Antiviral Compounds from Myxobacteria
Abstract
1. Introduction
2. Myxobacteria
3. Secondary Metabolites from Myxobacteria with Antiviral Activity
3.1. Human Immunodeficiency Virus (HIV)
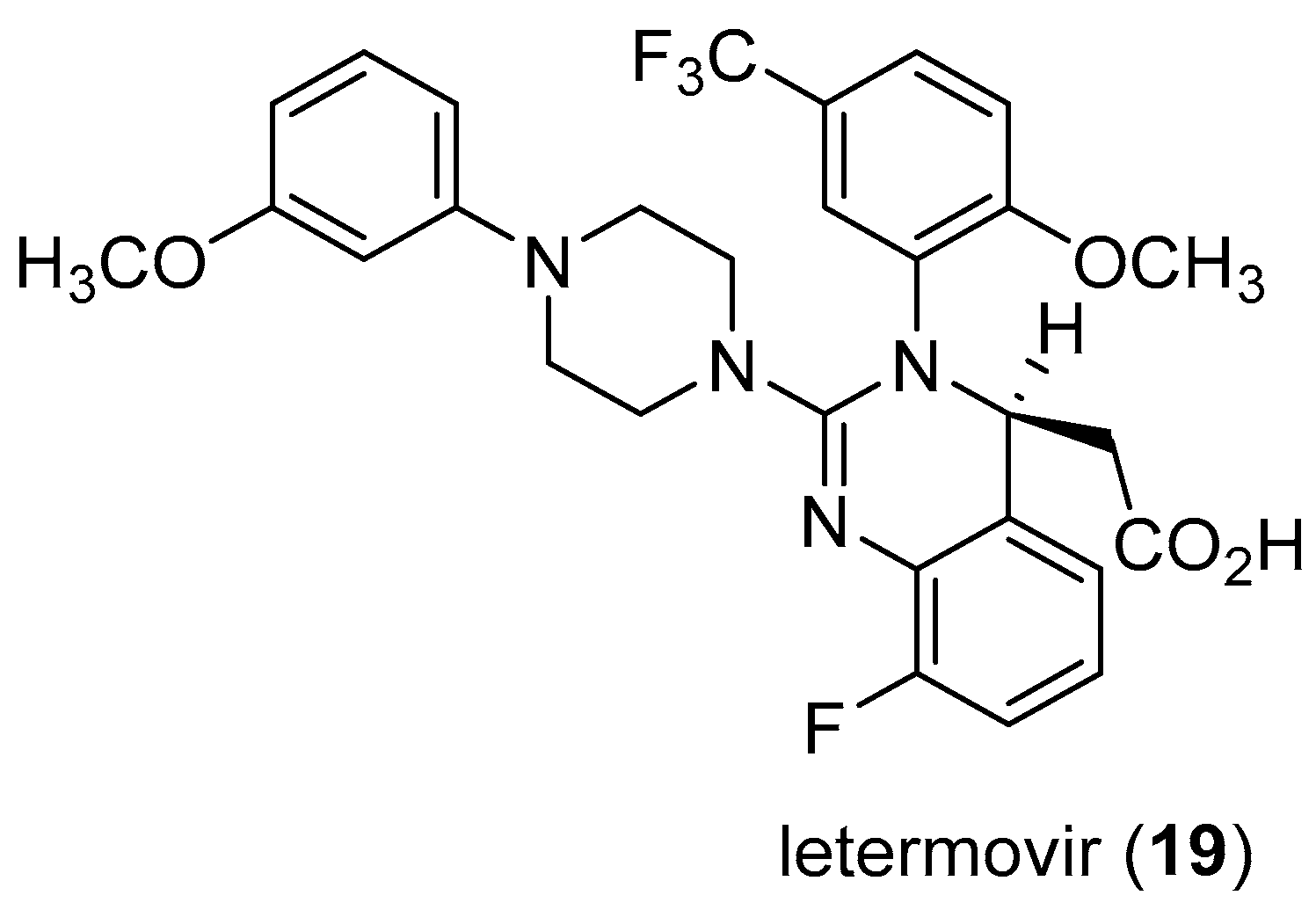
3.2. Human Cytomegalovirus (HCMV)
3.3. Ebola Virus Disease (EVD)
3.4. Hepatitis C Virus
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nübel, U. Emergence and spread of antimicrobial resistance: Recent insights from bacterial population genomics. In How to Overcome the Antibiotic Crisis—Facts, Challenges, Technologies & Future Perspective; Stadler, M., Dersch, P., Eds.; Springer: Berlin, Germany, 2017; Volume 398, pp. 35–53. [Google Scholar]
- Blair, W.; Cox, C. Current Landscape of Antiviral Drug Discovery. F1000 Rev. 2016, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Antimicrobial resistance. Available online: http://www.who.int/en/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 18 May 2018).
- Dziuban, E.J.; DeVos, J.; Ngeno, B.; Ngugi, E.; Zhang, G.; Sabatier, J.; Wagar, N.; Diallo, K.; Nganga, L.; Katana, A.; et al. High prevalence of Abacavir-associated L74V/I mutations in Kenyan children failing antiretroviral therapy. Pediatr. Infect. Dis. J. 2017, 36, 758–760. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. Antiretroviral Drugs Used in the Treatment of HIV Infection. Available online: https://www.fda.gov/ForPatients/Illness/HIVAIDS/Treatment/ucm118915.htm (accessed on 22 May 2018).
- National Institute of Allergy and Infectious Disease. Drugs That Fight HIV-1. Available online: https://aidsinfo.nih.gov/contentfiles/upload/HIV_Pill_Brochure.pdf (accessed on 22 May 2018).
