Seasonal Variation in In Hospite but Not Free-Living, Symbiodiniaceae Communities Around Hainan Island, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Location
2.2. Sample Collection and Environmental Factors Determination
2.3. DNA Extraction and Amplification of Symbiodiniaceae ITS2 rDNA
2.4. Sequencing and Bioinformatic Analysis
2.5. Statistical Analysis and Visualization
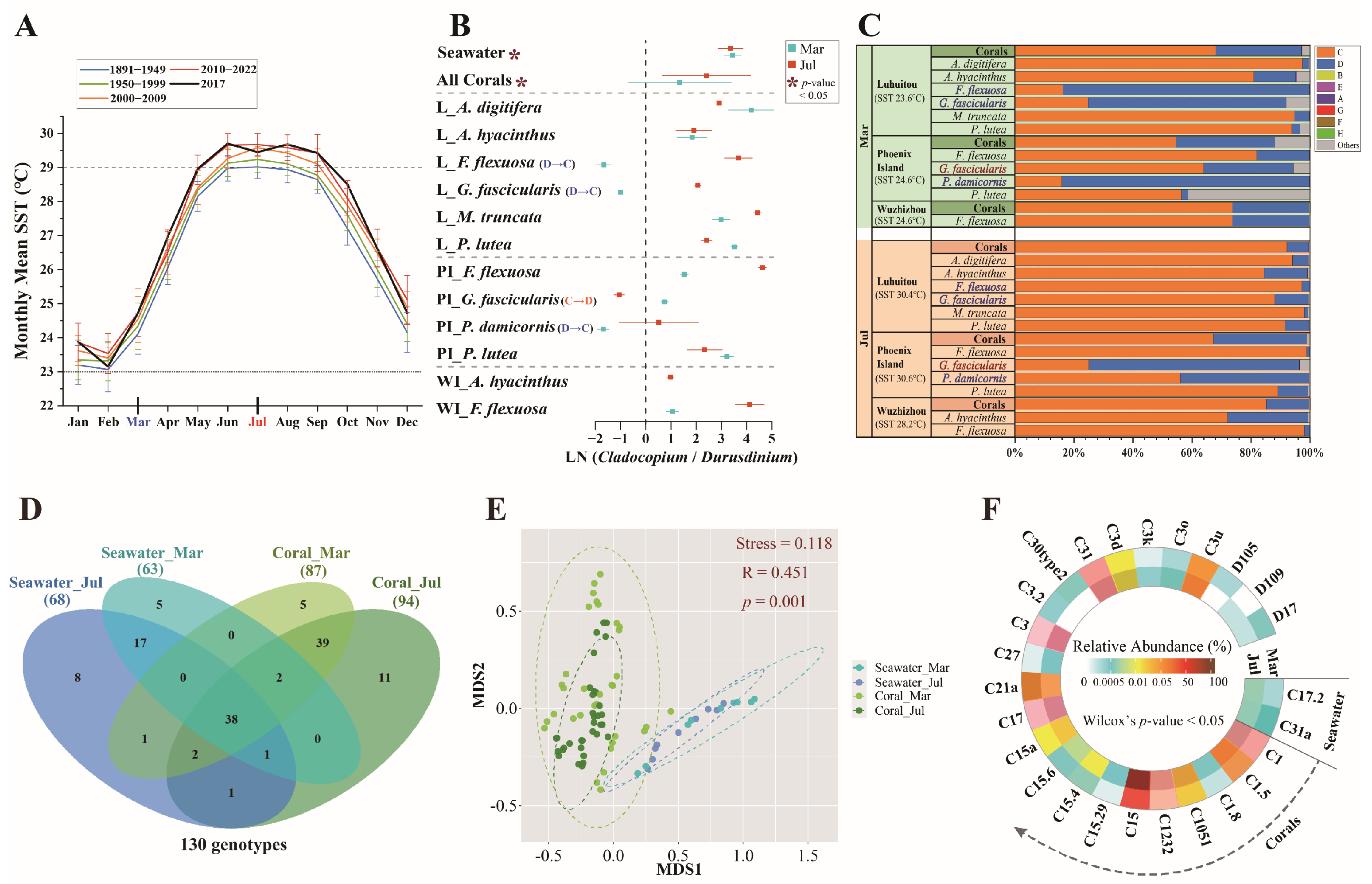
3. Results
3.1. Differences in Symbiodiniaceae Genetic Diversity Between Cold and Warm Months
3.2. Variations in Symbiodiniaceae Community Between Cold and Warm Months
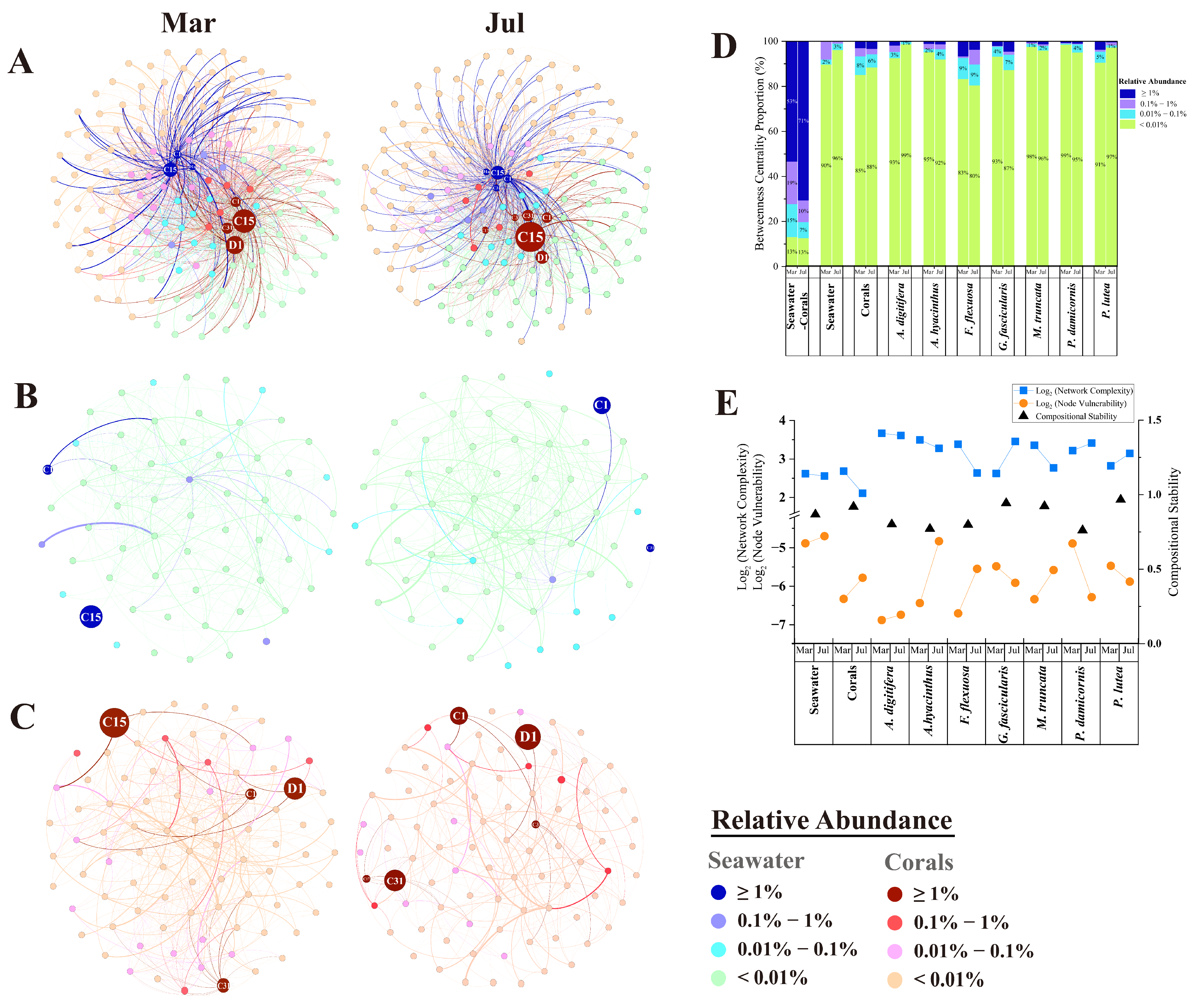
3.3. Network Analysis of Genotypes Within Symbiodiniaceae Community
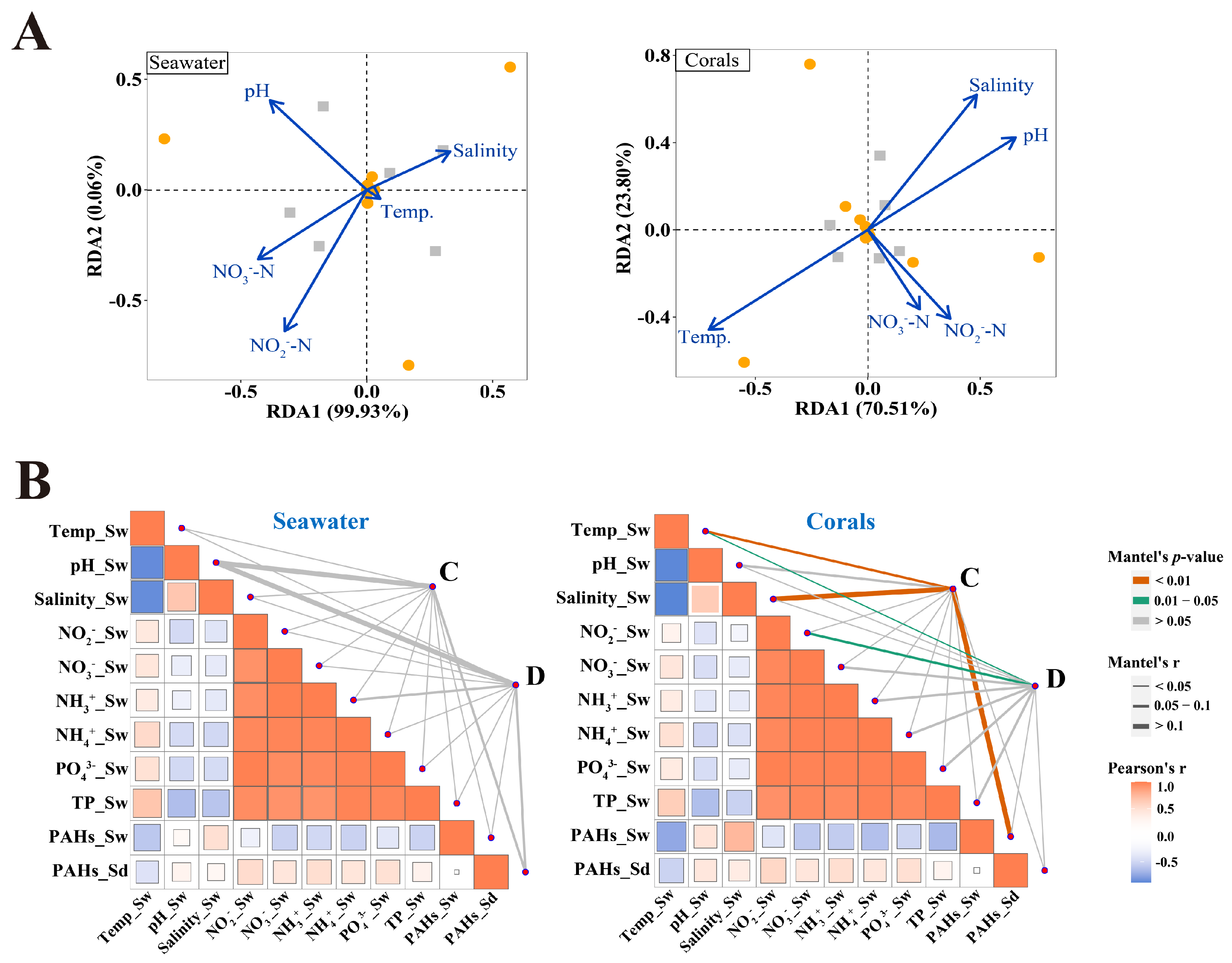
3.4. Correlations of Symbiodiniaceae Community with Environmental Factors
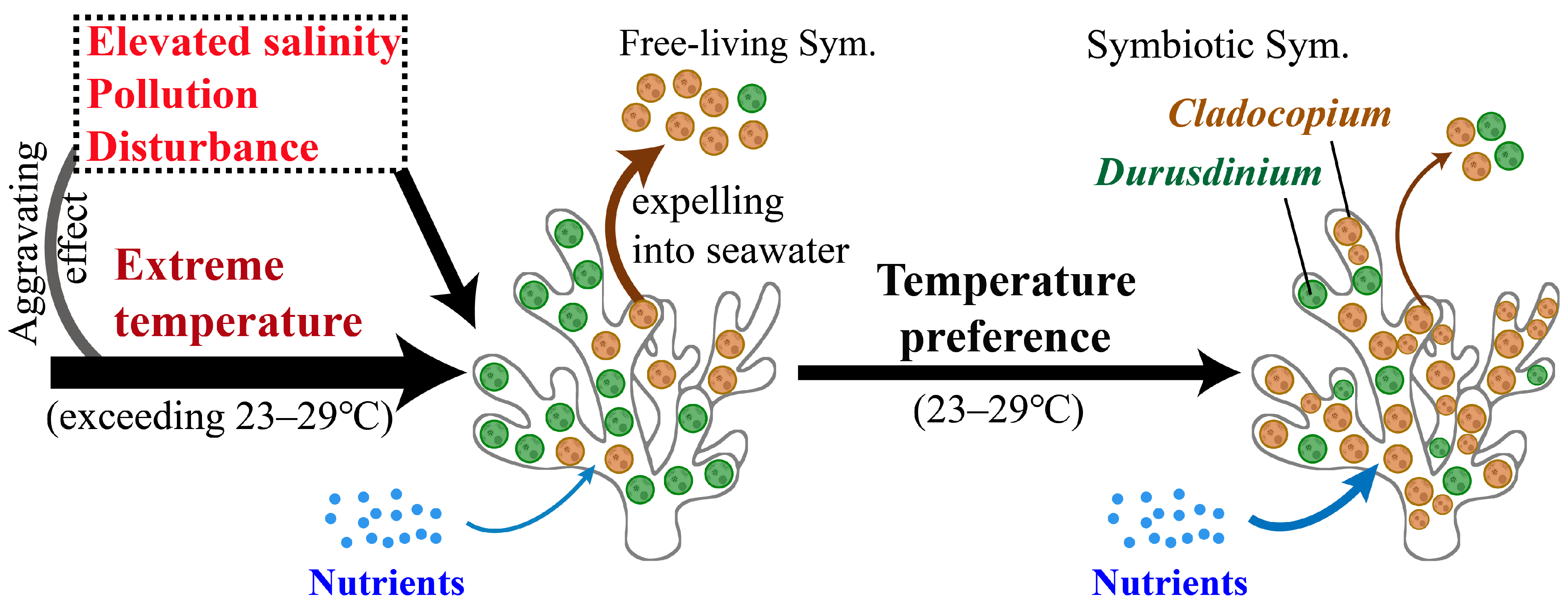
4. Discussion
4.1. Adaptability Regulation of Symbiodiniaceae Community Between Cold and Warm Months
4.2. Seasonal Changes in Symbiodiniaceae Interaction and Community Stability
4.3. Effects of Environmental Factors on Free-Living and In Hospite Symbiodiniaceae Community
4.4. Temperature-Driven Variations in In Hospite Symbiodiniaceae Community
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mayer, K.-I.; Colin, L.; Voolstra, C.R.; Cárdenas, A. Ecosystem Microbiology of Coral Reefs. In Coral Reef Microbiome; Springer: Berlin/Heidelberg, Germany, 2025; pp. 131–144. [Google Scholar]
- Hester, E.R.; Barott, K.L.; Nulton, J.; Vermeij, M.J.A.; Rohwer, F.L. Stable and sporadic symbiotic communities of coral and algal holobionts. ISME J. 2016, 10, 1157–1169. [Google Scholar] [CrossRef] [PubMed]
- Blackall, L.L.; Wilson, B.; van Oppen, M.J. Coral-the world’s most diverse symbiotic ecosystem. Mol. Ecol. 2015, 24, 5330–5347. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, C.; Lawrance, I.; Vasanth, A.J. Endosymbiotic Symbiodinium clades occurrence and influence on coral growth and resilience during stress. Symbiosis 2022, 86, 261–272. [Google Scholar] [CrossRef]
- Wall, C.B.; Kaluhiokalani, M.; Popp, B.N.; Donahue, M.J.; Gates, R.D. Divergent symbiont communities determine the physiology and nutrition of a reef coral across a light-availability gradient. ISME J. 2020, 14, 945–958. [Google Scholar] [CrossRef]
- Sun, Y.; Sheng, H.; Rädecker, N.; Lan, Y.; Tong, H.; Huang, L.; Jiang, L.; Diaz-Pulido, G.; Zou, B.; Zhang, Y.; et al. Symbiodiniaceae algal symbionts of Pocillopora damicornis larvae provide more carbon to their coral host under elevated levels of acidification and temperature. Commun. Biol. 2024, 7, 1528. [Google Scholar] [CrossRef]
- Cunning, R.; Yost, D.M.; Guarinello, M.L.; Putnam, H.M.; Gates, R.D. Variability of Symbiodinium Communities in Waters, Sediments, and Corals of Thermally Distinct Reef Pools in American Samoa. PLoS ONE 2015, 10, e0145099. [Google Scholar] [CrossRef]
- Palacio-Castro, A.M.; Smith, T.B.; Brandtneris, V.; Snyder, G.A.; van Hooidonk, R.; Maté, J.L.; Manzello, D.; Glynn, P.W.; Fong, P.; Baker, A.C. Increased dominance of heat-tolerant symbionts creates resilient coral reefs in near-term ocean warming. Proc. Natl. Acad. Sci. USA 2023, 120, e2202388120. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Alvarez-Noriega, M.; Alvarez-Romero, J.G.; Anderson, K.D.; Baird, A.H.; Babcock, R.C.; Beger, M.; Bellwood, D.R.; Berkelmans, R.; et al. Global warming and recurrent mass bleaching of corals. Nature 2017, 543, 373–377. [Google Scholar] [CrossRef]
- Baker, A.C.; Starger, C.J.; McClanahan, T.R.; Glynn, P.W.J.N. Corals’ adaptive response to climate change. Nature 2004, 430, 741. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Poloczanska, E.S.; Skirving, W.; Dove, S. Coral Reef Ecosystems under Climate Change and Ocean Acidification. Front. Mar. Sci. 2017, 4, 158. [Google Scholar] [CrossRef]
- Millán-Márquez, A.M.; Velasco-Montoya, D.A.; Terraneo, T.I.; Benzoni, F.; Bocanegra-Castaño, C.; Zapata, F.A. Symbiodiniaceae diversity in Pocillopora corals in different environments of the Colombian Eastern Pacific: Symbiont specificity in spite of coral-host flexibility. Coral Reefs 2024, 43, 1581–1597. [Google Scholar] [CrossRef]
- Quigley, K.M. Breeding and Selecting Corals Resilient to Global Warming. Annu. Rev. Anim. Biosci. 2024, 12, 209–332. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yu, K.; Liao, Z.; Chen, B.; Qin, Z.; Liang, J.; Gao, X. Adaptation strategies of relatively high-latitude marginal reef corals in response to severe temperature fluctuations. Sci. Total Environ. 2023, 903, 166439. [Google Scholar] [CrossRef] [PubMed]
- Garrido, A.G.; Machado, L.F.; Zilberberg, C.; Leite, D.C.d.A. Insights into ‘Symbiodiniaceae phycosphere’ in a coral holobiont. Symbiosis 2020, 83, 25–39. [Google Scholar] [CrossRef]
