Radial Extracorporeal Shock Wave Therapy against Cutibacterium acnes Implant-Associated Infections: An in Vitro Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Biomaterials
2.2. Susceptibility Assessment of C. Acnes Planktonic Form to rESWT
2.3. Mature Biofilm Production Confirmation
2.4. Susceptibility Assessment of C. acnes Biofilms to Radial Shockwaves
2.5. Treatment Arms
2.6. Sample Size Calculation and Statistical Analysis
3. Results
3.1. Antimicrobial Susceptibility of C. acnes Planktonic Form to Radial Shockwaves
3.2. Antibiofilm Activity of rESWT
4. Discussion
4.1. Biomaterial and Strain Selection
4.2. rESWT Application and Reliability in Cell Viability Measurements
4.3. Effect of rESWT against C. acnes Planktonic and Biofilm Forms
4.4. Study Limitations and Implications for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nelson, G.N.; Davis, D.E.; Namdari, S. Outcomes in the treatment of periprosthetic joint infection after shoulder arthroplasty: A systematic review. J Shoulder Elbow Surg. 2016, 25, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Klouche, S.; Sariali, E.; Mamoudy, P. Cost analysis of revisions of the total hip prostheses infected. Rev. Chir. Orthop. Traumatol. 2010, 96, 167–175. [Google Scholar]
- Achermann, Y.; Goldstein, E.J.; Coenye, T.; Shirtliff, M.E. Propionibacterium acnes: From commensal to opportunistic biofilm-associated implant pathogen. Clin. Microbiol. Rev. 2014, 27, 419–440. [Google Scholar] [CrossRef] [PubMed]
- Bayston, R.; Ashraf, W.; Barker-Davies, R.; Tucker, E.; Clement, R.; Clayton, J.; Freeman, B.J.; Nuradeen, B. Biofilm formation by Propionibacterium acnes on biomaterials in vitro and in vivo: Impact on diagnosis and treatment. J. Biomed. Mater. Res. 2007, 81, 705–709. [Google Scholar] [CrossRef]
- Trampuz, A.; Osmon, D.R.; Hanssen, A.D.; Steckelberg, J.M.; Patel, R. Molecular and antibiofilm approaches to prosthetic joint infection. Clin. Orthop. Relat. Res. 2003, 414, 69–88. [Google Scholar] [CrossRef]
- Aslam, S. Effect of antibacterials on biofilms. Am. J. Infect. Control. 2008, 36, S175.e9–S17511. [Google Scholar] [CrossRef]
- Zimmerli, W.; Trampuz, A.; Ochsner, P.E. Prosthetic-joint infections. N. Engl. J. Med. 2004, 351, 1645–1654. [Google Scholar] [CrossRef]
