Bacterial Consortium for Improved Maize (Zea mays L.) Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Rhizosphere Soil Sampling
Isolation of Bacteria from Rhizospheric Soils
2.2. In Vitro Screening of Isolates for Different Plant Growth-Promoting Activities
2.2.1. Phosphate Solubilization
2.2.2. Hydrogen Cyanide (HCN) Production
2.2.3. Ammonia Production
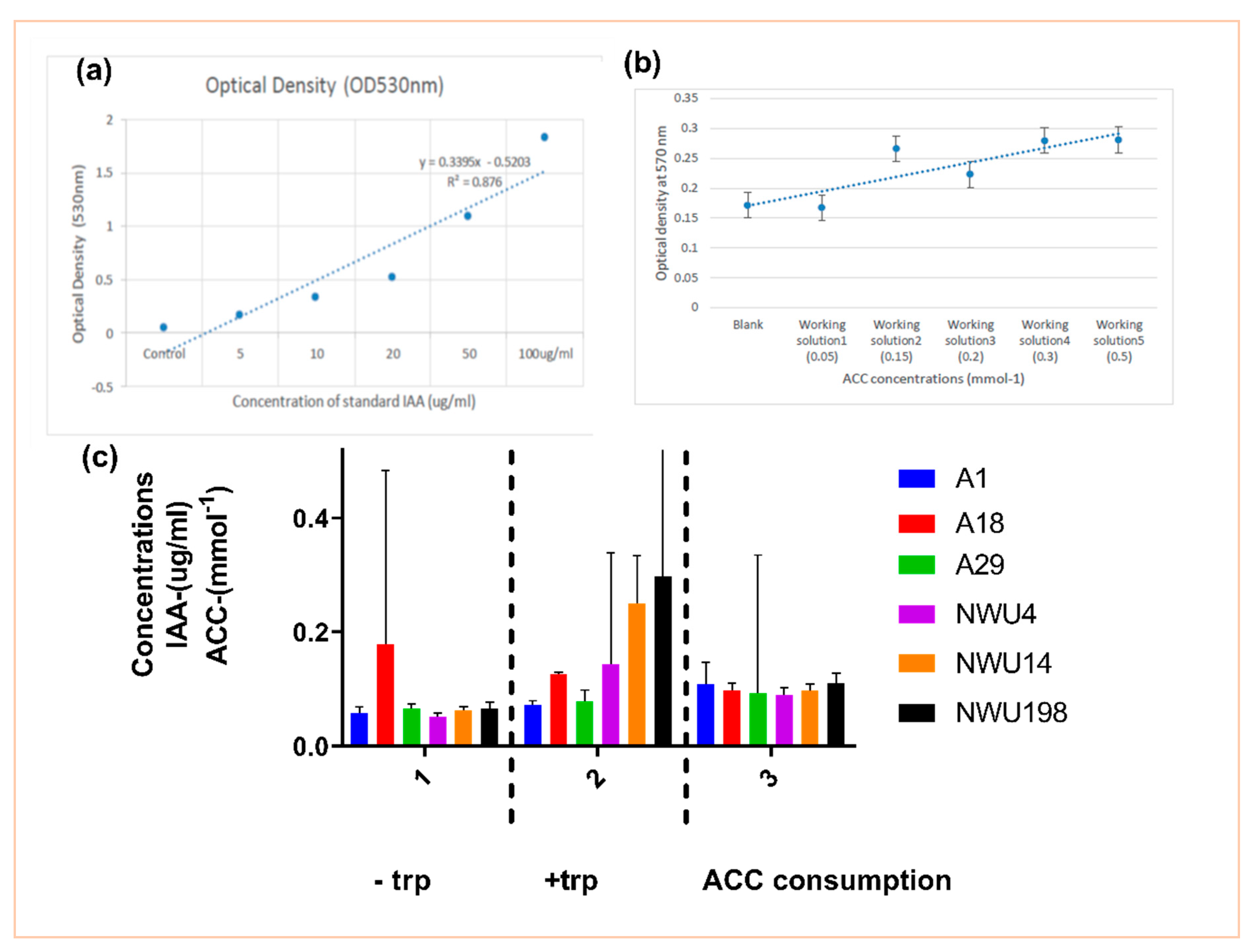
2.2.4. Detection of Indole Acetic Acid (IAA)
Quantification of IAA Production
2.2.5. 1-Aminocyclopropane-1-Carboxylic Acid (ACC) Utilization Assay
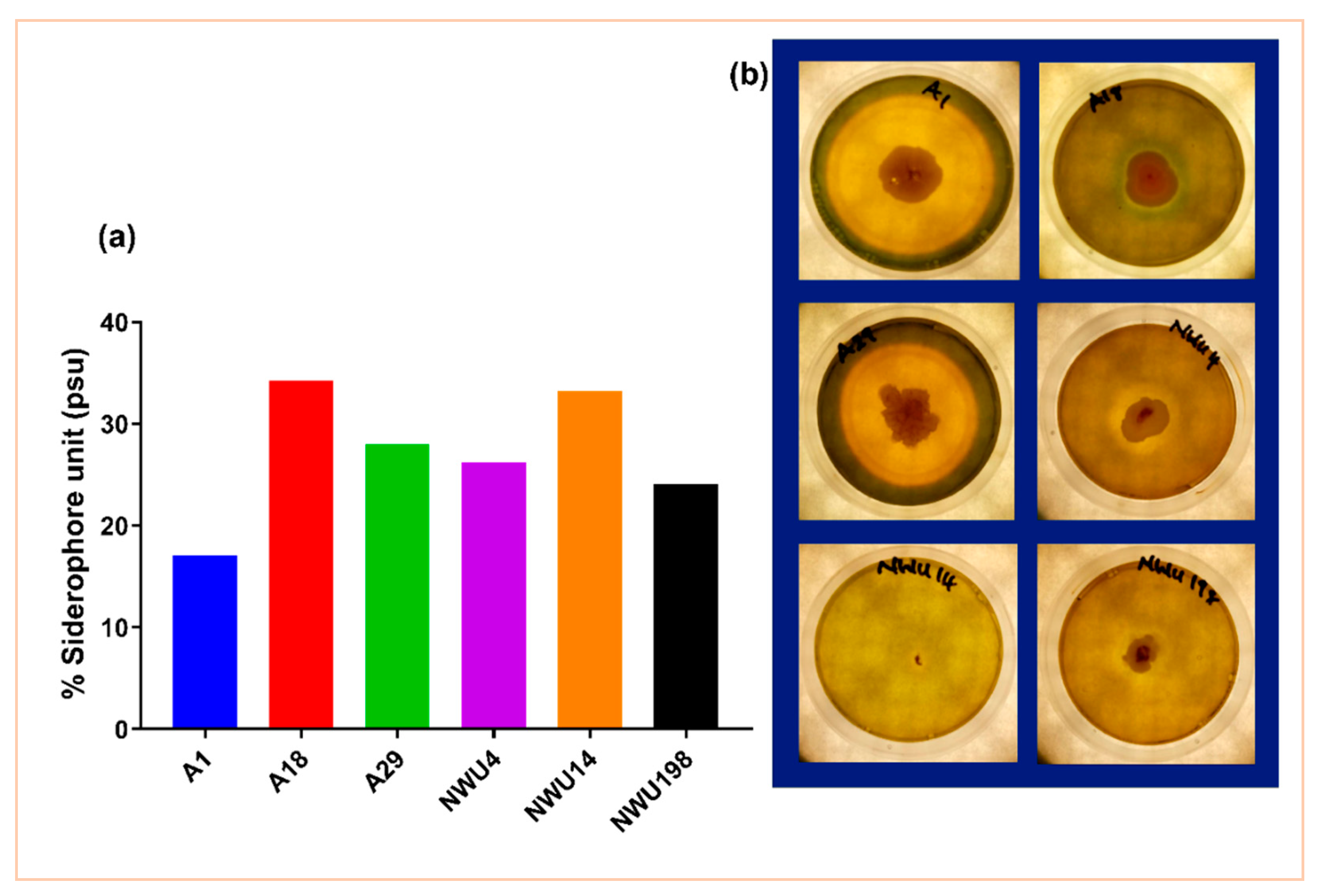
2.2.6. Siderophore Production
Detection in Plate Culture
Quantitative Estimation
2.3. Biochemical Activities of Isolates
2.3.1. Catalase Production
2.3.2. Protease Production
2.3.3. Oxidase Production
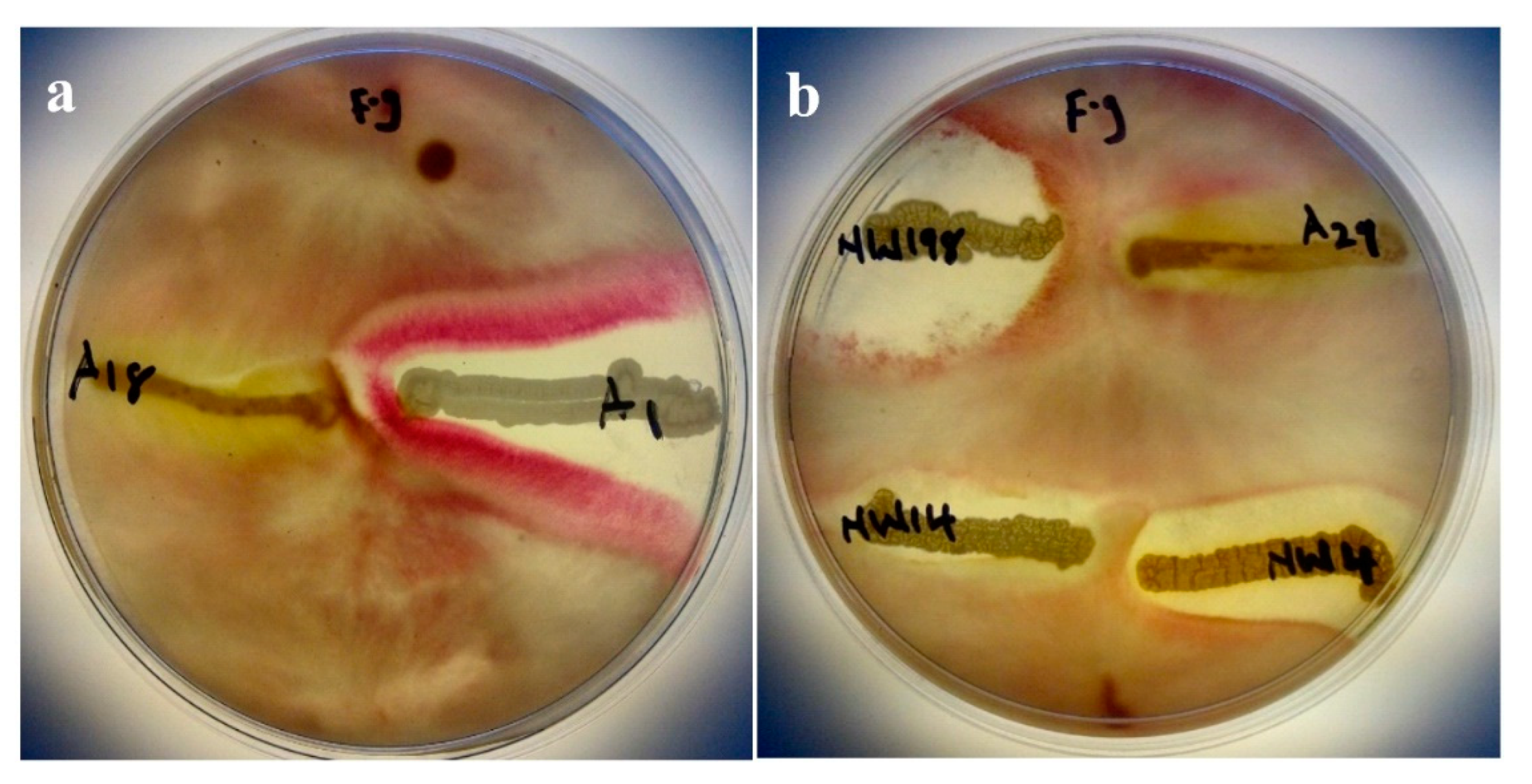
2.4. Antagonism Assay Against Phytopathogenic Fungi
2.5. Molecular Characterization of Bacterial Strains
2.5.1. DNA Extraction and 16S rDNA Gene Amplification
2.5.2. Sequence Alignment and Phylogenetic Analysis
2.6. Preparation of Inoculum and Seed Treatment for Field Trial
2.6.1. Preparation of Inoculum and Seed Treatment
2.6.2. Field Study
2.7. Statistical Analysis
3. Results
3.1. In Vitro Screening of Isolates for Plant Growth-Promoting Activities
3.1.1. IAA Production and ACC Utilization Assay
3.1.2. Siderophore Production
3.2. Biochemical Activities of Isolates
3.3. Antagonism Assay Against Phytopathogenic Fungi
3.4. Molecular Characterization of Bacterial Strains
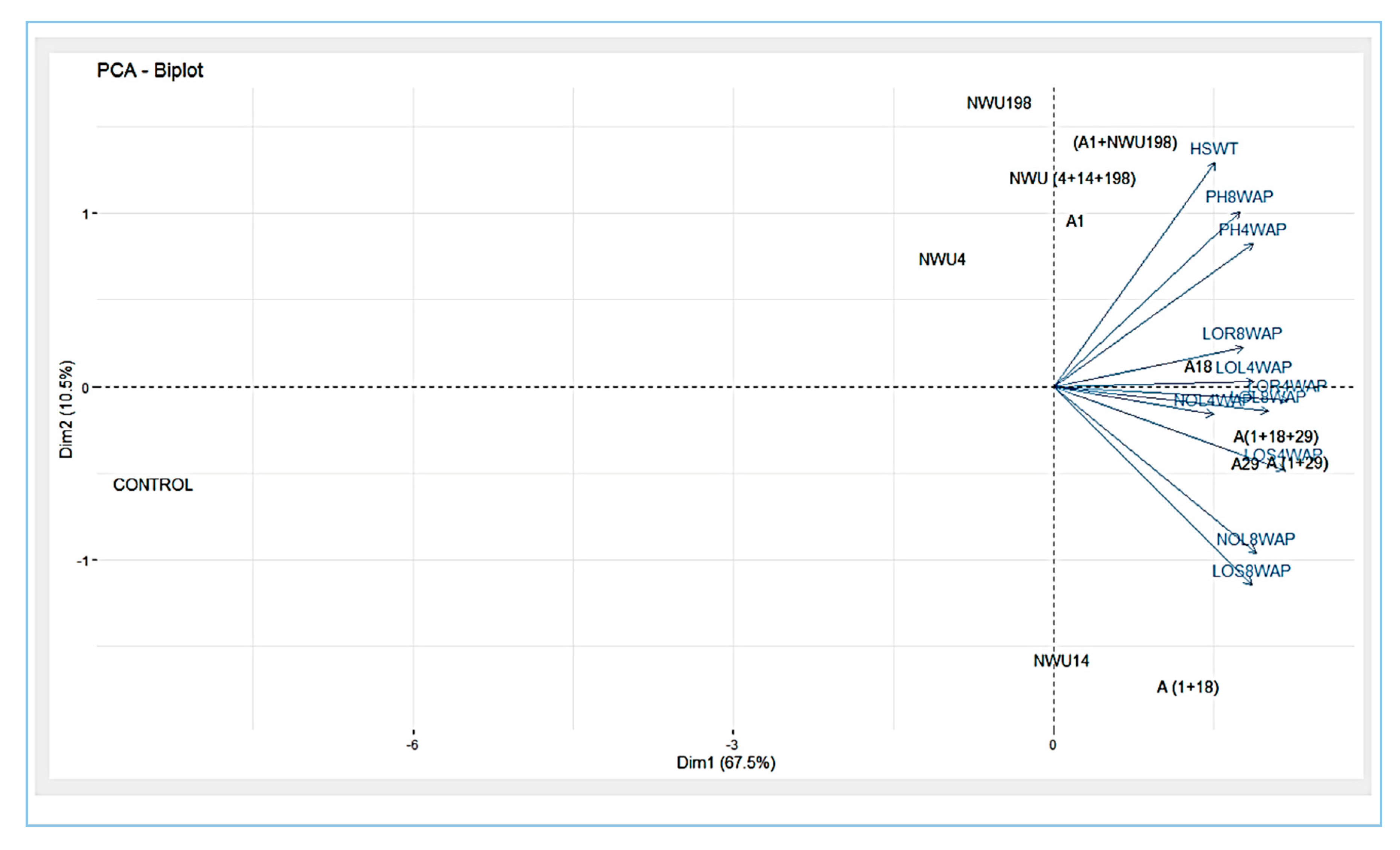
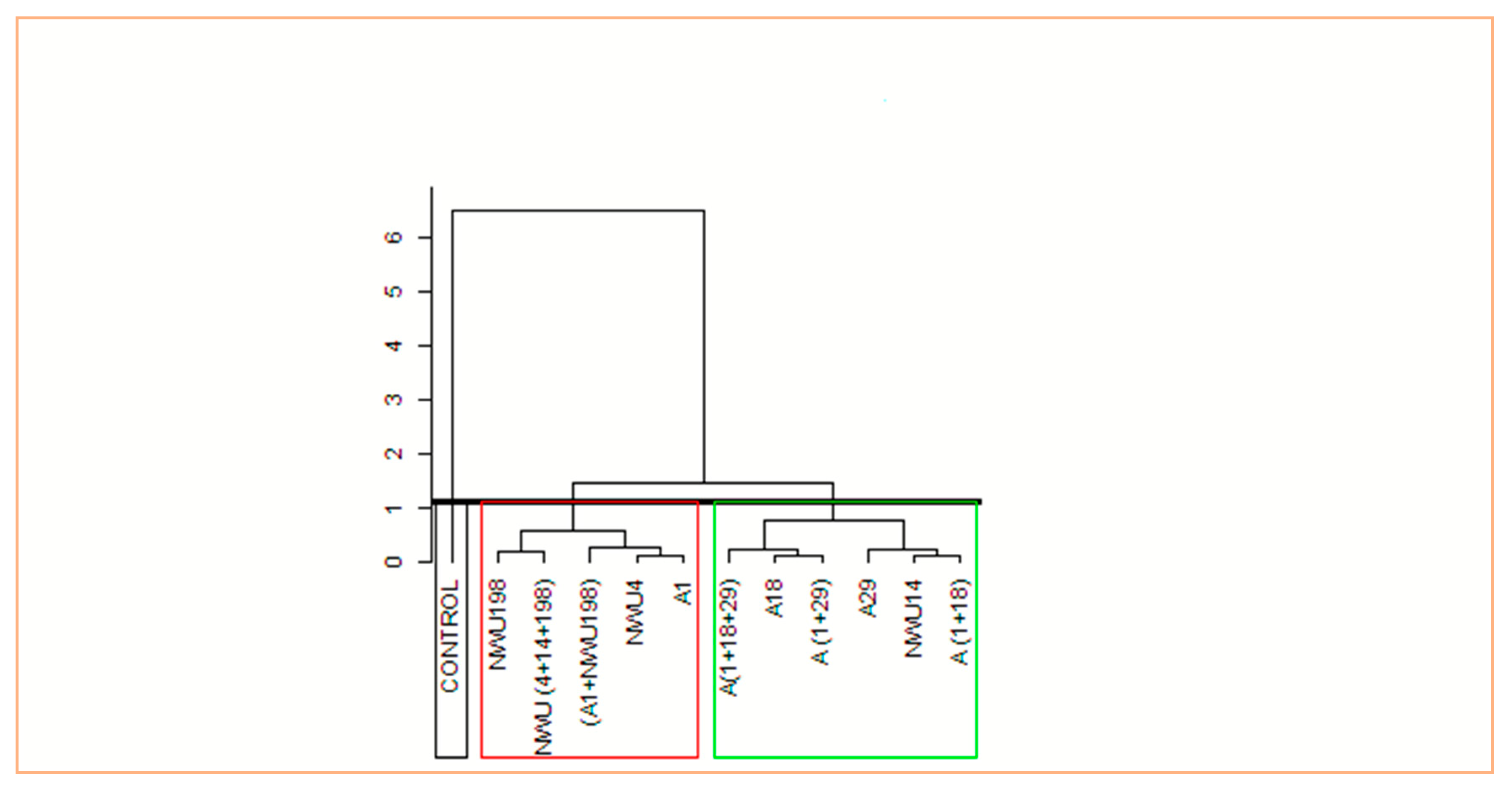
3.5. Plant Growth Promotion Assay
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saharan, B.S.; Nehra, V. Plant growth promoting rhizobacteria: A critical review. Life Sci. Med. Res. 2011, 21, 1–30. [Google Scholar]
- Olanrewaju, O.S.; Glick, B.R.; Babalola, O.O. Mechanisms of action of plant growth promoting bacteria. World J. Microbiol. Biotechnol. 2017, 33, 197. [Google Scholar] [CrossRef] [PubMed]
- Adjanohoun, A.; Allagbe, M.; Noumavo, P.; Gotoechan-Hodonou, H.; Sikirou, R.; Dossa, K.; GleleKakaï, R.; Kotchoni, S.; Baba-Moussa, L. Effects of plant growth promoting rhizobacteria on field grown maize. J. Anim. Plant Sci. 2011, 11, 1457–1465. [Google Scholar]
- Gopalakrishnan, S.; Srinivas, V.; Sree Vidya, M.; Rathore, A. Plant growth-promoting activities of Streptomyces sp. In sorghum and rice. SpringerPlus 2013, 2, 574. [Google Scholar] [CrossRef]
