The Effect of Nitrogen Content on Archaeal Diversity in an Arctic Lake Region
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Site Description and Sample Collection
2.2. Geochemical Properties of Soils and Lake Sediments
2.3. DNA Extraction, Polymerase Chain Reaction (PCR) Amplification, and Sequencing
2.3.1. DNA Extraction and PCR Amplification
2.3.2. PCR Product Quantification, Qualification, and Purification
2.3.3. Library Preparation and Sequencing
2.3.4. Processing of Sequencing Data
2.4. Statistical Analyses
3. Results
3.1. Geochemical Properties of Soil and Sediment Samples
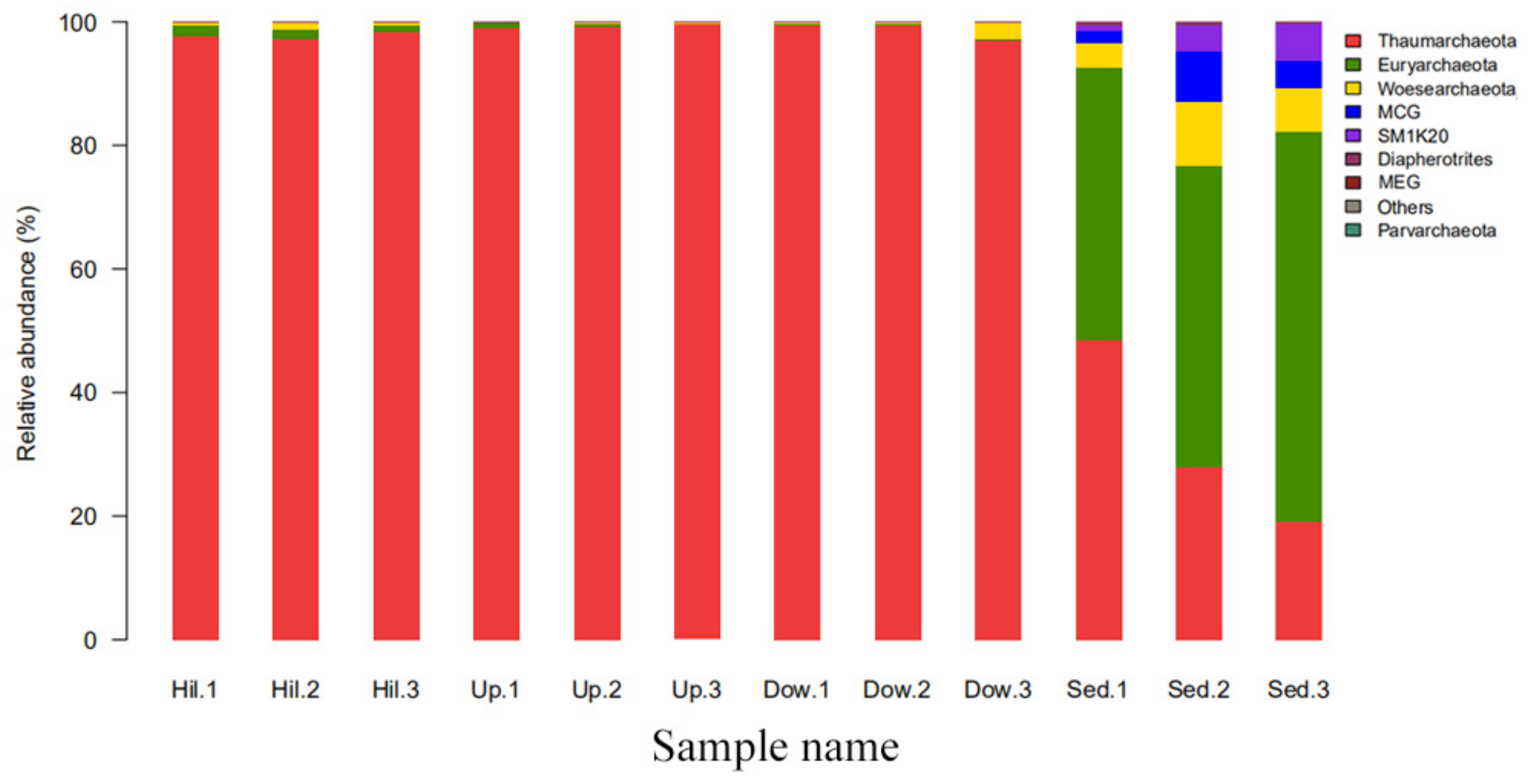
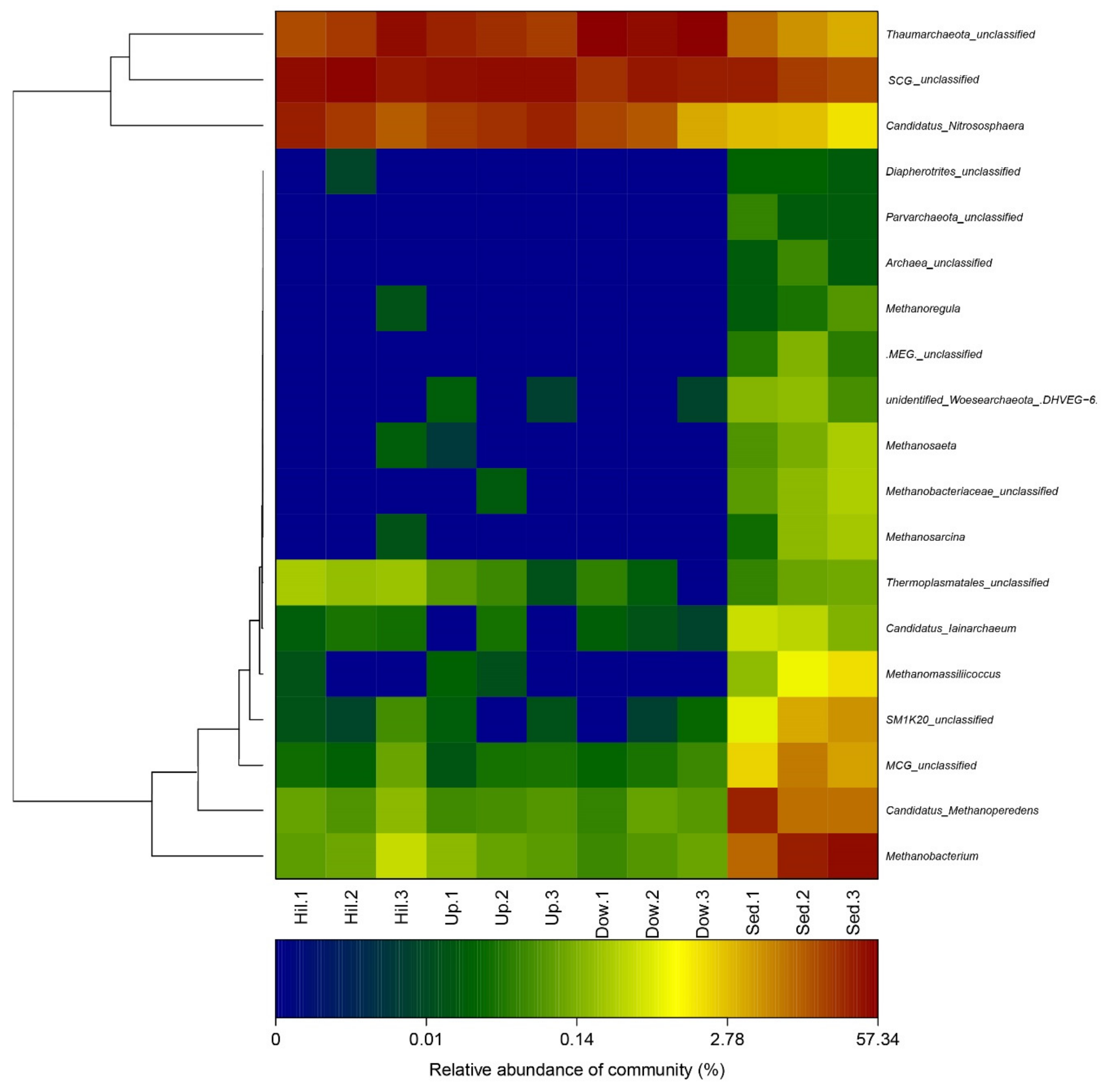
3.2. Archaeal Diversity and Community Composition
3.3. Correlations between Environmental Variables and Archaeal Community Structure
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kirtman, B.; Power, S.; Adedoyin, A.; Boer, G.; Bojariu, R.; Camilloni, I.; Doblas-Reyes, F.; Fiore, A.; Kimoto, M.; Meehl, G. Near-term climate change: Projections and predictability. In Climate Change 2013: The Physical Science Basis: Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Huang, J.; Zhang, X.; Zhang, Q.; Lin, Y.; Hao, M.; Luo, Y.; Zhao, Z.; Yao, Y.; Chen, X.; Wang, L.; et al. Recently amplified arctic warming has contributed to a continual global warming trend. Nat. Clim. Chang. 2017, 7, 875–879. [Google Scholar] [CrossRef]
- Vincent, W.F.; Lemay, M.; Allard, M.; Wolfe, B.B. Adapting to permafrost change: A science framework. Eos Trans. Am. Geophys. Union 2013, 94, 373–375. [Google Scholar] [CrossRef]
- Tripathi, B.M.; Kim, M.; Kim, Y.; Byun, E.; Yang, J.W.; Ahn, J.; Lee, Y.K. Variations in bacterial and archaeal communities along depth profiles of alaskan soil cores. Sci. Rep. 2018, 8, 504. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Jung, J.Y.; Tripathi, B.M.; Gockede, M.; Lee, Y.K.; Kim, M. Dynamics of microbial communities and CO2 and CH4 fluxes in the tundra ecosystems of the changing arctic. J. Microbiol. 2019, 57, 325–336. [Google Scholar] [CrossRef]
- Schuur, E.A.; Mack, M.C. Ecological response to permafrost thaw and consequences for local and global ecosystem services. Annu. Rev. Ecol. Evol. Syst. 2018, 49, 279–301. [Google Scholar] [CrossRef]
