Microbiota of the Therapeutic Euganean Thermal Muds with a Focus on the Main Cyanobacteria Species
Abstract
1. Introduction
2. Materials and Methods
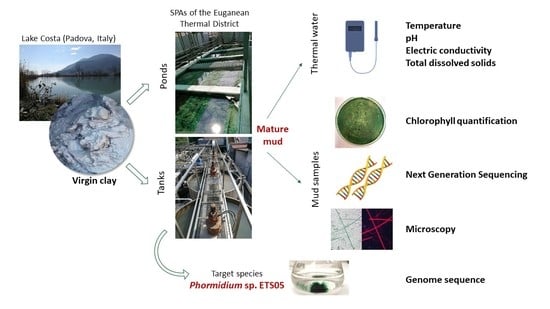
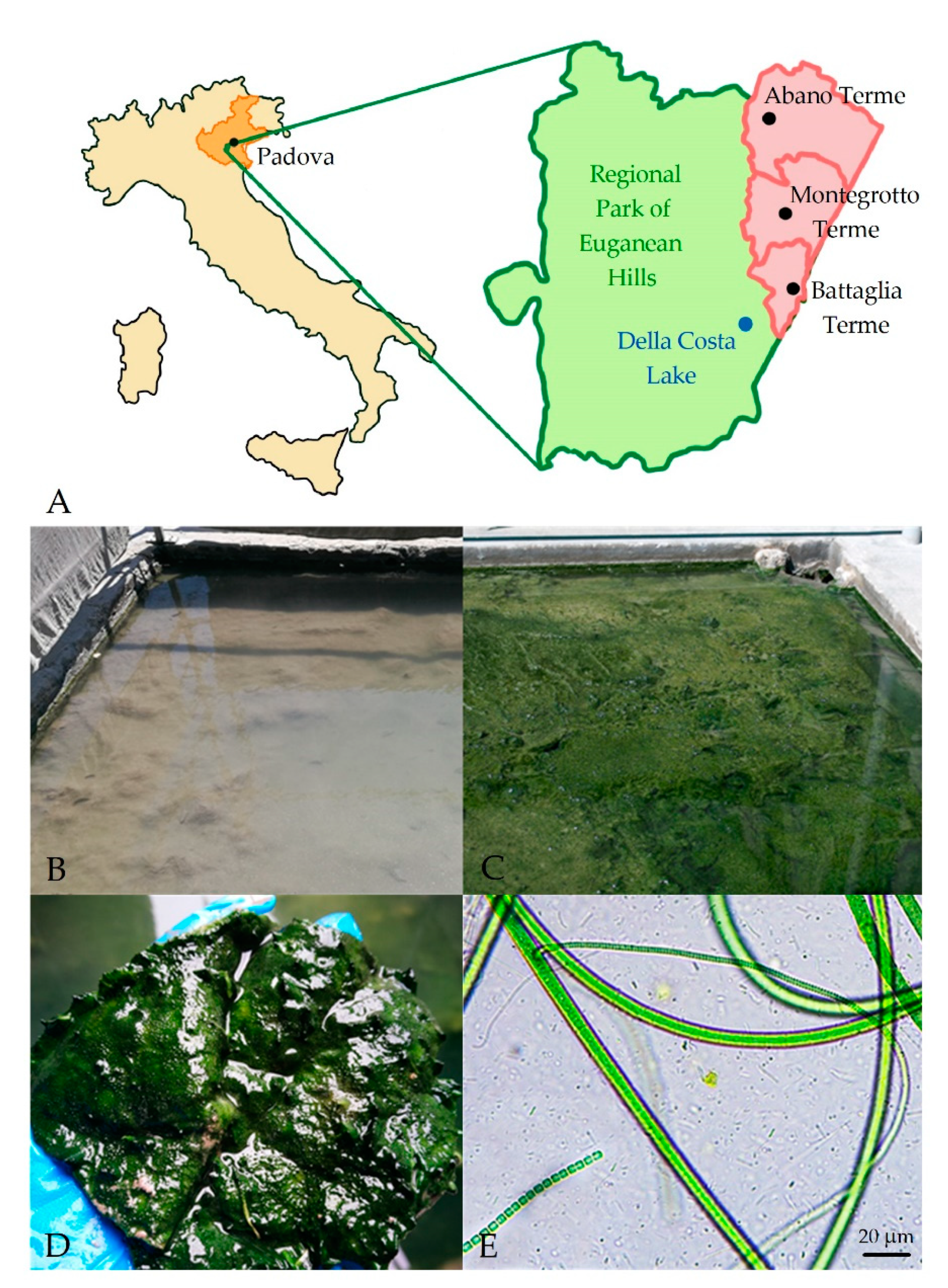
2.1. Study Site and Sample Collection
2.2. Pigments Extraction and Chlorophyll a (Chl a) Quantification
2.3. DNA Extraction, Amplification and Sequencing
2.4. Sequencing Data Analysis
2.5. Light and Fluorescence Microscopy Analyses
2.6. Phormidium sp. ETS-05 Cultivation, Genome Sequencing and Data Processing
2.7. Statistical Analyses
3. Results
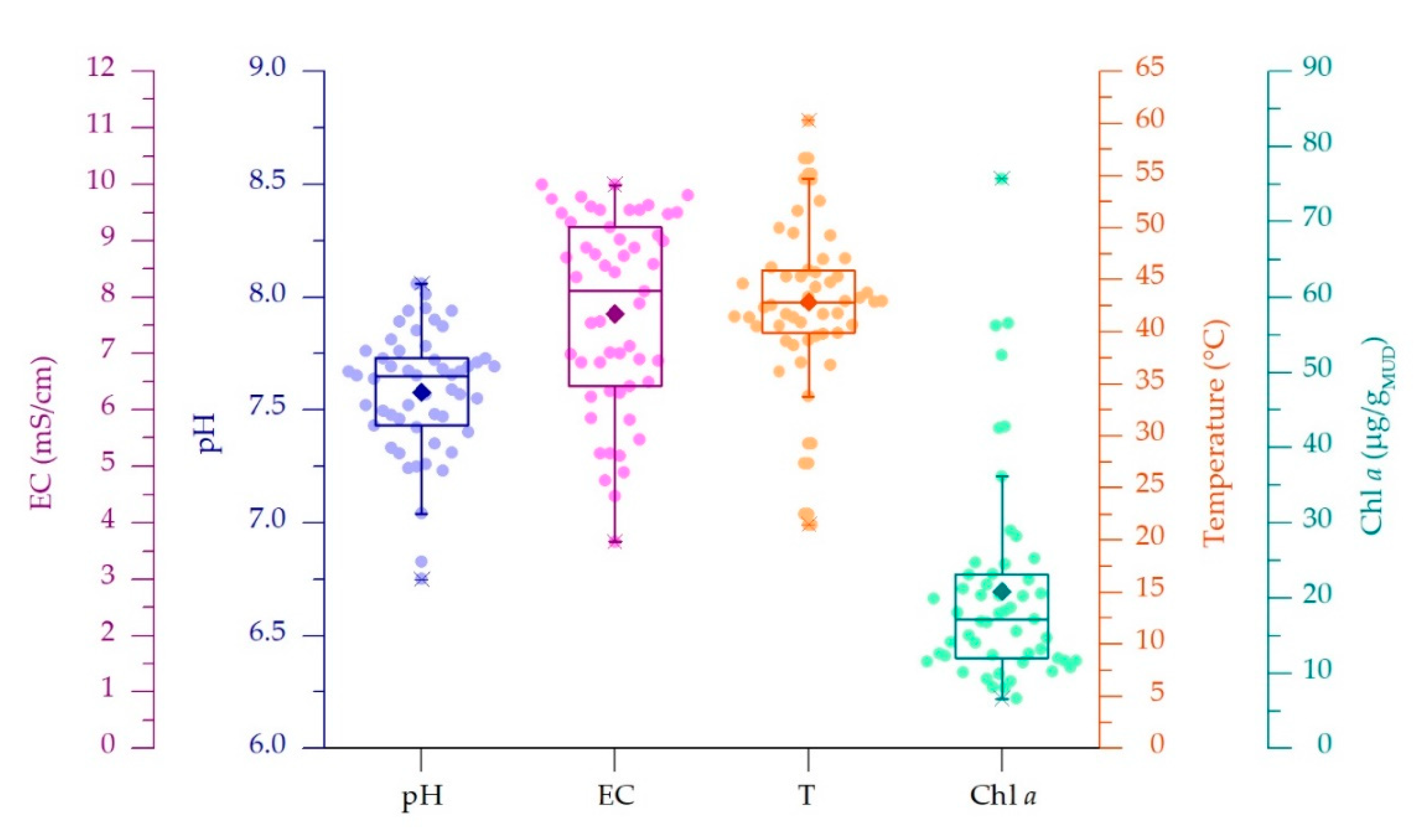
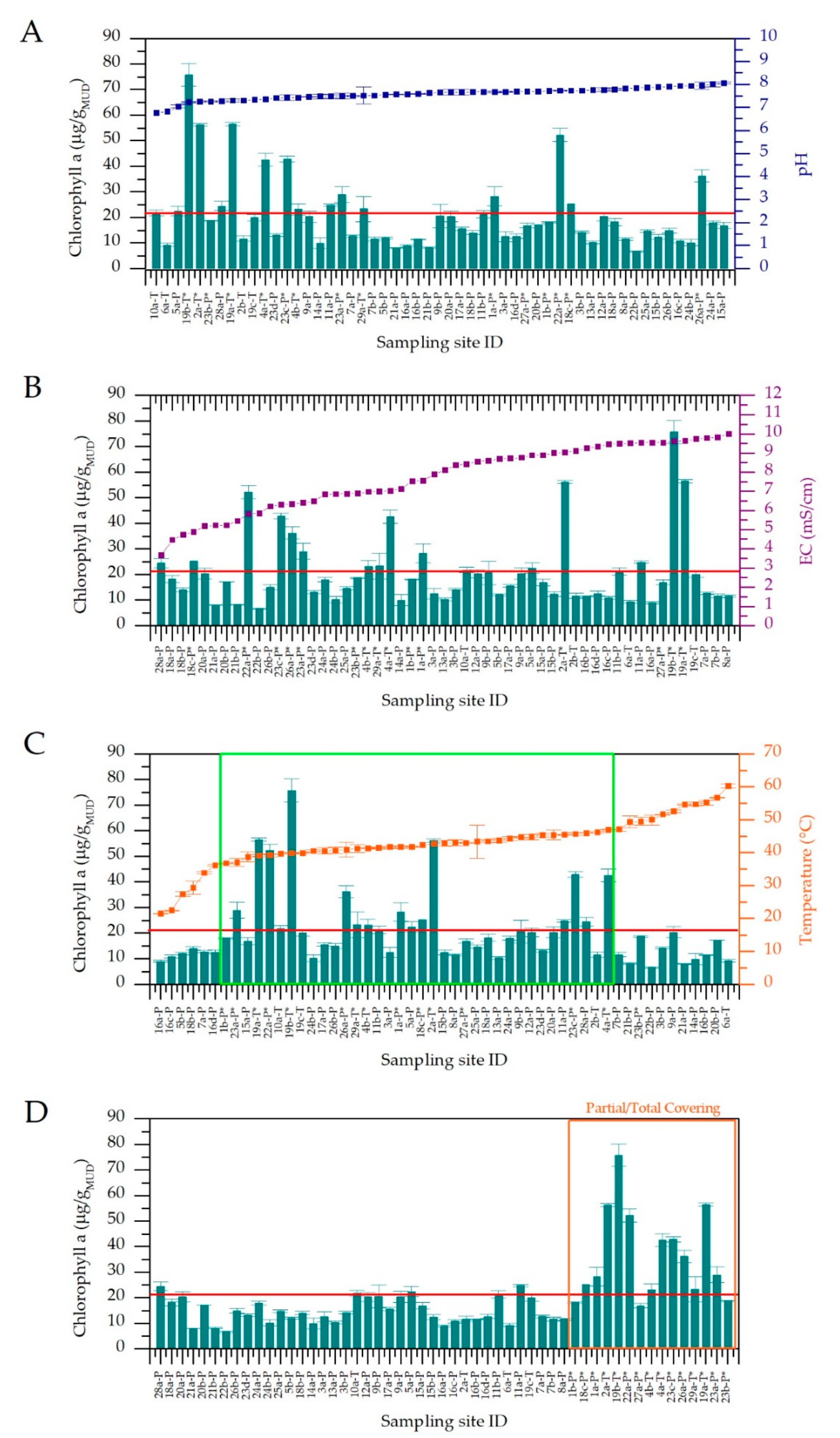
3.1. Physico-Chemical Parameters of Thermal Waters and Chlorophyll a Content of Muds
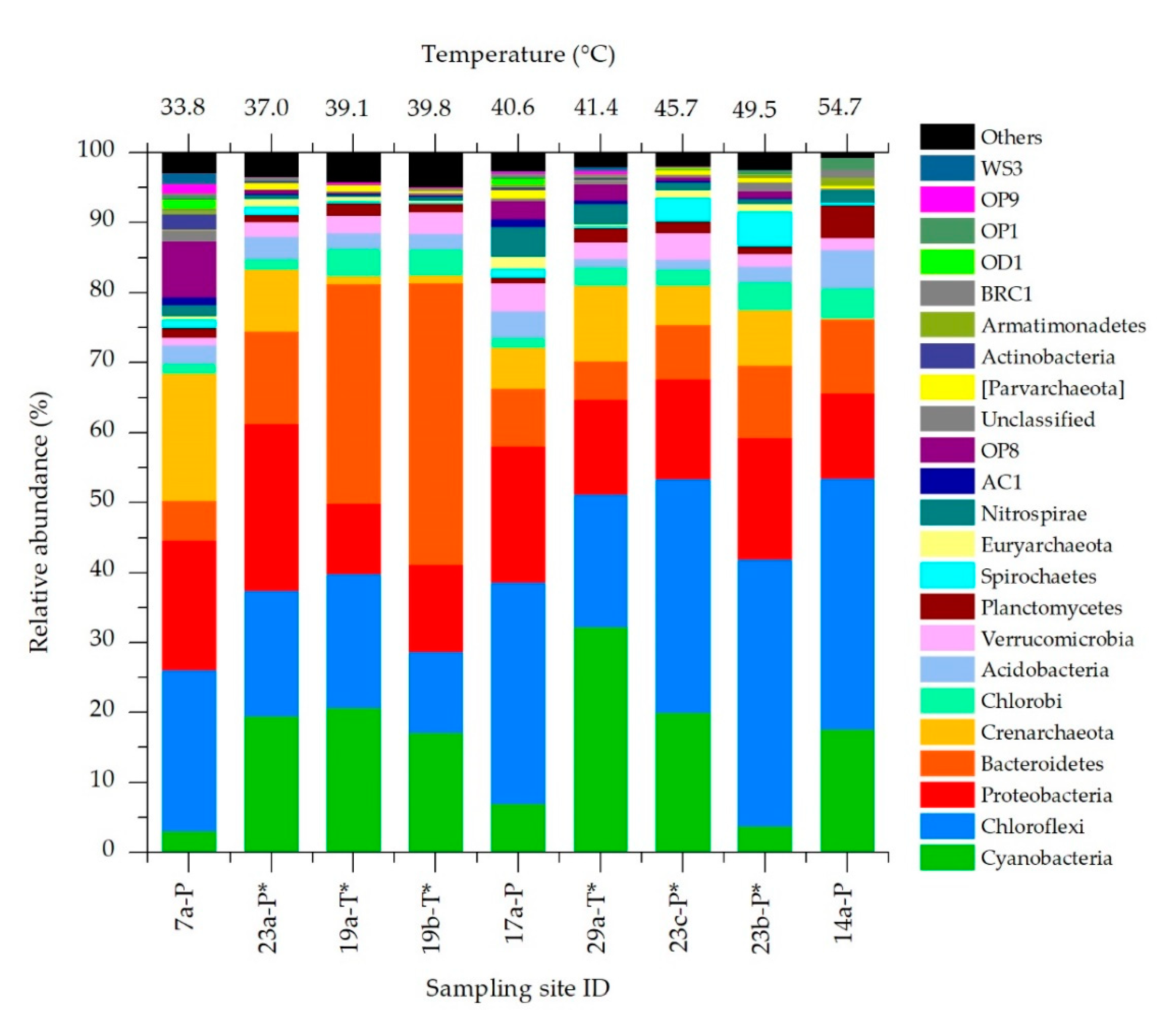
3.2. Bacteria and Archaea Community Composition
3.3. Effects of Temperature on Chloroflexi, Proteobacteria and Bacteroidetes Communities Structure
3.4. Temperature Effect on Cyanobacteria Community
3.5. Phormidium sp. ETS-05 Genome Sequence
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carretero, M.I. Clay minerals and their beneficial effects upon human health. A review. Appl. Clay Sci. 2002, 21, 155–163. [Google Scholar] [CrossRef]
- Carretero, M.I. Clays in pelotherapy. A review. Part II: Organic compounds, microbiology and medical applications. Appl. Clay Sci. 2020, 189, 105531. [Google Scholar] [CrossRef]
- Bellometti, S.; Cecchettin, M.; Galzigna, L. Mud pack therapy in osteoarthrosis Changes in serum levels of chondrocyte markers. Clin. Chim. Acta 1997, 268, 101–106. [Google Scholar] [CrossRef]
- Carretero, M.I. Clays in pelotherapy. A review. Part I: Mineralogy, chemistry, physical and physicochemical properties. Appl. Clay Sci. 2020, 189, 105526. [Google Scholar] [CrossRef]
- Pola, M.; Fabbri, P.; Piccinini, L.; Zampieri, D. Conceptual and numerical models of a tectonically-controlled geothermal system: A case study of the Euganean Geothermal System, Northern Italy. Cent. Eur. Geol. 2015, 58, 129–151. [Google Scholar] [CrossRef]
- Fabbri, P.; Pola, M.; Piccinini, L.; Zampieri, D.; Roghel, A.; Libera, N.D. Monitoring, utilization and sustainable development of a low-temperature geothermal resource: A case study of the Euganean Geothermal Field (NE, Italy). Geothermics 2017, 70, 281–294. [Google Scholar] [CrossRef]
- Calderan, A.; Carraro, A.; Honisch, C.; Lalli, A.; Ruzza, P.; Tateo, F. Euganean therapeutic mud (NE Italy): Chlorophyll a variations over two years and relationships with mineralogy and geochemistry. Appl. Clay Sci. 2020, 185, 105361. [Google Scholar] [CrossRef]
- Definizione e Aggiornamento dei Livelli Essenziali di Assistenza, di cui All’articolo 1, Comma 7, del Decreto Legislativo 30 Dicembre 1992, n. 502. (17A02015). Gazzetta Ufficiale della Repubblica Italiana, 18 March 2017.
- Carretero, M.I.; Gomes, C.S.F.; Tateo, F. Chapter 5.5—Clays, Drugs, and Human Health. In Developments in Clay Science; Bergaya, F., Lagaly, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 5, pp. 711–764. ISBN 1572-4352. [Google Scholar]
