Selection and Evaluation of Staphylococcus xylosus as a Biocontrol Agent against Toxigenic Moulds in a Dry-Cured Ham Model System
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacteria and Fungi
2.2. Staphylococcal Enterotoxin Analysis
2.3. 16S rDNA Sequence Analysis
2.4. Inocula Preparation
2.5. Effect of GCC+ on Growth of Ochratoxigenic Moulds
2.6. Effect of S. xylosus on Growth and Mycotoxins Production by Mycotoxigenic Moulds
2.6.1. Growth Assessment of Toxigenic Moulds
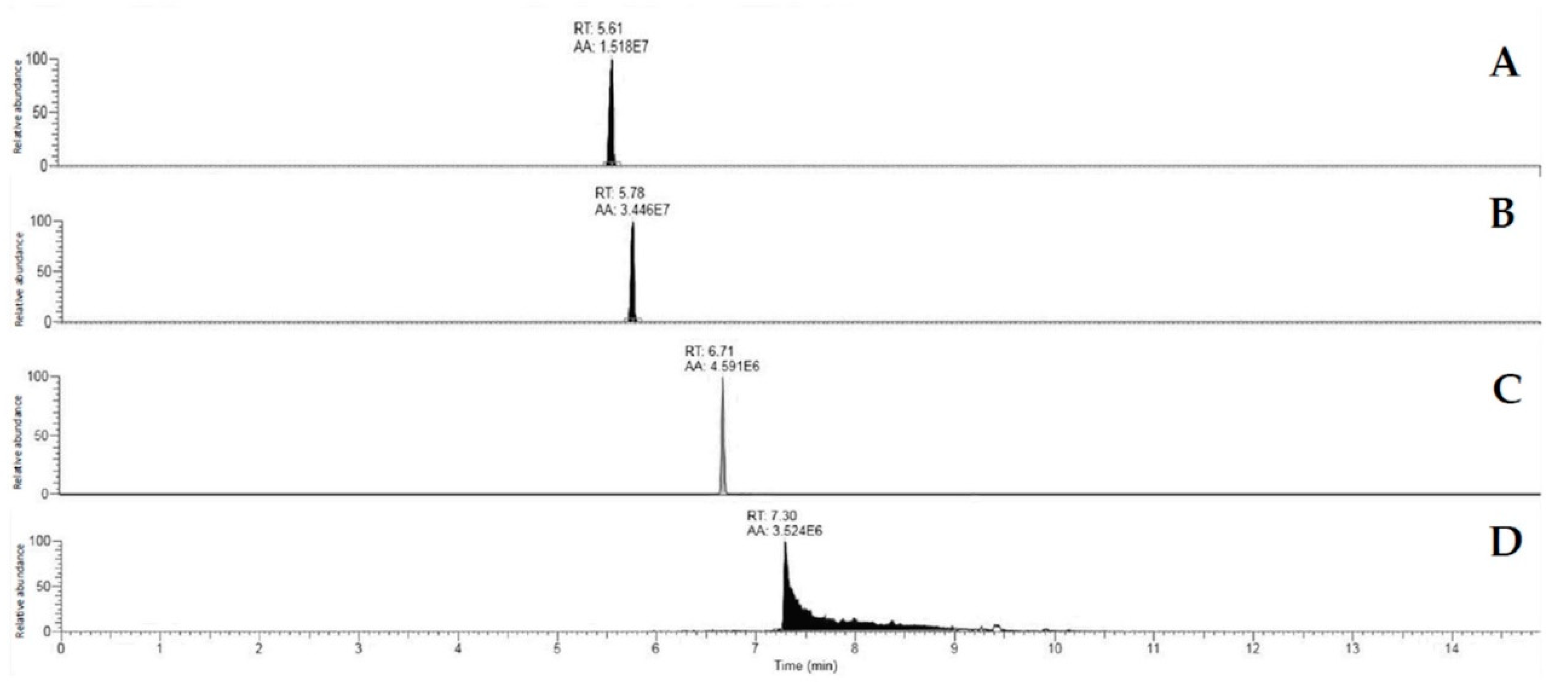
2.6.2. Extraction and Quantification of Mycotoxins
OTA Extraction
Aflatoxins Extraction
CPA Extraction
Recovery Experiments and Linearity Range
Mycotoxin Quantification by Orbitrap Q Exactive Plus Analysis
Mycotoxin Determination Sensitivity
2.7. Statistical Analysis
3. Results
3.1. Enterotoxigenic Potential of Gram-Positive, Catalase-Positive Cocci
3.2. Selection and Identification of Gram-Positive, Catalase-Positive Cocci Isolates with Antifungal Activity
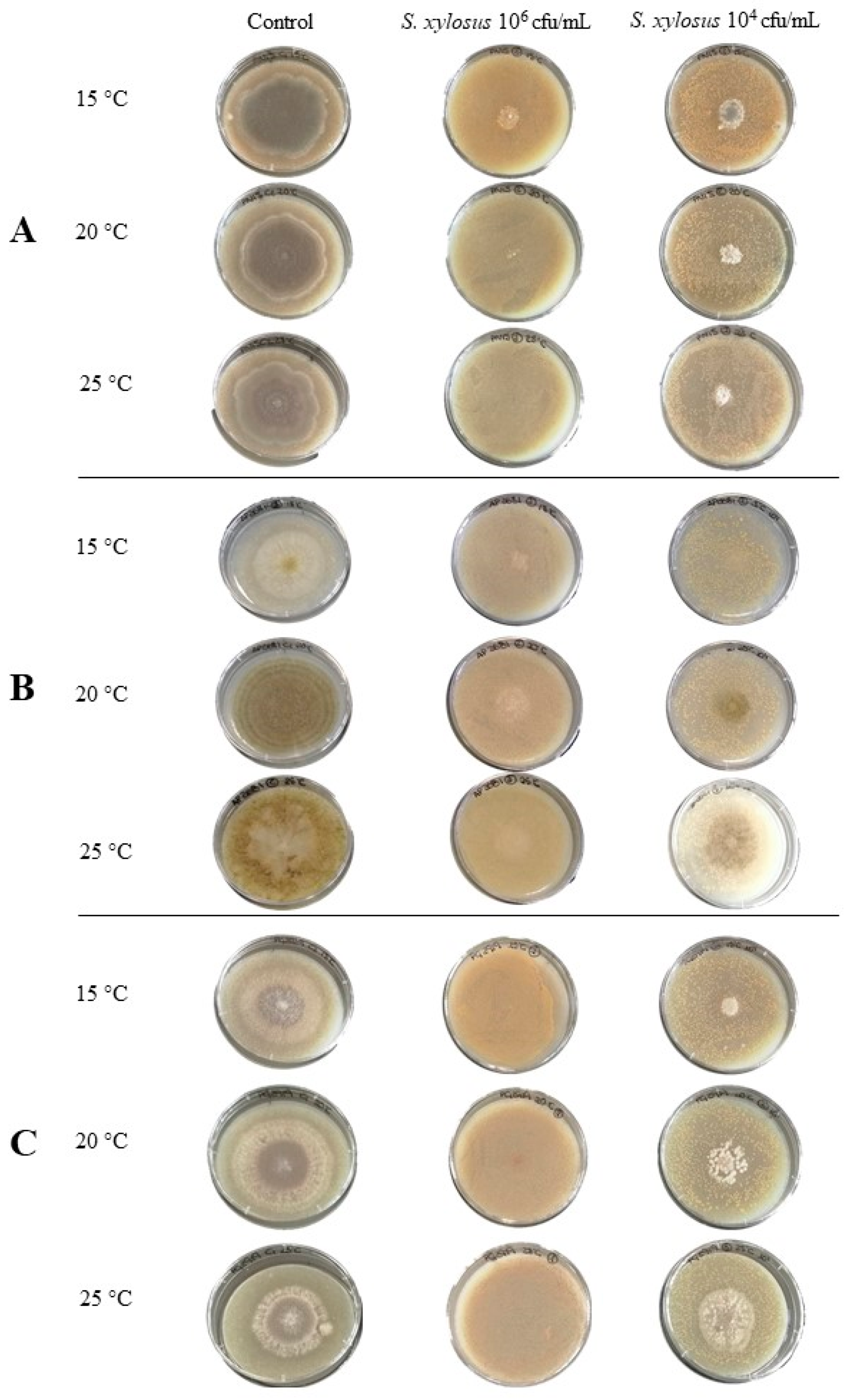
3.3. Antifungal Activity of S. xylosus in a Dry-Cured Ham Model System
3.3.1. Effect on Mould Growth
3.3.2. Effect on Mycotoxin Production
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Battilani, P.; Pietri, A.; Giorni, P.; Formenti, S.; Bertuzzi, T.; Toscani, T.; Virgili, R.; Kozakiewicz, Z. Penicillium populations in dry-cured ham manufacturing plants. J. Food Prot. 2007, 70, 975–980. [Google Scholar] [CrossRef] [PubMed]
- López-Díaz, T.-M.; Santos, J.-A.A.; García-López, M.-L.; Otero, A. Surface mycoflora of a Spanish fermented meat sausage and toxigenicity of Penicillium isolates. Int. J. Food Microbiol. 2001, 68, 69–74. [Google Scholar] [CrossRef]
- Núñez, F.; Rodríguez, M.M.; Bermúdez, M.E.; Córdoba, J.J.; Asensio, M.A. Composition and toxigenic potential of the mould population on dry-cured Iberian ham. Int. J. Food Microbiol. 1996, 32, 185–197. [Google Scholar] [CrossRef]
- Martín, A.; Córdoba, J.J.; Aranda, E.; Córdoba, M.G.; Asensio, M.A. Contribution of a selected fungal population to the volatile compounds on dry-cured ham. Int. J. Food Microbiol. 2006, 110, 8–18. [Google Scholar] [CrossRef]
- Martín, A.; Córdoba, J.J.; Núñez, F.; Benito, M.J.; Asensio, M.A. Contribution of a selected fungal population to proteolysis on dry-cured ham. Int. J. Food Microbiol. 2004, 94, 55–66. [Google Scholar] [CrossRef]
