Approaches to Dispersing Medical Biofilms
Abstract
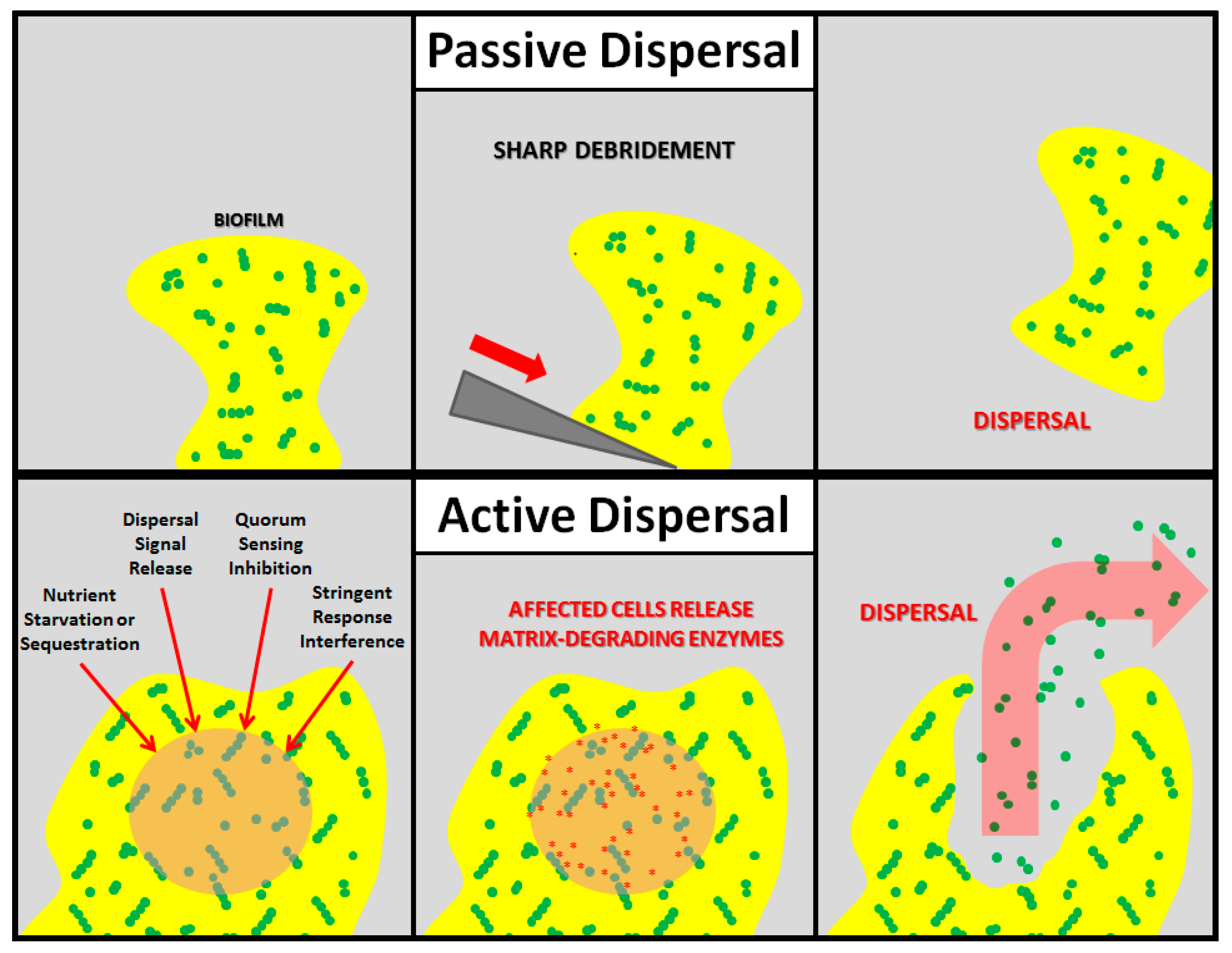
:1. Introduction
2. Enzymes
2.1. Proteases
2.2. Deoxyribonucleases
2.3. Glycoside Hydrolases
3. Antibiofilm Peptides
4. Dispersal Molecules
5. Hurdles to Development
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Rumbaugh, K.P.; Diggle, S.P.; Watters, C.M.; Ross-Gillespie, A.; Griffin, A.S.; West, S.A. Quorum sensing and the social evolution of bacterial virulence. Curr. Biol. 2009, 19, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Karatan, E.; Watnick, P. Signals, regulatory networks, and materials that build and break bacterial biofilms. Microbiol. Mol. Biol. Rev. 2009, 73, 310–347. [Google Scholar] [CrossRef] [PubMed]
- Attinger, C.; Wolcott, R. Clinically addressing biofilm in chronic wounds. Adv. Wound Care 2012, 1, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Romling, U.; Balsalobre, C. Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 2012, 272, 541–561. [Google Scholar] [CrossRef] [PubMed]
- Rogers, S.A.; Huigens, R.W., III; Cavanagh, J.; Melander, C. Synergistic effects between conventional antibiotics and 2-aminoimidazole-derived antibiofilm agents. Antimicrob. Agents Chemother. 2010, 54, 2112–2118. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K. Persister cells: Molecular mechanisms related to antibiotic tolerance. Handb. Exp. Pharmacol. 2012, 211, 121–133. [Google Scholar]
- Jiao, Y.; Cody, G.D.; Harding, A.K.; Wilmes, P.; Schrenk, M.; Wheeler, K.E.; Banfield, J.F.; Thelen, M.P. Characterization of extracellular polymeric substances from acidophilic microbial biofilms. Appl. Environ. Microbiol. 2010, 76, 2916–2922. [Google Scholar] [CrossRef] [PubMed]
- Lasa, I.; Penades, J.R. Bap: A family of surface proteins involved in biofilm formation. Res. Microbiol. 2006, 157, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Speziale, P.; Pietrocola, G.; Foster, T.J.; Geoghegan, J.A. Protein-based biofilm matrices in Staphylococci. Front. Cell. Infect. Microbiol. 2014, 4, 171. [Google Scholar] [CrossRef] [PubMed]
- Muthukrishnan, G.; Quinn, G.A.; Lamers, R.P.; Diaz, C.; Cole, A.L.; Chen, S.; Cole, A.M. Exoproteome of Staphylococcus aureus reveals putative determinants of nasal carriage. J. Proteome Res. 2011, 10, 2064–2078. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Bishop, P.L. Biodegradability of biofilm extracellular polymeric substances. Chemosphere 2003, 50, 63–69. [Google Scholar] [CrossRef]
- Kaplan, J.B. Biofilm dispersal: Mechanisms, clinical implications, and potential therapeutic uses. J. Dent. Res. 2010, 89, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Hobley, L.; Harkins, C.; MacPhee, C.E.; Stanley-Wall, N.R. Giving structure to the biofilm matrix: An overview of individual strategies and emerging common themes. FEMS Microbiol. Rev. 2015, 39, 649–669. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.; Golonka, E.; Potempa, J.; Foster, S.J. The role and regulation of the extracellular proteases of Staphylococcus aureus. Microbiology 2004, 150, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Marti, M.; Trotonda, M.P.; Tormo-Mas, M.A.; Vergara-Irigaray, M.; Cheung, A.L.; Lasa, I.; Penades, J.R. Extracellular proteases inhibit protein-dependent biofilm formation in Staphylococcus aureus. Microbes Infect. 2010, 12, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Abraham, N.M.; Jefferson, K.K. Staphylococcus aureus clumping factor B mediates biofilm formation in the absence of calcium. Microbiology 2012, 158, 1504–1512. [Google Scholar] [CrossRef] [PubMed]
- Mootz, J.M.; Malone, C.L.; Shaw, L.N.; Horswill, A.R. Staphopains modulate Staphylococcus aureus biofilm integrity. Infect. Immun. 2013, 81, 3227–3238. [Google Scholar] [CrossRef] [PubMed]
- Loughran, A.J.; Atwood, D.N.; Anthony, A.C.; Harik, N.S.; Spencer, H.J.; Beenken, K.E.; Smeltzer, M.S. Impact of individual extracellular proteases on Staphylococcus aureus biofilm formation in diverse clinical isolates and their isogenic sara mutants. Microbiologyopen 2014, 3, 897–909. [Google Scholar] [CrossRef] [PubMed]
- Gjermansen, M.; Nilsson, M.; Yang, L.; Tolker-Nielsen, T. Characterization of starvation-induced dispersion in Pseudomonas putida biofilms: Genetic elements and molecular mechanisms. Mol. Microbiol. 2010, 75, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Rao, T.S. Dispersal of Bap-mediated Staphylococcus aureus biofilm by proteinase K. J. Antibiot. 2013, 66, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, U.T.; Burrows, L.L. DNase I and proteinase K impair listeria monocytogenes biofilm formation and induce dispersal of pre-existing biofilms. Int. J. Food Microbiol. 2014, 187, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Ma, C.; Lin, L. Co-loaded proteinase K/thyme oil liposomes for inactivation of Escherichia coli O157:H7 biofilms on cucumber. Food Funct. 2016, 7, 4030–4040. [Google Scholar] [CrossRef] [PubMed]
- Chaignon, P.; Sadovskaya, I.; Ragunah, C.; Ramasubbu, N.; Kaplan, J.B.; Jabbouri, S. Susceptibility of staphylococcal biofilms to enzymatic treatments depends on their chemical composition. Appl. Microbiol. Biotechnol. 2007, 75, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.L.; Girerd, P.H.; Karjane, N.W.; Jefferson, K.K. Effect of biofilm phenotype on resistance of Gardnerella vaginalis to hydrogen peroxide and lactic acid. Am. J. Obstet. Gynecol. 2007, 197, 170.e1–170.e7. [Google Scholar] [CrossRef] [PubMed]
- Fredheim, E.G.; Klingenberg, C.; Rohde, H.; Frankenberger, S.; Gaustad, P.; Flaegstad, T.; Sollid, J.E. Biofilm formation by Staphylococcus haemolyticus. J. Clin. Microbiol. 2009, 47, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Izano, E.A.; Shah, S.M.; Kaplan, J.B. Intercellular adhesion and biocide resistance in nontypeable haemophilus influenzae biofilms. Microb. Pathog. 2009, 46, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Medina, A.A.; Kadouri, D.E. Biofilm formation of bdellovibrio bacteriovorus host-independent derivatives. Res. Microbiol. 2009, 160, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Boles, B.R.; Horswill, A.R. Agr-mediated dispersal of Staphylococcus aureus biofilms. PLoS Pathog. 2008, 4, e1000052. [Google Scholar] [CrossRef] [PubMed]
