Fate and Biofilm Formation of Wild-Type and Pressure-Stressed Pathogens of Public Health Concern in Surface Water and on Abiotic Surfaces
Abstract
1. Introduction
2. Materials and Methods
2.1. Planktonic Cells Preparations and Enumeration
2.2. Biofilm Fate and Formation
2.3. Neutralization, and Microbiological and pH Analyses
2.4. Experimental Design and Statistical Analyses
3. Results and Discussion
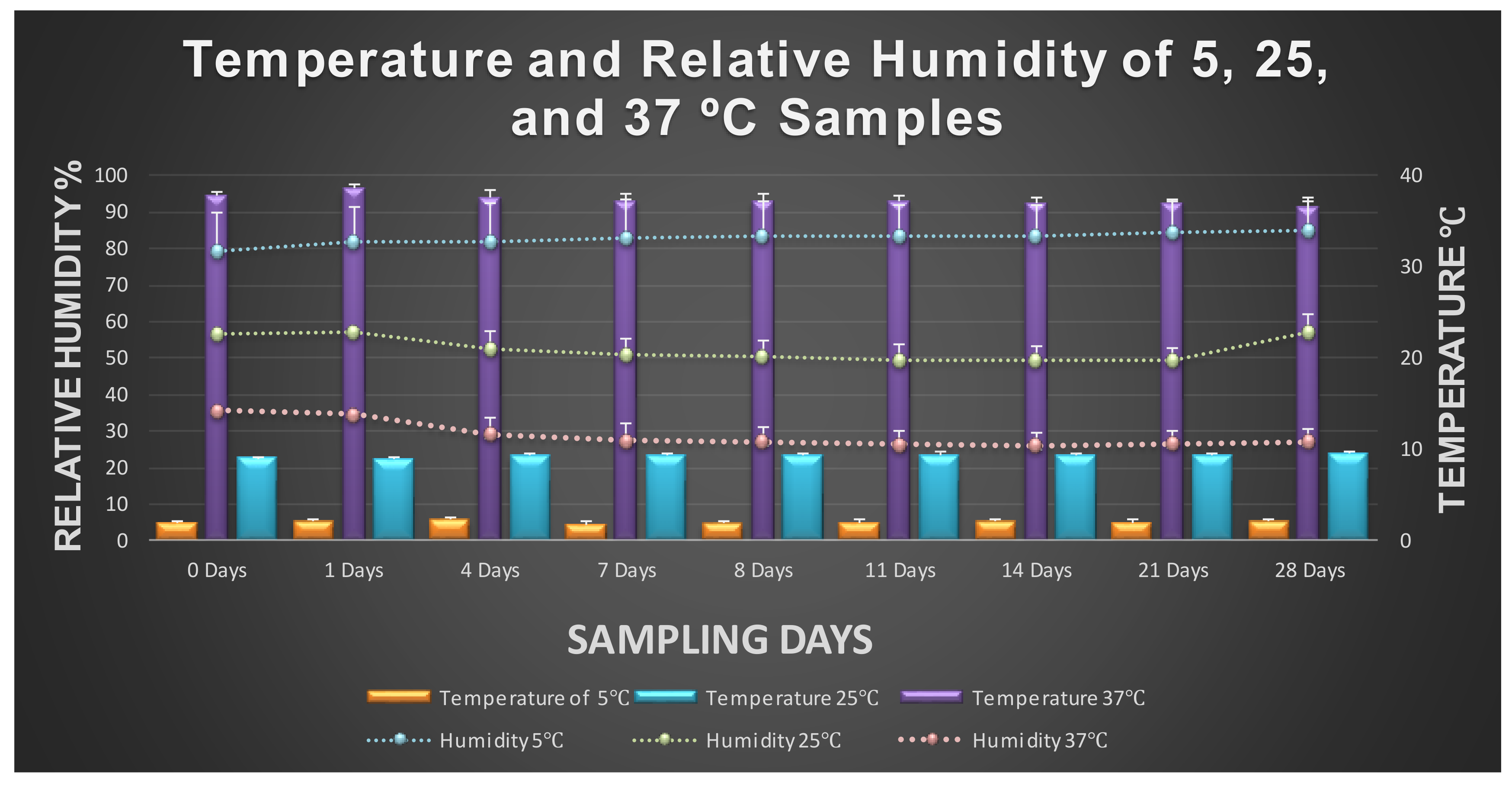
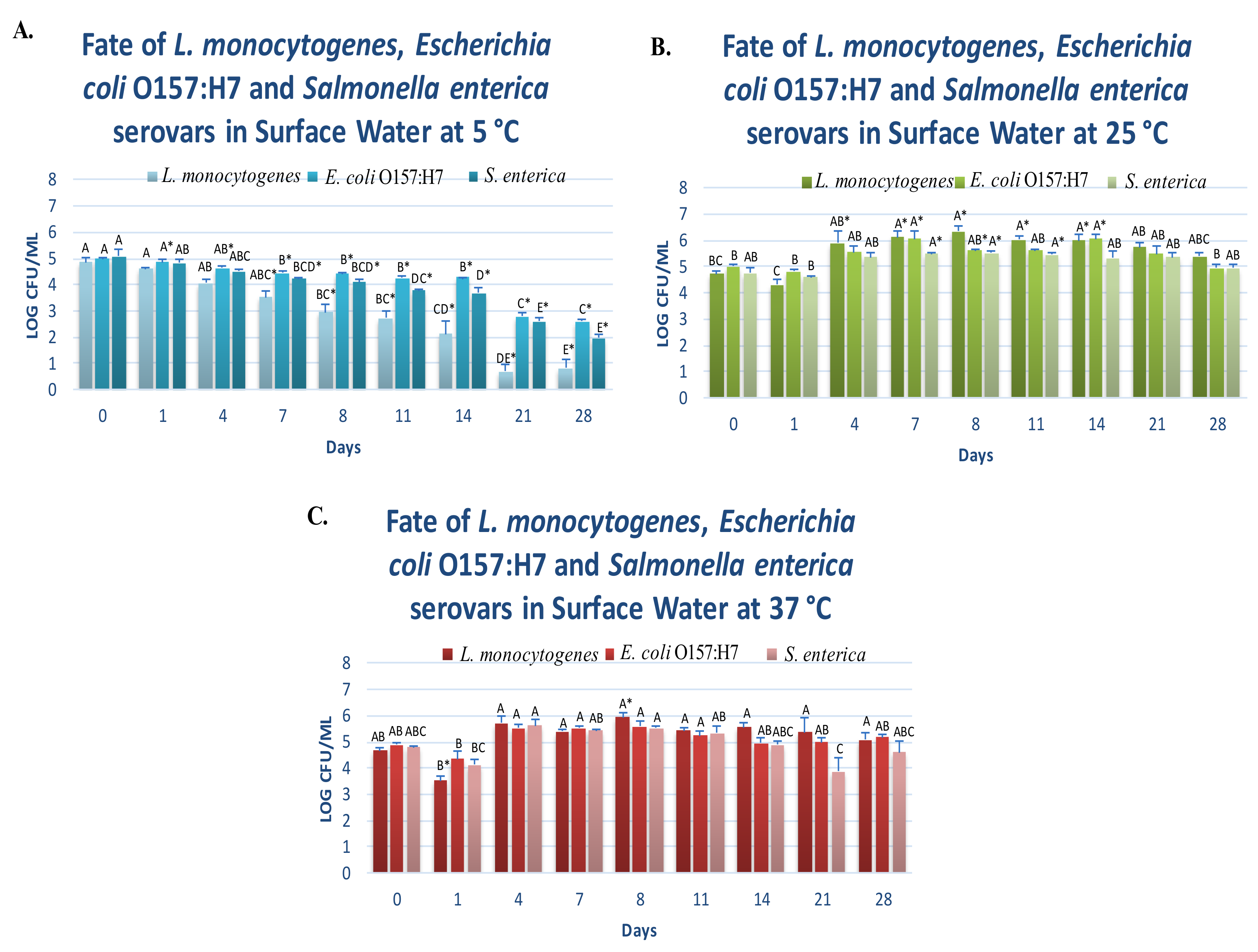
3.1. Fate of Planktonic Pathogens at 5 °C
3.2. Fate of Planktonic Pathogens at 25 and 37 °C
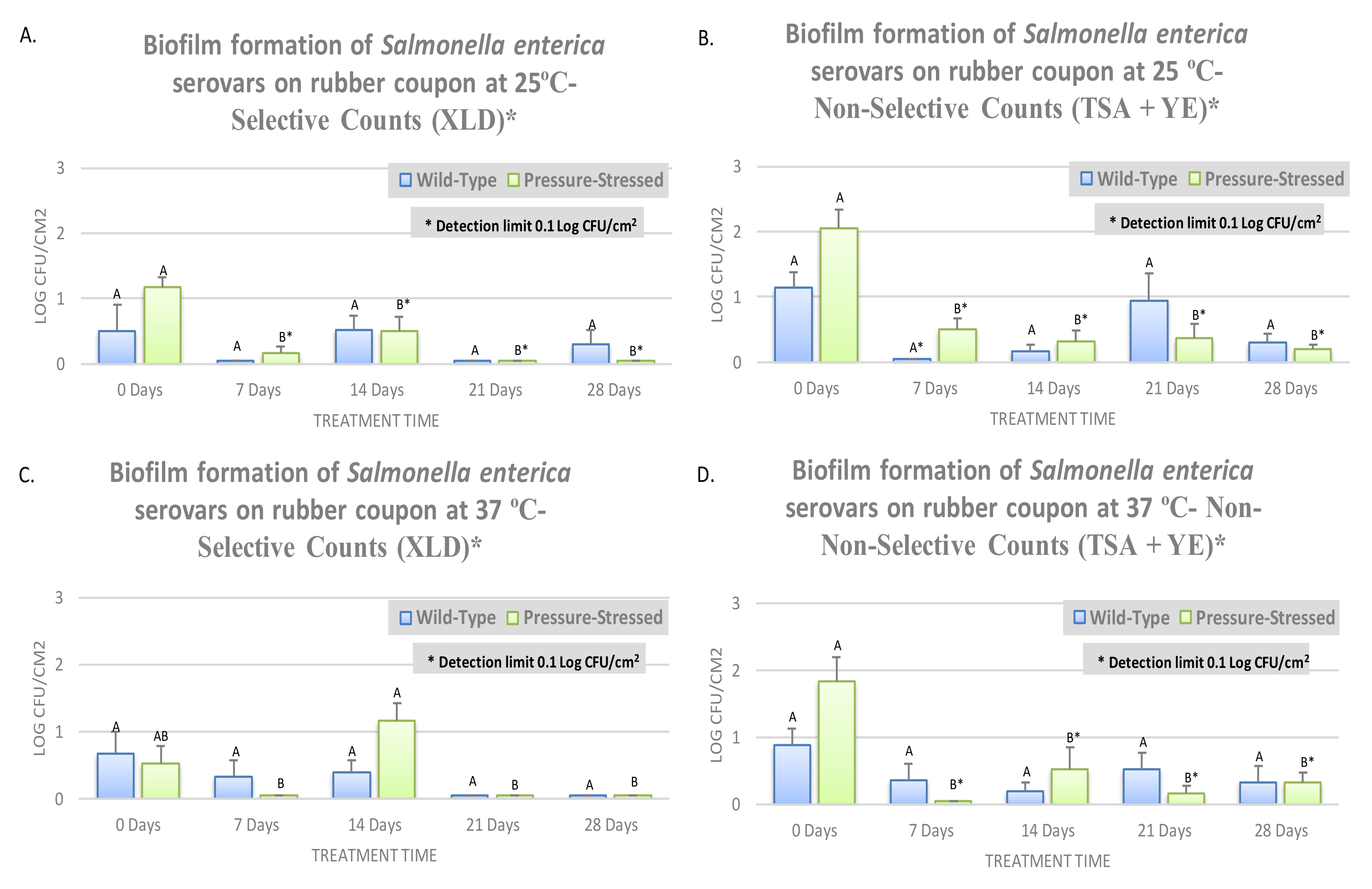
3.3. Biofilm Formation of Non-Typhoidal Salmonella at 25 and 37 °C
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnson, S. The ghost map: The story of London’s most terrifying epidemic--and how it changed science, cities, and the modern world; Penguin Books Ltd.: London, UK, 2006. [Google Scholar]
- Fouladkhah, A. The Need for evidence-based outreach in the current food safety regulatory landscape. J. Ext. 2017, 55, 2COM1. [Google Scholar]
- Fouladkhah, A.C.; Thompson, B.; Camp, J.S. Safety of Food and Water Supplies in the Landscape of Changing Climate. Microorganisms 2019, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Steele, M.; Odumeru, J. Irrigation water as source of foodborne pathogens on fruit and vegetables. J. Food Prot. 2004, 67, 2839–2849. [Google Scholar] [CrossRef] [PubMed]
