Maturational Changes Alter Effects of Dietary Phytase Supplementation on the Fecal Microbiome in Fattening Pigs
Abstract
1. Introduction
2. Materials and Methods
2.1. Diets
2.2. Animals and Housing
2.3. Chemical Analyses
2.4. DNA Extraction and 16S rRNA Gene Amplification
2.5. Bioinformatic Analysis and Statistical Analysis
3. Results
3.1. Feed Intake
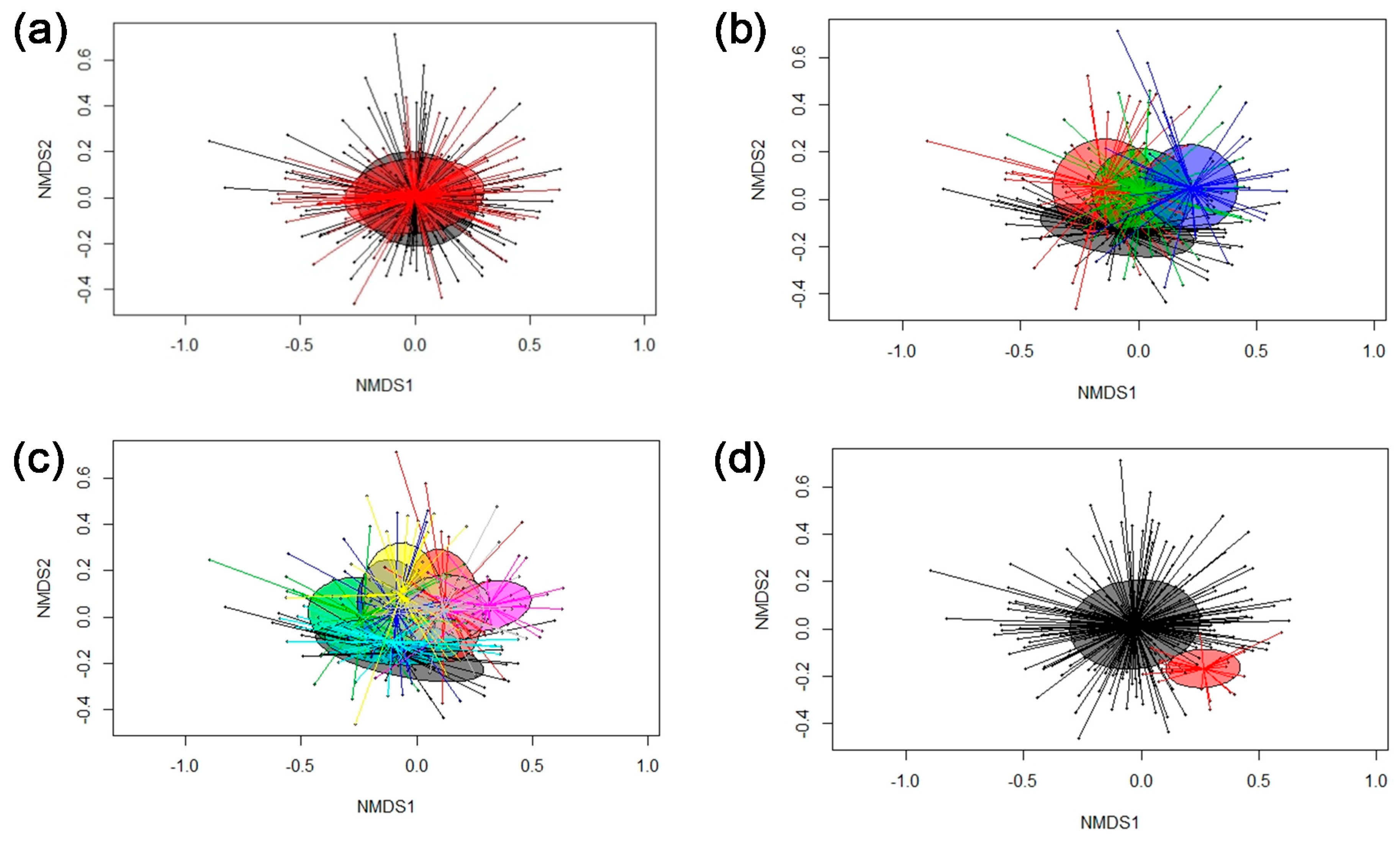
3.2. Microbiota Composition Analysis and Age-Related Changes
3.3. Cluster Analysis
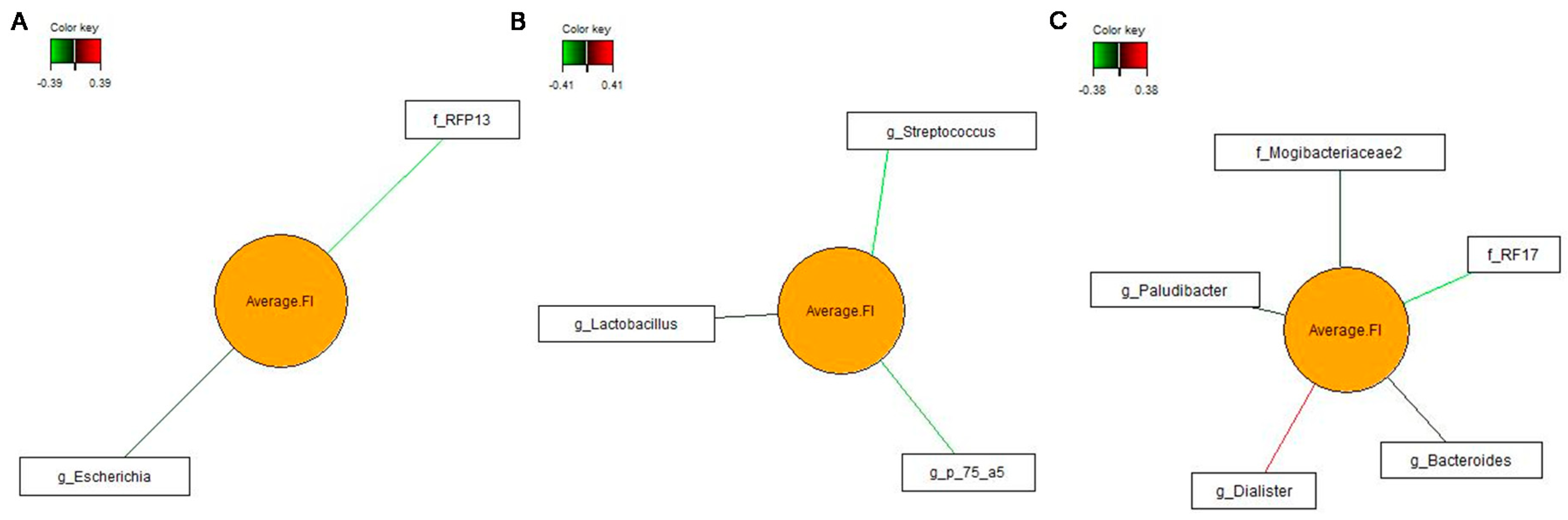
3.4. Associations between Bacterial Genera in Feces and Pig’s Feed Intake
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Links between diet, gut microbiota composition and gut metabolism. Proc. Nutr. Soc. 2015, 74, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Hrdina, J.; Banning, A.; Kippa, A.; Loh, G.; Blaut, M.; Brigelius-Flohé, R. The gastrointestinal microbiota affects the selenium status and selenoprotein expression in mice. J. Nutr. Biochem. 2009, 20, 638–648. [Google Scholar] [CrossRef] [PubMed]
- Durand, M.; Komisarczuk, S. Influence of Major Minerals on Rumen Microbiota. J. Nutr. 1988, 118, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Metzler-Zebeli, B.U.; Mann, E.; Schmitz-Esser, S.; Wagner, M.; Ritzmann, M.; Zebeli, Q. Changing dietary calcium-phosphorus level and cereal source selectively alters abundance of bacteria and metabolites in the upper gastrointestinal tracts of weaned pigs. Appl. Environ. Microbiol. 2013, 79, 7264–7272. [Google Scholar] [CrossRef] [PubMed]
- Mann, E.; Schmitz-Esser, S.; Zebeli, Q.; Wagner, M.; Ritzmann, M.; Metzler-Zebeli, B.U. Mucosa-associated bacterial microbiome of the gastrointestinal tract of weaned pigs and dynamics linked to dietary calcium-phosphorus. PLoS ONE 2014, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Dersjant-Li, Y.; Awati, A.; Schulze, H.; Partridge, G. Phytase in non-ruminant animal nutrition: A critical review on phytase activities in the gastrointestinal tract and influencing factors. J. Sci. Food Agric. 2015, 95, 878–896. [Google Scholar] [CrossRef]
- Oster, M.; Gerlinger, C.; Heide, K.; Just, F.; Borgelt, L.; Wolf, P.; Polley, C.; Vollmar, B.; Muráni, E.; Ponsuksili, S.; et al. Lower dietary phosphorus supply in pigs match both animal welfare aspects and resource efficiency. Ambio. 2017, 47, S20–S29. [Google Scholar] [CrossRef]
- Metzler, B.U.; Mosenthin, R.; Baumgärtel, T.; Rodehutscord, M. The effect of dietary phosphorus and calcium level, phytase supplementation, and ileal infusion of pectin on the chemical composition and carbohydrase activity of fecal bacteria and the level of microbial metabolites in the gastrointestinal tract of pigs. J. Anim. Sci. 2008, 86, 1544–1555. [Google Scholar] [CrossRef]
- Klinsoda, J.; Vötterl, J.C.; Zebeli, Q.; Metzler-Zebeli, B.U. Lactic acid treatment of cereals and dietary phytase modified fecal microbiome composition without affecting expression of virulence factor genes in growing pigs. Front. Microbiol. 2019, 10, 2345. [Google Scholar] [CrossRef]