- Marhoefer, S.R.; Zenger, K.R.; Strugnell, J.M.; Logan, M.; van Oppen, M.J.H.; Kenkel, C.D.; Bay, L.K. Signatures of Adaptation and Acclimatization to Reef Flat and Slope Habitats in the Coral Pocillopora damicornis. Front. Mar. Sci. 2021, 8, 704709. [Google Scholar] [CrossRef]
- Olivares-Cordero, D.; Timmons, C.; Kenkel, C.D.; Quigley, K.M. Symbiont Community Changes Confer Fitness Benefits for Larvae in a Vertically Transmitting Coral. Ecol. Evol. 2025, 15, e70839. [Google Scholar] [CrossRef]
- Loram, J.E.; Trapido-Rosenthal, H.G.; Douglas, A.E. Functional significance of genetically different symbiotic algae Symbiodinium in a coral reef symbiosis. Mol. Ecol. 2007, 16, 4849–4857. [Google Scholar] [CrossRef]
- Ferse, S.C.A.; Fabina, N.S.; Putnam, H.M.; Franklin, E.C.; Stat, M.; Gates, R.D. Transmission Mode Predicts Specificity and Interaction Patterns in Coral-Symbiodinium Networks. PLoS ONE 2012, 7, e44970. [Google Scholar] [CrossRef]
- Bernasconi, R.; Stat, M.; Koenders, A.; Huggett, M.J. Global Networks of Symbiodinium-Bacteria Within the Coral Holobiont. Microb. Ecol. 2019, 77, 794–807. [Google Scholar] [CrossRef]
- Chen, B.; Wei, Y.; Yu, K.; Liang, Y.; Yu, X.; Liao, Z.; Qin, Z.; Xu, L.; Bao, Z.J.A.; Microbiology, E. The microbiome dynamics and interaction of endosymbiotic Symbiodiniaceae and fungi are associated with thermal bleaching susceptibility of coral holobionts. Appl. Environ. Microbiol. 2024, 90, e01939-23. [Google Scholar] [CrossRef]
- Marzonie, M.R.; Nitschke, M.R.; Bay, L.K.; Bourne, D.G.; Harrison, H.B. Symbiodiniaceae diversity varies by host and environment across thermally distinct reefs. Mol. Ecol. 2024, 33, e17342. [Google Scholar] [CrossRef]
- Quigley, K.M.; Randall, C.J.; van Oppen, M.J.H.; Bay, L.K. Assessing the role of historical temperature regime and algal symbionts on the heat tolerance of coral juveniles. Biol. Open 2020, 9, bio047316. [Google Scholar] [CrossRef] [PubMed]
- Fabina, N.S.; Putnam, H.M.; Franklin, E.C.; Stat, M.; Gates, R.D. Symbiotic specificity, association patterns, and function determine community responses to global changes: Defining critical research areas for coral-Symbiodinium symbioses. Glob. Change Biol. 2013, 19, 3306–3316. [Google Scholar] [CrossRef] [PubMed]
- Zweifler, A.; Evans, R.D.; Browne, N.K.; Thomas, L. Symbiont community dynamics in the turbid reef specialist, Turbinaria reniformis, along a latitudinal and environmental gradient in Western Australia. Coral Reefs 2024, 43, 1161–1171. [Google Scholar] [CrossRef]
- Kemp, D.W.; Hoadley, K.D.; Lewis, A.M.; Wham, D.C.; Smith, R.T.; Warner, M.E.; LaJeunesse, T.C. Thermotolerant coral–algal mutualisms maintain high rates of nutrient transfer while exposed to heat stress. Proc. R. Soc. B Biol. Sci. 2023, 290, 20231403. [Google Scholar] [CrossRef]
- Thornhill, D.J.; Kemp, D.W.; Bruns, B.U.; Fitt, W.K.; Schmidt, G.W. Correspondence Between Cold Tolerance and Temperate Biogeography In a Western Atlantic Symbiodinium (Dinophyta) Lineage. J. Phycol. 2008, 44, 1126–1135. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, H.; Zhu, W.; Zhang, J.; Liu, X.; Zhu, M.; Wang, A.; Li, X. The impact of environmental conditions on the heat tolerance of Acropora hyacinthus. J. Exp. Mar. Biol. Ecol. 2025, 585, 152096. [Google Scholar] [CrossRef]
- Chen, R.-W.; Li, Z.; Huang, J.; Liu, X.; Zhu, W.; Li, Y.; Wang, A.; Li, X. The community stability of Symbiodiniaceae and bacteria of different morphological corals and linkages to coral susceptibility to anthropogenic disturbance. Coral Reefs 2024, 43, 467–481. [Google Scholar] [CrossRef]
- Swain, T.D.; Lax, S.; Gilbert, J.; Backman, V.; Marcelino, L.A. A Phylogeny-Informed Analysis of the Global Coral-Symbiodiniaceae Interaction Network Reveals that Traits Correlated with Thermal Bleaching Are Specific to Symbiont Transmission Mode. mSystems 2021, 6, 11. [Google Scholar] [CrossRef]
- Abbott, E.; Dixon, G.; Matz, M. Shuffling between Cladocopium and Durusdinium extensively modifies the physiology of each symbiont without stressing the coral host. Mol. Ecol. 2021, 30, 6585–6595. [Google Scholar] [CrossRef]
- Cunning, R.; Silverstein, R.N.; Baker, A.C. Investigating the causes and consequences of symbiont shuffling in a multi-partner reef coral symbiosis under environmental change. Proc. R. Soc. B Biol. Sci. 2015, 282, 20141725. [Google Scholar] [CrossRef]
- Buzzoni, D.; Cunning, R.; Baker, A.C. The role of background algal symbionts as drivers of shuffling to thermotolerant Symbiodiniaceae following bleaching in three Caribbean coral species. Coral Reefs 2023, 42, 1285–1295. [Google Scholar] [CrossRef]
- Logan, C.A.; Dunne, J.P.; Ryan, J.S.; Baskett, M.L.; Donner, S.D. Quantifying global potential for coral evolutionary response to climate change. Nat. Clim. Change 2021, 11, 537–542. [Google Scholar] [CrossRef]
