Evaluation of Multifarious Plant Growth Promoting Trials of Yeast Isolated from the Soil of Assam Tea (Camellia sinensis var. assamica) Plantations in Northern Thailand
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Yeast Isolation
2.3. Yeast Identification
2.4. Determination of Indole-3-Acetic Acid (IAA) Production
2.5. Determination of Siderophore Production
2.6. Determination of Ammonia Production
2.7. Determination of Mineral Solubilization Ability
2.8. Determination of Extracellular Enzyme Production
3. Results
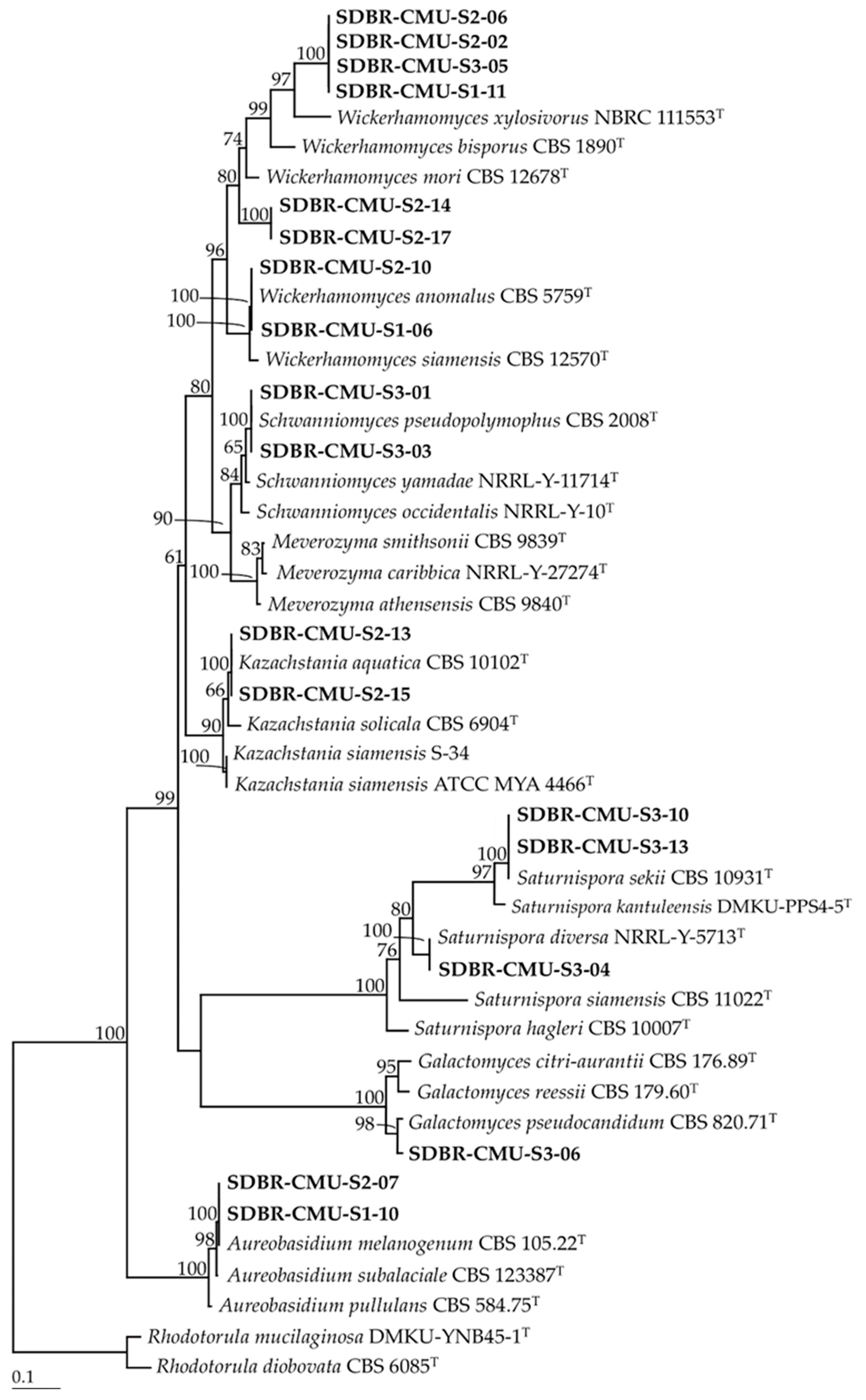
3.1. Yeast Isolation and Identification
3.2. Determination of IAA Production
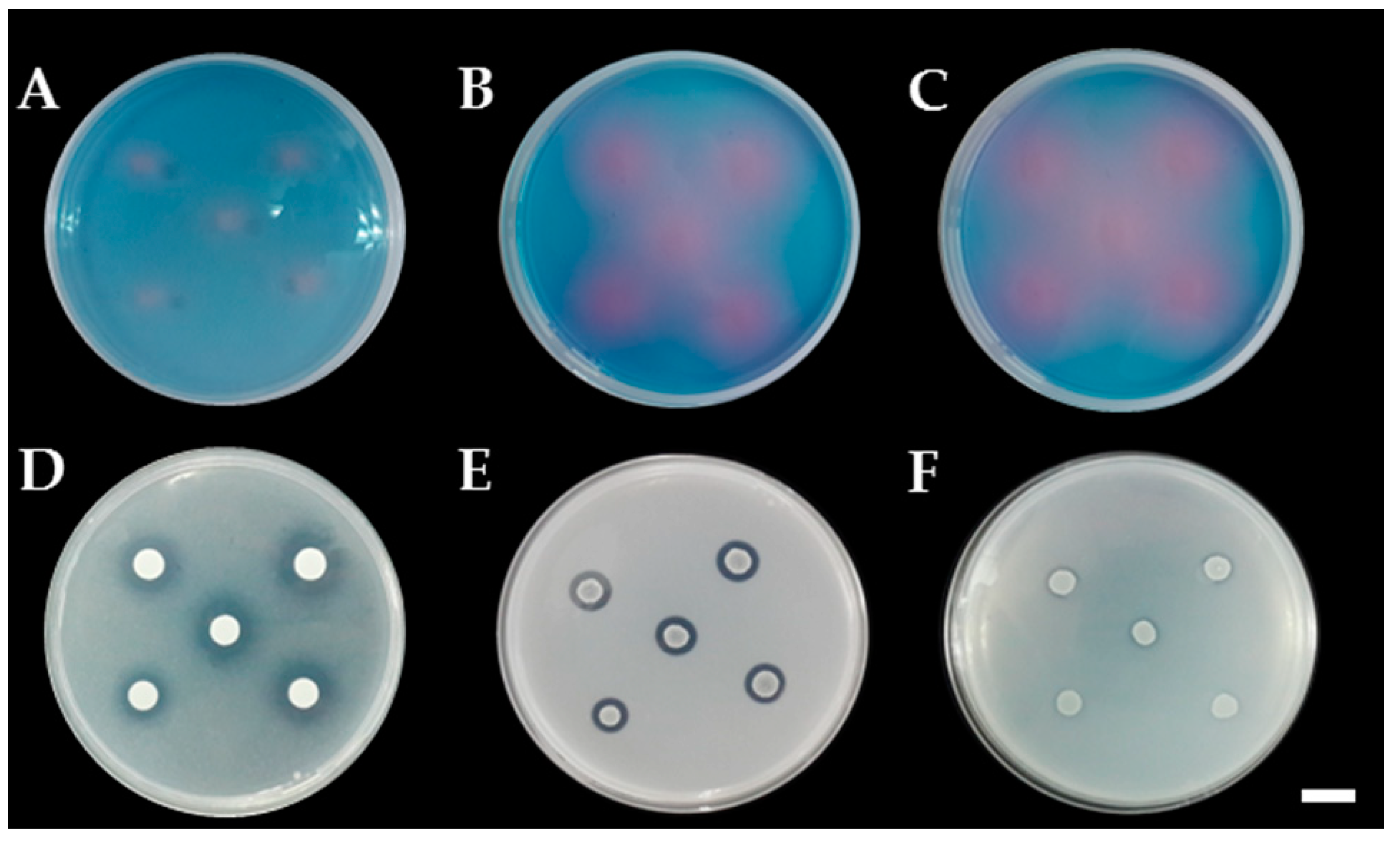
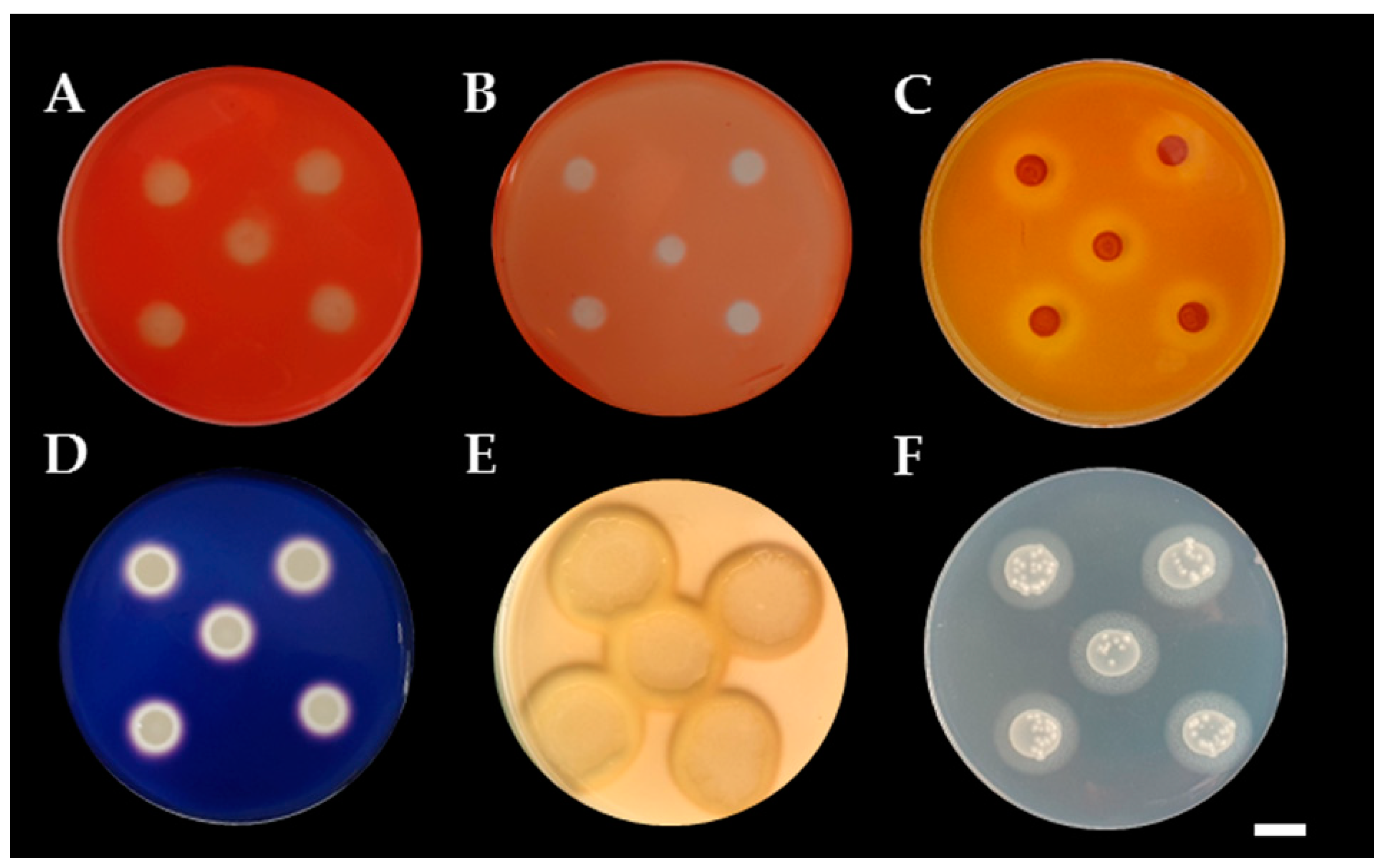
3.3. Determination of Siderophore and Ammonia Production
3.4. Determination of Mineral Solubilization Ability
3.5. Determination of Extracellular Enzyme Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wight, W.; Baruwa, P.K. What is tea? Nature 1957, 179, 506–507. [Google Scholar] [CrossRef]
- Ming, T.L. Monograph of the Genus Camellia; Yunnan Science and Technology Press: Kunming, China, 2000; 352p. [Google Scholar]
- Chen, J.; Wang, P.S.; Xia, Y.M.; Xu, M.; Pei, S.J. Genetic diversity and differentiation of Camellia sinensis L. (cultivated tea) and its wild relatives in Yunnan province of China, revealed by morphology, biochemistry and allozyme studies. Genet. Resour. Crop. Evol. 2005, 52, 41–52. [Google Scholar] [CrossRef]
- Korsamphan, C.; Boonma, W.; Sringarm, K. Diversity of local tea on highland of northern Thailand. J. Agric. 2010, 26, 93–99. [Google Scholar]
- Khanongnuch, C.; Unban, K.; Kanpeiengjai, A.; Saenjum, C. Recent research advances and ethno-botanical history of miang, a traditional fermented tea (Camellia sinensis var. assamica) of northern Thailand. J. Ethn. Foods 2017, 4, 135–144. [Google Scholar] [CrossRef]
- Kawakami, M.; Chairote, G.; Kobayashi, A. Flavor constituents of pickled tea, miang, in Thailand. Agric. Biol. Chem. 1987, 51, 1683–1687. [Google Scholar]
- Phromrukachat, S.; Tiengburanatum, N.; Meechui, J. Assessment of active ingredients in pickled tea. Asian J. Food Agro-Ind. 2010, 3, 312–318. [Google Scholar]
- Lin, W.; Lin, M.; Zhou, H.; Wu, H.; Li, Z.; Lin, W. The effects of chemical and organic fertilizer usage on rhizosphere soil in tea orchards. PLoS ONE 2019, 14, e0217018. [Google Scholar] [CrossRef]
- Gao, G. Experiment studies on the hygienics of AsBaCdPb in tea. Chin. J. Food. Hygiene. 2001, 13, 12–14. [Google Scholar]
- Li, Y.; Li, Z.; Arafat, Y.; Lin, W.; Jiang, Y.; Weng, B.; Lin, W. Characterizing rhizosphere microbial communities in long-term monoculture tea orchards by fatty acid profiles and substrate utilization. Eur. J. Soil Biol. 2017, 81, 48–54. [Google Scholar] [CrossRef]
- Maghanga, J.K.; Kituyi, J.L.; Kisinyo, P.O.; Ng’etich, W.K. Impact of nitrogen fertilizer applications on surface water nitrate levels within a kenyan tea plantation. J. Chem. 2012, 1, 196516. [Google Scholar] [CrossRef]
- Ruan, J.; Gerendas, J.; Haerdter, R.; Sattelmacher, B. Effect of root zone pH and form and concentration of nitrogen on accumulation of quality-related components in green tea. J. Sci. Food Agric. 2007, 87, 1505–1516. [Google Scholar] [CrossRef]
- Wang, Z.; Geng, Y.; Liang, T. Optimization of reduced chemical fertilizer use in tea gardens based on the assessment of related environmental and economic benefits. Sci. Total. Environ. 2020, 713, 136439. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Viveros, O.; Jorquera, M.A.; Crowlet, D.E.; Gajardo, G.; Mora, M.L. Mechanisms and practical considerations involved in plant growth promotion by rhizobacteria. J. Soil Sci. Plant Nutr. 2010, 10, 239–319. [Google Scholar] [CrossRef]