- Suslow, T.V.; Oria, M.P.; Beuchat, L.R.; Garrett, E.H.; Parish, M.E.; Harris, L.J.; Farber, J.N.; Busta, F.F. Production practices as risk factors in microbial food safety of fresh and fresh-cut produce. Compr. Rev. Food Sci. Food Saf. 2003, 2, 38–77. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC), National Outbreak Reporting System (NORS). 2019. Available online: https://wwwn.cdc.gov/norsdashboard/ (accessed on 4 December 2019).
- Lyautey, E.; Lapen, D.R.; Wilkes, G.; McCleary, K.; Pagotto, F.; Tyler, K.; Hartmann, A.; Piveteau, P.; Rieu, A.; Robertson, W.J.; et al. Distribution and characteristics of Listeria monocytogenes isolates from surface waters of the South Nation River watershed, Ontario, Canada. Appl. Environ. Microbiol. 2007, 73, 5401–5410. [Google Scholar] [CrossRef] [PubMed]
- Thurston-Enriquez, J.A.; Watt, P.; Dowd, S.E.; Enriquez, R.; Pepper, I.L.; Gerba, C.P. Detection of protozoan parasites and microsporidia in irrigation waters used for crop production. J. Food Prot. 2002, 65, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Haley, B.J.; Cole, D.J.; Lipp, E.K. Distribution, diversity, and seasonality of waterborne Salmonellae in a rural watershed. Appl. Environ. Microbiol. 2009, 75, 1248–1255. [Google Scholar] [CrossRef]