- DailyMed. Drug Listing Certification. Available online: https://dailymed.nlm.nih.gov/dailymed/index.cfm (accessed on 22 May 2018).
- Reust, C.E. Common adverse effects of antiretroviral therapy for HIV disease. Am. Fam. Phys. 2011, 83, 1443–1451. [Google Scholar]
- Tantillo, C.; Ding, J.; Jacobo-Molina, A.; Nanni, R.G.; Boyer, P.; Hughes, S.H.; Pauwels, R.; Andries, K.; Jansen, P.A.; Arnold, E. Locations of anti-AIDS drug binding sites and resistance mutations in the three-dimensional structure of HIV-1 reverse transcriptase: Implications for mechanisms of drug inhibition and resistance. J. Mol. Biol. 1994, 243, 369–387. [Google Scholar] [CrossRef] [PubMed]
- Pawlotsky, J. Treatment failure and resistance with direct-acting antiviral drugs against Hepatitis C virus. Hepatology 2011, 53, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
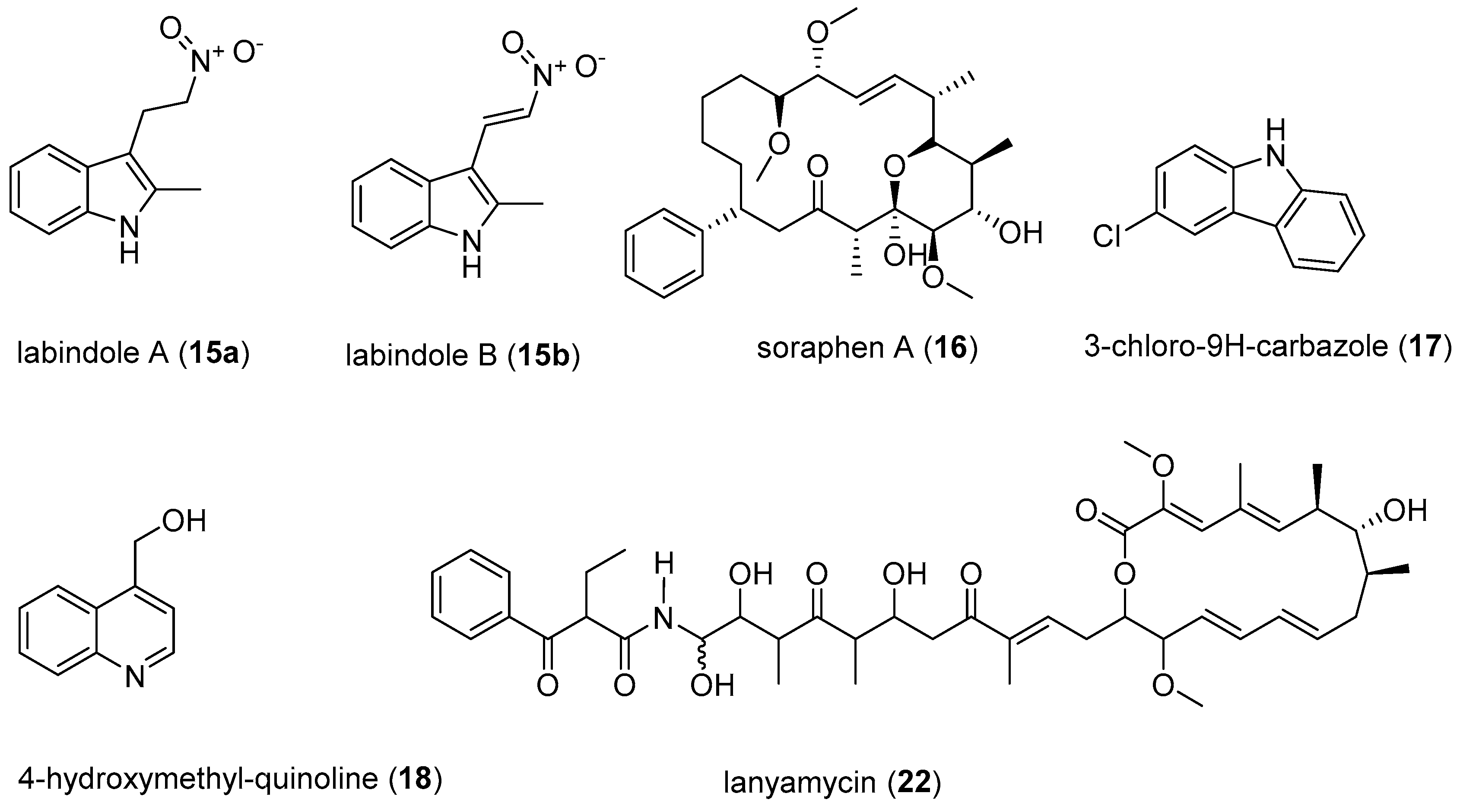
- Koutsoudakis, G.; Romero-Brey, I.; Berger, C.; Pérez-Vilaró, G.; Monteiro, P.P.; Vondran, F.W.R.; Kalesse, M.; Harmrolfs, K.; Müller, R.; Martinez, J.P.; et al. Soraphen A: A broad-spectrum antiviral natural product with potent anti-hepatitis C virus activity. J. Hepatol. 2015, 63, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, J.; Fayad, A.A.; Müller, R. Natural products from myxobacteria: Novel metabolites and bioactivities. Nat. Prod. Rep. 2017, 34, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Hüttel, S.; Testolin, G.; Herrmann, J.; Planke, T.; Gille, F.; Moreno, M.; Stadler, M.; Brönstrup, M.; Kirschning, A.; Müller, R. Discovery and total synthesis of natural cystobactamid derivatives with superior activity against Gram-negative pathogens. Angew. Chem. Int. Ed. 2017, 56, 12760–12764. [Google Scholar] [CrossRef] [PubMed]
- Gerth, K.; Bedorf, N.; Irschik, H.; Höfle, G.; Reichenbach, H. The soraphens: A family of novel antifungal compounds from Sorangium cellulosum (Myxobacteria). J. Antibiot. 1994, 47, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Reichenbach, H. Myxobacteria, producers of novel bioactive substances. J. Ind. Microbiol. Biotechnol. 2001, 27, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Gerth, K.; Pradella, S.; Perlova, O.; Beyer, S.; Müller, R. Myxobacteria: Proficient producers of novel natural products with various biological activities—Past and future biotechnological aspects with the focus on the genus Sorangium. J. Biotechnol. 2003, 106, 233–253. [Google Scholar] [CrossRef] [PubMed]
- Dawid, W. Biology and global distribution of myxobacteria in soils. FEMS Microbiol. 2000, 24, 403–427. [Google Scholar] [CrossRef]
- Mohr, K.I.; Moradi, A.; Glaeser, S.P.; Kämpfer, P.; Gemperlein, K.; Nübel, U.; Schumann, P.; Müller, R.; Wink, J. Nannocystis konarekensis sp. nov., a novel myxobacterium from an Iranian desert. Int. J. Syst. Evol. Microbiol. 2018, 68, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, S.C.; Müller, R. Myxobacteria—‘Microbial factories’ for the production of bioactive secondary metabolites. Mol. BioSyst. 2009, 5, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Weissman, K.J.; Müller, R. Myxobacterial secondary metabolites: Bioactivities and modes-of-action. Nat. Prod. Rep. 2010, 27, 1276–1295. [Google Scholar] [CrossRef] [PubMed]
