A Frameshift Mutation in wcaJ Associated with Phage Resistance in Klebsiella pneumoniae
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Bacteriophages, Plasmids, Oligos, and Growth Conditions
2.2. Phage Lysate Preparation
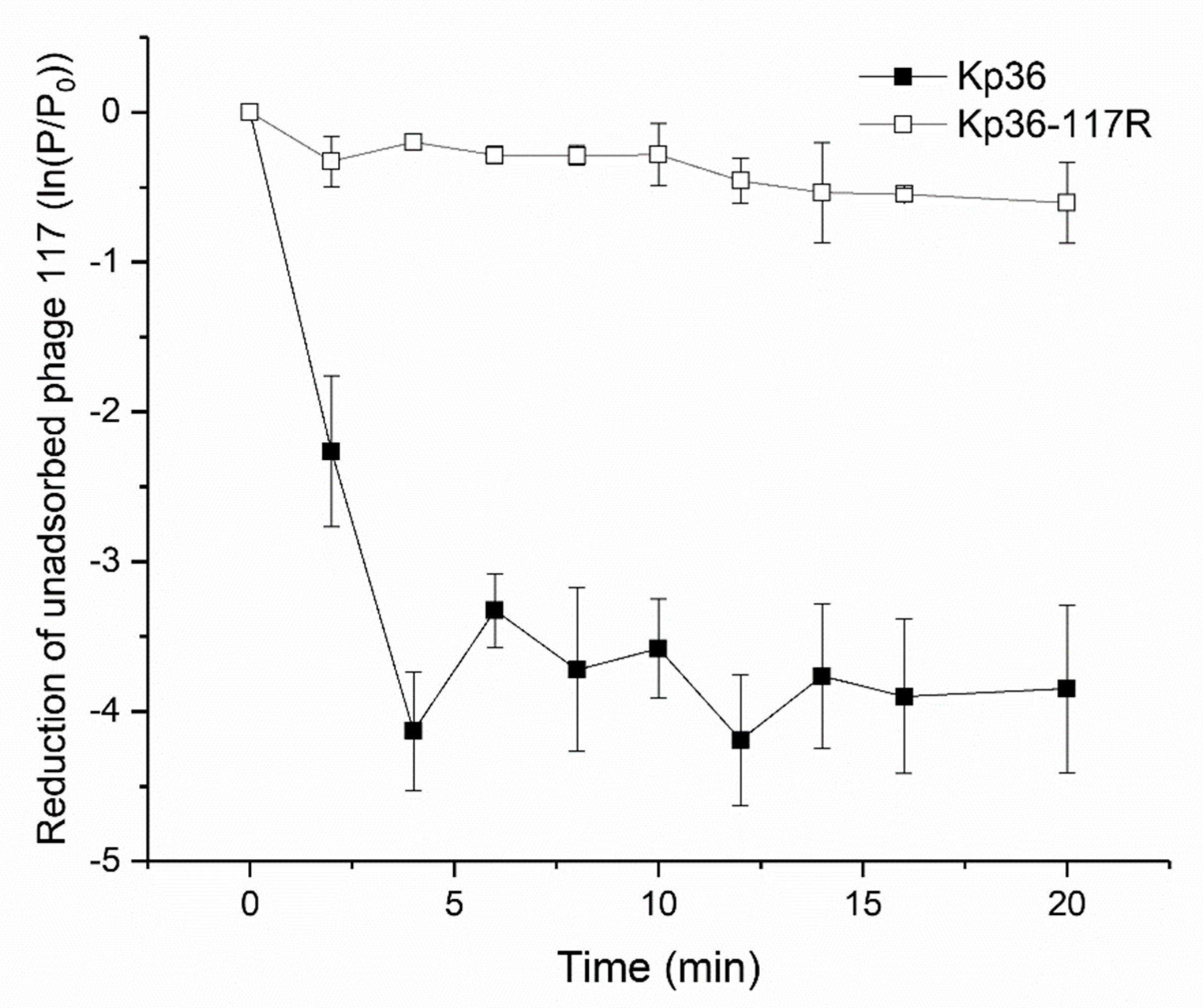
2.3. Phage 117 Adsorption Rate Measurements
2.4. Bioinformatic Analysis of Phage-Resistant Mutant
2.5. Construction and Characterization of Bacterial Mutants
2.6. Complementation Assay
2.7. Spot Tests and Efficiency of Plaquing (EOP) Assays
2.8. Growth Curves of K. Pneumoniae Strains and Competition Assay
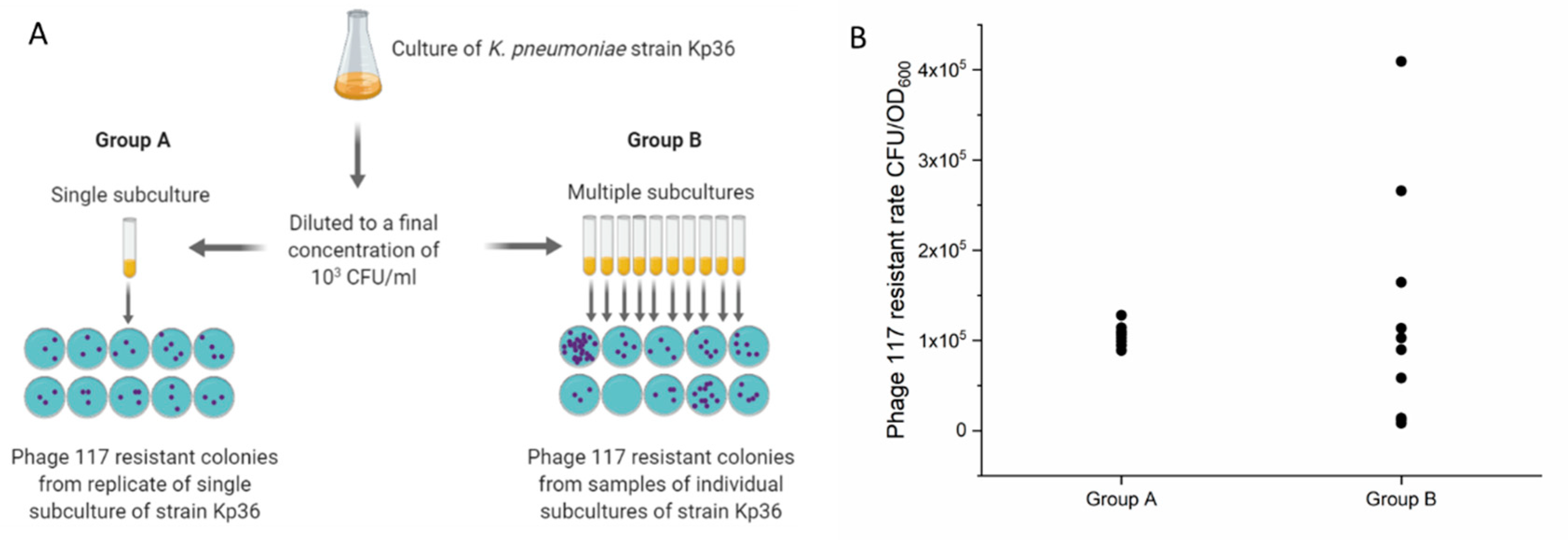
2.9. Luria–Delbrück Fluctuation Tests
3. Results
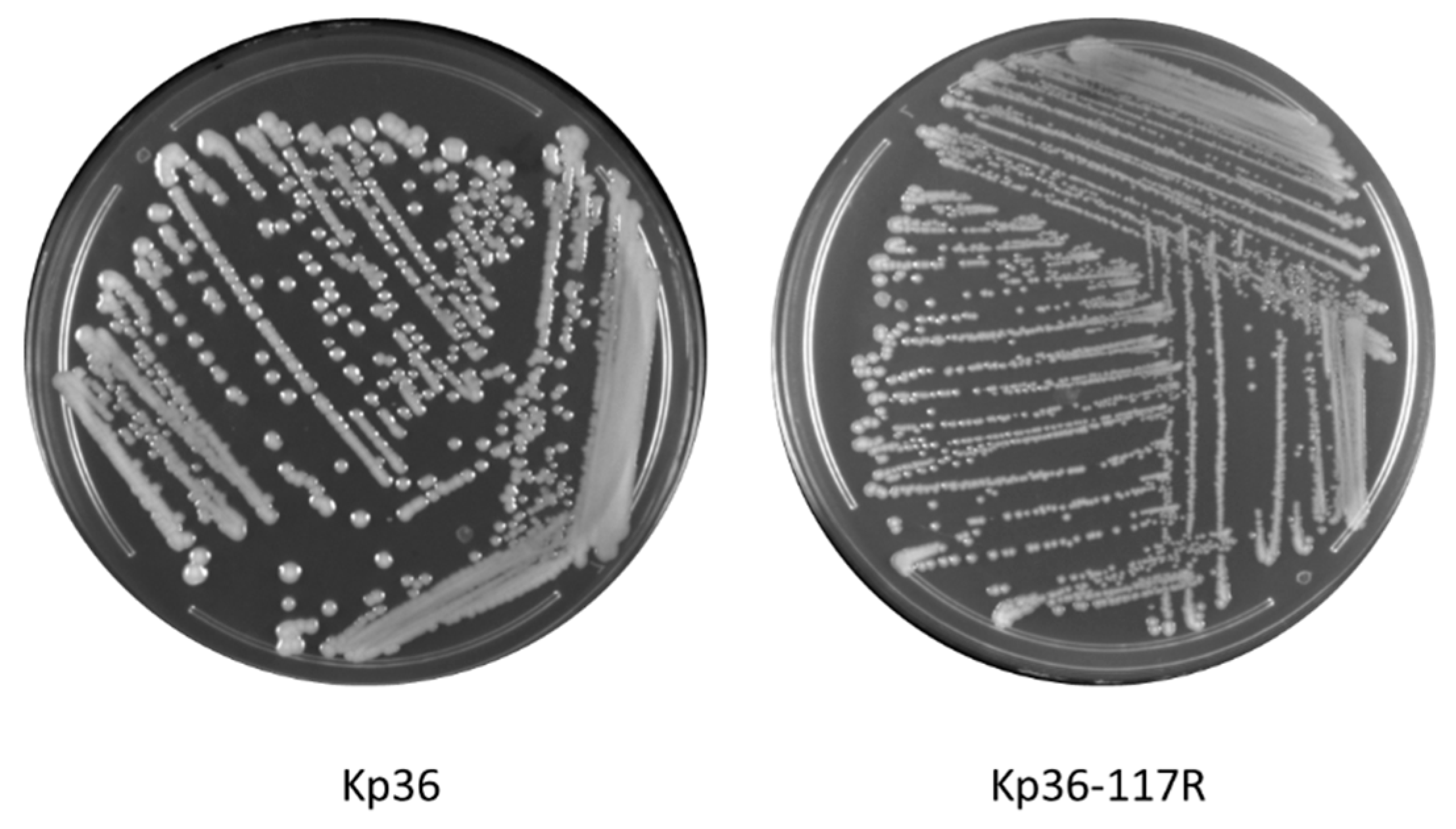
3.1. Morphological Differences between Wild-Type and Phage Resistant Mutants
3.2. Phage Adsorption Assay Indicates Development of Resistance to Phage 117 during Adsorption
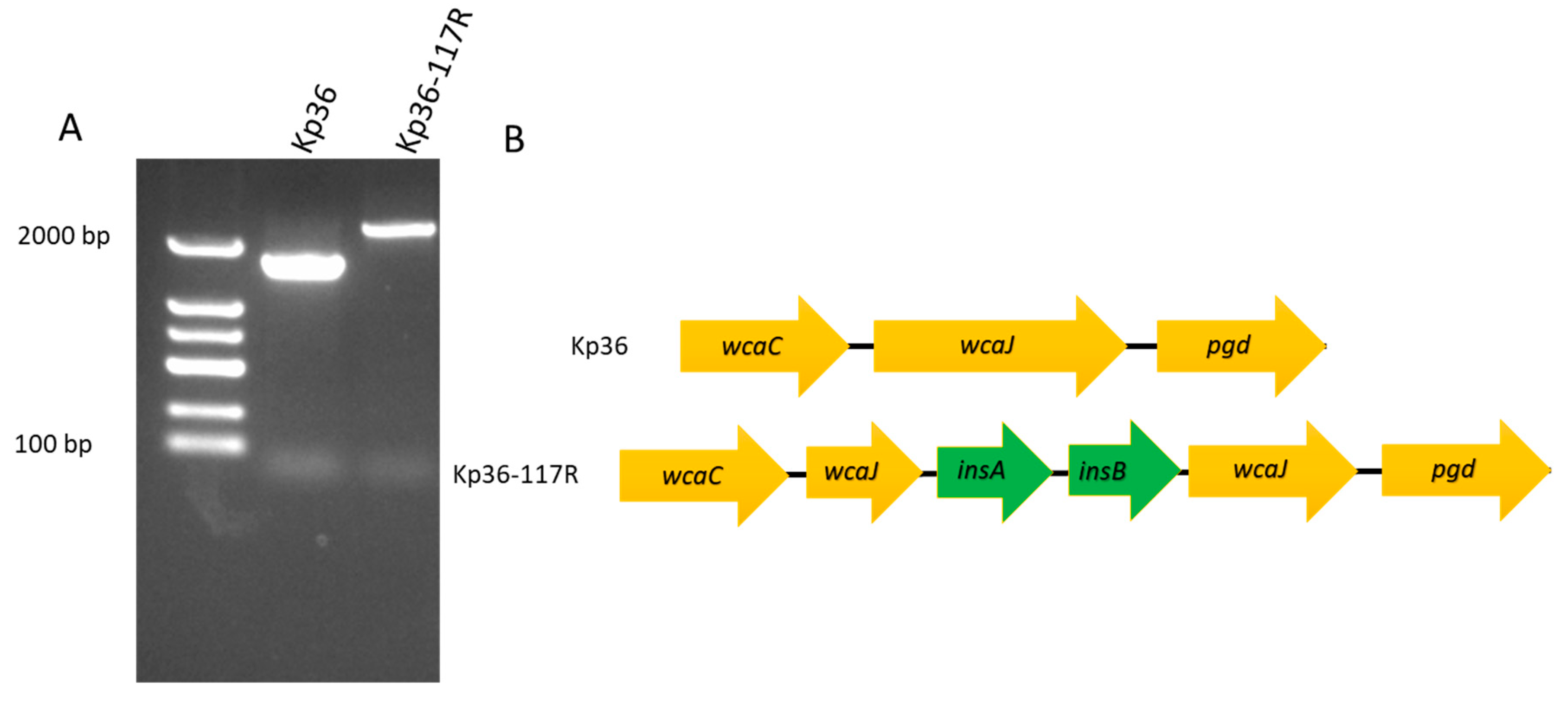
3.3. Characterization of the Phage Resistance Mechanism
3.4. Complementation of Strain Kp36-117R with Wild-Type wcaJ Restores Ф117 Sensitivity
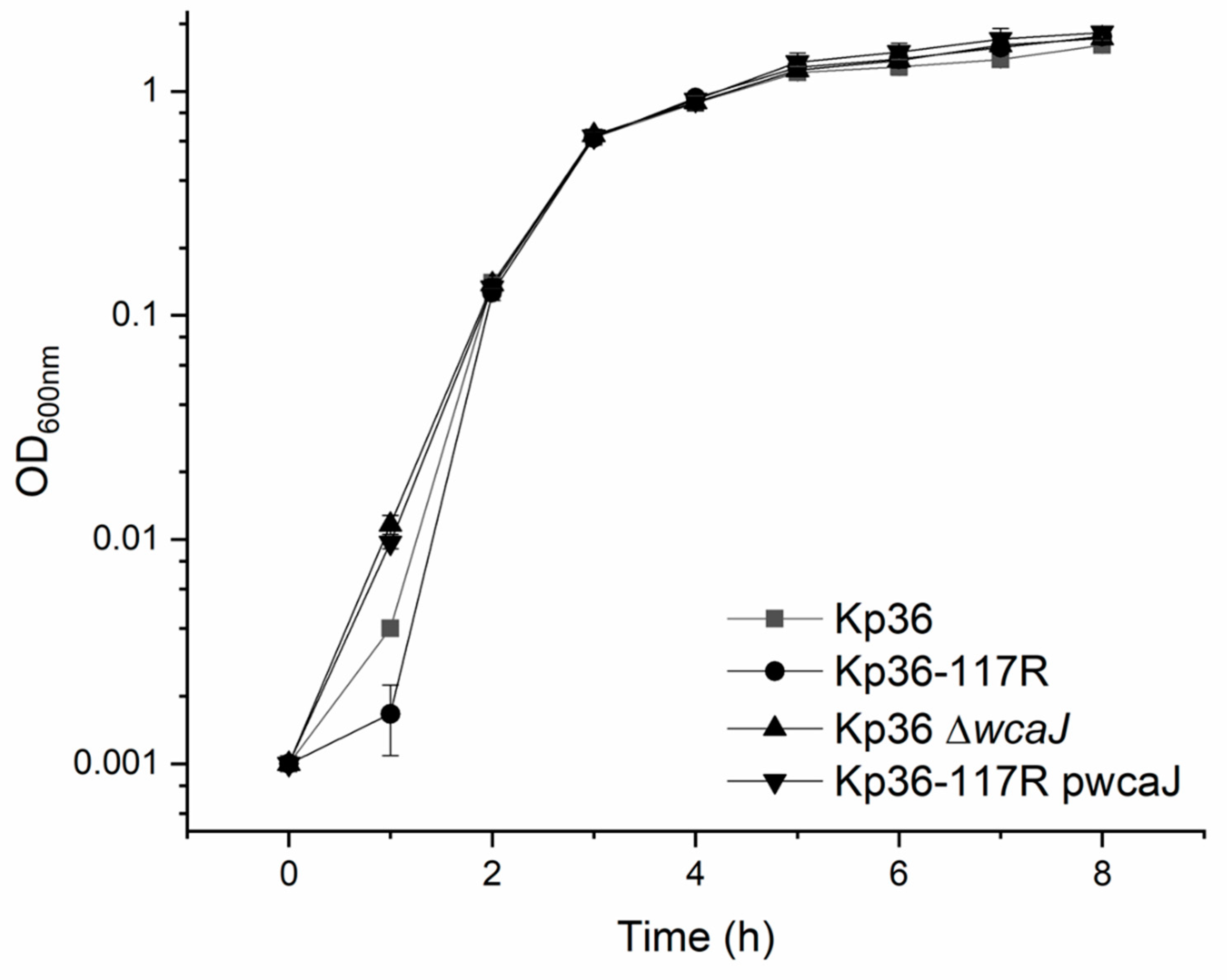
3.5. The wcaJ Mutation Does Not Affect Growth Rate and Competition Assays
3.6. Non-Mucoid Phage Resistant Mutants are Unlikely Induced by Phage 117
4. Discussion
4.1. wcaJ Mutation Provides K. pneumoniae Strain Kp36 Phage Resistance by the Loss of Phage Receptor (Colanic Acid) Biosynthesis
