Thymol and Carvacrol Downregulate the Expression of Salmonella typhimurium Virulence Genes during an In Vitro Infection on Caco-2 Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Culture Conditions
2.2. Chemicals and Test Solutions
2.3. Minimal Inhibitory Concentration of Bioactive Compounds
2.4. Time-Kill Curve of Thymol and Carvacrol
2.5. Effect of Thymol and Carvacrol on Intestinal Caco-2 Cells Monolayer Integrity
2.6. Determination of Salmonella Virulence mRNA Expression
2.7. Statistical Analysis
3. Results
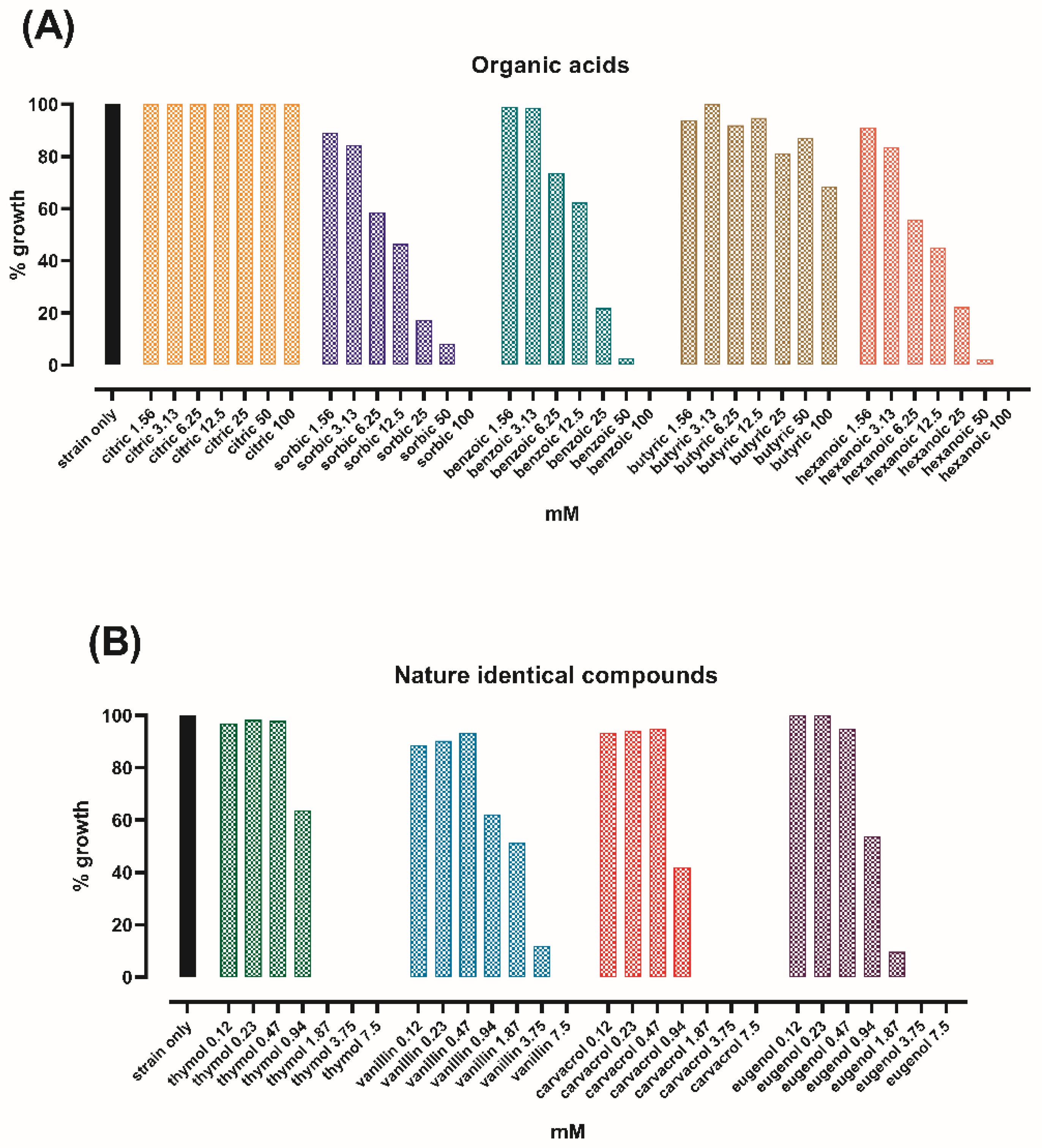
3.1. Thymol and Carvacrol Have the Lowest MIC among Tested Bioactive Compounds
3.2. The Bactericidal Effect of Thymol and Carvacrol Is Dose-Dependent
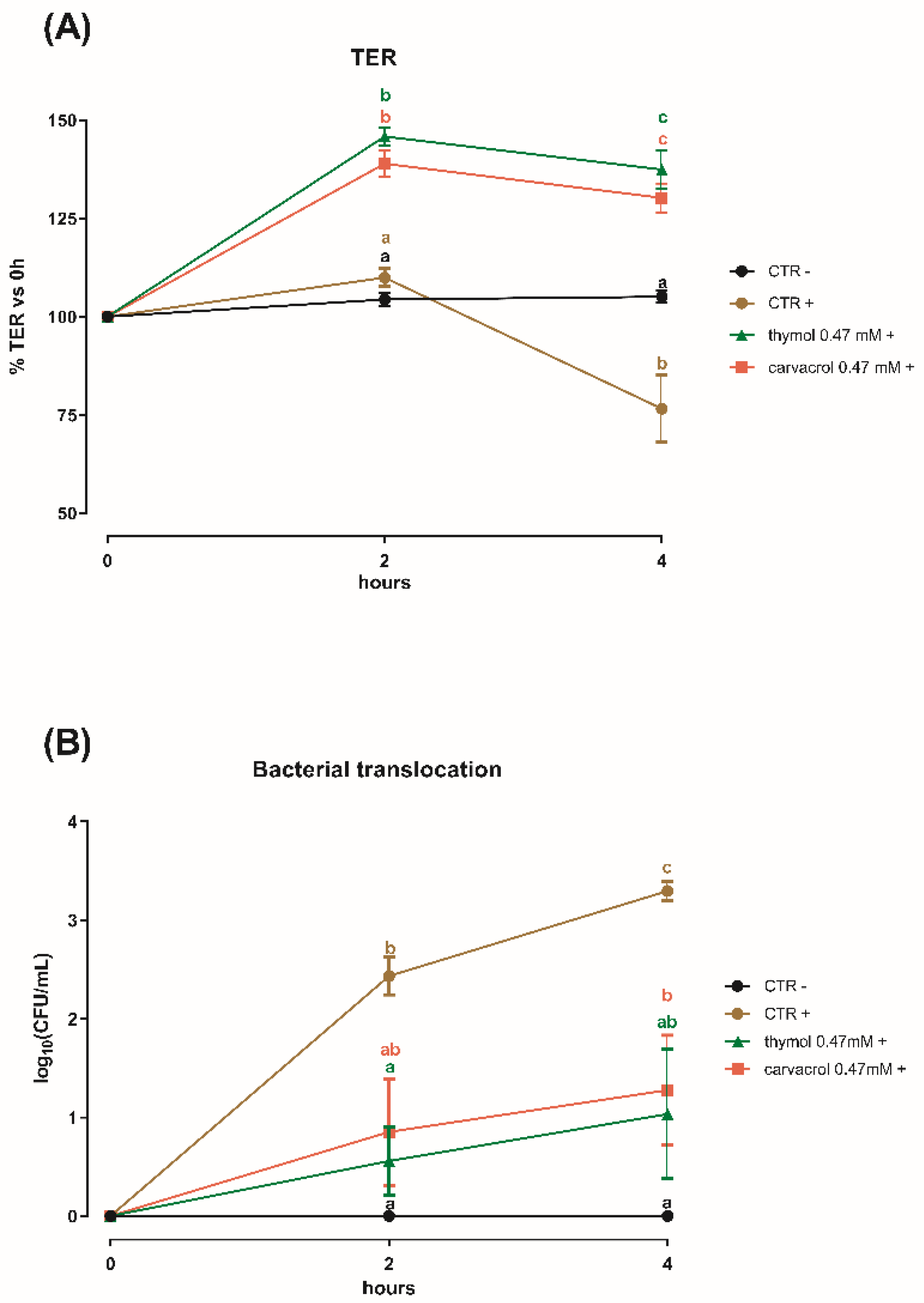
3.3. Thymol and Carvacrol at Sub-Lethal Concentrations Can Counteract a S. typhimurium Challenge on Caco-2 Cells