- Núñez, F.; Westphal, C.D.; Bermúdez, E.; Asensio, M.A. Production of secondary metabolites by some terverticillate penicillia on carbohydrate-rich and meat substrates. J. Food Prot. 2007, 70, 2829–2836. [Google Scholar] [CrossRef]
- Bertuzzi, T.; Gualla, A.; Morlacchini, M.; Pietri, A. Direct and indirect contamination with ochratoxin A of ripened pork products. Food Control 2013, 34, 79–83. [Google Scholar] [CrossRef]
- Rodríguez, A.; Rodríguez, M.; Martín, A.; Delgado, J.; Córdoba, J.J. Presence of ochratoxin A on the surface of dry-cured Iberian ham after initial fungal growth in the drying stage. Meat Sci. 2012, 92, 728–734. [Google Scholar] [CrossRef]
- Sánchez-Montero, L.; Córdoba, J.J.; Peromingo, B.; Álvarez, M.; Núñez, F. Effects of environmental conditions and substrate on growth and ochratoxin A production by Penicillium verrucosum and Penicillium nordicum: Relative risk assessment of OTA in dry-cured meat products. Food Res. Int. 2019, 121, 604–611. [Google Scholar] [CrossRef]
- Vipotnik, Z.; Rodríguez, A.; Rodrigues, P. Aspergillus westerdijkiae as a major ochratoxin A risk in dry-cured ham based-media. Int. J. Food Microbiol. 2017, 241, 244–251. [Google Scholar] [CrossRef]
- Sonjak, S.; Ličen, M.; Frisvad, J.C.; Gunde-Cimerman, N. Salting of dry-cured meat. A potential cause of contamination with the ochratoxin A-producing species Penicillium nordicum. Food Microbiol. 2011, 28, 1111–1116. [Google Scholar] [CrossRef] [PubMed]
- Virgili, R.; Simoncini, N.; Toscani, T.; Camardo Leggieri, M.; Formenti, S.; Battilani, P. Biocontrol of Penicillium nordicum growth and Ochratoxin A production by native yeasts of dry cured ham. Toxins 2012, 4, 68–82. [Google Scholar] [CrossRef] [PubMed]
- Ostry, V.; Malir, F.; Toman, J.; Grosse, Y. Mycotoxins as human carcinogens-the IARC Monographs classification. Mycotoxin Res. 2017, 33, 65–73. [Google Scholar] [CrossRef]
- Peromingo, B.; Rodríguez, A.; Bernáldez, V.; Delgado, J.; Rodríguez, M. Effect of temperature and water activity on growth and aflatoxin production by Aspergillus flavus and Aspergillus parasiticus on cured meat model systems. Meat Sci. 2016, 122, 76–83. [Google Scholar] [CrossRef]
- Pleadin, J.; Malenica, M.; Vah, N.; Milone, S.; Safti, L. Survey of aflatoxin B1 and ochratoxin A occurrence in traditional meat products coming from Croatian households and markets. Food Control 2015, 52, 71–77. [Google Scholar] [CrossRef]
