Gut Microbiota in Patients with Different Metabolic Statuses: Moscow Study
Abstract
1. Introduction
2. Materials and Methods
2.1. The Inclusion Criteria
2.2. The Exclusion Criteria
2.3. Ethical Aspects
2.4. Patients’ Preassessment
- Glucose metabolism impairment: fasting hyperglycemia (fasting venous plasma glucose ≥6.1 and <7.0 mmol/L) or impaired glucose tolerance (IGT) (fasting venous plasma glucose <7 mmol/L, ≥7.8 and <11.1 mg/dL 2 h after oral glucose tolerance test (OGTT)) or new onset DM-2 (fasting glucose ≥7 mmol/L or ≥11.1 mmol/L 2 h after OGTT);
- Lipid metabolism impairment: TC >5.0 mmol/L and/or LDL >3.0 mmol/L and/or HDL <1.0 mmol/L for men and <1.2 mmol/L for women and/or triglycerides >1.7 mmol/L;
- Body Mass Index (BMI) ≥ 30 kg/m2 (obesity) and/or waist circumference ≥94 cm for men and ≥80 cm for women (abdominal obesity);
- Arterial hypertension (AH) (systolic/diastolic blood pressure (BP) level of 140/90–159/99 mmHg). Patients with BP 160/100 mmHg and higher and those who required antihypertensive treatment were not included;
- Smoking (regardless of the numbers of cigarettes and years of smoking).
2.5. Basic Methods
2.5.1. Gut Microbiota Composition
2.5.2. Library Preparation
2.5.3. Blood Vessels Assessment
2.6. Bioinformatic Processing
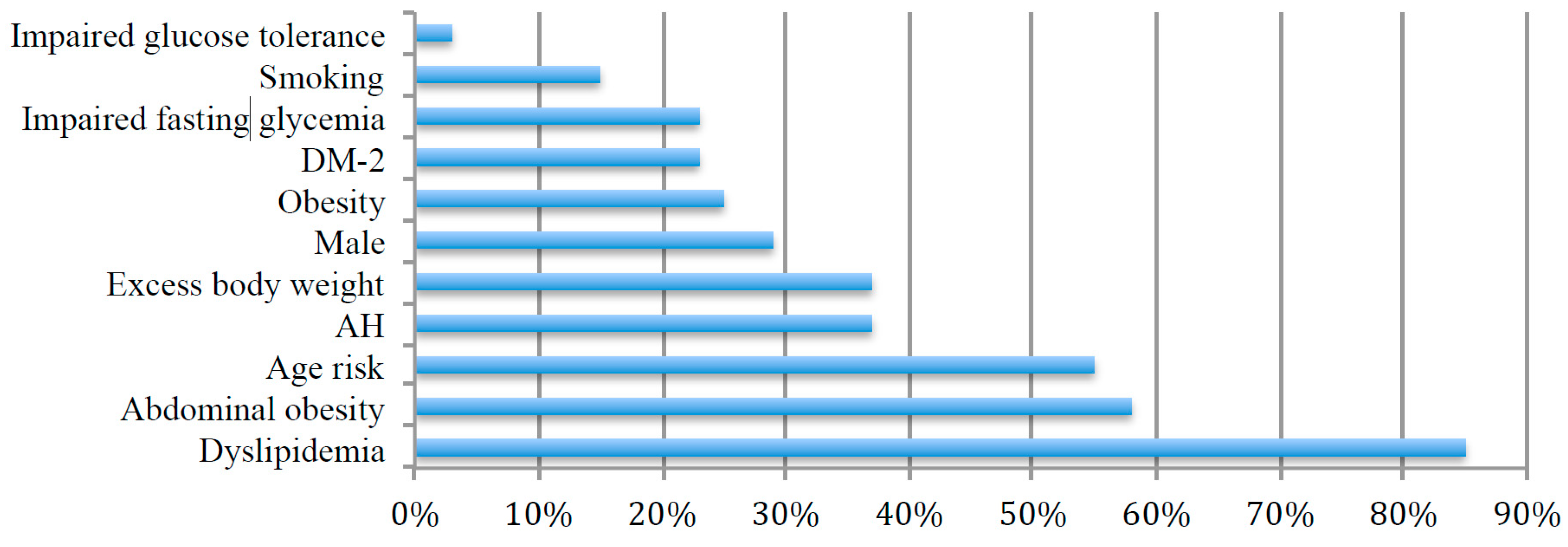
3. Results
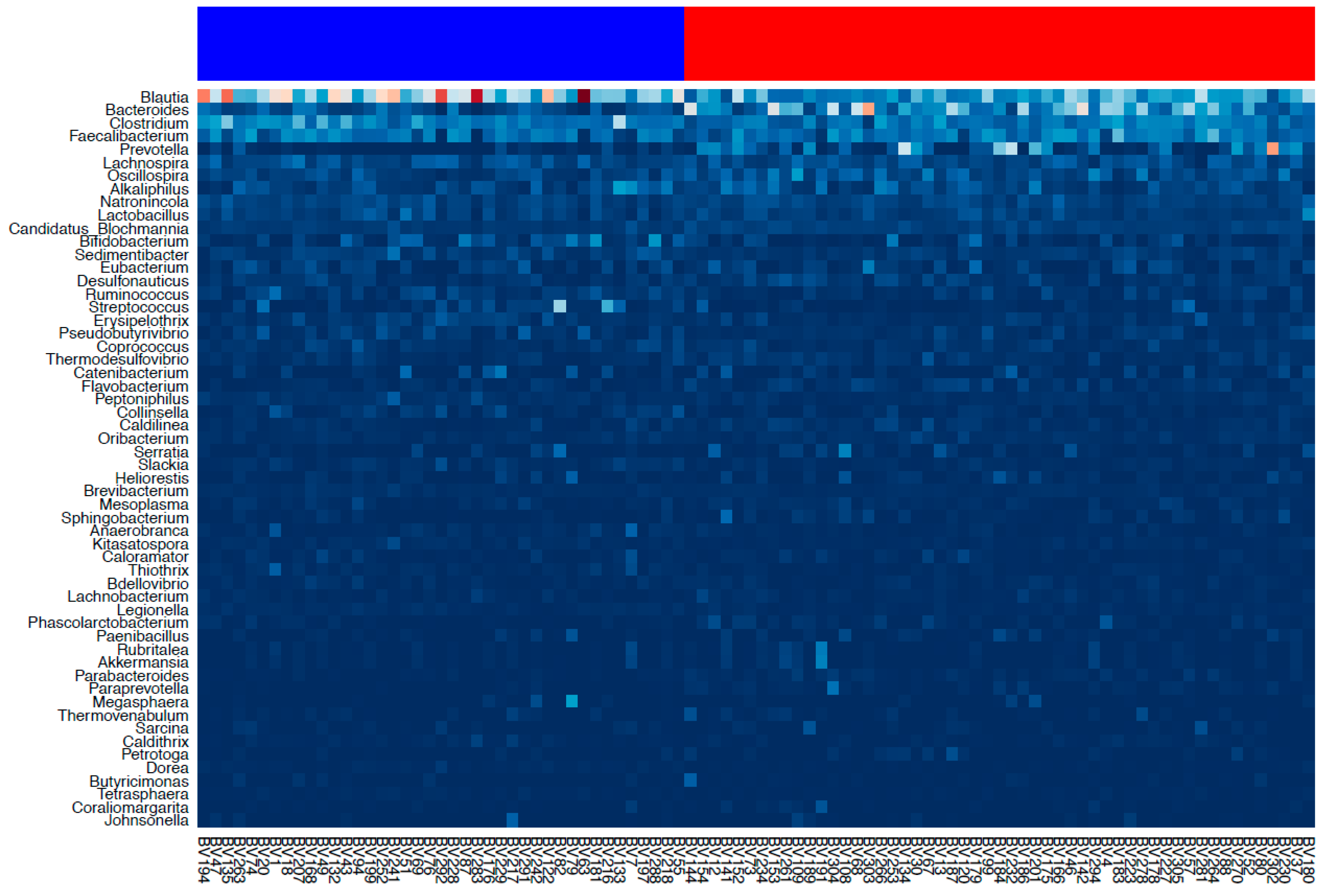
3.1. Gut Microbiota Assessment
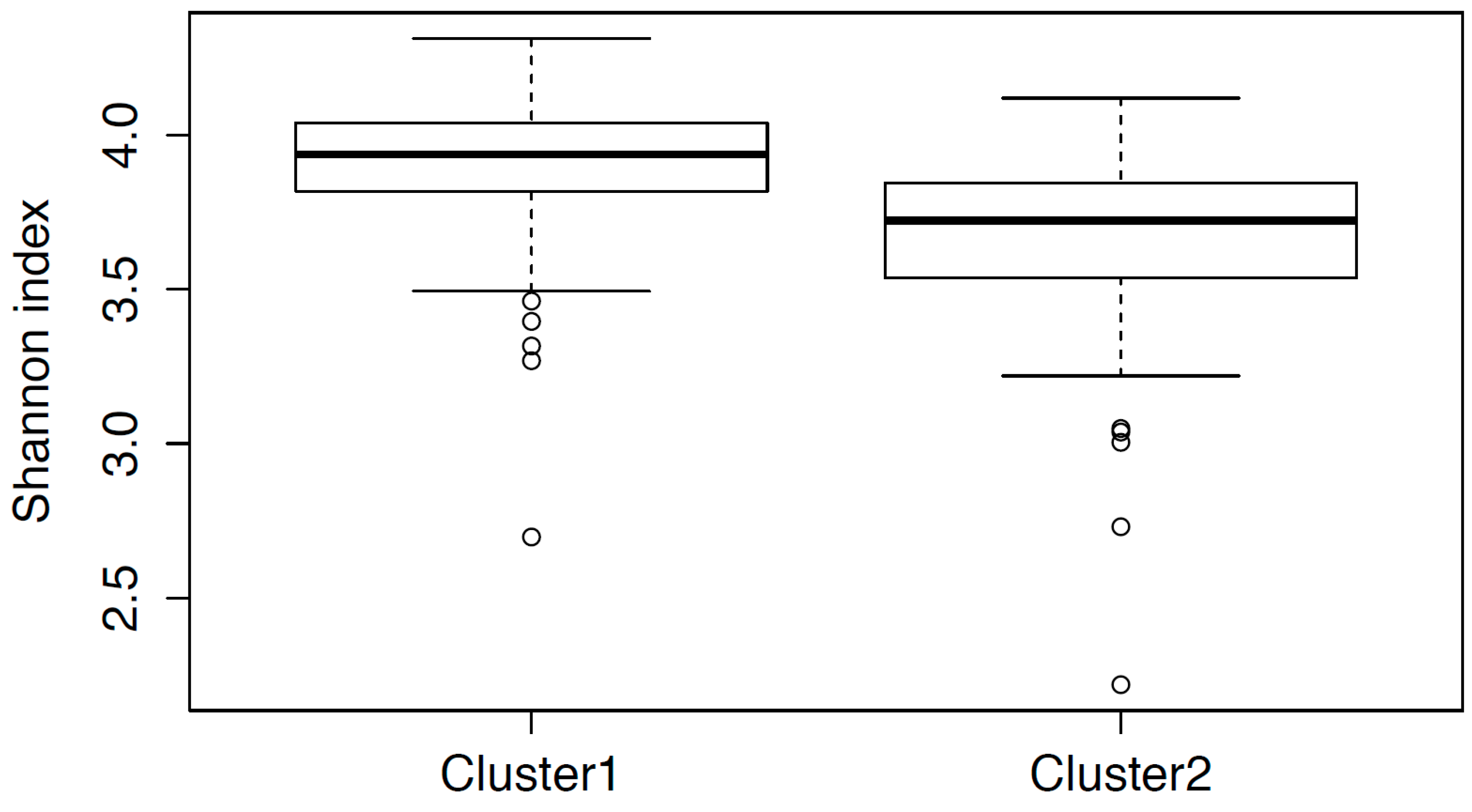
3.2. Cluster Analysis
3.3. Cardiovascular Risk Factors and Gut Microbiota Composition
3.3.1. Obesity and Gut Microbiota
3.3.2. Lipids and Gut Microbiota
3.3.3. Glucose Metabolism and Gut Microbiota
