Non-Sexually Transmitted Infection (STI)-Related Pelvic Inflammatory Disease (PID)
Abstract
1. Introduction
2. Epidemiology
3. Microbiology
4. Pathophysiology
5. Clinical Manifestations
6. Diagnosis
6.1. Medical History and Physical Examination
6.2. Diagnostic Workup
6.3. Microscopy
6.4. Imaging
6.4.1. Ultrasound
6.4.2. Computed Tomography (CT)
6.4.3. Magnetic Resonance Imaging (MRI)
6.5. Transcervical Endometrial Biopsy
6.6. Laparoscopy
6.7. Biomarkers
7. Management and Treatment
7.1. Empiric Treatment
7.1.1. Hospitalized Patients
7.1.2. Non-Hospitalized Patients
7.2. Enteric Pathogens
7.3. Anaerobic Pathogen Associated Infections
7.4. PID Due to Actinomycosis
7.5. PID Due to Tuberculosis
7.6. PID Due to CMV
7.7. PID Due to Parasitic Infection
7.8. Surgical Management
8. Complications and Prognosis
9. Follow-Up and Monitoring
10. Prevention
11. Future Directions
12. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AI | Artificial Intelligence |
| AIDS | Acquired Immunodeficiency Syndrome |
| BL/BLI | Beta-lactam/Beta-lactamase inhibitor |
| BV | Bacterial Vaginosis |
| CA-125 | Cancer Antigen 125 |
| CDC | Centers for Disease Control and Prevention |
| CMV | Cytomegalovirus |
| CPP | Chronic Pelvic Pain |
| CRP | C-reactive Protein |
| CRAB | Carbapenem Resistant Acinetobacter baumanii |
| CRE | Carbapenem resistant Enterobacteriales |
| CRPA | Carbapenem resistant Pseudomonas aeruginosa |
| CT | Computed Tomography |
| DNA | Deoxyribonucleic Acid |
| E. coli | Escherichia coli |
| ESBL | Extended-Spectrum β-Lactamase |
| ESR | Erythrocyte Sedimentation Rate |
| FGTB | Female Genital Tract Tuberculosis |
| GI | Gastrointestinal |
| GAS | Group A Streptococcus |
| H. influenzae | Haemophilus influenzae |
| HSG | Hysterosalpingography |
| HIV | Human Immunodeficiency Virus |
| HR | Rifampicin + Isoniazid regimen |
| HRZE | Isoniazid, Rifampicin, Pyrazinamide, and Ethambutol regimen |
| IBD | Inflammatory Bowel Disease |
| IDSA | Infectious Diseases Society of America |
| IFN-γ | Interferon Gamma |
| IgA | Immunoglobulin A |
| IgE | Immunoglobulin E |
| IL-1 | Interleukin-1 |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| IM | Intramuscular |
| IUD | Intrauterine Device |
| IV | Intravenous |
| K. pneumoniae | Klebsiella pneumoniae |
| LPS | Lipopolysaccharide |
| MBL | Metallo-β-lactamase |
| M. genitalium | Mycoplasma genitalium |
| M. hominis | Mycoplasma hominis |
| M. tuberculosis | Mycobacterium tuberculosis |
| MDR | Multidrug-Resistant |
| MMP-9 | Matrix Metalloproteinase-9 |
| MRI | Magnetic Resonance Imaging |
| NAAT | Nucleic Acid Amplification Test |
| NF-κB | Nuclear Factor Kappa-Light-Chain-Enhancer of Activated B Cells |
| NLR | Neutrophil-to-Lymphocyte Ratio |
| N. gonorrhoeae | Neisseria gonorrhoeae |
| N. meningitidis | Neisseria meningitidis |
| P. aeruginosa | Pseudomonas aeruginosa |
| PCR | Polymerase Chain Reaction |
| PID | Pelvic Inflammatory Disease |
| PCT | Procalcitonin |
| RNA | Ribonucleic Acid |
| S. aureus | Staphylococcus aureus |
| S. lugdunensis | Staphylococcus lugdunensis |
| S. pneumoniae | Streptococcus pneumoniae |
| S. viridans | Viridans Group Streptococci |
| STD | Sexually Transmitted Disease |
| STI | Sexually Transmitted Infection |
| TB | Tuberculosis |
| TGF-β | Transforming Growth Factor Beta |
| TLR | Toll-Like Receptor |
| TOA | Tubo-Ovarian Abscess |
| USPSTF | United States Preventive Services Task Force |
| WHO | World Health Organization |
| WBC | White Blood Cell |
References
- Workowski, K.A.; Bachmann, L.H.; Chan, P.A.; Johnston, C.M.; Muzny, C.A.; Park, I.; Reno, H.; Zenilman, J.M.; Bolan, G.A. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm. Rep. 2021, 70, 1–187. [Google Scholar] [CrossRef] [PubMed]
- Weström, L. Incidence, prevalence, and trends of acute pelvic inflammatory disease and its consequences in industrialized countries. Am. J. Obstet. Gynecol. 1980, 138 Pt 2, 880–892. [Google Scholar] [CrossRef]
- Brunham, R.C.; Gottlieb, S.L.; Paavonen, J. Pelvic inflammatory disease. N. Engl. J. Med. 2015, 372, 2039–2048. [Google Scholar] [CrossRef]
- He, D.; Wang, T.; Ren, W. Global burden of pelvic inflammatory disease and ectopic pregnancy from 1990 to 2019. BMC Public Health 2023, 23, 1894. [Google Scholar] [CrossRef] [PubMed]
- Papy, P.; Emmanuel, N.; Agwu, E.; Extension, K.P. Prevalence of Pelvic Inflammatory Disease among Women Attending the the Gynecology Clinic at Kampala International University Teaching Hospital, Uganda. IDOSR J. Sci. Technol. 2022, 9, 1–9. [Google Scholar]