- Arora, N.K. Agricultural sustainability and food security. Environ. Sustain. 2018, 1, 217–219. [Google Scholar] [CrossRef]
- Tamenkovic, S.; Beskoski, V.; Karabegovic, I.; Lazic, M.; Nikolic, N. Microbial fertilizers: A comprehensive review of current findings and future perspectives. Span. J. Agric. Res. 2018, 16, e09R01. [Google Scholar] [CrossRef]
- Naik, K.; Mishra, S.; Srichandan, H.; Singh, P.K.; Sarangi, K. Plant growth promoting microbes: Potential link to sustainable agriculture and environment. Biocatal. Agric. Biotechnol. 2019, 21, 101326. [Google Scholar] [CrossRef]
- Mishra, J.; Singh, R.; Arora, N.K. Plant growth-promoting microbes: Diverse roles in agriculture and environmental sustainability. In Probiotics and Plant Health; Kumar, V., Kumar, M., Sharma, S., Prasad, R., Eds.; Springer: Singapore, 2017; pp. 71–111. [Google Scholar]
- Abhilash, P.C.; Dubey, R.K.; Tripathi, V.; Gupta, V.K.; Singh, H.B. Plant growth-promoting microorganisms for environmental sustainability. Trends Biotechnol. 2016, 34, 847–850. [Google Scholar] [CrossRef]
- Rilling, J.; Acuña, J.; Nannipieri, P.; Cassan, F.; Maruyama, F.; Jorquera, M. Current opinion and perspectives on the methods for tracking and monitoring plant growth-promoting bacteria. Soil Biol. Biochem. 2019, 130, 205–219. [Google Scholar] [CrossRef]
- Wagi, S.; Ahmed, A. Phyllospheric plant growth promoting bacteria. J. Bacteriol. Mycol. 2017, 5, 215–216. [Google Scholar]
- Gupta, H.; Saini, R.V.; Pagadala, V.; Kumar, N.; Sharma, D.K.; Saini, A.K. Analysis of plant growth promoting potential of endophytes isolated from Echinacea purpurea and Lonicera japonica. J. Soil. Sci Plant Nutr. 2016, 16, 588–597. [Google Scholar] [CrossRef]
- Ignatova, L.V.; Brazhnikova, Y.V.; Berzhanova, R.Z.; Mukasheva, T.D. Plant growth-promoting and antifungal activity of yeasts from dark chestnut soil. Microbiol. Res. 2015, 175, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Yurkov, A.M. Yeasts of the soil - obscure but precious. Yeast 2018, 35, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Botha, A. The importance and ecology of yeasts in soil. Soil Biol. Biochem. 2011, 43, 1–8. [Google Scholar] [CrossRef]
- Fu, S.F.; Sun, P.F.; Lu, H.Y.; Wei, J.Y.; Xiao, H.S.; Fang, W.T.; Cheng, B.Y.; Chou, J.Y. Plant growth-promoting traits of yeasts isolated from the phyllosphere and rhizosphere of Drosera spatulata Lab. Fungal Biol. 2016, 120, 433–448. [Google Scholar] [CrossRef]
- Limtong, S.; Koowadjanakul, N. Yeasts from phylloplane and their capability to produce indole-3-acetic acid. World J. Microbiol. Biotechnol. 2012, 28, 3323–3335. [Google Scholar] [CrossRef]
- Jaiboon, K.; Lertwattanasakul, N.; Limtong, P.; Limtong, S. Yeasts from peat in a tropical peat swamp forest in Thailand and their ability to produce ethanol, indole-3-acetic acid and extracellular enzymes. Mycol. Prog. 2016, 15, 755–770. [Google Scholar] [CrossRef]
- Amprayn, K.; Rose, M.Y.; Kecskes, M.; Pereg, L.; Nguyen, H.T.; Kennedy, I.R. Plant growth promoting characteristics of soil yeast (Candida tropicalis HY) and its effectiveness for promoting rice growth. Appl. Soil Ecol. 2012, 61, 295–299. [Google Scholar] [CrossRef]
- Stielow, J.B.; Levesque, C.A.; Seifert, K.A.; Meyer, W.; Lriny, L.; Smits, D.; Renfurm, R.; Verkely, G.J.M.; Groenewald, M.; Chaduli, D.; et al. One fungus, which genes? Development and assessment of universal primers for potential secondary fungal DNA barcodes. Persoonia 2015, 35, 242–263. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML-VI-HPC: Maximum likelihood based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 2006, 22, 2688–2690. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence intervals on phylogenetics: An approach using bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Kumla, J.; Suwannarach, N.; Bussaban, B.; Matsui, K.; Lumyong, S. Indole-3-acetic acid production, solubilization of insoluble metal minerals and metal tolerance of some sclerodermatoid fungi collected from northern Thailand. Ann. Microbial. 2014, 64, 707–720. [Google Scholar] [CrossRef]
