Bacterial and Fungal Diversity Inside the Medieval Building Constructed with Sandstone Plates and Lime Mortar as an Example of the Microbial Colonization of a Nutrient-Limited Extreme Environment (Wawel Royal Castle, Krakow, Poland)
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Sample Collection
2.3. Cultivation of Microorganisms
2.4. Siderophore Detection Assay
2.5. Extraction of Total DNA, Marker Gene Amplifications and High-Throughput Sequencing of 16S rDNA and ITS2 Amplicons
2.6. Bioinformatic Processing of the Sequenced Data
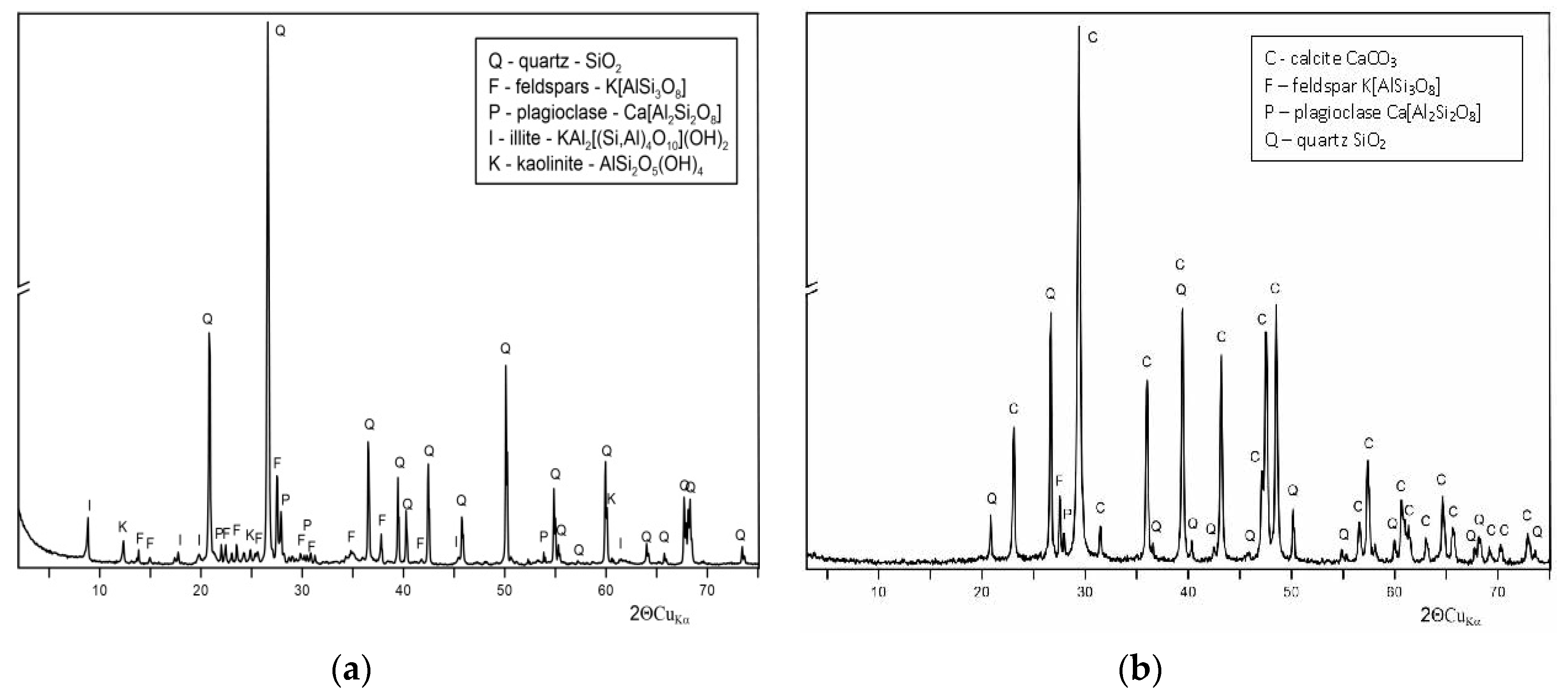
2.7. X-ray Powder Diffraction (XRD) Analysis
2.8. Measurements of the Chemical Composition of Air
2.9. Quantitative Analysis of Particulate Matter Concentrations in the Air
2.10. Scanning Electron Microscopy (SEM)
3. Results
3.1. Environmental Conditions in the Rotunda of Sts. Felix and Adauctus
3.2. Scanning Electron Microscopy (SEM)
3.3. Siderophores
3.4. Diversity Analysis with the Use of 16S rDNA and ITS2 Marker Gene Sequencing
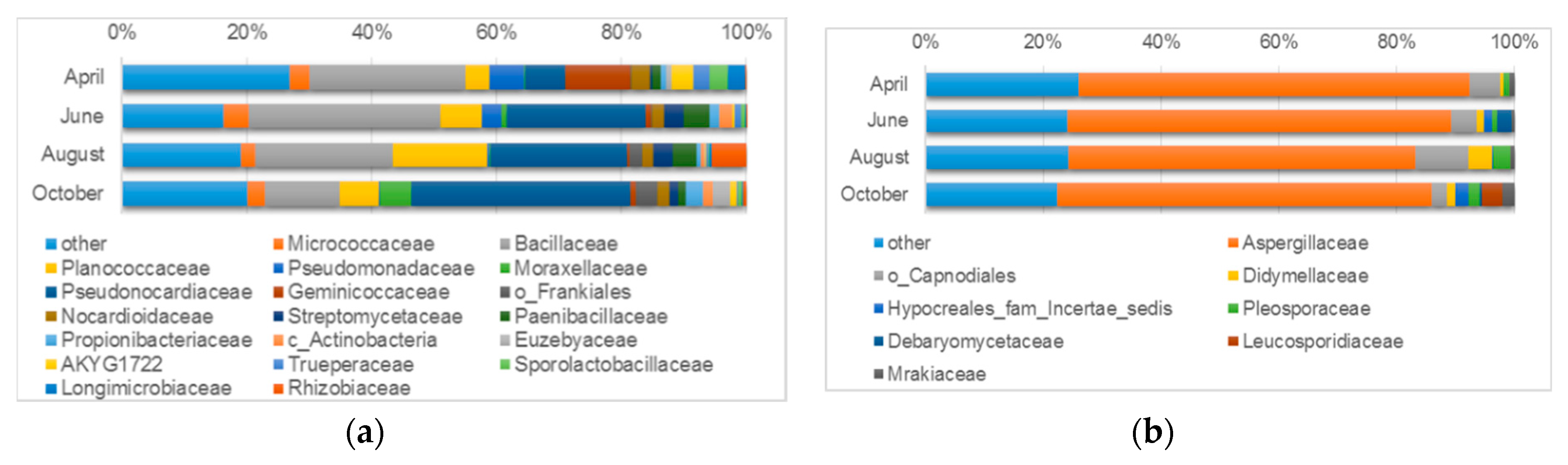
3.4.1. Microbiological Diversity of Ancient Wall Surfaces
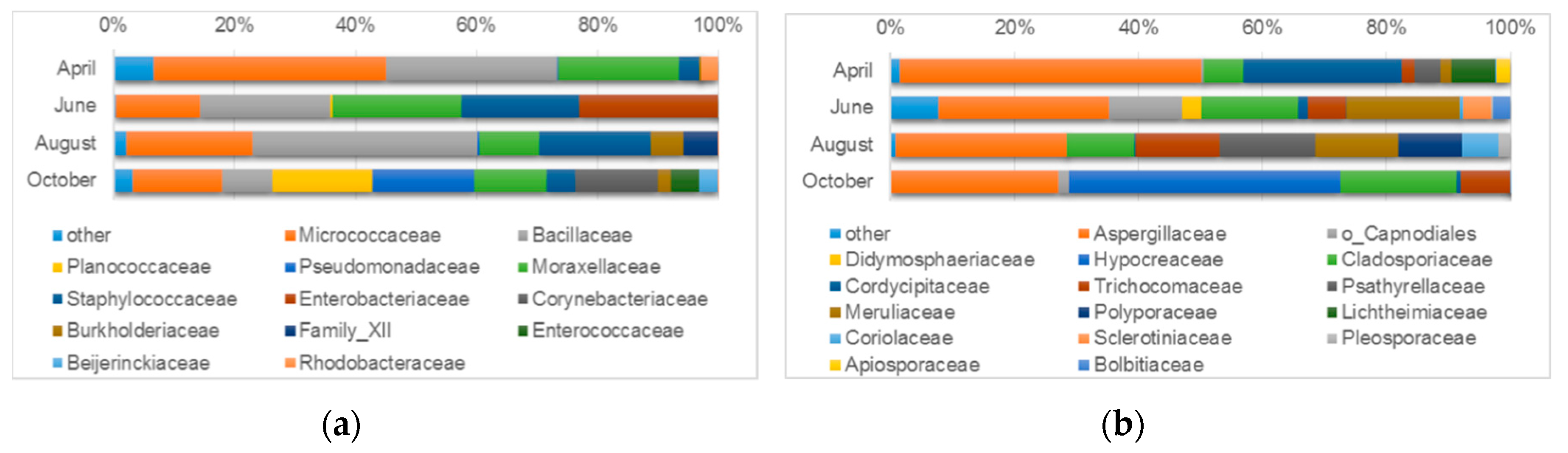
3.4.2. Microbiological Diversity of Indoor Air