- Kaltenrieder, P.; Procacci, G.; Vannière, B.; Tinner, W. Vegetation and fire history of the Euganean Hills (Colli Euganei) as recorded by Lateglacial and Holocene sedimentary series from Lago della Costa (northeastern Italy). Holocene 2010, 20, 679–695. [Google Scholar] [CrossRef]
- Centini, M.; Tredici, M.R.; Biondi, N.; Buonocore, A.; Maffei Facino, R.; Anselmi, C. Thermal mud maturation: Organic matter and biological activity. Int. J. Cosmet. Sci. 2015, 37, 339–347. [Google Scholar] [CrossRef]
- Tateo, F.; Ravaglioli, A.; Andreoli, C.; Bonina, F.; Coiro, V.; Degetto, S.; Giaretta, A.; Menconi Orsini, A.; Puglia, C.; Summa, V. The in-vitro percutaneous migration of chemical elements from a thermal mud for healing use. Appl. Clay Sci. 2009, 44, 83–94. [Google Scholar] [CrossRef]
- Fioravanti, A.; Cantarini, L.; Guidelli, G.M.; Galeazzi, M. Mechanisms of action of spa therapies in rheumatic diseases: What scientific evidence is there? Rheumatol. Int. 2011, 31, 1–8. [Google Scholar] [CrossRef]
- Tolomio, C.; De Appolonia, F.; Moro, I.; Berrini, C.C. Thermophilic microalgae growth on different substrates and at different temperatures in experimental tanks in Abano Terme (Italy). Algol. Stud. Für Hydrobiol. Suppl. Vol. 2004, 111, 145–157. [Google Scholar] [CrossRef]
- Galzigna, L.; Moretto, C.; Lalli, A. Physical and biochemical changes of thermal mud after maturation. Biomed. Pharmacother. 1996, 50, 306–308. [Google Scholar] [CrossRef]
- Marcolongo, G.; De Appolonia, F.; Venzo, A.; Berrie, C.P.; Carofiglio, T.; Ceschi Berrini, C. Diacylglycerolipids isolated from a thermophile cyanobacterium from the Euganean hot springs. Nat. Prod. Res. 2006, 20, 766–774. [Google Scholar] [CrossRef]
- Gris, B.; Sforza, E.; Morosinotto, T.; Bertucco, A.; La Rocca, N. Influence of light and temperature on growth and high-value molecules productivity from Cyanobacterium aponinum. J. Appl. Phycol. 2017, 29, 1781–1790. [Google Scholar] [CrossRef]
- Lenti, M.; Gentili, C.; Pianezzi, A.; Marcolongo, G.; Lalli, A.; Cancedda, R.; Cancedda, F.D. Monogalactosyldiacylglycerol anti-inflammatory activity on adult articular cartilage. Nat. Prod. Res. 2009, 23, 754–762. [Google Scholar] [CrossRef]
- Ulivi, V.; Lenti, M.; Gentili, C.; Marcolongo, G.; Cancedda, R.; Descalzi Cancedda, F. Anti-inflammatory activity of monogalactosyldiacylglycerol in human articular cartilage in vitro: Activation of an anti-inflammatory cyclooxygenase-2 (COX-2) pathway. Arthritis Res. Ther. 2011, 13, R92. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsdottir, A.B.; Omarsdottir, S.; Brynjolfsdottir, A.; Paulsen, B.S.; Olafsdottir, E.S.; Freysdottir, J. Exopolysaccharides from Cyanobacterium aponinum from the Blue Lagoon in Iceland increase IL-10 secretion by human dendritic cells and their ability to reduce the IL-17+RORγt+/IL-10+FoxP3+ ratio in CD4+ T cells. Immunol. Lett. 2015, 163, 157–162. [Google Scholar] [CrossRef]
