Separation, Characterization, and Handling of Microalgae by Dielectrophoresis
Abstract
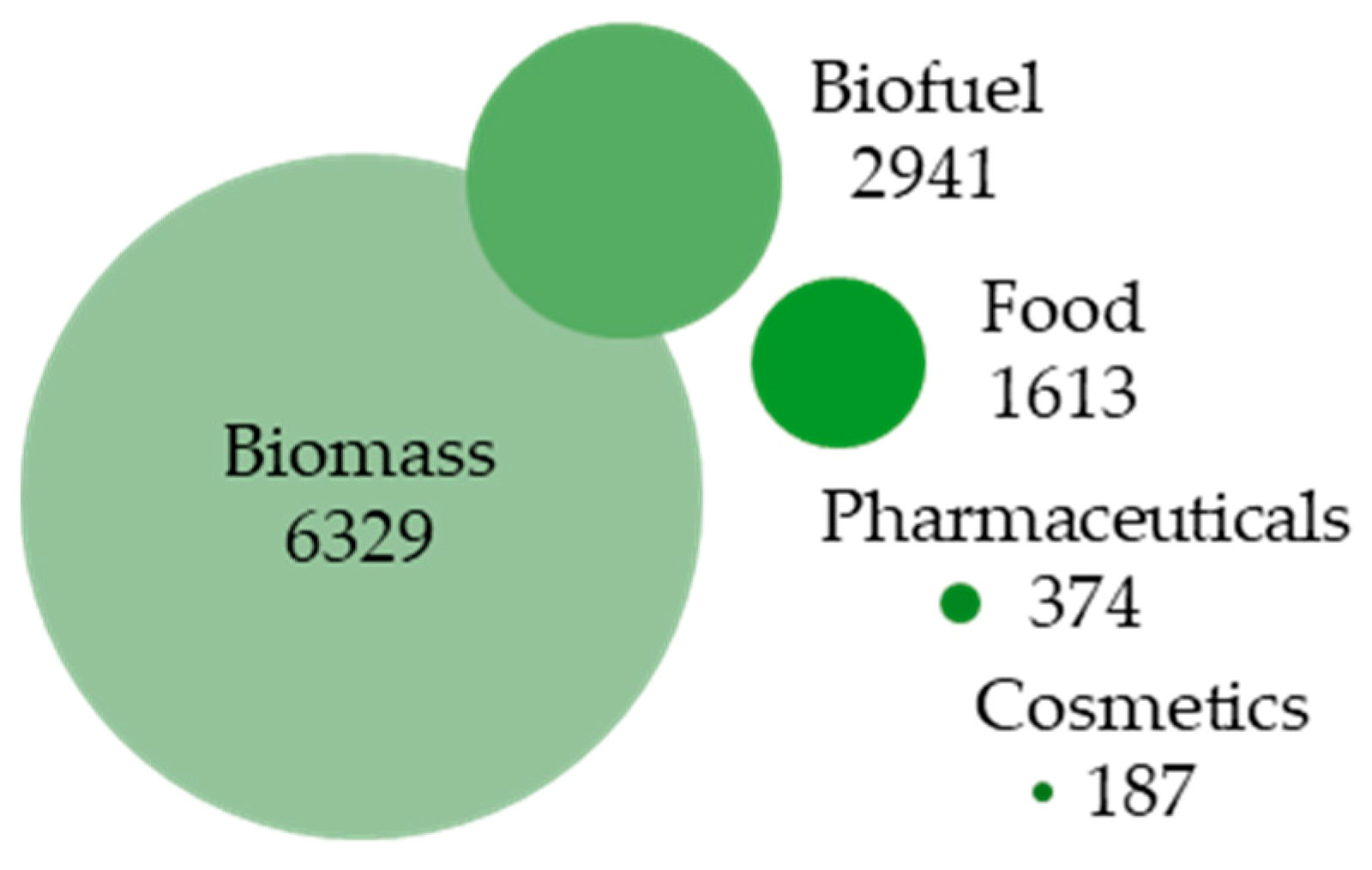
1. Recent Advances in Microalgae Research and Microalgae-Based Bioprocess Development
2. DEP Technology for Cell Population Analysis
- The particle diameter d is the single most important contributor to FDEP.
- FDEP depends on and of both the particle and media (index p and m). Changes of angular frequency may either cause a positive or negative DEP force, depending on whether holds.
- The local electrical field depends on the applied voltage V as well as the electrode design and geometry. The layout of the flow channel and the electrodes thus offer various degrees of freedom for optimizing the DEP force.
