Fitness Cost of blaNDM-5-Carrying p3R-IncX3 Plasmids in Wild-Type NDM-Free Enterobacteriaceae
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Strain Construction
2.2. Sampling of Recipients and Conjugative Transfer Assay
2.3. Whole-Genome Sequencing
2.4. Antimicrobial Susceptibility Testing
2.5. Plasmid Stability
2.6. Growth Kinetics
2.7. Biofilm Formation
3. Results and Discussion
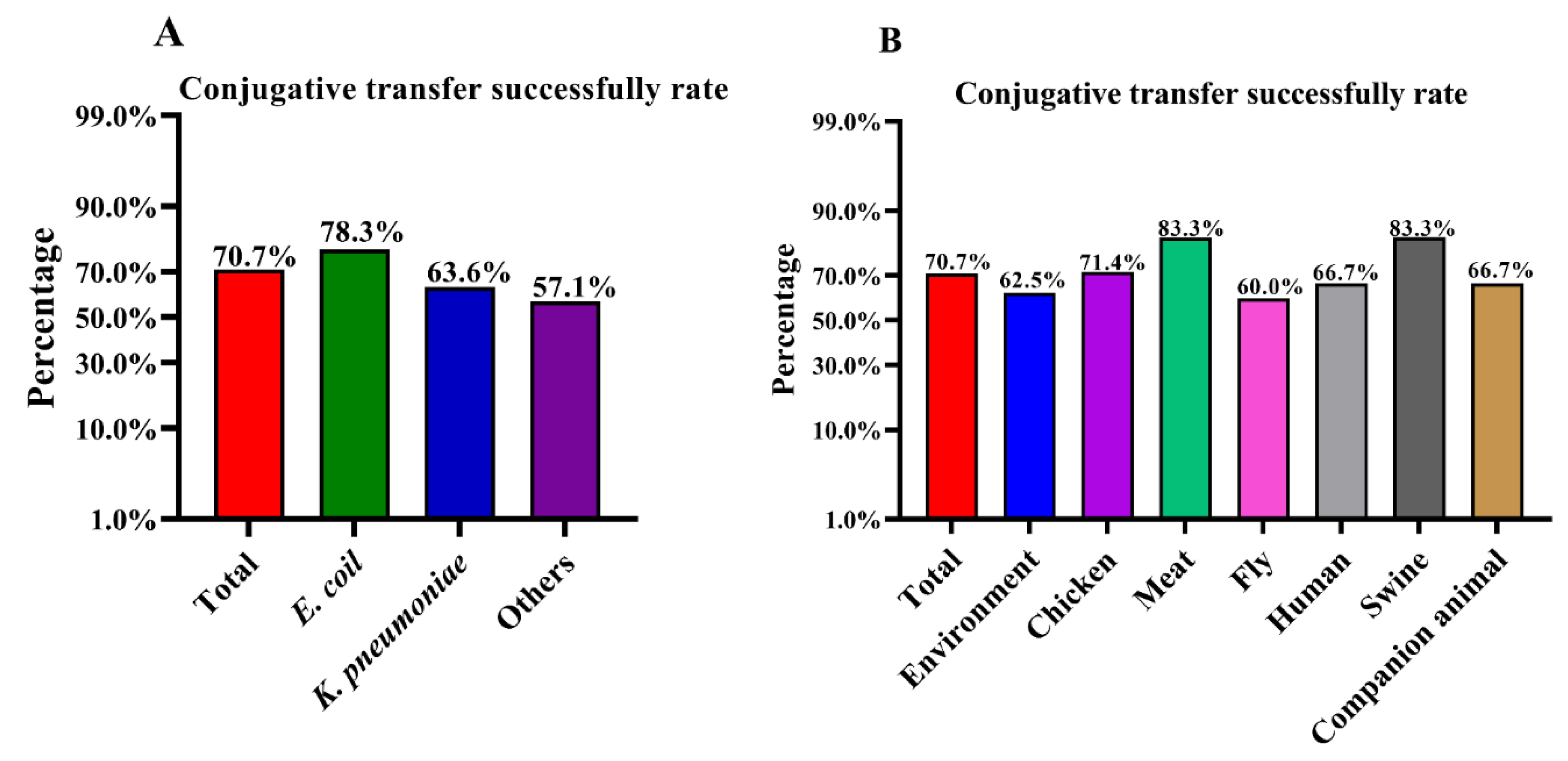
3.1. High Success Rates of Conjugative Transfer of the p3R-IncX3 Plasmid
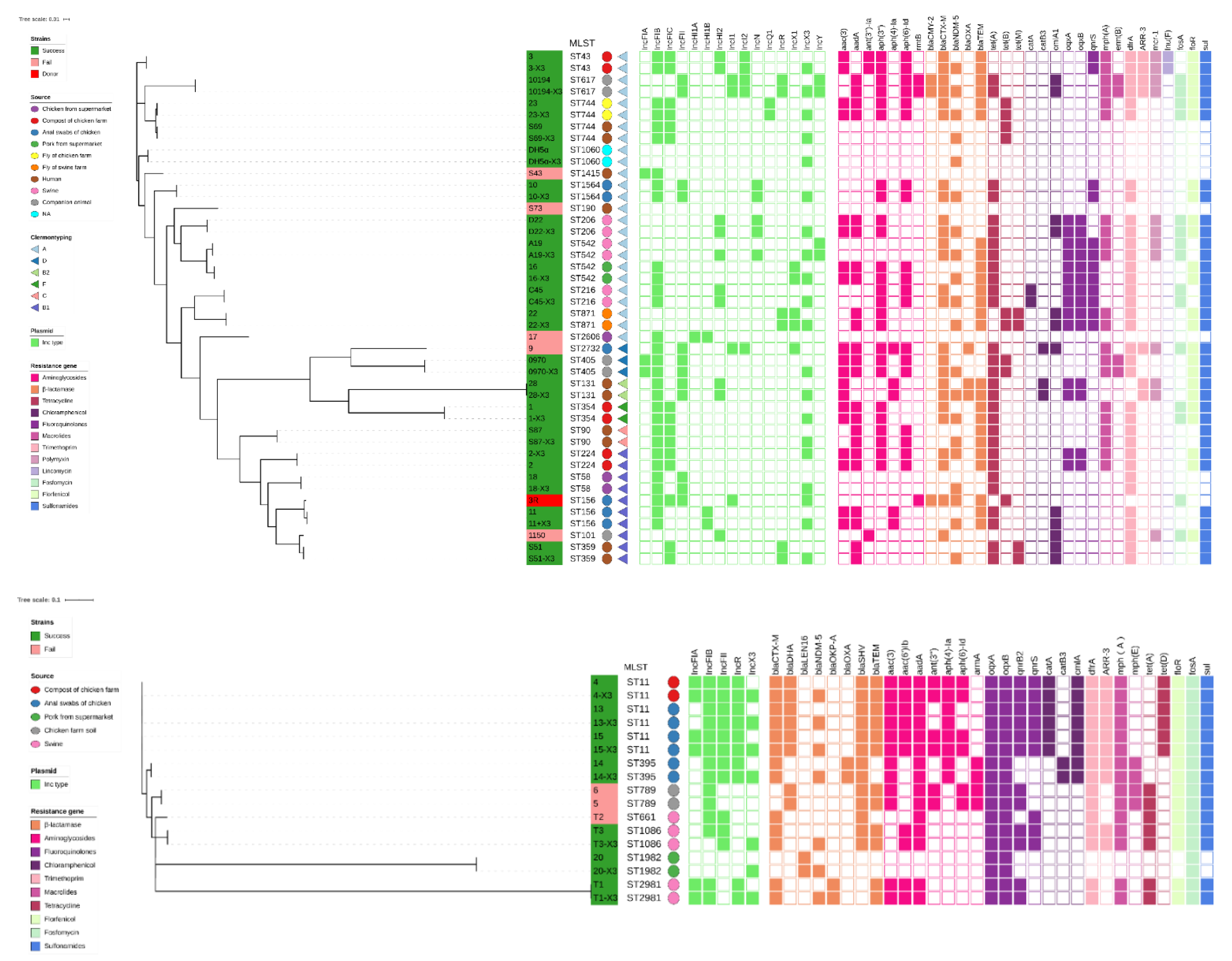
3.2. Phylogenetic Relationships among the Recipient Strains
3.3. Resistance Profiles and Stability of the Transconjugants Carrying the p3R-IncX3 Plasmid
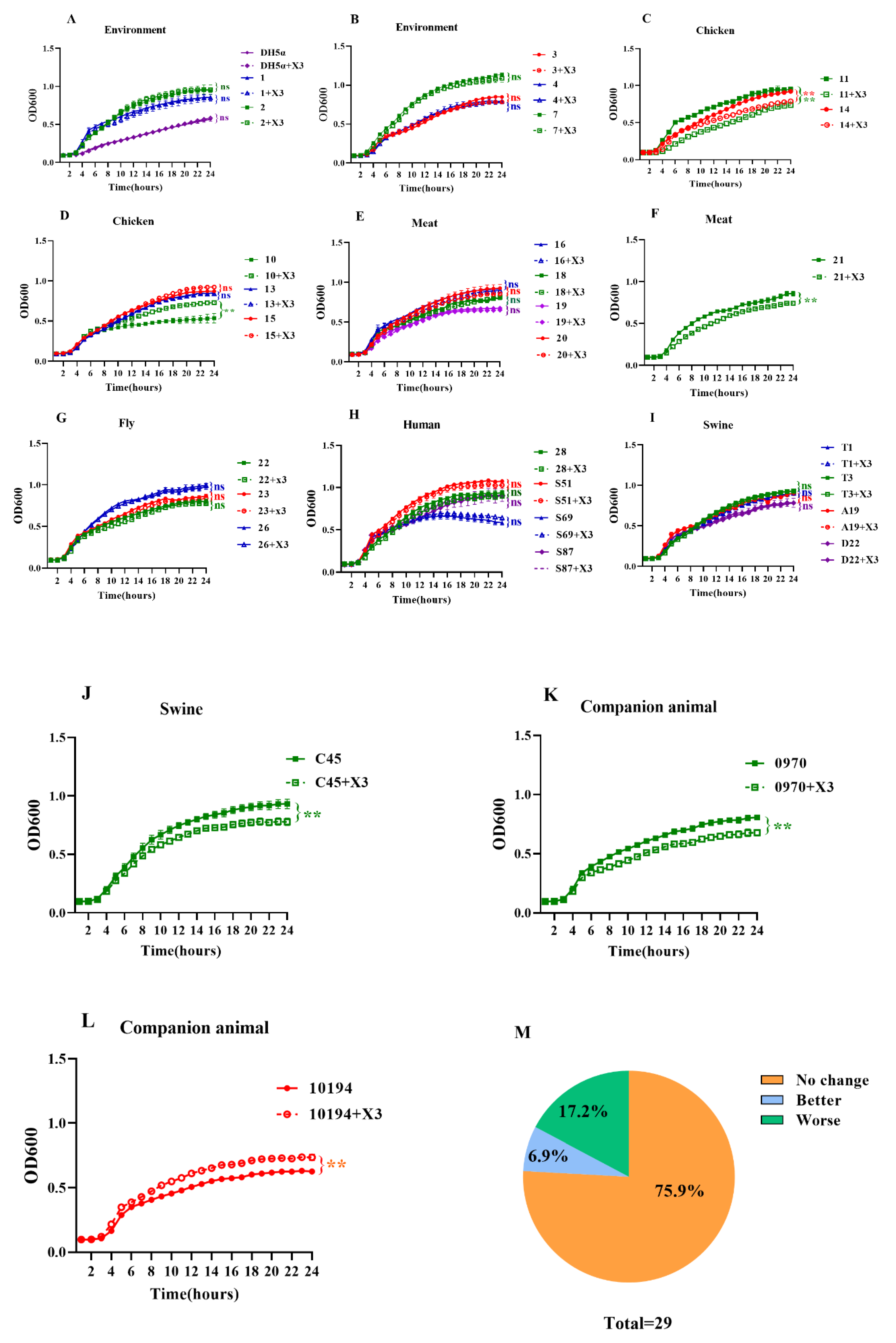
3.4. Growth Kinetics and Fitness Cost of the p3R-IncX3 Plasmid
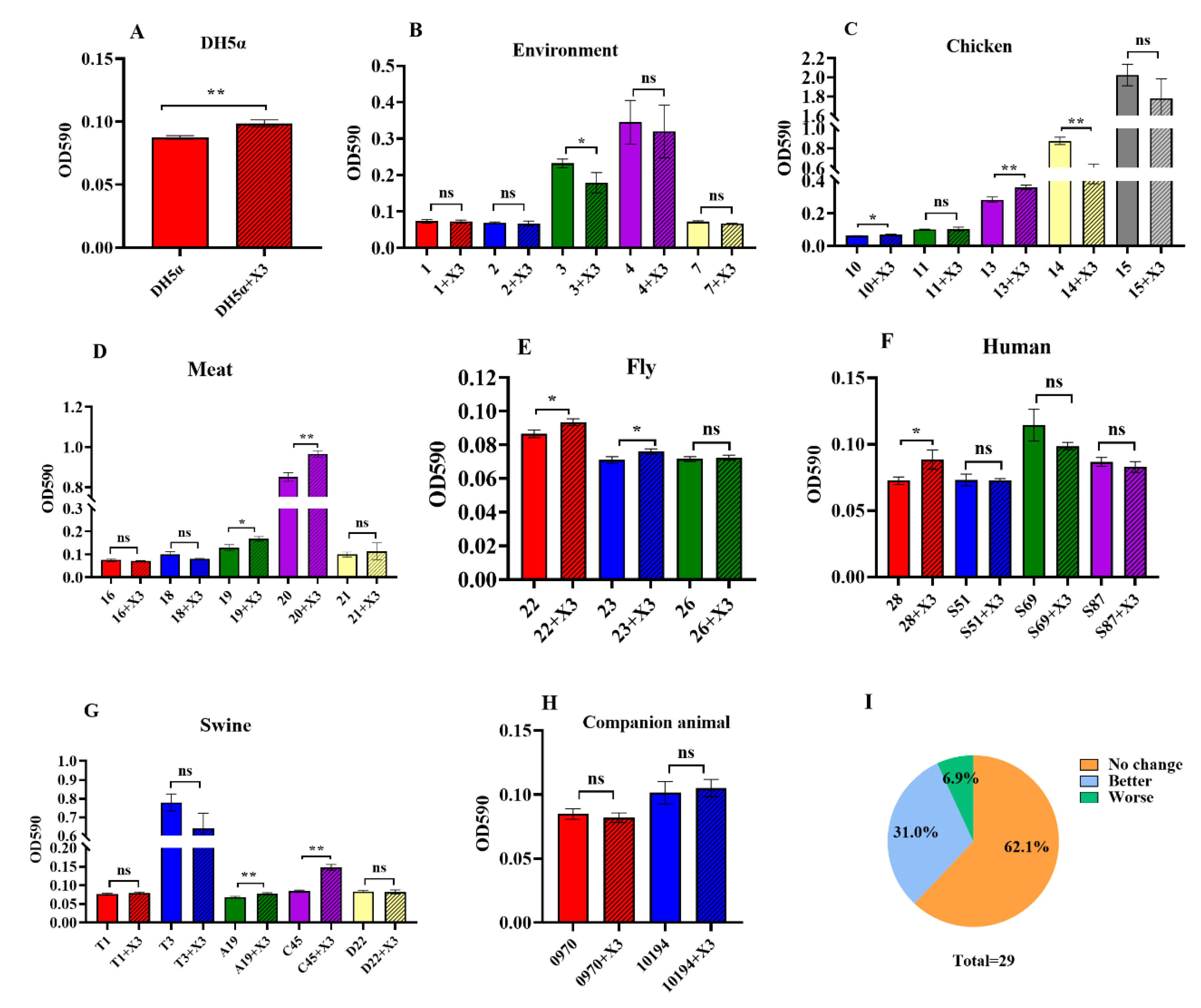
3.5. Changes in Biofilm Formation as a Result of p3R-IncX3 Plasmid Carriage
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Infectious Diseases Society of America. The 10×’20 Initiative: Pursuing a global commitment to develop 10 new antibacterial drugs by 2020. Clin. Infect. Dis. 2010, 50, 1081–1083. [Google Scholar] [CrossRef] [PubMed]
- Shankar, P.R.; Balasubramanium, R. Antimicrobial resistance: Global report on surveillance. Australas. Med. J. 2014, 7, 237. [Google Scholar]
- Yong, D.; Toleman, M.A.; Giske, C.G.; Cho, H.S.; Sundman, K.; Lee, K.; Walsh, T.R. Characterization of a new metallo-β-lactamase gene, blaNDM-1, and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob. Agents Chemother. 2009, 53, 5046–5054. [Google Scholar] [CrossRef] [PubMed]
- Schwaber, M.J.; Carmeli, Y. Carbapenem-resistant Enterobacteriaceae: A potential threat. JAMA 2008, 300, 2911–2913. [Google Scholar] [PubMed]
