Rhizophagy Cycle: An Oxidative Process in Plants for Nutrient Extraction from Symbiotic Microbes
Abstract
1. Introduction
2. The Symbiotic Bacteria
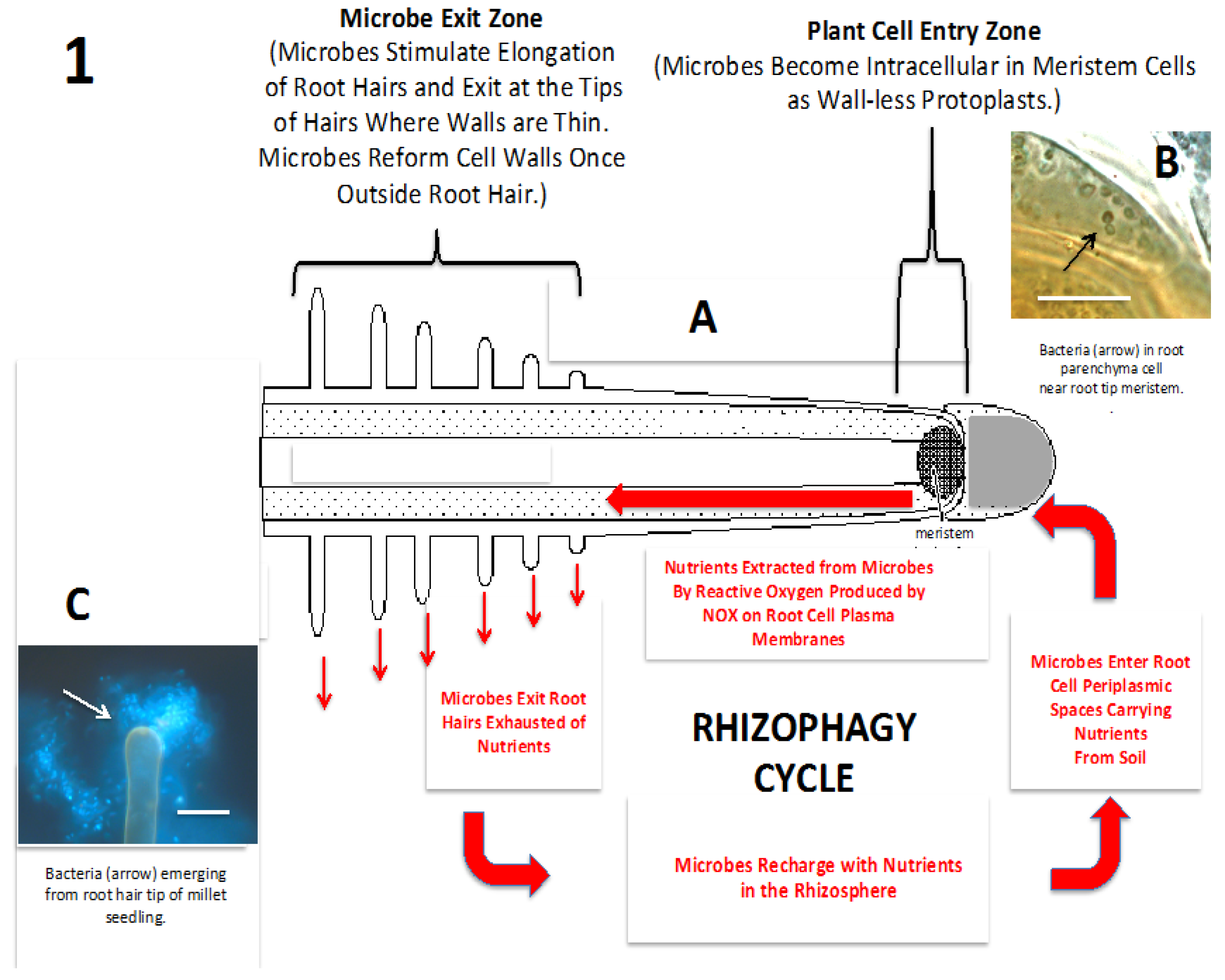
3. Plant ‘Farming’ of Rhizosphere Microbes
4. Nutrients Transferred to the Host Plant
5. Rhizophagy Microbes as Carriers of Micronutrients
6. Balance between Microbe-Oxidation Susceptibility and Resistance
7. Mechanisms for Microbe Entry into Roots
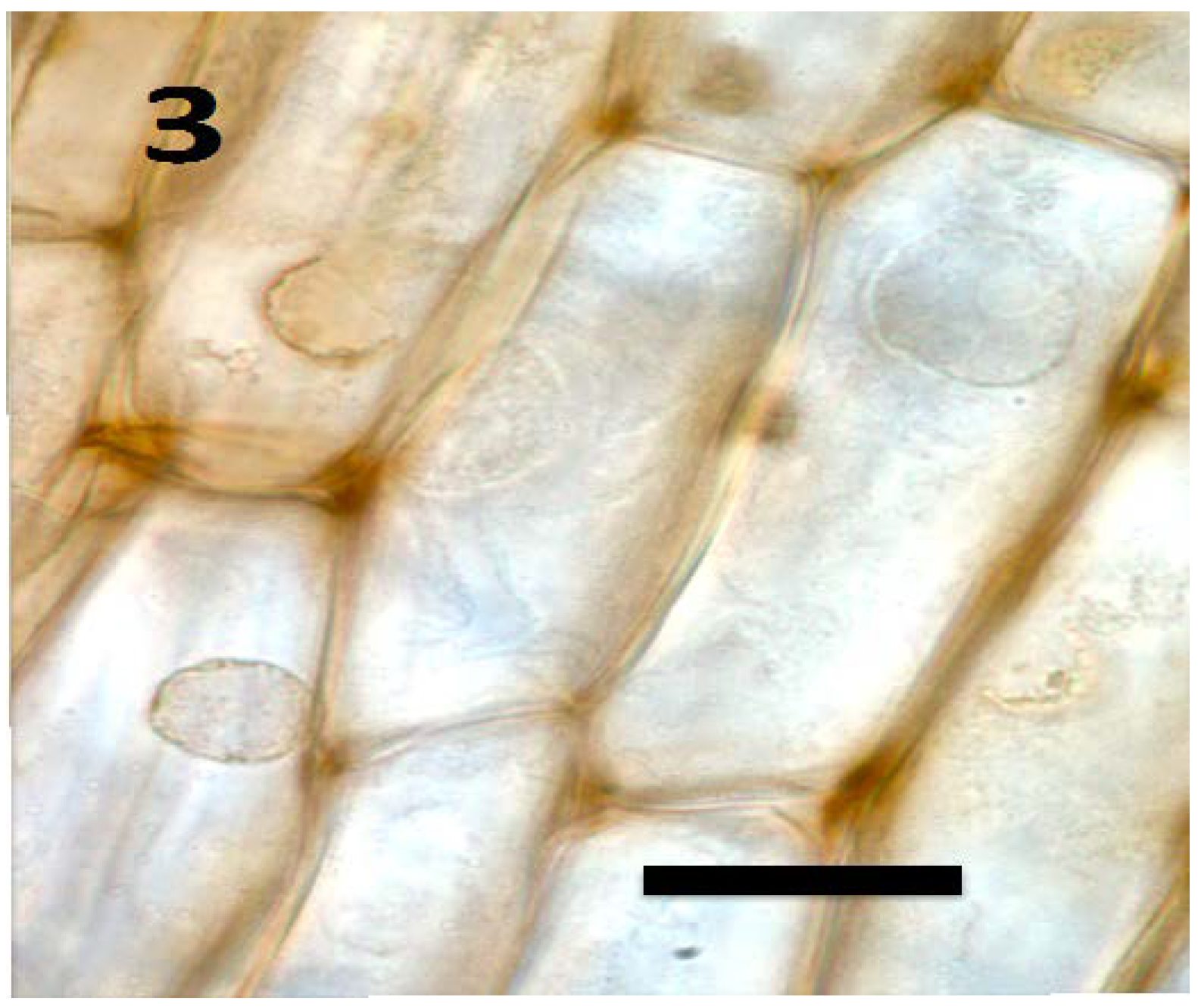
7.1. Endocytosis Hypothesis to Explain Microbe Entry into Root Meristem Cells
7.2. Hypothesized Role of Short-Chain Fatty Acids in Induction of Bacteria to Enter into Root Meristem Cells
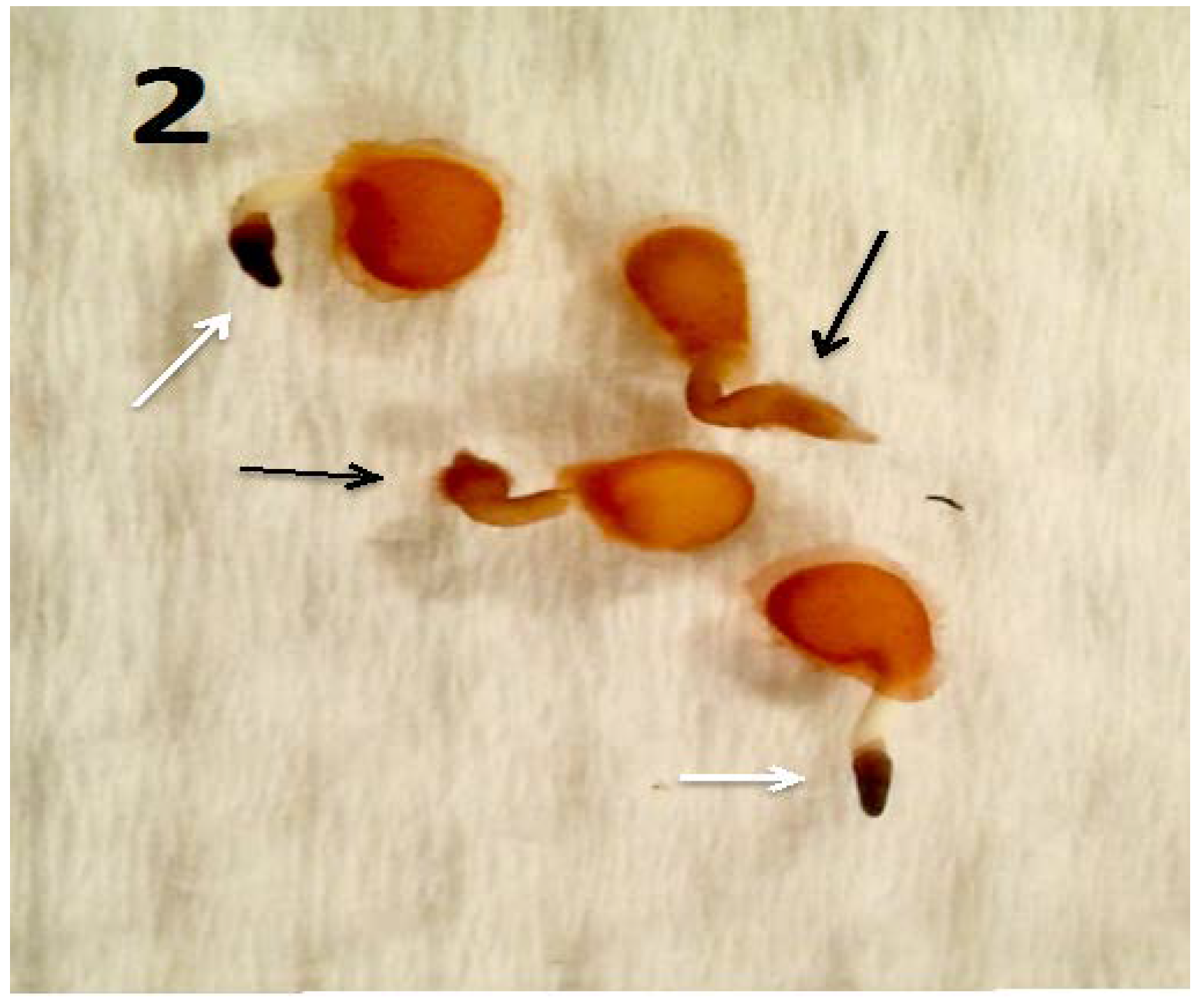
7.3. Propionic Acid and Butyric Acid Experiment to Test SCFA Removal-Induced Infection of Meristem Cells
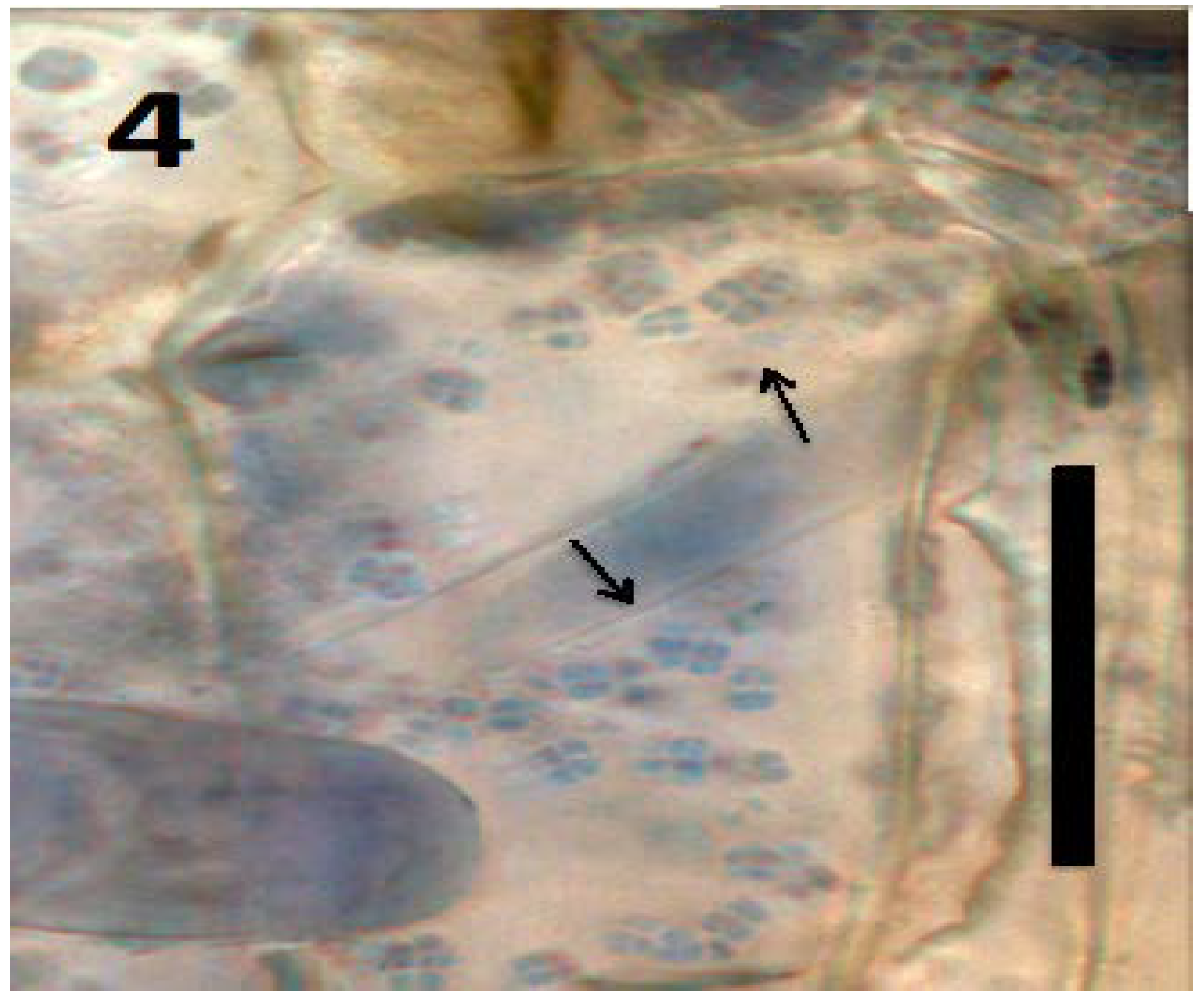
8. Loss of Cell Walls by Bacteria on Entry into the Periplasmic Space of Root Cells
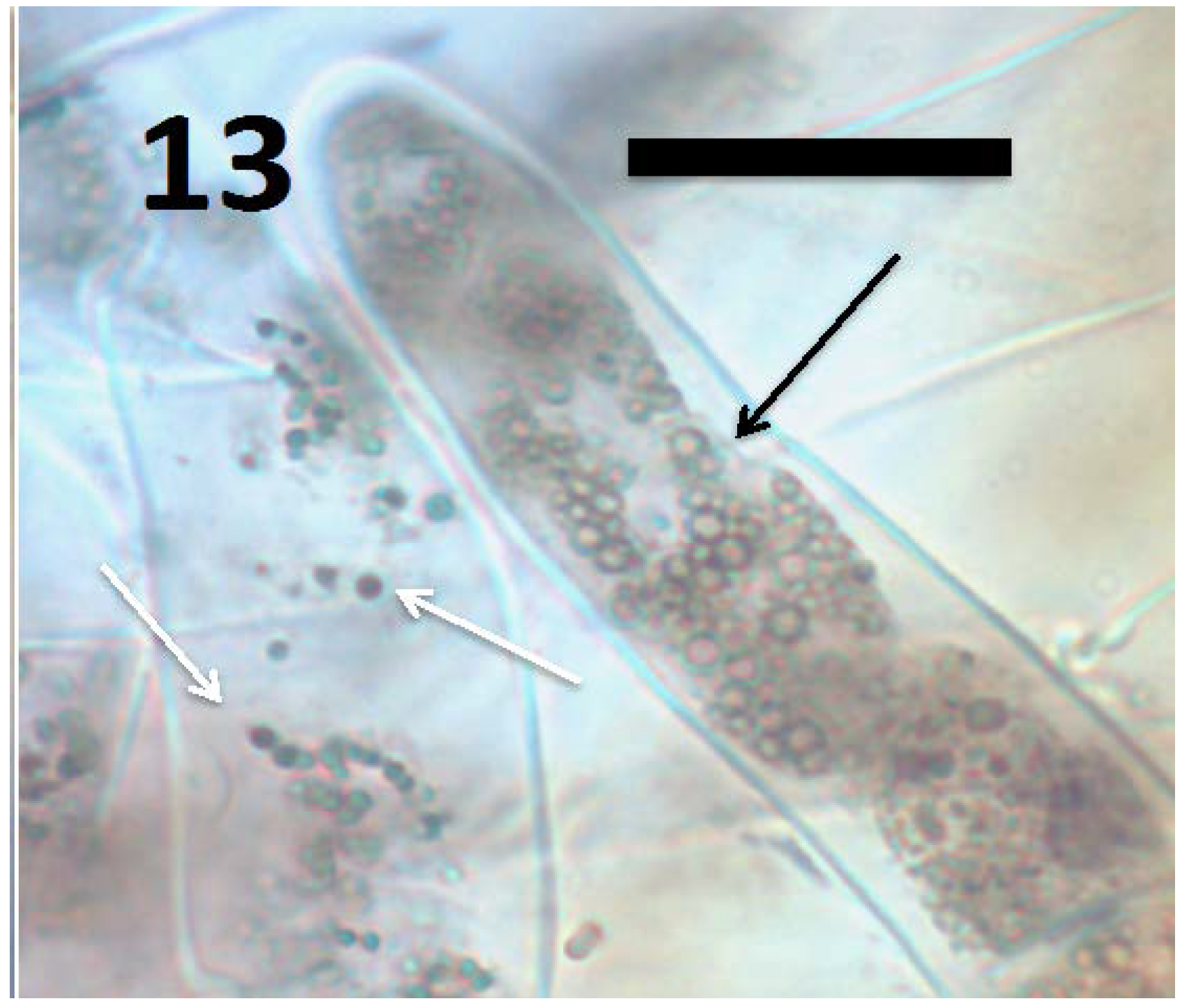
9. Intracellular Bacteria Exit Roots to Re-Enter Soil Populations
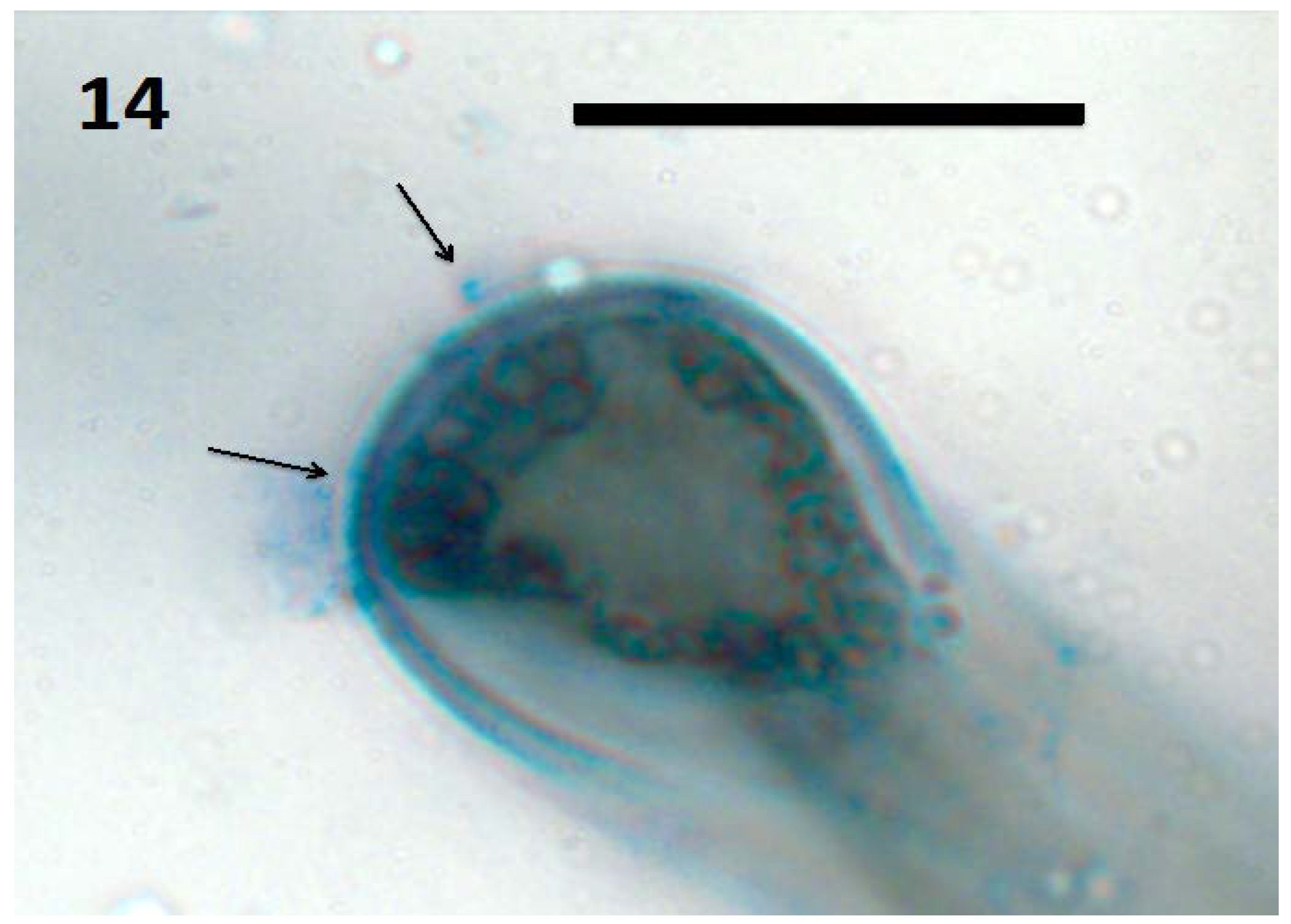
10. Do Fungi Also Function in the Rhizophagy Cycle?
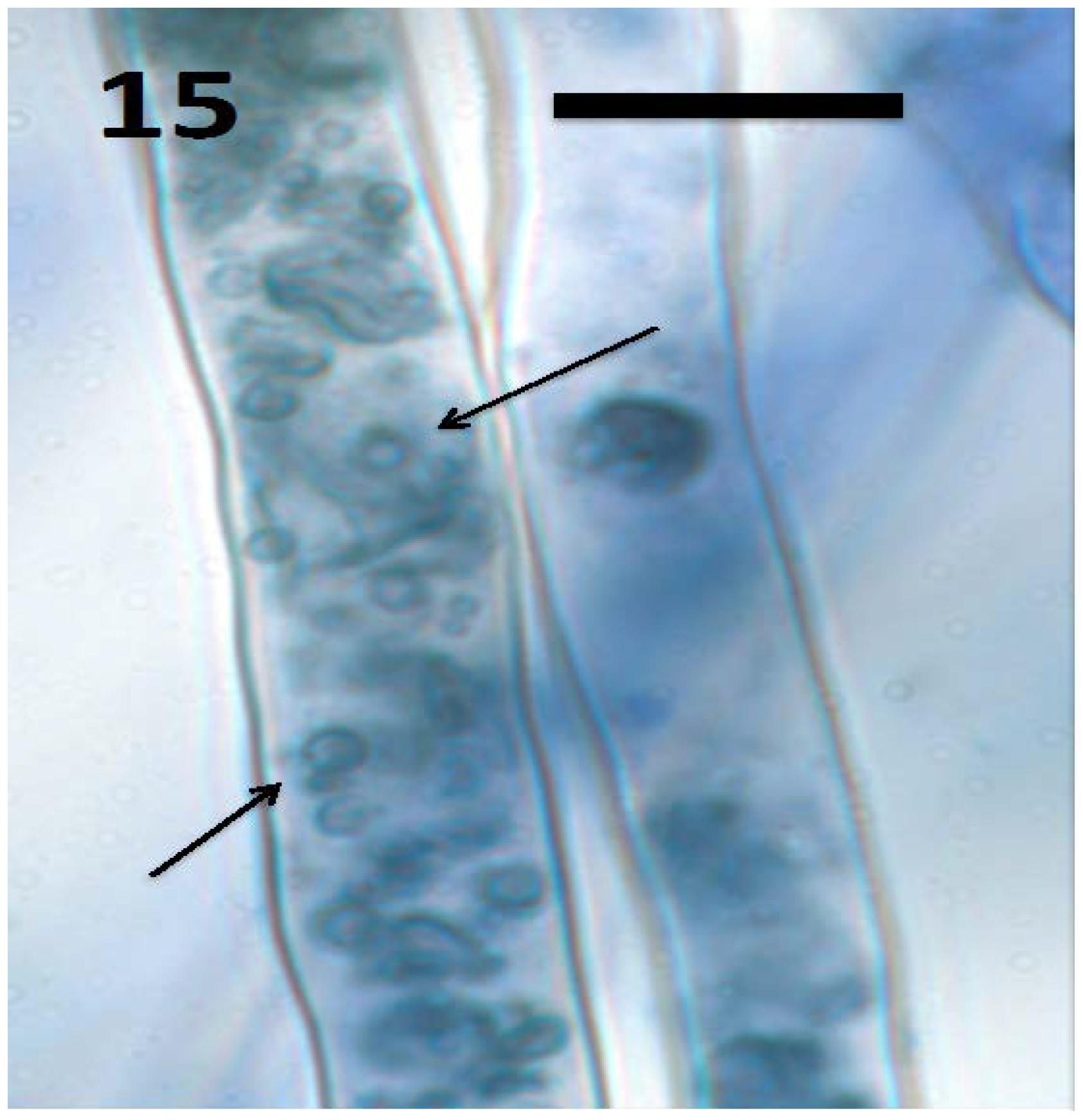
11. Non-Nutritive Functions of Rhizophagy Microbes
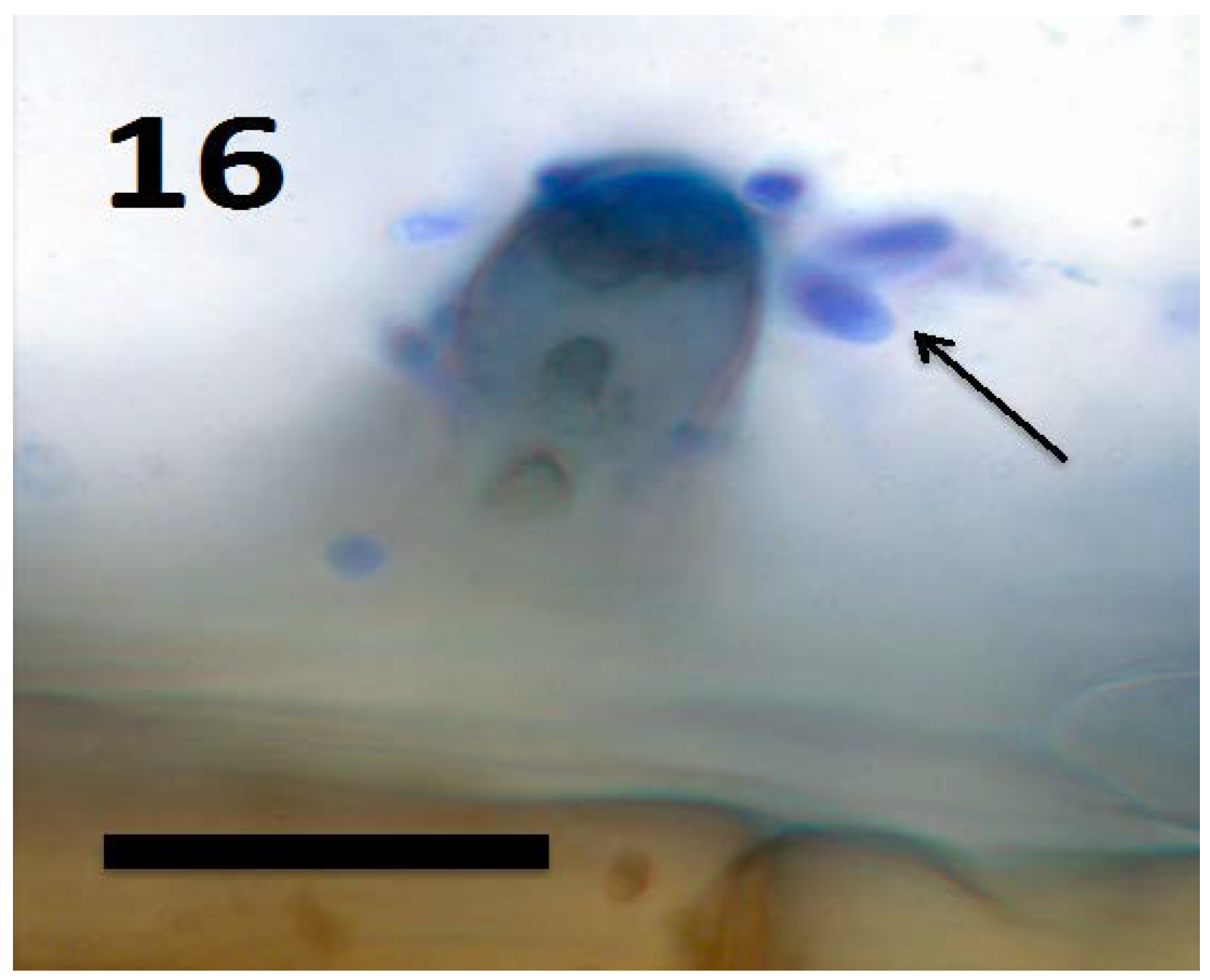
11.1. Modulation of Plant Development
11.2. Enhancement in Oxidative Stress Tolerance in Host Plants
11.3. Enhanced Disease Resistance Due to Endophytic Microbes
11.4. Endophyte-Mediated Suppression of Competitor Plant Species
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Manetas, Y. Alice in the Land of Plants: Biology of Plants and Their Importance for Planet; Springer: New York, NY, USA, 2012; p. 374. [Google Scholar]
- Pawlowski, K.; Demchenko, K.N. The diversity of actinorhizal symbiosis. Protoplasma 2012, 249, 967–979. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Hidalgo, P.; Hirsch, A.M. The nodule microbiome: N2-fixing rhizobia do not live alone. Phytobiomes 2017, 1, 70–82. [Google Scholar] [CrossRef]
- Santi, C.; Bogusz, D.; Franche, C. Biological nitrogen fixation in non-legume plants. Ann. Bot. 2013, 111, 743–767. [Google Scholar] [CrossRef] [PubMed]
- Coba de la Peña, T.; Fedorova, E.; Pueyo, J.J.; Lucas, M.M. The Symbiosome: Legume and Rhizobia Co-evolution toward a Nitrogen-Fixing Organelle? Front. Plant Sci. 2017, 8, 2229. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.M.; Kobayashi, H.; Davies, B.W.; Taga, M.E.; Walker, G.C. How rhizobial symbionts invade plants: The Sinorhizobium–Medicago model. Nat. Rev. Microbiol. 2007, 5, 619–633. [Google Scholar] [CrossRef] [PubMed]
- James, E.K.; Olivares, F.L. Infection and colonization of sugar cane and other graminaceous plants by endophytic diazotrophs. Crit. Rev. Plant Sci. 1998, 17, 77–119. [Google Scholar] [CrossRef]
- Schulz, B.; Römmert, A.K.; Dammann, U.; Aust, H.J.; Strack, D. The endophyte-host interaction: A balanced antagonism? Mycol. Res. 1999, 103, 1275–1283. [Google Scholar] [CrossRef]