- Bruno, A.; Rossi, C.; Marcolongo, G.; Di Lena, A.; Venzo, A.; Berrie, C.P.; Corda, D. Selective in vivo anti-inflammatory action of the galactolipid monogalactosyldiacylglycerol. Eur. J. Pharmacol. 2005, 524, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Zampieri, R.M.; Adessi, A.; Caldara, F.; Codato, A.; Furlan, M.; Rampazzo, C.; De Philippis, R.; La Rocca, N.; Dalla Valle, L. Anti-Inflammatory Activity of Exopolysaccharides from Phormidium sp. ETS05, the Most Abundant Cyanobacterium of the Therapeutic Euganean Thermal Muds, Using the Zebrafish Model. Biomolecules 2020, 10, 582. [Google Scholar] [CrossRef]
- Ceschi Berrini, C.; De Appolonia, F.; Valle, L.D.; Komárek, J.; Andreoli, C. Morphological and molecular characterization of a thermophilic cyanobacterium (Oscillatoriales) from the Euganean Thermal Springs (Padua, Italy). Algol. Stud. Für Hydrobiol. Suppl. Vol. 2004, 113, 73–85. [Google Scholar]
- Lalli, A.; Andreoli, C.; Ceschi Berrini, C.; De Appolonia, F.; Marcolongo, G. Anti-Inflammatory Active Principles in Euganean Thermal Mud. European Patent 1571203 (B1), 24 July 2013. [Google Scholar]
- Andreoli, C.; Rascio, N. The algal flora in the Thermal Baths of Montegrotto Terme (Padua). Its distribution over one-year period. Int. Rev. Gesamten Hydrobiol. Hydrogr. 1975, 60, 857–871. [Google Scholar] [CrossRef]
- Moro, I.; Di Bella, M.; Rascio, N.; La Rocca, N.; Andreoli, C. Conferva duplisecta Pollini: Rediscovery in Euganean Thermal Springs (Italy) and new assignment to the Oscillatoria genus. Caryologia 2007, 60, 133–136. [Google Scholar]
- Moro, I.; Rascio, N.; La Rocca, N.; Sciuto, K.; Albertano, P.; Bruno, L.; Andreoli, C. Polyphasic characterization of a thermo-tolerant filamentous cyanobacterium isolated from the Euganean thermal muds (Padua, Italy). Eur. J. Phycol. 2010, 45, 143–154. [Google Scholar] [CrossRef]
- Moran, R. Formulae for determination of chlorophyllous pigments extracted with n,n-dimethylformamide. Plant Physiol. 1982, 69, 1376–1381. [Google Scholar] [CrossRef]
- Kambura, A.K.; Mwirichia, R.K.; Kasili, R.W.; Karanja, E.N.; Makonde, H.M.; Boga, H.I. Bacteria and Archaea diversity within the hot springs of Lake Magadi and Little Magadi in Kenya. BMC Microbiol. 2016, 16, 136. [Google Scholar] [CrossRef]
- The Earth Microbiome Project Consortium; Thompson, L.R.; Sanders, J.G.; McDonald, D.; Amir, A.; Ladau, J.; Locey, K.J.; Prill, R.J.; Tripathi, A.; Gibbons, S.M.; et al. A communal catalogue reveals Earth’s multiscale microbial diversity. Nature 2017, 551, 457–463. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108, 4516–4522. [Google Scholar] [CrossRef]
- Treu, L.; Kougias, P.G.; de Diego-Díaz, B.; Campanaro, S.; Bassani, I.; Fernández-Rodríguez, J.; Angelidaki, I. Two-year microbial adaptation during hydrogen-mediated biogas upgrading process in a serial reactor configuration. Bioresour. Technol. 2018, 264, 140–147. [Google Scholar] [CrossRef]
- Kovalovszki, A.; Treu, L.; Ellegaard, L.; Luo, G.; Angelidaki, I. Modeling temperature response in bioenergy production: Novel solution to a common challenge of anaerobic digestion. Appl. Energy 2020, 263, 114646. [Google Scholar] [CrossRef]