- Walsh, T.R.; Weeks, J.; Livermore, D.M.; Toleman, M.A. Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: An environmental point prevalence study. Lancet Infect. Dis. 2011, 11, 355–362. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, R.; Li, J.; Wu, Z.; Yin, W.; Schwarz, S.; Tyrrell, J.M.; Zheng, Y.; Wang, S.; Shen, Z.; et al. Comprehensive resistome analysis reveals the prevalence of NDM and MCR-1 in Chinese poultry production. Nat. Microbiol. 2017, 2, 16260. [Google Scholar] [CrossRef]
- Shen, Z.; Hu, Y.; Sun, Q.; Hu, F.; Zhou, H.; Shu, L.; Ma, T.; Shen, Y.; Wang, Y.; Li, J.; et al. Emerging carriage of NDM-5 and MCR-1 in Escherichia coli from healthy people in multiple regions in China: A cross sectional observational study. EClinicalMedicine 2018, 6, 11–20. [Google Scholar] [CrossRef]
- Nordmann, P.; Poirel, L.; Walsh, T.R.; Livermore, D.M. The emerging NDM carbapenemases. Trends Microbiol. 2011, 19, 588–595. [Google Scholar] [CrossRef]
- Guducuoglu, H.; Gursoy, N.C.; Yakupogullari, Y.; Parlak, M.; Karasin, G.; Sunnetcioglu, M.; Otlu, B. Hospital outbreak of a colistin-resistant, NDM-1- and OXA-48-producing Klebsiella pneumoniae: High mortality from pandrug resistance. Microb. Drug Resist. 2018, 24, 966–972. [Google Scholar] [CrossRef]
- Wu, W.; Feng, Y.; Tang, G.; Qiao, F.; McNally, A.; Zong, Z. NDM metallo-β-lactamases and their bacterial producers in health care settings. Clin. Microbiol. Rev. 2019, 32, e00115-18. [Google Scholar] [CrossRef]
- Johnson, T.J.; Bielak, E.M.; Fortini, D.; Hansen, L.H.; Hasman, H.; Debroy, C.; Nolan, L.K.; Carattoli, A. Expansion of the IncX plasmid family for improved identification and typing of novel plasmids in drug-resistant Enterobacteriaceae. Plasmid 2012, 68, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tong, M.K.; Chow, K.H.; Cheng, V.C.C.; Tse, C.W.S.; Wu, A.K.L.; Lai, R.W.M.; Luk, W.K.; Tsang, D.N.C.; Ho, P.L. Occurrence of highly conjugative IncX3 epidemic plasmid carrying blaNDM in Enterobacteriaceae isolates in geographically widespread areas. Front. Microbiol. 