- Vereen, E., Jr.; Lowrance, R.R.; Jenkins, M.B.; Adams, P.; Rajeev, S.; Lipp, E.K. Landscape and seasonal factors influence Salmonella and Campylobacter prevalence in a rural mixed use watershed. Water Res. 2013, 47, 6075–6085. [Google Scholar] [CrossRef]
- Edge, T.A.; El-Shaarawi, A.; Gannon, V.; Jokinen, C.; Kent, R.; Khan, I.U.; Koning, W.; Lapen, D.; Miller, J.; Neumann, N.; et al. Investigation of an Escherichia coli environmental benchmark for waterborne pathogens in agricultural watersheds in Canada. J. Environ. Qual. 2012, 41, 21–30. [Google Scholar] [CrossRef]
- Capozzi, V.; Fiocco, D.; Amodio, M.L.; Gallone, A.; Spano, G. Bacterial stressors in minimally processed food. Int. J. Mol. Sci. 2009, 10, 3076–3105. [Google Scholar] [CrossRef] [PubMed]
- Aras, S.; Kabir, M.N.; Chowdhury, S.; Fouladkhah, A.C. Augmenting the Pressure-Based Pasteurization of Listeria monocytogenes by Synergism with Nisin and Mild Heat. Int. J. Environ. Res. Public Health 2020, 17, 563. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.N.; Aras, S.; Allison, A.; Adhikari, J.; Chowdhury, S.; Fouladkhah, A. Interactions of Carvacrol, Caprylic Acid, Habituation, and Mild Heat for Pressure-Based Inactivation of O157 and Non-O157 Serogroups of Shiga Toxin-Producing Escherichia coli in Acidic Environment. Microorganisms 2019, 7, 145. [Google Scholar] [CrossRef]