- Islam, F.; Yasmeen, T.; Ali, Q.; Ali, S.; Arif, M.S.; Hussain, S.; Rizvi, H. Influence of Pseudomonas aeruginosa as PGPR on oxidative stress tolerance in wheat under Zn stress. Ecotoxicol. Environ. Saf. 2014, 104, 285–293. [Google Scholar] [CrossRef]
- Alori, E.T.; Babalola, O.O.; Prigent-Combaret, C. Impacts of Microbial Inoculants on the Growth and Yield of Maize Plant. Open Agric. J. 2019, 13, 1–8. [Google Scholar] [CrossRef]
- Kumari, S.; Bharat, N.K. In vitro screening and efficacy of plant growth promoting rhizobacteria and biocontrol agents in bell pepper (Capsicum annuum L.). J. Pharmacogn. Phytochem. 2018, 7, 1472–1475. [Google Scholar]
- Toumatia, O.; Compant, S.; Yekkour, A.; Goudjal, Y.; Sabaou, N.; Mathieu, F.; Sessitsch, A.; Zitouni, A. Biocontrol and plant growth promoting properties of Streptomyces mutabilis strain IA1 isolated from a Saharan soil on wheat seedlings and visualization of its niches of colonization. S. Afr. J. Bot. 2016, 105, 234–239. [Google Scholar] [CrossRef]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L.M.P. Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Genet. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef]
- Jacobsen, S.-E.; Sørensen, M.; Pedersen, S.M.; Weiner, J. Feeding the world: Genetically modified crops versus agricultural biodiversity. Agron. Sust. Dev. 2013, 33, 651–662. [Google Scholar] [CrossRef]
- Arruda, L.; Beneduzi, A.; Martins, A.; Lisboa, B.; Lopes, C.; Bertolo, F.; Passaglia, L.M.P.; Vargas, L.K. Screening of rhizobacteria isolated from maize (Zea mays L.) in Rio Grande do Sul State (South Brazil) and analysis of their potential to improve plant growth. Appl. Soil Ecol. 2013, 63, 15–22. [Google Scholar] [CrossRef]
- Tang, M.; He, X.; Luo, Y.; Ma, L.; Tang, X.; Huang, K. Nutritional assessment of transgenic lysine-rich maize compared with conventional quality protein maize. J. Sci. Food Agric. 2013, 93, 1049–1054. [Google Scholar] [CrossRef] [PubMed]
- Ahemad, M.; Kibret, M. Mechanisms and applications of plant growth promoting rhizobacteria: Current perspective. J. King Saud Univ. Sci. 2014, 26, 1–20. [Google Scholar] [CrossRef]
- Olanrewaju, O.S.; Ayangbenro, A.S.; Glick, B.R.; Babalola, O.O. Plant health: Feedback effect of root exudates-rhizobiome interactions. Appl. Microbiol. Biotechnol. 2019, 103, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Pandey, P.; Dubeya, R.; Maheshwari, D. Bacteria consortium optimization improves nutrient uptake, nodulation, disease suppression and growth of the common bean (Phaseolus vulgaris) in both pot and field studies. Rhizosphere 2016, 2, 13–23. [Google Scholar] [CrossRef]