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hueck, H.J. The biodeterioration of materials—an appraisal. Int. Biodeterior. Biodegrad. 2001, 48, 5–11. [Google Scholar] [CrossRef]
- Di Carlo, E.; Barresi, G.; Palla, F. Biotechnology and Conservation of Cultural Heritage, 1st ed.; Springer: Cham, Switzerland, 2017; pp. 1–30. [Google Scholar]
- Pinheiro, A.C.; Mesquita, N.; Trovão, J.; Soares, F.; Tiago, I.; Coelho, C.; de Carvalho, H.P.; Gil, F.; Catarino, L.; Piñar, G.; et al. Limestone biodeterioration: A review on the Portuguese cultural heritage scenario. J. Cult. Herit. 2019, 36, 275–285. [Google Scholar] [CrossRef]
- Mansch, R.; Bock, E. Biodeterioration of natural stone with special reference to nitrifying bacteria. Biodegradation 1998, 9, 47–64. [Google Scholar] [CrossRef] [PubMed]
- Dakal, T.C.; Arora, P.K. Evaluation of potential of molecular and physical techniques in studying biodeterioration. Rev. Environ. Sci. Biotechnol. 2012, 11, 71–104. [Google Scholar] [CrossRef]
- Otlewska, A.; Adamiak, J.; Gutarowska, B. Application of molecular techniques for the assessment of microorganism diversity on cultural heritage objects. Acta Biochim. Pol. 2014, 61, 217–225. [Google Scholar] [CrossRef]