- Doney, S.C.; Ruckelshaus, M.; Duffy, J.E.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N. Climate change impacts on marine ecosystems. Ann. Rev. Mar. Sci. 2012, 4, 11–37. [Google Scholar] [CrossRef]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15, 365–377. [Google Scholar] [CrossRef]
- Singh, B.K.; Bardgett, R.D.; Smith, P.; Reay, D.S. Microorganisms and climate change: Terrestrial feedbacks and mitigation options. Nat. Rev. Microbiol. 2010, 8, 779–790. [Google Scholar] [CrossRef]
- Chonggang, X.; Chao, L.; Stan, W.; Cathy, W.; Nathan, M.D. Importance of feedback loops between soil inorganic nitrogen and microbial communities in the heterotrophic soil respiration response to global warming. Nat. Rev. Microbiol. 2011, 9, 222. [Google Scholar]
- Blaud, A.; Lerch, T.Z.; Phoenix, G.K.; Osborn, A.M. Arctic soil microbial diversity in a changing world. Res. Microbiol. 2015, 166, 796–813. [Google Scholar] [CrossRef]
- Høj, L.; Olsen, R.A.; Torsvik, V.L. Effects of temperature on the diversity and community structure of known methanogenic groups and other archaea in high arctic peat. ISME J. 2008, 2, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Schleper, C.; Nicol, G.W. Ammonia-oxidising archaea—Physiology, ecology and evolution. Adv. Microb. Physiol. 2010, 57, 1–41. [Google Scholar] [PubMed]
- Francis, C.A.; Roberts, K.J.; Beman, J.M.; Santoro, A.E.; Oakley, B.B. Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proc. Natl. Acad. Sci. USA 2005, 102, 14683–14688. [Google Scholar] [CrossRef] [PubMed]
- Hongyue, D.; Xiaoxia, Z.; Jin, S.; Tiegang, L.; Zhinan, Z.; Guanpin, Y. Diversity and spatial distribution of sediment ammonia-oxidizing crenarchaeota in response to estuarine and environmental gradients in the changjiang estuary and east china sea. Microbiology 2008, 154, 2084–2095. [Google Scholar]
- Vetriani, C.; Jannasch, H.W.; Macgregor, B.J.; Stahl, D.A.; Reysenbach, A.L. Population structure and phylogenetic characterization of marine benthic archaea in deep-sea sediments. Appl. Environ. Microbiol. 1999, 65, 4375–4384. [Google Scholar]