- Lauderdale, K.J.; Boles, B.R.; Cheung, A.L.; Horswill, A.R. Interconnections between sigma b, agr, and proteolytic activity in Staphylococcus aureus biofilm maturation. Infect. Immun. 2009, 77, 1623–1635. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.C.; Garbe, J.; Collin, M. Cysteine proteinase SpeB from Streptococcus pyogenes—A potent modifier of immunologically important host and bacterial proteins. Biol. Chem. 2011, 392, 1077–1088. [Google Scholar] [CrossRef] [PubMed]
- Connolly, K.L.; Roberts, A.L.; Holder, R.C.; Reid, S.D. Dispersal of group a streptococcal biofilms by the cysteine protease SpeB leads to increased disease severity in a murine model. PLoS ONE 2011, 6, e18984. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.F.; Li, Y.H.; Bowden, G.H. Detachment of Streptococcus mutans biofilm cells by an endogenous enzymatic activity. Infect. Immun. 1996, 64, 1035–1038. [Google Scholar] [PubMed]
- Banar, M.; Emaneini, M.; Satarzadeh, M.; Abdellahi, N.; Beigverdi, R.; Leeuwen, W.B.; Jabalameli, F. Evaluation of mannosidase and trypsin enzymes effects on biofilm production of Pseudomonas aeruginosa isolated from burn wound infections. PLoS ONE 2016, 11, e0164622. [Google Scholar] [CrossRef] [PubMed]
- Niazi, S.A.; Clark, D.; Do, T.; Gilbert, S.C.; Foschi, F.; Mannocci, F.; Beighton, D. The effectiveness of enzymic irrigation in removing a nutrient-stressed endodontic multispecies biofilm. Int. Endod. J. 2014, 47, 756–768. [Google Scholar] [CrossRef] [PubMed]
- McGavin, M.J.; Zahradka, C.; Rice, K.; Scott, J.E. Modification of the Staphylococcus aureus fibronectin binding phenotype by v8 protease. Infect. Immun. 1997, 65, 2621–2628. [Google Scholar] [PubMed]
- Okshevsky, M.; Meyer, R.L. The role of extracellular DNA in the establishment, maintenance and perpetuation of bacterial biofilms. Crit. Rev. Microbiol. 2015, 41, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Sehar, S.; Manefield, M. The roles of extracellular DNA in the structural integrity of extracellular polymeric substance and bacterial biofilm development. Environ. Microbiol. Rep. 2013, 5, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Jakubovics, N.S.; Shields, R.C.; Rajarajan, N.; Burgess, J.G. Life after death: The critical role of extracellular DNA in microbial biofilms. Lett. Appl. Microbiol. 2013, 57, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Whitchurch, C.B.; Tolker-Nielsen, T.; Ragas, P.C.; Mattick, J.S. Extracellular DNA required for bacterial biofilm formation. Science 2002, 295, 1487. [Google Scholar] [CrossRef] [PubMed]
- Alhede, M.; Bjarnsholt, T.; Givskov, M.; Alhede, M. Pseudomonas aeruginosa biofilms: Mechanisms of immune evasion. Adv. Appl. Microbiol. 2014, 86, 1–40. [Google Scholar] [PubMed]
- Seper, A.; Fengler, V.H.; Roier, S.; Wolinski, H.; Kohlwein, S.D.; Bishop, A.L.; Camilli, A.; Reidl, J.; Schild, S. Extracellular nucleases and extracellular DNA play important roles in Vibrio cholerae biofilm formation. Mol. Microbiol. 2011, 82, 1015–1037. [Google Scholar] [CrossRef] [PubMed]
- Tetz, V.V.; Tetz, G.V. Effect of extracellular DNA destruction by DNase I on characteristics of forming biofilms. DNA Cell Biol. 2010, 29, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Sahu, P.K.; Iyer, P.S.; Oak, A.M.; Pardesi, K.R.; Chopade, B.A. Characterization of eDNA from the clinical strain Acinetobacter baumannii AIIMS 7 and its role in biofilm formation. Sci. World J. 2012, 2012, 973436. [Google Scholar] [CrossRef] [PubMed]
- Tetz, G.V.; Artemenko, N.K.; Tetz, V.V. Effect of DNase and antibiotics on biofilm characteristics. Antimicrob. Agents Chemother. 2009, 53, 1204–1209. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Shingaki, R.; Sogawa, N.; Sogawa, C.A.; Asaumi, J.; Kokeguchi, S.; Fukui, K. Biofilm formation by a fimbriae-deficient mutant of Actinobacillus actinomycetemcomitans. Microbiol. Immunol. 2003, 47, 877–881. [Google Scholar] [CrossRef] [PubMed]
- Godeke, J.; Paul, K.; Lassak, J.; Thormann, K.M. Phage-induced lysis enhances biofilm formation in shewanella oneidensis MR-1. ISME J. 2011, 5, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Conover, M.S.; Mishra, M.; Deora, R. Extracellular DNA is essential for maintaining bordetella biofilm integrity on abiotic surfaces and in the upper respiratory tract of mice. PLoS ONE 2011, 6, e16861. [Google Scholar] [CrossRef] [PubMed]
- Svensson, S.L.; Davis, L.M.; MacKichan, J.K.; Allan, B.J.; Pajaniappan, M.; Thompson, S.A.; Gaynor, E.C. The cprs sensor kinase of the zoonotic pathogen Campylobacter jejuni influences biofilm formation and is required for optimal chick colonization. Mol. Microbiol. 2009, 71, 253–272. [Google Scholar] [CrossRef] [PubMed]
- Rice, K.C.; Mann, E.E.; Endres, J.L.; Weiss, E.C.; Cassat, J.E.; Smeltzer, M.S.; Bayles, K.W. The cida murein hydrolase regulator contributes to DNA release and biofilm development in Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2007, 104, 8113–8118. [Google Scholar] [CrossRef] [PubMed]
- Thomas, V.C.; Thurlow, L.R.; Boyle, D.; Hancock, L.E. Regulation of autolysis-dependent extracellular DNA release by Enterococcus faecalis extracellular proteases influences biofilm development. J. Bacteriol. 2008, 190, 5690–5698. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, M.; Lappann, M.; Knochel, S.; Molin, S. Role of extracellular DNA during biofilm formation by listeria monocytogenes. Appl. Environ. Microbiol. 2010, 76, 2271–2279. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.; Uppuluri, P.; Thomas, D.P.; Cleary, I.A.; Henriques, M.; Lopez-Ribot, J.L.; Oliveira, R. Presence of extracellular DNA in the candida albicans biofilm matrix and its contribution to biofilms. Mycopathologia 2010, 169, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, R.; Williams, C.; Lappin, D.F.; Millington, O.; Martins, M.; Ramage, G. Extracellular DNA release acts as an antifungal resistance mechanism in mature aspergillus fumigatus biofilms. Eukaryot. Cell 2013, 12, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Nemoto, K.; Hirota, K.; Murakami, K.; Taniguti, K.; Murata, H.; Viducic, D.; Miyake, Y. Effect of varidase (streptodornase) on biofilm formed by Pseudomonas aeruginosa. Chemotherapy 2003, 49, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Waryah, C.B.; Wells, K.; Ulluwishewa, D.; Chen-Tan, N.; Gogoi-Tiwari, J.; Ravensdale, J.; Costantino, P.; Gokcen, A.; Vilcinskas, A.; Wiesner, J.; et al. In vitro antimicrobial efficacy of tobramycin against Staphylococcus aureus biofilms in combination with or without DNase I and/or Dispersin B: A preliminary investigation. Microb. Drug Resist 2016. [Google Scholar] [CrossRef] [PubMed]