- Hurek, T.; Handley, L.L.; Reinhold-Hurek, B.; Piché, Y. Azoarcus grass endophytes contribute fixed nitrogen to the plant in an unculturable state. Mol. Plant Microbe Interact. 2002, 15, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Compant, S.; Duffy, B.; Nowak, J.; Clément, C.; Barka, E.A. Use of plant growth-promoting bacteria for biocontrol of plant diseases: Principles, mechanisms of action, and future prospects. Appl. Environ. Microbiol. 2005, 71, 4951–4959. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Reddy, K.M. Microscopic elucidation of abundant endophytic bacteria colonizing the cell wall-plasma membrane peri-space in the shoot-tip tissue of banana. AoB Plants 2013, 5, plt011. [Google Scholar] [CrossRef]
- Thomas, P.; Soly, T.A. Endophytic bacteria associated with growing shoot tips of banana (Musa sp.) cv. Grand Naine and the affinity of endophytes to the host. Microb. Ecol. 2009, 58, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Johnston-Monje, D.; Raizada, M.N. Conservation and diversity of seed associated endophyes in Zea across boundaries of evolution, ethnography and ecology. PLoS ONE 2011, 6, e20396. [Google Scholar] [CrossRef] [PubMed]
- Hardoim, P.R.; van Overbeek, L.S.; Berg, G.; Pirttilä, A.M.; Compant, S.; Campisano, A.; Döring, M.; Sessitsch, A. The hidden world within plants: Ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol. Rev. 2015, 79, 293–320. [Google Scholar] [CrossRef] [PubMed]
- Holland, M.A. Probiotics for Plants? What the PPFMs told us and some ideas about how to use them. J. Wash Acad. Sci. 2016, 102, 31. [Google Scholar]
- Herrera, S.D.; Grossi, C.; Zawoznik, M.; Groppa, M.D. Wheat seeds harbour bacterial endophytes with potential as plant growth promoters and biocontrol agents of Fusarium graminearum. Microbiol. Res. 2016, 186–187, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Shehata, H.R.; Dumigan, C.; Watts, S.; Raizada, M.N. An endophytic microbe from an unusual volcanic swamp corn seeks and inhabits root hair cells to extract rock phosphate. Sci. Rep. 2017, 7, 1347. [Google Scholar] [CrossRef] [PubMed]
- Kandel, S.L.; Joubert, P.M.; Doty, L.S. Bacterial endophyte colonization and distribution within plants. Microorganisms 2017, 5, 77. [Google Scholar] [CrossRef] [PubMed]
- Redman, R.S.; Sheehan, K.B.; Stout, R.G.; Rodriguez, R.J.; Henson, J.M. Thermotolerance generated by plant/fungal symbiosis. Science 2002, 298, 1581. [Google Scholar] [CrossRef] [PubMed]
- Waller, F.; Achatz, B.; Baltruschat, H.; Fodor, J.; Becker, K.; Fisher, M.; Heier, T.; Huckelhoven, R.; Neumann, C.; Wettstein, D.; et al. The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc. Natl. Acad. Sci. USA 2005, 102, 13386–13391. [Google Scholar] [CrossRef] [PubMed]
- Kuldau, G.; Bacon, C.W. Clavicipitaceous endophytes: Their ability to enhance grass resistance to multiple stresses. Biol. Control 2008, 46, 57–71. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; Woodward, C.; Kim, Y.O.; Redman, R.S. Habitat-Adapted Symbiosis as a Defense against Abiotic and Biotic Stresses; White, J.F., Torres, M.S., Eds.; CRC Press: Boca Raton, FL, USA, 2009; Defensive Mutualism in Microbial Symbiosis; pp. 335–346. [Google Scholar]