- Bultreys, A.; Gheysen, I. Production and comparison of peptide siderophores from strains of distantly related pathovars of Pseudomonas syringae and Pseudomonas viridiflava LMG 2352. Appl. Environ. Microbiol. 2000, 66, 325–331. [Google Scholar] [CrossRef]
- Schwyn, B.; Neilands, J.B. Universal Chemical Assay for the Detection Determination of Siderophores. Anal. Biochem. 1987, 56, 47–56. [Google Scholar] [CrossRef]
- Fekete, F.A.; Chandhoke, V.; Jellison, J. Iron-binding compounds produced by wood- decaying basidiomycetes. Appl. Environ. Microbiol. 1989, 55, 2720–2722. [Google Scholar]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Gonzalez-Peña, A.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, R.H.; Larsson, K.H.; Taylor, A.F.S.; Bengtsson-Palme, J.; Jeppesen, T.S.; Schigel, D.; Kennedy, P.; Picard, K.; Glöckner, F.O.; Tedersoo, L.; et al. The UNITE database for molecular identification of fungi: Handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res. 2019, 47, D259–D264. [Google Scholar] [CrossRef] [PubMed]
- Directive 2008/50/EC of the European Parliament and of the Council of 21 May 2008 on ambient air quality and cleaner air for Europe. Off. J. Eur. Union 2008, 152, 1–44. Available online: https://eur-lex.europa.eu/eli/dir/2008/50/oj.