- Yarza, P.; Yilmaz, P.; Pruesse, E.; Glöckner, F.O.; Ludwig, W.; Schleifer, K.-H.; Whitman, W.B.; Euzéby, J.; Amann, R.; Rosselló-Móra, R. Uniting the classification of cultured and uncultured bacteria and archaea using 16S rRNA gene sequences. Nat. Rev. Microbiol. 2014, 12, 635–645. [Google Scholar] [CrossRef]
- Brient, L.; Lengronne, M.; Bertrand, E.; Rolland, D.; Sipel, A.; Steinmann, D.; Baudin, I.; Legeas, M.; Le Rouzic, B.; Bormans, M. A phycocyanin probe as a tool for monitoring cyanobacteria in freshwater bodies. J. Environ. Monit. 2008, 10, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Amarouche-Yala, S.; Benouadah, A.; El Ouahab Bentabet, A.; López-García, P. Morphological and phylogenetic diversity of thermophilic cyanobacteria in Algerian hot springs. Extremophiles 2014, 18, 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Anagnostidis, K.; Komárek, J. Modern approach to the classification system of cyanophytes. 1-Introduction. Algol. Stud. Arch. Für Hydrobiol. Suppl. Vol. 1985, 38–39, 291–302. [Google Scholar]
- Anagnostidis, K.; Komárek, J. Modern approach to the classification system of cyanophytes. 3-Oscillatoriales. Algol. Stud. Arch. Für Hydrobiol. Suppl. Vol. 1988, 50–53, 327–472. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Modern approach to the classification system of cyanophytes. 2. Chroococcales. Algol. Stud. Arch. Für Hydrobiol. Suppl. Vol. 1986, 43, 157–226. [Google Scholar]
- Komárek, J.; Kaštovský, J.; Mareš, J.; Johansen, J.R. Taxonomic classification of cyanoprokaryotes (cyanobacterial genera) 2014, using a polyphasic approach. Preslia 2014, 86, 295–335. [Google Scholar]
- Komárek, J. Several problems of the polyphasic approach in the modern cyanobacterial system. Hydrobiologia 2018, 811, 7–17. [Google Scholar] [CrossRef]
- Rueden, C.T.; Schindelin, J.; Hiner, M.C.; DeZonia, B.E.; Walter, A.E.; Arena, E.T.; Eliceiri, K.W. ImageJ2: ImageJ for the next generation of scientific image data. BMC Bioinform. 2017, 18, 529. [Google Scholar] [CrossRef]
- Hillebrand, H.; Dürselen, C.-D.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Lawton, L.; Marsalek, B.; Padisák, J.; Chorus, I. Chapter 12. Determination of cyanobacteria in the laboratory. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; World Health Organization: London, UK, 1999. [Google Scholar]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Wick, R.R.; Judd, L.M.; Holt, K.E. Performance of neural network basecalling tools for Oxford Nanopore sequencing. Genome Biol. 2019, 20, 129. [Google Scholar] [CrossRef] [PubMed]
- Zimin, A.V.; Marçais, G.; Puiu, D.; Roberts, M.; Salzberg, S.L.; Yorke, J.A. The MaSuRCA genome assembler. Bioinformatics 2013, 29, 2669–2677. [Google Scholar] [CrossRef]
- Seemann, T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the Rapid Annotation of microbial genomes using Subsystems Technology (RAST). Nucleic Acids Res. 2014, 42, D206–D214. [Google Scholar] [CrossRef]