2018, 9, 2272. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Fu, Y.; Shen, M.; Huang, D.; Du, X.; Hu, Q.; Zhou, Y.; Wang, D.; Yu, Y. Dissemination of bla NDM-5 gene via an IncX3-type plasmid among non-clonal Escherichia coli in China. Antimicrob. Resist. Infect. 2018, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Krishnaraju, M.; Kamatchi, C.; Jha, A.K.; Devasena, N.; Vennila, R.; Sumathi, G.; Vaidyanathan, R. Complete sequencing of an IncX3 plasmid carrying blaNDM-5 allele reveals an early stage in the dissemination of the blaNDM gene. Indian J. Med. Microbiol. 2015, 33, 30–38. [Google Scholar] [PubMed]
- Zhang, Q.; Lv, L.; Huang, X.; Huang, Y.; Zhuang, Z.; Lu, J.; Liu, E.; Wan, M.; Xun, H.; Zhang, Z.; et al. Rapid increase in carbapenemase-producing enterobacteriaceae in retail meat driven by the spread of the blaNDM-5-carrying incX3 plasmid in China from 2016 to 2018. Antimicrob. Agents Chemother. 2019, 63, e00573-19. [Google Scholar] [CrossRef]
- Millan, A.S.; Maclean, R.C. Fitness costs of plasmids: A limit to plasmid transmission. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Baltrus, D.A. Exploring the costs of horizontal gene transfer. Trends Ecol. Evol. 2013, 28, 489–495. [Google Scholar] [CrossRef]
- Long, D.; Zhu, L.L.; Du, F.L.; Xiang, T.X.; Wan, L.G.; Wei, D.D.; Zhang, W.; Liu, Y. Phenotypical profile and global transcriptomic profile of Hypervirulent Klebsiella pneumoniae due to carbapenemase-encoding plasmid acquisition. BMC Genom. 2019, 20, 480. [Google Scholar] [CrossRef]
- Göttig, S.; Riedel-Christ, S.; Saleh, A.; Kempf, V.A.; Hamprecht, A. Impact of blaNDM-1 on fitness and pathogenicity of Escherichia coli and Klebsiella pneumoniae. Int. J. Antimicrob. Agents 2016, 47, 430–435. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef]