4.2. insA and insB Associated with a Frameshift Mutation in wcaJ
4.3. Phage 31 Infects Phage 117 Resistant Mutant
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Suttle, C.A. Viruses in the sea. Nature 2005, 437, 356. [Google Scholar] [CrossRef]
- Labrie, S.J.; Samson, J.E.; Moineau, S. Bacteriophage resistance mechanisms. Nat. Rev. Microbiol. 2010, 8, 317. [Google Scholar] [CrossRef]
- Castillo, D.; Rørbo, N.; Jørgensen, J.; Lange, J.; Tan, D.; Kalatzis, P.G.; Svenningsen, S.L.; Middelboe, M. Phage defense mechanisms and their genomic and phenotypic implications in the fish pathogen Vibrio anguillarum. FEMS Microbiol. Ecol. 2019, 95, fiz004. [Google Scholar] [CrossRef]
- Tan, D.; Svenningsen, S.L.; Middelboe, M. Quorum sensing determines the choice of antiphage defense strategy in Vibrio anguillarum. MBio 2015, 6, e00627–e00615. [Google Scholar] [CrossRef] [PubMed]
- Podschun, R.; Ullmann, U. Klebsiella spp. as nosocomial pathogens: Epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin. Microbiol. Rev. 1998, 11, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Zhang, Y.; Cheng, M.; Le, S.; Gu, J.; Bao, J.; Qin, J.; Guo, X.; Zhu, T. Characterization of Klebsiella pneumoniae ST11 Isolates and Their Interactions with Lytic Phages. Viruses 2019, 11, 1080. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-H.; Kuo, C.-F.; Wang, C.