- Korp, J.; Gurovic, M.S.V.; Nett, M. Antibiotics from predatory bacteria. Beilstein J. Org. Chem. 2016, 12, 594–607. [Google Scholar] [CrossRef] [PubMed]
- Goldman, B.S.; Nierman, W.C.; Kaiser, D.; Slater, S.C.; Durkin, A.S.; Eisen, J.A.; Ronning, C.M.; Barbazuk, W.B.; Blanchard, M.; Field, C.; et al. Evolution of sensory complexity recorded in a myxobacterial genome. Prod. Natl. Acad. Sci. USA 2006, 103, 15200–15205. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, S.C.; Müller, R. The impact of genomics on the exploitation of the myxobacterial secondary metabolome. Nat. Prod. Rep. 2009, 26, 1385–1407. [Google Scholar] [CrossRef] [PubMed]
- Céspedes, A.D.; Hufendiek, P.; Crüsemann, M.; Schäberle, T.F.; König, G.M. Marine-derived myxobacteria of the suborder Nannocystineae: An underexplored source of structurally intriguing and biologically active metabolites. Beilstein J. Org. Chem. 2016, 12, 969–984. [Google Scholar] [CrossRef] [PubMed]
- Schneiker, S.; Perlova, O.; Kaiser, O.; Gerth, K.; Alici, A.; Altmeyer, M.O.; Bartels, D.; Bekel, T.; Beyer, S.; Bode, E.; et al. Complete genome sequence of the myxobacterium Sorangium cellulosum. Nat. Biotechnol. 2007, 25, 1281–1289. [Google Scholar] [CrossRef] [PubMed]
- Schäberle, T.F.; Lohr, F.; Schmitz, A.; König, G.M. Antibiotics from myxobacteria. Nat. Prod. Rep. 2014, 31, 953–972. [Google Scholar] [CrossRef] [PubMed]
- Bode, H.B.; Bethe, B.; Höfs, R.; Zeeck, A. Big effects from small changes: Possible ways to explore nature’s chemical diversity. ChemBioChem 2002, 3, 619–627. [Google Scholar] [CrossRef]
- Seitz, R. Human Immunodeficiency Virus (HIV). Transfus. Med. Hemother. 2016, 43, 203–222. [Google Scholar]
- Curreli, F.; Kwon, Y.D.; Zhang, H.; Scacalossi, D.; Belov, D.S.; Tikhonov, A.A.; Andreev, A.I.; Altieri, A.; Kurkin, A.V.; Kwong, P.D.; et al. Structure-based design of a small molecule CD4-antagonist with broad spectrum anti-HIV-1 activity. J. Med. Chem. 2015, 58, 6909–6927. [Google Scholar] [CrossRef] [PubMed]
- Asim, K.D.; Lin, R.; Shibo, J. Structure-based identification of small molecule antiviral compounds targeted to the gp41 core structure of the Human Immunodeficiency Virus type 1. J. Med. Chem. 1999, 42, 3203–3209. [Google Scholar]
- Martinez, J.P.; Hinkelmann, B.; Fleta-Soriano, E.; Steinmetz, H.; Jansen, R.; Diez, J.; Frank, R.; Sasse, F.; Meyerhans, A. Identification of myxobacteria-derived HIV inhibitors by a high-throughput two-step infectivity assay. Microb. Cell Fact. 2013, 12. [Google Scholar] [CrossRef] [PubMed]
- Zander, W.; Irschik, H.; Augustiniak, H.; Herrmann, M.; Jansen, R.; Steinmetz, H.; Gerth, K.; Kessler, W.; Kalesse, M.; Höfle, G.; et al. Sulfangolids, macrolide sulfate esters from Sorangium cellulosum. Chem. Eur. J. 2012, 18, 6264–6271. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://scifinder.cas.org/scifinder/view/scifinder/scifinderExplore.jsf (accessed on 10 July 2018).