- Inouye, M.; Dashnow, H.; Raven, L.A.; Schultz, M.B.; Pope, B.J.; Tomita, T.; Zobel, J.; Holt, K.E. SRST2: Rapid genomic surveillance for public health and hospital microbiology labs. Genome Med. 2014, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Beghain, J.; Bridier-Nahmias, A.; Le Nagard, H.; Denamur, E.; Clermont, O. ClermonTyping: An easy-to-use and accurate in silico method for Escherichia genus strain phylotyping. Microb. Genom. 2018, 4, e000192. [Google Scholar] [CrossRef] [PubMed]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 26th ed.; CLSI supplement M100S; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2016. [Google Scholar]
- Sota, M.; Yano, H.; Hughes, J.M.; Daughdrill, G.W.; Abdo, Z.; Forney, L.J.; Top, E.M. Shifts in the host range of a promiscuous plasmid through parallel evolution of its replication initiation protein. ISME J. 2010, 4, 1568–1580. [Google Scholar] [CrossRef] [PubMed]
- The, K.H.; Flint, S.; French, N. Biofilm formation by Campylobacter jejuni in controlled mixed-microbial populations. Int. J. Food Microbiol. 2010, 143, 118–124. [Google Scholar]
- Nicolas-Chanoine, M.H.; Bertrand, X.; Madec, J.Y. Escherichia coli ST131, an intriguing clonal group. Clin. Microbiol. Rev. 2014, 27, 543–574. [Google Scholar] [CrossRef] [PubMed]
- Mathers, A.J.; Peirano, G.; Pitout, J.D. Escherichia coli ST131: The quintessential example of an international multiresistant high-risk clone. Adv. Appl. Microbiol. 2015, 90, 109–154. [Google Scholar] [PubMed]
- Liu, Y.Y.; Wang, Y.; Walsh, T.R.; Yi, L.X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Nicolau, D.P. Carbapenems: A potent class of antibiotics. Expert Opin. Pharmacother. 2008, 9, 3–37. [Google Scholar] [CrossRef]
- Andersson, D.I.; Hughes, D. Antibiotic resistance and its cost: Is it possible to reverse resistance? Nat. Rev. Microbiol. 2010, 8, 260. [Google Scholar] [CrossRef]
- Doyle, M.; Fookes, M.; Ivens, A.; Mangan, M.W.; Wain, J.; Dorman, C.J. An H-NS-like stealth protein aids horizontal DNA transmission in bacteria. Science 2007, 315, 251–252. [Google Scholar] [CrossRef]