- Shanmugam, V.; Thakur, H.; Kaur, J.; Gupta, S.; Rajkumar, S.; Dohroo, N.P. Genetic diversity of Fusarium spp. inciting rhizome rot of ginger and its management by PGPR consortium in the western Himalayas. Biol. Control. 2013, 66, 1–7. [Google Scholar] [CrossRef]
- Wang, C.-J.; Yang, W.; Wang, C.; Gu, C.; Niu, D.-D.; Liu, H.-X.; Wang, Y.-P.; Guo, J.-H. Induction of drought tolerance in cucumber plants by a consortium of three plant growth-promoting rhizobacterium strains. PLoS ONE 2012, 7, e52565. [Google Scholar] [CrossRef]
- Berendsen, R.L.; Vismans, G.; Yu, K.; Song, Y.; de Jonge, R.; Burgman, W.P.; Burmølle, M.; Herschend, J.; Bakker, P.A.; Pieterse, C.M. Disease-induced assemblage of a plant-beneficial bacterial consortium. ISME J. 2018, 12, 1496. [Google Scholar] [CrossRef]
- Sharma, C.K.; Vishnoi, V.K.; Dubey, R.; Maheshwari, D. A twin rhizospheric bacterial consortium induces systemic resistance to a phytopathogen Macrophomina phaseolina in mung bean. Rhizosphere 2018, 5, 71–75. [Google Scholar] [CrossRef]
- Verma, S.C.; Ladha, J.K.; Tripathi, A.K. Evaluation of plant growth promoting and colonization ability of endophytic diazotrophs from deep water rice. J. Biotechnol. 2001, 91, 127–141. [Google Scholar] [CrossRef]
- Castric, P.A. Hydrogen cyanide, a secondary metabolite of Pseudomonas aeruginosa. Can. J. Microbiol. 1975, 21, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Cappuccino, J.G.; Sherman, N. Biochemical activities of microorganisms. In Microbiology, a Laboratory Manual, 1st ed.; The Benjamin/Cummings Publishing Co.: San Francisco, CA, USA, 1992; pp. 105–300. [Google Scholar]
- Gordon, S.A.; Weber, R.P. Colorimetric estimation of indoleacetic acid. Plant. Physiol. 1951, 26, 192. [Google Scholar] [CrossRef] [PubMed]
- Patten, C.L.; Glick, B.R. Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl. Environ. Microbiol. 2002, 68, 3795–3801. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chang, S.; Lin, L.; Li, Y.; An, Q. A colorimetric assay of 1-aminocyclopropane-1-carboxylate (ACC) based on ninhydrin reaction for rapid screening of bacteria containing ACC deaminase. Lett. Appl. Microbiol. 2011, 53, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Schwyn, B.; Neilands, J. Universal chemical assay for the detection and determination of siderophores. Ana. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Clarke, P.H.; Cowan, S. Biochemical methods for bacteriology. J. Gen. Microbiol. 1952, 6, 187–197. [Google Scholar] [CrossRef]