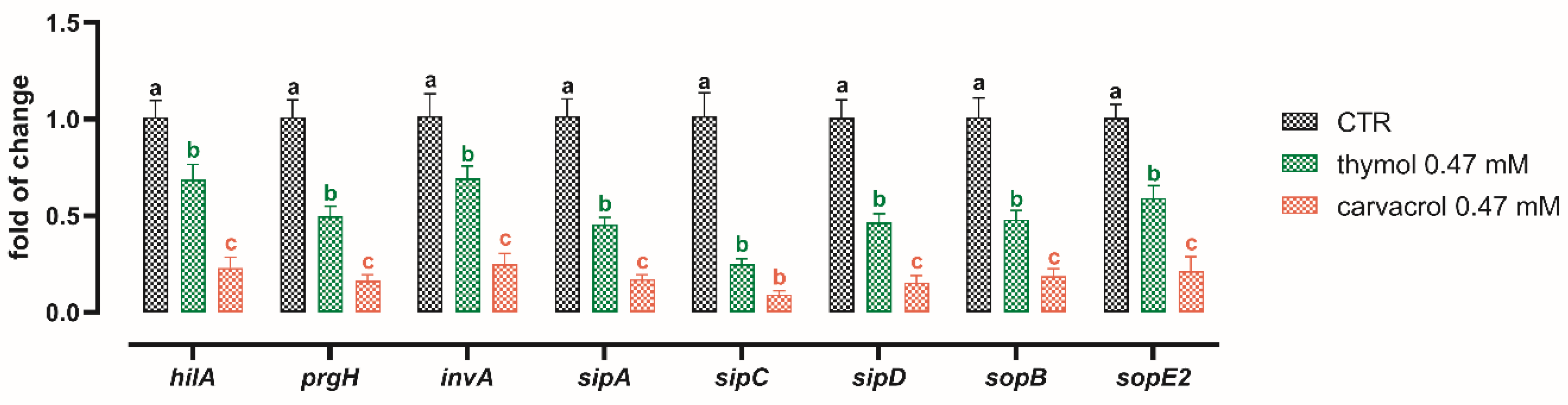
3.4. Sub-Lethal Concentrations of Thymol and Carvacrol Downregulate Virulence Genes of S. typhimurium
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EFSA. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA J. 2018, 16, e05500. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). National Salmonella Surveillance Annual Report, 2016; US Department of Health and Human Services, CDC: Atlanta, GA, USA, 2018.