- Di Luca, M.C.; Sørum, V.; Starikova, I.; Kloos, J.; Hülter, N.; Naseer, U.; Johnsen, P.; Samuelsen, Ø. Low biological cost of carbapenemase-encoding plasmids following transfer from Klebsiella pneumoniae to Escherichia coli. J. Antimicrob. Chemother. 2006, 72, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, B.; Thomson, N.R.; Thomas, C.M.; Brooks, C.M.; Sanders, M.; Delsol, A.A.; Ros, J.M.; Bennett, P.M.; Enne, V.I. Fitness of Escherichia coli strains carrying expressed and partially silent IncN and IncP1 plasmids. BMC Microbiol. 2012, 12, 53. [Google Scholar] [CrossRef] [PubMed]
- Rabin, N.; Zheng, Y.; Opoku-Temeng, C.; Du, Y.; Bonsu, E.; Sintim, H.O. Biofilm formation mechanisms and targets for developing antibiofilm agents. Future Med. Chem. 2015, 7, 493–512. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, N.M.; Martinez-Garcia, E.; Xavier, J.; Durham, W.M.; Kolter, R.; Kim, W.; Foster, K.R. Biofilm formation as a response to ecological competition. PLoS Biol. 2015, 13, e1002191. [Google Scholar] [CrossRef]
- Savage, V.J.; Chopra, I.; O’Neill, A.J. Staphylococcus aureus biofilms promote horizontal transfer of antibiotic resistance. Antimicrob. Agents Chemother. 2013, 57, 1968–1970. [Google Scholar] [CrossRef]
| No. | Species | Sources | MIC (mg/L) of Antimicrobial Agents | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MEM | CT | AMP | CRO | EFT | C | CN | AMC | TET | TGC | |||||||||||||
| + | − | + | − | + | − | + | − | + | − | + | − | + | − | + | − | + | − | + | − | |||
| DH5α | E. coli | TAKARA | 16 | 0.032 | 0.05 | 0.05 | >512 | 1 | 512 | 0.06 | 256 | 0.125 | 2 | 2 | 0.25 | 0.25 | 128/64 | 1/0.5 | 2 | 2 | 0.25 | 0.25 |
| 1 | E. coli | chicken manure compost | >128 | 0.032 | 2 | 2 | >512 | 1 | 512 | 128 | >256 | 64 | 128 | >128 | 16 | 16 | 128/64 | 16/8 | >128 | 128 | 1 | 0.5 |