- Rodríguez, A.; Rodríguez, M.; Martín, A.; Nuñez, F.; Córdoba, J.J. Evaluation of hazard of aflatoxin B1, ochratoxin A and patulin production in dry-cured ham and early detection of producing moulds by qPCR. Food Control 2012, 27, 118–126. [Google Scholar] [CrossRef]
- Rojas, F.J.; Jodral, M.; Gosalvez, F.; Pozo, R. Mycoflora and toxigenic Aspergillus flavus in Spanish dry-cured ham. Int. J. Food Microbiol. 1991, 13, 249–256. [Google Scholar] [CrossRef]
- Peromingo, B.; Rodríguez, M.; Núñez, F.; Silva, A.; Rodríguez, A. Sensitive determination of cyclopiazonic acid in dry-cured ham using a QuEChERS method and UHPLC–MS/MS. Food Chem. 2018, 263, 275–282. [Google Scholar] [CrossRef]
- Cvetnić, Z.; Pepeljnjak, S. Aflatoxin-producing potential of Aspergillus flavus and Aspergillus parasiticus isolated from samples of smoked-dried meat. Food Nahrung 1995, 39, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Markov, K.; Pleadin, J.; Bevardi, M.; Vahčić, N.; Sokolić-Mihalak, D.; Frece, J. Natural occurrence of aflatoxin B1, ochratoxin A and citrinin in Croatian fermented meat products. Food Control 2013, 34, 312–317. [Google Scholar] [CrossRef]
- IARC (International Agency for Research on Cancer). Aflatoxins. In Chemical Agents and Related Occupations. A Review of Human Carcinogens; IARC: Lyon, France, 2012; Volume 100 F, pp. 225–248. ISBN 978-92-832-1323-9. [Google Scholar]
- Alapont, C.; López-Mendoza, M.C.; Gil, J.V.; Martínez-Culebras, P.V. Mycobiota and toxigenic Penicillium species on two Spanish dry-cured ham manufacturing plants. Food Addit. Contam. Part A 2014, 31, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Ostry, V.; Toman, J.; Grosse, Y.; Malir, F. Cyclopiazonic acid: 50th anniversary of its discovery. World Mycotoxin J. 2018, 11, 1–14. [Google Scholar] [CrossRef]
- Andrade, M.J.; Thorsen, L.; Rodríguez, A.; Córdoba, J.J.; Jespersen, L. Inhibition of ochratoxigenic moulds by Debaryomyces hansenii strains for biopreservation of dry-cured meat products. Int. J. Food Microbiol. 2014, 170, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Cebrián, E.; Rodríguez, M.; Peromingo, B.; Bermúdez, E.; Núñez, F. Efficacy of the combined protective cultures of Penicillium chrysogenum and Debaryomyces hansenii for the control of ochratoxin a hazard in dry-cured ham. Toxins 2019, 11, 710. [Google Scholar] [CrossRef]