- Barilli, E.; Bacci, C.; StellaVilla, Z.; Merialdi, G.; D’Incau, M.; Brindani, F.; Vismarra, A. Antimicrobial resistance, biofilm synthesis and virulence genes in Salmonella isolated from pigs bred on intensive farms. Ital. J. Food Saf. 2018, 7, 7223. [Google Scholar] [CrossRef] [PubMed]
- Fedorka-Cray, P.J.; Gray, J.T.; Wray, C. Salmonella Infections in Pigs. In Salmonella in Domestic Animals; Wray, C., Wray, A., Eds.; CABI Publishing: New York, NY, USA, 2000; pp. 191–208. [Google Scholar]
- Farzan, A.; Friendship, R.M. A clinical field trial to evaluate the efficacy of vaccination in controlling Salmonella infection and the association of Salmonella-shedding and weight gain in pigs. Can. J. Vet. Res. 2010, 74, 258–263. [Google Scholar] [PubMed]
- Verbrugghe, E.; Van Parys, A.; Leyman, B.; Boyen, F.; Haesebrouck, F.; Pasmans, F. HtpG contributes to Salmonella Typhimurium intestinal persistence in pigs. Vet. Res. 2015, 46, 118. [Google Scholar] [CrossRef]
- Verbrugghe, E.; Boyen, F.; Van Parys, A.; Van Deun, K.; Croubels, S.; Thompson, A.; Shearer, N.; Leyman, B.; Haesebrouck, F.; Pasmans, F. Stress induced Salmonella Typhimurium recrudescence in pigs coincides with cortisol induced increased intracellular proliferation in macrophages. Vet. Res. 2011, 42, 118. [Google Scholar] [CrossRef]
- Grilli, E.; Foresti, F.; Tugnoli, B.; Fustini, M.; Zanoni, M.G.; Pasquali, P.; Callaway, T.R.; Piva, A.; Alborali, G.L. Microencapsulated sorbic acid and pure botanicals affect Salmonella Typhimurium shedding in pigs: A close-up look from weaning to slaughter in controlled and field conditions. Foodborne Pathog. Dis. 2015, 12, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Nieto, P.A.; Pardo-Roa, C.; Salazar-Echegarai, F.J.; Tobar, H.E.; Coronado-Arrázola, I.; Riedel, C.A.; Kalergis, A.M.; Bueno, S.M. New insights about excisable pathogenicity islands in Salmonella and their contribution to virulence. Microbes Infect. 2016, 18, 302–309. [Google Scholar] [CrossRef]
- Coburn, B.; Sekirov, I.; Finlay, B.B. Type III Secretion Systems and Disease. Clin. Microbiol. Rev. 2007, 20, 535–549. [Google Scholar] [CrossRef]
- Sukhan, A.; Kubori, T.; Wilson, J.; Galán, J.E. Genetic analysis of assembly of the Salmonella enterica serovar Typhimurium type III secretion-associated needle complex. J. Bacteriol. 2001, 183, 1159–1167. [Google Scholar] [CrossRef]
- Singh, Y.; Saxena, A.; Kumar, R.; Kumar Saxena, M. Virulence System of Salmonella with Special Reference to Salmonella enterica. In Salmonella A Re-Emerging Pathogen; Mascellino, M.T., Ed.; IntechOpen Limited: London, UK, 2018; pp. 41–53. [Google Scholar]
- Finlay, B.B.; Ruschkowski, S.; Dedhar, S. Cytoskeletal rearrangements accompanying salmonella entry into epithelial cells. J. Cell. Sci. 1991, 99, 283–296. [Google Scholar]
- Cruz, N.; Qi, L.; Alvarez, X.; Berg, R.D.; Deitch, E.A. The Caco-2 cell monolayer system as an in vitro model for studying bacterial-enterocyte interactions and bacterial translocation. J. Burn Care Rehabil. 1994, 15, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Finlay, B.B.; Falkow, S. Salmonella Interactions with Polarized Human Intestinal Caco-2 Epithelial Cells. J. Infect. Dis. 1990, 162, 1096–1106. [Google Scholar] [CrossRef] [PubMed]
- Boyle, E.C.; Brown, N.F.; Finlay, B.B. Salmonella enterica serovar Typhimurium effectors SopB, SopE, SopE2 and SipA disrupt tight junction structure and function. Cell. Microbiol. 2006, 8, 1946–1957. [Google Scholar] [CrossRef] [PubMed]