| 2 | E. coli | chicken manure compost | 64 | 0.06 | 1 | 2 | >512 | >512 | >512 | 512 | >256 | >256 | 128 | >128 | 128 | >128 | >128/64 | 32/16 | 64 | 64 | 0.5 | 0.5 |
| 3 | E. coli | chicken manure compost | 64 | 0.06 | 8 | 8 | >512 | >512 | >512 | 512 | >256 | >256 | >128 | >128 | 64 | 64 | 128/64 | 16/8 | 128 | 128 | 1 | 0.5 |
| 4 | K. pneumoniae | chicken feces | 128 | 4 | 16 | 16 | >512 | >512 | 512 | 256 | >256 | 256 | >128 | >128 | >128 | >128 | >128/64 | >128/64 | 128 | 128 | 8 | 8 |
| 7 | E. cloacae | chicken manure compost | 32 | 0.06 | 1 | 1 | >512 | 256 | 512 | 0.125 | 256 | 1 | 64 | 128 | 0.25 | 0.25 | >128/64 | 128/64 | >128 | 128 | 2 | 0.5 |
| 10 | E. coli | chicken cloaca | 64 | 0.06 | 4 | 4 | >512 | >512 | >512 | 128 | >256 | 128 | 128 | 128 | 0.5 | 0.5 | >128/64 | 16/8 | 128 | 128 | 0.125 | 0.125 |
| 11 | E. coli | chicken cloaca | 64 | 0.06 | 2 | 2 | >512 | >512 | >512 | 0.125 | >256 | 1 | 32 | 32 | 8 | 8 | >128/64 | 32/16 | 128 | 128 | 0.125 | 0.125 |
| 13 | K. pneumoniae | chicken cloaca | 64 | 4 | 4 | 4 | >512 | >512 | >512 | 256 | >256 | 256 | 128 | 128 | 16 | 16 | >128/64 | 128/64 | >128 | >128 | 0.25 | 0.25 |
| 14 | K. pneumoniae | chicken cloaca | >128 | 0.25 | 1 | 1 | >512 | >512 | >512 | 512 | >256 | 256 | >128 | >128 | >128 | >128 | >128/64 | >128/64 | 32 | 16 | 1 | 0.5 |
| 15 | K. pneumoniae | chicken cloaca | >128 | 0.125 | 16 | 16 | >512 | >512 | >512 | 256 | >256 | 256 | >128 | >128 | 16 | 16 | >128/64 | >128/64 | >128 | >128 | 8 | 4 |
| 16 | E. coli | pork, farmer market | 128 | 0.032 | 1 | 2 | >512 | 512 | 512 | 0.03 | >256 | 0.5 | 128 | >128 | 64 | 64 | >128/64 | 16/8 | 128 | 128 | 0.25 | 0.06 |
| 18 | E. coli | Chicken, supermarket | 64 | 0.032 | 1 | 1 | >512 | 32 | 512 | 0.06 | >256 | 0.5 | 4 | 4 | 0.5 | 0.5 | >128/64 | 2/1 | 128 | 128 | 0.5 | 0.5 |