3. Overview of DEP Microfluidic Systems for Microalgae Research
4. DEP Microfluidic Devices Categorized Based on Working Principles of Devices
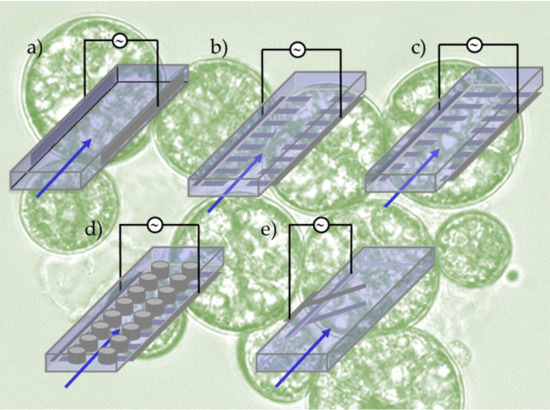
4.1. Trapping Designs Using pDEP Force
4.2. Planar Parallel Surface Electrodes for Cell Trapping
4.3. Micropost Electrode for Cell Trapping
4.4. Sharp-Tip Electrode Design for Cell Trapping
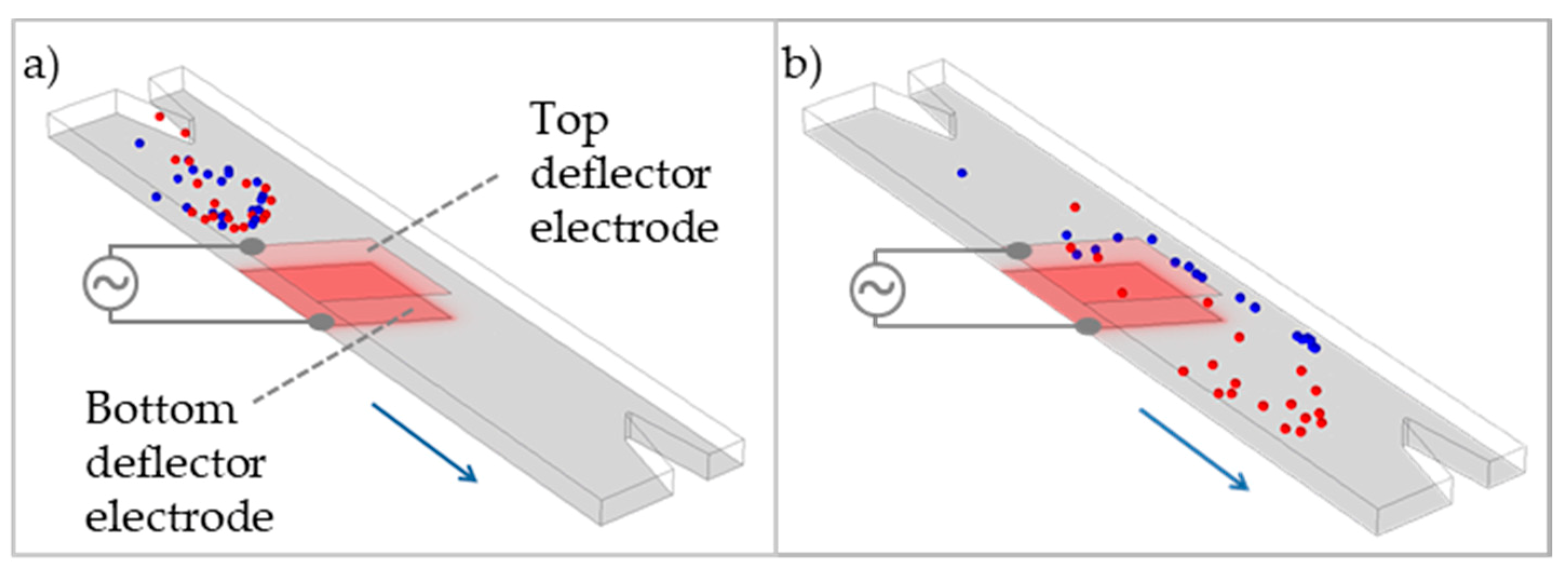
4.5. Flow-Through Deflection Structures for Cell Separation
4.6. Sharp-Tip Electrode for Cell Separation
4.7. Angled Electrodes for Cell Separation
5. DEP Microfluidic Devices Categorized Based on Their Applications
5.1. Cell Trapping and Concentration
5.2. Cell Separation Based on Intracellular Lipid Content
5.3. Cell Separation Based on Their Sizes or From Other Particles
5.4. Microalgae Analysis
5.5. Strain Selection Through Screening
6. Discussion
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Georgianna, D.R.; Mayfield, S.P. Exploiting diversity and synthetic biology for the production of algal biofuels. Nature 2012, 488, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Larkum, A.W.D.; Ross, I.L.; Kruse, O.; Hankamer, B. Selection, breeding and engineering of microalgae for bioenergy and biofuel production. Trends Biotechnol. 2012, 30, 198–204. [Google Scholar] [CrossRef]
- Lee, S.K.; Chou, H.; Ham, T.S.; Lee, T.S.; Keasling, J.D. Metabolic engineering of microorganisms for biofuels production: From bugs to synthetic biology to fuels. Curr. Opin. Biotechnol. 2008, 19, 556–563. [Google Scholar] [CrossRef]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development; General Assembley 70 Session: New York, NY, USA, 2015. [Google Scholar]
- European Comission. A Sustainable Bioeconomy for Europe: Strengthening the Connection between Economy, Society and the Environment; Updated Bioeconomy Strategy; European Comission: Brussels, Belgium, 2018. [Google Scholar]
- Ajjawi, I.; Verruto, J.; Aqui, M.; Soriaga, L.B.; Coppersmith, J.; Kwok, K.; Peach, L.; Orchard, E.; Kalb, R.; Xu, W.; et al. Lipid production in Nannochloropsis gaditana is doubled by decreasing expression of a single transcriptional regulator. Nat. Biotechnol. 2017, 35, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Bajhaiya, A.K.; Moreira, J.Z.; Pittman, J.K. Transcriptional Engineering of Microalgae: Prospects for High-Value Chemicals. Trends Biotechnol. 2017, 35, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as sources of high added-value compounds-a brief review of recent work. Biotechnol. Prog. 2011, 27, 597–613. [Google Scholar] [CrossRef]
- Saha, S.K.; McHugh, E.; Murray, P.; Walsh, D.J. Microalgae As a Source of Nutraceuticals. In Phycotoxins: Chemistry and Biochemistry, 2nd ed.; Botana, L.M., Alfonso, A., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; Volume 25, pp. 255–291. [Google Scholar]
- Hillig, F.; Annemüller, S.; Chmielewska, M.; Pilarek, M.; Junne, S.; Neubauer, P. Bioprocess development in single-use systems for heterotrophic marine microalgae. Chem. Ing. Tech. 2013, 85, 153–161. [Google Scholar] [CrossRef]
- Hillig, F.; Porscha, N.; Junne, S.; Neubauer, P. Growth and docosahexaenoic acid production performance of the heterotrophic marine microalgae Crypthecodinium cohnii in the wave-mixed single-use reactor CELL-tainer. Eng. Life Sci. 2014, 14, 254–263. [Google Scholar] [CrossRef]