- Nair, D.V.T.; Venkitanarayanan, K.; Kollanoor Johny, A. Antibiotic-Resistant Salmonella in the Food Supply and the Potential Role of Antibiotic Alternatives for Control. Foods 2018, 7, 167. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Weir, E.K.; Martin, L.C.; Poppe, C.; Coombes, B.K.; Boerlin, P. Subinhibitory concentrations of tetracycline affect virulence gene expression in a multi-resistant Salmonella enterica subsp. enterica serovar Typhimurium DT104. Microbes Infect. 2008, 10, 901–907. [Google Scholar] [CrossRef]
- Qi, Y.; Zhao, W.; Wang, T.; Pei, F.; Yue, M.; Li, F.; Liu, X.; Wang, X.; Li, H. Proteomic analysis of the antimicrobial effects of sublethal concentrations of thymol on Salmonella enterica serovar Typhimurium. Appl. Microbiol. Biotechnol. 2020, 104, 3493–3505. [Google Scholar] [CrossRef]
- Zhang, J.S.; Corredig, M.; Morales-Rayas, R.; Hassan, A.; Griffiths, M.W.; LaPointe, G. Downregulation of Salmonella Virulence Gene Expression During Invasion of Epithelial Cells Treated with Lactococcus lactis subsp. cremoris JFR1 Requires OppA. Probiotics Antimicrob. Proteins 2019. [Google Scholar] [CrossRef]
- Arguello, H.; Rubio, P.; Carvajal, A. Salmonella Control Measures at Farm in Swine Production. In Salmonella—Distribution, Adaptation, Control Measures and Molecular Technologies; Annous, B., Gurtler, J.B., Eds.; IntechOpen Limited: London, UK, 2012; pp. 99–122. [Google Scholar]
- Liu, Y.; Espinosa, C.D.; Abelilla, J.J.; Casas, G.A.; Lagos, L.V.; Lee, S.A.; Kwon, W.B.; Mathai, J.K.; Navarro, D.M.D.L.; Jaworski, N.W.; et al. Non-antibiotic feed additives in diets for pigs: A review. Anim. Nutr. 2018, 4, 113–125. [Google Scholar] [CrossRef]
- Costa, L.B.; Luciano, F.B.; Miyada, V.S.; Gois, F.D. Herbal extracts and organic acids as natural feed additives in pig diets. S. Afr. J. Anim. Sci. 2013, 43, 181–193. [Google Scholar]
- Van Immerseel, F.; Russell, J.B.; Flythe, M.D.; Gantois, I.; Timbermont, L.; Pasmans, F.; Haesebrouck, F.; Ducatelle, R. The use of organic acids to combat Salmonella in poultry: A mechanistic explanation of the efficacy. Avian Pathol. 2006, 35, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Windisch, W.; Schedle, K.; Plitzner, C.; Kroismayr, A. Use of phytogenic products as feed additives for swine and poultry. J. Anim. Sci. 2008, 86, E140–E148. [Google Scholar] [CrossRef] [PubMed]
- Gómez-García, M.; Sol, C.; De Nova, P.J.G.; Puyalto, M.; Mesas, L.; Puente, H.; Mencía-Ares, Ó.; Miranda, R.; Argüello, H.; Rubio, P.; et al. Antimicrobial activity of a selection of organic acids, their salts and essential oils against swine enteropathogenic bacteria. Porc. Health Manag. 2019, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Guillier, L.; Nazer, A.I.; Dubois-Brissonnet, F. Growth response of Salmonella typhimurium in the presence of natural and synthetic antimicrobials: Estimation of MICs from three different models. J. Food Prot. 2007, 70, 2243–2250. [Google Scholar] [CrossRef] [PubMed]
- Helander, I.M.; Alakomi, H.-L.; Latva-Kala, K.; Mattila-Sandholm, T.; Pol, I.; Smid, E.J.; Gorris, L.G.M.; Von Wright, A. Characterization of the Action of Selected Essential Oil Components on Gram-Negative Bacteria. J. Agric. Food Chem. 1998, 46, 3590–3595. [Google Scholar] [CrossRef]
- Bearson, S.; Bearson, B.; Foster, J.W. Acid stress responses in enterobacteria. FEMS Microbiol. Lett. 1997, 147, 173–180. [Google Scholar] [CrossRef]
- Pinto, M.; Robineleon, S.; Appay, M.D.; Kedinger, M.; Triadou, N.; Dussaulx, E.; Lacroix, B.; Simonassmann, P.; Haffen, K.; Fogh, J.; et al. Enterocyte-Like Differentiation and Polarization of the Human-Colon Carcinoma Cell-Line Caco-2 in Culture. Biol. Cell 1983. Available online: https://www.scienceopen.com/document?vid=10c4912d-182f-4361-98a7-50c52d5b3617 (accessed on 19 May 2020).