- Klinsoda, J.; Vötterl, J.C.; Zebeli, Q.; Metzler-Zebeli, B.U. Alterations of the viable ileal microbiota of gut-mucosa-lymph node axis in pigs fed phytase and lactic acid-treated cereals. Appl. Environ. Microbiol. 2020, 86. [Google Scholar] [CrossRef]
- Zhao, W.; Wang, Y.; Liu, S.; Huang, J.; Zhai, Z.; He, C.; Ding, J.; Wang, J.; Wang, H.; Fan, W.; et al. The dynamic distribution of porcine microbiota across different ages and gastrointestinal tract segments. PLoS ONE 2015, 10, e0117441. [Google Scholar] [CrossRef]
- Mann, E.; Dzieciol, M.; Pinior, B.; Neubauer, V.; Metzler-Zebeli, B.U.; Wagner, M.; Schmitz-Esser, S. High diversity of viable bacteria isolated from lymph nodes of slaughter pigs and its possible impacts for food safety. J. Appl. Microbiol. 2015, 119, 1420–1432. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, A.; Bahl, M.; Buschhardt, T.; Beck Hansen, T.; Abu Al-Soud, W.; Brejnrod, A.D.; Sørensen, S.J.; Nesbakken, T.; Aabo, S. Bacterial community analysis for investigating bacterial transfer from tonsils to the pig carcass. Int. J. Food Microbiol. 2019, 295, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Mach, N.; Berri, M.; Estellé, J.; Levenez, F.; Lemonnier, G.; Denis, C.; Leplat, J.; Chevaleyre, C.; Billon, Y.; Doré, J.; et al. Early-life establishment of the swine gut microbiome and impact on host phenotypes. Environ. Microbiol. Rep. 2015, 7, 554–569. [Google Scholar] [CrossRef] [PubMed]
- Gesellschaft fur Ernahrungsphysiologie [GfE]. Empfehlungen zur Energie- und Nährstoffversorgung von Schweinen; DLG: Frankfurt am Main, Germany, 2006. [Google Scholar]
- National Research Council [NRC]. Nutrient requirements of swine; Eleventh Revised Edition; The National Academies Press: Washington, DC, USA, 2012. [Google Scholar]
- Naumann, C.; Basler, R. Die Chemische Untersuchung von Futtermittenln; VDLUFA Verlag: Damstadt, Germany, 2012. [Google Scholar]
- Metzler-Zebeli, B.U.; Deckardt, K.; Schollenberger, M.; Rodehutscord, M.; Zebeli, Q. Lactic acid and thermal treatments trigger the hydrolysis of myo-inositol hexakisphosphate and modify the abundance of lower myo-inositol phosphates in barley (Hordeum vulgare L.). PLoS ONE 2014, 9, e101166. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Newman, M.A.; Ladinig, A.; Kandler, W.; Grüll, D.; Zebeli, Q. Transglycosylated starch accelerated intestinal transit and enhanced bacterial fermentation in the large intestine using a pig model. Br. J. Nutr. 2019, 122, 1–13. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Newman, M.A.; Grüll, D.; Zebeli, Q. Functional adaptations in the cecal and colonic metagenomes associated with the consumption of transglycosylated starch in a pig model. BMC Microbiol. 2019, 19, 87. [Google Scholar] [CrossRef]
- Callahan, B.; McMurdie, P.; Rosen, M.; Han, A.; Johnson, A.; Holmes, S. DADA2: High-resolution sample inference from Illumina amplicon data. Nature Meth. 2016, 13, 581–583. [Google Scholar] [CrossRef]
- García-Jiménez, B.; Wilkinson, M.D. Robust and automatic definition of microbiome states. Peer J. 2019, 7, e6657. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. egan: Community ecology package R package version 2.5-2. 2018. Available online: https://CRAN.R-project.org/package=vegan (accessed on 25 October 2018).