| 19 | R. ornithinolytica | Chicken, supermarket | 128 | 0.06 | 2 | 2 | >512 | 64 | 512 | 0.06 | 256 | 0.5 | 1 | 1 | 0.25 | 0.125 | >128/64 | 2/1 | 16 | 16 | 0.5 | 0.125 |
| 20 | K. variicola | Pork, retail store | 32 | 0.06 | 1 | 1 | >512 | 64 | >512 | 0.25 | >256 | 1 | 4 | 4 | 1 | 1 | >128/64 | 8/4 | 4 | 4 | 2 | 2 |
| 21 | C. freundii | Chicken, farmer market | 16 | 0.06 | 1 | 1 | >512 | 64 | 512 | 0.5 | 256 | 2 | 8 | 8 | 2 | 2 | >128/64 | 128/64 | >128 | >128 | 0.125 | 0.125 |
| 22 | E. coli | fly on pig farm | 32 | 0.032 | 1 | 1 | >512 | >512 | 512 | 0.25 | 256 | 0.25 | >128 | >128 | 0.25 | 1 | 128/64 | 8/4 | >128 | >128 | 0.25 | 0.125 |
| 23 | E. coli | fly on chicken farm | 64 | 0.06 | 1 | 2 | >512 | >512 | 512 | 128 | >256 | 128 | 128 | 64 | 64 | 16 | 128/64 | 16/8 | >128 | >128 | 0.25 | 0.125 |
| 26 | E. hormaechei | fly on pig farm | 64 | 0.06 | 1 | 1 | >512 | 512 | 512 | 0.25 | 256 | 1 | 128 | >128 | 2 | 2 | >128/64 | 128/64 | 128 | 128 | 0.5 | 0.5 |
| 28 | E. coli | human | 128 | 0.06 | 8 | 16 | >512 | >512 | >512 | 256 | >256 | 256 | 128 | >128 | 16 | 16 | >128/64 | 32/16 | 64 | 128 | 0.125 | 0.125 |
| T1 | K. quasipneumoniae | swine | 128 | 0.06 | 2 | 2 | >512 | >512 | >512 | 128 | >256 | 256 | 128 | >128 | 32 | 32 | >128/64 | 32/16 | 128 | 128 | 0.5 | 0.5 |
| T3 | K. pneumoniae | swine | >128 | 0.125 | 2 | 2 | >512 | >512 | >512 | 256 | >256 | 256 | >128 | >128 | 0.125 | 0.5 | >128/64 | 128/64 | >128 | >128 | 1 | 1 |
| A19 | E. coli | swine | 128 | 0.032 | 4 | 4 | >512 | 512 | 512 | 32 | >256 | 256 | 8 | 16 | 0.5 | 0.5 | >128/64 | 32/16 | 128 | 128 | 0.125 | 0.125 |
| C45 | E. coli | swine | 128 | 0.032 | 2 | 2 | >512 | 128 | 512 | 32 | >256 | 64 | >128 | 16 | 0.5 | 0.5 | >128/64 | 16/8 | 128 | 128 | 0.125 | 0.125 |
| D22 | E. coli | swine | 32 | 0.032 | 1 | 4 | >512 | 256 | 512 | 32 | 256 | 128 | >128 | >128 | 32 | 16 | 128/64 | 16/8 | 128 | 128 | 0.5 | 0.5 |