- Maurhofer, M.; Keel, C.; Haas, D.; Défago, G. Influence of plant species on disease suppression by Pseudomonas fluorescens strain CHAO with enhanced antibiotic production. Plant. Pathol. 1995, 44, 40–50. [Google Scholar] [CrossRef]
- Murray, R.; Costilow, R.N.; Nester, E.; Wood, W.A.; Krieg, N.R.; Phillips, G. Manual of methods for general bacteriology; American Society for Microbiology: Washington, DC, USA, 1981; pp. 409–433. [Google Scholar]
- Gopalakrishnan, S.; Pande, S.; Sharma, M.; Humayun, P.; Kiran, B.K.; Sandeep, D.; Vidya, M.S.; Deepthi, K.; Rupela, O. Evaluation of actinomycete isolates obtained from herbal vermicompost for the biological control of Fusarium wilt of chickpea. Crop. Prot. 2011, 30, 1070–1078. [Google Scholar] [CrossRef]
- Adegboye, M.F.; Babalola, O.O. Isolation, characterization and antibacterial activity of Streptomycetes from rhizosphere soils in North West Province, South Africa. Asia Life Sci. 2013, 9, 403–421. [Google Scholar]
- Olanrewaju, O.S.; Babalola, O.O. Streptomyces: Implications and interactions in plant growth promotion. Appl. Microbiol. Biotechnol. 2018, 103, 1179–1188. [Google Scholar] [CrossRef]
- Duncan, D.B. Multiple range and multiple F tests. Biometrics 1955, 11, 1–42. [Google Scholar] [CrossRef]
- Jha, C.K.; Saraf, M. Plant growth promoting rhizobacteria (PGPR): A review. J. Agric. Res. Dev. 2015, 5, 108–119. [Google Scholar]
- Verma, J.A.Y.; Jaiswal, D.K.M.; Prakash, S.; Yadav, J.; Singh, V. Characterization and screening of thermophilic Bacillus strains for developing plant growth promoting consortium from hot spring of Leh and Ladakh region of India. Front. Microbiol. 2018, 9, 1293. [Google Scholar] [CrossRef] [PubMed]
- Antoun, H. Beneficial Microorganisms for the Sustainable Use of Phosphates in Agriculture. Procedia Eng. 2012, 46, 62–67. [Google Scholar] [CrossRef]
- Kurabachew, H.; Wydra, K. Characterization of plant growth promoting rhizobacteria and their potential as bioprotectant against tomato bacterial wilt caused by Ralstonia solanacearum. Biol. Control. 2013, 67, 75–83. [Google Scholar] [CrossRef]
- Ahmad, F.; Ahmad, I.; Khan, M.S. Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiol. Res. 2008, 163, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Di, X.; Takken, F.L.; Tintor, N. How phytohormones shape interactions between plants and the soil-borne fungus Fusarium oxysporum. Front. Plant. Sci. 2016, 7, 170. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, M.; Singh, P.P.; Singh, S.K.; Singh, P.K.; Pandey, K.D. Isolation of plant growth promoting rhizobacteria and their impact on growth and curcumin content in Curcuma longa L. Biocatal. Agric. Biotechnol. 2016, 8, 1–7. [Google Scholar] [CrossRef]