- Eckhart, L.; Fischer, H.; Barken, K.B.; Tolker-Nielsen, T.; Tschachler, E. DNase1L2 suppresses biofilm formation by Pseudomonas aeruginosa and Staphylococcus aureus. Br. J. Dermatol. 2007, 156, 1342–1345. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.B.; LoVetri, K.; Cardona, S.T.; Madhyastha, S.; Sadovskaya, I.; Jabbouri, S.; Izano, E.A. Recombinant human DNase I decreases biofilm and increases antimicrobial susceptibility in Staphylococci. J. Antibiot. 2012, 65, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Hall-Stoodley, L.; Nistico, L.; Sambanthamoorthy, K.; Dice, B.; Nguyen, D.; Mershon, W.J.; Johnson, C.; Hu, F.Z.; Stoodley, P.; Ehrlich, G.D.; et al. Characterization of biofilm matrix, degradation by DNase treatment and evidence of capsule downregulation in Streptococcus pneumoniae clinical isolates. BMC Microbiol. 2008, 8, 173. [Google Scholar] [CrossRef] [PubMed]
- Shields, R.C.; Mokhtar, N.; Ford, M.; Hall, M.J.; Burgess, J.G.; ElBadawey, M.R.; Jakubovics, N.S. Efficacy of a marine bacterial nuclease against biofilm forming microorganisms isolated from chronic rhinosinusitis. PLoS ONE 2013, 8, e55339. [Google Scholar] [CrossRef] [PubMed]
- Nijland, R.; Hall, M.J.; Burgess, J.G. Dispersal of biofilms by secreted, matrix degrading, bacterial DNase. PLoS ONE 2010, 5, e15668. [Google Scholar] [CrossRef] [PubMed]
- Shakir, A.; Elbadawey, M.R.; Shields, R.C.; Jakubovics, N.S.; Burgess, J.G. Removal of biofilms from tracheoesophageal speech valves using a novel marine microbial deoxyribonuclease. Otolaryngol. Head Neck Surg. 2012, 147, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Wingender, J.; Strathmann, M.; Rode, A.; Leis, A.; Flemming, H.C. Isolation and biochemical characterization of extracellular polymeric substances from Pseudomonas aeruginosa. Methods Enzymol. 2001, 336, 302–314. [Google Scholar] [PubMed]
- Bales, P.M.; Renke, E.M.; May, S.L.; Shen, Y.; Nelson, D.C. Purification and characterization of biofilm-associated eps exopolysaccharides from eskape organisms and other pathogens. PLoS ONE 2013, 8, e67950. [Google Scholar] [CrossRef] [PubMed]
- Limoli, D.H.; Jones, C.J.; Wozniak, D.J. Bacterial extracellular polysaccharides in biofilm formation and function. Microbiol. Spectr. 2015, 3. [Google Scholar] [CrossRef]
- Watters, C.; Fleming, D.; Bishop, D.; Rumbaugh, K.P. Host responses to biofilm. Prog. Mol. Biol. Transl. Sci. 2016, 142, 193–239. [Google Scholar] [PubMed]
- Wang, G.; Li, X.; Wang, Z. Apd3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016, 44, D1087–D1093. [Google Scholar] [CrossRef] [PubMed]
- Overhage, J.; Campisano, A.; Bains, M.; Torfs, E.C.; Rehm, B.H.; Hancock, R.E. Human host defense peptide LL-37 prevents bacterial biofilm formation. Infect. Immun. 2008, 76, 4176–4182. [Google Scholar] [CrossRef] [PubMed]
- Lamppa, J.W.; Griswold, K.E. Alginate lyase exhibits catalysis-independent biofilm dispersion and antibiotic synergy. Antimicrob. Agents Chemother. 2013, 57, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Hisano, T.; Nishimura, M.; Yonemoto, Y.; Abe, S.; Yamashita, T.; Sakaguchi, K.; Kimura, A.; Murata, K. Bacterial alginate lyase highly active on acetylated alginates. J. Ferment. Bioeng. 1993, 75, 332–335. [Google Scholar] [CrossRef]
- Alkawash, M.A.; Soothill, J.S.; Schiller, N.L. Alginate lyase enhances antibiotic killing of mucoid Pseudomonas aeruginosa in biofilms. APMIS 2006, 114, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Bayer, A.S.; Speert, D.P.; Park, S.; Tu, J.; Witt, M.; Nast, C.C.; Norman, D.C. Functional role of mucoid exopolysaccharide (alginate) in antibiotic-induced and polymorphonuclear leukocyte-mediated killing of Pseudomonas aeruginosa. Infect. Immun. 1991, 59, 302–308. [Google Scholar] [PubMed]
- Kalpana, B.J.; Aarthy, S.; Pandian, S.K. Antibiofilm activity of α-amylase from Bacillus subtilis s8-18 against biofilm forming human bacterial pathogens. Appl. Biochem. Biotechnol. 2012, 167, 1778–1794. [Google Scholar] [CrossRef] [PubMed]
- Craigen, B.; Dashiff, A.; Kadouri, D.E. The use of commercially available alpha-amylase compounds to inhibit and remove Staphylococcus aureus biofilms. Open Microbiol. J. 2011, 5, 21–31. [Google Scholar] [PubMed]
- Watters, C.M.; Burton, T.; Kirui, D.K.; Millenbaugh, N.J. Enzymatic degradation of in vitro Staphylococcus aureus biofilms supplemented with human plasma. Infect. Drug Resist. 2016, 9, 71–78. [Google Scholar] [PubMed]
- Fleming, D.; Chahin, L.; Rumbaugh, K. Glycoside hydrolases degrade polymicrobial bacterial biofilms in wounds. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Izano, E.A.; Wang, H.; Ragunath, C.; Ramasubbu, N.; Kaplan, J.B. Detachment and killing of aggregatibacter actinomycetemcomitans biofilms by dispersin B and SDS. J. Dent. Res. 2007, 86, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.B.; Ragunath, C.; Velliyagounder, K.; Fine, D.H.; Ramasubbu, N. Enzymatic detachment of staphylococcus epidermidis biofilms. Antimicrob. Agents Chemother. 2004, 48, 2633–2636. [Google Scholar] [CrossRef] [PubMed]
- Gawande, P.V.; Leung, K.P.; Madhyastha, S. Antibiofilm and antimicrobial efficacy of DispersinB®-KSL-W peptide-based wound gel against chronic wound infection associated bacteria. Curr. Microbiol. 2014, 68, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Fazekas, E.; Kandra, L.; Gyemant, G. Model for beta-1,6-n-acetylglucosamine oligomer hydrolysis catalysed by dispersinb, a biofilm degrading enzyme. Carbohydr. Res. 2012, 363, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Yakandawala, N.; Gawande, P.V.; LoVetri, K.; Cardona, S.T.; Romeo, T.; Nitz, M.; Madhyastha, S. Characterization of the poly-β-1,6-N-acetylglucosamine polysaccharide component of Burkholderia biofilms. Appl. Environ. Microbiol. 2011, 77, 8303–8309. [Google Scholar] [CrossRef] [PubMed]