- Chan, W.Y.; Meyers, L.; Rudd, D.; Topa, S.H.; van Oppen, M.J.H. Heat-evolved algal symbionts enhance bleaching tolerance of adult corals without trade-off against growth. Glob. Change Biol. 2023, 29, 6945–6968. [Google Scholar] [CrossRef]
- Qin, Z.; Yu, K.; Chen, B.; Wang, Y.; Liang, J.; Luo, W.; Xu, L.; Huang, X. Diversity of Symbiodiniaceae in 15 Coral Species from the Southern South China Sea: Potential Relationship with Coral Thermal Adaptability. Front. Microbiol. 2019, 10, 2343. [Google Scholar] [CrossRef]
- Chakravarti, L.J.; Beltran, V.H.; van Oppen, M.J.H. Rapid thermal adaptation in photosymbionts of reef-building corals. Glob. Change Biol. 2017, 23, 4675–4688. [Google Scholar] [CrossRef] [PubMed]
- LaJeunesse, T.C.; Parkinson, J.E.; Gabrielson, P.W.; Jeong, H.J.; Reimer, J.D.; Voolstra, C.R.; Santos, S.R. Systematic Revision of Symbiodiniaceae Highlights the Antiquity and Diversity of Coral Endosymbionts. Curr. Biol. 2018, 28, 2570–2580.E6. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, R.N.; Cunning, R.; Baker, A.C. Tenacious D: Symbiodinium in clade D remain in reef corals at both high and low temperature extremes despite impairment. J. Exp. Biol. 2017, 220, 1192–1196. [Google Scholar] [CrossRef] [PubMed]
- Turnham, K.E.; Aschaffenburg, M.D.; Pettay, D.T.; Paz-García, D.A.; Reyes-Bonilla, H.; Pinzón, J.; Timmins, E.; Smith, R.T.; McGinley, M.P.; Warner, M.E.; et al. High physiological function for corals with thermally tolerant, host-adapted symbionts. Proc. R. Soc. B Biol. Sci. 2023, 290, 20231021. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, X.; Li, Y.; Sun, D.; Huang, W.; Shi, T. Symbiont shuffling dynamics associated with photodamage during temperature stress in coral symbiosis. Ecol. Indic. 2022, 145, 109706. [Google Scholar] [CrossRef]
- Foreman, A.D.; Duprey, N.N.; Yuval, M.; Dumestre, M.; Leichliter, J.N.; Rohr, M.C.; Dodwell, R.C.A.; Dodwell, G.A.S.; Clua, E.E.G.; Treibitz, T.; et al. Severe cold-water bleaching of a deep-water reef underscores future challenges for Mesophotic Coral Ecosystems. Sci. Total Environ. 2024, 951, 175210. [Google Scholar] [CrossRef] [PubMed]
- Goreau, T.J.; Hayes, R.L. 2023 Record marine heat waves: Coral reef bleaching HotSpot maps reveal global sea surface temperature extremes, coral mortality, and ocean circulation changes. Oxf. Open Clim. Change 2024, 4, kgae005, Erratum in Oxf. Open Clim. Change 2025, 5, kgaf006. [Google Scholar] [CrossRef]
- Leiva, C.; Torda, G.; Zhou, C.; Pan, Y.; Harris, J.; Xiang, X.; Tan, S.; Tian, W.; Hume, B.; Miller, D.J.; et al. Rapid Evolution in Action: Environmental Filtering Supports Coral Adaptation to a Hot, Acidic, and Deoxygenated Extreme Habitat. Glob. Change Biol. 2025, 31, e70103. [Google Scholar] [CrossRef]
- Quigley, K.M.; Ramsby, B.; Laffy, P.; Harris, J.; Mocellin, V.J.L.; Bay, L.K. Symbioses are restructured by repeated mass coral bleaching. Sci. Adv. 2022, 8, eabq8349. [Google Scholar] [CrossRef]
- Howe-Kerr, L.I.; Bachelot, B.; Wright, R.M.; Kenkel, C.D.; Bay, L.K.; Correa, A.M.S. Symbiont community diversity is more variable in corals that respond poorly to stress. Glob. Change Biol. 2020, 26, 2220–2234. [Google Scholar] [CrossRef]
- Cunning, R.; Lenz, E.A.; Edmunds, P.J. Measuring multi-year changes in the Symbiodiniaceae algae in Caribbean corals on coral-depleted reefs. PeerJ 2024, 12, e17358. [Google Scholar] [CrossRef]
- Ziegler, M.; Eguiluz, V.M.; Duarte, C.M.; Voolstra, C.R. Rare symbionts may contribute to the resilience of coral-algal assemblages. ISME J. 2018, 12, 161–172. [Google Scholar] [CrossRef]
- Li, J.; Shao, Z.; Cheng, K.; Yang, Q.; Ju, H.; Tang, X.; Zhang, S.; Li, J. Coral-associated Symbiodiniaceae exhibit host specificity but lack phylosymbiosis, with Cladocopium and Durusdinium showing different cophylogenetic patterns. New Phytol. 2025, 247, 309–324. [Google Scholar] [CrossRef]
- Quigley, K.M.; Willis, B.L.; Kenkel, C.D. Transgenerational inheritance of shuffled symbiont communities in the coral Montipora digitata. Sci. Rep. 2019, 9, 13328. [Google Scholar] [CrossRef]
- Huang, J.; Wang, F.; Zhao, H.; Xu, H.; Liu, S.; Xu, Q.; Wang, A.; Li, X. Reef benthic composition and coral communities at the Wuzhizhou Island in the south China sea: The impacts of anthropogenic disturbance. Estuar. Coast. Shelf Sci. 2020, 243, 106863. [Google Scholar] [CrossRef]
- Yu, K. Coral reefs in the South China Sea: Their response to and records on past environmental changes. Sci. China Earth Sci. 2012, 55, 1217–1229. [Google Scholar] [CrossRef]
- Zuo, X.; Su, F.; Wu, W.; Chen, Z.; Shi, W. Spatial and temporal variability of thermal stress to China’s coral reefs in South China Sea. Chin. Geogr. Sci. 2015, 25, 159–173. [Google Scholar] [CrossRef]