- Sessitsch, A.; Hardoim, P.; Doering, J.; Weilharter, A.; Krause, A.; Woyke, T.; Mitter, B.; Hauberg-Lotte, L.; Friedrich, F.; Rahalkar, M.; et al. Functional characteristics of an endophyte community colonizing rice roots as revealed by metagenomic analysis. Mol. Plant Microbe Interact. 2012, 25, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Fürnkranz, M.; Lukesch, B.; Müller, H.; Huss, H.; Grube, M.; Berg, G. Microbial diversity inside pumpkins: Microhabitat-specific communities display a high antagonistic potential against phytopathogens. Microb. Ecol. 2012, 63, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Gond, S.K.; Bergen, M.S.; Torres, M.S.; White, J.F. Endophytic Bacillus spp. produce antifungal lipopeptides and induce host defence gene expression in maize. Microbiol. Res. 2014, 172, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Gond, S.K.; Bergen, M.; Torres, M.S.; White, J.F. Effect of bacterial endophyte on expression of defense genes in Indian popcorn against Fusarium moniliforme. Symbiosis 2015, 66, 133–140. [Google Scholar] [CrossRef]
- Soares, M.A.; Li, H.; Bergen, M.; White, J.F. Functional role of an endophytic Bacillus amyloliquefaciens in enhancing growth and disease protection of invasive English ivy (Hedera helix L.). Plant Soil 2015, 405, 107–123. [Google Scholar] [CrossRef]
- Soares, M.A.; Li, H.Y.; Kowalski, K.P.; Bergen, M.; Torres, M.S.; White, J.F. Functional roles of bacteria from invasive Phragmites australis in promotion of host growth. Microb. Ecol. 2016, 72, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Lata, R.; Choudhury, S.; Gond, S.K.; White, J.F. Induction of abiotic stress tolerance in plants by endophytic microbes. Lett. Appl. Microbiol. 2018, 66, 268–276. [Google Scholar] [CrossRef] [PubMed]
- James, E.K. Nitrogen fixation in endophytic and associative symbiosis. Field Crops Res. 2000, 65, 197–209. [Google Scholar] [CrossRef]
- Roley, S.S.; Duncan, D.S.; Liang, D.; Garoutte, A.; Jackson, R.D.; Tiedje, J.M.; Philip Robertson, G. Associative nitrogen fixation (ANF) in switchgrass (Panicum virgatum) across a nitrogen input gradient. PLoS ONE 2018, 13, e0197320. [Google Scholar] [CrossRef] [PubMed]
- Hurek, T.; Reinhold, B.; Grimm, B.; Fendrik, I.; Niemann, E.G. Occurrence of effective nitrogen scavenging bacteria in the rhizosphere of kallar grass. Plant Soil 1988, 110, 339–348. [Google Scholar] [CrossRef]
- Glick, B.R. The enhancement of plant-growth by free-living bacteria. Can. J. Microbiol. 1995, 41, 109–117. [Google Scholar] [CrossRef]
- Holland, M.A. Occam’s razor applied to hormonology. Are cytokinins produced by plants? Plant Physiol. 1997, 115, 865–868. [Google Scholar] [CrossRef] [PubMed]
- Sergeeva, E.; Hirkala, D.L.M.; Nelson, L.M. Production of indole-3-acetic acid, aromatic amino acid aminotransferase activities and plant growth promotion by Pantoea agglomerans rhizosphere isolates. Plant Soil 2007, 297, 1–13. [Google Scholar] [CrossRef]