- Izano, E.A.; Sadovskaya, I.; Vinogradov, E.; Mulks, M.H.; Velliyagounder, K.; Ragunath, C.; Kher, W.B.; Ramasubbu, N.; Jabbouri, S.; Perry, M.B.; et al. Poly-n-acetylglucosamine mediates biofilm formation and antibiotic resistance in actinobacillus pleuropneumoniae. Microb. Pathog. 2007, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Itoh, Y.; Wang, X.; Hinnebusch, B.J.; Preston, J.F., III; Romeo, T. Depolymerization of beta-1,6-N-acetyl-d-glucosamine disrupts the integrity of diverse bacterial biofilms. J. Bacteriol. 2005, 187, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Ibberson, C.B.; Parlet, C.P.; Kwiecinski, J.; Crosby, H.A.; Meyerholz, D.K.; Horswill, A.R. Hyaluronan modulation impacts Staphylococcus aureus biofilm infection. Infect. Immun. 2016, 84, 1917–1929. [Google Scholar] [CrossRef] [PubMed]
- Pecharki, D.; Petersen, F.C.; Scheie, A.A. Role of hyaluronidase in Streptococcus intermedius biofilm. Microbiology 2008, 154, 932–938. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.; Hill, P.J.; Snarr, B.D.; Alnabelseya, N.; Pestrak, M.J.; Lee, M.J.; Jennings, L.K.; Tam, J.; Melnyk, R.A.; Parsek, M.R.; et al. Exopolysaccharide biosynthetic glycoside hydrolases can be utilized to disrupt and prevent Pseudomonas aeruginosa biofilms. Sci. Adv. 2016, 2, e1501632. [Google Scholar] [CrossRef] [PubMed]
- Barraud, N.; Kelso, M.J.; Rice, S.A.; Kjelleberg, S. Nitric oxide: A key mediator of biofilm dispersal with applications in infectious diseases. Curr. Pharm. Des. 2015, 21, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Romling, U.; Galperin, M.Y.; Gomelsky, M. Cyclic di-GMP: The first 25 years of a universal bacterial second messenger. Microbiol. Mol. Biol. Rev. 2013, 77, 1–52. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente-Nunez, C.; Reffuveille, F.; Haney, E.F.; Straus, S.K.; Hancock, R.E. Broad-spectrum anti-biofilm peptide that targets a cellular stress response. PLoS Pathog. 2014, 10, e1004152. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; de la Fuente-Nunez, C.; Shen, Y.; Haapasalo, M.; Hancock, R.E. Treatment of oral multispecies biofilms by an anti-biofilm peptide. PLoS ONE 2015, 10, e0132512. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente-Nunez, C.; Korolik, V.; Bains, M.; Nguyen, U.; Breidenstein, E.B.; Horsman, S.; Lewenza, S.; Burrows, L.; Hancock, R.E. Inhibition of bacterial biofilm formation and swarming motility by a small synthetic cationic peptide. Antimicrob. Agents Chemother. 2012, 56, 2696–2704. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.; Golla, R.M.; Lau, K.; Lushnikova, T.; Wang, G. Anti-staphylococcal biofilm effects of human cathelicidin peptides. ACS Med. Chem. Lett. 2016, 7, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wang, W.; Fan, M.; Tong, Z.; Kuang, R.; Jiang, W.; Ni, L. Antimicrobial and anti-biofilm effect of Bac8c on major bacteria associated with dental caries and Streptococcus mutans biofilms. Peptides 2014, 52, 61–67. [Google Scholar] [CrossRef] [PubMed]
- De Zoysa, G.H.; Cameron, A.J.; Hegde, V.V.; Raghothama, S.; Sarojini, V. Antimicrobial peptides with potential for biofilm eradication: Synthesis and structure activity relationship studies of battacin peptides. J. Med. Chem. 2015, 58, 625–639. [Google Scholar] [CrossRef] [PubMed]
- Pompilio, A.; Scocchi, M.; Pomponio, S.; Guida, F.; Di Primio, A.; Fiscarelli, E.; Gennaro, R.; Di Bonaventura, G. Antibacterial and anti-biofilm effects of cathelicidin peptides against pathogens isolated from cystic fibrosis patients. Peptides 2011, 32, 1807–1814. [Google Scholar] [CrossRef] [PubMed]
- Scarsini, M.; Tomasinsig, L.; Arzese, A.; D’Este, F.; Oro, D.; Skerlavaj, B. Antifungal activity of cathelicidin peptides against planktonic and biofilm cultures of candida species isolated from vaginal infections. Peptides 2015, 71, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Dosler, S.; Mataraci, E. In vitro pharmacokinetics of antimicrobial cationic peptides alone and in combination with antibiotics against methicillin resistant Staphylococcus aureus biofilms. Peptides 2013, 49, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Dosler, S.; Karaaslan, E. Inhibition and destruction of Pseudomonas aeruginosa biofilms by antibiotics and antimicrobial peptides. Peptides 2014, 62, 32–37. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente-Nunez, C.; Reffuveille, F.; Mansour, S.C.; Reckseidler-Zenteno, S.L.; Hernandez, D.; Brackman, G.; Coenye, T.; Hancock, R.E. d-enantiomeric peptides that eradicate wild-type and multidrug-resistant biofilms and protect against lethal Pseudomonas aeruginosa infections. Chem. Biol. 2015, 22, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, S.M.; de la Fuente-Nunez, C.; Baquir, B.; Faria-Junior, C.; Franco, O.L.; Hancock, R.E. Antibiofilm peptides increase the susceptibility of carbapenemase-producing klebsiella pneumoniae clinical isolates to β-lactam antibiotics. Antimicrob. Agents Chemother. 2015, 59, 3906–3912. [Google Scholar] [CrossRef] [PubMed]
- Nagant, C.; Pitts, B.; Nazmi, K.; Vandenbranden, M.; Bolscher, J.G.; Stewart, P.S.; Dehaye, J.P. Identification of peptides derived from the human antimicrobial peptide LL-37 active against biofilms formed by Pseudomonas aeruginosa using a library of truncated fragments. Antimicrob. Agents Chemother. 2012, 56, 5698–5708. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Sambanthamoorthy, K.; Palys, T.; Paranavitana, C. The human antimicrobial peptide LL-37 and its fragments possess both antimicrobial and antibiofilm activities against multidrug-resistant Acinetobacter baumannii. Peptides 2013, 49, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Dean, S.N.; Bishop, B.M.; van Hoek, M.L. Susceptibility of Pseudomonas aeruginosa biofilm to alpha-helical peptides: d-enantiomer of LL-37. Front. Microbiol. 2011, 2, 128. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Gao, Y.; Lu, Z.; Yang, L. Effect of antibacterial peptide LL-37 on the integrity of Acinetobacter baumannii biofilm. Nan Fang Yi Ke Da Xue Xue Bao 2014, 34, 426–429. [Google Scholar] [PubMed]