- Louden, B.C.; Haarmann, D.; Lynne, A.M. Use of blue agar CAS Assay for siderophore detection. J. Microbiol. Biol. Educ. 2011, 12, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Cappuccino, J.G.; Sherman, N. Microbiology: A Laboratory Manual, 6th ed.; Pearson Education: London, UK, 2002. [Google Scholar]
- Fomina, M.A.; Alexander, I.J.; Colpaert, J.V.; Gadd, G.M. Solubilization of toxic metal minerals and metal tolerance of mycorrhizal fungi. Soil Biol. Biochem. 2005, 37, 851–866. [Google Scholar] [CrossRef]
- Nosrati, R.; Owlia, P.; Saderi, H.; Rasooli, I.; Malboobi, M.A. Phosphate solubilization characteristics of efficient nitrogen fixing soil Azotobacter strains. Iran J. Microbiol. 2014, 6, 285–295. [Google Scholar]
- Hankin, L.; Anagnostakis, S.L. The use of solid media for the detection of enzyme production by fungi. Mycologia 1975, 67, 567–607. [Google Scholar] [CrossRef]
- Kasana, R.C.; Salwan, R.; Dhar, H.; Dutt, S.; Gulati, A. A rapid and easy method for the detection of microbial cellulases on agar plates using Gram’s iodine. Curr. Microbiol. 2008, 57, 503–507. [Google Scholar] [CrossRef]
- Lopes, F.; Motta, F.; Andrade, C.C.P.; Rodrigues, M.I.; Maugeri-filho, F. Thermo-stable xylanases from non-conventional yeasts. J. Microbiol. Biochem. Technol. 2011, 3, 36–42. [Google Scholar]
- Otero, D.M.; Cadaval, C.L.; Teixeira, L.M.; Rosa, C.A.; Sanzo, A.V.L.; Kalil, S.J. Screening of yeasts capable of producing cellulase-free xylanase. Afr. J. Biotechnol. 2015, 14, 1961–1969. [Google Scholar] [CrossRef]
- Haile, M.; Kang, W.H. Isolation, identification, and characterization of pectinolytic yeasts for starter culture in coffee fermentation. Microorganism 2019, 7, 401. [Google Scholar] [CrossRef]
- Yalçin, T.; Çorbacı, C. Isolation and characterization of amylase producing yeasts and improvement of amylase production. Turk. J. Biochem. 2013, 38, 101–108. [Google Scholar] [CrossRef]
- Charoenchai, C.; Fleet, G.H.; Henschke, P.A.; Todd, B.E.N.T. Screening of non-Saccharomyces wine yeasts for the presence of extracellular hydrolytic enzymes. Aust. J. Grape Wine Res. 1997, 3, 2–8. [Google Scholar] [CrossRef]
- Elegado, F.; Legaspi, C.L.; Paet, J.M.; Querubin, F.; Tolentino, J.E.; Vilela, J.; Paguio, A.; Maloles, J.; Zarate, J. Screening, identification and optimization of extracellular lipase production of yeast (Cryptococus flavescens) isolated from a tree canopy fern in the Mount Makiling Forest Reserve, Philippines. AIP Conf. Proc. 2019, 2155, 020029. [Google Scholar]
- Moreira, G.A.M.; do Vale, H.M.M. Occurrence of yeast species in soils under native and modified vegetation in an iron mining area. Rev. Bras. Cienc. Solo 2018, 42, e0170375. [Google Scholar] [CrossRef]
- Mohamed, H.M.; El-Homosy, R.F.; Abd-Ellatef, A.H.; Salh, F.M.; Hussein, M.Y. Identification of yeast strains isolated from agricultural soils for releasing potassium-bearing minerals. Geomicrobiol. J. 2017, 34, 261–266. [Google Scholar] [CrossRef]
- Diaz, P.E.; Aranda, C.; Martinez, O.; Godoy, R.; Gonzales, A.; Valenzuela, E. Characterization of yeast in hapludands soil with biotechnological potential. J. Soil Sci. Plant Nutr. 2017, 17, 948–965. [Google Scholar] [CrossRef]
- Liu, X.Z.; Wang, Q.M.; Göker, M.; Groenewald, M.; Kachalkin, A.V.; Lumbsch, H.T.; Bai, F.Y. Towards an integrated phylogenetic classification of the Tremellomycetes. Stud. Mycol. 2015, 81, 85–147. [Google Scholar] [CrossRef]