- Kumawat, K.; Sharma, P.; Sirari, A.; Singh, I.; Gill, B.; Singh, U.; Saharan, K. Synergism of Pseudomonas aeruginosa (LSE-2) nodule endophyte with Bradyrhizobium sp. (LSBR-3) for improving plant growth, nutrient acquisition and soil health in soybean. World J. Microbiol. Biotechnol. 2019, 35, 47. [Google Scholar] [CrossRef]
- Glick, B.R. Bacterial ACC Deaminase and the Alleviation of Plant Stress. In Advances in Applied Microbiology; Academic Press: Cambridge, MA, USA, 2004; pp. 291–312. [Google Scholar]
- Maheshwari, R.; Bhutani, N.; Suneja, P. Screening and characterization of siderophore producing endophytic bacteria from Cicer arietinum and Pisum sativum plants. J. Appl. Biol. Biotechnol. 2019, 7, 7–14. [Google Scholar]
- Arya, N.; Rana, A.; Rajwar, A.; Sahgal, M.; Sharma, A. Biocontrol efficacy of siderophore producing indigenous Pseudomonas strains against Fusarium wilt in Tomato. Nat. Acad. Sci. Lett. 2018, 41, 133–136. [Google Scholar] [CrossRef]
- Verma, V.; Singh, S.; Prakash, S. Bio-control and plant growth promotion potential of siderophore producing endophytic Streptomyces from Azadirachta indica A. Juss. J. Basic Microbiol. 2011, 51, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Tortora, M.L.; Díaz-Ricci, J.C.; Pedraza, R.O. Azospirillum brasilense siderophores with antifungal activity against Colletotrichum acutatum. Arch. Microbiol. 2011, 193, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, G.; Maheshwari, H.; Sanket, J.; Josh, I. Isolation and characterization of nodule associated Exiguobacterium sp. from the root nodules of Fenugreek (Trigonella foenumgraecum) and their possible role in plant growth promotion. Int. J. Microbiol. 2012, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Douriet-Gámez, N.R.; Maldonado-Mendoza, I.E.; Ibarra-Laclette, E.; Blom, J.; Calderón-Vázquez, C.L. Genomic analysis of Bacillus sp. Strain B25, a biocontrol agent of maize pathogen Fusarium verticillioides. Curr. Microbiol. 2018, 75, 247–255. [Google Scholar] [CrossRef]
- Chakraborty, K.; Thilakan, B.; Kizhakkekalam, V.K. Antibacterial aryl-crowned polyketide from Bacillus subtilis associated with seaweed Anthophycus longifolius. J. Appl. Microbiol. 2018, 124, 108–125. [Google Scholar] [CrossRef]