- Long, H.H.; Schmidt, D.D.; Baldwin, I.T. Native bacterial endophytes promote host growth in a species-specific manner; Phytohormone manipulations do not result in common growth responses. PLoS ONE 2008, 3, e2702. [Google Scholar] [CrossRef] [PubMed]
- Castanheira, N.; Dourado, A.C.; Alves, P.I.; Cortés-Pallero, A.; Delgado-Rodríguez, A.I.; Prazeres, A.; Borges, N.; Sánchez, C. Annual ryegrass—associated bacteria with potential for plant growth promotion. Microbiol. Res. 2014, 169, 768–779. [Google Scholar] [CrossRef] [PubMed]
- Bacon, C.W.; White, J.F. Functions, mechanisms and regulation of endophytic and epiphytic microbial communities of plants. Symbiosis 2016, 68, 87–98. [Google Scholar] [CrossRef]
- Paungfoo-Lonhienne, C.; Rentsch, D.; Robatzrk, S.; Webb, R.I.; Sagulenko, E.; Nasholm, T.; Schmidt, S.; Lonhienne, T.G.A. Turning the table: Plants consume microbes as a source of nutrients. PLoS ONE 2010, 5, e11915. [Google Scholar] [CrossRef] [PubMed]
- Paungfoo-Lonhienne, C.; Schmidt, S.; Webb, R.; Lonhienne, T. Molecular Microbial Ecology of the Rhizosphere; Wiley-Blackwell: Hoboken, NJ, USA, 2013; pp. 1199–1207. [Google Scholar]
- Adamczyk, B.; Smolander, A.; Kitunen, V.; Godlewski, M. Proteins as nitrogen source for plants: A short story about exudation of proteases by plant roots. Plant Signal. Behav. 2010, 5, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Paungfoo-Lonhienne, C.; Lonhienne, T.G.A.; Rentsch, D.; Robinson, N.; Christie, M.; Webb, R.I.; Gamage, H.K.; Carroll, B.J.; Schenk, P.M.; Schmidt, S. Plants can use protein as a nitrogen source without assistance of other organisms. Proc. Natl. Acad. Sci. USA 2008, 105, 4524–4529. [Google Scholar] [CrossRef] [PubMed]
- White, J.F.; Crawford, H.; Torres, M.S.; Mattera, R.; Irizarry, I.; Bergen, M. A proposed mechanism for nitrogen acquisition by grass seedlings through oxidation of symbiotic bacteria. Symbiosis 2012, 57, 161–171. [Google Scholar] [CrossRef] [PubMed]
- White, J.F.; Torres, M.S.; Somu, M.P.; Johnson, H.; Irizarry, I.; Chen, Q.; Zhang, N.; Walsh, E.; Tadych, M.; Bergen, M. Hydrogen peroxide staining to visualize bacterial infections of seedling root cells. Microsc. Res. Tech. 2014, 77, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Beltran-Garcia, M.; White, J.F.; Prado, F.M.; Prieto, K.R.; Yamaguchi, L.F.; Torres, M.S.; Kato, M.J.; Medeiros, M.H.G.; Di Mascio, P. Nitrogen acquisition in Agave tequilana from degradation of endophytic bacteria. Sci. Rep. 2014, 4, 6938. [Google Scholar] [CrossRef] [PubMed]
- White, J.F.; Torres, M.S.; Verma, S.K.; Elmore, M.T.; Kowalski, K.P.; Kingsley, K.L. Evidence for Widespread Microbivory of Endophytic Bacteria in Roots of Vascular Plants through Oxidative Degradation in Root Cell Periplasmic Spaces; Kumar, A., Singh, A., Singh, V., Eds.; Elsevier: New York, NY, USA, 2018. [Google Scholar]
