Impact of Aquaculture Practices on Intestinal Bacterial Profiles of Pacific Whiteleg Shrimp Litopenaeus vannamei
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Harvesting of Shrimp Intestinal Tissue
2.2. Study Site for the Indoor-Raised Shrimp
2.3. Pond-Raised Shrimp
2.4. Wild-Caught Shrimp
2.5. Microbial DNA Isolation and PCR Amplification
2.6. Computational Analysis of PCR Generated 16S rRNA Amplicon Sequences
2.7. Computational Analysis for Alpha and Beta Diversity
2.8. Statistical Analysis
2.9. Accession Numbers for Next Generation Sequencing Data
3. Results
3.1. Comparative Analysis by Taxonomic Composition
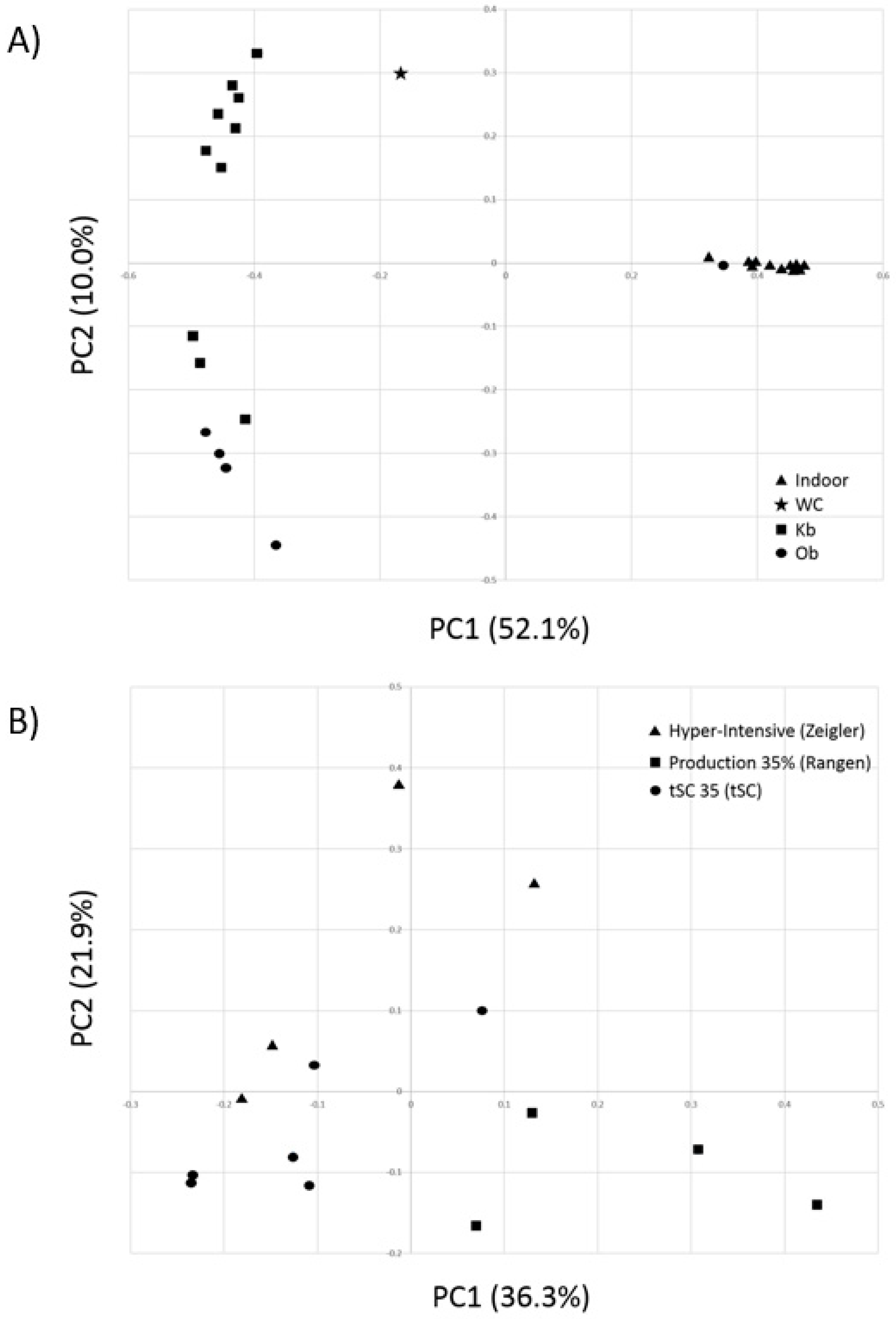
3.2. Comparative Alpha and Beta Diversity Analyses
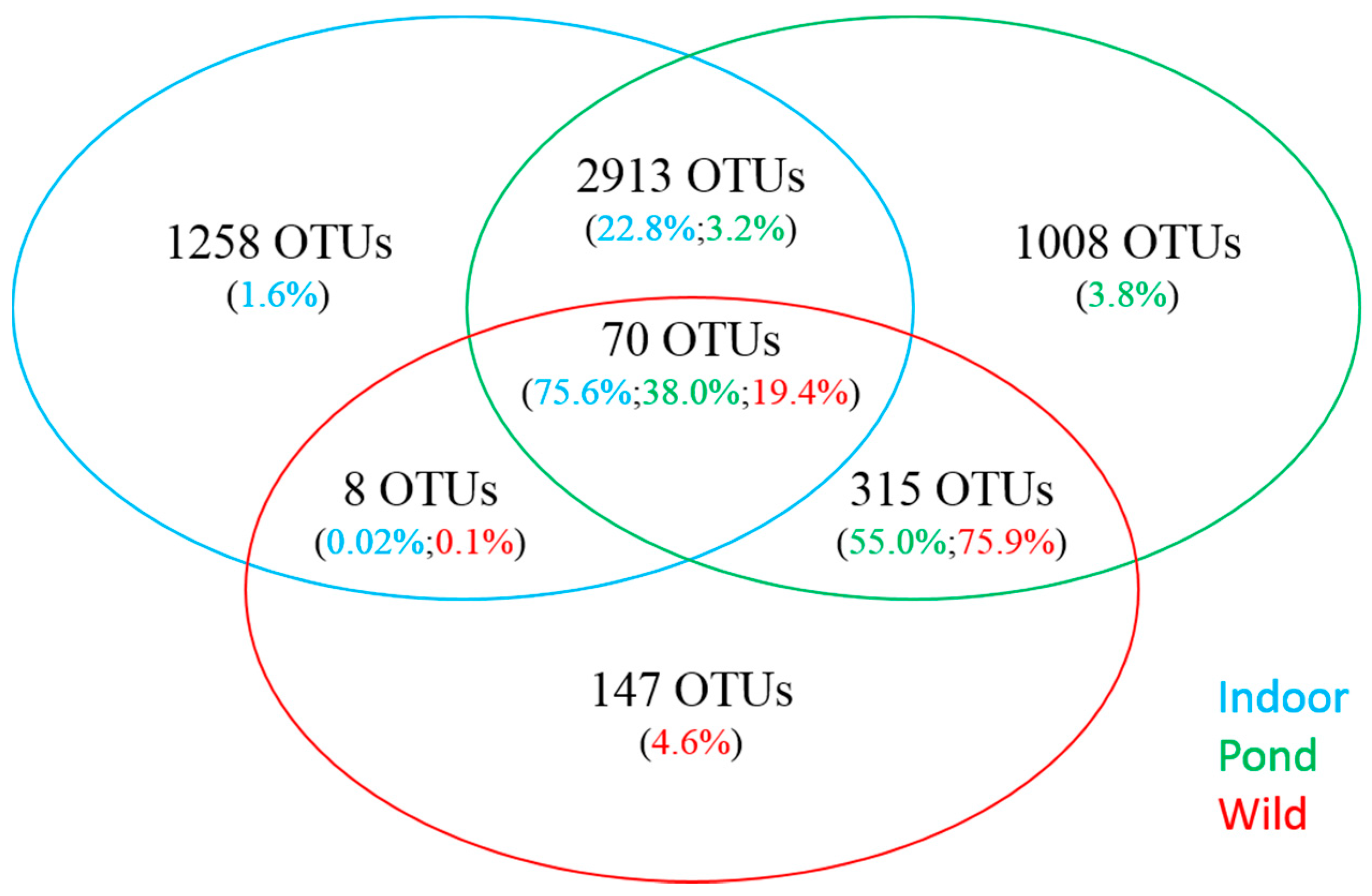
3.3. Comparative Analysis of Prominent OTUs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018; p. 227. [Google Scholar]
- Tidwell, J.H. Aquaculture Production Systems, 1st ed.; Wiley-Blackwell: AMes, IA, USA, 2012; p. 440. [Google Scholar]
- Gillett, R. Global Study of Shrimp Fisheries. FAO Fisheries; Food & Agriculture Organization of the United Nations (FAO): Rome, Italy, 2008. [Google Scholar]
- Gaille, B. 21 Shrimp Industry Statistics and Trends. 2018. Available online: Brandongaille.com (accessed on 7 November 2018).