- Vadkertiová, R.; Dudášová, H.; Balaščáková, M. Yeasts in agricultural and managed soils. In Yeasts in Natural Ecosystems: Ecology; Buzzini, P., Lachance, M.A., Yurkov, A.M., Eds.; Springer: Heidelberg, Germany, 2017; pp. 117–144. [Google Scholar]
- Yurkov, A.M. Yeasts in forest soils. In Yeasts in Natural Ecosystems: Ecology; Buzzini, P., Lachance, M.A., Yurkov, A.M., Eds.; Springer: Heidelberg, Germany, 2017; pp. 87–116. [Google Scholar]
- Slavikova, E.; Vandkertiova, R. The diversity of yeast in the agricultural soil. J. Basic Microbiol. 2003, 43, 430–436. [Google Scholar] [CrossRef]
- Birkhofer, K.; Schöning, I.; Alt, F.; Herold, N.; Klarner, B.; Maraun, M.; Schrumpf, M. General relationships between abiotic soil properties and soil biota across spatial scales and different land-use types. PLoS ONE 2012, 7, e43292. [Google Scholar] [CrossRef]
- Glushakova, A.; Maksimova1, I.; Morozova, A.; Kachalkin, A. Distribution features of yeasts in soils of South Vietnam (case study of the biogeocenoses of the National Park Cát Tiên). IOP Conf. Ser. Earth Environ. Sci. 2019, 368, 012012. [Google Scholar] [CrossRef]
- van Nieuwenhuijzen, E.J.; Houbraken, J.A.; Meijer, M.; Adan, O.C.; Samson, R.A. Aureobasidium melanogenum: A native of dark biofinishes on oil treated wood. Anton Leeuw. 2016, 109, 661–683. [Google Scholar] [CrossRef] [PubMed]
- Haq, I.; Nawaz, A.; Manzoor, Z.; Rehman, A.; Mukhtar, H.; Aftab, M.; Arshad, Y. A new multi-stress resistant Wickerhamomyces anomalus: Isolation, identification and bioethanol fermentation potential. Rev. Mex. Ing. Quim. 2019, 18, 841–849. [Google Scholar] [CrossRef]
- Quint, M.; Gray, W.M. Auxin signaling. Curr. Opin. Plant Biol. 2006, 9, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y. Auxin biosynthesis and its role in plant development. Annu. Rev. Plant Biol. 2010, 61, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Spaepen, S.; Vanderleyden, J. Auxin and plant-microbe interactions. Cold Spring Harb. Perspect. Biol. 2011, 3, a001438. [Google Scholar] [CrossRef]
- Spaepen, S.; Vanderleyden, J.; Remans, R. Indole-3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol. Rev. 2007, 31, 425–448. [Google Scholar] [CrossRef]
- Niemi, K.; Vuorinen, T.; Ernstsen, A.; Haggman, H. Ectomycorrhizal fungi and exogenous auxins influence root and mycorrhiza formation of Scots pine hypocotyl cuttings in vitro. Tree Physiol. 2002, 22, 1231–1239. [Google Scholar] [CrossRef]
- Kravchenko, L.V.; Azarova, T.S.; Makarova, N.M.; Tikhonovich, I.A. The effect of tryptophan present in plant root exudates on the phytostimulating activity of rhizobacteria. Microbiology 2004, 73, 156–158. [Google Scholar] [CrossRef]
- Sun, P.F.; Fang, W.T.; Shin, L.Y.; Wei, J.Y.; Fu, S.F.; Chou, J.Y. Indole-3-acetic acid-producing yeasts in the phyllosphere of the carnivorous plant Drosera indica L. PLoS ONE 2014, 9, 1–12. [Google Scholar] [CrossRef]
- Nutaratat, P.; Srisuk, N.; Arunrattiyakorn, P.; Limtong, S. Plant growth-promoting traits of epiphytic and endophytic yeasts isolated from rice and sugar cane leaves in Thailand. Fungal Biol. 2014, 118, 683–694. [Google Scholar] [CrossRef]
- Tsavkelova, E.A.; Cherdyntseva, T.A.; Botina, S.G.; Netrusov, A.I. Bacteria associated with orchid roots and microbial production of auxin. Microbiol. Res. 2007, 162, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Vitorino, L.C.; Silva, F.G.; Soares, M.A.; Souchie, E.L.; Costa, A.C.; Lima, W.C. Solubilization of calcium and iron phosphate and in vitro production of indole acetic acid by endophytic isolates of Hyptis marrubioides Epling (Lamiaceae). Int. Res. J. Biotechnol. 2012, 3, 47–54. [Google Scholar]
- Chutima, R.; Lumyong, S. Production of indole-3-acetic acid by Thai native orchid-associated fungi. Symbiosis 2014, 56, 35–44. [Google Scholar] [CrossRef]