- Arenas, E.G.; Rodriguez Palacio, M.C.; Juantorena, A.U.; Fernando, S.E.L.; Sebastian, P.J. Microalgae as a potential source for biodiesel production: Techniques, methods, and other challenges. Int. J. Energy Res. 2017, 41, 761–789. [Google Scholar] [CrossRef]
- Duong, V.T.; Thomas-Hall, S.R.; Schenk, P.M. Growth and lipid accumulation of microalgae from fluctuating brackish and sea water locations in South East Queensland—Australia. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef]
- Doan, T.T.Y.; Sivaloganathan, B.; Obbard, J.P. Screening of marine microalgae for biodiesel feedstock. Biomass Bioenergy 2011, 35, 2534–2544. [Google Scholar] [CrossRef]
- National Research Council. Sustainable Development of Algal Biofuels in the United States; The National Academies Press: Washington, DC, USA, 2012. [Google Scholar]
- Acheampong, M.; Ertem, F.C.; Kappler, B.; Neubauer, P. In pursuit of Sustainable Development Goal (SDG) number 7: Will biofuels be reliable? Renew. Sustain. Energy Rev. 2017, 75, 927–937. [Google Scholar] [CrossRef]
- Chisti, Y. Constraints to commercialization of algal fuels. J. Biotechnol. 2013, 167, 201–214. [Google Scholar] [CrossRef]
- Hyka, P.; Lickova, S.; Přibyl, P.; Melzoch, K.; Kovar, K. Flow cytometry for the development of biotechnological processes with microalgae. Biotechnol. Adv. 2013, 31, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Hsu, S.-C.; Han, S.-I.; Thapa, H.R.; Guzman, A.R.; Browne, D.R.; Mehmet Tatli, M.; Devarenne, T.P.; Stern, D.B.; Han, A. High-throughput droplet microfluidics screening platform for selecting fast-growing and high lipid-producing microalgae from a mutant library. Plant Direct 2017, 1, e00011. [Google Scholar] [CrossRef] [PubMed]
- Bippes, M.; Brauer, T.; Brück, T.; Buchholz, R.; Cotta, F.; Friedl, T.; Griehl, C.; Griesbeck, C.; Kistenmacher, H.; Heckenberger, U.; et al. Mikroalgen-Biotechnologie-Gegenwärtiger Stand, Herausforderungen, Ziele; Theodor-Heuss-Allee: Frankfurt, Germany, 2016. [Google Scholar]
- DOE (U.S. Department of Energy). National Algal Biofuels Technology Review; U.S. Department of Energy: Washington, DC, USA, 2016.
- Pierobon, S.C.; Cheng, X.; Graham, P.J.; Nguyen, B.; Karakolis, E.G.; Sinton, D. Emerging microalgae technology: A review. Sustain. Energy Fuels 2017, 1–26. [Google Scholar] [CrossRef]
- Miao, X.; Wu, Q. Biodiesel production from heterotrophic microalgal oil. Bioresour. Technol. 2006, 97, 841–846. [Google Scholar] [CrossRef]
- Lu, Y.; Yang, L.; Wei, W.; Shi, Q. Microchip-based single-cell functional proteomics for biomedical applications. Lab Chip 2017, 17, 1250–1263. [Google Scholar] [CrossRef]
- Hümmer, D.; Kurth, F.; Naredi-Rainer, N.; Dittrich, P.S. Single cells in confined volumes: Microchambers and microdroplets. Lab Chip 2016, 16, 447–458. [Google Scholar] [CrossRef]
- Avesar, J.; Ben Arye, T.B.; Levenberg, S. Frontier microfluidic techniques for short and long-term single cell analysis. Lab Chip 2014, 14, 2161–2167. [Google Scholar] [CrossRef]
- Han, A.; Hou, H.; Li, L.; Kim, H.S.; de Figueiredo, P. Microfabricated devices in microbial bioenergy sciences. Trends Biotechnol. 2013, 31, 225–232. [Google Scholar] [CrossRef]
- Kim, H.S.; Devarenne, T.P.; Han, A. Microfluidic systems for microalgal biotechnology: A review. Algal Res. 2018, 30, 149–161. [Google Scholar] [CrossRef]