- Shen, Y.; Volrath, S.L.; Weatherly, S.C.; Elich, T.D.; Tong, L. A mechanism for the potent inhibition of eukaryotic acetyl-coenzyme A carboxylase by soraphen A, a macrocyclic polyketide natural product. Mol. Cell 2004, 16, 881–891. [Google Scholar] [CrossRef] [PubMed]
- Mühlradt, P.F.; Sasse, F. Epothilone B stabilizes microtubuli of macrophages like taxol without showing taxol-like endotoxin activity. Cancer Res. 1997, 57, 3344–3346. [Google Scholar] [PubMed]
- Goodin, S.; Kane, M.P.; Rubin, E.H. Epothilones: Mechanism of action and biologic activity. J. Clin. Oncol. 2004, 22, 2015–2025. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.K.; Tucker, C.; Favours, E.; Cheshire, P.J.; Creech, J.; Billups, C.A.; Smykla, R.; Lee, F.Y.F.; Houghton, P.J. In vivo evaluation of Ixabepilone (BMS247550), A novel epothilone B derivative, against pediatric cancer models. Clin. Cancer Res. 2005, 11, 6950–6958. [Google Scholar] [CrossRef] [PubMed]
- Reboll, M.R.; Ritter, B.; Sasse, F.; Niggemann, J.; Frank, R.; Nourbakhsh, M. The myxobacterial compounds spirangien A and spirangien M522 are potent inhibitors of IL-8 expression. ChemBioChem 2012, 13, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Jurkliewicz, E.; Jansen, R.; Kunze, B.; Trowitzsch-Klenast, W.; Porche, E.; Reichenbach, H.; Höfle, G.; Hunsmann, G. Three new potent HIV-1 inhibitors from myxobacteria. Antivir. Chem. Chemother. 1992, 3, 189–193. [Google Scholar] [CrossRef]
- Trowitzsch-Kienast, W.; Forche, E.; Wray, V.; Reichenbach, H.; Jurkiewicz, E.; Hunsmann, G.; Höfle, G. Phenalamide, neue HIV-1-Inhibitoren aus Myxococcus stipitatus Mx s40. Liebigs Ann. Chem. 1992, 1992, 659–664. [Google Scholar] [CrossRef]
- Garcia, R.; Stadler, M.; Gemperlein, K.; Müller, R. Aetherobacter fasciculatus gen. nov., sp. nov. and Aetherobacter rufus sp. nov., novel myxobacteria with promising biotechnological applications. Int. J. Syst. Evol. Microbiol. 2016, 66, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Plaza, A.; Garcia, R.; Bifulco, G.; Martinez, J.P.; Hüttel, S.; Sasse, F.; Meyerhans, A.; Stadler, M.; Müller, R. Aetheramides A and B, potent HIV-inhibitory depsipeptides from a Myxobacterium of the new genus “Aetherobacter”. Org. Lett. 2012, 14, 2854–2857. [Google Scholar] [CrossRef] [PubMed]
- Gerth, K.; Schummer, D.; Höfle, G.; Irschik, H.; Reichenbach, H. Ratjadon: A new antifungal compound from Sorangium cellulosum (Myxobacteria) Production, physico-chemical and biological properties. J. Antibiot. 1995, 48, 787–792. [Google Scholar] [CrossRef]
- Fleta-Soriano, E.; Martinez, J.P.; Hinkelmann, B.; Gerth, K.; Washausen, P.; Diez, J.; Frank, R.; Sasse, F.; Meyerhans, A. The myxobacterial metabolite ratjadone A inhibits HIV infection by blocking the Rev/CRM1-mediated nuclear export pathway. Microb. Cell Fact. 2014, 13, 17. [Google Scholar] [CrossRef] [PubMed]
- ICTV. Taxonomy. Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 30 April 2018).
- Britt, W.J.; Vugler, L.; Butfiloski, E.D.; Stevens, E.B. Cell surface expression of Human Cytomegalovirus (HCMV): Use of HCMV-recombinant vaccinia virus-infected cells in analysis of the human neutralizing antibody response. J. Virol. 1990, 64, 1079–1085. [Google Scholar] [PubMed]
- Arvin, A.M.; Alford, C.A. Chronic Intrauterine and Perinatal Infections. In Antiviral Agents and Viral Diseases of Man; Galassov, G.J., Whitley, R.J., Merigan, T.C., Eds.; Raven Press: New York, NY, USA, 1990; Volume 3, pp. 497–580. [Google Scholar]
- Field, A.K. Human cytomegalovirus: Challenges opportunities and new drug development. Antivir. Chem. Chemother. 1999, 10, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.; Martinez, A. Novel agents for the treatment of human cytomegalovirus infection. Exp. Opin. Ther. Pat. 2000, 10, 165–177. [Google Scholar] [CrossRef]
- Xiong, X.; Smith, J.L.; Chen, M.S. Effect of incorporation of Cidofovir into DNA by Human Cytomegalovirus DNA polymerase on DNA elongation. Antimicrob. Agents Chemother. 1997, 24, 594–599. [Google Scholar]
- Boivin, G.; Chou, S.; Quirk, M.R.; Alejo, E.; Jordan, M.C. Detection of ganciclovir resistance mutations and quantitation of cytomegalovirus (CMV) DNA in leucocytes of patients with fatal disseminated CMV disease. J. Infect. Dis. 1996, 173, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Nassir, M.R.; Emerson, S.G.; Devivar, R.V.; Townsend, L.B.; Drach, J.C.; Taichman, R. Comparison of benzimidazole nucleosides ganciclovir on the in vitro proliferation and colony formation of human marrow progenitor cells. Br. J. Haematol. 1996, 93, 273–279. [Google Scholar] [CrossRef]
- Kaul, D.R.; Stoelben, S.; Cober, E.; Ojo, T.; Sandusky, E.; Lischka, P.; Zimmermann, H.; Rübsamen-Schaeff, H. First report of successful treatment of multidrug-resistant cytomegalovirus disease with the novel anti-CMV compound AIC246. Am. J. Transpl. 2011, 11, 1079–1084. [Google Scholar] [CrossRef] [PubMed]
- NEJM Journal Watch. Letermovir Approved to Prevent CMV Infection and Disease in Transplant Patients. Available online: https://www.jwatch.org/na45554/2017/12/07/letermovir-approved-prevent-cmv-infection-and-disease (accessed on 17 June 2018).