-H.; Wu, C.-M.; Tsao, N. Experimental phage therapy in treating Klebsiella pneumoniae-mediated liver abscesses and bacteremia in mice. Antimicrob. Agents Chemother. 2011, 55, 1358–1365. [Google Scholar] [CrossRef]
- Corbellino, M.; Kieffer, N.; Kutateladze, M.; Balarjishvili, N.; Leshkasheli, L.; Askilashvili, L.; Tsertsvadze, G.; Rimoldi, S.G.; Nizharadze, D.; Hoyle, N. Eradication of a Multidrug-Resistant, Carbapenemase-Producing Klebsiella pneumoniae Isolate Following Oral and Intra-rectal Therapy With a Custom Made, Lytic Bacteriophage Preparation. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Loc-Carrillo, C.; Abedon, S.T. Pros and cons of phage therapy. Bacteriophage 2011, 1, 111–114. [Google Scholar] [CrossRef]
- Kim, H.; Kim, M.; Bai, J.; Lim, J.-A.; Heu, S.; Ryu, S. Colanic Acid Is a Novel Phage Receptor of Pectobacterium carotovorum subsp. carotovorum Phage POP72. Front. Microbiol. 2019, 10, 143. [Google Scholar] [CrossRef]
- Patel, K.B.; Toh, E.; Fernandez, X.B.; Hanuszkiewicz, A.; Hardy, G.G.; Brun, Y.V.; Bernards, M.A.; Valvano, M.A. Functional characterization of UDP-glucose: Undecaprenyl-phosphate glucose-1-phosphate transferases of Escherichia coli and Caulobacter crescentus. J. Bacteriol. 2012, 194, 2646–2657. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Wang, G.; Le, S.; Wu, M.; Cheng, M.; Guo, Z.; Ji, Y.; Xi, H.; Zhao, C.; Wang, X. Three capsular polysaccharide synthesis-related glucosyltransferases, GT-1, GT-2 and WcaJ, are associated with virulence and phage sensitivity of Klebsiella pneumoniae. Front. Microbiol. 2019, 10, 1189. [Google Scholar] [CrossRef] [PubMed]
- Goldfarb, T.; Sberro, H.; Weinstock, E.; Cohen, O.; Doron, S.; Charpak-Amikam, Y.; Afik, S.; Ofir, G.; Sorek, R. BREX is a novel phage resistance system widespread in microbial genomes. Embo J. 2015, 34, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Hegstad, K.; Mikalsen, T.; Coque, T.; Werner, G.; Sundsfjord, A. Mobile genetic elements and their contribution to the emergence of antimicrobial resistant Enterococcus faecalis and Enterococcus faecium. Clin. Microbiol. Infect. 2010, 16, 541–554. [Google Scholar] [CrossRef] [PubMed]
- Guinane, C.M.; Kent, R.M.; Norberg, S.; Hill, C.; Fitzgerald, G.F.; Stanton, C.; Ross, R.P. Host specific diversity in Lactobacillus johnsonii as evidenced by a major chromosomal inversion and phage resistance mechanisms. PLoS ONE 2011, 6, e18740. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Wang, S.; Chen, W.; Song, L.; Zhang, Y.; Shen, Z.; Yu, F.; Li, M.; Ji, Q. CRISPR-Cas9 and CRISPR-assisted cytidine deaminase enable precise and efficient genome editing in Klebsiella pneumoniae. Appl. Environ. Microbiol. 2018, 84, e01834–e01818. [Google Scholar] [CrossRef]