- Srinivasan, B.; Kolli, A.R.; Esch, M.B.; Abaci, H.E.; Shuler, M.L.; Hickman, J.J. TEER measurement techniques for in vitro barrier model systems. J. Lab. Autom. 2015, 20, 107–126. [Google Scholar] [CrossRef]
- Rossi, B.; Toschi, A.; Piva, A.; Grilli, E. Single components of botanicals and nature-identical compounds as a non-antibiotic strategy to ameliorate health status and improve performance in poultry and pigs. Nutr. Res. Rev. 2020, 1–17. [Google Scholar] [CrossRef]
- Inamuco, J.; Veenendaal, A.; Burt, S.; Post, J.; Bokhoven, J.; Haagsman, H.; Veldhuizen, E. Sub-lethal levels of carvacrol reduce Salmonella Typhimurium motility and invasion of porcine epithelial cells. Vet. Microbiol. 2011, 157, 200–207. [Google Scholar] [CrossRef]
- Burt, S.A.; Adolfse, S.J.M.; Ahad, D.S.A.; Tersteeg-Zijderveld, M.H.G.; Jongerius-Gortemaker, B.G.M.; Post, J.A.; Brüggemann, H.; Santos, R.R. Cinnamaldehyde, Carvacrol and Organic Acids Affect Gene Expression of Selected Oxidative Stress and Inflammation Markers in IPEC-J2 Cells Exposed to Salmonella typhimurium. Phytother. Res. 2016, 30, 1988–2000. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Qiu, J.; Luo, Z.-Q.; Deng, X. The Herbal Compound Thymol Protects Mice From Lethal Infection by Salmonella Typhimurium. Front Microbiol. 2018, 9, 1022. [Google Scholar] [CrossRef]
- Grantcharova, N.; Peters, V.; Monteiro, C.; Zakikhany, K.; Römling, U. Bistable Expression of CsgD in Biofilm Development of Salmonella enterica Serovar Typhimurium. J. Bacteriol. 2009, 192, 456–466. [Google Scholar] [CrossRef]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils—Present Status and Future Perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef]
- Yuan, W.; Yuk, H.-G. Effects of Sublethal Thymol, Carvacrol, and trans-Cinnamaldehyde Adaptation on Virulence Properties of Escherichia coli O157:H7. Appl. Environ. Microbiol. 2019, 85, 1–11. [Google Scholar]
- Nazzaro, F.; Fratianni, F.; D’Acierno, A.; Coppola, R.; Ayala-Zavala, F.J.; Da Cruz, A.G.; Feo, V.D. Essential Oils and Microbial Communication. In Essential Oils—Oils of Nature; El-Shemy, H., Ed.; IntechOpen Limited: London, UK, 2019; pp. 1–26. [Google Scholar]
- Rutherford, S.T.; Bassler, B.L. Bacterial quorum sensing: Its role in virulence and possibilities for its control. Cold Spring Harb. Perspect Med. 2012, 2, 1–25. [Google Scholar] [CrossRef]
- Choi, J.; Shin, D.; Ryu, S. Implication of Quorum Sensing in Salmonella enterica Serovar Typhimurium Virulence: The luxS Gene Is Necessary for Expression of Genes in Pathogenicity Island 1. Infect Immun. 2007, 75, 4885–4890. [Google Scholar] [CrossRef]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Hautefort, I.; Thompson, A.; Hinton, J.C.; Immerseel, F.V. Butyrate Specifically Down-Regulates Salmonella Pathogenicity Island 1 Gene Expression. Appl. Environ. Microbiol. 2006, 72, 946–949. [Google Scholar] [CrossRef]
- Boyen, F.; Haesebrouck, F.; Vanparys, A.; Volf, J.; Mahu, M.; Van Immerseel, F.; Rychlik, I.; Dewulf, J.; Ducatelle, R.; Pasmans, F. Coated fatty acids alter virulence properties of Salmonella Typhimurium and decrease intestinal colonization of pigs. Vet. Microbiol. 2008, 132, 319–327. [Google Scholar] [CrossRef]