- Iacumin, L.; Manzano, M.; Andyanto, D.; Comi, G. Biocontrol of ochratoxigenic moulds (Aspergillus ochraceus and Penicillium nordicum) by Debaryomyces hansenii and Saccharomycopsis fibuligera during speck production. Food Microbiol. 2017, 62, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Peromingo, B.; Núñez, F.; Rodríguez, A.; Alía, A.; Andrade, M.J. Potential of yeasts isolated from dry-cured ham to control ochratoxin A production in meat models. Int. J. Food Microbiol. 2018, 268, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Peromingo, B.; Andrade, M.J.; Delgado, J.; Sánchez-Montero, L.; Núñez, F. Biocontrol of aflatoxigenic Aspergillus parasiticus by native Debaryomyces hansenii in dry-cured meat products. Food Microbiol. 2019, 82, 269–276. [Google Scholar] [CrossRef]
- Bernáldez, V.; Rodríguez, A.; Martín, A.; Lozano, D.; Córdoba, J.J. Development of a multiplex qPCR method for simultaneous quantification in dry-cured ham of an antifungal-peptide Penicillium chrysogenum strain used as protective culture and aflatoxin-producing moulds. Food Control 2014, 36, 257–265. [Google Scholar] [CrossRef]
- Delgado, J.; Peromingo, B.; Rodríguez, A.; Rodríguez, M. Biocontrol of Penicillium griseofulvum to reduce cyclopiazonic acid contamination in dry-fermented sausages. Int. J. Food Microbiol. 2019, 293, 1–6. [Google Scholar] [CrossRef]
- Rodríguez, A.; Bernáldez, V.; Rodríguez, M.; Andrade, M.J.; Núñez, F.; Córdoba, J.J. Effect of selected protective cultures on ochratoxin A accumulation in dry-cured Iberian ham during its ripening process. LWT-Food Sci. Technol. 2015, 60, 923–928. [Google Scholar] [CrossRef]
- Álvarez, M.; Rodríguez, A.; Peromingo, B.; Núñez, F.; Rodríguez, M. Enterococcus faecium: A promising protective culture to control growth of ochratoxigenic moulds and mycotoxin production in dry-fermented sausages. Mycol. Res. 2020, 36, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Delgado, J.; Rodríguez, A.; García, A.; Núñez, F.; Asensio, M. Inhibitory effect of PgAFP and protective cultures on Aspergillus parasiticus growth and aflatoxins production on dry-fermented sausage and cheese. Microorganisms 2018, 6, 69. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.; Núñez, F.; Córdoba, J.J.; Bermúdez, E.; Asensio, M.A. Gram-positive, catalase-positive cocci from dry cured Iberian ham and their enterotoxigenic potential. Appl. Environ. Microbiol. 1996, 62, 1897–1902. [Google Scholar] [CrossRef]