- Rohart, F.; Gautier, B.; Singh, A.; Lê Cao, K.A. MixOmics: A R package for ‘omics feature selection and multiple data integration. PLoS Comp. Biol. 2017, 13, e1005752. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. B. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Wang, X.; Tsai, T.; Deng, F.; Wei, X.; Chai, J.; Knapp, J.; Apple, J.; Maxwell, C.; Lee, J.; Li, Y.; et al. Longitudinal investigation of the swine gut microbiome from birth to market reveals stage and growth performance associated bacteria. Microbiome. 2019, 7, 109. [Google Scholar] [CrossRef]
- Metzler, B.U.; Mosenthin, R. A review of interactions between dietary fiber and the gastrointestinal microbiota and their consequences on intestinal phosphorus metabolism in growing pigs. Asian-Austr. J. Anim. Sci. 2008, 21, 4603–4615. [Google Scholar] [CrossRef]
- Broom, L.; Kogut, M. Gut immunity: Its development and reasons and opportunities for modulation in monogastric production animals. Anim. Health Res. Rev. 2018, 19, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Metzler-Zebeli, B.U.; Schmitz-Esser, S.; Mann, E.; Grüll, D.; Molnar, T.; Zebeli, Q. Adaptation of the cecal bacterial microbiome of growing pigs in response to resistant starch type 4. Appl. Environ. Microbiol. 2015, 81, 8489–8499. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, R.; Middelkoop, A.; Bolhuis, J.; Kleerebezem, M. Legitimate and Reliable Determination of the Age-Related Intestinal Microbiome in Young Piglets; Rectal Swabs and Fecal Samples Provide Comparable Insights. Front. Microbiol. 2019, 10, 1886. [Google Scholar] [CrossRef] [PubMed]
- Guevarra, R.; Lee, J.; Lee, S.; Seok, M.; Kim, D.; Kang, B.; Johnson, T.; Isaacson, R.; Kim, H. Piglet gut microbial shifts early in life: Causes and effects. J. Anim. Sci. Biotechnol. 2019, 10, 1. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Vahjen, W.; Baumgärtel, T.; Rodehutscord, M.; Mosenthin, R. Ileal microbiota of growing pigs fed different dietary calcium phosphate levels and phytase content and subjected to ileal pectin infusion. J. Anim. Sci. 2010, 88, 147–158. [Google Scholar] [CrossRef]
- Lopez-Siles, M.; Khan, T.M.; Duncan, S.H.; Harmsen, H.J.; Garcia-Gil, L.J.; Flint, H.J. Cultured representatives of two major phylogroups of human colonic Faecalibacterium prausnitzii can utilize pectin, uronic acids, and host-derived substrates for growth. Appl. Environ. Microbiol. 2012, 78, 420–428. [Google Scholar] [CrossRef]
- Guevarra, R.B.; Hong, S.H.; Cho, J.H.; Kim, B.R.; Shin, J.; Lee, J.H.; Kim, H.B. The dynamics of the piglet gut microbiome during the weaning transition in association with health and nutrition. J. Anim. Sci. Biotechnol. 2018, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, D.; Falony, G.; Vieira-Silva, S.; Tito, R.Y.; Joossens, M.; Raes, J. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut Microbiota 2016, 65, 57. [Google Scholar] [CrossRef] [PubMed]