- Huang, H.; Song, C.-C.; Yang, Z.-L.; Dong, Y.; Hu, Y.-Z.; Gao, F. Identification of the Replication Origins from Cyanothece ATCC 51142 and Their Interactions with the DnaA Protein: From In Silico to In Vitro Studies. Front. Microbiol. 2015, 6, 1370. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Zhang, C.-T.; Gao, F. Ori-Finder 2, an integrated tool to predict replication origins in the archaeal genomes. Front. Microbiol. 2014, 5, 482. [Google Scholar] [CrossRef]
- Bosi, E.; Donati, B.; Galardini, M.; Brunetti, S.; Sagot, M.-F.; Lió, P.; Crescenzi, P.; Fani, R.; Fondi, M. MeDuSa: A multi-draft based scaffolder. Bioinformatics 2015, 31, 2443–2451. [Google Scholar] [CrossRef]
- Rutherford, K.; Parkhill, J.; Crook, J.; Horsnell, T.; Rice, P.; Rajandream, M.-A.; Barrell, B. Artemis: Sequence visualization and annotation. Bioinformatics 2000, 16, 944–945. [Google Scholar] [CrossRef]
- Kougias, P.G.; Campanaro, S.; Treu, L.; Zhu, X.; Angelidaki, I. A novel archaeal species belonging to Methanoculleus genus identified via de-novo assembly and metagenomic binning process in biogas reactors. Anaerobe 2017, 46, 23–32. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y. KEGG Mapper for inferring cellular functions from protein sequences. Protein Sci. 2020, 29, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Gurevich, A.; Saveliev, V.; Vyahhi, N.; Tesler, G. QUAST: Quality assessment tool for genome assemblies. Bioinformatics 2013, 29, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Campanaro, S.; Treu, L.; Rodriguez-R, L.M.; Kovalovszki, A.; Ziels, R.M.; Maus, I.; Zhu, X.; Kougias, P.G.; Basile, A.; Luo, G.; et al. New insights from the biogas microbiome by comprehensive genome-resolved metagenomics of nearly 1600 species originating from multiple anaerobic digesters. Biotechnol. Biofuels 2020, 13, 25. [Google Scholar] [CrossRef]
- Hyatt, D.; Chen, G.-L.; LoCascio, P.F.; Land, M.L.; Larimer, F.W.; Hauser, L.J. Prodigal: Prokaryotic gene recognition and translation initiation site identification. BMC Bioinform. 2010, 11, 119. [Google Scholar] [CrossRef]
- Segata, N.; Börnigen, D.; Morgan, X.C.; Huttenhower, C. PhyloPhlAn is a new method for improved phylogenetic and taxonomic placement of microbes. Nat. Commun. 2013, 4, 2304. [Google Scholar] [CrossRef]
- Huson, D.H.; Richter, D.C.; Rausch, C.; Dezulian, T.; Franz, M.; Rupp, R. Dendroscope: An interactive viewer for large phylogenetic trees. BMC Bioinform. 2007, 8, 460. [Google Scholar] [CrossRef]
- Olm, M.R.; Brown, C.T.; Brooks, B.; Banfield, J.F. dRep: A tool for fast and accurate genomic comparisons that enables improved genome recovery from metagenomes through de-replication. ISME J. 2017, 11, 2864–2868. [Google Scholar] [CrossRef] [PubMed]
- Varghese, N.J.; Mukherjee, S.; Ivanova, N.; Konstantinidis, K.T.; Mavrommatis, K.; Kyrpides, N.C.; Pati, A. Microbial species delineation using whole genome sequences. Nucleic Acids Res. 2015, 43, 6761–6771. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Shih, P.M.; Wu, D.; Latifi, A.; Axen, S.D.; Fewer, D.P.; Talla, E.; Calteau, A.; Cai, F.; Tandeau de Marsac, N.; Rippka, R.; et al. Improving the coverage of the cyanobacterial phylum using diversity-driven genome sequencing. Proc. Nat. Acad. Sci. USA 2013, 110, 1053–1058. [Google Scholar] [CrossRef]