- Joutey, T.N.; Bahafid, W.; Sayel, H.; El Ghachtouli, N. Biodegradation: Involved Microorganisms and Genetically Engineered Microorganisms, 1st ed.; IntechOpen: London, UK, 2013; pp. 289–320. [Google Scholar]
- Perito, B.; Cavalieri, D. Innovative metagenomic approaches for detection of microbial communities involved in biodeteriorattion of cultural heritage. IOP Conf. Ser. Mater. Sci. Eng. 2018, 364, 012074. [Google Scholar] [CrossRef]
- Niesler, A.; Górny, R.L.; Wlazlo, A.; Ludzeń-Izbińska, B.; Lawniczek-Walczyk, A.; Golofit-Szymczak, M.; Meres, Z.; Kasznia-Kocot, J.; Harkawy, A.; Lis, D.O.; et al. Microbial contamination of storerooms at the Auschwitz-Birkenau Museum. Aerobiologia 2010, 26, 125–133. [Google Scholar] [CrossRef]
- Konhauser, K.O. Introduction to Geomicrobiology, 1st ed.; Blackwell Science Ltd.: Oxford, UK, 2007; pp. 196–203. ISBN 978-0-632-05454-1. [Google Scholar]
- Dakal, T.C.; Cameotra, S.S. Microbially induced deterioration of architectural heritages: Routes and mechanisms involved. Environ. Sci. Eur. 2012, 24, 1–13. [Google Scholar] [CrossRef]
- Drewniak, Ł.; Skłodowska, A.; Manecki, M.; Bajda, T. Solubilization of Pb-bearing apatite Pb5(PO4)3Cl by bacteria isolated from polluted environment. Chemosphere 2017, 171, 302–307. [Google Scholar] [CrossRef]
- Zanardini, E.; May, E.; Inkpen, R.; Cappitelli, F.; Murrell, J.C.; Purdy, K.J. Diversity of archaeal and bacterial communities on exfoliated sandstone from Portchester Castle (UK). Int. Biodeterior. Biodegrad. 2016, 109, 78–87. [Google Scholar] [CrossRef]
- Gutarowska, B.; Celikkol-Aydin, S.; Bonifay, V.; Otlewska, A.; Aydin, E.; Oldham, A.L.; Brauer, J.I.; Duncan, K.E.; Adamiak, J.; Sunner, J.A.; et al. Metabolomic and high-throughput sequencing analysis-modern approach for the assessment of biodeterioration of materials from historic buildings. Front. Microbiol. 2015, 6, 1–13. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, B.; He, Z.; Yang, X. Distribution and diversity of bacteria and fungi colonization in stone monuments analyzed by high-throughput sequencing. PLoS ONE 2016, 11, 1–17. [Google Scholar] [CrossRef]
- Chimienti, G.; Piredda, R.; Pepe, G.; van der Werf, I.D.; Sabbatini, L.; Crecchio, C.; Ricciuti, P.; D’Erchia, A.M.; Manzari, C.; Pesole, G. Profile of microbial communities on carbonate stones of the medieval church of San Leonardo di Siponto (Italy) by Illumina-based deep sequencing. Appl. Microbiol. Biotechnol. 2016, 100, 8537–8548. [Google Scholar] [CrossRef] [PubMed]
- Dyda, M.; Decewicz, P.; Romaniuk, K.; Wojcieszak, M.; Sklodowska, A.; Dziewit, L.; Drewniak, L.; Laudy, A. Application of metagenomic methods for selection of an optimal growth medium for bacterial diversity analysis of microbiocenoses on historical stone surfaces. Int. Biodeterior. Biodegrad. 2018, 131, 2–10. [Google Scholar] [CrossRef]
- Sun, W.; Yu, G.; Louie, T.; Liu, T.; Zhu, C.; Xue, G.; Gao, P. From mesophilic to thermophilic digestion: The transitions of anaerobic bacterial, archaeal, and fungal community structures in sludge and manure samples. Appl. Microbiol. Biotechnol. 2015, 99, 10271–10282. [Google Scholar] [CrossRef] [PubMed]
