Characterization of Antimicrobial Resistance in Serratia spp. and Citrobacter spp. Isolates from Companion Animals in Japan: Nosocomial Dissemination of Extended-Spectrum Cephalosporin-Resistant Citrobacter freundii
Abstract
:1. Introduction
2. Materials and Methods
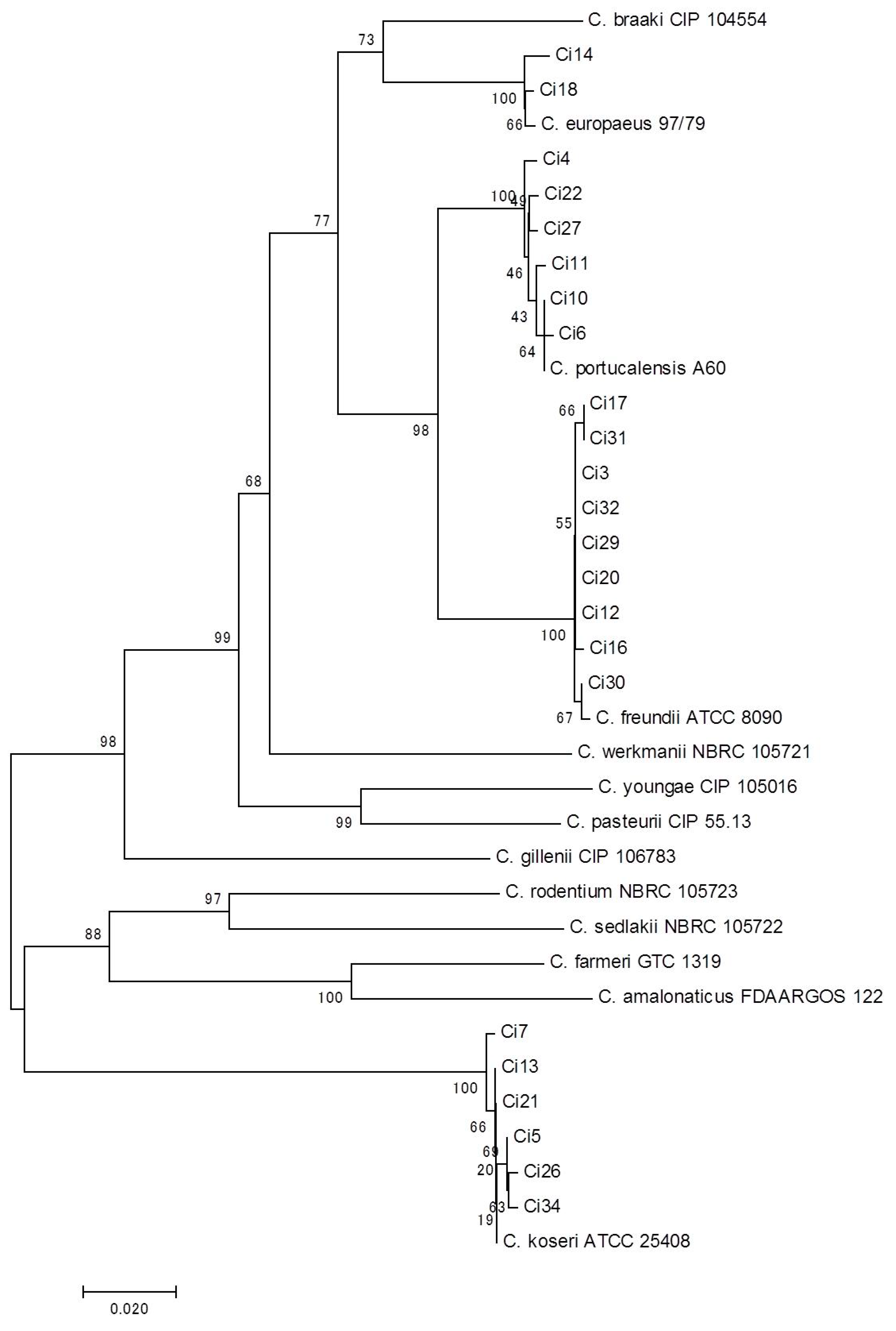
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Choi, S.H.; Lee, J.E.; Park, S.J.; Kim, M.N.; Choo, E.J.; Kwak, Y.G.; Jeong, J.Y.; Woo, J.H.; Kim, N.J.; Kim, Y.S. Prevalence, microbiology, and clinical characteristics of extended-spectrum β-lactamase-producing Enterobacter spp., Serratia marcescens, Citrobacter freundii, and Morganella morganii in Korea. Eur. J. Clin. Microbiol. Infect. Dis. 2007, 26, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, H.; Yano, H.; Hirakata, Y.; Endo, S.; Arai, K.; Ogawa, M.; Shimojima, M.; Aiyagi, T.; Hatta, M.; Yamada, M.; et al. High prevalence of extended-spectrum β-lactamases and qnr determinants in Citrobacter species from Japan: dissemination of CTX-M-2. J. Antimicrob. Chemother. 2011, 66, 2255–2262. [Google Scholar] [CrossRef] [PubMed]
- Wiebe, V.J. Drug Therapy for Infectious Diseases of the Dog and Cat; Wiley Blackwell: Ames, IA, USA, 2015; pp. 49–108. [Google Scholar]
- Brenner, D.J.; O’Hara, C.M.; Grimont, P.A.D.; Janda, J.M.; Falsen, E.; Aldova, E.; Ageron, E.; Schindler, J.; Abbott, S.L.; Steigerwalt, A.G. Biochemical identification of Citrobacter species defined by DNA hybridization and description of Citrobacter gillenii sp. nov. (formerly Citrobacter Genomospecies 10) and Citrobacter murliniae sp. nov. (Formerly Citrobacter Genomospecies 11). J. Clin. Microbiol. 1999, 37, 2619–2624. [Google Scholar] [PubMed]
- Clermont, D.; Motreff, L.; Passet, V.; Fernandez, J.C.; Bizet, C.; Brisse, S. Multilocus sequence analysis of the genus Citrobacter and description of Citrobacter pasteurii sp. nov. Int. J. Syst. Evol. Microbiol. 2015, 65, 1486–1490. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, T.G.; Gonçalves, B.R.; da Silva, M.S.; Novais, Â.; Machado, E.; Carriço, J.A.; Peixe, L. Citrobacter portucalensis sp. nov., isolated from an aquatic sample. Int. J. Syst. Evol. Microbiol. 2017, 67, 3513–3517. [Google Scholar] [CrossRef] [PubMed]