- Marba-Ardebol, A.M.; Turon, X.; Neubauer, P.; Junne, S. Application of flow cytometry analysis to elucidate the impact of scale-down conditions in Escherichia coli cultivations P. Gil Salvador 2013 Award in Bioengineering category. (November 22, 2013 in the Annual General Assembly of the AIQS). Afinidad 2016, 73, 7–15. [Google Scholar]
- De La Jara, A.; Mendoza, H.; Martel, A.; Molina, C.; Nordströn, L.; De La Rosa, V.; Díaz, R. Flow cytometric determination of lipid content in a marine dinoflagellate, Crypthecodinium cohnii. Environ. Boil. Fishes 2003, 15, 433–438. [Google Scholar]
- Montero, M.F.; Aristizábal, M.; García Reina, G. Isolation of high-lipid content strains of the marine microalga Tetraselmis suecica for biodiesel production by flow cytometry and single-cell sorting. J. Appl. Phycol. 2011, 23, 1053–1057. [Google Scholar] [CrossRef][Green Version]
- Neofotis, P.; Huang, A.; Sury, K.; Chang, W.; Joseph, F.; Gabr, A.; Twary, S.; Qiu, W.; Holguin, O.; Polle, J.E.W. Characterization and classification of highly productive microalgae strains discovered for biofuel and bioproduct generation. Algal Res. 2016, 15, 164–178. [Google Scholar] [CrossRef]
- Neofotis, P.; Huang, A.; Chang, W.; Joseph, F.; Polle, J.E.W. Microalgae Strain Isolation, Screening, and Identification for biofuels and High-Value Products. In Microalgal Production for Biomass and High-Value Products; CRC Press: Boca Raton, FL, USA, 2016; pp. 63–89. [Google Scholar]
- Pohl, H.A. Dielectrophoresis; 579 Volumes; Cambridge University Press: Cambridge, UK, 1978. [Google Scholar]
- Birkholz, M. Crystal-field induced dipole moments in heteropolar crystals—I: Concept. Zeitschrift für Physik B 1995, 96, 325–332. [Google Scholar] [CrossRef]
- Gagnon, Z.R. Cellular dielectrophoresis: Applications to the characterization, manipulation, separation and patterning of cells. Electrophoresis 2011, 32, 2466–2487. [Google Scholar] [CrossRef]
- Çetin, B.; Li, D. Dielectrophoresis in microfluidics technology. Electrophoresis 2011, 32, 2410–2427. [Google Scholar] [CrossRef]
- Lee, H.-B.; Jeong, M.; Chung, J.-H. Dielectrophoretic sensitivity analysis of cell characterization. Int. J. Precis. Eng. Manuf. 2017, 18, 747–754. [Google Scholar] [CrossRef]
- Pethig, R. Review—Where is Dielectrophoresis (DEP) Going? J. Electrochem. Soc. 2017, 164, B3049–B3055. [Google Scholar] [CrossRef]
- Hughes, M.P. Fifty years of dielectrophoretic cell separation technology. Biomicrofluidics 2016, 10, 1–9. [Google Scholar] [CrossRef]
- Li, M.; Anand, R.K. Cellular dielectrophoresis coupled with single-cell analysis. Anal. Bioanal. Chem. 2018, 410, 2499–2515. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zheng, X.-L.; Hu, N.; Yang, J.; Luo, H.-Y.; Jiang, F.; Liao, Y.-J. Research Progress on Microfluidic Chip of Cell Separation Based on Dielectrophoresis. Chin. J. Anal. Chem. 2015, 43, 300–309. [Google Scholar] [CrossRef]
- Birkholz, M.; Mai, A.; Wenger, C.; Meliani, C.; Scholz, R. Technology modules from micro-and nano-electronics for the life sciences. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 355–377. [Google Scholar] [CrossRef] [PubMed]
- Viefhues, M.; Eichhorn, R. DNA dielectrophoresis: Theory and applications a review. Electrophoresis 2017, 38, 1483–1506. [Google Scholar] [CrossRef]
- Lee, N.; Hwang, B.; Kim, B. The potential of a dielectrophoresis activated cell sorter (DACS) as a next generation cell sorter. Micro Nano Syst. Lett. 2016, 4, 92. [Google Scholar] [CrossRef]