- Zhao, M.; Zhang, H.; Zhong, Y.; Jiang, D.; Liu, G.; Yan, H.; Zhang, H.; Guo, P.; Li, C.; Yang, H.; et al. The Status of Coral Reefs and Its Importance for Coastal Protection: A Case Study of Northeastern Hainan Island, South China Sea. Sustainability 2019, 11, 4354. [Google Scholar] [CrossRef]
- Han, M.; Zhang, R.; Yu, K.; Li, A.; Wang, Y.; Huang, X. Polycyclic aromatic hydrocarbons (PAHs) in corals of the South China Sea: Occurrence, distribution, bioaccumulation, and considerable role of coral mucus. J. Hazard. Mater. 2020, 384, 121299. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Yan, Z.; Tang, K.; Xing, Q.; Lin, J.; Su, H.; Wu, Z.; Wu, G.; Yang, C.; Tang, J.; et al. Threats of BaA-SM2 as Key Bioaccumulated PAH and Antibiotic Components to Coral Energy Dynamics and Symbiosis Stability. Water Res. 2025, 287, 124297. [Google Scholar] [CrossRef]
- Cao, X.; Wang, L.; Lin, J.; Wu, G.; Tang, K.; Tang, J.; Yan, Z.; An, M.; Liu, Z.; Zhou, Z. Differential bioaccumulation and tolerances of massive and branching scleractinian corals to polycyclic aromatic hydrocarbons in situ. Sci. Total Environ. 2024, 931, 172920. [Google Scholar] [CrossRef]
- Yin, K.; Zhang, X.; Pei, Y.; Fu, Y.; Zhang, X.; Li, Y.; Li, X.; Wang, X.; Zhou, H. Unraveling the differential tolerance mechanisms of Acropora formosa and Montipora digitata to Benzo[a]pyrene (BaP) exposure via 4D proteomics. J. Hazard. Mater. 2025, 492, 138213. [Google Scholar] [CrossRef]
- GB/T 12763.4-2007; Specification of Oceanographic Survey-Part 4: Survey of Chemical Parameters in Sea Water. Standards Press of China: Beijing, China, 2007. (In Chinese)
- Yang, T.; Cheng, H.; Wang, H.; Drews, M.; Li, S.; Huang, W.; Zhou, H.; Chen, C.M.; Diao, X. Comparative study of polycyclic aromatic hydrocarbons (PAHs) and heavy metals (HMs) in corals, surrounding sediments and surface water at the Dazhou Island, China. Chemosphere 2019, 218, 157–168. [Google Scholar] [CrossRef]
- Yuan, J.; Li, M.; Lin, S. An Improved DNA Extraction Method for Efficient and Quantitative Recovery of Phytoplankton Diversity in Natural Assemblages. PLoS ONE 2015, 10, e0133060. [Google Scholar] [CrossRef]
- Stat, M.; Pochon, X.; Cowie, R.O.M.; Gates, R.D. Specificity in communities of Symbiodinium in corals from Johnston Atoll. Mar. Ecol. Prog. Ser. 2009, 386, 83–96. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Arif, C.; Daniels, C.; Bayer, T.; Banguera-Hinestroza, E.; Barbrook, A.; Howe, C.J.; LaJeunesse, T.C.; Voolstra, C.R. Assessing Symbiodinium diversity in scleractinian corals via next-generation sequencing-based genotyping of the ITS2 rDNA region. Mol. Ecol. 2014, 23, 4418–4433. [Google Scholar] [CrossRef]
- Glassman, S.I.; Martiny, J.B.H. Broadscale Ecological Patterns Are Robust to Use of Exact Sequence Variants versus Operational Taxonomic Units. mSphere 2018, 3, e00148-18. [Google Scholar] [CrossRef]
- Hunter, R.L.; LaJeunesse, T.C.; Santos, S.R. Structure and Evolution of the Rdna Internal Transcribed Spacer (Its) Region 2 In the Symbiotic Dinoflagellates (Symbiodinium, Dinophyta). J. Phycol. 2007, 43, 120–128. [Google Scholar] [CrossRef]
- Pochon, X.; Garcia-Cuetos, L.; Baker, A.C.; Castella, E.; Pawlowski, J. One-year survey of a single Micronesian reef reveals extraordinarily rich diversity of Symbiodinium types in soritid foraminifera. Coral Reefs 2007, 26, 867–882. [Google Scholar] [CrossRef]
- Quigley, K.M.; Davies, S.W.; Kenkel, C.D.; Willis, B.L.; Matz, M.V.; Bay, L.K. Deep-sequencing method for quantifying background abundances of symbiodinium types: Exploring the rare symbiodinium biosphere in reef-building corals. PLoS ONE 2014, 9, e94297. [Google Scholar] [CrossRef]
- Cunning, R.; Gates, R.D.; Edmunds, P.J. Using high-throughput sequencing of ITS2 to describe Symbiodinium metacommunities in St. John, US Virgin Islands. PeerJ 2017, 5, e3472. [Google Scholar] [CrossRef]
- Franklin, E.C.; Stat, M.; Pochon, X.; Putnam, H.M.; Gates, R.D. GeoSymbio: A hybrid, cloud-based web application of global geospatial bioinformatics and ecoinformatics for Symbiodinium-host symbioses. Mol. Ecol. Resour. 2012, 12, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, Y.; Zhang, F. Abundant and rare bacteria possess different diversity and function in crop monoculture and rotation systems across regional farmland. Soil Biol. Biochem. 2022, 171, 108742. [Google Scholar] [CrossRef]
- Mo, Y.; Zhang, W.; Yang, J.; Lin, Y.; Yu, Z.; Lin, S. Biogeographic patterns of abundant and rare bacterioplankton in three subtropical bays resulting from selective and neutral processes. ISME J. 2018, 12, 2198–2210. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.M.; Guo, X.; Wu, L.; Zhang, Y.; Xiao, N.; Ning, D.; Shi, Z.; Zhou, X.; Wu, L.; Yang, Y.; et al. Climate warming enhances microbial network complexity and stability. Nat. Clim. Change 2021, 11, 343–348. [Google Scholar] [CrossRef]