- McCormack, U.; Curião, T.; Buzoianu, S.; Prieto, M.L.; Ryan, T.; Varley, P.; Crispie, F.; Magowan, E.; Metzler-Zebeli, B.U.; Berry, D.; et al. Exploring a possible link between the intestinal microbiota and feed efficiency in pigs. Appl. Environ. Microbiol. 2017, 83, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.; Scott, K.; Louis, P.; Duncan, S. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastro. Hepat. 2012, 9, 577–589. [Google Scholar] [CrossRef]
- Kovatcheva-Datchary, P.; Nilsson, A.; Akrami, R.; Lee, Y.S.; De Vadder, F.; Arora, T.; Hallen, A.; Martens, E.; Björck, I.; Bäckhed, F. Dietary fiber-induced improvement in glucose metabolism is associated with increased abundance of Prevotella. Cell Metab. 2015, 22, 971–982. [Google Scholar] [CrossRef]
- Vötterl, J.C.; Zebeli, Q.; Hennig-Pauka, I.; Metzler-Zebeli, B.U. Soaking in lactic acid lowers the phytate-phosphorus content and increases the resistant starch in wheat and corn grains. Anim. Feed Sci. Technol. 2019, 252, 115–125. [Google Scholar] [CrossRef]
- She, Y.; Sparks, J.C.; Stein, H.H. Effects of increasing concentrations of an Escherichia coli phytase on the apparent ileal digestibility of amino acids and the apparent total tract digestibility of energy and nutrients in corn-soybean meal diets fed to growing pigs. J. Anim. Sci. 2018, 96, 2804–2816. [Google Scholar] [CrossRef]
- Rose, R. The role of calcium in oral streptococcal aggregation and the implications for biofilm formation and retention. BBA-Gen. Subjects 2000, 1475, 76–82. [Google Scholar] [CrossRef]
- Rigden, D.J.; Jedrzejas, M.J.; Galperin, M.Y. An extracellular calcium-binding domain in bacteria with a distant relationship to EF-hands. FEMS Microbiol. Lett. 2003, 221, 103–110. [Google Scholar] [CrossRef]
- Geesey, G.; Wigglesworth-Cooksey, B.; Cooksey, K.E. Influence of calcium and other cations on surface adhesion of bacteria and diatoms: A review. Biofouling 2000, 15, 195–205. [Google Scholar] [CrossRef]
- Lim, S.M.; Ahn, D.H. Factors affecting adhesion of lactic acid bacteria to caco-2 cells and inhibitory effect on infection of Salmonella Typhimurium. J. Microbiol. Biotechnol. 2012, 22, 1731–1739. [Google Scholar] [CrossRef]
- Govers, M.; van der Meer, R. Effects of dietary calcium and phosphate on the intestinal interactions between calcium, phosphate, fatty acids, and bile acids. Gut Microbiota. 1993, 34, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Mäkinen, K.K.; Mäkinen, P.L. The peptidolytic capacity of the spirochete system. Med. Microbiol. Immunol. 1996, 185, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mesina, V.G.R.; Lagos, L.V.; Sulabo, R.C.; Walk, C.L.; Stein, H.H. Effects of microbial phytase on mucin synthesis, gastric protein hydrolysis, and degradation of phytate along the gastrointestinal tract of growing pigs. J. Anim. Sci. 2019, 97, 756–767. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.; Daly, K.; Moran, A.; Ryan, S.; Bravo, D.; Shirazi-Beechey, S. Composition and diversity of mucosa-associated microbiota along the entire length of the pig gastrointestinal tract; dietary influences. Environ. Microbiol. 2017, 19, 1425–1438. [Google Scholar] [CrossRef] [PubMed]
| Ingredients (%) | Control Diet | Phytase Diet |
|---|---|---|
| Barley | 45.06 | 45.00 |
| Wheat (11% CP) | 35.51 | 35.46 |
| Soybean meal HP (47% CP) | 8.11 | 8.10 |