- Dosler, S.; Karaaslan, E.; Alev Gerceker, A. Antibacterial and anti-biofilm activities of melittin and colistin, alone and in combination with antibiotics against Gram-negative bacteria. J. Chemother. 2016, 28, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Haisma, E.M.; de Breij, A.; Chan, H.; van Dissel, J.T.; Drijfhout, J.W.; Hiemstra, P.S.; El Ghalbzouri, A.; Nibbering, P.H. LL-37-derived peptides eradicate multidrug-resistant Staphylococcus aureus from thermally wounded human skin equivalents. Antimicrob. Agents Chemother. 2014, 58, 4411–4419. [Google Scholar] [CrossRef] [PubMed]
- Haisma, E.M.; Goblyos, A.; Ravensbergen, B.; Adriaans, A.E.; Cordfunke, R.A.; Schrumpf, J.; Limpens, R.W.; Schimmel, K.J.; den Hartigh, J.; Hiemstra, P.S.; et al. Antimicrobial peptide P60.4Ac-containing creams and gel for eradication of methicillin-resistant Staphylococcus aureus from cultured skin and airway epithelial surfaces. Antimicrob. Agents Chemother. 2016, 60, 4063–4072. [Google Scholar] [CrossRef] [PubMed]
- Boles, B.R.; Thoendel, M.; Singh, P.K. Rhamnolipids mediate detachment of Pseudomonas aeruginosa from biofilms. Mol. Microbiol. 2005, 57, 1210–1223. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, A.; Nusca, T.D.; Hochbaum, A.I. Rhamnolipids mediate an interspecies biofilm dispersal signaling pathway. ACS Chem. Biol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Quinn, G.A.; Maloy, A.P.; Banat, M.M.; Banat, I.M. A comparison of effects of broad-spectrum antibiotics and biosurfactants on established bacterial biofilms. Curr. Microbiol. 2013, 67, 614–623. [Google Scholar] [CrossRef] [PubMed]
- De Rienzo, M.A.; Martin, P.J. Effect of mono and di-rhamnolipids on biofilms pre-formed by Bacillus subtilis BBK006. Curr. Microbiol. 2016, 73, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Yang, Z.; Pu, M.; Peti, W.; Wood, T.K. Engineering a novel c-di-GMP-binding protein for biofilm dispersal. Environ. Microbiol. 2011, 13, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Lord, D.M.; Baran, A.U.; Wood, T.K.; Peti, W.; Page, R. BdcA, a protein important for Escherichia coli biofilm dispersal, is a short-chain dehydrogenase/reductase that binds specifically to NADPH. PLoS ONE 2014, 9, e105751. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Zhang, G.; Wood, T.K. Escherichia coli BdcA controls biofilm dispersal in Pseudomonas aeruginosa and Rhizobium meliloti. BMC Res. Notes 2011, 4, 447. [Google Scholar] [CrossRef] [PubMed]
- Dean, S.N.; Chung, M.C.; van Hoek, M.L. Burkholderia diffusible signal factor signals to francisella novicida to disperse biofilm and increase siderophore production. Appl. Environ. Microbiol. 2015, 81, 7057–7066. [Google Scholar] [CrossRef] [PubMed]
- Dow, J.M.; Crossman, L.; Findlay, K.; He, Y.Q.; Feng, J.X.; Tang, J.L. Biofilm dispersal in Xanthomonas campestris is controlled by cell-cell signaling and is required for full virulence to plants. Proc. Natl. Acad. Sci. USA 2003, 100, 10995–11000. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.G.; Marques, C.N. A fatty acid messenger is responsible for inducing dispersion in microbial biofilms. J. Bacteriol. 2009, 191, 1393–1403. [Google Scholar] [CrossRef] [PubMed]
- Sepehr, S.; Rahmani-Badi, A.; Babaie-Naiej, H.; Soudi, M.R. Unsaturated fatty acid, cis-2-decenoic acid, in combination with disinfectants or antibiotics removes pre-established biofilms formed by food-related bacteria. PLoS ONE 2014, 9, e101677. [Google Scholar] [CrossRef] [PubMed]
- Rahmani-Badi, A.; Sepehr, S.; Mohammadi, P.; Soudi, M.R.; Babaie-Naiej, H.; Fallahi, H. A combination of cis-2-decenoic acid and antibiotics eradicates pre-established catheter-associated biofilms. J. Med. Microbiol. 2014, 63, 1509–1516. [Google Scholar] [CrossRef] [PubMed]
- Rahmani-Badi, A.; Sepehr, S.; Babaie-Naiej, H. A combination of cis-2-decenoic acid and chlorhexidine removes dental plaque. Arch. Oral Biol. 2015, 60, 1655–1661. [Google Scholar] [CrossRef] [PubMed]
- Martinez, L.R.; Mihu, M.R.; Han, G.; Frases, S.; Cordero, R.J.; Casadevall, A.; Friedman, A.J.; Friedman, J.M.; Nosanchuk, J.D. The use of chitosan to damage Cryptococcus neoformans biofilms. Biomaterials 2010, 31, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Orgaz, B.; Lobete, M.M.; Puga, C.H.; San Jose, C. Effectiveness of chitosan against mature biofilms formed by food related bacteria. Int. J. Mol. Sci. 2011, 12, 817–828. [Google Scholar] [CrossRef] [PubMed]
- Martinez, L.R.; Mihu, M.R.; Tar, M.; Cordero, R.J.; Han, G.; Friedman, A.J.; Friedman, J.M.; Nosanchuk, J.D. Demonstration of antibiofilm and antifungal efficacy of chitosan against candidal biofilms, using an in vivo central venous catheter model. J. Infect. Dis. 2010, 201, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Mu, H.; Zhang, W.; Cui, G.; Zhu, J.; Duan, J. Chitosan coupling makes microbial biofilms susceptible to antibiotics. Sci. Rep. 2013, 3, 3364. [Google Scholar] [CrossRef] [PubMed]
- Mu, H.; Guo, F.; Niu, H.; Liu, Q.; Wang, S.; Duan, J. Chitosan improves anti-biofilm efficacy of gentamicin through facilitating antibiotic penetration. Int. J. Mol. Sci. 2014, 15, 22296–22308. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.J., Jr.; Akers, K.S.; Romano, D.R.; Woodbury, R.L.; Hardy, S.K.; Murray, C.K.; Wenke, J.C. d-amino acids enhance the activity of antimicrobials against biofilms of clinical wound isolates of Staphylococcus aureus and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2014, 58, 4353–4361. [Google Scholar] [CrossRef] [PubMed]
- Leiman, S.A.; May, J.M.; Lebar, M.D.; Kahne, D.; Kolter, R.; Losick, R. d-amino acids indirectly inhibit biofilm formation in Bacillus subtilis by interfering with protein synthesis. J. Bacteriol. 2013, 195, 5391–5395. [Google Scholar] [CrossRef] [PubMed]
- Kolodkin-Gal, I.; Romero, D.; Cao, S.; Clardy, J.; Kolter, R.; Losick, R. d-amino acids trigger biofilm disassembly. Science 2010, 328, 627–629. [Google Scholar] [CrossRef] [PubMed]
- Harmata, A.J.; Ma, Y.; Sanchez, C.J.; Zienkiewicz, K.J.; Elefteriou, F.; Wenke, J.C.; Guelcher, S.A. d-amino acid inhibits biofilm but not new bone formation in an ovine model. Clin. Orthop. Relat. Res. 2015, 473, 3951–3961. [Google Scholar] [CrossRef] [PubMed]