- White, J.F.; Kingsley, K.L.; Kowalski, K.P.; Irizarry, I.; Micci, A.; Soares, M.A.; Bergen, M.S. Disease protection and allelopathic interactions of seed-transmitted endophytic Pseudomonads of invasive seed grass (Phragmites australis). Plant Soil 2017, 422, 195–208. [Google Scholar] [CrossRef]
- Verma, S.; Kingsley, K.; Irizarry, I.; Bergen, M.; Kharwar, R.; White, J.F. Seed vectored endophytic bacteria modulate development of rice seedlings. J. Appl. Microbiol. 2017, 122, 1680–1691. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Kingsley, K.; Bergen, M.; English, C.; Elmore, M.; Kharwar, R.N.; White, J.F. Bacterial endophytes from rice cut grass (Leersia oryzoides L.) increase growth, promote root gravitropic response, stimulate root hair formation, and protect rice seedlings from disease. Plant Soil 2018, 422, 223–238. [Google Scholar] [CrossRef]
- Verma, S.K.; White, J.F. Indigenous endophytic seed bacteria promote seedling development and defend against fungal disease in browntop millet (Urochloa ramose L.). J. Appl. Microbiol. 2018, 124, 764–778. [Google Scholar] [CrossRef] [PubMed]
- Prieto, K.R.; Echaide-Aquino, F.; Huerta-Robles, A.; Valerio, H.P.; Macedo-Raygoza, G.; Prado, F.M.; Medeiros, M.; Brito, H.F.; da Silva, I.; Felinto, M.C.F.; et al. Plant Macronutrient Use Efficiency; Hossain, M., Kamiya, T., Burritt, D., Tram, L.-S.P., Fujiwara, T., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 285–302. [Google Scholar]
- White, J.F.; Kingsley, K.; Harper, C.J.; Verma, S.K.; Brindisi, L.; Chen, Q.; Chang, X.; Micci, A.; Bergen, M. Transformative Paleobotany: Papers to Commemorate the Life and Legacy of Thomas N. Taylor; Krings, M., Harper, C.J., Cuneo, N.R., Rothwell, G.W., Eds.; Elsevier: New York, NY, USA, 2018. [Google Scholar]
- White, J.F.; Kingsley, K.L.; Butterworth, S.; Brindisi, L.; Gatei, J.W.; Elmore, M.T.; Verma, S.K.; Yao, X.; Kowalski, K.P. Seed Endophytes: Biology and Biotechnology; Verma, S.K., White, J.F., Eds.; Springer: Cham, Switzerland, 2018; in press. [Google Scholar]
- White, J.F. Syntrophic imbalance and the etiology of bacterial endoparasitism diseases. Med. Hypotheses 2017, 107, 14–15. [Google Scholar] [CrossRef] [PubMed]
- White, J.F.; Chen, Q.; Torres, M.; Mattera, R.; Irizarry, I.; Tadych, M.; Bergen, M. Collaboration between grass seedlings and rhizobacteria to scavenge organic nitrogen in soils. AoB Plants 2015, 7, plu093. [Google Scholar] [CrossRef] [PubMed]
- Irizarry, I.; White, J.F. Application of bacteria from non-cultivated plants to promote growth, alter root architecture and alleviate salt stress of cotton. J. Appl. Microbiol. 2017, 122, 1110–1120. [Google Scholar] [CrossRef] [PubMed]
- Irizarry, I.; White, J. Bacillus amyloliquefaciens alters gene expression, ROS production and lignin synthesis in cotton seedling roots. J. Appl. Microbiol. 2018, 124, 1589–1603. [Google Scholar] [CrossRef] [PubMed]
- Funk-Jensen, D.; Hockenhull, J. Root exudation, rhizosphere microorganisms and disease control. Växtskyddsnotiser 1984, 48, 49–54. [Google Scholar]