- Segreti, J.; Nelson, J.A.; Trenholme, G.M. Prolonged suppressive antibiotic therapy for infected orthopedic prostheses. Clin. Infect. Dis. 1998, 27, 711–713. [Google Scholar] [CrossRef]
- Høiby, N.; Bjarnsholt, T.; Moser, C.; Bassi, G.L.; Coenye, T.; Donelli, G.; Hall-Stoodley, L.; Hola, V.; Imbert, C.; Kirketerp-Moller, K.; et al. ESCMID guideline for the diagnosis and treatment of biofilm infections 2014. Clin. Microbiol. Infect. 2015, 21 (Suppl. 1), S1–S25. [Google Scholar] [CrossRef]
- Hsu, J.E.; Bumgarner, R.E.; Matsen, F.A., 3rd. Propionibacterium in Shoulder Arthroplasty: What We Think We Know Today. J. Bone Joint Surg. Am. 2016, 98, 597–606. [Google Scholar] [CrossRef]
- Gerdesmeyer, L.; von Eiff, C.; Horn, C.; Henne, M.; Roessner, M.; Diehl, P.; Gollwitzer, H. Antibacterial effects of extracorporeal shock waves. Ultrasound Med. Biol. 2005, 31, 115–119. [Google Scholar] [CrossRef]
- Gollwitzer, H.; Horn, C.; von Eiff, C.; Henne, M.; Gerdesmeyer, L. Antibakterieller Effekt hochenergetischer extrakorporaler Stoßwellen: Ein in vitro Nachweis. Z. Orthop. Ihre Grenzgeb. 2004, 142, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Reilly, J.M.; Bluman, E.; Tenforde, A.S. Effect of Shockwave Treatment for Management of Upper and Lower Extremity Musculoskeletal Conditions: A Narrative Review. PMR 2018, 10, 1385–1403. [Google Scholar] [CrossRef] [PubMed]
- Horn, C.; Mengele, K.; Gerdesmeyer, L.; Gradinger, R.; Gollwitzer, H. The effect of antibacterial acting extracorporeal shockwaves on bacterial cell integrity. Med. Sci. Monit. 2009, 15, BR364–BR369. [Google Scholar] [PubMed]
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.F.; Alarcon, E.I. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, e01067. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J.; Huang, H.Y.; Yang, K.; Wang, F.S.; Wong, M. Pathomechanism of shock wave injuries on femoral artery, vein and nerve. An experimental study in dogs. Injury 2002, 33, 439–446. [Google Scholar] [CrossRef]
- Gollwitzer, H.; Roessner, M.; Langer, R.; Gloeck, T.; Diehl, P.; Horn, C.; Stemberger, A.; von Eiff, C.; Gerdesmeyer, L. Safety and effectiveness of extracorporeal shockwave therapy: Results of a rabbit model of chronic osteomyelitis. Ultrasound Med. Biol. 2009, 35, 595–602. [Google Scholar] [CrossRef]