- Allison, A.; Chowdhury, S.; Fouladkhah, A. Synergism of mild heat and high-pressure pasteurization against Listeria monocytogenes and natural microflora in phosphate-buffered saline and raw milk. Microorganisms 2018, 6, 102. [Google Scholar] [CrossRef] [PubMed]
- Allison, A.; Fouladkhah, A. Sensitivity of Salmonella serovars and natural microflora to high-pressure pasteurization: Open access data for risk assessment and practitioners. Data Brief 2018, 21, 480–484. [Google Scholar] [CrossRef]
- Fouladkhah, A.; Geornaras, I.; Sofos, J.N. Biofilm formation of O157 and Non-O157 shiga toxin-producing Escherichia coli and multidrug-resistant and susceptible Salmonella Typhimurium and Newport and their inactivation by sanitizers. J. Food Sci. 2013, 78, M880–M886. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, C.S.; Dourou, D.; Geornaras, I.; Yoon, Y.; Scanga, J.A.; Belk, K.E.; Smith, G.C.; Nychas, G.E.; Sofos, J.N. Transfer, attachment, and formation of biofilms by Escherichia coli O157 on meat-contact surface material. J. Food Sci. 2012, 77, 343–347. [Google Scholar] [CrossRef]
- Stopforth, J.D.; Samelis, J.; Sofos, J.N.; Kendall, P.A.; Smith, G.C. Biofilm formation by acid-adapted and nonadapted Listeria monocytogenes in fresh beef decontamination washings and its subsequent inactivation with sanitizers. J. Food Prot. 2002, 65, 1717–1727. [Google Scholar] [CrossRef]