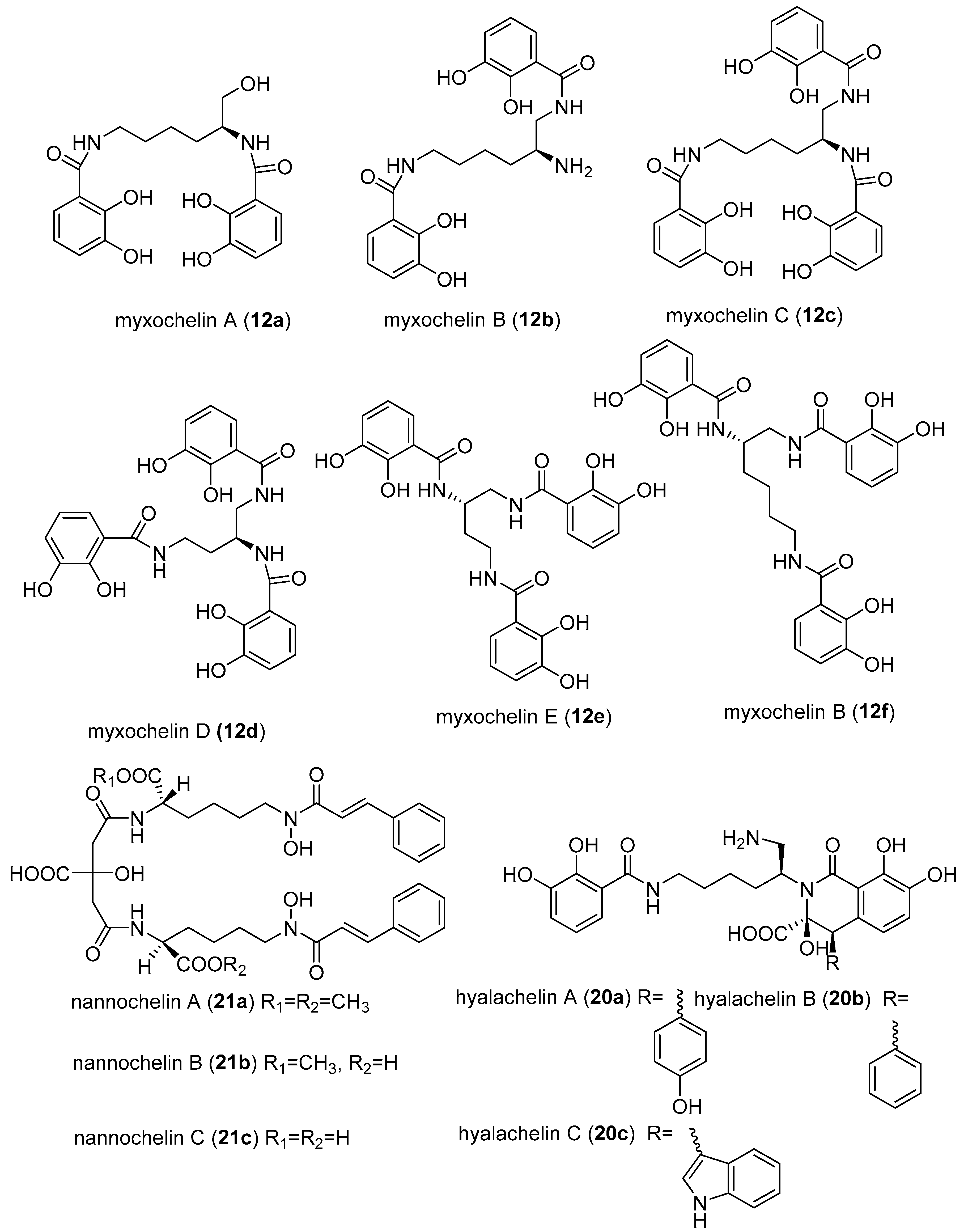
- Schieferdecker, S.; König, S.; Koeberle, A.; Dahse, H.M.; Werz, O.; Nett, M. Myxochelins Target Human 5-Lipoxygenase. J. Nat. Prod. 2015, 78, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Nagoba, B.; Vedpathak, D. Medical applications of siderophores. Eur. J. Gen. Med. 2011, 8, 229–235. [Google Scholar] [CrossRef]
- Miyanaga, S.; Obata, T.; Onaka, H.; Fujita, T.; Saito, N.; Sakurai, H.; Saiki, I.; Furumai, T.; Igarashi, Y. Absolute configuration and antitumor activity of myxochelin a produced by Nonomuraea pusilla TP-A0861. J. Antibiot. 2006, 59, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Korp, J.; König, S.; Schieferdecker, S.; Dahse, H.M.; König, G.M.; Werz, O.; Nett, M. Harnessing enzymatic promiscuity in myxochelins biosynthesis for the production of 5-lipoxygenase inhibitors. ChemBioChem 2015, 16, 2445–2450. [Google Scholar] [CrossRef] [PubMed]
- Schieferdecker, S.; König, S.; Simona Pace, S.; Werz, O.; Nett, M. Myxochelin-inspired 5-lipoxygenase inhibitors: Synthesis and biological evaluation. ChemMedChem 2017, 12, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Miyanga, S.; Sakurai, H.; Saiki, I.; Onaka, H.; Igarashi, Y. Synthesis and evaluation of myxochelins analogues as antimetastatic agents. Biol. Med. Chem. 2009, 17, 2724–2732. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, H.D.; Hartmann, V.; Pistorius, D.; Reissbrodt, R.; Trowitzsch-Kienast, W. Myxochelins B, C, D, E and F: A new structural principle for powerful siderophores imitating Nature. Eur. J. Org. Chem. 1998, 541–551. [Google Scholar] [CrossRef]
- Gaitatzis, N.; Kunze, B.; Müller, R. Novel insights into siderophore formation in myxobacteria. Chembiochem 2005, 6, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Nadmid, S.; Plaza, A.; Lauro, G.; Garcia, R.; Bifulco, G.; Müller, R. Hyalachelins A–C, Unusual Siderophores Isolated from the terrestrial Myxobacterium Hyalangium minutum. Org. Lett. 2014, 16, 4130–4133. [Google Scholar] [CrossRef] [PubMed]