- Van der Worp, H.; van den Akker-Scheek, I.; van Schie, H.; Zwerver, J. ESWT for tendinopathy: Technology and clinical implications. Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 1451–1458. [Google Scholar] [CrossRef]
- Howlin, R.P.; Winnard, C.; Angus, E.M.; Frapwell, C.J.; Webb, J.S.; Cooper, J.J.; Aiken, S.S.; Bishop, J.Y.; Stoodley, P. Prevention of Propionibacterium acnes biofilm formation in prosthetic infections in vitro. J. Shoulder Elbow Surg. 2017, 26, 553–563. [Google Scholar] [CrossRef]
- Ravn, C.; Furustrand Tafin, U.; Bétrisey, B.; Overgaard, S.; Trampuz, A. Reduced ability to detect surface-related biofilm bacteria after antibiotic exposure under in-vitro conditions. Acta Orthop. 2016, 87, 644–650. [Google Scholar] [CrossRef]
- Tafin, U.F.; Corvec, S.; Betrisey, B.; Zimmerli, W.; Trampuz, A. Role of rifampin against Propionibacterium acnes biofilm in vitro and in an experimental foreign-body infection model. Antimicrob. Agents Chemother. 2012, 56, 1885–1891. [Google Scholar] [CrossRef] [PubMed]
- Roehm, N.W.; Rodgers, G.H.; Hatfield, S.M.; Glasebrook, A.L. An improved colorimetric assay for cell proliferation and viability utilizing the tetrazolium salt XTT. J. Immunol. Methods 1991, 142, 257–265. [Google Scholar] [CrossRef]
- Bjerkan, G.; Witsø, E.; Bergh, K. Sonication is superior to scraping for retrieval of bacteria in biofilm on titanium and steel surfaces in vitro. Acta Orthop. 2009, 80, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Oethinger, M.; Tuohy, M.J.; Procop, G.W.; Bauer, T.W. Improved detection of biofilm-formative bacteria by vortexing and sonication: A pilot study. Clin. Orthop. Relat. Res. 2009, 467, 1360–1364. [Google Scholar] [CrossRef]
- Faggion, C.M., Jr. Guidelines for reporting pre-clinical in vitro studies on dental materials. J. Evid. Based Dent. Pract. 2012, 12, 182–189. [Google Scholar] [CrossRef]
- Fink, B.; Grossmann, A.; Fuerst, M.; Schäfer, P.; Frommelt, L. Two-stage cementless revision of infected hip endoprostheses. Clin. Orthop. Relat. Res. 2009, 467, 1848–1858. [Google Scholar] [CrossRef]
- Hofmann, A.A.; Goldberg, T.D.; Tanner, A.M.; Cook, T.M. Ten-year experience using an articulating antibiotic cement hip spacer for the treatment of chronically infected total hip. J. Arthroplasty 2005, 20, 874–879. [Google Scholar] [CrossRef]