3.3.4. First Grade Hypertension and Gut Microbiota
3.3.5. Smoking and Gut Microbiota
3.3.6. Diet and Gut Microbiota
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Sekirov, I.; Russell, S.L.; Antunes, L.C.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; West, N.P.; Cripps, A.W. Obesity, inflammation, and the gut microbiota. Lancet Diabetes Endocrinol. 2015, 3, 207–215. [Google Scholar] [CrossRef]
- Stein, J.H.; Korcarz, C.E.; Hurst, R.T.; Lonn, E.; Kendall, C.B.; Mohler, E.R.; Post, W.S. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: A consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. Endorsed by the Society for Vascular Medicine. J. Am. Soc. Echocardiogr. 2008, 21, 93–111. [Google Scholar] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Huttley, G.A. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Lu, N.; Feng, C.; Thurston, S.W.; Xia, Y.; Zhu, L.; Tu, X.M. On fitting generalized linear mixed-effects models for binary responses using different statistical packages. Stat. Med. 2011, 30, 2562–2572. [Google Scholar] [CrossRef] [PubMed]
- Egshatyan, L.; Kashtanova, D.; Popenko, A.; Tkacheva, O.; Tyakht, A.; Alexeev, D.; Boytsov, S. Gut microbiota and diet in patients with different glucose tolerance. Endocr. Connect. 2016, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Bertalan, M. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Xu, S. Principles of Statistical Genomics, 1st ed.; Springer: New York, NY, USA, 2013; p. XVI, 428. [Google Scholar]