- Qian, H.; Yu, H.; Li, P.; Zhu, E.; Yao, Q.; Tai, C.; Deng, Z.; Gerdes, K.; He, X.; Gan, J. Toxin–antitoxin operon kacAT of Klebsiella pneumoniae is regulated by conditional cooperativity via a W-shaped KacA–KacT complex. Nucleic Acids Res. 2019, 47, 7690–7702. [Google Scholar] [CrossRef]
- Koonin, E.V.; Wolf, Y.I. Is evolution Darwinian or/and Lamarckian? Biol. Direct 2009, 4, 42. [Google Scholar] [CrossRef]
- Murray, A. Salvador Luria and Max Delbrück on random mutation and fluctuation tests. Genetics 2016, 202, 367. [Google Scholar] [CrossRef][Green Version]
- Luria, S.E.; Delbrück, M. Mutations of bacteria from virus sensitivity to virus resistance. Genetics 1943, 28, 491. [Google Scholar]
- Bertozzi Silva, J.; Storms, Z.; Sauvageau, D. Host receptors for bacteriophage adsorption. FEMS Microbiol. Lett. 2016, 363, fnw002. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, G.; Andrianopoulos, K.; Hobbs, M.; Reeves, P.R. Organization of the Escherichia coli K-12 gene cluster responsible for production of the extracellular polysaccharide colanic acid. J. Bacteriol. 1996, 178, 4885–4893. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, N.; Sorokin, A.; Anderson, I.; Galleron, N.; Candelon, B.; Kapatral, V.; Bhattacharyya, A.; Reznik, G.; Mikhailova, N.; Lapidus, A. Genome sequence of Bacillus cereus and comparative analysis with Bacillus anthracis. Nature 2003, 423, 87. [Google Scholar] [CrossRef] [PubMed]