- Paez, C.; Juanola-Feliu, E.; Punter-Villagrasa, J.; Zamora, B.D.M.; Corbera, A.H.; Colomer-Farrarons, J.; Miribel-Català, P.L.; Martí, J.S. Combined Dielectrophoresis and Impedance Systems for Bacteria Analysis in Microfluidic On-Chip Platforms. Sensors 2016, 16, 1514. [Google Scholar] [CrossRef]
- Pethig, R. Dielectrophoresis: Theory, Methodology and Biological Applications; Wiley: Hoboken, NJ, USA, 2017. [Google Scholar]
- Pohl, H.A.; Kaler, K. Continuous dielectrophoretic separation of cell mixtures. Cell Biophys. 1979, 1, 15–28. [Google Scholar] [CrossRef]
- Pohl, H.A.; Kaler, K.; Pollock, K. The continuous positive and negative dielectrophoresis of microorganisms. Bliss 1981, 9, 67–86. [Google Scholar] [CrossRef]
- Müller, T.; Schnelle, T.; Fuhr, G. Dielectric single cell spectra in snow algae. Polar Biol. 1998, 20, 303–310. [Google Scholar] [CrossRef]
- Hübner, Y.; Hoettges, K.F.; Hughes, M.P. Water quality test based on dielectrophoretic measurements of fresh water algae Selenastrum capricornutum. J. Environ. Monit. JEM 2003, 5, 861–864. [Google Scholar] [CrossRef]
- Wang, J.; Sukhorukov, V.L.; Djuzenova, C.S.; Zimmermann, U.; Müller, T.; Fuhr, G. Electrorotational spectra of protoplasts generated from the giant marine algaValonia utricularis. Protoplasma 1997, 196, 123–134. [Google Scholar] [CrossRef]
- Wu, Y.; Huang, C.; Wang, L.; Miao, X.; Xing, W.; Cheng, J. Electrokinetic system to determine differences of electrorotation and traveling-wave electrophoresis between autotrophic and heterotrophic algal cells. Colloids Surf. A Phys. Eng. Asp. 2005, 262, 57–64. [Google Scholar] [CrossRef]
- Ogata, S.; Yasukawa, T.; Matsue, T. Dielectrophoretic manipulation of a single chlorella cell with dual-microdisk electrode. Bioelectrochemistry 2001, 54, 33–37. [Google Scholar] [CrossRef]
- Marbà-Ardébol, A.M.; Emmerich, J.; Neubauer, P.; Junne, S. Single-cell-based monitoring of fatty acid accumulation in Crypthecodinium cohnii with three-dimensional holographic and in situ microscopy. Process. Biochem. 2017, 52, 223–232. [Google Scholar] [CrossRef]
- Siebman, C.; Velev, O.D.; Slaveykova, V.I. Alternating current-dielectrophoresis collection and chaining of phytoplankton on chip: Comparison of individual species and artificial communities. Biosensors 2017, 7, 10004. [Google Scholar] [CrossRef]
- Song, Y.; Yang, J.; Shi, X.; Jiang, H.; Wu, Y.; Peng, R.; Wang, Q.; Gong, N.; Pan, X.; Sun, Y.; et al. DC dielectrophoresis separation of marine algae and particles in a microfluidic chip. Sci. China Ser. B Chem. 2012, 55, 524–530. [Google Scholar] [CrossRef]
- Deng, Y.L.; Chang, J.S.; Juang, Y.J. Separation of microalgae with different lipid contents by dielectrophoresis. Bioresour. Technol. 2012, 135, 137–141. [Google Scholar] [CrossRef]
- Hadady, H.; Redelman, D.; Hiibel, S.R.; Geiger, E.J. Continuous-flow sorting of microalgae cells based on lipid content by high frequency dielectrophoresis. AIMS Biophys. 2016, 3, 398–414. [Google Scholar] [CrossRef]
- Suscillon, C.; Velev, O.D.; Slaveykova, V.I. Alternating current-dielectrophoresis driven on-chip collection and chaining of green microalgae in freshwaters. Biomicrofluidics 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Siebman, C.; Velev, O.D.; Slaveykova, V.I. Two-dimensional algal collection and assembly by combining AC-dielectrophoresis with fluorescence detection for contaminant-induced oxidative stress sensing. Biosensors 2015, 5, 319–336. [Google Scholar] [CrossRef] [PubMed]