- Kida, Y.; Inoue, H.; Shimizu, T.; Kuwano, K. Serratia marcescens serralysin induces inflammatory responses through protease-activated receptor 2. Infect. Immun. 2007, 75, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Leite, A.Z.; Rodrigues, N.C.; Gonzaga, M.I.; Paiolo, J.C.C.; de Souza, C.A.; Stefanutto, N.A.V.; Mariano, V.S. Detection of Increased Plasma Interleukin-6 Levels and Prevalence of Prevotella copri and Bacteroides vulgatus in the Feces of Type 2 Diabetes Patients. Front. Immunol. 2017, 8, 1107. [Google Scholar] [CrossRef] [PubMed]
- Tuovinen, E.; Keto, J.; Nikkila, J.; Matto, J.; Lahteenmaki, K. Cytokine response of human mononuclear cells induced by intestinal Clostridium species. Anaerobe 2013, 19, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Klimenko, N.S.; Tyakht, A.V.; Popenko, A.S.; Vasiliev, A.S.; Altukhov, I.A.; Ischenko, D.S.; Musienko, S.V. Microbiome Responses to an Uncontrolled Short-Term Diet Intervention in the Frame of the Citizen Science Project. Nutrients 2018, 10, 576. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Darveau, R.P.; Curtis, M.A. The keystone-pathogen hypothesis. Nat. Rev. Microbiol. 2012, 10, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Tims, S.; Derom, C.; Jonkers, D.M.; Vlietinck, R.; Saris, W.H.; Kleerebezem, M.; Zoetendal, E.G. Microbiota conservation and BMI signatures in adult monozygotic twins. ISME J. 2013, 7, 707–717. [Google Scholar] [CrossRef] [PubMed]
| Genus | Mann–Whitney U Test, FDR Adjusted p-Value | |
|---|---|---|
| 1 | Prevotella * | 0.0001 |
| 2 | Oscillospira * | 0.005 |
| 3 | Ruminococcus | 0.008 |
| 4 | Flavobacterium * | 0.005 |
| 5 | Peptoniphilus | 0.005 |
| 6 | Sphingobacterium * | 0.0003 |
| 7 | Thiothrix | 0.005 |
| 8 | Legionella | 0.005 |
| 9 | Parabacteroides * | 0.002 |