- Brochier-Armanet, C.; Boussau, B.; Gribaldo, S.; Forterre, P. Mesophilic crenarchaeota: Proposal for a third archaeal phylum, the thaumarchaeota. Nat. Rev. Microbiol. 2008, 6, 245. [Google Scholar] [CrossRef]
- Kessler, P.; Blank, C.; Leigh, J.A. The nif gene operon of the methanogenic archaeon methanococcus maripaludis. J. Bacteriol. 1998, 180, 1504. [Google Scholar]
- Magingo, F.S.S.; Stumm, C.K. Nitrogen fixation by methanobacterium formicicum. FEMS Microbiol. Lett. 1991, 81, 273–277. [Google Scholar] [CrossRef][Green Version]
- Leigh, J.A. Nitrogen fixation in methanogens: The archaeal perspective. Curr. Issues Mol. Biol. 2000, 2, 125–131. [Google Scholar]
- Nordin, A.; Schmidt, I.K.; Shaver, G.R. Nitrogen uptake by arctic soil microbes and plants in relation to soil nitrogen supply. Ecology 2004, 85, 955–962. [Google Scholar] [CrossRef]
- Shaver, G.R.; Chapin, F.S., III. Response to fertilization by various plant growth forms in an alaskan tundra: Nutrient accumulation and growth. Ecology 1980, 61, 662–675. [Google Scholar] [CrossRef]
- Falkowski, P.G. Evolution of the nitrogen cycle and its influence on the biological sequestration of co2 in the ocean. Nature 1997, 387, 272–275. [Google Scholar] [CrossRef]
- Chen, L.; Liu, L.; Mao, C.; Qin, S.; Wang, J.; Liu, F.; Blagodatsky, S.; Yang, G.; Zhang, Q.; Zhang, D. Nitrogen availability regulates topsoil carbon dynamics after permafrost thaw by altering microbial metabolic efficiency. Nat. Commun. 2018, 9, 3951. [Google Scholar] [CrossRef] [PubMed]
- Finger, R.A.; Turetsky, M.R.; Kielland, K.; Ruess, R.W.; Mack, M.C.; Euskirchen, E.S. Effects of permafrost thaw on nitrogen availability and plant-soil interactions in a boreal alaskan lowland. J. Ecol. 2016, 104, 1542–1554. [Google Scholar] [CrossRef]
- Palis, R.G.; Okwach, G.; Rose, C.W.; Saffigna, P.G. Soil erosion processes and nutrient loss. I. The interpretation of enrichment ratio and nitrogen loss in runoff sediment. Aust. J. Soil Res. 1990, 28, 623–639. [Google Scholar] [CrossRef]
- Douglas, C.L.; King, K.A.; Zuzel, J.F. Nitrogen and phosphorus in surface runoff and sediment from a wheat-pea rotation in northeastern oregon. J. Environ. Qual. 1998, 27, 1170–1177. [Google Scholar] [CrossRef]