- Zhou, G.; Cai, L.; Li, Y.; Tong, H.; Jiang, L.; Zhang, Y.; Lei, X.; Guo, M.; Liu, S.; Qian, P.Y.; et al. Temperature-Driven Local Acclimatization of Symbiodnium Hosted by the Coral Galaxea fascicularis at Hainan Island, China. Front. Microbiol. 2017, 8, 2487. [Google Scholar] [CrossRef]
- Chen, C.A.; Lam, K.K.; Nakano, Y.; Tsai, W.S. A stable association of the stress-tolerant zooxanthellae, clade D, with the low-temperature-tolerant coral, (Scleractinia: Faviidae) in subtropical non-reefal coral communities. Zool. Stud. 2003, 42, 540–550. [Google Scholar]
- Naugle, M.S.; Oliver, T.A.; Barshis, D.J.; Gates, R.D.; Logan, C.A. Variation in Coral Thermotolerance Across a Pollution Gradient Erodes as Coral Symbionts Shift to More Heat-Tolerant Genera. Front. Mar. Sci. 2021, 8, 760891. [Google Scholar] [CrossRef]
- Li, Z.; Li, Y.; Zhu, W.; Liu, X.; Chen, R.-W.; Wang, A.; Li, X. The Symbiodiniaceae and Bacterial Dynamic Composition of the Coral Echinopora gemmacea on Wuzhizhou Island. J. Mar. Sci. Eng. 2023, 11, 2262. [Google Scholar] [CrossRef]
- LaJeunesse, T.C.; Wham, D.C.; Pettay, D.T.; Parkinson, J.E.; Keshavmurthy, S.; Chen, C.A. Ecologically differentiated stress-tolerant endosymbionts in the dinoflagellate genusSymbiodinium (Dinophyceae) Clade D are different species. Phycologia 2014, 53, 305–319. [Google Scholar] [CrossRef]
- Kao, K.W.; Keshavmurthy, S.; Tsao, C.H.; Wang, J.T.; Chen, C.A. Repeated and Prolonged Temperature Anomalies Negate Symbiodiniaceae Genera Shuffling in the Coral Platygyra verweyi (Scleractinia; Merulinidae). Zool. Stud. 2018, 57, e55. [Google Scholar] [CrossRef]
- Turnham, K.E.; Lewis, A.M.; Kemp, D.W.; Warner, M.E.; Wham, D.F.; Smith, R.T.; LaJeunesse, T.C. Limited persistence of the heat-tolerant zooxanthella Durusdinium trenchii, in corals transplanted to a barrier reef where it is rare among natal colonies. Coral Reefs 2025, 44, 555–570. [Google Scholar] [CrossRef]
- Bassim, K.; Sammarco, P. Effects of temperature and ammonium on larval development and survivorship in a scleractinian coral (Diploria strigosa). Mar. Biol. 2003, 142, 241–252. [Google Scholar] [CrossRef]
- Chen, Y.; Ma, J.; Huang, H.; Chen, P.; Chao, F.; Luo, W.J.A.B. Effects of Temperature, pH, Ammonia Nitrogen Content and Salinity on Coral (Acropora millepora & Palythoa sp.) Bleaching. Agric. Biotechnol. 2017, 6, 52–59. [Google Scholar]
- NOAA. In What Types of Water Do Corals Live? Available online: https://oceanservice.noaa.gov/facts/coralwaters.html (accessed on 8 December 2022).
- Russnak, V.; Rodriguez-Lanetty, M.; Karsten, U. Photophysiological Tolerance and Thermal Plasticity of Genetically Different Symbiodiniaceae Endosymbiont Species of Cnidaria. Front. Mar. Sci. 2021, 8, 657348. [Google Scholar] [CrossRef]
- Li, J.; Shao, Z.; Cheng, K.; Yang, Q.; Ju, H.; Tang, X.; Cai, W.; Yu, K.; Li, J. Higher genotypic diversity and distinct assembly mechanism of free-living Symbiodiniaceae assemblages than sympatric coral endosymbiotic assemblages in a tropical coral reef. New Phytol. 2024, 12, e00514-24. [Google Scholar] [CrossRef]
- Huang, H.; Zhou, G.; Yang, J.; Liu, S.; You, F.; Lei, X. Diversity of free-living and symbioticSymbiodiniumin the coral reefs of Sanya, South China Sea. Mar. Biol. Res. 2013, 9, 117–128. [Google Scholar] [CrossRef]
- Pochon, X.; Stat, M.; Takabayashi, M.; Chasqui, L.; Chauka, L.J.; Logan, D.D.K.; Gates, R.D. Comparison of Endosymbiotic and Free-Living Symbiodinium (Dinophyceae) Diversity in a Hawaiian Reef Environment1. J. Phycol. 2010, 46, 53–65. [Google Scholar] [CrossRef]
- Cunning, R.; Silverstein, R.N.; Baker, A.C. Symbiont shuffling linked to differential photochemical dynamics of Symbiodinium in three Caribbean reef corals. Coral Reefs 2017, 37, 145–152. [Google Scholar] [CrossRef]
- Yang, T. Distribution of Polycyclic Aromatic Hydrocarbons in Coral Reefs and Response of Symbiotic Symbiodiniaceae to Benzo[a]pyrene Stress. Ph.D. Thesis, Hainan University, Hainan, China, 2020. (In Chinese). [Google Scholar]
- Ros, M.; Suggett, D.J.; Edmondson, J.; Haydon, T.; Hughes, D.J.; Kim, M.; Guagliardo, P.; Bougoure, J.; Pernice, M.; Raina, J.-B.; et al. Symbiont shuffling across environmental gradients aligns with changes in carbon uptake and translocation in the reef-building coral Pocillopora acuta. Coral Reefs 2021, 40, 595–607. [Google Scholar] [CrossRef]
- Silverstein, R.N.; Cunning, R.; Baker, A.C. Change in algal symbiont communities after bleaching, not prior heat exposure, increases heat tolerance of reef corals. Glob. Change Biol. 2015, 21, 236–249. [Google Scholar] [CrossRef]