- Limtong, S.; Kaewwichian, R.; Yongmanitchai, W.; Kawasaki, H. Diversity of culturable yeasts in phylloplane of sugarcane in Thailand and their capability to produce indole-3-acetic acid. World. J. Microbiol. Biotechnol. 2014, 30, 1785–1796. [Google Scholar] [CrossRef]
- Nassar, A.H.; El-Tarabily, K.A.; Sivasithamparam, K. Promotion of plant growth by an auxin-producing isolate of the yeast Williopsis saturnus endophytic in maize (Zea mays L.) roots. Biol. Fertil. Soils 2005, 42, 97–108. [Google Scholar] [CrossRef]
- Ahmed, E.; Holmstrom, S.J.M. Siderophores in environmental research: Roles and applications. Microb. Biotechnol. 2014, 7, 196–208. [Google Scholar] [CrossRef]
- D’Onofrio, A.; Crawford, J.M.; Stewart, E.J.; Witt, K.; Gavrish, E.; Epstein, S.; Clardy, J.; Lewis, K. Siderophores from neighboring organisms promote the growth of uncultured bacteria. Chem. Biol. 2010, 17, 254–264. [Google Scholar]
- Saha, R.; Saha, N.; Donofrio, R.S.; Bestervelt, L.L. Microbial siderophores: A mini review. J. Basic Microbiol. 2013, 53, 301–317. [Google Scholar] [CrossRef]
- Thanh, V.N.; van Dyk, M.S.; Wingfield, M.J. Debaryomyces mycophilus sp. nov., a siderophore-dependent yeast isolated from woodlice. FEMS Yeast Res. 2002, 2, 415–427. [Google Scholar]
- Mpanga, I.K.; Nkebiwe, P.M.; Kuhlmann, M.; Cozzolino, V.; Piccolo, A.; Geistlinger, J.; Berger, N.; Ludewig, U.; Neumann, G. The form of N supply determines plant growth promotion by P-solubilizing microorganisms in maize. Microorganisms 2019, 7, 38. [Google Scholar] [CrossRef]
- Bartelme, R.P.; Oyserman, B.O.; Blom, J.E.; Sepulveda-Villet, O.J.; Newton, R.J. Stripping away the soil: Plant growth promoting microbiology opportunities in aquaponics. Front. Microbiol. 2018, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–645. [Google Scholar] [CrossRef] [PubMed]
- Mapelli, F.; Marasco, R.; Balloi, A.; Rolli, E.; Cappitelli, F.; Daffonchio, D.; Borin, S. Mineral-microbe interactions: Biotechnological potential of bioweathering. J. Biotechnol. 2012, 157, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Alori, E.T.; Glick, B.R.; Babalola, O.O. Microbial phosphorus solubilization and its potential for use in sustainable agriculture. Front. Microbiol. 2017, 8, 971. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.M.; Metwally, A.K. Effect of combined inoculation of Rhizobium with soil yeasts on nodulation, growth and yield of common bean (Phaseolus vulgaris L.) under field condition. Am. J. Plant Nutr. Fert. Technol. 2014, 4, 1–10. [Google Scholar] [CrossRef]
- Into, P.; Khunnamwong, P.; Jindamoragot, S.; Am-in, S.; Intanoo, W.; Limtong, S. Yeast associated with rice phylloplane and their contribution to control of rice sheath blight disease. Microorganisms 2020, 8, 362. [Google Scholar] [CrossRef]
- Gizaw, B.; Tsegay, Z.; Tefera, G.; Aynalem, E. Phosphate solubilizing yeast isolated and characterized from teff rhizosphere soil collected from gojam; Ethiopia. J. Bacteriol. Mycol. Open Access 2017, 5, 218–223. [Google Scholar] [CrossRef][Green Version]
- Mestre, M.C.; Fontenla, S.; Bruzone, M.C.; Fernández, N.V.; Dames, J. Detection of plant growth enhancing features in psychrotolerant yeasts from Patagonia (Argentina). J. Basic Microbiol. 2016, 56, 1098–1106. [Google Scholar] [CrossRef]
- Fernandez-San, M.A.; Farran, I.; Larraya, L.; Ancin, M.; Arregui, L.M.; Veramendi, J. Plant growth-promoting traits of yeasts isolated from Spanish vineyards: Benefits for seedling development. Microbiol. Res. 2020, 237, 1–16. [Google Scholar] [CrossRef]
- Jadhv, H.P.; Sayyed, R.Z. Hydrolytic enzymes of rhizospheric microbes in crop protection. Cell Sci. Rep. 2016, 3, 135–136. [Google Scholar]