- Kunze, B.; Trowitzsch-Kienast, W.; Hofle, G.; Reichenbach, H. Nannochelins A, B and C, new iron-chelating compounds from Nannocystis exedens (myxobacteria) production, isolation, physico-chemical and biological properties. J. Antibiot. 1991, 45, 147–150. [Google Scholar] [CrossRef]
- Saha, M.; Sarkar, S.; Sarkar, B.; Sharma, B.K.; Bhattacharjee, S.; Prosun, T. Microbial siderophores and their potential applications: A review. Environ. Sci. Pollut. Res. 2016, 23, 3984–3999. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Ebola virus Disease. Available online: http://www.who.int/en/news-room/fact-sheets/detail/ebola-virus-disease (accessed on 5 May 2018).
- Beck, S.; Henß, L.; Weidner, T.; Herrmann, J.; Müller, R.; Chao, Y.; Weber, C.; Sliva, K.; Schnierle, S. Identification of inhibitors of Ebola virus pseudotyped vectors from a myxobacterial compound library. Antivir. Res. 2016, 132, 85–91. [Google Scholar] [CrossRef] [PubMed]
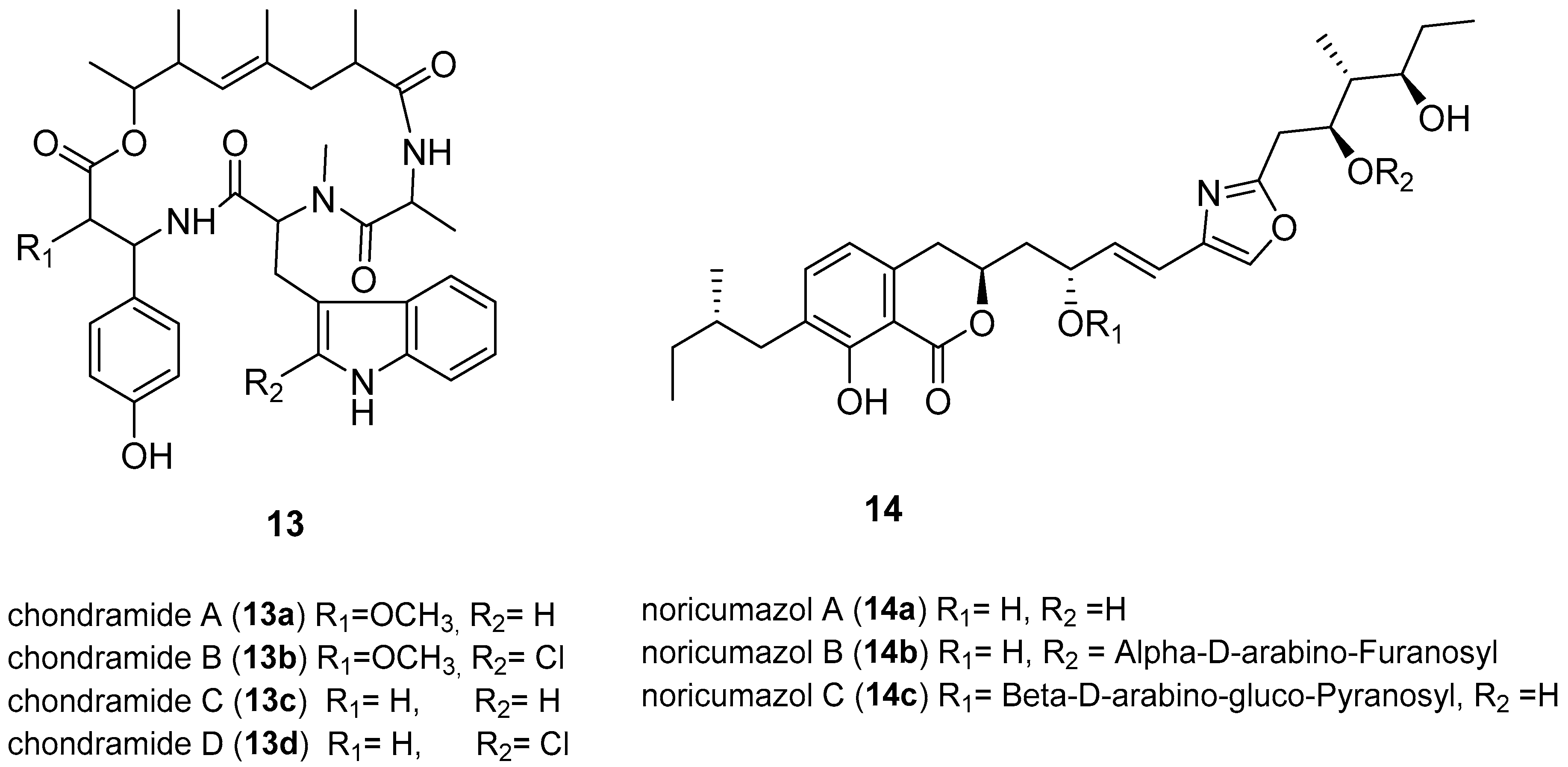
- Kunze, B.; Jansen, R.; Sasse, F.; Höflen, G.; Reichenbach, H.; Chondramides, A.D. New antifungal and cytostatic depsipeptides from Chondromyces crocatus (Myxobacteria). Production, physico-chemical and biological Properties. J. Antibiot. 1995, 48, 1262–1266. [Google Scholar] [CrossRef] [PubMed]
- Reichenbach, H. Myxobacteria: A source of new antibiotics. Trends Biotechnol. 1988, 6, 115–121. [Google Scholar] [CrossRef]
- Barbier, J.; Jansen, R.; Irschik, H.; Benson, S.; Gerth, K.; Böhlendorf, B.; Höfle, G.; Reichenbach, H.; Wegner, J.; Zeilinger, C.; et al. Isolation and total synthesis of icumazoles and noricumazoles—Antifungal antibiotics and cation-channel blockers from Sorangium cellulosum. Angew. Chem. Int. Ed. 2012, 51, 1256–1260. [Google Scholar] [CrossRef] [PubMed]