- Someya, N.; Tsuchiya, K.; Yoshida, T.; Noguchi, M.T.; Akutsu, K.; Sawada, H. Co-inoculation of an antibiotic-producing bacterium and a lytic enzyme-producing bacterium for the biocontrol of tomato wilt caused by Fusarium oxysporum f. sp. lycopersici. Biocont. Sci. 2007, 12, 1–6. [Google Scholar] [CrossRef][Green Version]
- Thakkar, A.; Saraf, M. Development of microbial consortia as a biocontrol agent for effective management of fungal diseases in Glycine max L. Arch. Phytopathol. Plant. Prot. 2015, 48, 459–474. [Google Scholar] [CrossRef]
- Beukes, I.; Rose, L.J.; van Coller, G.J.; Viljoen, A. Disease development and mycotoxin production by the Fusarium graminearum species complex associated with South African maize and wheat. Eur. J. Plant. Pathol. 2017, 150, 893–910. [Google Scholar] [CrossRef]
- Harris, L.J.; Balcerzak, M.; Johnston, A.; Schneiderman, D.; Ouellet, T. Host-preferential Fusarium graminearum gene expression during infection of wheat, barley, and maize. Fungal Biol. 2016, 120, 111–123. [Google Scholar] [CrossRef]
- Quesada-Ocampo, L.M.; Al-Haddad, J.; Scruggs, A.C.; Buell, C.R.; Trail, F. Susceptibility of maize to stalk rot caused by Fusarium graminearum deoxynivalenol and zearalenone mutants. Phytopathology 2016, 106, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Mousa, W.K.; Shearer, C.R.; Limay-Rios, V.; Zhou, T.; Raizada, M.N. Bacterial endophytes from wild maize suppress Fusarium graminearum in modern maize and inhibit mycotoxin accumulation. Front. Plant. Sci. 2015, 6, 805. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Selvaraj, J.N.; Xing, F.; Zhou, L.; Wang, Y.; Song, H.; Tan, X.; Sun, L.; Sangare, L.; Folly, Y.M.E. Antagonistic action of Bacillus subtilis strain SG6 on Fusarium graminearum. PLoS ONE 2014, 9, e92486. [Google Scholar] [CrossRef] [PubMed]
- Bressan, W.; Figueiredo, J.E.F. Efficacy and dose-response relationship in biocontrol of Fusarium disease in maize by Streptomyces spp. Eur. J. Plant. Pathol. 2008, 120, 311–316. [Google Scholar] [CrossRef]
- Vijayabharathi, R.; Gopalakrishnan, S.; Sathya, A.; Vasanth Kumar, M.; Srinivas, V.; Mamta, S. Streptomyces sp. as plant growth-promoters and host-plant resistance inducers against Botrytis cinerea in chickpea. Biocont. Sci. Technol. 2018, 28, 1140–1163. [Google Scholar] [CrossRef]
- Cheng, G.; Huang, Y.; Yang, H.; Liu, F. Streptomyces felleus YJ1: Potential biocontrol agents against the sclerotinia stem rot (Sclerotinia sclerotiorum) of oilseed rape. J. Agric. Sci. 2014, 6, 91. [Google Scholar] [CrossRef]
- Babalola, O.O. Beneficial bacteria of agricultural importance. Biotechnol. Lett. 2010, 32, 1559–1570. [Google Scholar] [CrossRef]