- Galzigna, L.; Ceschi-Berrini, C.; Moschin, E.; Tolomio, C. Thermal mud-pack as an anti-inflammatory treatment. Biomed. Pharmacother. 1998, 52, 408–409. [Google Scholar] [CrossRef]
- Tolomio, C.; Ceschi-Berrini, C.; Moschin, E.; Galzigna, L. Colonization by diatoms and antirheumatic activity of thermal mud. Cell Biochem. Funct. 1999, 17, 29–33. [Google Scholar] [CrossRef]
- Baschini, M.T.; Pettinari, G.R.; Vallés, J.M.; Aguzzi, C.; Cerezo, P.; López-Galindo, A.; Setti, M.; Viseras, C. Suitability of natural sulphur-rich muds from Copahue (Argentina) for use as semisolid health care products. Appl. Clay Sci. 2010, 49, 205–212. [Google Scholar] [CrossRef]
- da Silva, P.S.C.; Torrecilha, J.K.; de Macedo Gouvea, P.F.; Máduar, M.F.; de Oliveira, S.M.B.; Scapin, M.A. Chemical and radiological characterization of Peruíbe Black Mud. Appl. Clay Sci. 2015, 118, 221–230. [Google Scholar] [CrossRef]
- Çelik Karakaya, M.; Karakaya, N.; Sarıoğlan, Ş.; Koral, M. Some properties of thermal muds of some spas in Turkey. Appl. Clay Sci. 2010, 48, 531–537. [Google Scholar] [CrossRef]
- Gálvez, I.; Torres-Piles, S.; Ortega, E. Innate/inflammatory bioregulation and clinical effectiveness of whole-body hyperthermia (balneotherapy) in elderly patients with osteoarthritis. Int. J. Hyperth. 2018, 35, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Capdepuy, M.; Goya, O.; Quentin-Noury, C. Sanitary aspects of the thermal muds in Aquitaine. Int. J. Environ. Health Res. 1994, 4, 1–6. [Google Scholar] [CrossRef]
- Ma’or, Z.; Henis, Y.; Alon, Y.; Orlov, E.; Sorensen, K.B.; Oren, A. Antimicrobial properties of Dead Sea black mineral mud. Int. J. Dermatol. 2006, 45, 504–511. [Google Scholar] [CrossRef]
- Ramata-Stunda, A.; Petrina, Z.; Mekss, P.; Kizane, G.; Silamikele, B.; Muiznieks, I.; Nikolajeva, V. Microbiological characterization and sterilization-induced changes in the profile of the hydrophobic organic substances in Latvian balneological peat. Int. J. Environ. Sci. Technol. 2015, 12, 2371–2380. [Google Scholar] [CrossRef]
- Moro, I.; Rascio, N.; La Rocca, N.; Di Bella, M.; Andreoli, C. Cyanobacterium aponinum, a new Cyanoprokaryote from the microbial mat of Euganean thermal springs (Padua, Italy). Algol. Stud. 2007, 123, 1–15. [Google Scholar] [CrossRef]
- Sciuto, K.; Moro, I. Detection of the new cosmopolitan genus Thermoleptolyngbya (Cyanobacteria, Leptolyngbyaceae) using the 16S rRNA gene and 16S–23S ITS region. Mol. Phylogenet. Evol. 2016, 105, 15–35. [Google Scholar] [CrossRef]
- Miller, S.R.; Strong, A.L.; Jones, K.L.; Ungerer, M.C. Bar-Coded Pyrosequencing Reveals Shared Bacterial Community Properties along the Temperature Gradients of Two Alkaline Hot Springs in Yellowstone National Park. Appl. Environ. Microbiol. 2009, 75, 4565–4572. [Google Scholar] [CrossRef] [PubMed]
- Portillo, M.C.; Sririn, V.; Kanoksilapatham, W.; Gonzalez, J.M. Differential microbial communities in hot spring mats from Western Thailand. Extremophiles 2009, 13, 321–331. [Google Scholar] [CrossRef]