- Whipps, J.M. Microbial interactions and biocontrol in the rhizosphere. J. Exp. Bot. 2001, 52, 487–511. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, M.; Villarreal, P.; Barahona, S.; Alcaíno, J.; Cifuentes, V.; Baeza, M. Screening and characterization of amylase and cellulase activities in psychrotolerant yeasts. BMC. Microbiol. 2016, 16, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Giese, E.C.; Dussán, K.J.; Pierozzi, M.; Chandel, A.K.; Pagnocca, F.C.; Da Silva, S.S. Cellulase production by Trichosporon laibachii. Orbital Electron. J. Chem. 2017, 4, 271–278. [Google Scholar] [CrossRef]
- Adelabu, B.; Kareem, S.O.; Adeogun, A.I.; Wakil, S.M. Optimization of cellulase enzyme from sorghum straw by yeasts isolated from plant feeding-termite Zonocerus variegatus. Food. Biosci. 2019, 7, 81–101. [Google Scholar]
- Escribano, R.; González-Arenzana, L.; Garijo, P.; Berlanas, C.; López-Alfaro, I.; López, R.; Gutie’rrez, A.R.; Santamaría, P. Screening of enzymatic activities within different enological non-Saccharomyces yeasts. J. Food Sci. Technol. 2017, 54, 1555–1564. [Google Scholar] [CrossRef] [PubMed]
- Gomes, F.C.; Safar, S.V.; Marques, A.R.; Medeiros, A.O.; Santos, A.R.; Carvalho, C.; Lachance, M.A.; Sampaio, J.P.; Rosa, C.A. The diversity and extracellular enzymatic activities of yeasts isolated from water tanks of Vriesea minarum, an endangered bromeliad species in Brazil, and the description of Occultifur brasiliensis f.a., sp. nov. Anton. Leeuw. 2015, 107, 597–611. [Google Scholar] [CrossRef]
- Moller, L.; Kessler, K.D.; Steyn, A.; Valentine, A.J. The role of Cryptococcus laurentii and mycorrhizal fungi in the nutritional physiology of Lupinus angustifolius L. hosting N2-fixing nodules. Plant Soil 2016, 409, 345–360. [Google Scholar] [CrossRef]
- Yilmaz-Semerci, S.; Demirel, G.; Tastekin, A. Wickerhamomyces anomalus blood stream infection in a term newborn with pneumonia. Turk. J. Pediatr. Dis. 2017, 59, 349–351. [Google Scholar] [CrossRef]
- Molina-Leyva, A.; Ruiz-Carrascosa, J.C.; Leyva-Garcia, A.; Husein-Elanmed, H. Cutaneous Cryptococcus laurentii infection in an immunocompetent child. Int. J. Infect. Dis. 2013, 17, 1232–1233. [Google Scholar] [CrossRef]
- Vilchez, J.I.; Navas, A.; Gonzalez-Lopez, J.; Acros, S.C.; Manzanera, M. Biosafety test for plant growth-promoting bacteria: Proposed environmental and human safety index (EHSI) protocol. Front. Microbiol. 2015, 6, 1514. [Google Scholar] [CrossRef]
- Sundh, I.; Hökeberg, M.; Levenfors, J.J.; Nilsson, A.I. Safety assessment of biocontrol and plant growth-promoting pseudomonads useful in crop production. Ann. Appl. Biol. 2011, 159, 291–301. [Google Scholar] [CrossRef]
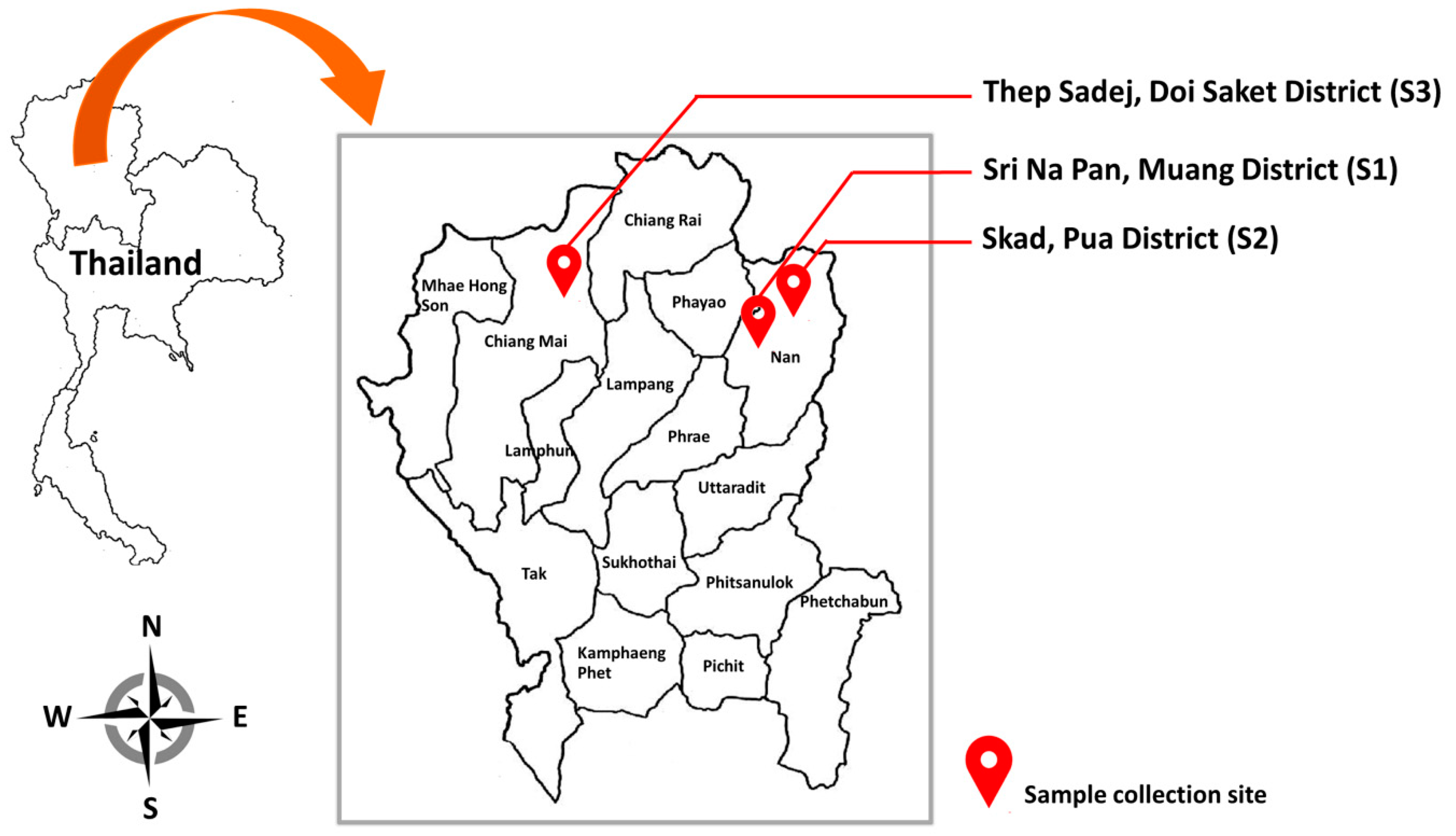

| Site | Location | Soil Type | pH Value | EC (µS/cm) |
|---|---|---|---|---|
| S1 | Skad, Pua District, Nan Province | Sandy clay loam | 6.13 | 0.048 |
| S2 | Sri Na Pan, Muang District, Nan Province | Sandy clay loam | 6.06 | 0.046 |
| S3 | Thep Sadej, Doi Saket District, Chiang Mai Province | Sandy clay loam | 6.54 | 0.049 |
| Site | Strain SDBR-CMU | GenBank Number | Closeted Type Species/Similarity (%) | Identification |
|---|---|---|---|---|
| S1 | S1-01 | MT613405 | Trichosporon asahii CBS 2479/100 | T. asahii |
| S1-02 | MT613406 | Papiliotrema laurentii CBS 139 99.87 | P. laurentii | |
| S1-03 | MT613407 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae | |
| S1-04 | MT623567 | Trichosporon asahii CBS 2479/100 | T. asahii | |
| S1-05 | MT623565 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae | |
| S1-06 | MT613408 | Wickerhamomyces anomalus CBS 5759/100 | W. anomalus | |
| S1-07 | MT613409 | Trichosporon asahii CBS 2479/100 | T. asahii | |
| S1-08 | MT623566 | Trichosporon asahii CBS 2479/100 | T. asahii | |
