 ± 176.55 | 1434.89 ± 24.48 | 2.71 | 0.88 | 8.05 |
| 3748 | 77 | 8.0 | −310 | N64.73644/W172.85123 | 401 | 7 | 87 | 7 | 20 | 7 | 1148.64 ± 147.97 | 1089.69 ± 20.16 | 1.93 | 0.78 | 4.62 |
| 3714 | 79 | 6.7 | −316 | N65.806362/W173.39634 | 854 | 16 | 69 | 12 | 25 | 11 | 3683.68 ± 396.69 | 4252.5 ± 41.12 | 2.86 | 0.92 | 12.05 |
| 3710 | 84 | 7.0 | −40 | N65.806271/W173.39639 | 562 | 14 | 70 | 16 | 28 | 15 | 2533.27 ± 354.46 | 3666.54 ± 30.06 | 3.25 | 0.92 | 12.08 |
| 3730 | 94 | 6.9 | −360 | N65.805844/W173.39485 | 403 | 8 | 148 | 9 | 23 | 7 | 1964 ± 433.64 | 1511.87 ± 24.28 | 2.83 | 0.81 | 5.4 |
| 3751a | 75 | 7.5 | +30/+80 | N64.435798/W172.52126 | 709 | 18 | 135 | 17 | 19 | 10 | 4378.12 ± 704.19 | 3836.49 ± 34.84 | 3.77 | 0.94 | 15.39 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kochetkova, T.V.; Toshchakov, S.V.; Zayulina, K.S.; Elcheninov, A.G.; Zavarzina, D.G.; Lavrushin, V.Y.; Bonch-Osmolovskaya, E.A.; Kublanov, I.V. Hot in Cold: Microbial Life in the Hottest Springs in Permafrost. Microorganisms 2020, 8, 1308. https://doi.org/10.3390/microorganisms8091308
Kochetkova TV, Toshchakov SV, Zayulina KS, Elcheninov AG, Zavarzina DG, Lavrushin VY, Bonch-Osmolovskaya EA, Kublanov IV. Hot in Cold: Microbial Life in the Hottest Springs in Permafrost. Microorganisms. 2020; 8(9):1308. https://doi.org/10.3390/microorganisms8091308
Chicago/Turabian StyleKochetkova, Tatiana V., Stepan V. Toshchakov, Kseniya S. Zayulina, Alexander G. Elcheninov, Daria G. Zavarzina, Vasiliy Yu. Lavrushin, Elizaveta A. Bonch-Osmolovskaya, and Ilya V. Kublanov. 2020. "Hot in Cold: Microbial Life in the Hottest Springs in Permafrost" Microorganisms 8, no. 9: 1308. https://doi.org/10.3390/microorganisms8091308
APA StyleKochetkova, T. V., Toshchakov, S. V., Zayulina, K. S., Elcheninov, A. G., Zavarzina, D. G., Lavrushin, V. Y., Bonch-Osmolovskaya, E. A., & Kublanov, I. V. (2020). Hot in Cold: Microbial Life in the Hottest Springs in Permafrost. Microorganisms, 8(9), 1308. https://doi.org/10.3390/microorganisms8091308