- Poszytek, K.; Pyzik, A.; Sobczak, A.; Lipinski, L.; Sklodowska, A.; Drewniak, L. The effect of the source of microorganisms on adaptation of hydrolytic consortia dedicated to anaerobic digestion of maize silage. Anaerobe 2017, 46, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Wojcieszak, M.; Pyzik, A.; Poszytek, K.; Krawczyk, P.S.; Sobczak, A.; Lipinski, L.; Roubinek, O.; Palige, J.; Sklodowska, A.; Drewniak, L. Adaptation of methanogenic inocula to anaerobic digestion of maize silage. Front. Microbiol. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.; Chen, S.; Zhao, Z.; Feng, J.; Zhang, Z.; Lu, K.; Jia, J. Water bacterial and fungal community compositions associated with urban lakes, Xi’an, China. Int. J. Environ. Res. Public Health 2018, 15, 469. [Google Scholar] [CrossRef]
- Douterelo, I.; Fish, K.E.; Boxall, J.B. Succession of bacterial and fungal communities within biofilms of a chlorinated drinking water distribution system. Water Res. 2018, 141, 74–85. [Google Scholar] [CrossRef]
- Zhang, Y.; Dong, S.; Gao, Q.; Liu, S.; Ganjurjav, H.; Wang, X.; Su, X.; Wu, X. Soil bacterial and fungal diversity differently correlated with soil biochemistry in alpine grassland ecosystems in response to environmental changes. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Wang, B.; Adachi, Y.; Sugiyama, S. Soil productivity and structure of bacterial and fungal communities in unfertilized arable soil. PLoS ONE 2018, 13, 1–17. [Google Scholar] [CrossRef]
- Ziganshina, E.E.; Mohammed, W.S.; Shagimardanova, E.I.; Vankov, P.Y.; Gogoleva, N.E.; Ziganshin, A.M. Fungal, Bacterial, and Archaeal Diversity in the Digestive Tract of Several Beetle Larvae (Coleoptera). Biomed. Res. Int. 2018, 2018, 1–15. [Google Scholar] [CrossRef]
- Yun, J.H.; Jung, M.J.; Kim, P.S.; Bae, J.W. Social status shapes the bacterial and fungal gut communities of the honey bee. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kemp, P.F.; Aller, J.Y. Bacterial diversity in aquatic and other environments: What 16S rDNA libraries can tell us. FEMS Microbiol. Ecol. 2004, 47, 161–177. [Google Scholar] [CrossRef]
- Orellana, S.C.; Leite, L.R.; Smith, A.; Medeiros, J.D.; Badotti, F.; Fonseca, P.L.; BM Vaz, A.; Oliveira, G.; Góes-Neto, A. Assessment of Fungal Diversity in the Environment using Metagenomics: A Decade in Review. Fungal Genomics Biol. 2013, 03, 1000110. [Google Scholar] [CrossRef]
- Laiz, L.; Piñar, G.; Lubitz, W.; Saiz-Jimenez, C. Monitoring the colonization of monuments by bacteria: Cultivation versus molecular methods. Environ. Microbiol. 2003, 5, 72–74. [Google Scholar] [CrossRef]
- Warscheid, T.; Braams, J. Biodeterioration of stone: A review. Int. Biodeterior. Biodegrad. 2000, 46, 343–368. [Google Scholar] [CrossRef]
- Saarela, M.; Alakomi, H.L.; Suihko, M.L.; Maunuksela, L.; Raaska, L.; Mattila-Sandholm, T. Heterotrophic microorganisms in air and biofilm samples from Roman catacombs, with special emphasis on actinobacteria and fungi. Int. Biodeterior. Biodegrad. 2004, 54, 27–37. [Google Scholar] [CrossRef]