- Samonis, G.; Karageorgopoulos, D.E.; Kofteridis, D.P.; Matthaiou, D.K.; Sidiropoulou, V.; Maraki, S.; Falagas, M.E. Citrobacter infections in a general hospital: characteristics and outcomes. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Samonis, G.; Vouloumanou, E.K.; Christofaki, M.; Dimopoulou, D.; Maraki, S.; Triantafyllow, E.; Kofteridis, D.P.; Falagas, M.E. Serratia infections in a general hospital: characteristics and outcomes. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 653–660. [Google Scholar] [CrossRef] [PubMed]
- MacDougall, C. Beyond susceptible and resistant, Part I: Treatment of infections due to gram-negative organisms with inducible β-lactamases. J. Pediatr. Phrmacol. Ther. 2011, 16, 23–30. [Google Scholar]
- Deshpande, L.M.; Jones, R.N.; Fritsche, T.R.; Sader, H.S. Occurrence and characterization of carbapenemase-producing Enterobacteriaceae: report from the SENTRY antimicrobial surveillance program (2000–2004). Microb. Drug Resist. 2006, 12, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, T.G.; Novais, Å.; Branquinho, R.; Machado, E.; Peixe, L. Phylogeny and comparative genomics unveil independent diversification trajectories of qnrB and genetic platforms within particular Citrobacter species. Antimicrob. Agents Chemother. 2015, 59, 5951–5958. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular Evolutionary Genetics Analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Boil. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Kimura, M. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals. Approved Standard-Fourth Edition; CLSI Document VET01-A4; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2013. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twntieth Informational Supplement; CLSI document M100-S20; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2010. [Google Scholar]
- EUCAST. EUCAST Guidelines for Detection of Resistance Mechanisms and Specific Resistances of Clinical and/or Epidemiological Importance. Available online: http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Resistance_mechanisms/EUCAST_detection_of_resistance_mechanisms_v1.0_20131211.pdf. (accessed on 27 September 2017).