- Sekine, Y.; Ohtsubo, E. Frameshifting is required for production of the transposase encoded by insertion sequence 1. Proc. Natl. Acad. Sci. USA 1989, 86, 4609–4613. [Google Scholar] [CrossRef]
- Zaleski, P.; Wojciechowski, M.; Piekarowicz, A. The role of Dam methylation in phase variation of Haemophilus influenzae genes involved in defence against phage infection. Microbiology 2005, 151, 3361–3369. [Google Scholar] [CrossRef]
- Seed, K.D.; Faruque, S.M.; Mekalanos, J.J.; Calderwood, S.B.; Qadri, F.; Camilli, A. Phase variable O antigen biosynthetic genes control expression of the major protective antigen and bacteriophage receptor in Vibrio cholerae O1. Plos Pathog. 2012, 8, e1002917. [Google Scholar] [CrossRef]
- Jacks, T.; Madhani, H.D.; Masiarz, F.R.; Varmus, H.E. Signals for ribosomal frameshifting in the Rous sarcoma virus gag-pol region. Cell 1988, 55, 447–458. [Google Scholar] [CrossRef]
- Ranjit, D.K.; Young, K.D. Colanic acid intermediates prevent de novo shape recovery of Escherichia coli spheroplasts, calling into question biological roles previously attributed to colanic acid. J. Bacteriol. 2016, 198, 1230–1240. [Google Scholar] [CrossRef]
- Hanna, A.; Berg, M.; Stout, V.; Razatos, A. Role of capsular colanic acid in adhesion of uropathogenic Escherichia coli. Appl. Environ. Microbiol. 2003, 69, 4474–4481. [Google Scholar] [CrossRef]