- Siebman, C.; Velev, O.; Slaveykova, V. Probing Contaminant-Induced Alterations in Chlorophyll Fluorescence by AC-Dielectrophoresis-Based 2D-Algal Array. Biosensors 2018, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Gringel, F.; Abt, V.; Neubauer, P.; Birkholz, M. Numerical Analysis of Dielectrophoresis for Separation of Microalgae. In Proceedings of the Frühjahrstagung der DPG, Berlin, Germany, 26 February–2 March 2018. [Google Scholar]
- Bono, M.S.; Ahner, B.A.; Kirby, B.J. Detection of algal lipid accumulation due to nitrogen limitation via dielectric spectroscopy of Chlamydomonas reinhardtii suspensions in a coaxial transmission line sample cell. Bioresour. Technol. 2013, 143, 623–631. [Google Scholar] [CrossRef]
- Kim, H.S.; Guzman, A.R.; Thapa, H.R.; Devarenne, T.P.; Han, A. A droplet microfluidics platform for rapid microalgal growth and oil production analysis: Droplet Microfluidics Algal Growth and Oil Analysis. Biotechnol. Bioeng. 2016, 113, 1691–1701. [Google Scholar] [CrossRef]
- Kumar, R.T.K.; Kanchustambham, P.; Kinnamon, D.; Prasad, S. 2D dielectrophoretic signature of Coscinodiscus wailesii algae in non-uniform electric fields. Algal Res. 2017, 27, 109–114. [Google Scholar] [CrossRef]
- Graham, D.M.; Messerli, M.A.; Pethig, R. Spatial manipulation of cells and organelles using single electrode dielectrophoresis. BioTechniques 2012, 52, 39–43. [Google Scholar] [CrossRef]
- Bahi, M.M.; Tsaloglou, M.-N.; Mowlem, M.; Morgan, H. Electroporation and lysis of marine microalga Karenia brevis for RNA extraction and amplification. J. R. Soc. Interface 2011, 8, 601–608. [Google Scholar] [CrossRef]
- Gallo-Villanueva, R.C.; Jesús-Pérez, N.M.; Martínez-López, J.I.; Pacheco, A.; Lapizco-Encinas, B.H. Assessment of microalgae viability employing insulator-based dielectrophoresis. Microfluid. Nanofluidics 2011, 10, 1305–1315. [Google Scholar] [CrossRef]
- Bunthawin, S.; Kongklaew, J.; Tuantranont, A.; Jaruwongrangsri, K.; Maturos, T. Microchip Electrode Development for Traveling wave Dielectrophoresis of Non-Spherical Cell Suspensions. Engineering 2012, 4, 88–93. [Google Scholar] [CrossRef]
- Bunthawin, S.; Ritchie, R.J.; Wanichapichart, P. Dielectrophoresis of Tetraselmis sp., a unicellular green alga, in travelling electric fields analyzed using the RC model for a spheroid. Songklanakarin J. Sci. Technol. 2011, 33, 585–597. [Google Scholar]
- Miled, A.; Sawan, M. Reconfigurable Lab-on-Chip Platform for Algae Cell Manipulation. In Proceedings of the IEEE International Symposium on Circuits and Systems, Melbourne, Australia, 1–5 June 2014; pp. 646–649. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Wu, X.; Jiang, Z.; Wang, W. Dielectrophoretic separation of microalgae cells in ballast water in a microfluidic chip. Electrophoresis 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Castro-Muñoz, R.; Yáñez-Fernández, J.; Fíla, V. Phenolic compounds recovered from agro-food by-products using membrane technologies: An overview. Food Chem. 2016, 213, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Montes, E.; Castro-Muñoz, R. Metabolites recovery from fermentation broths via pressure-driven membrane processes. Asia Pac. J. Chem. Eng. 2019, 14, e2332. [Google Scholar] [CrossRef]
- Castro-Muñoz, R.; Boczkaj, G.; Gontarek, E.; Cassano, A.; Fíla, V. Membrane technologies assisting plant-based and agro-food by-products processing: A comprehensive review. Trends Food Sci. Technol. 2020, 95, 219–232. [Google Scholar] [CrossRef]