- Sasse, F.; Steinmetz, H.; Heil, J.; Höfle, G.; Reichenbach, H. Tubulysins, new cytostatic peptides from Myxobacteria acting on microtubuli. Production, isolation, physico-chemical and biological properties. J. Antibiot. 2000, 53, 879–885. [Google Scholar] [CrossRef] [PubMed]
- El-Serag, H.B. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 2012, 142, 1264–1273. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Hepatitis C. Available online: http://www.who.int/en/news-room/fact-sheets/detail/hepatitis-c (accessed on 10 May 2018).
- Shepard, C.W.; Finelli, L.; Alter, M.J. Global epidemiology of hepatitis C virus infection. Lancet Infect. Dis. 2005, 5, 558–567. [Google Scholar] [CrossRef]
- Mulwa, L.; Jansen, R.; Praditya, D.; Mohr, K.; Wink, J.; Steinmann, E.; Stadler, M. Six heterocyclic metabolites from the Myxobacterium Labilithrix luteola. Molecules 2018, 23, 542. [Google Scholar] [CrossRef] [PubMed]
- Bedorf, N.; Schomburg, D.; Gerth, K.; Reichenbach, H.; Höfle, G.; Bedorf, N.; Schomburg, D.; Gerth, K.; Reichenbach, H.; Höfle, G. Isolation and structure elucidation of soraphen A1α, a novel antifungal macrolide from Sorangium cellulosum. Liebigs Ann. Chem. 1993, 1993, 1017–1021. [Google Scholar] [CrossRef]
- Singaravelu, R.; Desrochers, G.F.; Srinivasan, P.; O’Hara, S.; Lyn, R.; Müller, R.; Jones, D.M.; Russell, R.; Pezacki, J.P. Soraphen A: A probe for investigating the role of de novo lipogenesis during viral infection. ACS Infect. Dis. 2015, 1, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Gentzsch, J.; Hinkelmann, B.; Kaderali, L.; Irschik, H.; Jansen, R.; Sasse, F.; Frank, R.; Pietschmann, T. Hepatitis C virus complete life cycle screen for identification of small molecules with pro- or antiviral activity. Antiviral Res. 2011, 89, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Mulwa, L.S.; Jansen, R.; Praditya, D.F.; Mohr, K.I.; Okanya, P.W.; Wink, J.; Steinmann, E.; Stadler, M. Lanyamycin, a macrolide antibiotic from Sorangium cellulosum, strain Soce 481 (Myxobacteria). Beilstein J. Org. Chem. 2018, 14, 1554–1562. [Google Scholar] [CrossRef] [PubMed]