- Hallstrom, K.; McCormick, B.A. Salmonella Interaction with and Passage through the Intestinal Mucosa: Through the Lens of the Organism. Front. Microbiol. 2011, 2, 1–10. [Google Scholar] [CrossRef]
- Tafazoli, F.; Magnusson, K.-E.; Zheng, L. Disruption of Epithelial Barrier Integrity by Salmonella enterica Serovar Typhimurium Requires Geranylgeranylated Proteins. Infect Immun. 2003, 71, 872–881. [Google Scholar] [CrossRef]
- Köhler, H.; Sakaguchi, T.; Hurley, B.P.; Kase, B.A.; Kase, B.J.; Reinecker, H.-C.; McCormick, B.A. Salmonella enterica serovar Typhimurium regulates intercellular junction proteins and facilitates transepithelial neutrophil and bacterial passage. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, G178–G187. [Google Scholar] [CrossRef]

| Gene | Function | Sequence (5′ → 3′) | Product Length (bp) | Accession Number | Reference |
|---|---|---|---|---|---|
| hilA | Transcriptional regulator of the SPI-1 gene expression | F: CTGTACGGACAGGGCTATCG R: GCAGACTCTCGGATTGAACC | 130 | U25352.1 | [19] |
| prgH | Assembly of base ring structure of T3SS | F: CGCTGCGCAAAATGAAAGAG R: TTACGCGGCTCATCGAAATG | 177 | U21676 | [20] |
| invA | Assembly of needle-like structure of T3SS | F: TCTGGATGGTATGCCCGGTA R: TCATCGCACCGTCAAAGGAA | 140 | M90846.1 | This study |
| sipA | Actin rearrangement | F: GTCATTCGCGTGTGGATTCG R: TTCGGATGAAGCGTTGGTCA | 143 | U40013.1 | This study |
| sipC | Actin rearrangement | F: ACGGGCAGAATAGCGTCAAA R: ATACCCAGACTTTCCGTGGC | 150 | U25631.1 | This study |
| sipD | Translocation of effector proteins | F: ATTCCGCTTCTCCTCATCCG R: ACCGCGATGTTCTGTGGTAG | 107 | U40013.1 | This study |
| sopB | Invasion, generation, and maintenance of SCV | F: GATGGCGGCGAACCCTATAA R: GAAGACTACCAGGCGCACTT | 181 | AF213335.2 | This study |
| sopE2 | Actin rearrangement | F: GAACGCTTCTGAGGGTAGGG R: CGAGCATAGGCCGGATCTTT | 117 | AF200952.1 | This study |
| rpoD | Housekeeping | F: GTGAAATGGGCACTGTTGAACTG R: TTCCAGCAGATAGGTAATGGCTTC | 131 | NC_003197.2 | [21] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giovagnoni, G.; Rossi, B.; Tugnoli, B.; Ghiselli, F.; Bonetti, A.; Piva, A.; Grilli, E. Thymol and Carvacrol Downregulate the Expression of Salmonella typhimurium Virulence Genes during an In Vitro Infection on Caco-2 Cells. Microorganisms 2020, 8, 862. https://doi.org/10.3390/microorganisms8060862
Giovagnoni G, Rossi B, Tugnoli B, Ghiselli F, Bonetti A, Piva A, Grilli E. Thymol and Carvacrol Downregulate the Expression of Salmonella typhimurium Virulence Genes during an In Vitro Infection on Caco-2 Cells. Microorganisms. 2020; 8(6):862. https://doi.org/10.3390/microorganisms8060862
Chicago/Turabian StyleGiovagnoni, Giulia, Barbara Rossi, Benedetta Tugnoli, Federico Ghiselli, Andrea Bonetti, Andrea Piva, and Ester Grilli. 2020. "Thymol and Carvacrol Downregulate the Expression of Salmonella typhimurium Virulence Genes during an In Vitro Infection on Caco-2 Cells" Microorganisms 8, no. 6: 862. https://doi.org/10.3390/microorganisms8060862
APA StyleGiovagnoni, G., Rossi, B., Tugnoli, B., Ghiselli, F., Bonetti, A., Piva, A., & Grilli, E. (2020). Thymol and Carvacrol Downregulate the Expression of Salmonella typhimurium Virulence Genes during an In Vitro Infection on Caco-2 Cells. Microorganisms, 8(6), 862. https://doi.org/10.3390/microorganisms8060862