- Bowsher, A.W.; Ali, R.; Harding, S.A.; Tsai, C.-J.; Donovan, L.A. Evolutionary Divergences in root exudate composition among ecologically-contrasting Helianthus species. PLoS ONE 2016, 11, e0148280. [Google Scholar] [CrossRef] [PubMed]
- Broeckling, C.D.; Broz, A.K.; Bergelson, J.; Manter, D.K.; Vivanco, J.M. Root exudates regulate soil fungal community composition and diversity. Appl. Environ. Microbiol. 2008, 74, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Ortíz-Castro, R.; Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; López-Bucio, J. The role of microbial signals in plant growth and development. Plant Signal. Behav. 2009, 4, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Rudrappa, T.; Czymmek, K.J.; Paré, P.W.; Bais, H.P. Root-secreted malic acid recruits beneficial soil bacteria. Plant Physiol. 2008, 148, 1547–1556. [Google Scholar] [CrossRef] [PubMed]
- Badri, D.V.; Vivanco, J.M. Regulation and function of root exudates. Plant Cell Environ. 2009, 32, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Muangthong, A.; Youpensuk, S.; Rerkasem, B. Isolation and characterization of endophytic nitrogen fixing bacteria in sugarcane. Trop. Life Sci. Res. 2015, 26, 41–51. [Google Scholar] [PubMed]
- Gallon, J.R. The oxygen sensitivity of nitrogenase: A problem for biochemists and micro-organisms. Trends Biochem. Sci. 1981, 6, 19–23. [Google Scholar] [CrossRef]
- Singh, S.; Gupta, P.; Bajaj, B.K. Characterization of a robust serine protease from Bacillus subtilis K-1. J. Basic Microbiol. 2017, 58, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Hill, P.W.; Marsden, K.A.; Jones, D.L. How significant to plant N nutrition is the direct consumption of soil microbes by roots? New Phytol. 2013, 199, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.B.; Sayyed, R.Z.; Trivedi, M.H.; Gobi, T.A. Phosphate solubilizing microbes: Sustainable approach for managing phosphorus deficiency in agricultural soils. SpringerPlus 2013, 2, 587. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, T.C.; Nolan, E.M. Beyond Iron: Non-classical biological functions of bacterial siderophores. Daltan Trans. 2015, 44, 6320–6339. [Google Scholar] [CrossRef] [PubMed]
- Bar-Ness, E.; Chen, Y.; Hadar, H.; Marschner, H.; Römheld, V. Siderophores of Pseudomonas putida as an iron source for dicot and monocot plants. Plant Soil 1991, 130, 231–241. [Google Scholar] [CrossRef]
- Bar-Ness, E.; Hadar, Y.; Chen, Y.; Shanzer, A.; Libman, J. Iron uptake by plants from microbial siderophores: A study with 7-Nitrobenz-2 Oxa-1,3-Diazole-Desferrioxamine as fluorescent ferrioxamine B analog. Plant Physiol. 1992, 99, 1329–1335. [Google Scholar] [CrossRef] [PubMed]
- Mohana, D.C.; Thippeswamy, S.; Abishek, R.U. Antioxidant, antibacterial, and ultraviolet protective properties of carotenoids isolated from Micrococcus spp. Radiat. Prot. Environ. 2013, 36, 168–174. [Google Scholar] [CrossRef]