- Sherrell, J.C.; Fehring, T.K.; Odum, S.; Hansen, E.; Zmistowski, B.; Dennos, A.; Kalore, N.; Periprosthetic Infection Consortium. The Chitranjan Ranawat Award: Fate of two-stage reimplantation after failed irrigation and débridement for periprosthetic knee infection. Clin. Orthop. Relat. Res 2011, 469, 18–25. [Google Scholar] [CrossRef]
- Day, J.S.; Lau, E.; Ong, K.L.; Williams, G.R.; Ramsey, M.L.; Kurtz, S.M. Prevalence and projections of total shoulder and elbow arthroplasty in the United States to 2015. J. Shoulder Elbow Surg. 2010, 19, 1115–1120. [Google Scholar] [CrossRef]
- Isefuku, S.; Joyner, C.J.; Simpson, A.H. Gentamicin may have an adverse effect on osteogenesis. J. Orthop. Trauma 2003, 17, 212–216. [Google Scholar] [CrossRef]
- Richter-Hintz, D.; Rieker, J.; Rauch, L.; Homey, B. Sensitivity to constituents of bone cement in a patient with joint prosthesis. Hautarzt 2004, 55, 987–989. [Google Scholar] [PubMed]
- Sterling, G.J.; Crawford, S.; Potter, J.H.; Koerbin, G.; Crawford, R. The pharmacokinetics of Simplex-tobramycin bone cement. J. Bone Joint Surg. Br. 2003, 85, 646–649. [Google Scholar] [CrossRef]
- Thomes, B.; Murray, P.; Bouchier-Hayes, D. Development of resistant strains of Staphylococcus epidermidis on gentamicin-loaded bone cement in vivo. J. Bone Joint Surg. Br. 2002, 84, 758–760. [Google Scholar]
- Potera, C. Antibiotic Resistance: Biofilm Dispersing Agent Rejuvenates Older Antibiotics. Environ. Health Perspect. 2010, 118, A288. [Google Scholar] [CrossRef]
- Geethaa, M.; Singh, A.K.; Asokamania, R.; Gogiac, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Prog. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Truong, V.K.; Lapovok, R.; Estrin, Y.S.; Rundell, S.; Wang, J.Y.; Fluke, C.J.; Crawford, R.J.; Ivanova, E.P. The influence of nano-scale surface roughness on bacterial adhesion to ultrafine-grained titanium. Biomaterials 2010, 31, 3674–3683. [Google Scholar] [CrossRef] [PubMed]
- Rupp, F.; Scheideler, L.; Olshanska, N.; de Wild, M.; Wieland, M.; Geis-Gerstorfer, J. Enhancing surface free energy and hydrophilicity through chemical modification of microstructured titanium implant surfaces. J. Biomed. Mater. Res. Part A 2006, 76, 323–334. [Google Scholar] [CrossRef]
- Hetrick, E.M.; Schoenfisch, M.H. Reducing implant-related infections: Active release strategies. Chem. Soc. Rev. 2006, 35, 780–789. [Google Scholar] [CrossRef]