- Ellis, A. Evaluating the Impacts of Production Process and Product Origin on Willingness to Pay for Shrimp. Master’s Thesis, Clemson University, Clemson, SC, USA, 2011; p. 148. [Google Scholar]
- Greenberg, P. The Not-So-Simple Life of Shrimp. Prevention 24 July 2012. Available online: https://www.prevention.com/food-nutrition/healthy-eating/a20459712/how-to-choose-sustainable-shrimp/ (accessed on 7 November 2018).
- Li, E.; Wang, X.; Chen, K.; Xu, C.; Qin, J.G.; Chen, L. Physiological change and nutritional requirement of Pacific white shrimp Litopenaeus vannamei at low salinity. Rev. Aquac. 2017, 9, 57–75. [Google Scholar] [CrossRef]
- Cheng, K.M.; Hu, C.Q.; Liu, Y.N.; Zheng, S.X.; Qi, X.J. Effects of dietary calcium, phosphorus and calcium/phosphorus ratio on the growth and tissue mineralization of Litopenaeus vannamei reared in low-salinity water. Aquaculture 2006, 251, 472–483. [Google Scholar] [CrossRef]
- Dubay, K.; Tokuoka, S.; Gereffi, G. A Value Chain Analysis of the Sinaloa, Mexico Shrimp Fishery; Fund, E.D., Ed.; Center on Globalization, Governance & Competitiveness, Duke University: Durham, NC, USA, 2010. [Google Scholar]
- Alday-Sanz, V. The Shrimp Book; Nottingham University Press: Nottingham, UK, 2010; p. 200. [Google Scholar]
- Samocha, T.M.; Lawrence, A.L.; Collins, C.A.; Castille, F.L.; Bray, W.A.; Davies, C.J.; Lee, P.G.; Wood, G.F. Production of the Pacific White Shrimp, Litopenaeus vannamei, in High-Density Greenhouse-Enclosed Raceways Using Low Salinity Groundwater. J. Appl. Aquac. 2004, 15, 1–19. [Google Scholar] [CrossRef]
- Li, E.; Xu, C.; Wang, X.; Wang, S.; Zhao, Q.; Zhang, M.; Qin, J.G.; Chen, L. Gut Microbiota and its Modulation for Healthy Farming of Pacific White Shrimp Litopenaeus vannamei. Rev. Fish. Sci. Aquac. 2018, 26, 381–399. [Google Scholar] [CrossRef]
- Leung, P. Shrimp Culture Economics, Market & Trade; Wiley-Blackwell: Ames, IA, USA, 2006; p. 335. [Google Scholar]
- Huang, Z.; Li, X.; Wang, L.; Shao, Z. Changes in the intestinal bacterial community during the growth of white shrimp, Litopenaeus vannamei. Aquac. Res. 2016, 47, 1737–1746. [Google Scholar] [CrossRef]
- Cornejo-Granados, F.; Lopez-Zavala, A.A.; Gallardo-Becerra, L.; Mendoza-Vargas, A.; Sánchez, F.; Vichido, R.; Brieba, L.G.; Viana, M.T.; Sotelo-Mundo, R.R.; Ochoa-Leyva, A. Microbiome of Pacific Whiteleg shrimp reveals differential bacterial community composition between Wild, Aquacultured and AHPND/EMS outbreak conditions. Sci. Rep. 2017, 7, 11783. [Google Scholar] [CrossRef] [PubMed]
- Califano, G.; Castanho, S.; Soares, F.; Ribeiro, L.; Cox, C.J.; Mata, L.; Costa, R. Molecular Taxonomic Profiling of Bacterial Communities in a Gilthead Seabream (Sparus aurata) Hatchery. Front. Microbiol. 2017, 8, 204. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Zhu, J.; Dai, W.; Dong, C.; Qiu, Q.; Li, C. Integrating gut microbiota immaturity and disease-discriminatory taxa to diagnose the initiation and severity of shrimp disease. Env. Microbiol. 2017, 19, 1490–1501. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Morrison, M. Improved extraction of PCR-quality community DNA from digesta and fecal samples. Biotechniques 2004, 36, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Edwards, U.; Rogall, T.; Blöcker, H.; Emde, M.; Böttger, E.C. Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res. 1989, 17, 7843–7853. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.J.; Pace, B.; Olsen, G.J.; Stahl, D.A.; Sogin, M.L.; Pace, N.R. Rapid determination of 16S ribosomal RNA sequences for phylogenetic analyses. Proc. Natl. Acad. Sci. USA 1985, 82, 6955–6959. [Google Scholar] [CrossRef] [PubMed]