- Li, M.; Li, W.H.; Zhang, J.; Alici, G.; Wen, W. A review of microfabrication techniques and dielectrophoretic microdevices for particle manipulation and separation. J. Phys. D Appl. Phys. 2014, 47. [Google Scholar] [CrossRef]
- Pohl, H.A.; Hawk, I. Separation of Living and Dead Cells by Dielectrophoresis. Science 1966, 152, 647–649. [Google Scholar] [CrossRef]
- Wang, C.; Lee, K.A.; Choi, E.; Lee, K.-Y.; Lee, S.-Y.; Park, J.; Jung, K.-H. Enhancement of radionuclide bio-decontamination by screening highly efficient microalgae for Sr biomineralization. Lab Chip 2018, 18, 2270–2278. [Google Scholar] [CrossRef]
- Deng, Y.L.; Kuo, M.Y.; Juang, Y.J. Development of flow through dielectrophoresis microfluidic chips for biofuel production: Sorting and detection of microalgae with different lipid contents. Biomicrofluidics 2014, 8, 1–10. [Google Scholar] [CrossRef]
- Michael, K.A.; Hiibel, S.R.; Geiger, E.J. Dependence of the dielectrophoretic upper crossover frequency on the lipid content of microalgal cells. Algal Res. 2014, 6, 17–21. [Google Scholar] [CrossRef]
- Hackett, J.; Anderson, D.; Erdner, D.; Bhattacharya, D. Dinoflagellates: A remarkable evolutionary experiment. Am. J. Bot. 2004, 91, 1523–1534. [Google Scholar] [CrossRef] [PubMed]

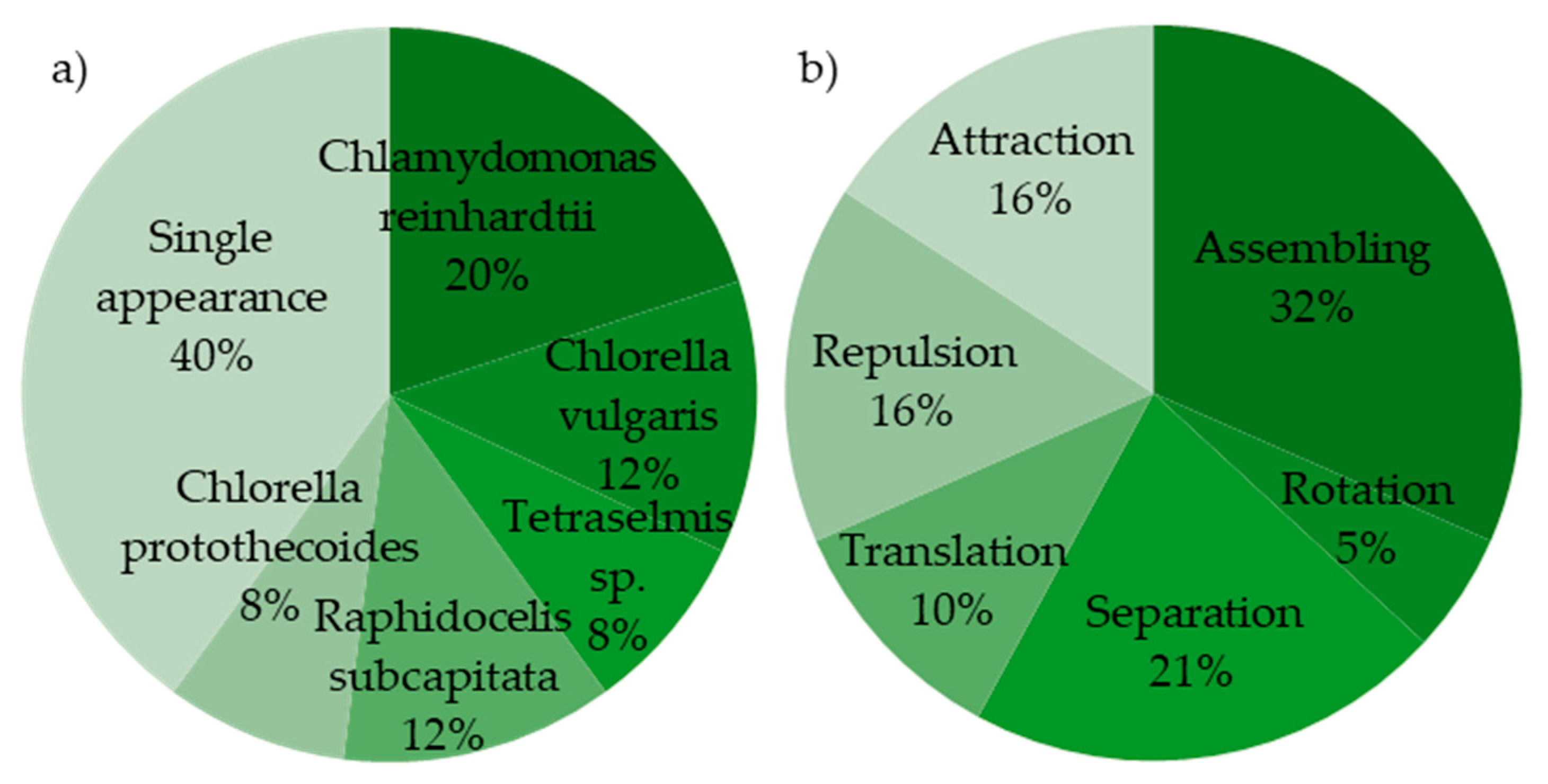
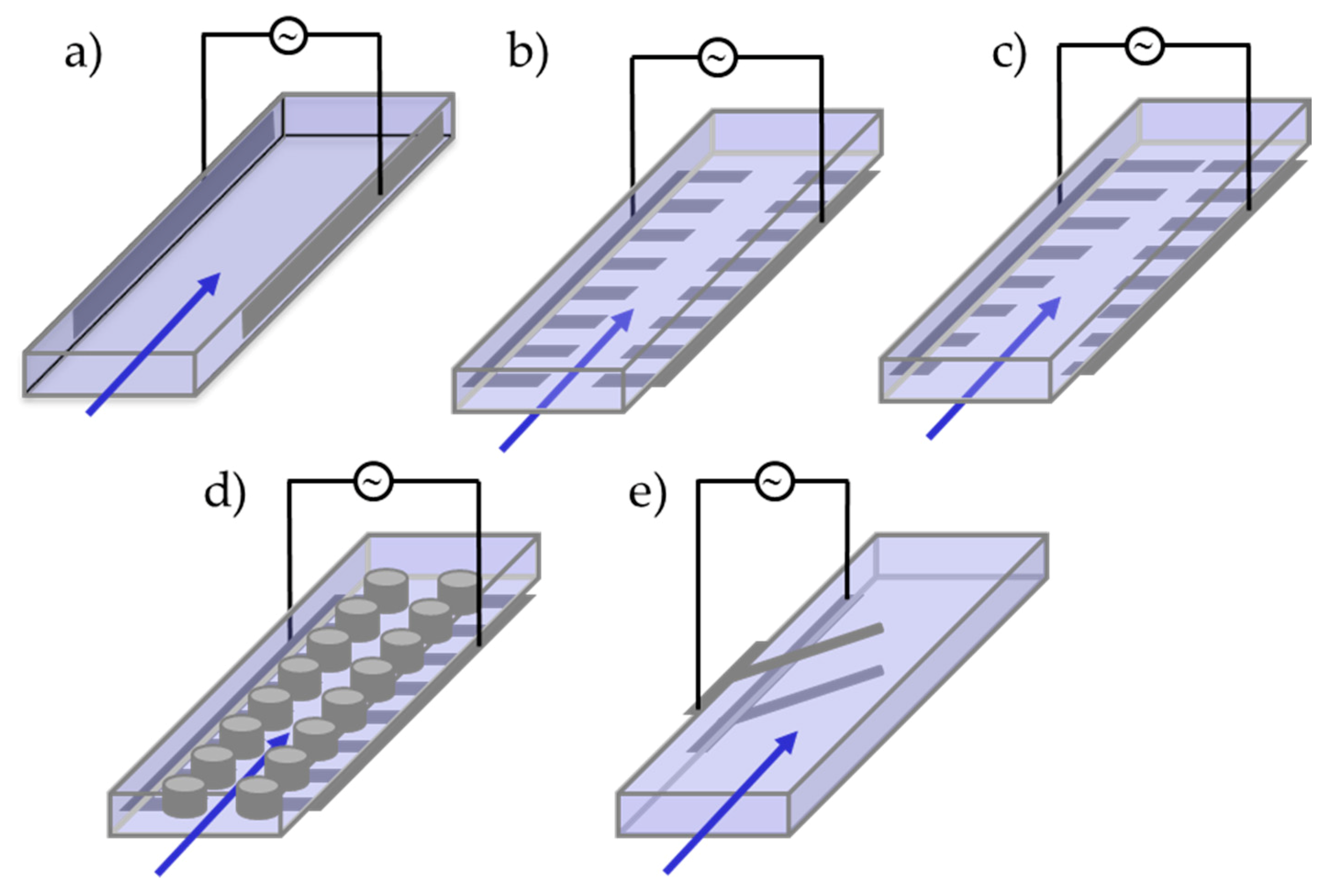
| Algal Species | Taxonomy and No. Flagella | Application Type and Description | Device Structure | Electric Field, Flow Rate, and Cell Concentration | Dielectric Properties of the Medium | Dielectric Properties of Microalgae | Ref. | |
|---|---|---|---|---|---|---|---|---|
| (a) Chlamydomonas reinhardtii (b) Synechocystis sp. (c) Cyclotella meneghiniana | (a) green alga, 2 (b) cyanobac., 0 (c) diatom, 0 | ass. rot. | measuring effects of AC field intensity, frequency and duration on chaining efficiency and chain lengths | chamber , coplanar electrodes | Geneva lake water | [56] | ||
| (a) Chlorella vulgaris (b) Raphidocelis subcapitata (c) Dunaliella salina | (a) green alga, 0 (b) green alga, 0 (c) green alga, 2 | sep. | separation by size and species | PDMS channels with overall field gradient | sodium borate buffer solution | [57] | ||
| Chlamydomonas reinhardtii (a) high lipid (b) low lipid | green alga, 2 | sep. | high-frequency DEP in continuous-flow cell screening device for separation based on lipid content | 4 interdigitated electrode arrays 10 electrodes each by etching | [58] | |||
| Chlamydomonas reinhardtii | green alga, 2 | ass. | high-frequency DEP to determine upper crossover frequency of cells with varying lipid content | glass slide with needle patterned electrodes (Au) | [59] | |||
| Chlamydomonas reinhardtii | green alga, 2 | ass. | characterization of effects of freshwater composition on the DEP response | , coplanar electrodes by vapor deposition | [60] | |||
| Chlamydomonas reinhardtii | green alga, 2 | ass. | rapid tool for capture and screen with fluorescence for the effect of contaminants | water; 0.0001 M MOPS +various contaminants | [61,62] | |||
| Chlorella vulgaris | green alga, 0 | sep. | studies on solution conductivity and lipid content, microfluidic chip to sort the microalgae with different lipid contents | by spin coating, symmetrical deflector electrodes by vapor deposition and photolithography | [63,64] | |||