- Allen-Waller, L.; Barott, K.L. Symbiotic dinoflagellates divert energy away from mutualism during coral bleaching recovery. Symbiosis 2023, 89, 173–186. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, X.; Kvitt, H.; Sheng, H.; Sun, D.; Niu, G.; Tchernov, D.; Shi, T. Lineage-specific symbionts mediate differential coral responses to thermal stress. Microbiome 2023, 11, 211. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.M.; Andras, J.P.; Jordan-Garza, A.G.; Fogel, M.L. Nitrate competition in a coral symbiosis varies with temperature among Symbiodinium clades. ISME J. 2013, 7, 1248–1251. [Google Scholar] [CrossRef] [PubMed]
- Cunning, R.; Gillette, P.; Capo, T.; Galvez, K.; Baker, A.C. Growth tradeoffs associated with thermotolerant symbionts in the coral Pocillopora damicornis are lost in warmer oceans. Coral Reefs 2014, 34, 155–160. [Google Scholar] [CrossRef]
- Yuyama, I.; Nakamura, T.; Higuchi, T.; Hidaka, M. Different Stress Tolerances of Juveniles of the Coral Acropora tenuis Associated with Clades C1 and D Symbiodinium. Zool. Stud. 2016, 55, e19. [Google Scholar] [CrossRef]
- Stat, M.; Gates, R.D. Clade D Symbiodinium in Scleractinian Corals: A “Nugget” of Hope, a Selfish Opportunist, an Ominous Sign, or All of the Above? J. Mar. Biol. 2011, 2011, 730715. [Google Scholar] [CrossRef]
- Matsuda, S.B.; Opalek, M.L.; Ritson-Williams, R.; Gates, R.D.; Cunning, R. Symbiont-mediated tradeoffs between growth and heat tolerance are modulated by light and temperature in the coral Montipora capitata. Coral Reefs 2023, 42, 1385–1394. [Google Scholar] [CrossRef]
- Wang, Q.; Zheng, X.; Zhou, X.; Zhang, H.; Cai, L.; Leung, J.Y.S.; Huang, L. Symbiont genus determines the trophic strategy of corals: Implications for intraspecific competition for energy sources in coral reefs. Ecol. Indic. 2024, 158, 111477. [Google Scholar] [CrossRef]
- Butler, C.C.; Turnham, K.E.; Lewis, A.M.; Nitschke, M.R.; Warner, M.E.; Kemp, D.W.; Hoegh-Guldberg, O.; Fitt, W.K.; van Oppen, M.J.H.; LaJeunesse, T.C. Formal recognition of host-generalist species of dinoflagellate (Cladocopium, Symbiodiniaceae) mutualistic with Indo-Pacific reef corals. J. Phycol. 2023, 59, 698–711. [Google Scholar] [CrossRef]
- Swain, T.D.; Chandler, J.; Backman, V.; Marcelino, L.; Blanckenhorn, W. Consensus thermotolerance ranking for 110 Symbiodinium phylotypes: An exemplar utilization of a novel iterative partial-rank aggregation tool with broad application potential. Funct. Ecol. 2016, 31, 172–183. [Google Scholar] [CrossRef]
- Zhu, W.; Liu, X.; Zhu, M.; Li, X.; Yin, H.; Huang, J.; Wang, A.; Li, X. Responses of Symbiodiniaceae Shuffling and Microbial Community Assembly in Thermally Stressed Acropora hyacinthus. Front. Microbiol. 2022, 13, 832081. [Google Scholar] [CrossRef]
- Pinsky, M.L.; Hillebrand, H.; Chase, J.M.; Antão, L.H.; Hirt, M.R.; Brose, U.; Michael, T.B.; Benoit, G.; Benjamin, R.; Blowes, S.A. Warming and cooling catalyse widespread temporal turnover in biodiversity. Nature 2025, 638, 995–999. [Google Scholar] [CrossRef]
- Stat, M.; Loh, W.K.W.; LaJeunesse, T.C.; Hoegh-Guldberg, O.; Carter, D.A. Stability of coral–endosymbiont associations during and after a thermal stress event in the southern Great Barrier Reef. Coral Reefs 2009, 28, 709–713. [Google Scholar] [CrossRef]
- Qi, Z. Interspecific and Spatial Changes of Coral Microbiome in Coastal Hainan Characteristics and Responses to Environmental Stress. Ph.D. Thesis, Hainan University, Haikou, China, 2023. [Google Scholar]
- Boulotte, N.M.; Dalton, S.J.; Carroll, A.G.; Harrison, P.L.; Putnam, H.M.; Peplow, L.M.; van Oppen, M.J. Exploring the Symbiodinium rare biosphere provides evidence for symbiont switching in reef-building corals. ISME J. 2016, 10, 2693–2701. [Google Scholar] [CrossRef]
- Fujise, L.; Suggett, D.J.; Stat, M.; Kahlke, T.; Bunce, M.; Gardner, S.G.; Goyen, S.; Woodcock, S.; Ralph, P.J.; Seymour, J.R.; et al. Unlocking the phylogenetic diversity, primary habitats, and abundances of free-living Symbiodiniaceae on a coral reef. Mol. Ecol. 2020, 30, 343–360. [Google Scholar] [CrossRef] [PubMed]
- Hume, B.C.C.; Voolstra, C.R.; Arif, C.; D’Angelo, C.; Burt, J.A.; Eyal, G.; Loya, Y.; Wiedenmann, J. Ancestral genetic diversity associated with the rapid spread of stress-tolerant coral symbionts in response to Holocene climate change. Proc. Natl. Acad. Sci. USA 2016, 113, 4416–4421. [Google Scholar] [CrossRef] [PubMed]
- Shade, A.; Jones, S.E.; Caporaso, J.G.; Handelsman, J.; Knight, R.; Fierer, N.; Gilbert, J.A.; Dubilier, N. Conditionally Rare Taxa Disproportionately Contribute to Temporal Changes in Microbial Diversity. mBio 2014, 5, mbio.01371-14. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Lin, Z.J.; Cai, Z.H.; Zeng, Y.H.; Zhu, J.M.; Du, X.P. Opportunistic bacteria use quorum sensing to disturb coral symbiotic communities and mediate the occurrence of coral bleaching. Environ. Microbiol. 2020, 22, 1944–1962. [Google Scholar] [CrossRef]