| S1-09 | MT623568 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae | |
| S1-10 | MT613410 | Aureobasidium melanogenum CBS 105.22/100 | A. melanogenum | |
| S1-11 | MT613411 | Wickerhamomyces xylosivorus NBRC 111553/89.85 | Wickerhamomyces sp. 1 | |
| S2 | S2-01 | MT613683 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae |
| S2-02 | MT623569 | Wickerhamomyces xylosivorus NBRC 111553/89.85 | Wickerhamomyces sp. 1 | |
| S2-03 | MT623575 | Trichosporon asahii CBS 2479/100 | T. asahii | |
| S2-04 | MT613721 | Papiliotrema laurentii CBS 139/99.87 | P. laurentii | |
| S2-05 | MT623576 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae | |
| S2-06 | MT613722 | Wickerhamomyces xylosivorus NBRC 111553/89.85 | Wickerhamomyces sp. 1 | |
| S2-07 | MT623572 | Aureobasidium melanogenum CBS 105.22/100 | A. melanogenum | |
| S2-08 | MT623573 | Trichosporon asahii CBS 2479/100 | T. asahii | |
| S2-09 | MT613858 | Curvibasidium pallidicorallinum CBS 9091/100 | C. pallidicorallinum | |
| S2-10 | MT623570 | Wickerhamomyces anomalus CBS 5759/100 | W. anomalus | |
| S2-11 | MT623574 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae | |
| S2-12 | MT613867 | Curvibasidium pallidicorallinum CBS 9091/100 | C. pallidicorallinum | |
| S2-13 | MT613870 | Kazachstania aquatica CBS 10102/100 | K. aquatica | |
| S2-14 | MT623571 | Wickerhamomyces mori CBS 12678/90.97 | Wickerhamomyces sp. 2 | |
| S2-15 | MT613872 | Kazachstania aquatica CBS 10102/100 | K. aquatica | |
| S2-16 | MT613876 | Papiliotrema laurentii CBS 139/99.85 | P. laurentii | |
| S2-17 | MT613875 | Wickerhamomyces mori CBS 12678/90.97 | Wickerhamomyces sp. 2 | |
| S3 | S3-01 | MT626064 | Schwanniomyces pseudopolymorphus CBS:2008/100 | S. pseudopolymorphus |
| S3-02 | MT626065 | Apiotrichum scarabaeorum CBS 5601/99.84 | Ap. scarabaeorum | |
| S3-03 | MT632028 | Schwanniomyces pseudopolymorphus CBS:2008/100 | S. pseudopolymorphus | |
| S3-04 | MT626066 | Saturnispora diversa NRRL Y-5713/100 | Sat. diversa | |
| S3-05 | MT639220 | Wickerhamomyces xylosivorus NBRC 111553/89.85 | Wickerhamomyces sp. 1 | |
| S3-06 | MT626068 | Galactomyces pseudocandidum CBS 820.71/97.87 | Galactomyces sp. | |
| S3-07 | MT632029 | Papiliotrema laurentii CBS 139/99.86 | P. laurentii | |
| S3-08 | MT626069 | Trichosporon coremiiforme CBS 2482/99.87 | T. coremiiforme | |
| S3-09 | MT622026 | Trichosporon asahii CBS 2479/100 | T. asahii | |
| S3-10 | MT626071 | Saturnispora sekii CBS 10931/100 | Sat. sekii | |
| S3-11 | MT632027 | Rhodosporidiobolus ruineniae CBS 5001/100 | R. ruineniae | |
| S3-12 | MT632031 | Trichosporon coremiiforme CBS 2482/99.87 | T. coremiiforme | |
| S3-13 | MT632030 | Saturnispora sekii CBS 10931/100 | Sat. sekii | |
| S3-14 | MT632025 | Trichosporon asahii CBS 2479/100 | T. asahii |
| Yeast Species | Strain SDBR-CMU | Amount of IAA (mg/L) * |
|---|---|---|
| Trichosporon asahii | S1-01 | 3.21 ± 2.19 |
| S1-04 | 3.65 ± 1.34 | |
| S1-07 | 2.12 ± 1.35 | |
| S1-08 | 4.21 ± 1.87 | |
| S2-03 | 2.53 ± 1.46 | |
| S2-08 | 5.02 ± 2.05 | |
| S3-09 | 3.82 ± 2.14 | |
| S3-14 | 2.35 ± 0.92 | |
| Papiliotrema laurentii | S1-02 | 2.35 ± 1.97 |
| S2-04 | 3.54 ± 1.06 | |
| S2-16 | 2.78 ± 1.53 | |
| S3-07 | 2.93 ± 2.04 | |
| Rhodosporidiobolus ruineniae | S1-03 | 37.32 ± 3.05 |
| S1-05 | 20.45 ± 2.67 | |
| S1-09 | 18.38 ± 1.28 | |
| S2-01 | 26.73 ± 3.24 | |
| S2-05 | 17.45 ± 2.03 | |
| S2-11 | 14.63 ± 2.15 | |
| S3-11 | 23.45 ± 3.04 | |
| Wickerhamomyces sp. 1 | S1-11 | 17.36 ± 2.12 |
| S2-02 | 12.45 ± 3.16 | |
| S2-06 | 10.38 ± 2/18 | |
| S3-05 | 16.63 ± 1.96 | |
| Curvibasidium pallidicorallinum | S2-09 | 8.61 ± 1.37 |
| S2-12 | 3.62 ± 1.75 | |
| Apiotrichum scarabaeorum | S3-02 | 4.24 ± 1.48 |
| Trichosporon coremiiforme | S3-08 | 4.56 ± 2.42 |