- Harris, L.G.; Meredith, D.O.; Eschbach, L.; Richards, R.G. Staphylococcus aureus adhesion to standard micro-rough and electropolished implant materials. J. Mater. Sci. Mater. Med. 2007, 18, 1151–1156. [Google Scholar] [CrossRef]
- Ivanova, E.P.; Mitik-Dineva, N.; Wang, J.; Pham, D.K.; Wright, J.P.; Nicolau, D.V.; Mocanasu, R.C.; Crawford, R.J. Staleya guttiformis attachment on poly(tert-butylmethacrylate) polymeric surfaces. Micron 2008, 39, 1197–1204. [Google Scholar] [CrossRef]
- Mitik-Dineva, N.; Wang, J.; Truong, V.K.; Stoddart, P.; Malherbe, F.; Crawford, R.J.; Ivanova, E.P. Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus attachment patterns on glass surfaces with nanoscale roughness. Curr. Microbiol. 2009, 58, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Wang, H.; Ni, M.; Rui, Y.; Cheng, T.Y.; Cheng, C.K.; Pan, X.; Li, G.; Lin, C. Enhanced osteointegration of medical titanium implant with surface modifications in micro/nanoscale structures. J. Orthop. Transl. 2017, 2, 35–42. [Google Scholar] [CrossRef]
- Wennerberg, A.; Albrektsson, T. Effects of titanium surface topography on bone integration: A systematic review. Clin. Oral Implants Res. 2009, 20 (Suppl. 4), 172–184. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, A.; Lood, R.; Mörgelin, M.; Söderquist, B.; Holst, E.; Collin, M.; Christensson, B.; Rasmussen, M. Biofilm formation by Propionibacterium acnes is a characteristic of invasive isolates. Clin. Microbiol. Infect. 2009, 15, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.I.; Nagahori, R.; Yamada, S.; Sugimoto, S.; Sato, C.; Sato, M.; Iwase, T.; Hashimoto, K.; Mizunoe, Y. The Composition and Structure of Biofilms Developed by Propionibacterium acnes Isolated from Cardiac Pacemaker Devices. Front. Microbiol. 2018, 9, 182. [Google Scholar] [CrossRef] [PubMed]
- Peeters, E.; Nelis, H.J.; Coenye, T. Comparison of multiple methods for quantification of microbial biofilms grown in microtiter plates. J. Microbiol. Methods 2008, 72, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Pettit, R.K.; Weber, C.A.; Kean, M.J.; Hoffmann, H.; Pettit, G.R.; Tan, R.; Franks, K.S.; Horton, M.L. Microplate Alamar blue assay for Staphylococcus epidermidis biofilm susceptibility testing. Antimicrob. Agents Chemother. 2005, 49, 2612–2617. [Google Scholar] [CrossRef]