- Cuecas, A.; Portillo, M.C.; Kanoksilapatham, W.; Gonzalez, J.M. Bacterial Distribution Along a 50 °C Temperature Gradient Reveals a Parceled Out Hot Spring Environment. Microb. Ecol. 2014, 68, 729–739. [Google Scholar] [CrossRef]
- Ghosh, D.; Bal, B.; Kashyap, V.K.; Pal, S. Molecular Phylogenetic Exploration of Bacterial Diversity in a Bakreshwar (India) Hot Spring and Culture of Shewanella-Related Thermophiles. Appl. Environ. Microbiol. 2003, 69, 4332–4336. [Google Scholar] [CrossRef]
- Hanada, S.; Takaichi, S.; Matsuura, K.; Nakamura, K. Roseiflexus castenholzii gen. nov., sp. nov., a thermophilic, filamentous, photosynthetic bacterium that lacks chlorosomes. Int. J. Syst. Evol. Microbiol. 2002, 52, 187–193. [Google Scholar] [CrossRef]
- Hirayama, H.; Takai, K.; Inagaki, F.; Nealson, K.H.; Horikoshi, K. Thiobacter subterraneus gen. nov., sp. nov., an obligately chemolithoautotrophic, thermophilic, sulfur-oxidizing bacterium from a subsurface hot aquifer. Int. J. Syst. Evol. Microbiol. 2005, 55, 467–472. [Google Scholar] [CrossRef][Green Version]
- Albuquerque, L.; Rainey, F.A.; Nobre, M.F.; da Costa, M.S. Schleiferia thermophila gen. nov., sp. nov., a slightly thermophilic bacterium of the phylum ‘Bacteroidetes’ and the proposal of Schleiferiaceae fam. nov. Int. J. Syst. Evol. Microbiol. 2011, 61, 2450–2455. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baena, S.; Perdomo, N.; Carvajal, C.; Díaz, C.; Patel, B.K.C. Desulfosoma caldarium gen. nov., sp. nov., a thermophilic sulfate-reducing bacterium from a terrestrial hot spring. Int. J. Syst. Evol. Microbiol. 2011, 61, 732–736. [Google Scholar] [CrossRef]
- Tang, K.-H.; Barry, K.; Chertkov, O.; Dalin, E.; Han, C.S.; Hauser, L.J.; Honchak, B.M.; Karbach, L.E.; Land, M.L.; Lapidus, A.; et al. Complete genome sequence of the filamentous anoxygenic phototrophic bacterium Chloroflexus aurantiacus. BMC Genom. 2011, 12, 334. [Google Scholar] [CrossRef]
- Grégoire, P.; Fardeau, M.-L.; Guasco, S.; Lagière, J.; Cambar, J.; Michotey, V.; Bonin, P.; Ollivier, B. Desulfosoma profundi sp. nov., a thermophilic sulfate-reducing bacterium isolated from a deep terrestrial geothermal spring in France. Antonie van Leeuwenhoek 2012, 101, 595–602. [Google Scholar] [CrossRef]
- Boden, R.; Hutt, L.P.; Rae, A.W. Reclassification of Thiobacillus aquaesulis (Wood & Kelly, 1995) as Annwoodia aquaesulis gen. nov., comb. nov., transfer of Thiobacillus (Beijerinck, 1904) from the Hydrogenophilales to the Nitrosomonadales, proposal of Hydrogenophilalia class. nov. within the ‘Proteobacteria’, and four new families within the orders Nitrosomonadales and Rhodocyclales. Int. J. Syst. Evol. Microbiol. 2017, 67, 1191–1205. [Google Scholar] [PubMed]
- Baldovin, T.; Amoruso, I.; Caldara, F.; Buja, A.; Baldo, V.; Cocchio, S.; Bertoncello, C. Microbiological Hygiene Quality of Thermal Muds: A Pilot Study in Pelotherapy Facilities of the Euganean Thermal District (NE Italy). Int. J. Environ. Res. Public. Health 2020, 17, 5040. [Google Scholar] [CrossRef] [PubMed]