- Opdahl, L.J.; Gonda, M.G.; St-Pierre, B. Identification of Uncultured Bacterial Species from Firmicutes, Bacteroidetes and Candidatus Saccharibacteria as Candidate Cellulose Utilizers from the Rumen of Beef Cows. Microorganisms 2018, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Parte, A.C. LPSN—List of Prokaryotic names with Standing in Nomenclature (bacterio.net), 20 years on. Int. J. Syst. Evol. Microbiol. 2018, 68, 1825–1829. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Xuan, Y.; Zhang, H.; Jiang, M.; Pan, Y.; Zhang, Y.; Gong, Y.; Lu, X.; Yu, D.; Xue, R.; et al. Bacterial diversity in the Penaeus vannamei Boone intestine and aquaculture environment. J. Fish. Sci. China 2016, 32, 594–605. [Google Scholar]
- Qiao, F.; Liu, Y.K.; Sun, Y.H.; Wang, X.D.; Chen, K.; Li, T.Y.; Li, E.C.; Zhang, M.L. Influence of different dietary carbohydrate sources on the growth and intestinal microbiota of Litopenaeus vannamei at low salinity. Aquac. Nutr. 2017, 23, 444–452. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Chen, J.C.; Tseng, K.C.; Lin, Y.C.; Huang, C.L. Activation of immunity, immune response, antioxidant ability, and resistance against Vibrio alginolyticus in white shrimp Litopenaeus vannamei decrease under long-term culture at low pH. Fish Shellfish Immunol. 2015, 46, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Santhiya, A.; Sivasankar, P. Occurrence of Vibrio species in shrimp farming systems in the costal farms of Tamil Nadu, India. J. Aquac. Trop. 2015, 30, 63–70. [Google Scholar]

| Tank | Diet | Time Point (day) | Sample | Average Weight (g) | Average Length (cm) |
|---|---|---|---|---|---|
| ST1 | Rangen | 34 | I-R-34 a | 1.304 | 5.2 |
| ST2 | Zeigler | 34 | I-Z-34 a | 1.453 | 5.3 |
| ST3 | tSC | 34 | I-t-34 a | 1.248 | 5.0 |
| STA | Rangen | 34 | I-R-34 b | 1.872 | 5.8 |
| STB | Zeigler | 34 | I-Z-34 b | 1.278 | 5.1 |
| STC | tSC | 34 | I-t-34 b | 1.258 | 5.1 |
| ST1 | Rangen | 44 | I-R-44 a | 2.357 | 6.3 |
| ST2 | Zeigler | 44 | I-Z-44 a | 2.381 | 6.3 |
| ST3 | tSC | 44 | I-t-44 a | 2.693 | 6.6 |
| STA | Rangen | 44 | I-R-44 b | 2.387 | 6.3 |
| STB | Zeigler | 44 | I-Z-44 b | 2.428 | 6.4 |
| STC | tSC | 44 | I-t-44 b | 2.039 | 6.0 |
| ST3 | tSC | 70 | I-t-70 a | 7.256 | 8.8 |
| STC | tSC | 70 | I-t-70 b | 8.868 | 9.2 |
| Diet Name | Protein b | Fat | Fiber | Ash | Manufacturer |
|---|---|---|---|---|---|
| Hyper-Intensive 35 | 35 (M,P) | 7 | 2 | 13 | Zeigler |
| Production 35% | 35 (M,P) | 8 | 3 | 15 | Rangen |
| tSC 35 | 35 (P) | 9 | 2 | 12 | tSC |
| Bumper Crop | 35 (M) | 8 | 3 | 12 | Vimifos |
| Taxonomic Affiliation | Indoor a | Ponds a | Wild |
|---|---|---|---|
| Proteobacteria # | 88.6 ± 3.8 | 51.8 ± 5.4 | 60.0 |
| Rhodobacteraceae # | 84.4 ± 3.8 | 5.1 ± 5.1 | 2.7 |
| Vibrionaceae # | 0.03 ± 0.01 | 44.8 ± 5.9 | 53.5 |