| Soybean meal (42% CP) | 7.01 | 7.00 |
| Calcium carbonate | 1.28 | 1.28 |
| Rapeseed oil | 1.00 | 1.00 |
| Monocalcium phosphate | 0.46 | 0.46 |
| Salt | 0.46 | 0.46 |
| Lysine-HCL 98 | 0.41 | 0.41 |
| Premix 1,2 | 0.39 | 0.39 |
| l-Threonine | 0.13 | 0.13 |
| Magnesiumoxide | 0.10 | 0.10 |
| DL-Methionine | 0.08 | 0.08 |
| Phytase (phytase units/kg) 3 | - | 650 |
| Analyzed chemical composition (dry matter basis) | ||
| Dry matter | 89.86 | 89.80 |
| Crude ash | 5.05 | 5.03 |
| Crude protein | 18.40 | 18.86 |
| aNDFOM | 13.82 | 14.07 |
| ADFOM | 5.75 | 5.73 |
| Starch | 69.20 | 69.10 |
| Sugar | 5.40 | 5.60 |
| Calcium | 7.48 | 7.50 |
| Phosphorus | 5.68 | 5.59 |
| Calcium:phosphorus | 1.32 | 1.34 |
| Diet | Control | Phytase | p-value | |||||
|---|---|---|---|---|---|---|---|---|
| Sex | f | m | f | m | SEM | Sex | Diet | Sex × Diet |
| Week 3 | 1.91 | 2.25 | 1.89 | 2.18 | 0.054 | <0.001 | 0.398 | 0.667 |
| Week 5 | 2.03 | 2.26 | 2.08 | 2.22 | 0.054 | 0.001 | 0.938 | 0.412 |
| Week 10 | 2.35 | 2.78 | 2.56 | 2.69 | 0.095 | 0.005 | 0.533 | 0.131 |
| Covariables | df | SS | R2 | F | p-Value |
|---|---|---|---|---|---|
| Cluster | 1 | 0.83 | 0.05 | 17.41 | 0.001 |
| Diet | 1 | 0.93 | 0.05 | 19.59 | 0.001 |
| Week | 1 | 2.31 | 0.13 | 48.69 | 0.001 |
| Sex | 1 | 0.07 | 0.004 | 1.43 | 0.178 |
| Pen | 4 | 1.54 | 0.09 | 8.08 | 0.001 |
| Cluster × diet | 1 | 0.09 | 0.005 | 1.81 | 0.079 |
| Cluster × week | 1 | 0.26 | 0.01 | 5.37 | 0.002 |
| Diet × week | 1 | 0.10 | 0.006 | 2.14 | 0.049 |
| Cluster × sex | 1 | 0.02 | 0.001 | 0.48 | 0.830 |
| Diet × sex | 1 | 0.05 | 0.003 | 1.13 | 0.272 |
| Week × sex | 1 | 0.06 | 0.003 | 1.23 | 0.250 |
| Cluster × pen | 3 | 0.21 | 0.01 | 1.47 | 0.115 |
| Week × pen | 4 | 0.47 | 0.03 | 2.47 | 0.001 |
| Sex × pen | 4 | 0.31 | 0.02 | 1.65 | 0.046 |
| Cluster × diet × week | 1 | 0.04 | 0.002 | 0.81 | 0.531 |
| Cluster × diet × sex | 1 | 0.03 | 0.002 | 0.58 | 0.742 |
| Diet × week × sex | 1 | 0.04 | 0.002 | 0.83 | 0.494 |
| Cluster × week × pen | 2 | 0.07 | 0.004 | 0.76 | 0.653 |
| Cluster × sex × pen | 2 | 0.06 | 0.003 | 0.66 | 0.783 |
| Week × sex × pen | 4 | 0.17 | 0.009 | 0.88 | 0.575 |
| Residual | 219 | 10.44 | 0.58 | ||
| Total | 255 | 18.12 | 1.00 |
| Week | 0 | 0 | 3 | 3 | 5 | 5 | 10 | 10 | p-value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diet | Con | Phy | SEM | Con | Phy | SEM | Con | Phy | SEM | Con | Phy | SEM | Week | Diet | Week × Diet |
| Chao1 | 663 | 618 | 23.2 | 413b | 503a | 23.2 | 484 | 530 | 23.2 | 470 | 455 | 23.2 | <0.001 | 0.376 | 0.005 |
| Shannon | 5.320 | 5.267 | 0.049 | 4.960 | 5.053 | 0.049 | 5.092 | 5.102 | 0.049 | 4.939 | 4.891 | 0.049 | <0.001 | 0.912 | 0.305 |
| Simpson | 0.984 | 0.983 | 0.002 | 0.982 a | 0.978 b | 0.002 | 0.983 a | 0.977 b | 0.002 | 0.976 | 0.972 | 0.002 | <0.001 | 0.014 | 0.446 |
| Week 3 | Week 5 | Week 10 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genus | Mean 1 | log2 Fold Change 2 | SE | p-Value | q-Value 3 | Mean 1 | log2 Fold Change 2 | SE | p-Value | q-Value 3 | Mean 1 | log2 Fold Change2 | SE | p-Value | q-Value 3 |
| Prevotella | 4129 | −0.86 | 0.191 | <0.001 | <0.001 | 3307 | −0.60 | 0.205 | 0.003 | 0.018 | 2041 | −0.66 | 0.228 | 0.004 | 0.017 |