- Grant, R.F. Ecosystem co2 and ch4 exchange in a mixed tundra and a fen within a hydrologically diverse arctic landscape: 2. Modeled impacts of climate change. J. Geophys. Res. Biogeosci. 2015, 120, 1388–1406. [Google Scholar] [CrossRef]
- Wang, N.F.; Zhang, T.; Yang, X.; Wang, S.; Yu, Y.; Dong, L.L.; Guo, Y.D.; Ma, Y.X.; Zang, J.Y. Diversity and composition of bacterial community in soils and lake sediments from an arctic lake area. Front. Microbiol. 2016, 7, 1170. [Google Scholar] [CrossRef]
- Dombrowski, N.; Lee, J.-H.; Williams, T.A.; Offre, P.; Spang, A. Genomic diversity, lifestyles and evolutionary origins of dpann archaea. FEMS Microbiol. Lett. 2019, 366, fnz008. [Google Scholar] [CrossRef]
- Roland, H.; Lebedeva, E.V.; Eva, S.; Kilian, S.; Andreas, R.; Holger, D.; Michael, W. A moderately thermophilic ammonia-oxidizing crenarchaeote from a hot spring. Proc. Natl. Acad. Sci. USA 2008, 105, 2134–2139. [Google Scholar]
- Li-Mei, Z.; Offre, P.R.; Ji-Zheng, H.; Verhamme, D.T.; Nicol, G.W.; Prosser, J.I. Autotrophic ammonia oxidation by soil thaumarchaea. Proc. Natl. Acad. Sci. USA 2010, 107, 17240–17245. [Google Scholar]
- Miranda, K.M.; Ridnour, L.; Esprey, M.; Citrin, D.; Thomas, D.; Mancardi, D.; Donzelli, S.; Wink, D.A.; Katori, T.; Tocchetti, C.G. Comparison of the chemical biology of NO and HNO: An inorganic perspective. Prog. Inorg. Chem. 2005, 54, 349–384. [Google Scholar]
- Michael, P.; Christa, S.; Michael, W. The thaumarchaeota: An emerging view of their phylogeny and ecophysiology. Curr. Opin. Microbiol. 2011, 14, 300–306. [Google Scholar]
- Jennifer, P.; Dumont, M.G.; Ralf, C. Ammonia oxidation coupled to co2 fixation by archaea and bacteria in an agricultural soil. Proc. Natl. Acad. Sci. USA 2011, 108, 4170–4175. [Google Scholar]
- Lone, H.; Marte, R.; Lars Egil, H.; Olsen, R.A.; Torsvik, V.L. Effects of water regime on archaeal community composition in arctic soils. Environ. Microbiol. 2010, 8, 984–996. [Google Scholar]
- Kayhanian, M. Performance of a high--solids anaerobic digestion process under various ammonia concentrations. J. Chem. Technol. Biotechnol. 2010, 59, 349–352. [Google Scholar] [CrossRef]