- Brindle, E.R.; Miller, D.A.; Stewart, P.S. Hydrodynamic deformation and removal of Staphylococcus epidermidis biofilms treated with urea, chlorhexidine, iron chloride, or dispersinb. Biotechnol. Bioeng. 2011, 108, 2968–2977. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Stewart, P.S. Biofilm removal caused by chemical treatments. Water Res. 2000, 34, 4229–4233. [Google Scholar] [CrossRef]
- Banin, E.; Brady, K.M.; Greenberg, E.P. Chelator-induced dispersal and killing of Pseudomonas aeruginosa cells in a biofilm. Appl. Environ. Microbiol. 2006, 72, 2064–2069. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lin, Y.; Lu, Q.; Li, F.; Yu, J.; Wang, Z.; He, Y.; Song, C. In vitro and in vivo activity of EDTA and antibacterial agents against the biofilm of mucoid Pseudomonas aeruginosa. Infection 2017, 45, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Jothiprakasam, V.; Sambantham, M.; Chinnathambi, S.; Vijayaboopathi, S. Candida tropicalis biofilm inhibition by ZnO nanoparticles and EDTA. Arch. Oral Biol. 2016, 73, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Maisetta, G.; Grassi, L.; Di Luca, M.; Bombardelli, S.; Medici, C.; Brancatisano, F.L.; Esin, S.; Batoni, G. Anti-biofilm properties of the antimicrobial peptide temporin 1Tb and its ability, in combination with EDTA, to eradicate Staphylococcus epidermidis biofilms on silicone catheters. Biofouling 2016, 32, 787–800. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, J.; Hoogenkamp, M.; Felippe, W.T.; Crielaard, W.; van der Waal, S.V. Effectiveness of EDTA and modified salt solution to detach and kill cells from Enterococcus faecalis biofilm. J. Endod. 2016, 42, 320–323. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, E.; Vighetto, C.; Di Martino, P.; Larreta Garde, V.; Seyer, D. Synergistic antibiofilm efficacy of various commercial antiseptics, enzymes and EDTA: A study of Pseudomonas aeruginosa and Staphylococcus aureus biofilms. Int. J. Antimicrob. Agents 2016, 48, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Ammons, M.C.; Copie, V. Mini-review: Lactoferrin: A bioinspired, anti-biofilm therapeutic. Biofouling 2013, 29, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.R.; Silva, M.G.; Rocas, I.N.; Siqueira, J.F., Jr. Biofilm biomass disruption by natural substances with potential for endodontic use. Braz. Oral Res. 2013, 27, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Brackman, G.; Coenye, T. Quorum sensing inhibitors as anti-biofilm agents. Curr. Pharm. Des. 2015, 21, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Uppuluri, P.; Lopez-Ribot, J.L. Go forth and colonize: Dispersal from clinically important microbial biofilms. PLoS Pathog. 2016, 12, e1005397. [Google Scholar] [CrossRef] [PubMed]
- Chua, S.L.; Liu, Y.; Yam, J.K.; Chen, Y.; Vejborg, R.M.; Tan, B.G.; Kjelleberg, S.; Tolker-Nielsen, T.; Givskov, M.; Yang, L. Dispersed cells represent a distinct stage in the transition from bacterial biofilm to planktonic lifestyles. Nat. Commun. 2014, 5, 4462. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Tan, K.S. The danger signal extracellular ATP is an inducer of Fusobacterium nucleatum biofilm dispersal. Front. Cell. Infect. Microbiol. 2016, 6, 155. [Google Scholar] [CrossRef] [PubMed]
- Wolcott, R.D.; Kennedy, J.P.; Dowd, S.E. Regular debridement is the main tool for maintaining a healthy wound bed in most chronic wounds. J. Wound Care 2009, 18, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Markakis, K.; Bowling, F.L.; Boulton, A.J. The diabetic foot in 2015: An overview. Diabetes Metab. Res. Rev. 2016, 32 (Suppl. 1), 169–178. [Google Scholar] [CrossRef] [PubMed]
| Enzyme | Summary | References |
|---|---|---|
| Aureolysin (Aur) | A staphylococcal metalloprotease that has been shown to disrupt S. aureus biofilms by degrading Bap and clumping factor b. | [17,20] |
| LapG Protease | A protease produced by Pseudomonas putida that has been shown to trigger dispersal through modification of the outer membrane-associated, exopolysaccharide-binding protein, LapA. | [21] |
| Proteinase K | A highly reactive and stable serine protease that exhibits a broad range of cleavage by targeting peptide bonds adjacent to the carboxylic group of aliphatic and aromatic amino acids. It is active against the biofilms produced by a range of bacteria, including S. aureus, Listeria monocytogenes, Staphylococcus lugdunensis, Staphylococcus heamolyticus, Gardnerella vaginalis, and Escherichia coli, Heamophilus influenza, and Bdellovibrio bacteriovorus. | [22,23,24,25,26,27,28,29] |
| Spl Proteases | A group of six Staphylococcal serine proteases that have been shown to be involved in S. aureus biofilm dispersal, possibly by cleaving the cell wall-associated protein, EbpS. | [30,31] |
| Staphopain A (ScpA), Staphopain B (SspB) | Staphylococcal cysteine proteases that have been shown to disperse S. aureus biofilms through degradation of (an) unknown target(s). | [19,20] |
| Streptococcal Cysteine Protease (SpeB) | A Streptococcus pyogenes cysteine protease that is historically known to be involved in immune evasion by the pathogen, owing to its cleavage of host immune molecules, as well as tissue invasion by way of host ECM degradation. However, SpeB has more recently been shown to be involved in dispersal of in vivo S. pyogenes biofilms via hydrolysis of surface proteins M and F1, which are hypothesized to be involved in microcolony formation. | [32,33] |
| Surface-protein-releasing enzyme (SPRE) | An endogenous Streptococcal protease that has been shown to cause Streptococcus mutans monolayer biofilm detachment from a colonized surface via the release of the surface protein, antigen P1. | [34] |
| Trypsin | A pancreatic serine protease that cleaves peptides at the carboxyl side of lysine or arginine. It is active against biofilms made by multiple bacterial species, including Pseudomonas aeruginosa, Streptococcus mitis, Actinomyces radicidentis, Staphylococcus epidermidis, and Gardnerella vaginalis. | [25,26,35,36] |
| V8 Serine Protease (SspA) | A staphylococcal serine protease that degrades fibronectin binding proteins and Bap in S. aureus biofilms. | [17,37] |