| Clostridiaceae | 1358 | 0.75 | 0.206 | <0.001 | 0.003 | 1739 | 0.67 | 0.206 | 0.001 | 0.009 | 1751 | 0.14 | 0.160 | 0.397 | 0.569 |
| Ruminococcaceae | 1350 | 0.51 | 0.128 | <0.001 | 0.001 | 1559 | 0.31 | 0.118 | 0.009 | 0.041 | 1217 | 0.26 | 0.081 | 0.001 | 0.009 |
| Lactobacillus | 1223 | −1.02 | 0.420 | 0.015 | 0.044 | 928 | −0.68 | 0.424 | 0.111 | 0.242 | 1047 | −0.61 | 0.342 | 0.075 | 0.175 |
| SMB53 | 902 | 0.64 | 0.182 | <0.001 | 0.004 | 957 | 0.48 | 0.171 | 0.005 | 0.023 | 763 | 0.01 | 0.147 | 0.945 | 0.961 |
| Megasphaera | 722 | −1.39 | 0.240 | <0.001 | <0.001 | 412 | −1.83 | 0.384 | <0.001 | <0.001 | 147 | −2.37 | 0.657 | <0.001 | 0.004 |
| Oscillospira | 705 | 0.69 | 0.177 | <0.001 | 0.001 | 864 | 0.18 | 0.138 | 0.199 | 0.373 | 646 | 0.21 | 0.088 | 0.016 | 0.051 |
| Ruminococcus | 576 | 0.25 | 0.186 | 0.175 | 0.324 | 619 | −0.29 | 0.157 | 0.068 | 0.165 | 361 | −0.01 | 0.139 | 0.951 | 0.961 |
| Treponema | 568 | −0.60 | 0.230 | 0.009 | 0.033 | 726 | −0.54 | 0.217 | 0.013 | 0.051 | 597 | −0.06 | 0.235 | 0.809 | 0.863 |
| S24-7 | 532 | −0.33 | 0.198 | 0.097 | 0.206 | 699 | 0.20 | 0.187 | 0.283 | 0.480 | 694 | −0.43 | 0.137 | 0.002 | 0.010 |
| Lachnospiraceae | 470 | −0.14 | 0.199 | 0.489 | 0.643 | 568 | −0.06 | 0.193 | 0.742 | 0.854 | 512 | −0.22 | 0.175 | 0.214 | 0.373 |
| Roseburia | 431 | −0.96 | 0.316 | 0.003 | 0.011 | 258 | −1.08 | 0.311 | <0.001 | 0.007 | 150 | −0.76 | 0.382 | 0.046 | 0.118 |
| Bacteroidales | 415 | 0.08 | 0.157 | 0.631 | 0.755 | 580 | 0.42 | 0.166 | 0.012 | 0.049 | 517 | 0.40 | 0.159 | 0.011 | 0.040 |
| Veillonellaceae | 385 | −1.36 | 0.332 | <0.001 | 0.001 | 216 | −1.27 | 0.412 | 0.002 | 0.012 | 57 | −1.67 | 0.569 | 0.003 | 0.016 |
| Streptococcus | 375 | −1.19 | 0.362 | 0.001 | 0.006 | 608 | −0.24 | 0.365 | 0.507 | 0.687 | 487 | −0.08 | 0.315 | 0.806 | 0.863 |
| Dialister | 347 | −0.98 | 0.372 | 0.008 | 0.033 | 219 | −0.83 | 0.489 | 0.088 | 0.203 | 60 | −1.64 | 0.783 | 0.036 | 0.099 |
| Coprococcus | 341 | 0.19 | 0.115 | 0.093 | 0.201 | 396 | −0.12 | 0.118 | 0.320 | 0.520 | 279 | −0.13 | 0.089 | 0.137 | 0.280 |
| Clostridiales | 326 | 0.86 | 0.165 | <0.001 | < 0.001 | 395 | 0.36 | 0.167 | 0.032 | 0.099 | 291 | 0.45 | 0.141 | 0.001 | 0.009 |
| Blautia | 325 | −0.20 | 0.159 | 0.210 | 0.372 | 261 | −0.72 | 0.180 | <0.001 | 0.001 | 156 | −1.07 | 0.321 | 0.001 | 0.008 |
| Phascolarctobacterium | 278 | 0.36 | 0.206 | 0.077 | 0.174 | 321 | 0.39 | 0.212 | 0.064 | 0.159 | 208 | −0.12 | 0.219 | 0.596 | 0.744 |
| Succinivibrio | 262 | 0.24 | 0.434 | 0.579 | 0.726 | 150 | −0.74 | 0.394 | 0.060 | 0.152 | 88 | −1.52 | 0.450 | 0.001 | 0.007 |
| [Prevotella] | 259 | −0.80 | 0.239 | 0.001 | 0.006 | 235 | −0.27 | 0.218 | 0.208 | 0.384 | 146 | −0.51 | 0.302 | 0.094 | 0.208 |
| Faecalibacterium | 243 | 0.13 | 0.273 | 0.628 | 0.755 | 169 | −0.59 | 0.250 | 0.019 | 0.066 | 80 | −1.17 | 0.354 | 0.001 | 0.008 |
| [Mogibacteriaceae] | 133 | 0.26 | 0.215 | 0.221 | 0.372 | 146 | −0.19 | 0.195 | 0.326 | 0.524 | 113 | −0.27 | 0.140 | 0.050 | 0.124 |
| Anaerovibrio | 130 | −0.81 | 0.380 | 0.034 | 0.092 | 85 | −0.25 | 0.407 | 0.537 | 0.708 | 46 | −1.42 | 0.552 | 0.010 | 0.039 |