- Izdebski, R.; Baraniak, A.; Herda, M.; Fiett, J.; Bonten, M.J.; Carmeli, Y.; Goossens, H.; Hryniewicz, W.; Brun-Buisson, C.; Gniadkowski, M. MOSAR WP2, WP3 and WP5 Study Groups. MLST reveals potentially high-risk international clones of Enterobacter cloacae. J. Antimicrob. Chemother. 2015, 70, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Kojima, A.; Ishii, Y.; Ishihara, K.; Esaki, H.; Asai, T.; Oda, C.; Tamura, Y.; Takahashi, T.; Yamaguchi, K. Extended-spectrum-β-lactamase-producing Escherichia coli strains isolated from farm animals from 1999 to 2002: report from the Japanese Veterinary Antimicrobial Resistance Monitoring Program. Antimicrob. Agents Chemother. 2005, 49, 3533–3537. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Pérez, F.J.; Hanson, N.D. Detection of plasmid-mediated AmpC β-lactamase genes in clinical isolates by using multiplex-PCR. J. Clin. Microbiol. 2002, 40, 2153–2162. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.J.; Ko, W.C.; Jung, Y.C.; Chuang, C.L.; Wu, J.J. Emergence of Klebsiella pneumoniae isolates producing inducible DHA-1 β-lactamase in a university hospital in Taiwan. J. Clin. Microbiol. 2002, 40, 3121–3126. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ensor, V.; Gossain, S.; Nye, K.; Hawkey, P. Rapid and simple detection of blaCTX-M genes by multiplex PCR assay. J. Med. Microbiol. 2007, 54, 1183–1187. [Google Scholar] [CrossRef] [PubMed]
- Usui, M.; Hiki, M.; Murakami, K.; Ozawa, M.; Nagai, H.; Asai, T. Evaluation of transferability of R-plasmid in bacteriocin-producing donors to bacteriocin-resistant recipients. Jpn. J. Infect. Dis. 2012, 65, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, H.; Matsuoka, Y.; Nakagawa, E.; Murase, T. Characteristics of Escherichia coli isolated from broiler chickens with colibacillosis in commercial farms from a common hatchery. Poult. Sci. 2017, 96, 3717–3724. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Lan, R.; Liu, L.; Wang, Y.; Zhang, Y.; Wang, Y.; Xu, J. Antimicrobial resistance and cytotoxicity of Citrobacter spp. in Maanshan Anhui Province, China. Front. Microbiol. 2017, 8, 1357. [Google Scholar] [CrossRef] [PubMed]
- Giani, T.; Sennati, S.; Antonelli, A.; Di Pilato, V.; di Maggio, T.; Mantella, A.; Niccolai, C.; Spinicci, M.; Monasterio, J.; Castellanos, P.; et al. High prevalence of carriage of mcr-1-positive enteric bacteria among healthy children from rural communities in the Chaco region, Bolivia, September to October 2016. Eur. Surveill. 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Ding, H.; Shi, Y.; Zhao, Y.; Hu, X.; Ren, J.; Huang, G.; Wu, R.; Zhao, Z. Further spread of a blaKPC-harboring untypeable plasmid in Enterobacteriaceae in China. Front. Microbiol. 2018, 9, 1938. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Shimizu, T.; Mukai, Y.; Kuwajima, K.; Sato, T.; Kajino, A.; Usui, M.; Tamura, Y.; Kimura, Y.; Miyamoto, T.; et al. Phenotypic and molecular characterization of antimicrobial resistance in Enterobacter spp. isolates from companion animals in Japan. PLoS ONE 2017, 12, e0174178. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Arima, S.; Niina, A.; Kataoka, Y.; Takahashi, T. Characterization of Pseudomonas aeruginosa isolates from dogs and cats in Japan: current status of antimicrobial resistance and prevailing resistance mechanisms. Microbiol. Immunol. 2012, 56, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Harada, K.; Shimizu, T.; Sato, T.; Kajino, A.; Usui, M.; Tamura, Y.; Tsuyuki, Y.; Miyamoto, T.; Ohki, A.; et al. Species distribution, virulence factors, and antimicrobial resistance of Acinetobacter spp. isolates from dogs and cats: a preliminary study. Microbiol. Immunol. 2018, 62, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Hrabák, J.; Chudáčková, E.; Papagiannitsis, C.C. Detection of carbapenemases in Enterobacteriaceae: a challenge for diagnostic microbiological laboratories. Clin. Microbiol. Infect. 2014, 20, 839–853. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Harada, K.; Tsuyuki, Y.; Kimura, Y.; Miyamoto, T.; Hatoya, S.; Hikasa, Y. In vitro efficacy of 16 antimicrobial drugs against a large collection of β-lactamase-producing isolates of extraintestinal pathogenic Escherichia coli from dogs and cats. J. Med. Microbiol. 2017, 66, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Shimizu, T.; Mukai, Y.; Kuwajima, K.; Sato, T.; Usui, M.; Tamura, Y.; Kimura, Y.; Miyamoto, T.; Tsuyuki, Y.; et al. Phenotypic and molecular characterization of antimicrobial resistance in Klebsiella spp. isolates from companion animals in Japan: clonal dissemination of multidrug-resistant extended-spectrum β-lactamase-producing Klebsiella pneumoniae. Front. Microbiol. 2016, 7, 1021. [Google Scholar] [CrossRef] [PubMed]
- Ewers, C.; Stamm, I.; Pfeifer, Y.; Wieler, L.H.; Kopp, P.A.; Schønning, K.; Prenger-Berninghoff, E.; Scheufen, S.; Stolle, I.; Günther, S.; et al. Clonal spread of highly successful ST15-CTX-M-15 Klebsiella pneumoniae in companion animals and horses. J. Antimicrob. Chemother. 2014, 69, 2676–2680. [Google Scholar] [CrossRef] [PubMed]
- Tamang, M.D.; Nam, H.M.; Jang, G.C.; Kim, S.R.; Chae, M.H.; Jung, S.C.; Byun, J.W.; Park, Y.H.; Lim, S.K. Molecular characterization of extended-spectrum-β-lactamase-producing and plasmid-mediated AmpC β-Lactamase-producing Escherichia coli isolated from stray dogs in South Korea. Antimicrob. Agents Chemother. 2012, 56, 2705–2712. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zeng, Z.; Chen, S.; Ma, J.; He, L.; Liu, Y.; Deng, Y.; Lei, T.; Zhao, J.; Liu, J.H. High prevalence of blaCTX-M extended-spectrum β-lactamase genes in Escherichia coli isolates from pets and emergence of CTX-M-64 in China. Clin. Microbiol. Infect. 2010, 16, 1475–1481. [Google Scholar] [CrossRef] [PubMed]
- Ewers, C.; Bethe, A.; Wieler, L.H.; Guenther, S.; Stamm, I.; Kopp, P.A.; Grobbel, M. Companion animals: a relevant source of extended-spectrum β-lactamase-producing fluoroquinolone-resistant Citrobacter freundii. Int. J. Antimicrob. Agents 2011, 37, 86–87. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Nakai, Y.; Kataoka, Y. Mechanisms of resistance to cephalosporin and emergence of O25b-ST131 clone harboring CTX-M-27 β-lactamase in extraintestinal pathogenic Escherichia coli from dogs and cats in Japan. Microbiol. Immunol. 