| Enzyme | Summary | References |
|---|---|---|
| DNase I | A pancreatic DNase that has been shown to deconstruct the established biofilms of a wide range of microbes, including P. aeruginosa, Vibrio cholerae, E. coli, S. pyogenes, Klebsiella pneumoniae, Acinetobacter baumannii, Aggregatibacter actinomycetemcomitans, Shewanella oneidensis, S. heamolyticus, Bordetella pertussis, Bordetella bronchiseptica, Campylobacter jejuni, H. influenza, B. bacteriovorus, S. aureus, Enterococcus faecalis, Listeria monocytogenes, Candida albicans, and Aspergillus fumigatus. | [27,28,29,41,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57] |
| DNase 1L2 | A human DNase found in keratinocytes that has been shown to degrade the established biofilms of P. aeruginosa and S. aureus. | [58] |
| Dornase alpha | A highly purified form of recombinant human DNase I (rhDNase I) that has been shown to be effective against the established biofilms of S. aureus and Streptococcus pneumoniae. | [59,60] |
| λ Exonuclease | A viral DNase that disrupts established V. cholerae biofilms. | [43] |
| NucB | A bacterial DNase produced by the marine bacterium, Bacillus licheniformis, which has been show able to degrade the established biofilms of multiple bacterial species, including B. licheniformis, S. aureus, S. epidermidis, Staphylococcus salivarius, Staphylococcus constellatus, S. Staphylococcus lugdunesis, Staphylococcus anginosus, E. coli, Streptococcus intermedius, Micrococcus luteus, and Bacillus subtilis. | [61,62,63] |
| Streptodornase | A streptococcal DNase that disrupts the established biofilms of P. aeruginosa. | [56] |
| Enzyme | Summary | References |
|---|---|---|
| Alginate lyase | A glycoside hydrolase that that degrades the exopolysaccharide, alginate, common in mucoid P. aeruginosa biofilms, causing bacterial cell dispersal and increasing antibiotic efficacy and phagocytosis. | [70,71,72,73] |
| α-amylase | A glycoside hydrolase that hydrolyzes α(1,4) glycosidic linkages and is derived from multiple sources, such as certain microbes and the mammalian pancreas. It has exhibited dispersal of mature biofilms formed by V. cholerae, S. aureus and P. aeruginosa. | [74,75,76,77] |
| α-mannosidase | An acid hydrolase that has been shown to disrupt P. aeruginosa biofilms. | [35] |
| β-mannosidase | A glycoside hydrolase that targets β(1,4)-linked terminal mannose residues, and disrupts P. aeruginosa biofilms. | [35] |
| Cellulase | A glycoside hydrolase produced by multiple microbes that hydrolyzes the β(1,4) glycosidic linkage. It has been shown to cause the dispersal of S. aureus and P. aeruginosa biofilms. | [77] |
| Dispersin B | A glycoside hydrolase produced by the bacterium, A. actinomycetemcomitans, that has been shown to degrade the polysaccharide, poly(1,6)-N-acetyl-d-glucosamine (PNAG), by hydrolyzing β(1,6) glycosidic linkages. This enzyme has been effective against the biofilms made by multiple bacteria, including S. aureus, A. actinomycetemcomitans, S. epidermidis, A. baumannii, K. pneumoniae, E. coli, Burkholderia spp., A. pleuropneumoniae, Yersinia pestis and Pseudomonas fluorescens. | [57,78,79,80,81,82,83,84] |
| Hyaluronidase | An enzyme that cleaves hyaluronic acid (HA), which has been found to be incorporated into the biofilms made by multiple pathogens, including S. aureus, and S. intermedius in vivo. When utilized against HA-containing biofilms, dispersal has been observed. | [85,86] |
| PelAh | A glycoside hydrolase that disrupts the P. aeruginosa polysaccharide, Pel, causing dispersal of mature biofilms. | [87] |
| PslGH | A glycoside hydrolase that disrupts the P. aeruginosa polysaccharide, Psl, causing dispersal of mature biofilms. | [87] |
| Peptide | Summary | References |
|---|---|---|
| 1018 | A synthetic, modified form of the cationic antimicrobial peptide bactenecin, which triggers the degradation of the (p)ppGpp bacterial stringent response signal. This peptide has been shown to be effective at disrupting the established biofilms of P. aeruginosa, E. coli, A. baumannii, K. pneumoniae, S. aureus, Salmonella Typhimurium, and Burkholderia cenocepacia. | [90,91] |
| 1037 | A 9-amino-acid, synthetic, cationic peptide derived from the human cathelicidin LL-37, which has demonstrated efficacy against biofilms made by P. aeruginosa, B. cenocepacia, and L. monocytogenes. | [92] |
| 17BIPHE2 | A 17-amino-acid derivative of the human cathelicidin, LL-37, that has had exhibited efficacy in disrupting S. aureus biofilms. | [93] |
| Bac8c | A 12-amino-acid, synthetic peptide modified from bactenecin that has exhibited efficacy against S. mutans biofilms. | [94] |
| Battacin | A native, cyclic lipopeptide produced by Paneibacillus tianmunesis, whose synthetic derivatives, especially lipopeptide 17, have been shown to degrade P. aeruginosa and S. aureus biofilms. | [95] |
| BMAP-27 | A synthetic, bovine cathelicidin-derived peptide that has exhibited efficacy against S. aureus, P. aeruginosa, and Stenotrophomonas maltophilia biofilms. | [96] |
| BMAP-28 | A synthetic, bovine cathelicidin-derived peptide that has exhibited efficacy against S. aureus, P. aeruginosa, and S. maltophilia biofilms. | [96,97] |
| CAMA | A hybrid, synthetic peptide that combines amino acid sequences from the silk moth peptide, cecropin-A, and the bee venom peptide, melittin. It has exhibited the ability to degrade P. aeruginosa and S. aureus biofilms. | [98,99] |
| DJK-5 | A synthetic, D-enantiomeric, protease-resistant peptide that works, in part, by degrading the (p)ppGpp bacterial stringent response signal. It has been shown to be effective at disrupting P. aeruginosa, A. baumannii, Salmonella enterica and K. pneumoniae biofilms. | [100] |
| DJK-6 | A synthetic, D-enantiomeric, protease-resistant peptide that works, in part, by degrading the (p)ppGpp bacterial stringent response signal. It has been shown to be effective at P. aeruginosa, A. baumannii, S. enterica and K. pneumoniae biofilms. | [100,101] |
| GF-17 | A 17-amino-acid derivative of the human cathelicidin, LL-37, that has exhibited efficacy in disrupting S. aureus biofilms. | [93] |