- Glick, B.R. Plant Growth-Promoting Bacteria: Mechanisms and Applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef]
- Argaw, A. Evaluation of co-inoculation of Bradyrhizobium japonicum and phosphate solubilizing Pseudomonas sp. effect on soybean (Glycine max L. (Merr.)) in Assossa area. J. Agric. Res. 2012, 14, 213–224. [Google Scholar]
- Akhtar, N.; Naveed, M.; Khalid, M.; Ahmad, N.; Rizwan, M.; Siddique, S. Effect of bacterial consortia on growth and yield of maize grown in Fusarium infested soil. Soil Environ. 2018, 37, 35–44. [Google Scholar] [CrossRef]

| Bacterial Isolates | Ammonia Production | HCN Production | IAA Production | Phosphate Solubilization |
|---|---|---|---|---|
| A1, A4, A6 | ++ | − | − | + |
| A2, NWU 4 | + | + | − | − |
| A3 | + | + | − | + |
| A5, A7, A9, A10, A13, A16, A19, A21, A25, A28, A30, NWU 14, NWU 198 | + | − | − | − |
| A8 | − | − | − | − |
| A11, A23 | ++ | − | + | − |
| A12, A14 | + | − | + | − |
| A15, A17 | ++ | − | + | + |
| A18 | ++ | + | + | ++ |
| A20 | ++ | ++ | + | ++ |
| A22, A24, A27, A31 | ++ | − | − | − |
| A26 | − | ++ | + | ++ |
| A29 | ++ | − | + | + |
| Bacterial Isolates | Catalase Production | Oxidase Production | Protease Production |
|---|---|---|---|
| A1, A2 | + | + | + |
| A3, A18 | + | ++ | − |
| A4, NWU14, NWU198 | ++ | + | − |
| A5, A6, A10, A24, NWU4 | + | + | − |
| A7, A9, A16, A25 | + | − | + |
| A8, A26 | − | − | − |
| A11, A23 | + | ++ | + |
| A12, A14, A15, A19, A31 | + | − | − |
| A13, A17, A20, A21 | ++ | − | − |
| A22, A29 | ++ | ++ | − |
| A27, A28 | ++ | + | + |
| A30 | ++ | ++ | + |
| Treatments | LOR4WAP (cm) | LOR8WAP (cm) | LOL4WAP (cm) | LOL8WAP (cm) | LOS4WAP (cm) | LOS8WAP (cm) | NOL4W AP | NOL8W AP | PH4WAP (cm) | PH8WAP (cm) | HSWT (g) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | 0.84 ± 0.06 e | 1.08 ± 0.06 f | 0.97 ± 0.01 c | 1.16 ± 0.04 b | 1.53 ± 0.01 d | 1.62 ± 0.03 e | 0.92 ± 0.03 d | 0.95 ± 0 b | 1.63 ± 0.05 d | 1.86 ± 0.04 c | 1.35 ± 0.02d |
| A1 | 1.14 ± 0.05 bcd | 1.22 ± 0.03 ed | 1.31 ± 0.02 ab | 1.34 ± 0.02 a | 1.74 ± 0.03 abc | 1.77 ± 0.07 cd | 0.98 ± 0.03 abcd | 1.01 ± 0.05ab | 1.87 ± 0.12 abc | 2.04 ± 0.03 ab | 1.47 ± 0.03 abc |
| A18 | 1.2 ± 0.03 ab | 1.27 ± 0.02 bc | 1.31 ± 0.04 b | 1.37 ± 0.04 a | 1.69 ± 0.03 abc | 1.78 ± 0.02 bc | 1.02 ± 0.06 abc | 1.05 ± 0.02 ab | 1.94 ± 0.01 a | 2.03 ± 0.02 ab | 1.44 ± 0.04 bc |
| A29 | 1.23 ± 0.01 a | 1.26 ± 0.02 cd | 1.34 ± 0.04 a | 1.37 ± 0.06 a | 1.79 ± 0.03 ab | 1.77 ± 0.02 bc | 0.97 ± 0.06 abcd | 1.09 ± 0.04 a | 1.88 ± 0.04 abc | 2.04 ± 0.03 ab | 1.45 ± 0.05 bc |