- Jin, R.; Huang, G.; Ma, C.; Yang, G. Ammonia inhibition of anaerobic digestion. Ind. Water Treat. 2010, 30, 9–12. [Google Scholar]
- Liu, X.; Li, M.; Castelle, C.J.; Probst, A.J.; Zhou, Z.; Pan, J.; Liu, Y.; Banfield, J.F.; Gu, J.D. Insights into the ecology, evolution, and metabolism of the widespread woesearchaeotal lineages. Microbiome 2018, 6, 102. [Google Scholar] [CrossRef]
- Cui, M.; Ma, A.; Qi, H.; Zhuang, X.; Zhuang, G. Anaerobic oxidation of methane: An “active” microbial process. Microbiologyopen 2015, 4, 1–11. [Google Scholar] [CrossRef]
- Vaksmaa, A.; Guerrero-Cruz, S.; Alen, T.A.V.; Cremers, G.; Ettwig, K.F.; Lüke, C.; Jetten, M.S.M. Enrichment of anaerobic nitrate-dependent methanotrophic ‘candidatus methanoperedens nitroreducens’ archaea from an italian paddy field soil. Appl. Microbiol. Biotechnol. 2017, 101, 7075–7084. [Google Scholar] [CrossRef]
- Conrad, R. The global methane cycle: Recent advances in understanding the microbial processes involved. Environ. Microbiol. Rep. 2010, 1, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Molénat, J.; Durand, P.; Gascuel-Odoux, C.; Davy, P.; Gruau, G. Mechanisms of nitrate transfer from soil to stream in an agricultural watershed of french brittany. Water Air Soil Pollut. 2002, 133, 161–183. [Google Scholar] [CrossRef]
- Sharma, B.M.; Tayal, S.; Chakraborty, P.; Bharat, G.K. Chemical characterization of meltwater from east rathong glacier vis-à-vis Western Himalayan Glaciers. In Dynamics of Climate Change and Water Resources of Northwestern Himalaya; Springer: Cham, Switzerland, 2015. [Google Scholar]
- Cao, L.K.; Chen, G.J.; Lu, Y.T. Nitrogen leaching in vegetable fields in the suburbs of shanghai. Pedosphere 2005, 15, 641–645. [Google Scholar]
- Tranvik, L.J.; Downing, J.A.; Cotner, J.B.; Loiselle, S.A.; Striegl, R.G.; Ballatore, T.J.; Dillon, P.; Finlay, K.; Fortino, K.; Knoll, L.B. Lakes and reservoirs as regulators of carbon cycling and climate. Limnol. Oceanogr. 2009, 54, 2298–2314. [Google Scholar] [CrossRef]
- Muster, S.; Langer, M.; Heim, B.; Westermann, S.; Boike, J. Subpixel heterogeneity of ice-wedge polygonal tundra: A multi-scale analysis of land cover and evapotranspiration in the lena river delta, siberia. Tellus Ser. B Chem. Phys. Meteorol. 2012, 64, 1–14. [Google Scholar] [CrossRef]