| RFP12 | 127 | −0.79 | 0.304 | 0.009 | 0.033 | 197 | −0.14 | 0.255 | 0.573 | 0.733 | 196 | −0.13 | 0.293 | 0.646 | 0.764 |
| Dorea | 127 | −0.18 | 0.207 | 0.385 | 0.565 | 117 | −0.25 | 0.184 | 0.180 | 0.349 | 71 | −0.35 | 0.141 | 0.012 | 0.043 |
| Bulleidia | 123 | −0.13 | 0.188 | 0.494 | 0.643 | 104 | 0.23 | 0.172 | 0.185 | 0.353 | 53 | −0.33 | 0.240 | 0.170 | 0.333 |
| Christensenellaceae | 119 | 0.13 | 0.360 | 0.712 | 0.821 | 213 | 0.78 | 0.324 | 0.016 | 0.056 | 272 | 0.93 | 0.286 | 0.001 | 0.009 |
| Butyricicoccus | 115 | 0.31 | 0.179 | 0.083 | 0.184 | 115 | −0.04 | 0.195 | 0.832 | 0.906 | 68 | 0.09 | 0.220 | 0.684 | 0.791 |
| [Ruminococcus] | 113 | −0.12 | 0.222 | 0.595 | 0.731 | 88 | −0.40 | 0.207 | 0.056 | 0.148 | 48 | −0.64 | 0.261 | 0.014 | 0.046 |
| CF231 | 112 | −0.29 | 0.226 | 0.197 | 0.359 | 145 | −0.16 | 0.224 | 0.464 | 0.663 | 127 | −0.86 | 0.203 | <0.001 | 0.001 |
| Sarcina | 111 | 0.61 | 0.380 | 0.109 | 0.220 | 105 | 0.24 | 0.312 | 0.439 | 0.651 | 72 | −0.20 | 0.394 | 0.605 | 0.744 |
| Peptostreptococcacae | 104 | 0.66 | 0.219 | 0.003 | 0.011 | 65 | 0.46 | 0.184 | 0.013 | 0.051 | 46 | −0.21 | 0.224 | 0.342 | 0.529 |
| [Eubacterium] | 97 | −0.46 | 0.223 | 0.040 | 0.099 | 66 | −0.17 | 0.227 | 0.443 | 0.651 | 27 | −0.75 | 0.217 | 0.001 | 0.007 |
| Turicibacter | 97 | 0.47 | 0.286 | 0.104 | 0.215 | 131 | 0.78 | 0.234 | 0.001 | 0.008 | 117 | 0.22 | 0.186 | 0.242 | 0.400 |
| Clostridium | 49 | 1.69 | 0.568 | 0.003 | 0.013 | 113 | 1.20 | 0.494 | 0.015 | 0.055 | 154 | 0.04 | 0.442 | 0.927 | 0.961 |
| Parabacteroides | 41 | 0.85 | 0.438 | 0.053 | 0.125 | 70 | 0.86 | 0.330 | 0.009 | 0.041 | 45 | 0.58 | 0.256 | 0.023 | 0.064 |
| [Paraprevotella] | 38 | −0.18 | 0.298 | 0.552 | 0.708 | 57 | −0.25 | 0.221 | 0.264 | 0.467 | 40 | 0.00 | 0.205 | 0.986 | 0.986 |
| Escherichia | 35 | 2.74 | 0.759 | 0.000 | 0.003 | 40 | 0.76 | 0.456 | 0.095 | 0.215 | 35 | 0.33 | 0.436 | 0.446 | 0.629 |
| Genus | Mean 1 | log2 Fold Change 2 | SE | p-Value | q-Value 3 |
|---|---|---|---|---|---|
| Prevotella | 4411 | −1.13 | 0.223 | <0.001 | <0.001 |
| Clostridiaceae | 1747 | −0.06 | 0.169 | 0.712 | 0.794 |
| Ruminococcaceae | 1592 | 0 | 0.069 | 0.948 | 0.965 |
| Lactobacillus | 1297 | −1.28 | 0.350 | <0.001 | 0.001 |
| SMB53 | 959 | −0.40 | 0.142 | 0.005 | 0.013 |
| Oscillospira | 791 | −0.21 | 0.100 | 0.032 | 0.068 |
| S24-7 | 759 | 0.11 | 0.145 | 0.452 | 0.590 |
| Megasphaera | 713 | −2.90 | 0.444 | <0.001 | <0.001 |
| Treponema | 680 | 0.04 | 0.190 | 0.845 | 0.883 |
| Bacteroidales | 606 | 0.62 | 0.115 | <0.001 | <0.001 |
| Ruminococcus | 569 | −0.44 | 0.135 | 0.001 | 0.004 |
| Streptococcus | 550 | −1.54 | 0.324 | <0.001 | <0.001 |
| Faecalibacterium | 548 | 0.66 | 0.163 | <0.001 | <0.001 |
| Roseburia | 471 | −1.73 | 0.347 | <0.001 | <0.001 |
| Lachnospiraceae | 423 | −0.25 | 0.112 | 0.028 | 0.060 |
| RFP12 | 421 | −2.06 | 0.465 | <0.001 | <0.001 |
| CF231 | 385 | 0.30 | 0.121 | 0.012 | 0.029 |
| Blautia | 331 | −1.04 | 0.224 | <0.001 | <0.001 |
| Phascolarctobacterium | 328 | −0.24 | 0.182 | 0.190 | 0.294 |
| [Prevotella] | 306 | 0.29 | 0.230 | 0.200 | 0.302 |