| 0970 | E. coli | companion animal | >128 | 0.125 | 2 | 2 | >512 | >512 | >512 | 256 | >256 | >256 | 128 | >128 | >128 | >128 | >128/64 | 16/8 | >128 | >128 | 0.25 | 0.125 |
| 10194 | E. coli | companion animal | 128 | 0.125 | 4 | 4 | >512 | >512 | >512 | >256 | >256 | >256 | 128 | >128 | >128 | >128 | >128/64 | >128/64 | 128 | 128 | 0.25 | 0.125 |
| S51 | E. coli | human | 32 | 0.125 | 1 | 1 | >512 | 4 | 512 | 0.06 | 256 | 1 | 4 | 4 | 1 | 1 | >128/64 | 2/1 | 128 | 128 | 0.25 | 0.125 |
| S69 | E. coli | human | 64 | 0.032 | 1 | 2 | >512 | 1 | 512 | 0.03 | 256 | 0.5 | 2 | 2 | 1 | 1 | 128/64 | 2/1 | 128 | >128 | 0.125 | 0.125 |
| S87 | E. coli | human | 64 | 0.032 | 1 | 2 | >512 | >512 | 512 | 256 | 256 | 1 | 4 | 4 | 1 | 1 | >128/64 | 16/8 | 128 | 128 | 0.25 | 0.125 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, T.; Fu, J.; Xie, N.; Ma, S.; Lei, L.; Zhai, W.; Shen, Y.; Sun, C.; Wang, S.; Shen, Z.; et al. Fitness Cost of blaNDM-5-Carrying p3R-IncX3 Plasmids in Wild-Type NDM-Free Enterobacteriaceae. Microorganisms 2020, 8, 377. https://doi.org/10.3390/microorganisms8030377
Ma T, Fu J, Xie N, Ma S, Lei L, Zhai W, Shen Y, Sun C, Wang S, Shen Z, et al. Fitness Cost of blaNDM-5-Carrying p3R-IncX3 Plasmids in Wild-Type NDM-Free Enterobacteriaceae. Microorganisms. 2020; 8(3):377. https://doi.org/10.3390/microorganisms8030377
Chicago/Turabian StyleMa, Tengfei, Jiani Fu, Ning Xie, Shizhen Ma, Lei Lei, Weishuai Zhai, Yingbo Shen, Chengtao Sun, Shaolin Wang, Zhangqi Shen, and et al. 2020. "Fitness Cost of blaNDM-5-Carrying p3R-IncX3 Plasmids in Wild-Type NDM-Free Enterobacteriaceae" Microorganisms 8, no. 3: 377. https://doi.org/10.3390/microorganisms8030377
APA StyleMa, T., Fu, J., Xie, N., Ma, S., Lei, L., Zhai, W., Shen, Y., Sun, C., Wang, S., Shen, Z., Wang, Y., Walsh, T. R., & Shen, J. (2020). Fitness Cost of blaNDM-5-Carrying p3R-IncX3 Plasmids in Wild-Type NDM-Free Enterobacteriaceae. Microorganisms, 8(3), 377. https://doi.org/10.3390/microorganisms8030377