- Edmonds, J.W.; Weston, N.B.; Joye, S.B.; Mou, X.; Moran, M.A. Microbial community response to seawater amendment in low-salinity tidal sediments. Microb. Ecol. 2009, 58, 558–568. [Google Scholar] [CrossRef] [PubMed]





| Site | Sample | Water Content | pH | Organic Carbon (OrC) % | NH4+–N (μg/g) | SiO42−–Si (μg/g) | NO3−–N (μg/g) | NO2−–N (μg/g) |
|---|---|---|---|---|---|---|---|---|
| Hill | Hill.1 | 0.14 | 7.63 | 1.18 | 0.94 | 2.25 | 0.06 | 0.04 |
| Hill.2 | 0.13 | 7.97 | 1.00 | 1.89 | 2.32 | 0.30 | 0.22 | |
| Hill.3 | 0.14 | 8.03 | 1.06 | 0.50 | 1.85 | 0.62 | 0.04 | |
| average | 0.14 ± 0.01 | 7.88 ± 0.22 | 1.08 ± 0.09 | 1.11 ± 0.71 | 2.14 ± 0.25 | 0.32 ± 0.28 | 0.10 ± 0.10 | |
| Up | Up.1 | 0.16 | 7.62 | 1.53 | 2.85 | 1.43 | 0.62 | 0.23 |
| Up.2 | 0.11 | 7.68 | 1.16 | 1.85 | 1.75 | 0.52 | 0.15 | |
| Up.3 | 0.10 | 7.85 | 0.12 | 0.75 | 2.19 | 0.03 | 0.04 | |
| average | 0.12 ± 0.03 | 7.72 ± 12 | 0.94 ± 0.73 | 1.82 ± 1.05 | 1.79 ± 0.38 | 0.39 ± 0.32 | 0.14 ± 0.10 | |
| Down | Down.1 | 0.11 | 8.13 | 0.25 | 1.15 | 4.36 | 0.20 | 0.03 |
| Down.2 | 0.11 | 8.22 | 0.21 | 0.93 | 3.11 | 0.20 | 0.03 | |
| Down.3 | 0.08 | 8.11 | 0.16 | 1.39 | 4.50 | 0.44 | 0.06 | |
| average | 0.10 ± 0.02 | 8.15 ± 0.06 | 0.21 ± 0.05 | 1.16 ± 0.23 | 3.99 ± 0.77 | 0.28 ± 0.14 | 0.04 ± 0.02 | |
| Sediment | Sedi.1 | 0.18 | 7.91 | 0.11 | 3.27 | 3.11 | 0.05 | 0.01 |
| Sedi.2 | 0.23 | 7.84 | 0.51 | 4.08 | 4.29 | 0.01 | 0.01 | |
| Sedi.3 | 0.19 | 7.90 | 0.31 | 3.44 | 3.06 | 0.01 | 0.01 | |
| average | 0.20 ± 0.03 | 7.88 ± 0.04 | 0.31 ± 0.2 | 3.60 ± 0.42 | 3.49 ± 0.70 | 0.02 ± 0.02 | 0.01 ± 0 |
| Sample Name | Raw Tag | Effective | OTUs | Shannon (H’) | Pielou (J’) | Simpson (1-D) | chao1 | ACE | Good’s Coverage |
|---|---|---|---|---|---|---|---|---|---|
| Hill.1 | 29217 | 23991 | 75 | 2.491 | 0.426 | 0.773 | 83 | 90.254 | 0.999 |
| Hill.2 | 21558 | 17420 | 74 | 2.629 | 0.450 | 0.796 | 65.882 | 71.368 | 0.999 |
| Hill.3 | 25608 | 18117 | 87 | 2.248 | 0.385 | 0.707 | 122.667 | 123.325 | 0.998 |
| Up.1 | 26854 | 23756 | 56 | 2.564 | 0.439 | 0.787 | 66 | 74.721 | 0.999 |