- Stewart, P.S.; Franklin, M.J. Physiological heterogeneity in biofilms. Nat. Rev. Microbiol. 2008, 6, 199. [Google Scholar] [CrossRef]
- Brockhurst, M.A.; Rainey, P.B.; Buckling, A. The effect of spatial heterogeneity and parasites on the evolution of host diversity. Proc. R. Soc. Lond. 2004, 271, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Heilmann, S.; Sneppen, K.; Krishna, S. Coexistence of phage and bacteria on the boundary of self-organized refuges. Proc. Natl. Acad. Sci. USA 2012, 109, 12828–12833. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, R.S.; Svenningsen, S.L.; Sneppen, K.; Mitarai, N. A growing microcolony can survive and support persistent propagation of virulent phages. Proc. Natl. Acad. Sci. USA 2018, 115, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Scholl, D.; Adhya, S.; Merril, C. Escherichia coli K1’s capsule is a barrier to bacteriophage T7. Appl. Environ. Microbiol. 2005, 71, 4872–4874. [Google Scholar] [CrossRef]
- Schrag, S.; Mittler, J. Host-parasite coexistence: The role of spatial refuges in stabilizing bacteria-phage interactions. Am. Nat. 1996, 148, 348–377. [Google Scholar] [CrossRef]

| Relative Efficiency of Plating | |||
|---|---|---|---|
| Klebsiella pneumoniae strains | Phage 117 (Podoviridae) | Phage 31 (Podoviridae) | Plaque morphology |