- Leroy, S.; Vermassen, A.; Ras, G.; Talon, R. Insight into the genome of Staphylococcus xylosus, a ubiquitous species well adapted to meat products. Microorganisms 2017, 5, 52. [Google Scholar] [CrossRef] [PubMed]
- Martín, B.; Garriga, M.; Hugas, M.; Bover-Cid, S.; Veciana-Nogués, M.T.; Aymerich, T. Molecular, technological and safety characterization of Gram-positive catalase-positive cocci from slightly fermented sausages. Int. J. Food Microbiol. 2006, 107, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.; Núñez, F.; Córdoba, J.J.; Sanabria, C.; Bermúdez, E.; Asensio, M.A. Characterization of Staphylococcus spp. and Micrococcus spp. isolated from Iberian ham throughout the ripening process. Int. J. Food Microbiol. 1994, 24, 329–335. [Google Scholar] [CrossRef]
- Fiorentini, Â.M.; Sawitzki, M.C.; Bertol, T.M.; Sant’Anna, E.S. Viability of Staphylococcus xylosus isolated from artisanal sausages for application as starter cultures in meat products. Braz. J. Microbiol. 2009, 129–133. [Google Scholar] [CrossRef]
- Simonová, M.; Strompfová, V.; Marciňáková, M.; Lauková, A.; Vesterlund, S.; Moratalla, M.L.; Bover-Cid, S.; Vidal-Carou, C. Characterization of Staphylococcus xylosus and Staphylococcus carnosus isolated from Slovak meat products. Meat Sci. 2006, 73, 559–564. [Google Scholar] [CrossRef]
- Vermassen, A.; de la Foye, A.; Loux, V.; Talon, R.; Leroy, S. Transcriptomic analysis of Staphylococcus xylosus in the presence of nitrate and nitrite in meat reveals its response to nitrosative stress. Front. Microbiol. 2014, 5, 1–15. [Google Scholar] [CrossRef]
- Corbiere Morot-Bizot, S.; Leroy, S.; Talon, R. Monitoring of staphylococcal starters in two French processing plants manufacturing dry fermented sausages. J. Appl. Microbiol. 2007, 102, 238–244. [Google Scholar] [CrossRef]
- Kamala, A.; Ortiz, J.; Kimanya, M.; Haesaert, G.; Donoso, S.; Tiisekwa, B.; De Meulenaer, B. Multiple mycotoxin co-occurrence in maize grown in three agro-ecological zones of Tanzania. Food Control 2015, 54, 208–215. [Google Scholar] [CrossRef]
- Sánchez-Montero, L.; Córdoba, J.J.; Alía, A.; Peromingo, B.; Núñez, F. Effect of Spanish smoked paprika “Pimentón de La Vera” on control of ochratoxin A and aflatoxins production on a dry-cured meat model system. Int. J. Food Microbiol. 2019, 308, 108303. [Google Scholar] [CrossRef] [PubMed]