| Up.2 | 26503 | 22461 | 60 | 2.392 | 0.410 | 0.775 | 54.667 | 56.963 | 0.999 |
| Up.3 | 23635 | 20012 | 69 | 2.213 | 0.379 | 0.756 | 62.545 | 66.666 | 0.999 |
| Down.1 | 28575 | 25156 | 42 | 1.803 | 0.309 | 0.632 | 36.25 | 36.296 | 1 |
| Down.2 | 27944 | 22992 | 50 | 2.187 | 0.374 | 0.715 | 40.909 | 43.927 | 1 |
| Down.3 | 26607 | 23313 | 65 | 2.114 | 0.362 | 0.643 | 53.077 | 57.351 | 0.999 |
| Sedi.1 | 27538 | 20514 | 196 | 3.09 | 0.529 | 0.789 | 191.091 | 195.725 | 0.998 |
| Sedi.2 | 31650 | 23542 | 276 | 4.116 | 0.705 | 0.851 | 280.41 | 284.337 | 0.997 |
| Sedi.3 | 19396 | 14710 | 206 | 3.414 | 0.585 | 0.757 | 190.634 | 194.981 | 0.999 |
| RDA1 | RDA2 | r2 | Pr (>r) | ||
|---|---|---|---|---|---|
| Water content | −0.963478 | 0.267788 | 0.6685 | 0.008 | ** |
| pH | 0.123615 | −0.992330 | 0.0209 | 0.908 | |
| OrC | 0.897715 | 0.440576 | 0.3345 | 0.149 | |
| NH4+–N | −0.995861 | 0.090889 | 0.6897 | 0.008 | ** |
| SiO42−–Si | −0.985292 | −0.170879 | 0.1667 | 0.431 | |
| NO3−–N | 0.989857 | 0.142067 | 0.3876 | 0.108 | |
| NO2−–N | 0.999554 | −0.029876 | 0.2080 | 0.362 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, J.; Liu, F.; Han, W.; Wang, Y.; Zhu, Q.; Zang, J.; Wang, S.; Zhang, B.; Wang, N. The Effect of Nitrogen Content on Archaeal Diversity in an Arctic Lake Region. Microorganisms 2019, 7, 543. https://doi.org/10.3390/microorganisms7110543
Lv J, Liu F, Han W, Wang Y, Zhu Q, Zang J, Wang S, Zhang B, Wang N. The Effect of Nitrogen Content on Archaeal Diversity in an Arctic Lake Region. Microorganisms. 2019; 7(11):543. https://doi.org/10.3390/microorganisms7110543
Chicago/Turabian StyleLv, Jinjiang, Feng Liu, Wenbing Han, Yu Wang, Qian Zhu, Jiaye Zang, Shuang Wang, Botao Zhang, and Nengfei Wang. 2019. "The Effect of Nitrogen Content on Archaeal Diversity in an Arctic Lake Region" Microorganisms 7, no. 11: 543. https://doi.org/10.3390/microorganisms7110543
APA StyleLv, J., Liu, F., Han, W., Wang, Y., Zhu, Q., Zang, J., Wang, S., Zhang, B., & Wang, N. (2019). The Effect of Nitrogen Content on Archaeal Diversity in an Arctic Lake Region. Microorganisms, 7(11), 543. https://doi.org/10.3390/microorganisms7110543