| Kp36 (WT) | 1 | 0 | Clear |
| Kp36-117R (phage 117-resistant mutant) | 0 | 1 | Clear |
| Kp36 ΔwcaJ (wcaJ in-frame deletion) | 0 | 0.70 ± 0.06 | Clear |
| Kp36-117R pBAD (empty plasmid) | 0 | 0.47 ± 0.07 | Clear |
| Kp36-117R pwcaJ (Plasmid with complementary wcaJ) | 0.66 ± 0.15 | 0 | Turbid |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, D.; Zhang, Y.; Qin, J.; Le, S.; Gu, J.; Chen, L.-k.; Guo, X.; Zhu, T. A Frameshift Mutation in wcaJ Associated with Phage Resistance in Klebsiella pneumoniae. Microorganisms 2020, 8, 378. https://doi.org/10.3390/microorganisms8030378
Tan D, Zhang Y, Qin J, Le S, Gu J, Chen L-k, Guo X, Zhu T. A Frameshift Mutation in wcaJ Associated with Phage Resistance in Klebsiella pneumoniae. Microorganisms. 2020; 8(3):378. https://doi.org/10.3390/microorganisms8030378
Chicago/Turabian StyleTan, Demeng, Yiyuan Zhang, Jinhong Qin, Shuai Le, Jingmin Gu, Li-kuang Chen, Xiaokui Guo, and Tongyu Zhu. 2020. "A Frameshift Mutation in wcaJ Associated with Phage Resistance in Klebsiella pneumoniae" Microorganisms 8, no. 3: 378. https://doi.org/10.3390/microorganisms8030378
APA StyleTan, D., Zhang, Y., Qin, J., Le, S., Gu, J., Chen, L.-k., Guo, X., & Zhu, T. (2020). A Frameshift Mutation in wcaJ Associated with Phage Resistance in Klebsiella pneumoniae. Microorganisms, 8(3), 378. https://doi.org/10.3390/microorganisms8030378