- Suihko, M.L.; Alakomi, H.L.; Gorbushina, A.; Fortune, I.; Marquardt, J.; Saarela, M. Characterization of aerobic bacterial and fungal microbiota on surfaces of historic Scottish monuments. Syst. Appl. Microbiol. 2007, 30, 494–508. [Google Scholar] [CrossRef]
- Gutarowska, B.; Czyżowska, A. The ability of filamentous fungi to produce acids on indoor building materials. Ann. Microbiol. 2009, 59, 807–813. [Google Scholar] [CrossRef]
- Crook, B.; Burton, N.C. Indoor moulds, Sick Building Syndrome and building related illness. Fungal Biol. Rev. 2010, 24, 106–113. [Google Scholar] [CrossRef]
- Sterflinger, K.; Piñar, G. Microbial deterioration of cultural heritage and works of art - Tilting at windmills? Appl. Microbiol. Biotechnol. 2013, 97, 9637–9646. [Google Scholar] [CrossRef]
- Valentin, N. Microbial Contamination in Archives and Museums: Health Hazards and Preventative Strategies Using Air Ventilation Systems; The Getty Conservation Institute: Los Angeles, CA, USA, 2007; pp. 1–26. [Google Scholar]
- Mazzoli, R.; Giuffrida, M.G.; Pessione, E. Back to the past: “find the guilty bug—microorganisms involved in the biodeterioration of archeological and historical artifacts”. Appl. Microbiol. Biotechnol. 2018, 102, 6393–6407. [Google Scholar] [CrossRef] [PubMed]
- Byrd, J.J.; Xu, H.-S.; Colwell, R.R. Viable but nonculturable bacteria in drinking water. Appl. Environ. Microbiol. 1991, 57, 875–878. [Google Scholar] [PubMed]
- Davis, C. Enumeration of probiotic strains: Review of culture-dependent and alternative techniques to quantify viable bacteria. J. Microbiol. Methods 2014, 103, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Eom, H.J.; Park, C.; Jung, J.; Shin, B.; Kim, W.; Chung, N.; Choi, I.G.; Park, W. Calcium carbonate precipitation by Bacillus and sporosarcina strains isolated from concrete and analysis of the bacterial community of concrete. J. Microbiol. Biotechnol. 2016, 26, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Singh, P.; Lal, R. Genetics and Genomics of the Genus Amycolatopsis. Indian, J. Microbiol. 2016, 56, 233–246. [Google Scholar] [CrossRef]
- Sangal, V.; Goodfellow, M.; Blom, J.; Tan, G.Y.A.; Klenk, H.-P.; Sutcliffe, I.C. Revisiting the Taxonomic Status of the Biomedically and Industrially Important Genus Amycolatopsis, using a Phylogenomic Approach. Front. Microbiol. 2018, 9, 1–9. [Google Scholar] [CrossRef]
- Piñar, G.; Kraková, L.; Pangallo, D.; Piombino-Mascali, D.; Maixner, F.; Zink, A.; Sterflinger, K. Halophilic bacteria are colonizing the exhibition areas of the Capuchin Catacombs in Palermo, Italy. Extremophiles 2014, 18, 677–691. [Google Scholar] [CrossRef]
- Otlewska, A.; Adamiak, J.; Stryszewska, T.; Kańka, S.; Gutarowska, B. Factors Determining the Biodiversity of Halophilic Microorganisms on Historic Masonry Buildings. Microbes Environ. 2017, 32, 164–173. [Google Scholar] [CrossRef]
- Mitchell, R.; Gu, J.-D. Changes in the biofilm microflora of limestone caused by atmospheric pollutants. Int. Biodeterior. Biodegrad. 2000, 46, 299–303. [Google Scholar] [CrossRef]
- Jouzani, G.S.; Sharafi, R. New “Omics” Technologies and Biogas Production. In Biogas. Biofuel and Biorefinery Technologies, 1st ed.; Springer: Cham, Switzerland, 2018; Volume 6, pp. 419–436. [Google Scholar]