- Nana, A.; Nelson, S.B.; McLaren, A.; Chen, A.F. What’s New in Musculoskeletal Infection: Update on Biofilms. J. Bone Joint Surg. Am. 2016, 98, 1226–1234. [Google Scholar] [CrossRef]
- Lovati, A.B.; Bottagisio, M.; de Vecchi, E.; Gallazzi, E.; Drago, L. Animal Models of Implant-Related Low-Grade Infections. A Twenty-Year Review. Adv. Exp. Med. Biol. 2017, 97, 29–50. [Google Scholar] [CrossRef]
- Chen, X.; Li, X. Extracorporeal shock wave therapy could be a potential adjuvant treatment for orthopaedic implant-associated infections. J. Med. Hypotheses Ideas 2013, 7, 54–58. [Google Scholar] [CrossRef]
- Leopold, S.S. Editorial: When “safe and effective” becomes dangerous. Clin. Orthop. Relat. Res. 2014, 472, 1999–2001. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Zhao, Y.; Zhang, J.; Han, D.; Chen, C.; Huang, Y.; Chen, X.; Zhang, X.; Wang, T.; Li, X. Increased effects of extracorporeal shock waves combined with gentamicin against staphylococcus aureus biofilms in vitro and in vivo. Ultrasound Med. Biol. 2016, 42, 2245–2252. [Google Scholar] [CrossRef] [PubMed]
- Wanner, S.; Gstöttner, M.; Meirer, R.; Hausdorfer, J.; Fille, M.; Stöckl, B. Low-energy shock waves enhance the susceptibility of staphylococcal biofilms to antimicrobial agents in vitro. J. Bone Joint Surg. Br. 2011, 93, 824–827. [Google Scholar] [CrossRef] [PubMed]

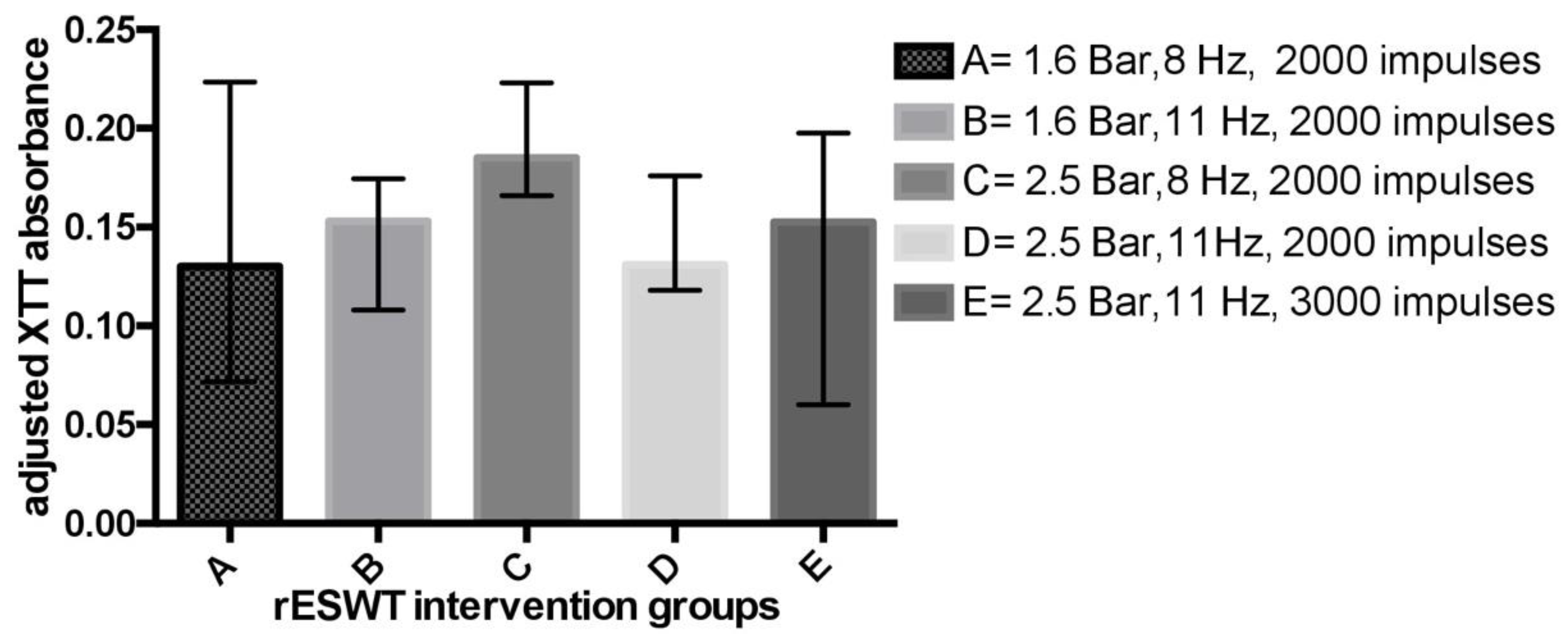
| Treatment Group (n) | Mean Absorbance Difference * (SE) | p Value for Parametric Tests † | Median Absorbance (IQR) | p Value for Non-Parametric Tests † | % Reduction in Bacterial Load Based on Negative Control Measurements (Median) | |
|---|---|---|---|---|---|---|
| Intervention Group | Positive Control Group | |||||
| A (20) (8 Hz, 1.6 Bar, 2000 impulses) | 0.1995 (0.044) | 0.000175 | 0.7705 (0.712 to 0.864) | 0.9245 (0.888 to 0.104) | 0.001 | 54% (0.64) |
| B (20) (11 Hz, 1.6 Bar, 2000 impulses) | N/A | N/A | 0.7937 (0.748 to 0.815) | 0.9245 (0.888 to 0.104) | 0.001 | 46% (0.64) |
| C (20) (8 Hz, 2.5 Bar, 2000 impulses) | 0.138 (0.346) | 0.001 | 0.8255 (0.807 to 0.863) | 0.9245 (0.888 to 0.104) | 0.001 | 35% (0.64) |
| D (20) (11 Hz, 2.5 Bar, 2000 impulses) | 0.188 (0.037) | p = 0.000044 | 0.7720 (0.758 to 0.817) | 0.9245 (0.888 to 0.104) | 0.0003 | 46% (0.64) |
| E (20) (11 Hz, 2.5 Bar, 3000 impulses) | 0.255 (0.055) | p = 0.000121 | 0.8870 (0.795 to 0.932) | 1.0900 (1.014 to 1.240) | 0.001 | 42% (0.61) |