- Núñez, F.; Lara, M.S.; Peromingo, B.; Delgado, J.; Sánchez-Montero, L.; Andrade, M.J. Selection and evaluation of Debaryomyces hansenii isolates as potential bioprotective agents against toxigenic penicillia in dry-fermented sausages. Food Microbiol. 2015, 46, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Bernáldez, V.; Córdoba, J.J.; Rodríguez, M.; Cordero, M.; Polo, L.; Rodríguez, A. Effect of Penicillium nalgiovense as protective culture in processing of dry-fermented sausage “salchichón”. Food Control 2013, 32, 69–76. [Google Scholar] [CrossRef]
- dos Cruxen, C.E.S.; Funck, G.D.; da Dannenberg, G.S.; Haubert, L.; de Marques, J.L.; Kroning, I.S.; Chaves, F.C.; da Silva, W.P.; Fiorentini, Â.M. Characterization of Staphylococcus xylosus LQ3 and its application in dried cured sausage. LWT-Food Sci. Technol. 2017, 86, 538–543. [Google Scholar] [CrossRef]
- Olesen, P.T.; Meyer, A.S.; Stahnke, L.H. Generation of flavour compounds in fermented sausages-the influence of curing ingredients, Staphylococcus starter culture and ripening time. Meat Sci. 2004, 66, 675–687. [Google Scholar] [CrossRef]
- Gong, A.D.; Sun, G.J.; Zhao, Z.Y.; Liao, Y.C.; Zhang, J.B. Staphylococcus saprophyticus L-38 produces volatile 3,3-dimethyl-1,2-epoxybutane with strong inhibitory activity against Aspergillus flavus germination and aflatoxin production. World Mycotoxin J. 2019, 1–12. [Google Scholar] [CrossRef]
- Alijani, Z.; Amini, J.; Ashengroph, M.; Bahramnejad, B. Antifungal activity of volatile compounds produced by Staphylococcus sciuri strain MarR44 and its potential for the biocontrol of Colletotrichum nymphaeae, causal agent strawberry anthracnose. Int. J. Food Microbiol. 2019, 307, 108276. [Google Scholar] [CrossRef]
- Asensio, M.A.; Núñez, F.; Delgado, J.; Bermúdez, E. Control of toxigenic molds in food processing. In Microbial Food Safety and Preservation Techniques; Rai, V.R., Bai, A.J., Eds.; CRC Press (Taylor and Francis): Boca Raton, FL, USA, 2014; pp. 329–357. [Google Scholar]
- Dalié, D.K.D.; Deschamps, A.M.; Richard-Forget, F. Lactic acid bacteria-Potential for control of mould growth and mycotoxins: A review. Food Control 2010, 21, 370–380. [Google Scholar] [CrossRef]
- Delgado, J.; Acosta, R.; Rodríguez-Martín, A.; Bermúdez, E.; Núñez, F.; Asensio, M.A. Growth inhibition and stability of PgAFP from Penicillium chrysogenum against fungi common on dry-ripened meat products. Int. J. Food Microbiol. 2015, 205, 23–29. [Google Scholar] [CrossRef]