| Compound | April | June | August | October |
|---|---|---|---|---|
| carbon dioxide | 0.14 | 0,12 | 0.12 | 0.16 |
| nitric oxide (NO) | 0.00 | 0.25 | 5.27 | 0.00 |
| acetic acid | 0.00 | 0.00 | 0.06 | 1.75 |
| methane | 1.59 | 1.05 | 0.73 | 1.90 |
| sulfur dioxide | 0.00 | 0.00 | 0.00 | 0.95 |
| ammonia | 0.00 | 0.02 | 0.02 | 0.04 |
| carbonyl sulfide | 0.00 | 1.47 | 1.51 | 0.00 |
| hydrogen cyanide | 0.00 | 1.10 | 0.97 | 0.00 |
| cyclohexane | 0.07 | 0.26 | 0.21 | 0.34 |
| formaldehyde | 0.16 | 0.04 | 0.08 | 0.00 |
| benzene | 0.83 | 0.68 | 5.21 | 1.85 |
| toluene | 0.11 | 0.00 | 0.00 | 0.42 |
| m-xylene | 0.00 | 0.00 | 0.00 | 0.00 |
| acetone | 1.04 | 0.51 | 1.25 | 0.70 |
| methyl isobutyl ketone | 0.00 | 0.00 | 0.00 | 0.00 |
| ethane | 0.56 | 0.00 | 0.41 | 0.00 |
| ethanol | 0.87 | 1.28 | 0.85 | 0.73 |
| acetaldehyde | 0.50 | 0.41 | 0.51 | 1.82 |
| diethyl ether | 0.06 | 0.07 | 0.14 | 0.00 |
| 1,2,4-trimetylbenzene | 0.39 | 0.50 | 0.00 | 0.00 |
| octane | 0.00 | 0.48 | 0.54 | 0.33 |
| ethylene | 0.00 | 0.42 | 0.04 | 0.23 |
| 1,3-butadiene | 1.46 | 0.00 | 1.03 | 0.50 |
| styrene | 2.55 | 0.00 | 0.00 | 0.00 |
| cumene | 0.00 | 0.28 | 0.15 | 0.24 |
| Month | Temperature (°C) | Dew Point (°C) | Wet Bulb (°C) | Humidity (%) | PM 2.5 (µg/m3) | PM 10 (µg/m3) |
|---|---|---|---|---|---|---|
| April | 19.3 | 7.9 | 15.2 | 34.3 | 7 ± 0% | 21 ± 3% |
| June | 21.3 | 12 | 17.7 | 47.3 | 4 ± 12% | 20 ± 4% |
| August | 24.6 | 18.6 | 20.6 | 63 | 9 ± 7% | 22 ± 2% |
| October | 22.1 | 12.5 | 16.8 | 51.3 | 36 ± 4% | 86 ± 3% |
| Sample | April | June | August | October | |
|---|---|---|---|---|---|
| Sand-like/dust samples from ancient walls | Bacteria | Bacillus (20.2%) Pseudomonas (5.6%) Virgibacillus (3.2%) Crossiella (3.1%) Paenisporosarcina (3.1%) Truepera (2.7%) Arthrobacter (2.7%) Genera uncultured or unspecified (41.8%) | Bacillus (19.7%) Crossiella (9.8%) Amycolatopsis (5.5%) Paenisporosarcina (3.4%) Pseudomonas (3.3%) Streptomyces (3.2%) Virgibacillus (3.1%) Prauserella (2.9%) Arthrobacter (2.9%) Paenibacillus (2.1%) Planococcus (2.1%) Genera uncultured or unspecified (23.7%) | Bacillus (14.9%) Paenisporosarcina (14.0%) Crossiella (9.6%) Mesorhizobium (5.4%) Amycolatopsis (4.3%) Virgibacillus (4.0%) Prauserella (3.9%) Streptomyces (3.0%) Saccharopolyspora (2.2%) Genera uncultured or unspecified (15.6%) | Crossiella (14.5%) Amycolatopsis (10.9%) Bacillus (10.3%) Prauserella (5.2%) Acinetobacter (5.1%) Paenisporosarcina (3.1%) Genera uncultured or unspecified (27.3%) |
| Fungi | Aspergillus (8.8%) Genera unidentified or unspecified (67.3%) | Aspergillus (20.7%) Penicillium (7.0%) Phialosimplex (2.7%) Debaryomyces (2.5%) Genera unidentified or unspecified (44.3%) | Aspergillus (12.1%) Alternaria (2.6%) Genera unidentified or unspecified (62.9%) | Aspergillus (10.3%) Phialosimplex (4.3%) Leucosporidium (3.4%) Acremonium (2.1%) Genera unidentified or unspecified (56.5%) | |