| Other Proteobacteria # | 4.2 ± 0.9 | 1.8 ± 0.7 | 3.8 |
| Firmicutes # | 0.7 ± 0.1 | 36.0 ± 5.9 | 18.7 |
| Fusobacteria # | 0.0 ± 0.0 | 7.9 ± 2.4 | 3.2 |
| Bacteroidetes | 2.2 ± 2.0 | 1.6 ± 0.5 | 2.2 |
| Candidatus Saccharibacteria | 2.7 ± 2.2 | 0.5 ± 0.05 | 0.0 |
| Cyanobacteria # | 0.001 ± 0.001 | 1.6 ± 0.8 | 7.6 |
| Actinobacteria # | 3.0 ± 1.1 | 0.06 ± 0.05 | 0.2 |
| Other phyla | 1.6 ± 1.2 | 0.3 ± 0.2 | 6.0 |
| Unclassified bacteria | 1.2 ± 0.3 | 0.8 ± 0.2 | 2.3 |
| Index | Indoor | Ponds | P Value | Wild |
|---|---|---|---|---|
| Observed OTUs | 252 ± 27 | 177 ± 30 | 0.0746 | 422 |
| Ace | 1488 ± 327 | 788 ± 258 | 0.1082 | 660 |
| Chao1 | 724 ± 119 | 462 ± 129 | 0.1441 | 609 |
| Shannon | 2.21 ± 0.12 | 2.13 ± 0.13 | 0.6403 | 3.00 |
| Simpson | 0.28 ± 0.02 | 0.28 ± 0.03 | 0.9682 | 0.23 |
| Coverage (%) | 96.7 ± 0.4 | 97.9 ± 0.5 | 0.0685 | 96.1 |
| OTUs | Indoor a | Ponds a | Wild | Closest Valid Taxon (id%) |
|---|---|---|---|---|
| Proteobacteria | ||||
| SD_Shr-00001 # | 37.9 ± 4.9 | 1.5 ± 1.5 | 0.07 | Phaeobacter piscinae (98.5%) |
| SD_Shr-00002 # | 23.8 ± 3.1 | 2.3 ± 2.2 | 0.2 | Thalassobacter stenotrophicus (98.5%) |
| SD_Shr-00004 # | 0.02 ± 0.007 | 26.8 ± 4.6 | 1.8 | Vibrio alginolyticus (99.1%) |
| SD_Shr-00005 # | 0.0 ± 0.0 | 6.4 ± 2.3 | 46.4 | Photobacterium damselae (99.1%) |
| SD_Shr-00006 # | 7.8 ± 2.1 | 0.2 ± 0.1 | 0.4 | Ruegeria profundi (99.4%) |
| SD_Shr-00009 # | 2.7 ± 0.8 | 0.1 ± 0.1 | 0.09 | Roseovarius pacificus (99.6%) |
| Firmicutes | ||||
| SD_Shr-00003 # | 0.0 ± 0.0 | 28.2 ± 6.2 | 0.5 | Oceanobacillus iheyensis (80.9%) |
| SD_Shr-00008 # | 0.0 ± 0.0 | 3.5 ± 1.1 | 0.09 | Oceanobacillus iheyensis (80.6%) |
| SD_Shr-00046 # | 0.003 ± 0.001 | 0.02 ± 0.004 | 12.2 | Romboutsia lituseburensis (98.2%) |
| Fusobacteria | ||||
| SD_Shr-00007 # | 0.0 ± 0.0 | 4.4 ± 1.7 | 0.1 | Propionigenium maris (96.4%) |
| SD_Shr-00015 # | 0.0 ± 0.0 | 1.7 ± 0.6 | 3.0 | Propionigenium modestum (91.3%) |
| Cyanobacteria | ||||
| SD_Shr-00021 | 0.0 ± 0.0 | 0.8 ± 0.5 | 2.5 | Gloeobacter kilaueensis (86.2%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Landsman, A.; St-Pierre, B.; Rosales-Leija, M.; Brown, M.; Gibbons, W. Impact of Aquaculture Practices on Intestinal Bacterial Profiles of Pacific Whiteleg Shrimp Litopenaeus vannamei. Microorganisms 2019, 7, 93. https://doi.org/10.3390/microorganisms7040093
Landsman A, St-Pierre B, Rosales-Leija M, Brown M, Gibbons W. Impact of Aquaculture Practices on Intestinal Bacterial Profiles of Pacific Whiteleg Shrimp Litopenaeus vannamei. Microorganisms. 2019; 7(4):93. https://doi.org/10.3390/microorganisms7040093
Chicago/Turabian StyleLandsman, Angela, Benoit St-Pierre, Misael Rosales-Leija, Michael Brown, and William Gibbons. 2019. "Impact of Aquaculture Practices on Intestinal Bacterial Profiles of Pacific Whiteleg Shrimp Litopenaeus vannamei" Microorganisms 7, no. 4: 93. https://doi.org/10.3390/microorganisms7040093
APA StyleLandsman, A., St-Pierre, B., Rosales-Leija, M., Brown, M., & Gibbons, W. (2019). Impact of Aquaculture Practices on Intestinal Bacterial Profiles of Pacific Whiteleg Shrimp Litopenaeus vannamei. Microorganisms, 7(4), 93. https://doi.org/10.3390/microorganisms7040093