- Álvarez, M.; Núñez, F.; Delgado, J.; Andrade, M.J.; Rodríguez, M.; Rodríguez, A. Competitiveness of three biocontrol candidates against ochratoxigenic Penicillium nordicum under dry-cured meat environmental and nutritional conditions. Fungal Biol. 2020, in press. [Google Scholar] [CrossRef]
- Delgado, J.; Núñez, F.; Asensio, M.A.; Owens, R.A. Quantitative proteomics of Penicillium nordicum profiles and ochratoxin A repression by protective cultures. Int. J. Food Microbiol. 2019, 305, 108243. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, A.; Medina, Á.; Córdoba, J.J.; Magan, N. The influence of salt (NaCl) on ochratoxin A biosynthetic genes, growth and ochratoxin A production by three strains of Penicillium nordicum on a dry-cured ham-based medium. Int. J. Food Microbiol. 2014, 178, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Meftah, S.; Abid, S.; Dias, T.; Rodrigues, P. Mechanisms underlying the effect of commercial starter cultures and a native yeast on ochratoxin A production in meat products. LWT 2020, 117, 108611. [Google Scholar] [CrossRef]
- Luz, C.; Ferrer, J.; Mañes, J.; Meca, G. Toxicity reduction of ochratoxin A by lactic acid bacteria. Food Chem. Toxicol. 2018, 112, 60–66. [Google Scholar] [CrossRef]


| Isolate | Identification | Accession Number | % Identity |
|---|---|---|---|
| Ku13 | K. uropygioeca | NR_157676.1 | 95.88 |
| Ku15 | K. uropygioeca | NR_157676.1 | 98.72 |
| Ku40 | K. uropygioeca | NR_157676.1 | 100 |
| Se28 | S. equorum | MK015791.1 | 99.86 |
| Se29 | S. equorum | MN229550.1 | 99.79 |
| Se31 | S. equorum | MN229551.1 | 99.91 |
| Se32 | S. equorum | MN758799.1 | 98.28 |
| Se39 | S. equorum | KY940339.1 | 99.85 |
| Se41 | S. equorum | MK796070.1 | 99.92 |
| Se47 | S. equorum | MN758799.1 | 99.64 |
| Se50 | S. equorum | KP224447.1 | 99.56 |
| Se53 | S. equorum | KY940339.1 | 99.86 |
| Ss21 | S. saprophyticus | KU922358.1 | 99.02 |
| Ss42 | S. saprophyticus | MF457583.1 | 99.4 |
| Ss46 | S. saprophyticus | MF457583.1 | 99.7 |
| Ss7 | S. saprophyticus | MN229561.1 | 98.52 |
| Ss9 | S. saprophyticus | KT717631.1 | 99 |
| Sv33 | S. vitulinus | KM378591.1 | 99.69 |
| Sw16 | S. warneri | MK414943.1 | 98.8 |
| Sx8 | S. xylosus | HM854231.1 | 99 |
| S17 | S. pasteuri/S. warneri | MH930440.1/MN181250.1 | 99.79 |
| S19 | S. pasteuri/S. warneri | MH930440.1/MN421516.1 | 99.79 |
| 15 °C | 20 °C | 25 °C | ||||
|---|---|---|---|---|---|---|
| Mould | Control | Sx8 | Control | Sx8 | Control | Sx8 |