- Giaouris, E.; Chorianopoulous, N.; Nychas, G.E. Effect of temperature, pH, and water activity on biofilm formation of Salmonella enteric Enteritidis PT4 on stainless steel surfaces as indicated by the bead vortexing method and conductance measurement. J. Food Prot. 2005, 68, 2149–2154. [Google Scholar] [CrossRef]
- Henry-Smith, M. Control of Biofilm and Planktonic Cells of Cronobacter sakazakii, Listeria monocytogenes, Shiga Toxin-Producing Escherichia coli, and Salmonella Serovars Using Pressure-Based and Quaternary Ammonium Compound-Based Sanitizers. Master’s Thesis, Tennessee State University, Nashville, TN, USA, 2019. [Google Scholar]
- FDA. BBB. 2017 Second Edition. Available online: https://www.fda.gov/food/foodborne-pathogens/bad-bug-book-second-edition (accessed on 12 February 2020).
- CDC. Multistate Outbreak of Listeriosis Linked to Whole Cantaloupes from Jensen Farms, Colorado (FINAL UPDATE). 2012. Available online: https://www.cdc.gov/listeria/outbreaks/cantaloupes-jensen-farms/index.html (accessed on 12 March 2020).
- WHO. Listeriosis—South Africa. 2018. Available online: https://www.who.int/csr/don/28-march-2018-listeriosis-south-africa/en/ (accessed on 12 March 2020).