- Ohwada, T.; Shirakawa, Y.; Kusumoto, M.; Masuda, H.; Sato, T. Susceptibility to hydrogen peroxide and catalase activity of root nodule bacteria. Biosci. Biotechnol. Biochem. 2014, 63, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.J.; Chang, A.C.; Crowley, D.E.; Page, A.L. Characterization of organic acids recovered from rhizosphere of corn grown on biosolids treated media. Comm. Soil Sci. Plant Anal. 2006, 37, 871–887. [Google Scholar] [CrossRef]
- Sun, Y.; O’Riordan, M. Regulation of bacterial pathogenesis by intestinal short-chain fatty acids. Adv. Appl. Microbiol. 2013, 85, 93–118. [Google Scholar] [PubMed]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Cox, N.A.; McHan, F.; Bailey, J.S.; Shotts, E.B. Effect of butyric or lactic acid on the in vivo colonization of Salmonella typhimurium. J. Appl. Poult. Res. 1994, 3, 315–318. [Google Scholar] [CrossRef]
- Van Immerseel, F.; Fievez, V.; de Buck, J.; Pasmans, F.; Martel, A.; Haesebrouck, F.; Ducatelle, R. Microencapsulated short-chain fatty acids in feed modify colonization and invasion early after infection with Salmonella enteritidis in young chickens. Poult. Sci. 2004, 83, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Tramontano, W.A.; Scanlon, C. Cell cycle inhibition by butyrate in legume root meristems. Phytochemistry 1996, 41, 85–88. [Google Scholar] [CrossRef]
- Lanzagorta, J.M.A.; de la Torre, C.; Aller, P. The effect of butyrate on cell cycle progression in Allium cepa root meristems. Physiol. Plant. 1988, 72, 775–781. [Google Scholar] [CrossRef]
- Errington, J.; Mickiewicz, K.; Kaeai, Y.; Wu, L.J. L-form bacteria, chronic diseases and the origins of life. Phil. Trans. R. Soc. B 2016, 371, 20150494. [Google Scholar] [CrossRef] [PubMed]
- Kawai, Y.; Mercier, R.; Wu, L.J.; Domínguez-Cuevas, P.; Oshima, T.; Errington, J. Cell growth of wall-free L-form bacteria is limited by oxidative damage. Curr. Biol. 2015, 25, 1613–1618. [Google Scholar] [CrossRef] [PubMed]
- Allan, E.J.; Hoischen, C.; Gumpert, J. Bacterial L-forms. Adv. Appl. Microbiol. 2009, 68, 1–39. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, C.M.J.; Booth, N.A.; Allan, E.J. An ELISA for the detection of Bacillus subtilis L-form bacteria confirms their symbiosis in strawberry. Lett. Appl. Microbiol. 2000, 31, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Dimova, T.; Terzieva, A.; Djerov, L.; Dimitrova, V.; Nikolov, A.; Grozdanov, P.; Markova, N. Mother-to-newborn transmission of mycobacterial L-forms and Vδ2 T-cell response in placentobiome of BCG-vaccinated pregnant women. Sci. Rep. 2017, 7, 17366. [Google Scholar] [CrossRef] [PubMed]
- Atsatt, P.R.; Whiteside, M.D. Novel symbiotic protoplasts formed by endophytic fungi explain their hidden existence, lifestyle switching, and diversity within the plant kingdom. PLoS ONE 2014, 9, e95266. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R. Modulation of plant ethylene levels by the bacterial enzyme ACC deaminase. FEMS Microbiol. Lett. 2005, 251, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hardoim, P.R.; Van Overbeek, L.S.; Van Elsas, J.D. Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol. 2008, 16, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.; Redman, R. Balancing the generation and elimination of reactive oxygen species. Proc. Natl. Acad. Sci. USA 2005, 102, 3175–3176. [Google Scholar] [CrossRef] [PubMed]