- Williams, S.D.; Patterson, M.R. Resistance and robustness of the global coral–symbiont network. Ecology 2020, 101, e02990. [Google Scholar] [CrossRef]
- Faust, K.; Raes, J. Microbial interactions: From networks to models. Nat. Rev. Microbiol. 2012, 10, 538–550. [Google Scholar] [CrossRef]
- Montoya, J.M.; Pimm, S.L.; Solé, R.V. Ecological networks and their fragility. Nature 2006, 442, 259–264. [Google Scholar] [CrossRef]
- Shinzato, C.; Takeuchi, T.; Yoshioka, Y.; Tada, I.; Kanda, M.; Broussard, C.; Iguchi, A.; Kusakabe, M.; Marin, F.; Satoh, N.; et al. Whole-Genome Sequencing Highlights Conservative Genomic Strategies of a Stress-Tolerant, Long-Lived Scleractinian Coral, Porites australiensis Vaughan, 1918. Genome Biol. Evol. 2021, 13, evab270. [Google Scholar] [CrossRef]
- Alderdice, R.; Hume, B.C.C.; Kühl, M.; Pernice, M.; Suggett, D.J.; Voolstra, C.R. Disparate Inventories of Hypoxia Gene Sets Across Corals Align with Inferred Environmental Resilience. Front. Mar. Sci. 2022, 9, 834332. [Google Scholar] [CrossRef]
- Manullang, C.; Millyaningrum, I.H.; Iguchi, A.; Miyagi, A.; Tanaka, Y.; Nojiri, Y.; Sakai, K. Responses of branching reef corals Acropora digitifera and Montipora digitata to elevated temperature and pCO2. PeerJ 2020, 8, e10562. [Google Scholar] [CrossRef] [PubMed]
- Marshall, P.A.; Baird, A.H. Bleaching of corals on the Great Barrier Reef: Differential susceptibilities among taxa. Coral Reefs 2000, 19, 155–163. [Google Scholar] [CrossRef]
- Edwards, A.J.; Clark, S.; Zahir, H.; Rajasuriya, A.; Naseer, A.; Rubens, J. Coral bleaching and mortality on artificial and natural reefs in Maldives in 1998, sea surface temperature anomalies and initial recovery. Mar. Pollut. Bull. 2001, 42, 7–15. [Google Scholar] [CrossRef]
- Li, S.; Yu, K.; Shi, Q.; Chen, T.; Zhao, M.; Yan, H. Experimental study of stony coral response to the high temperature in Luhuitou of Hainan Island. Trop. Geogr. 2008, 28, 534–539. [Google Scholar]
- Li, S.; Yu, K.; Shi, Q.; Chen, T.; Zhao, M. Low water temperature tolerance and responding mode of scleractinian corals in Sanya Bay. Chin. J. Appl. Ecol. 2009, 20, 2289–2295. [Google Scholar]
- Liang, J.; Yu, K.; Wang, Y.; Huang, X.; Huang, W.; Qin, Z.; Pan, Z.; Yao, Q.; Wang, W.; Wu, Z. Distinct Bacterial Communities Associated with Massive and Branching Scleractinian Corals and Potential Linkages to Coral Susceptibility to Thermal or Cold Stress. Front. Microbiol. 2017, 8, 979. [Google Scholar] [CrossRef]
- Wang, J.; Chen, J.; Wang, S.; Li, F.; Fu, C.; Wang, Y. Monoclonal Culture and Characterization of Symbiodiniaceae C1 Strain from the Scleractinian Coral Galaxea fascicularis. Front. Physiol. 2021, 11, 621111. [Google Scholar] [CrossRef]
- Guan, Y.; Hohn, S.; Merico, A. Suitable environmental ranges for potential coral reef habitats in the tropical ocean. PLoS ONE 2015, 10, e0128831. [Google Scholar] [CrossRef]
- Hall, E.R.; Muller, E.M.; Goulet, T.; Bellworthy, J.; Ritchie, K.B.; Fine, M. Eutrophication may compromise the resilience of the Red Sea coral Stylophora pistillata to global change. Mar. Pollut. Bull. 2018, 131, 701–711. [Google Scholar] [CrossRef]
- Yang, T.; Diao, X.; Cheng, H.; Wang, H.; Zhou, H.; Zhao, H.; Chen, C.M. Comparative study of polycyclic aromatic hydrocarbons (PAHs) and heavy metals (HMs) in corals, sediments and seawater from coral reefs of Hainan, China. Environ. Pollut. 2020, 264, 114719. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Qi, Z.; Yin, L.; Chang, P.; Ju, H.; Diao, X. Physiological responses of different corals under single and combined stress of acidification and benzo[a]pyrene. Asian J. Ecotoxicol. 2023, 18, 456–464. (In Chinese) [Google Scholar] [CrossRef]
- Claar, D.C.; Starko, S.; Tietjen, K.L.; Epstein, H.E.; Cunning, R.; Cobb, K.M.; Baker, A.C.; Gates, R.D.; Baum, J.K. Dynamic symbioses reveal pathways to coral survival through prolonged heatwaves. Nat. Commun. 2020, 11, 6097. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Xu, D.; Tian, W.; Lu, Z.; Zhou, Y.; Chu, M.; Zhao, J.; Liu, B.; Cao, H.; Zhang, R.; et al. The effect of bioturbation on the release behavior of polycyclic aromatic hydrocarbons from sediments: A sediment-seawater microcosm experiment combined with a fugacity model. Environ. Pollut. 2024, 349, 123933. [Google Scholar] [CrossRef]
- Lo, H.S.; Chaumet, B.; Azaroff, A.; Sobek, A.; Jonsson, S.; Gorokhova, E. Disentangling the Impacts of PAHs, Microplastics, and Sediment Resuspension on Algal Physiology: A Partial Least Squares Structural Equation Modeling Approach. ACS Environ. Au 2025. [Google Scholar] [CrossRef]