| Pn15 | 3.93 ± 1.36 | <LOQ *,a | 18.76 ± 3.22 | <LOQ * | 66.59 ± 19.52 | <LOQ * |
| Pn92 | 2.75 ± 0.49 | <LOD *,b | 26.61 ± 4.82 | <LOD * | 89.53 ± 14.90 | <LOD * |
| Pn856 | 31.54 ± 0.07 | 4.23 ± 0.35 * | 260.64 ± 71.93 | 64.26 ± 11.72 * | 389.73 ± 57.65 | 860.13 ± 357.03 * |
| 15 °C | 20 °C | 25 °C | ||||
|---|---|---|---|---|---|---|
| Mould | Control | Sx8 | Control | Sx8 | Control | Sx8 |
| Af65 | 2.09 ± 0.25 | 0.18 ± 0.03 * | 41.06 ± 6.23 | 0.28 ± 0.09 * | 28.49 ± 3.07 | 2.08 ± 2.21 * |
| Ap2681 | 0.08 ± 0.02 | 0.01 ± 0.01 * | 0.06 ± 0.05 | 0.01 ± 0.01 | 0.10 ± 0.02 | <LOD *,a |
| 15 °C | 20 °C | 25 °C | ||||
|---|---|---|---|---|---|---|
| Mould | Control | Sx8 | Control | Sx8 | Control | Sx8 |
| Ap2681 | 0.73 ± 0.34 | <LOD *,a | 4.85 ± 8.24 | <LOD * | 0.91 ± 0.30 | <LOD * |
| 15 °C | 20 °C | 25 °C | ||||
|---|---|---|---|---|---|---|
| Mould | Control | SX8 | Control | SX8 | Control | SX8 |
| Pg2919 | 1270.4 ± 431.9 | 888.5 ± 258.3 | 2427.8 ± 724.8 | 1793.9 ± 34.7 * | 4492.9 ± 227.0 | 2694.1 ± 881.9 * |
| Pg14319 | 2804.2 ± 802.8 | 1160.0 ± 165.0 * | 4120.4 ± 944.7 | 2932.9 ± 651.9 | 6298.7 ± 2.9 | 5358.9 ± 382.4 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cebrián, E.; Núñez, F.; Gálvez, F.J.; Delgado, J.; Bermúdez, E.; Rodríguez, M. Selection and Evaluation of Staphylococcus xylosus as a Biocontrol Agent against Toxigenic Moulds in a Dry-Cured Ham Model System. Microorganisms 2020, 8, 793. https://doi.org/10.3390/microorganisms8060793
Cebrián E, Núñez F, Gálvez FJ, Delgado J, Bermúdez E, Rodríguez M. Selection and Evaluation of Staphylococcus xylosus as a Biocontrol Agent against Toxigenic Moulds in a Dry-Cured Ham Model System. Microorganisms. 2020; 8(6):793. https://doi.org/10.3390/microorganisms8060793
Chicago/Turabian StyleCebrián, Eva, Félix Núñez, Fernando J. Gálvez, Josué Delgado, Elena Bermúdez, and Mar Rodríguez. 2020. "Selection and Evaluation of Staphylococcus xylosus as a Biocontrol Agent against Toxigenic Moulds in a Dry-Cured Ham Model System" Microorganisms 8, no. 6: 793. https://doi.org/10.3390/microorganisms8060793
APA StyleCebrián, E., Núñez, F., Gálvez, F. J., Delgado, J., Bermúdez, E., & Rodríguez, M. (2020). Selection and Evaluation of Staphylococcus xylosus as a Biocontrol Agent against Toxigenic Moulds in a Dry-Cured Ham Model System. Microorganisms, 8(6), 793. https://doi.org/10.3390/microorganisms8060793