- Bravakos, P.; Kotoulas, G.; Skaraki, K.; Pantazidou, A.; Economou-Amilli, A. A polyphasic taxonomic approach in isolated strains of Cyanobacteria from thermal springs of Greece. Mol. Phylogenet. Evol. 2016, 98, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Palinska, K.A.; Vogt, J.C.; Surosz, W. Biodiversity analysis of the unique geothermal microbial ecosystem of the Blue Lagoon (Iceland) using next-generation sequencing (NGS). Hydrobiologia 2018, 811, 93–102. [Google Scholar] [CrossRef]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. The halotolerance and phylogeny of cyanobacteria with tightly coiled trichomes (Spirulina Turpin) and the description of Halospirulina tapeticola gen. nov., sp. nov. Int. J. Syst. Evol. Microbiol. 2000, 50, 1265–1277. [Google Scholar]
- Ferris, M.J.; Ruff-Roberts, A.L.; Kopczynski, E.D.; Bateson, M.M.; Ward, D.M. Enrichment culture and microscopy conceal diverse thermophilic Synechococcus populations in a single hot spring microbial mat habitat. Appl. Environ. Microbiol. 1996, 62, 1045–1050. [Google Scholar] [CrossRef]
- Buch, B.; Martins, M.D.; Branco, L.H.Z. A widespread cyanobacterium supported by polyphasic approach: Proposition of Koinonema pervagatum gen. & sp. nov. (Oscillatoriales)1. J. Phycol. 2017, 53, 1097–1105. [Google Scholar]
- Komárek, J. A polyphasic approach for the taxonomy of cyanobacteria: Principles and applications. Eur. J. Phycol. 2016, 51, 346–353. [Google Scholar] [CrossRef]


| Taxonomy Based on ANI | Cyanobacteria; Oscillatoriophycideae; Oscillatoriales; Oscillatoriaceae; Oscillatoria |
|---|---|
| Best hit 16S rRNA nrNCBI (similarity %) | Phormidium sp. ElfPHct20 (98.26) |
| Genome size (Mbp) | 6.29 |
| GC content (%) | 59.09 |
| CDSs | 5535 |
| CDSs assigned to FIGfams | 3140 |
| NCBI BioProject | PRJNA622676 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gris, B.; Treu, L.; Zampieri, R.M.; Caldara, F.; Romualdi, C.; Campanaro, S.; La Rocca, N. Microbiota of the Therapeutic Euganean Thermal Muds with a Focus on the Main Cyanobacteria Species. Microorganisms 2020, 8, 1590. https://doi.org/10.3390/microorganisms8101590
Gris B, Treu L, Zampieri RM, Caldara F, Romualdi C, Campanaro S, La Rocca N. Microbiota of the Therapeutic Euganean Thermal Muds with a Focus on the Main Cyanobacteria Species. Microorganisms. 2020; 8(10):1590. https://doi.org/10.3390/microorganisms8101590
Chicago/Turabian StyleGris, Barbara, Laura Treu, Raffaella Margherita Zampieri, Fabrizio Caldara, Chiara Romualdi, Stefano Campanaro, and Nicoletta La Rocca. 2020. "Microbiota of the Therapeutic Euganean Thermal Muds with a Focus on the Main Cyanobacteria Species" Microorganisms 8, no. 10: 1590. https://doi.org/10.3390/microorganisms8101590
APA StyleGris, B., Treu, L., Zampieri, R. M., Caldara, F., Romualdi, C., Campanaro, S., & La Rocca, N. (2020). Microbiota of the Therapeutic Euganean Thermal Muds with a Focus on the Main Cyanobacteria Species. Microorganisms, 8(10), 1590. https://doi.org/10.3390/microorganisms8101590