- Berrios-Rolon, P.J.; Cotto, M.C.; Marquez, F. Polycyclic Aromatic Hydrocarbons (PAHs) in Freshwater Systems: A Comprehensive Review of Sources, Distribution, and Ecotoxicological Impacts. Toxics 2025, 13, 321. [Google Scholar] [CrossRef]
- Xiang, N.; Jiang, C.; Huang, W.; Nordhaus, I.; Zhou, H.; Drews, M.; Diao, X. The impact of acute benzo(a)pyrene on antioxidant enzyme and stress-related genes in tropical stony corals (Acropora spp.). Sci. Total Environ. 2019, 694, 133474. [Google Scholar] [CrossRef]
- de Souza, M.R.; Caruso, C.; Ruiz-Jones, L.; Drury, C.; Gates, R.; Toonen, R.J. Community composition of coral-associated Symbiodiniaceae differs across fine-scale environmental gradients in Kāne‘ohe Bay. R. Soc. Open Sci. 2022, 9, 212042. [Google Scholar] [CrossRef]
- Pratomo, A.; Bengen, D.G.; Zamani, N.P.; Lane, C.; Humphries, A.T.; Borbee, E.; Subhan, B.; Madduppa, H. Diversity and distribution of Symbiodiniaceae detected on coral reefs of Lombok, Indonesia using environmental DNA metabarcoding. PeerJ 2022, 10, e14006. [Google Scholar] [CrossRef]
- Saxby, T.; Dennison, W.C.; Hoegh-Guldberg, O. Photosynthetic responses of the coral Montipora digitata to cold temperature stress. Mar. Ecol. Prog. Ser. 2003, 248, 85–97. [Google Scholar] [CrossRef]
- Berkelmans, R.; van Oppen, M.J.H. The role of zooxanthellae in the thermal tolerance of corals: A ‘nugget of hope’ for coral reefs in an era of climate change. Proc. R. Soc. B Biol. Sci. 2006, 273, 2305–2312. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-Y.; Carballo-Bolaños, R.; Kuo, C.-Y.; Keshavmurthy, S.; Chen, C.A. Leptoria phrygia in Southern Taiwan shuffles and switches symbionts to resist thermal-induced bleaching. Sci. Rep. 2020, 10, 7808. [Google Scholar] [CrossRef] [PubMed]
- Cunning, R.; Baker, A.C. Thermotolerant coral symbionts modulate heat stress-responsive genes in their hosts. Mol. Ecol. 2020, 29, 2940–2950. [Google Scholar] [CrossRef]
- Brener-Raffalli, K.; Clerissi, C.; Vidal-Dupiol, J.; Adjeroud, M.; Bonhomme, F.; Pratlong, M.; Aurelle, D.; Mitta, G.; Toulza, E. Thermal regime and host clade, rather than geography, drive Symbiodinium and bacterial assemblages in the scleractinian coral Pocillopora damicornis sensu lato. Microbiome 2018, 6, 39. [Google Scholar] [CrossRef]
- Zhao, H.; Yuan, M.; Strokal, M.; Wu, H.C.; Liu, X.; Murk, A.; Kroeze, C.; Osinga, R. Impacts of nitrogen pollution on corals in the context of global climate change and potential strategies to conserve coral reefs. Sci. Total Environ. 2021, 774, 145017. [Google Scholar] [CrossRef]
- Donovan, M.K.; Adam, T.C.; Shantz, A.A.; Speare, K.E.; Munsterman, K.S.; Rice, M.M.; Schmitt, R.J.; Holbrook, S.J.; Burkepile, D.E. Nitrogen pollution interacts with heat stress to increase coral bleaching across the seascape. Proc. Natl. Acad. Sci. USA 2020, 117, 5351–5357. [Google Scholar] [CrossRef]
- Rotchell, J.M.; Ostrander, G.K. Molecular Toxicology of Corals: A Review. J. Toxicol. Environ. Health Part B 2011, 14, 571–592. [Google Scholar] [CrossRef]
- Overmans, S.; Nordborg, M.; Diaz-Rua, R.; Brinkman, D.L.; Negri, A.P.; Agusti, S. Phototoxic effects of PAH and UVA exposure on molecular responses and developmental success in coral larvae. Aquat. Toxicol. 2018, 198, 165–174. [Google Scholar] [CrossRef]
- Xiang, N.; Jiang, C.; Yang, T.; Li, P.; Wang, H.; Xie, Y.; Li, S.; Zhou, H.; Diao, X. Occurrence and distribution of Polycyclic aromatic hydrocarbons (PAHs) in seawater, sediments and corals from Hainan Island, China. Ecotoxicol. Environ. Saf. 2018, 152, 8–15. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, T.; Qi, Z.; Wang, H.; Zheng, P.; Kao, S.-J.; Diao, X. Seasonal Variation in In Hospite but Not Free-Living, Symbiodiniaceae Communities Around Hainan Island, China. Microorganisms 2025, 13, 1958. https://doi.org/10.3390/microorganisms13081958
Yang T, Qi Z, Wang H, Zheng P, Kao S-J, Diao X. Seasonal Variation in In Hospite but Not Free-Living, Symbiodiniaceae Communities Around Hainan Island, China. Microorganisms. 2025; 13(8):1958. https://doi.org/10.3390/microorganisms13081958
Chicago/Turabian StyleYang, Tinghan, Zhao Qi, Haihua Wang, Pengfei Zheng, Shuh-Ji Kao, and Xiaoping Diao. 2025. "Seasonal Variation in In Hospite but Not Free-Living, Symbiodiniaceae Communities Around Hainan Island, China" Microorganisms 13, no. 8: 1958. https://doi.org/10.3390/microorganisms13081958
APA StyleYang, T., Qi, Z., Wang, H., Zheng, P., Kao, S.-J., & Diao, X. (2025). Seasonal Variation in In Hospite but Not Free-Living, Symbiodiniaceae Communities Around Hainan Island, China. Microorganisms, 13(8), 1958. https://doi.org/10.3390/microorganisms13081958