| Air sampled onto agar plates | Bacteria | Bacillus (28.4%) Psychrobacter (19.9%) Pseudarthrobacter (16.2%) Micrococcus (14.5%) Arthrobacter (3.7%) Kocuria (3.4%) Staphylococcus (3.3%) Paracoccus (2.2%) Genera uncultured or unspecified (0.8%) | Bacillus (21.4%) Staphylococcus (19.4%) Enhydrobacter (10.9%) Acinetobacter (10.6%) Micrococcus (7.7%) Kocuria (5.5%) Genera uncultured or unspecified (23.2%) | Bacillus (37.3%) Micrococcus (20.1%) Staphylococcus (18.5%) Acinetobacter (8.2%) Exiguobacterium (5.7%) Massilia (5.3%) Genera uncultured or unspecified (0.2%) | Pseudomonas (16.8%) Corynebacterium 1 (13.8%) Micrococcus (12.2%) Acinetobacter (10.7%) Bacillus (8.4%) Lysinibacillus (5.8%) Staphylococcus (4.8%) Enterococcus (4.8%) Kocuria (2.5%) Microvirga (2.2%) Massilia (2.0%) Genera uncultured or unspecified (10.8%) |
| Fungi | Penicillium (41.9%) Beauveria (25.5%) Cladosporium (6.4%) Circinella (7.1%) Aspergillus (6.9%) Coprinellus (4.3%) Talaromyces (2.1%) Genera unidentified or unspecified (3.0%) | Penicillium (22.1%) Cladosporium (15.8%) Porostereum (11.8%) Talaromyces (6.1%) Aspergillus (5.3%) Bjerkandera (5.9%) Botrytis (4.5%) Paraconiothyrium (3.1%) Agrocybe (2.8%) Genera unidentified or unspecified (12.7%) | Penicillium (20.6%) Coprinellus (15.6%) Talaromyces (13.5%) Cladosporium (11.0%) Porostereum (10.5%) Coriolopsis (9.8%) Aspergillus (6.9%) Trametes (5.9%) Bjerkandera (2.7%) Alternaria (2.0%) Genera unidentified or unspecified (0.8%) | Trichoderma (43.7%) Penicillium (26.8%) Cladosporium (18.8%) Talaromyces (8.0%) Genera unidentified or unspecified (2.7%) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dyda, M.; Pyzik, A.; Wilkojc, E.; Kwiatkowska-Kopka, B.; Sklodowska, A. Bacterial and Fungal Diversity Inside the Medieval Building Constructed with Sandstone Plates and Lime Mortar as an Example of the Microbial Colonization of a Nutrient-Limited Extreme Environment (Wawel Royal Castle, Krakow, Poland). Microorganisms 2019, 7, 416. https://doi.org/10.3390/microorganisms7100416
Dyda M, Pyzik A, Wilkojc E, Kwiatkowska-Kopka B, Sklodowska A. Bacterial and Fungal Diversity Inside the Medieval Building Constructed with Sandstone Plates and Lime Mortar as an Example of the Microbial Colonization of a Nutrient-Limited Extreme Environment (Wawel Royal Castle, Krakow, Poland). Microorganisms. 2019; 7(10):416. https://doi.org/10.3390/microorganisms7100416
Chicago/Turabian StyleDyda, Magdalena, Adam Pyzik, Ewa Wilkojc, Beata Kwiatkowska-Kopka, and Aleksandra Sklodowska. 2019. "Bacterial and Fungal Diversity Inside the Medieval Building Constructed with Sandstone Plates and Lime Mortar as an Example of the Microbial Colonization of a Nutrient-Limited Extreme Environment (Wawel Royal Castle, Krakow, Poland)" Microorganisms 7, no. 10: 416. https://doi.org/10.3390/microorganisms7100416
APA StyleDyda, M., Pyzik, A., Wilkojc, E., Kwiatkowska-Kopka, B., & Sklodowska, A. (2019). Bacterial and Fungal Diversity Inside the Medieval Building Constructed with Sandstone Plates and Lime Mortar as an Example of the Microbial Colonization of a Nutrient-Limited Extreme Environment (Wawel Royal Castle, Krakow, Poland). Microorganisms, 7(10), 416. https://doi.org/10.3390/microorganisms7100416