| Dialister | 302 | −4.98 | 0.488 | <0.001 | <0.001 |
| Coprococcus | 243 | −1.35 | 0.295 | <0.001 | <0.001 |
| Escherichia | 219 | 1.10 | 0.253 | <0.001 | <0.001 |
| Succinivibrio | 213 | −1.35 | 0.383 | <0.001 | 0.002 |
| Christensenellaceae | 201 | 0.30 | 0.316 | 0.344 | 0.469 |
| Anaerovibrio | 160 | −1.08 | 0.404 | 0.008 | 0.020 |
| Bulleidia | 154 | 0.65 | 0.188 | 0.001 | 0.002 |
| [Mogibacteriaceae] | 152 | 0.43 | 0.133 | 0.001 | 0.005 |
| Clostridium | 142 | 1.24 | 0.412 | 0.003 | 0.008 |
| Dorea | 133 | −0.29 | 0.162 | 0.071 | 0.132 |
| Butyricicoccus | 128 | −0.28 | 0.176 | 0.107 | 0.175 |
| Turicibacter | 126 | 0.22 | 0.211 | 0.289 | 0.414 |
| Veillonellaceae | 126 | 0.19 | 0.460 | 0.673 | 0.777 |
| Peptostreptococcacae | 116 | −0.99 | 0.196 | <0.001 | <0.001 |
| [Ruminococcus] | 107 | −1.49 | 0.230 | <0.001 | <0.001 |
| Mitsuokella | 92 | −3.40 | 0.599 | <0.001 | <0.001 |
| [Eubacterium] | 92 | −1.31 | 0.258 | <0.001 | <0.001 |
| Sarcina | 89 | −1.54 | 0.464 | 0.001 | 0.003 |
| Pirellulaceae | 88 | −0.30 | 0.184 | 0.105 | 0.173 |
| Oribacterium | 68 | −2.18 | 0.250 | <0.001 | 0.000 |
| Coriobacteriaceae | 67 | −0.33 | 0.242 | 0.176 | 0.277 |
| RFN20 | 66 | −0.12 | 0.221 | 0.572 | 0.679 |
| Campylobacter | 64 | −26.11 | 1.632 | <0.001 | <0.001 |
| Parabacteroides | 62 | 0.08 | 0.303 | 0.785 | 0.851 |
| Clostridiales | 58 | −1.88 | 0.572 | 0.001 | 0.004 |
| Catenibacterium | 56 | −3.95 | 0.555 | <0.001 | <0.001 |
| RF16 | 55 | 1.84 | 0.487 | <0.001 | 0.001 |
| BS11 | 54 | 2.02 | 1.031 | 0.051 | 0.099 |
| Acidaminococcus | 54 | −5.52 | 1.137 | <0.001 | <0.001 |
| WPS-2 | 52 | 0.88 | 0.444 | 0.047 | 0.094 |
| Lachnospira | 52 | −0.97 | 0.338 | 0.004 | 0.012 |
| Paludibacter | 52 | 0.95 | 0.310 | 0.002 | 0.007 |
| [Paraprevotellaceae] | 51 | −0.16 | 0.193 | 0.416 | 0.549 |
| Methanobrevibacter | 51 | −0.42 | 0.452 | 0.352 | 0.475 |
| p-75-a5 | 48 | 0.72 | 0.204 | <0.001 | 0.002 |
| Shuttleworthia | 48 | −3.77 | 0.781 | <0.001 | <0.001 |
| Collinsella | 44 | −1.26 | 0.327 | <0.001 | 0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Metzler-Zebeli, B.U.; Klinsoda, J.; Vötterl, J.C.; Verhovsek, D. Maturational Changes Alter Effects of Dietary Phytase Supplementation on the Fecal Microbiome in Fattening Pigs. Microorganisms 2020, 8, 1073. https://doi.org/10.3390/microorganisms8071073
Metzler-Zebeli BU, Klinsoda J, Vötterl JC, Verhovsek D. Maturational Changes Alter Effects of Dietary Phytase Supplementation on the Fecal Microbiome in Fattening Pigs. Microorganisms. 2020; 8(7):1073. https://doi.org/10.3390/microorganisms8071073
Chicago/Turabian StyleMetzler-Zebeli, Barbara U., Jutamat Klinsoda, Julia C. Vötterl, and Doris Verhovsek. 2020. "Maturational Changes Alter Effects of Dietary Phytase Supplementation on the Fecal Microbiome in Fattening Pigs" Microorganisms 8, no. 7: 1073. https://doi.org/10.3390/microorganisms8071073
APA StyleMetzler-Zebeli, B. U., Klinsoda, J., Vötterl, J. C., & Verhovsek, D. (2020). Maturational Changes Alter Effects of Dietary Phytase Supplementation on the Fecal Microbiome in Fattening Pigs. Microorganisms, 8(7), 1073. https://doi.org/10.3390/microorganisms8071073