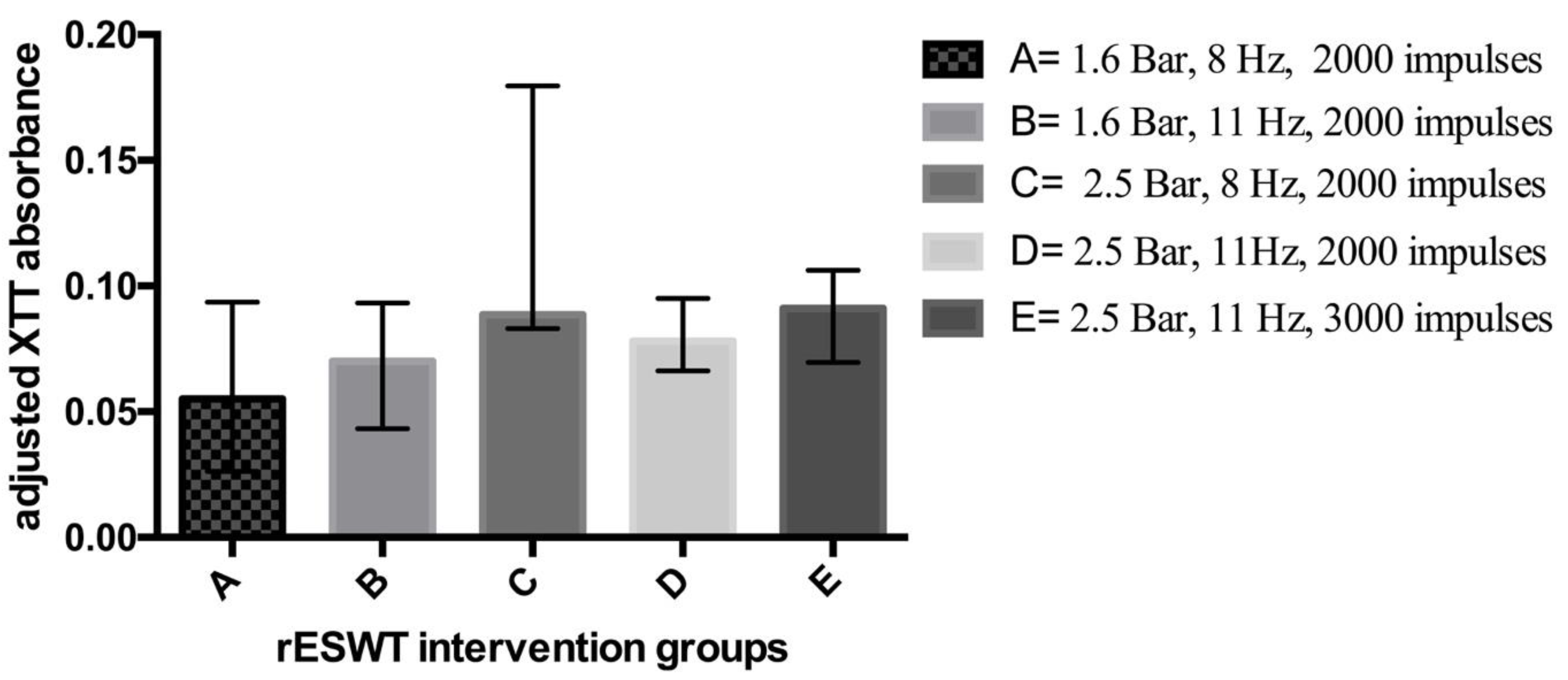
| Treatment Group (n) | Mean Absorbance Difference (SD) | p Value for Parametric Test † | Median Absorbance (IQR) | p Value for Nonparametric Test † | % Reduction in Biofilm Based on Negative Control Measurements (Median) | |
|---|---|---|---|---|---|---|
| Intervention Group | Untreated Control Group | |||||
| A (24) (8 Hz, 1.6 Bar, 2000 impulses) | N/A | N/A | 0.244 (0.215–0.282) | 0.298 (0.247–0.307) | 0.033 | 49% (0.188) |
| B (24) (11 Hz, 1.6 Bar, 2000 impulses) | 0.0005 (0.084) | 0.973 | 0.255 (0.228–0.278) | 0.266 (0.246–0.277) | 0.571 | 14% (0.187) |
| C (24) (8 Hz, 2.5 Bar, 2000 impulses) | N/A | N/A | 0.280 (0.275–0.371) | 0.308 (0.264–0.369) | 0.421 | 25% (0.196) |
| D (24) (11 Hz, 2.5 Bar, 2000 impulses) | N/A | N/A | 0.266 (0.254–0.283) | 0.297 (0.252–0.33) | 0.306 | 29% (0.19) |
| E (24) (11 Hz, 2.5 Bar, 3000 impulses) | N/A | N/A | 0.331 (0.31–0.346) | 0.365 (0.340–0.377) | 0.112 | 24% (0.223) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsikopoulos, K.; Drago, L.; Koutras, G.; Givissis, P.; Vagdatli, E.; Soukiouroglou, P.; Papaioannidou, P. Radial Extracorporeal Shock Wave Therapy against Cutibacterium acnes Implant-Associated Infections: An in Vitro Trial. Microorganisms 2020, 8, 743. https://doi.org/10.3390/microorganisms8050743
Tsikopoulos K, Drago L, Koutras G, Givissis P, Vagdatli E, Soukiouroglou P, Papaioannidou P. Radial Extracorporeal Shock Wave Therapy against Cutibacterium acnes Implant-Associated Infections: An in Vitro Trial. Microorganisms. 2020; 8(5):743. https://doi.org/10.3390/microorganisms8050743
Chicago/Turabian StyleTsikopoulos, Konstantinos, Lorenzo Drago, Georgios Koutras, Panagiotis Givissis, Eleni Vagdatli, Prodromos Soukiouroglou, and Paraskevi Papaioannidou. 2020. "Radial Extracorporeal Shock Wave Therapy against Cutibacterium acnes Implant-Associated Infections: An in Vitro Trial" Microorganisms 8, no. 5: 743. https://doi.org/10.3390/microorganisms8050743
APA StyleTsikopoulos, K., Drago, L., Koutras, G., Givissis, P., Vagdatli, E., Soukiouroglou, P., & Papaioannidou, P. (2020). Radial Extracorporeal Shock Wave Therapy against Cutibacterium acnes Implant-Associated Infections: An in Vitro Trial. Microorganisms, 8(5), 743. https://doi.org/10.3390/microorganisms8050743