| Chlorella vulgaris | green alga, 0 | att. rep. | screening for highest radionuclide bio-decontamination by n- and p-DEP | on glass by lithography | [65] | |||
| Coscinodiscus wailesii | diatom, 0 | att. rep. | 2D dielectrophoretic signature | PDMS microfluidic well, interdigitated electrode pattern | [66] | |||
| Eremosphaera viridis | green alga, 0 | att. rep. | tool for spatial manipulation | commercially available single electrode, etched elgiloy tip with porous metal-oxide coating | [67] | |||
| Karenia brevis | dinoflagellate, 2 | ass. | dielectrophoretic concentration | [68] | ||||
| Raphidocelis subcapitata | green alga, 0 | sep. | concentrate and separate live and dead cells | by wet etching, overall field gradient | [69] | |||
| Tetraselmis sp. | green alga, 4 | trans. | twDEP used to estimate the dielectric properties | glass slide, octa-pairs interdigitated electrode by photolithography and wet-etching | [70] | |||
| Tetraselmis sp. (a) control (b) As treated (c) boiled | green alga, 4 | trans. | determination of dielectric properties and effects of arsenic | glass slide, octa-pair interdigitated electrodes ) | [71] | |||
| heterogeneous population | various | ass. | technique to monitor the concentration of algae in fresh water to avoid mass contaminations | chamber, four electrodes | [72] | |||
| (a) Platymonas sp. (b) Closterium sp. | (a) green alga, 2 (b) green alga, 0 | sep. | continuous separation of different microalgae from microplastics by multi-electrode n- and p-DEP | [73] | ||||
| Term | Meaning | Unit | Term | Meaning | Unit/Value | Term | Meaning | Unit |
|---|---|---|---|---|---|---|---|---|
| Cell diameter | Suspending medium conductivity | Cell wall thickness | ||||||
| Electrode width | Cell membrane conductivity | Cell membrane thickness | ||||||
| Electrode height | Cell interior conductivity | Cell concentration | ||||||
| Electrode length | Cell lipid body conductivity | Cell number | - | |||||
| Electrode diameter | Cell wall conductivity | Flow rate | ||||||
| Gap to next electrode | Suspending medium relative permittivity | - | frequency | |||||
| Channel/chamber width | Cell interior relative permittivity | - | Field strength | |||||
| Channel/chamber height | Cell lipid body relative permittivity | - | Voltage | |||||
| Channel/chamber length | Cell wall relative permittivity | |||||||
| Channel/chamber length | Permittivity of free space | |||||||
| Channel/chamber diameter |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abt, V.; Gringel, F.; Han, A.; Neubauer, P.; Birkholz, M. Separation, Characterization, and Handling of Microalgae by Dielectrophoresis. Microorganisms 2020, 8, 540. https://doi.org/10.3390/microorganisms8040540
Abt V, Gringel F, Han A, Neubauer P, Birkholz M. Separation, Characterization, and Handling of Microalgae by Dielectrophoresis. Microorganisms. 2020; 8(4):540. https://doi.org/10.3390/microorganisms8040540
Chicago/Turabian StyleAbt, Vinzenz, Fabian Gringel, Arum Han, Peter Neubauer, and Mario Birkholz. 2020. "Separation, Characterization, and Handling of Microalgae by Dielectrophoresis" Microorganisms 8, no. 4: 540. https://doi.org/10.3390/microorganisms8040540
APA StyleAbt, V., Gringel, F., Han, A., Neubauer, P., & Birkholz, M. (2020). Separation, Characterization, and Handling of Microalgae by Dielectrophoresis. Microorganisms, 8(4), 540. https://doi.org/10.3390/microorganisms8040540