| S3-12 | 3.52 ± 1.87 |
| Yeast Species | Strain SDBR-CMU | Solubilization Index */Solubilization Activities | ||
|---|---|---|---|---|
| Ca3(PO4)2 | ZnO | ZnCO3 | ||
| Papiliotrema laurentii | S1-02 | 1.32 ± 0.94/Medium | 2.13 ± 0.92/High | – |
| S2-04 | 1.04 ± 0.54/Medium | 2.05 ± 0.32/High | – | |
| S2-16 | 1.26 ± 0.72/Medium | 2.08 ± 0.24/High | – | |
| S3-07 | 1.26 ± 0.36/Medium | 2.07 ± 0.36/High | – | |
| Wickerhamomyces anomalus | S1-06 | 1.56 ± 0.61/Medium | 1.34 ± 0.21/Medium | 1.52 ± 0.31/Medium |
| S2-10 | 1.48 ± 0.17/Medium | 1.27 ± 0.35/Medium | 1.38 ± 0.52/Medium | |
| Yeast Species | Strain SDBR-CMU | Enzyme Activity Index * | |||||
|---|---|---|---|---|---|---|---|
| Amylase | Endoglucanase | Lipase | Pectinase | Protease | Xylanase | ||
| Trichosporon asahii | S1-01 | – | 1.57 | 1.25 | – | – | – |
| S1-04 | – | 1.43 | 1.28 | – | – | – | |
| S1-07 | – | 1.03 | 1.27 | – | – | – | |
| S1-08 | – | 1.34 | 1.17 | – | – | – | |
| S2-03 | – | 1.06 | 1.24 | – | – | – | |
| S2-08 | – | 1.13 | 1.07 | – | – | – | |
| S3-09 | – | 1.15 | 1.04 | – | – | – | |
| S3-14 | – | 1.23 | 1.08 | – | – | – | |
| Papiliotrema laurentii | S1-02 | 1.01 | – | 1.30 | 1.63 | 1.04 | 1.01 |
| S2-04 | 1.13 | – | 1.21 | 1.54 | 1.09 | 1.02 | |
| S2-16 | 1.08 | – | 1.33 | 1.46 | 1.12 | 1.03 | |
| S3-07 | 1.05 | – | 1.08 | 1.38 | 1.07 | 1.06 | |
| Rhodosporidiobolus ruineniae | S1-03 | – | 1.07 | 1.09 | – | – | 1.01 |
| S1-05 | – | 1.04 | 1.09 | – | – | 1.06 | |
| S1-09 | – | 1.34 | 1.07 | – | – | 1.04 | |
| S2-01 | – | 1.12 | 1.37 | – | – | 1.02 | |
| S2-05 | – | 1.13 | 1.07 | – | – | 1.08 | |
| S2-11 | – | 1.09 | 1.07 | – | – | 1.13 | |
| S3-11 | – | 1.04 | 1.10 | – | – | 1.03 | |
| Apiotrichum scarabaeorum | S3-02 | – | – | – | – | – | 1.04 |
| Trichosporon coremiiforme | S3-08 | – | 1.07 | 1.13 | – | – | – |
| S3-12 | – | 1.05 | 1.32 | – | – | – | |
| Wickerhamomyces anomalus | S1-06 | – | 1.01 | – | – | 1.03 | – |
| S2-10 | – | 1.06 | – | – | 1.24 | – | |
| Kazachstania aquatica | S2-13 | 1.18 | 1.37 | – | – | – | – |
| S2-15 | 1.12 | 1.32 | – | – | – | – | |
| Schwanniomyces pseudopolymorphus | S3-01 | 1.05 | 1.16 | – | – | – | – |
| S3-03 | 1.04 | 1.12 | – | – | – | – | |
| Aureobasidium melanogenum | S1-10 | – | – | – | – | – | 1.07 |
| S2-07 | – | – | – | – | – | 1.05 | |
| Galactomyces sp. | S3-06 | – | – | – | – | – | 1.01 |
| Saturnispora diversa | S3-04 | – | – | 1.20 | – | – | – |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumla, J.; Nundaeng, S.; Suwannarach, N.; Lumyong, S. Evaluation of Multifarious Plant Growth Promoting Trials of Yeast Isolated from the Soil of Assam Tea (Camellia sinensis var. assamica) Plantations in Northern Thailand. Microorganisms 2020, 8, 1168. https://doi.org/10.3390/microorganisms8081168
Kumla J, Nundaeng S, Suwannarach N, Lumyong S. Evaluation of Multifarious Plant Growth Promoting Trials of Yeast Isolated from the Soil of Assam Tea (Camellia sinensis var. assamica) Plantations in Northern Thailand. Microorganisms. 2020; 8(8):1168. https://doi.org/10.3390/microorganisms8081168
Chicago/Turabian StyleKumla, Jaturong, Supakorn Nundaeng, Nakarin Suwannarach, and Saisamorn Lumyong. 2020. "Evaluation of Multifarious Plant Growth Promoting Trials of Yeast Isolated from the Soil of Assam Tea (Camellia sinensis var. assamica) Plantations in Northern Thailand" Microorganisms 8, no. 8: 1168. https://doi.org/10.3390/microorganisms8081168
APA StyleKumla, J., Nundaeng, S., Suwannarach, N., & Lumyong, S. (2020). Evaluation of Multifarious Plant Growth Promoting Trials of Yeast Isolated from the Soil of Assam Tea (Camellia sinensis var. assamica) Plantations in Northern Thailand. Microorganisms, 8(8), 1168. https://doi.org/10.3390/microorganisms8081168