- Hoffmann, S.; Maculloch, B.; Batz, M. Economic Burden of Major Foodborne Illnesses Acquired in the United States. U.S. Department of Agriculture, Economic Research Service: Washington, DC, USA, 2015; EIB-140. [Google Scholar]
- Scallan, E.; Hoekstra, R.M.; Mahon, B.E.; Jones, T.F.; Griffin, P.M. An assessment of the human health impact of seven leading foodborne pathogens in the United States using disability adjusted life years. Epidemiol. Infect. 2015, 143, 2795–2804. [Google Scholar] [CrossRef]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—Major pathogens. Emerg. Infect. Dis. 2011, 17, 7. [Google Scholar] [CrossRef]
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 2007, 113, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Gholipour, S.; Nikaeen, M.; Farhadkhani, M.; Nikmanesh, B. Survey of Listeria monocytogenes contamination of various environmental samples and associated health risks. Food Control 2020, 108, 106843. [Google Scholar] [CrossRef]
- Tuttle, J.; Gomez, T.; Doyle, M.P.; Wells, J.G.; Zhao, T.; Tauxe, R.V.; Griffin, P.M. Lessons from a large outbreak of Escherichia coli O157: H7 infections: Insights into the infectious dose and method of widespread contamination of hamburger patties. Epidemiol. Infect. 1999, 122, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Ryser, E.T.; Marth, E.H. Listeria, Listeriosis, and Food Safety; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Kroupitski, Y.; Pinto, R.; Brandl, M.T.; Belausov, E.; Sela, S. Interactions of Salmonella enterica with lettuce leaves. J. Appl. Microbiol. 2009, 106, 1876–1885. [Google Scholar] [CrossRef] [PubMed]
- Ge, C.; Lee, C.; Lee, J. The impact of extreme weather events on Salmonella internalization in lettuce and green onion. Food Res. Int. 2012, 45, 1118–1122. [Google Scholar] [CrossRef]
- Deering, A.J.; Mauer, L.J.; Pruitt, R.E. Internalization of E. coli O157: H7 and Salmonella spp. in plants: A review. Food Res. Int. 2012, 45, 567–575. [Google Scholar] [CrossRef]
- Gómez-López, V.M.; Marín, A.; Allende, A.; Beuchat, L.R.; Gil, M.I. Postharvest handling conditions affect internalization of Salmonella in baby spinach during washing. J. Food Prot. 2013, 76, 1145–1151. [Google Scholar] [CrossRef]
- Lapidot, A.; Yaron, S. Transfer of Salmonella enterica serovar Typhimurium from contaminated irrigation water to parsley is dependent on curli and cellulose, the biofilm matrix components. J. Food Prot. 2009, 72, 618–623. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kabir, M.N.; Aras, S.; Wadood, S.; Chowdhury, S.; Fouladkhah, A.C. Fate and Biofilm Formation of Wild-Type and Pressure-Stressed Pathogens of Public Health Concern in Surface Water and on Abiotic Surfaces. Microorganisms 2020, 8, 408. https://doi.org/10.3390/microorganisms8030408
Kabir MN, Aras S, Wadood S, Chowdhury S, Fouladkhah AC. Fate and Biofilm Formation of Wild-Type and Pressure-Stressed Pathogens of Public Health Concern in Surface Water and on Abiotic Surfaces. Microorganisms. 2020; 8(3):408. https://doi.org/10.3390/microorganisms8030408
Chicago/Turabian StyleKabir, Md Niamul, Sadiye Aras, Sabrina Wadood, Shahid Chowdhury, and Aliyar Cyrus Fouladkhah. 2020. "Fate and Biofilm Formation of Wild-Type and Pressure-Stressed Pathogens of Public Health Concern in Surface Water and on Abiotic Surfaces" Microorganisms 8, no. 3: 408. https://doi.org/10.3390/microorganisms8030408
APA StyleKabir, M. N., Aras, S., Wadood, S., Chowdhury, S., & Fouladkhah, A. C. (2020). Fate and Biofilm Formation of Wild-Type and Pressure-Stressed Pathogens of Public Health Concern in Surface Water and on Abiotic Surfaces. Microorganisms, 8(3), 408. https://doi.org/10.3390/microorganisms8030408