| Genus | Estimate | Standart Error | z Value | FDR | |
|---|---|---|---|---|---|
| Obesity | Serratia | 0.64 | 0.17 | 3.85 | 0.003 |
| Obesity | Prevotella | 0.56 | 0.12 | 4.54 | 0.0003 |
| Abdominal obesity | Prevotella | 0.49 | 0.12 | 4.19 | 0.0008 |
| Abdominal obesity | Oscillospira | −0.51 | 0.11 | −4.44 | 0.0005 |
| Abdominal obesity | Serratia | 1.20 | 0.33 | 3.67 | 0.004 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kashtanova, D.A.; Tkacheva, O.N.; Doudinskaya, E.N.; Strazhesko, I.D.; Kotovskaya, Y.V.; Popenko, A.S.; Tyakht, A.V.; Alexeev, D.G. Gut Microbiota in Patients with Different Metabolic Statuses: Moscow Study. Microorganisms 2018, 6, 98. https://doi.org/10.3390/microorganisms6040098
Kashtanova DA, Tkacheva ON, Doudinskaya EN, Strazhesko ID, Kotovskaya YV, Popenko AS, Tyakht AV, Alexeev DG. Gut Microbiota in Patients with Different Metabolic Statuses: Moscow Study. Microorganisms. 2018; 6(4):98. https://doi.org/10.3390/microorganisms6040098
Chicago/Turabian StyleKashtanova, Daria A., Olga N. Tkacheva, Ekaterina N. Doudinskaya, Irina D. Strazhesko, Yulia V. Kotovskaya, Anna S. Popenko, Alexander V. Tyakht, and Dmitry G. Alexeev. 2018. "Gut Microbiota in Patients with Different Metabolic Statuses: Moscow Study" Microorganisms 6, no. 4: 98. https://doi.org/10.3390/microorganisms6040098
APA StyleKashtanova, D. A., Tkacheva, O. N., Doudinskaya, E. N., Strazhesko, I. D., Kotovskaya, Y. V., Popenko, A. S., Tyakht, A. V., & Alexeev, D. G. (2018). Gut Microbiota in Patients with Different Metabolic Statuses: Moscow Study. Microorganisms, 6(4), 98. https://doi.org/10.3390/microorganisms6040098