- Yeganeh, B.; Ghavami, S.; Kroeker, A.L.; Mahood, T.H.; Stelmack, G.L.; Klonisch, T.; Coombs, K.M.; Halayko, A.J. Suppression of influenza A virus replication in human lung epithelial cells by noncytotoxic concentrations bafilomycin A1. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 308, 270–286. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, T.; Krug, D.; Bozkurt, N.; Duddela, S.; Jansen, R.; Garcia, R.; Gerth, K.; Steinmetz, H.; Müller, R. Correlating chemical diversity with taxonomic distance for discovery of natural products in myxobacteria. Nat. Commum. 2018, 9, 803. [Google Scholar] [CrossRef] [PubMed]
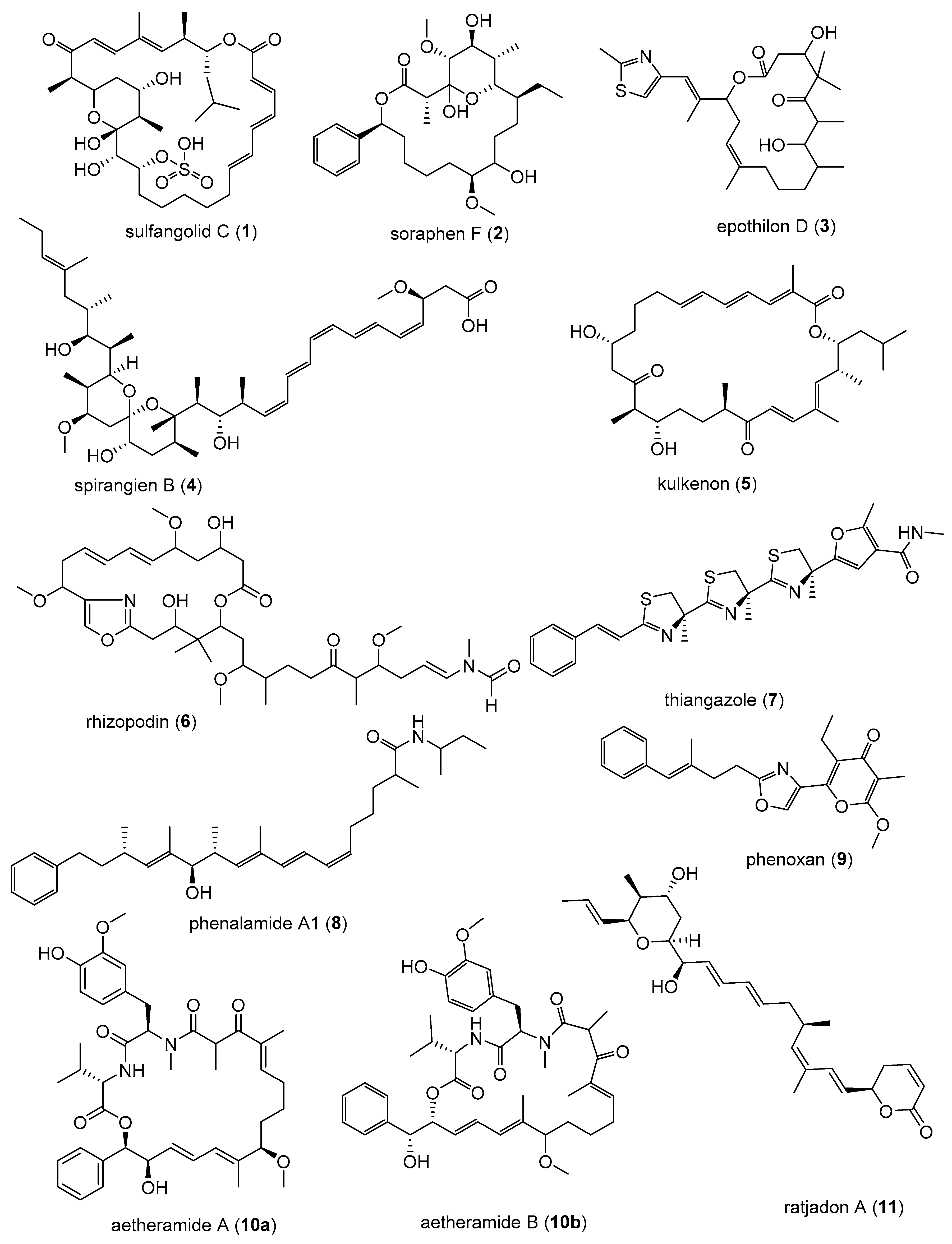
| Compound | MW 1 | EC50 (µM) | CC50 (µM) | SI ** |
|---|---|---|---|---|
| Nevirapine * | 266 | 0.07 | 81.8 | >103 |
| sulfangolid C (1) | 682 | 0.41 | 8.18 | 20.2 |
| soraphen F (2) | 522 | 0.30 | 5.02 | 16.5 |
| epothilon D (3) | 491 | 0.0005 | 0.012 | 24.4 |
| spirangien B (4) | 717 | 0.007 | 0.35 | 52 |
| kulkenon (5) | 734 | 0.07 | 0.36 | 5.3 |
| Compound | MW 1 | Tx 2 (nM) | AE 3 (µM) | SI ** |
|---|---|---|---|---|
| AZT * | 267 | 250,000 | 25 | 104 |
| thiangazole (7) | 539 | >4700 | 0.0047 | >106 |
| phenalamide A1 (8) | 491 | 102,000 | 1.02 | 105 |
| Phenoxan (9) | 379 | >6600 | 6.6 | >103 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mulwa, L.S.; Stadler, M. Antiviral Compounds from Myxobacteria. Microorganisms 2018, 6, 73. https://doi.org/10.3390/microorganisms6030073
Mulwa LS, Stadler M. Antiviral Compounds from Myxobacteria. Microorganisms. 2018; 6(3):73. https://doi.org/10.3390/microorganisms6030073
Chicago/Turabian StyleMulwa, Lucky S., and Marc Stadler. 2018. "Antiviral Compounds from Myxobacteria" Microorganisms 6, no. 3: 73. https://doi.org/10.3390/microorganisms6030073
APA StyleMulwa, L. S., & Stadler, M. (2018). Antiviral Compounds from Myxobacteria. Microorganisms, 6(3), 73. https://doi.org/10.3390/microorganisms6030073