2012, 56, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Avison, M.B.; Underwood, S.; Okazaki, A.; Walsh, T.R.; Bennett, P.M. Analysis of AmpC β-lactamase expression and sequence in biochemically atypical ceftazidime-resistant Enterobacteriaceae from paediatric patients. J. Antimicrob. Chemother. 2004, 53, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. AmpC β-lactamases. Clin. Microbiol. Rev. 2009, 22, 161–182. [Google Scholar] [CrossRef] [PubMed]
- Hammerum, A.M.; Hansen, F.; Nielsen, H.L.; Jakobsen, L.; Stegger, M.; Andersen, P.S.; Jensen, P.; Nielsen, T.K.; Hansen, L.H.; Hasman, H.; et al. Use of WGS data for investigation of a long-term NDM-1-producing Citrobacter freundii outbreak and secondary in vivo spread of blaNDM-1 to Escherichia coli, Klebsiella pneumoniae and Klebsiella oxytoca. J. Antimicrob. Chemother. 2016, 71, 3117–3124. [Google Scholar] [CrossRef] [PubMed]
- Villa, J.; Arana, D.M.; Viedma, E.; Perez-Montarelo, D.; Chaves, F. Characterization of mobile genetic elements carrying VIM-1 and KPC-2 carbapenemases in Citrobacter freundii isolates in Madrid. Int. J. Med. Microbiol. 2017, 307, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Kukla, R.; Chudejova, K.; Papagiannitsis, C.C.; Medvecky, M.; Habalova, K.; Hobzova, L.; Bolehovska, R.; Pliskova, L.; Hrabak, J.; Zemlickova, H. Characterization of KPC-encoding plasmids from Enterobacteriaceae isolated in a Czech Hospital. Antimicrob Agents Chemother. 2018, 62. [Google Scholar] [CrossRef] [PubMed]

| Agents | Genera (No. of Isolates) | MIC (µg/mL) | No. of Resistance (%) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ≤0.031 | 0.063 | 0.125 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | 16 | 32 | 64 | 128 | 256 | >256 | |||
| CPL | Serratia (30) | 30 | 30 (100) * | ||||||||||||||
| Citrobacter (23) | 2 | 2 | 1 | 1 | 4 | 2 | 1 | 10 | 18 (78.3) | ||||||||
| CMZ | Serratia (30) | 2 | 9 | 10 | 5 | 4 | 4 (13.3) | ||||||||||
| Citrobacter (23) | 1 | 3 | 1 | 1 | 5 | 5 | 6 | 1 | 7 (30.4) | ||||||||
| CTX | Serratia (30) | 2 | 14 | 13 | 1 | 0 (0.0) | |||||||||||
| Citrobacter (23) | 3 | 6 | 6 | 1 | 1 | 1 | 2 | 1 | 2 | 8 (34.8) * | |||||||
| MPM | Serratia (30) | 4 | 25 | 1 | 0 (0.0) | ||||||||||||
| Citrobacter (23) | 19 | 3 | 1 | 0 (0.0) | |||||||||||||
| TET | Serratia (30) | 1 | 1 | 2 | 7 | 14 | 5 | 26 (86.7) * | |||||||||
| Citrobacter (23) | 1 | 15 | 1 | 2 | 1 | 1 | 2 | 4 (17.4) | |||||||||
| AMK | Serratia (30) | 4 | 3 | 20 | 3 | 0 (0.0) | |||||||||||
| Citrobacter (23) | 2 | 5 | 13 | 2 | 1 | 0 (0.0) | |||||||||||
| CHL | Serratia (30) | 2 | 14 | 10 | 2 | 1 | 1 | 4 (13.3) | |||||||||
| Citrobacter (23) | 17 | 4 | 1 | 1 | 2 (8.7) | ||||||||||||
| CIP | Serratia (30) | 4 | 4 | 16 | 4 | 1 | 1 | 0 (0.0) | |||||||||
| Citrobacter (23) | 6 | 2 | 2 | 3 | 1 | 1 | 2 | 2 | 3 | 1 | 6 (26.1) * | ||||||
| 0.125/0.063 | 0.25/0.125 | 0.5/0.25 | 1/0.5 | 2/1 | 4/2 | 8/4 | 8/16 | 32/16 | 64/32 | 128/64 | 256/128 | >256/128 | |||||
| ACV | Serratia (30) | 1 | 3 | 1 | 2 | 15 | 8 | 25 (83.3) | |||||||||
| Citrobacter (23) | 3 | 2 | 4 | 5 | 9 | 14 (60.9) | |||||||||||