- White, J.F.; Torres, M.S. Is endophyte-mediated defensive mutualism oxidative stress protection? Physiol. Plant. 2010, 138, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.S.; White, J.F.; Zhang, X.; Hinton, D.M.; Bacon, C.W. Endophyte-mediated adjustments in host morphology and physiology and effects on host fitness traits in grasses. Fungal Ecol. 2012, 5, 322–330. [Google Scholar] [CrossRef]
- Hamilton, C.E.; Gundel, P.E.; Helander, M.; Saikkonen, K. Endophytic mediation of reactive oxygen species and antioxidant activity in plants: A review. Fungal Divers. 2012, 54, 1–10. [Google Scholar] [CrossRef]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L.M.P. Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Genet. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Kingsley, K.L.; Bergen, M.S.; Kowalski, K.P.; White, J.F. Fungal disease protection in rice (Oryza sativa) seedlings by growth promoting seed-associated endophytic bacteria from invasive Phragmites australis. Microorganisms 2018, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, D.K.; Prakash, A.; Johri, B.N. Induced systemic resistance (ISR) in plants: Mechanism of action. Indian J. Microbiol. 2007, 47, 289–297. [Google Scholar] [CrossRef] [PubMed]
- León, M.; Yaryura, P.M.; Montecchia, M.S.; Hernández, A.I.; Correa, O.S.; Pucheu, N.L.; García, A.F. Antifungal activity of selected indigenous Pseudomonas and Bacillus from the soybean rhizosphere. Int. J. Microbiol. 2009, 572049. [Google Scholar] [CrossRef]
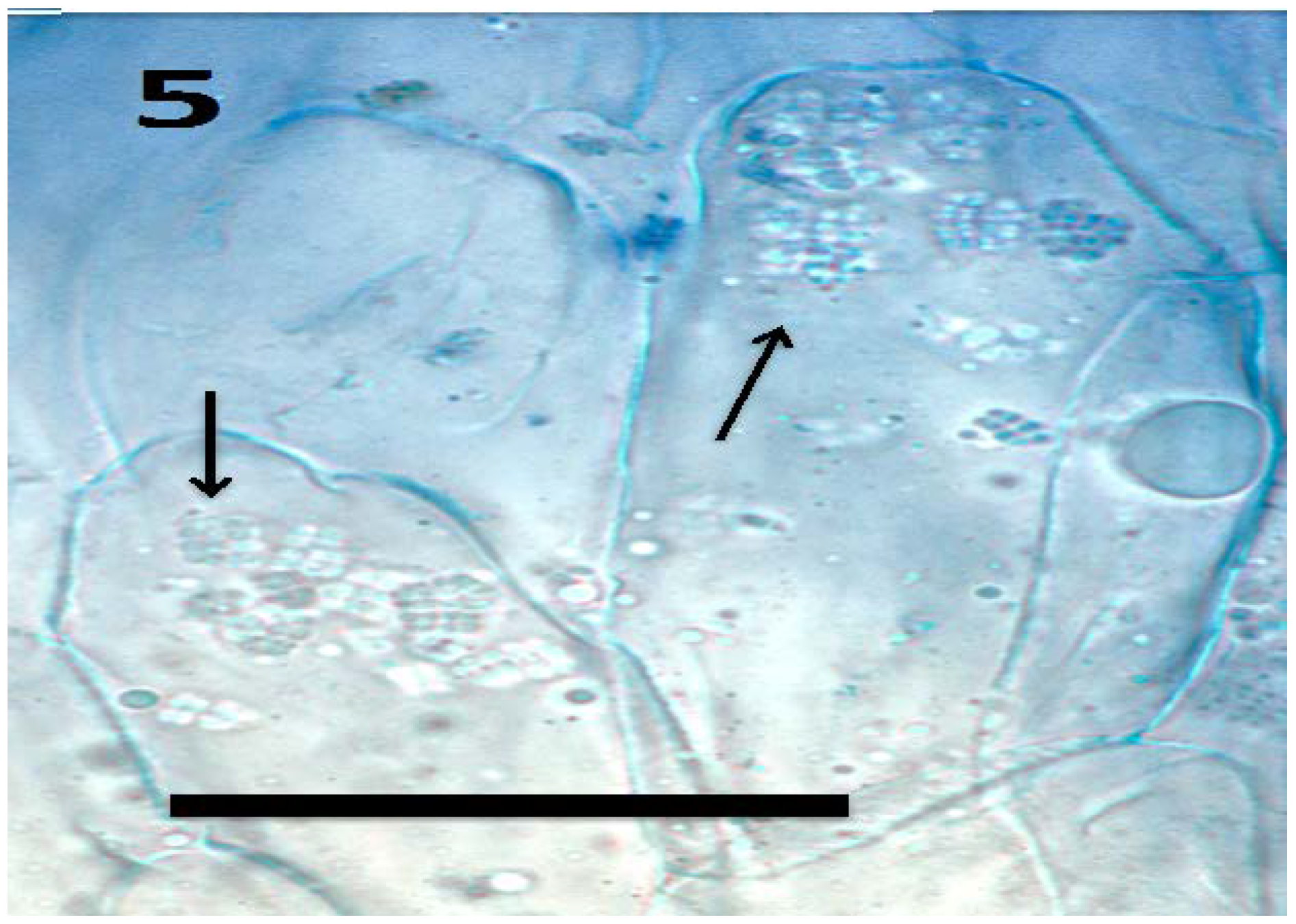
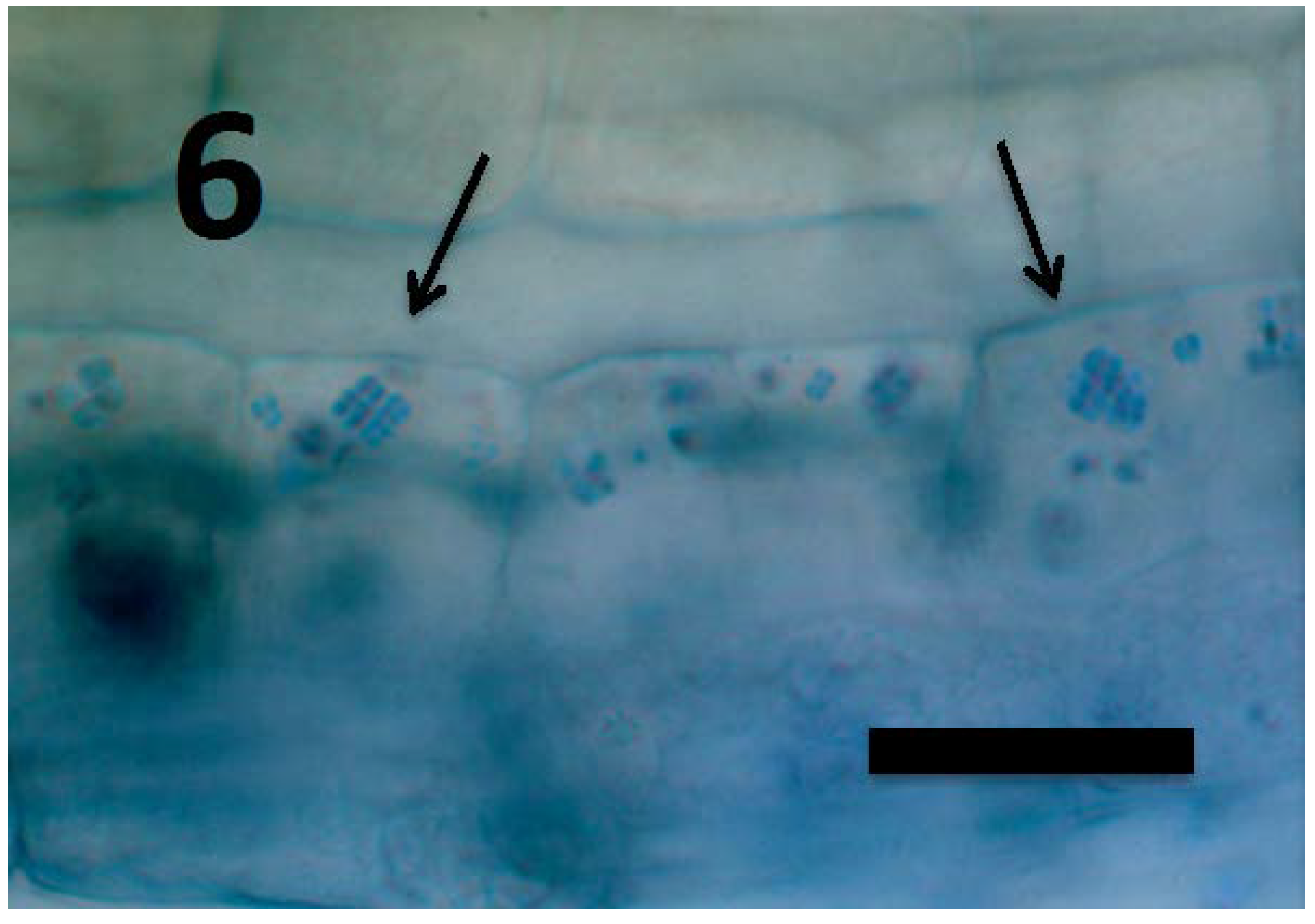
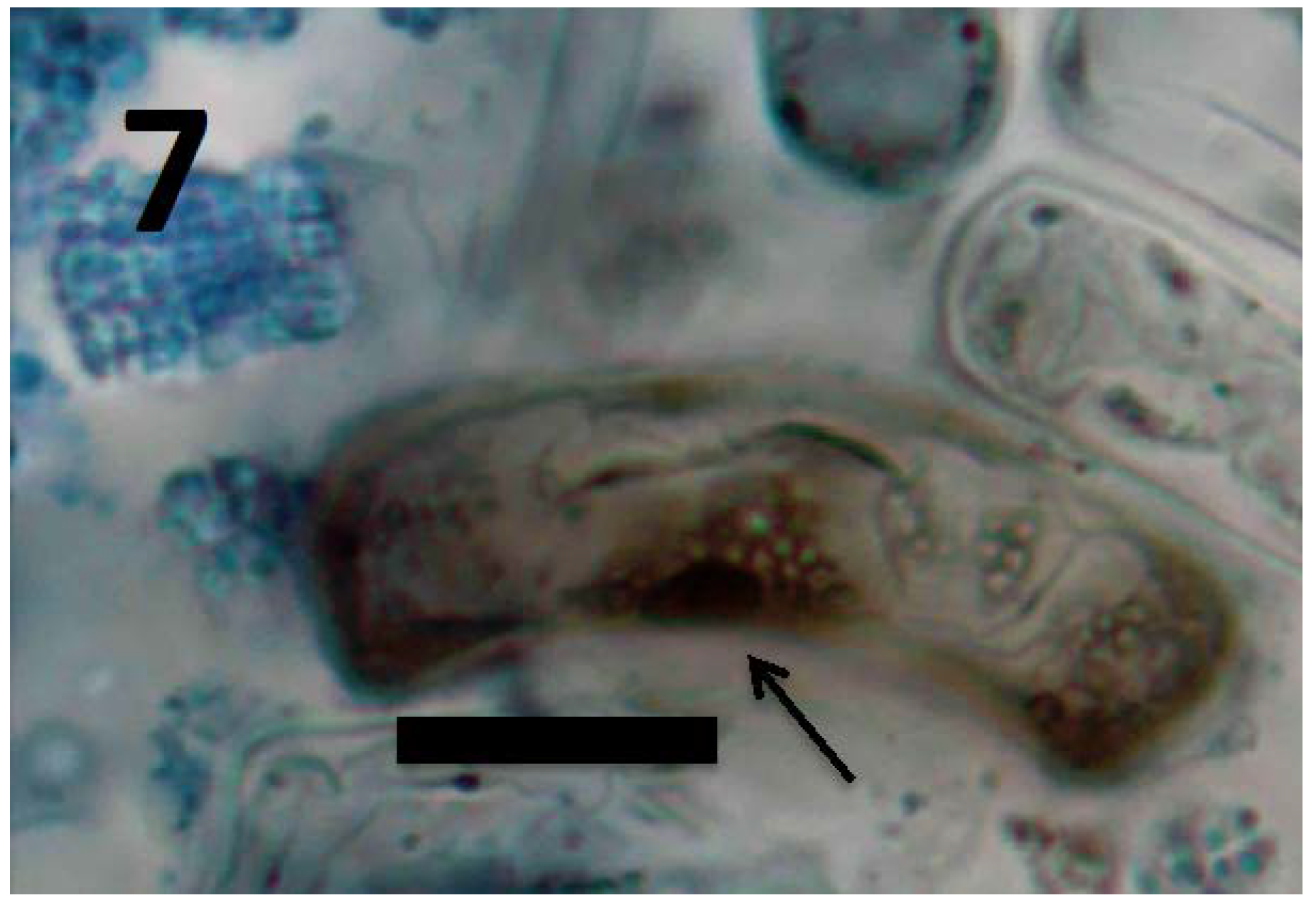
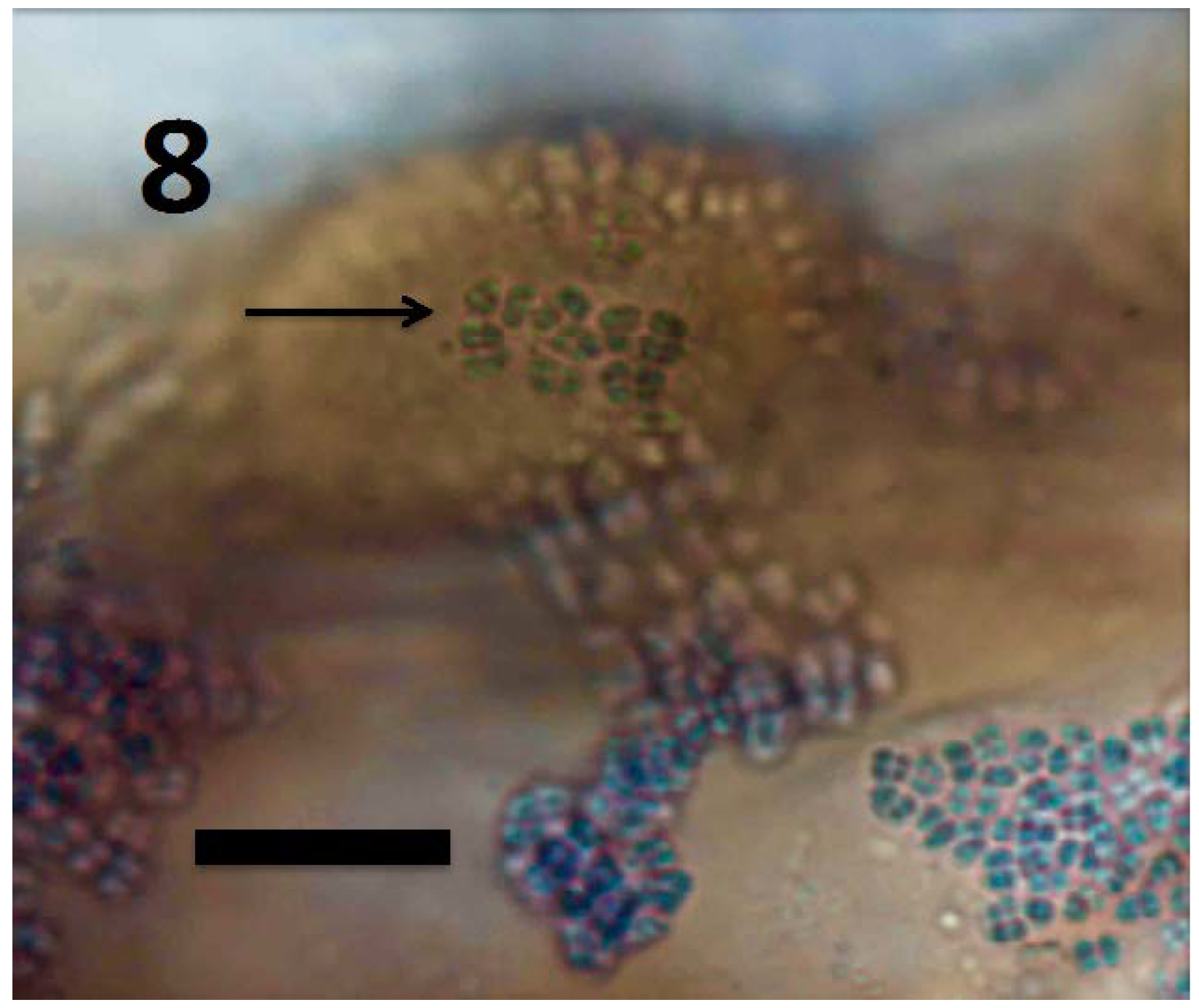
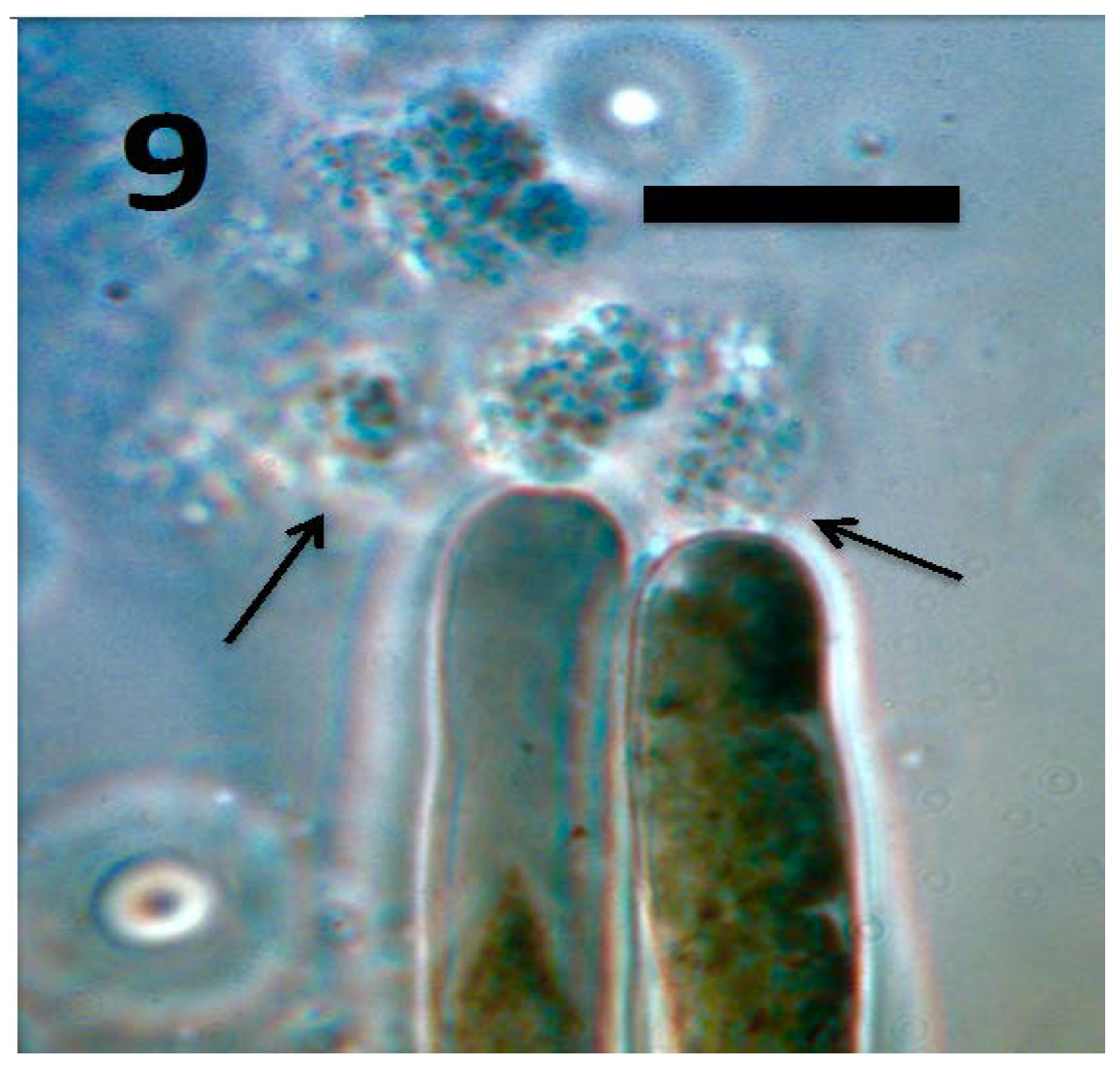
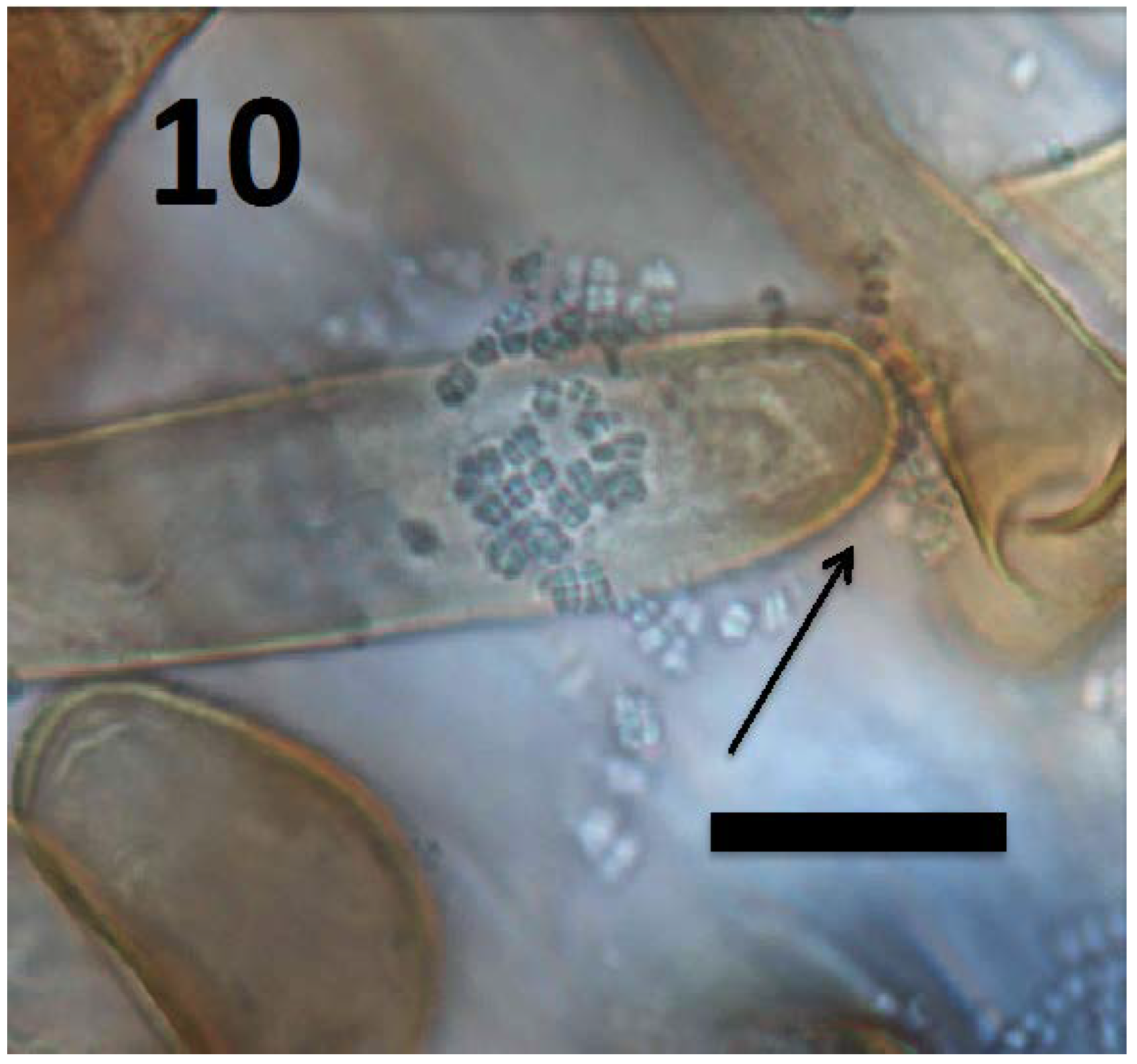
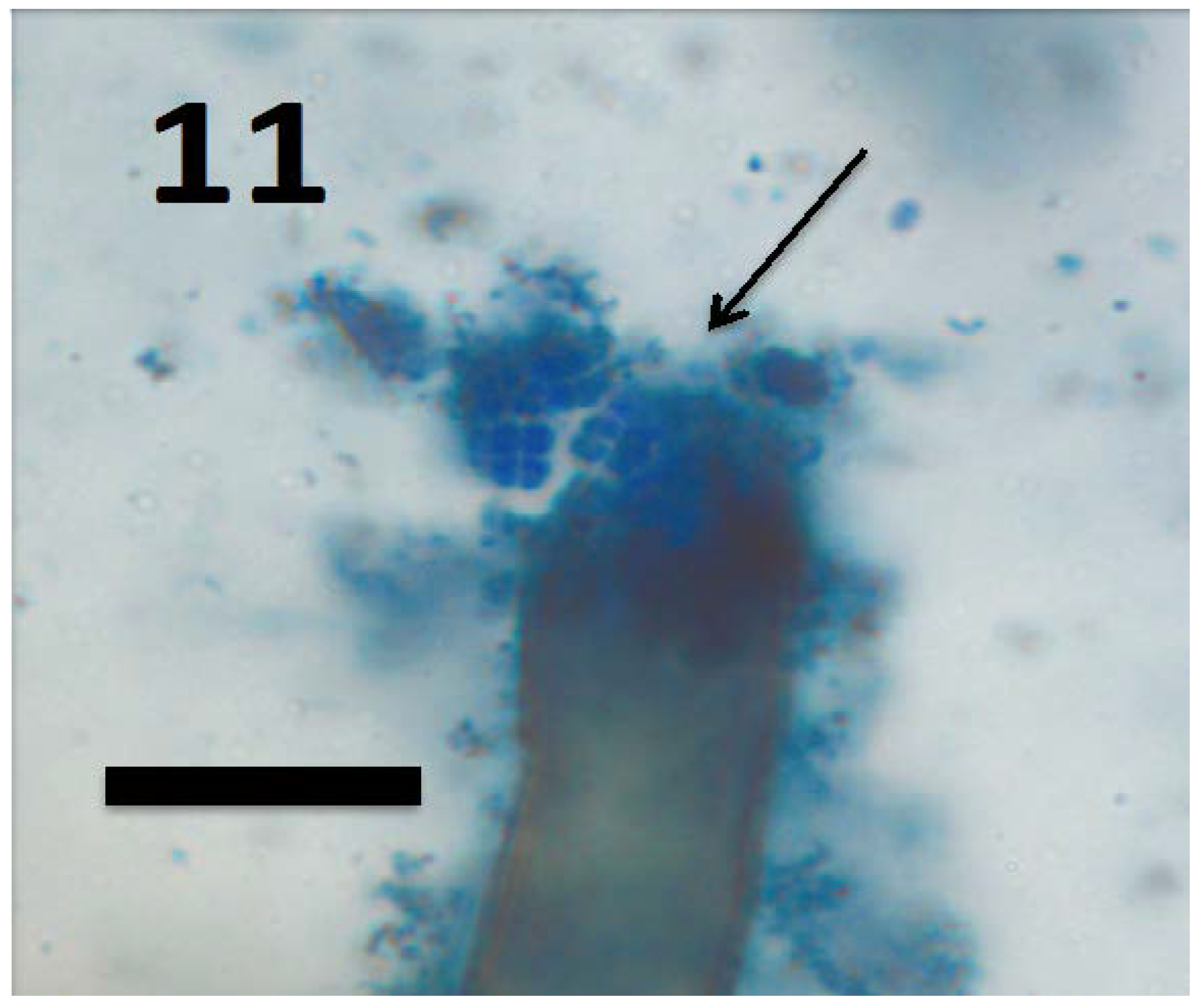
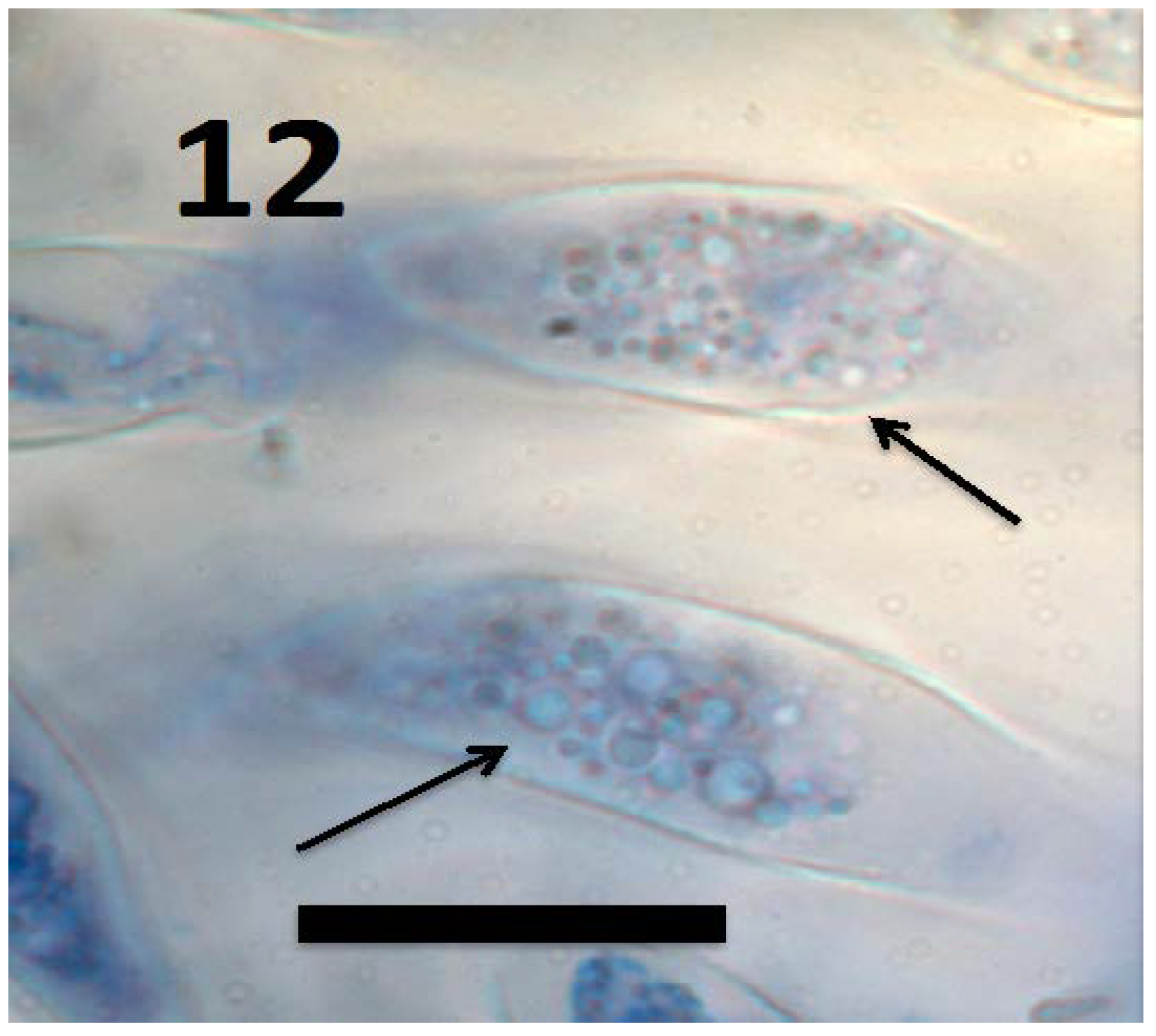
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
White, J.F.; Kingsley, K.L.; Verma, S.K.; Kowalski, K.P. Rhizophagy Cycle: An Oxidative Process in Plants for Nutrient Extraction from Symbiotic Microbes. Microorganisms 2018, 6, 95. https://doi.org/10.3390/microorganisms6030095
White JF, Kingsley KL, Verma SK, Kowalski KP. Rhizophagy Cycle: An Oxidative Process in Plants for Nutrient Extraction from Symbiotic Microbes. Microorganisms. 2018; 6(3):95. https://doi.org/10.3390/microorganisms6030095
Chicago/Turabian StyleWhite, James F., Kathryn L. Kingsley, Satish K. Verma, and Kurt P. Kowalski. 2018. "Rhizophagy Cycle: An Oxidative Process in Plants for Nutrient Extraction from Symbiotic Microbes" Microorganisms 6, no. 3: 95. https://doi.org/10.3390/microorganisms6030095
APA StyleWhite, J. F., Kingsley, K. L., Verma, S. K., & Kowalski, K. P. (2018). Rhizophagy Cycle: An Oxidative Process in Plants for Nutrient Extraction from Symbiotic Microbes. Microorganisms, 6(3), 95. https://doi.org/10.3390/microorganisms6030095