| LL-31 | A synthetic fragment of the human cathelicidin, LL-37, in which the last 6 amino acid residues are removed. The peptide has been shown to disrupt P. aeruginosa biofilms. | [102] |
| LL-37 | A 37-amino-acid, native human cathelicidin that has been shown to disrupt A. baumannii and P. aeruginosa biofilms. | [69,103,104,105] |
| LL7-31 | A synthetic fragment of the human cathelicidin, LL-37, in which the first 6, and last 6, amino acid residues are removed. The peptide has been shown to disrupt P. aeruginosa biofilms. | [102] |
| LL7-37 | A synthetic fragment of the human cathelicidin, LL-37, in which the first 6 amino acid residues are removed. The peptide has been shown to disrupt P. aeruginosa biofilms. | [102] |
| Melittin | A native, 26-amino-acid, haemolytic peptide, isolated from the venom of the European honey bee, Apis mellifer. The peptide has been efficacious against P. aeruginosa, E. coli and K. pneumonia biofilms. | [106] |
| P10 | A synthetic, 24-amino-acid peptide derived from the P60.4AC (which itself is a derivative of the human cathelicidin, LL-37) that has been shown to degrade S. aureus biofilms. | [107] |
| P60.4Ac | A synthetic, 24-amino-acid peptide derived from the human cathelicidin, LL-37, which has been shown to be effective at degrading S. aureus biofilms. | [107,108] |
| SMAP-29 | A synthetic, sheep cathelicidin-derived peptide that has exhibited efficacy against S. aureus, P. aeruginosa, and Stenotrophomonas maltophilia biofilms. | [96] |
| Molecule | Summary | References |
|---|---|---|
| Dispersal Signals | ||
| Cis-2-decenoic acid (CDA) | A type of fatty acid cross-kingdom signaling molecule, also known as a diffusible signal factor (DSF), which was originally found to be produced by P. aeruginosa. This particular DSF has been shown to trigger the dispersal of P. aeruginosa, E. coli, K. pneumoniae, P. mirabilis, S. pyogenes, B. subtilis, S. aureus, C. albicans, S. enterica, and S. mutans biofilms. It should be noted that other DSF’s, such as Burkholderia diffusible signal factor (BDSF) [116] and Xanthomonas diffusible signal factor (XDSF) [117], have been isolated and exhibit similar inductions of dispersal events. | [118,119,120,121] |
| Nitric oxide | An endogenously produced dispersal signal that is highly conserved across a wide range of microbial species. It has been shown to be involved in the dispersal of biofilms formed by P. aeruginosa, E. coli, Fusobacterium nucleatum, Serratia marcescens, V. cholerae, B. licheniformis, Shewanella woodyi, N. gonorrhoeae, Pseudoalteromonas, Vibrio fischeri, S. aureus, B. subtilis, Legionella pneumophila, Nitrosomonas europaea, P. putida, C. albicans, Candida tropicalis, and Ulva linza. | [88] |
| Anti-Matrix Molecules | ||
| Chitosan | A polycationic macromolecule derived from the polysaccharide, chitin that has been shown to penetrate and possibly disrupt Cryptococcus neoformans, L. monocytogenes, P. fluorescens, Bacillus cereus, S. enterica, C. albicans, and P. aeruginosa biofilms. It is important to note that it has not been proven that chitosan has any direct effect on the biofilm matrix, and it is possible that the molecule achieves biofilm disruption by penetrating the matrix and acting on the microbes themselves. | [122,123,124,125,126] |
| d-amino acids | d-isoforms of certain amino acids, including d-Leu, d-Met, d-Trp, d-Tyr, and d-Phe have been shown to cause the disassembly of biofilms though to multiple, hypothesized mechanisms, including (1) inhibition of genes involved in EPS production; and (2) incorporation of d-amino acids into the bacterial cell wall, resulting in the loss of cell-surface fibers vital to biofilm formation. d-amino acids have exhibited efficacy against S. aureus, P. aeruginosa, and B. subtilis biofilms. | [127,128,129,130] |
| Rhamnolipids | A microbial-produced surfactant that, at normal levels, is important for biofilm maturation in the form of fluid channel maintenance and cellular migration. At elevated levels, however, these rhamnolipids have been shown to trigger the dispersal of P. aeruginosa, E. coli, S. aureus, B. subtilis, M. luteus, and Yarrowia lipolytica biofilms. | [109,110,111,112] |
| Urea | An amide that is theorized to break down biofilms by disrupting the hydrogen bonds that are vital for EPS mechanical stability. The compound has exhibited dispersal ability against S. epidermidis, P. aeruginosa and K. pneumoniae biofilms. | [131,132] |
| Sequestration Molecules | ||
| BdcA | A protein that directly reduces unbound c-di-GMP concentrations by binding, but not degrading, the molecules, causing biofilm-related cellular mechanisms not to be activated. BdcA has been shown to cause the dispersal of E. coli, P. aeruginosa, P. fluorescens, and Rhizobium meliloti biofilms. | [113,114,115] |
| EDTA | Ethylenediaminetetraacetic acid (EDTA) is a metal-ion chelator that can sequester integral, EPS-matrix-stabilizing ions, and triggers dispersal in P. aeruginosa, H. influenzae, S. epidermidis, C. tropicalis, and E. faecalis biofilms. | [133,134,135,136,137,138] |
| Lactoferrin | An iron-binding protein from the innate immune system that is found in a variety of bodily fluids. By chelating iron, an essential bacterial nutrient and global regulator of a variety of functions, including biofilm development and growth, lactoferrin can trigger active dispersal. It has been shown to be effective against P. aeruginosa, E. coli, S. aureus, E. faecalis and S. epidermidis biofilms. | [139,140] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fleming, D.; Rumbaugh, K.P. Approaches to Dispersing Medical Biofilms. Microorganisms 2017, 5, 15. https://doi.org/10.3390/microorganisms5020015
Fleming D, Rumbaugh KP. Approaches to Dispersing Medical Biofilms. Microorganisms. 2017; 5(2):15. https://doi.org/10.3390/microorganisms5020015
Chicago/Turabian StyleFleming, Derek, and Kendra P. Rumbaugh. 2017. "Approaches to Dispersing Medical Biofilms" Microorganisms 5, no. 2: 15. https://doi.org/10.3390/microorganisms5020015
APA StyleFleming, D., & Rumbaugh, K. P. (2017). Approaches to Dispersing Medical Biofilms. Microorganisms, 5(2), 15. https://doi.org/10.3390/microorganisms5020015