| ≤0.03/0.59 | 0.06/1.19 | 0.13/2.38 | 0.25/4.75 | 0.5/9.5 | 1/19 | 2/38 | 4/76 | 8/152 | 16/304 | 32/608 | 64/1216 | >64/1216 | |||||
| TMS | Serratia (30) | 4 | 5 | 18 | 3 | 0 (0.0) | |||||||||||
| Citrobacter (23) | 9 | 5 | 1 | 2 | 1 | 1 | 1 | 3 | 4 (17.4) * | ||||||||
| Strain | Year | Host | Origin | ST | AmpC Overexpression | ESBLs | qAmpCs | Other β-lactamases | MIC (μg/mL)b | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ACV | CPL | CMZ | CTX | MPM | TET | CHL | AMK | CIP | TMS | |||||||||
| C. freundii (n = 5) | ||||||||||||||||||
| Ci17 | 2016 | Cat | Urine | 18 | + | CMY-117 | TEM-1 | 64/32 | >256 | 64 | 32 | 0.063 | 1 | 16 | 2 | 8 | 8/152 | |
| Ci20 | 2016 | Cat | Urine | 156 * | - | CTX-M-3 | CMY-78-like | TEM-1 | 16/8 | >256 | 16 | 256 | 0.031 | 1 | 8 | 4 | 2 | 0.06/1.19 |
| Ci29 | 2016 | Cat | Urine | 156 * | - | CTX-M-3 | CMY-78-like | TEM-1 | 16/8 | >256 | 8 | >256 | 0.031 | 4 | 16 | 2 | 4 | 0.25/4.75 |
| Ci31 | 2016 | Cat | Urine | 18 | + | CMY-117 | 64/32 | >256 | 64 | 16 | 0.063 | 128 | 8 | 1 | 8 | >64/1216 | ||
| Ci32 | 2016 | Cat | Urine | 156 * | - | CTX-M-3 | CMY-78-like | TEM-1 | 32/16 | >256 | 16 | >256 | 0.031 | 32 | 16 | 8 | 8 | 2/38 |
| C. portucalensis (n = 2) | ||||||||||||||||||
| Ci10 | 2015 | Dog | Urine | NA | + | DHA-1, CMY-37-like | 64/32 | >256 | 64 | 8 | 0.125 | 128 | 128 | 2 | 16 | >64/1216 | ||
| Ci27 | 2016 | Dog | Nasal | NA | + | CMY-13 | 64/32 | >256 | 128 | 32 | 0.063 | 1 | 8 | 4 | 4 | ≤0.03/0.59 | ||
| C. koseri (n = 1) | ||||||||||||||||||
| Ci7 | 2015 | Dog | Urine | NA | NA | DHA-1 | 64/32 | >256 | 64 | 4 | 0.015 | 1 | 8 | 1 | 0.25 | 0.5/9.5 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harada, K.; Shimizu, T.; Ozaki, H.; Kimura, Y.; Miyamoto, T.; Tsuyuki, Y. Characterization of Antimicrobial Resistance in Serratia spp. and Citrobacter spp. Isolates from Companion Animals in Japan: Nosocomial Dissemination of Extended-Spectrum Cephalosporin-Resistant Citrobacter freundii. Microorganisms 2019, 7, 64. https://doi.org/10.3390/microorganisms7030064
Harada K, Shimizu T, Ozaki H, Kimura Y, Miyamoto T, Tsuyuki Y. Characterization of Antimicrobial Resistance in Serratia spp. and Citrobacter spp. Isolates from Companion Animals in Japan: Nosocomial Dissemination of Extended-Spectrum Cephalosporin-Resistant Citrobacter freundii. Microorganisms. 2019; 7(3):64. https://doi.org/10.3390/microorganisms7030064
Chicago/Turabian StyleHarada, Kazuki, Takae Shimizu, Hiroichi Ozaki, Yui Kimura, Tadashi Miyamoto, and Yuzo Tsuyuki. 2019. "Characterization of Antimicrobial Resistance in Serratia spp. and Citrobacter spp. Isolates from Companion Animals in Japan: Nosocomial Dissemination of Extended-Spectrum Cephalosporin-Resistant Citrobacter freundii" Microorganisms 7, no. 3: 64. https://doi.org/10.3390/microorganisms7030064
APA StyleHarada, K., Shimizu, T., Ozaki, H., Kimura, Y., Miyamoto, T., & Tsuyuki, Y. (2019). Characterization of Antimicrobial Resistance in Serratia spp. and Citrobacter spp. Isolates from Companion Animals in Japan: Nosocomial Dissemination of Extended-Spectrum Cephalosporin-Resistant Citrobacter freundii. Microorganisms, 7(3), 64. https://doi.org/10.3390/microorganisms7030064