| NWU4 | 1.10 ± 0.04 d | 1.19 ± 0.01 e | 1.28 ± 0.06 ab | 1.34 ± 0.05 a | 1.73 ± 0.06 abc | 1.73 ± 0.04 cd | 0.95 ± 0.10 bcd | 1.01 ± 0.02 ab | 1.87 ± 0.01 abc | 2.01 ± 0.072 ab | 1.46 ± 0.04 bc |
| NWU14 | 1.15 ± 0.02 abcd | 1.27 ± 0.02 bcd | 1.31 ± 0.09 ab | 1.43 ± 0.06 a | 1.74 ± 0.02 abc | 1.76 ± 0.05 ab | 0.98 ± 0.03 abcd | 1.05 ± 0.08 ab | 1.81 ± 0.01 c | 1.98 ± 0.02 b | 1.42 ± 0.03 c |
| NWU198 | 1.12 ± 0.02 cd | 1.31 ± 0.02 ab | 1.24 ± 0.08 b | 1.28 ± 0.06 a | 1.69 ± 0.04 c | 1.76 ± 0.02 d | 0.97 ± 0.03 abcd | 1.01 ± 0.05 ab | 1.88 ± 0.05 abc | 2.01 ± 0.05 ab | 1.49 ± 0.02ab |
| A (1 + 18) | 1.19 ± 0.04 abc | 1.26 ± 0.03 bcd | 1.36 ± 0.01 b | 1.46 ± 0.04 a | 1.71 ± 0.06 a | 1.76 ± 0.01 a | 0.97 ± 0.07 abcd | 1.09 ± 0.02 a | 1.88 ± 0.03 abc | 1.99 ± 0.05 b | 1.44 ± 0.03 bc |
| A (1 + 29) | 1.2 ± 0.05 abc | 1.34 ± 0.04 a | 1.35 ± 0.01 ab | 1.44 ± 0.03 a | 1.74 ± 0.01 a | 1.75 ± 0.01 ab | 1.04 ± 0.04 ab | 1.06 ± 0.06 a | 1.90 ± 0.04 abc | 2.02 ± 0.02 ab | 1.47 ± 0.03 abc |
| (A1 + NWU198) | 1.15 ± 0.01 abcd | 1.25 ± 0.01 cd | 1.29 ± 0.06 b | 1.34 ± 0.06 a | 1.72 ± 0.05 abc | 1.72 ± 0.07 cd | 1.01 ± 0.02 abcd | 1.04 ± 0.04 ab | 1.92 ± 0.02 ab | 2.08 ± 0.04 a | 1.47 ± 0.01 abc |
| A (1 + 18 + 29) | 1.22 ± 0.02 a | 1.34 ± 0.016 a | 1.32 ± 0.02 b | 1.43 ± 0.01 a | 1.70 ± 0.03 abc | 1.79 ± 0.02 ab | 1.05 ± 0.02 a | 1.05 ± 0.08 ab | 1.83 ± 0.07 bc | 2.01 ± 0.05ab | 1.50 ± 0.02ab |
| NWU (4 + 14 + 198) | 1.2 ± 0.07 abc | 1.33 ± 0.03 a | 1.25 ± 0.04 b | 1.36 ± 0.04 a | 1.71 ± 0.03 bc | 1.72 ± 0.01 bcd | 0.94 ± 0.03 cd | 1.04 ± 0.08 ab | 1.88 ± 0.02 abc | 1.99 ± 0.02 b | 1.52 ± 0.01 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olanrewaju, O.S.; Babalola, O.O. Bacterial Consortium for Improved Maize (Zea mays L.) Production. Microorganisms 2019, 7, 519. https://doi.org/10.3390/microorganisms7110519
Olanrewaju OS, Babalola OO. Bacterial Consortium for Improved Maize (Zea mays L.) Production. Microorganisms. 2019; 7(11):519. https://doi.org/10.3390/microorganisms7110519
Chicago/Turabian StyleOlanrewaju, Oluwaseyi Samuel, and Olubukola Oluranti Babalola. 2019. "Bacterial Consortium for Improved Maize (Zea mays L.) Production" Microorganisms 7, no. 11: 519. https://doi.org/10.3390/microorganisms7110519
APA StyleOlanrewaju, O. S., & Babalola, O. O. (2019). Bacterial Consortium for Improved Maize (Zea mays L.) Production. Microorganisms, 7(11), 519. https://doi.org/10.3390/microorganisms7110519