- Yagur, Y.; Weitzner, O.; Barchilon Tiosano, L.; Paitan, Y.; Katzir, M.; Schonman, R.; Klein, Z.; Miller, N. Characteristics of pelvic inflammatory disease caused by sexually transmitted disease—An epidemiologic study. J. Gynecol. Obstet. Human. Reprod. 2021, 50, 102176. [Google Scholar] [CrossRef]
- Surd, A.; Mureșan, R.; Oprea, A.; Snakovszki, K.; Sur, L.M.; Usatiuc, L.-O.; Ciongradi, C.-I.; Sârbu, I. Diagnostic Challenges and Management Strategies of Pelvic Inflammatory Disease in Sexually Inactive Pediatric and Adolescent Patients: A Systematic Review of Case Reports. J. Clin. Med. 2025, 14, 3971. [Google Scholar] [CrossRef]
- Cho, H.-W.; Koo, Y.-J.; Min, K.-J.; Hong, J.-H.; Lee, J.-K. Pelvic Inflammatory Disease in Virgin Women With Tubo-ovarian Abscess: A Single-Center Experience and Literature Review. J. Pediatr. Adolesc. Gynecol. 2017, 30, 203–208. [Google Scholar] [CrossRef]
- Haggerty, C.L.; Peipert, J.F.; Weitzen, S.; Hendrix, S.L.; Holley, R.L.; Nelson, D.B.; Randall, H.; Soper, D.E.; Wiesenfeld, H.C.; Ness, R.B. Predictors of chronic pelvic pain in an urban population of women with symptoms and signs of pelvic inflammatory disease. Sex. Transm. Dis. 2005, 32, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Weström, L.; Joesoef, R.; Reynolds, G.; Hagdu, A.; Thompson, S.E. Pelvic inflammatory disease and fertility. A cohort study of 1,844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex. Transm. Dis. 1992, 19, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Abatangelo, L.; Okereke, L.; Parham-Foster, C.; Parrish, C.; Scaglione, L.; Zotte, D.; Taub, L.F. If pelvic inflammatory disease is suspected empiric treatment should be initiated. J. Am. Acad. Nurse Pract. 2010, 22, 117–122. [Google Scholar] [CrossRef]
- Ness, R.B.; Hillier, S.L.; Kip, K.E.; Soper, D.E.; Stamm, C.A.; McGregor, J.A.; Bass, D.C.; Sweet, R.L.; Rice, P.; Richter, H.E. Bacterial vaginosis and risk of pelvic inflammatory disease. Obstet. Gynecol. 2004, 104, 761–769. [Google Scholar] [CrossRef]
- Cohen, C.R.; Lingappa, J.R.; Baeten, J.M.; Ngayo, M.O.; Spiegel, C.A.; Hong, T.; Donnell, D.; Celum, C.; Kapiga, S.; Delany, S.; et al. Bacterial vaginosis associated with increased risk of female-to-male HIV-1 transmission: A prospective cohort analysis among African couples. PLoS Med. 2012, 9, e1001251. [Google Scholar] [CrossRef]
- Wiesenfeld, H.C.; Sweet, R.L.; Ness, R.B.; Krohn, M.A.; Amortegui, A.J.; Hillier, S.L. Comparison of acute and subclinical pelvic inflammatory disease. Sex. Transm. Dis. 2005, 32, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.M.; Anyalechi, G.E.; Cohen, C.R.; Haggerty, C.L.; Manhart, L.E.; Hillier, S.L. Etiology and Diagnosis of Pelvic Inflammatory Disease: Looking Beyond Gonorrhea and Chlamydia. J. Infect. Dis. 2021, 224 (Suppl. 2), S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Kreisel, K.M.; Llata, E.; Haderxhanaj, L.; Pearson, W.S.; Tao, G.; Wiesenfeld, H.C.; Torrone, E.A. The Burden of and Trends in Pelvic Inflammatory Disease in the United States, 2006-2016. J. Infect. Dis. 2021, 224 (Suppl. 2), S103–S112. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.S.; Horner, P.J.; Price, M.J.; Mitchell, H.D.; Soldan, K. What Do Diagnoses of Pelvic Inflammatory Disease in Specialist Sexual Health Services in England Tell Us About Chlamydia Control? J. Infect. Dis. 2021, 224 (Suppl. 2), S113–S120. [Google Scholar] [CrossRef] [PubMed]
- Al-Kuran, O.A.; Al-Mehaisen, L.; Al-Karablieh, M.; Abu Ajamieh, M.; Flefil, S.; Al-Mashaqbeh, S.; Albustanji, Y.; Al-Kuran, L. Gynecologists and pelvic inflammatory disease: Do we actually know what to do?: A cross-sectional study in Jordan. Medicine 2023, 102, e35014. [Google Scholar] [CrossRef] [PubMed]
- Goller, J.L.; De Livera, A.M.; Fairley, C.K.; Guy, R.J.; Bradshaw, C.S.; Chen, M.Y.; Hocking, J.S. Characteristics of pelvic inflammatory disease where no sexually transmitted infection is identified: A cross-sectional analysis of routinely collected sexual health clinic data. Sex. Transm. Infect. 2017, 93, 68–70. [Google Scholar] [CrossRef]
- Sweeney, S.; Bateson, D.; Fleming, K.; Huston, W. Factors associated with pelvic inflammatory disease: A case series analysis of family planning clinic data. Womens Health 2022, 18, 17455057221112263. [Google Scholar] [CrossRef]
- Metreau, E.; Elizabeth, K.; Shwetha, Y.; Eapen, G. World Bank Country Classifications by Income Level for 2024–2025. Available online: https://blogs.worldbank.org/en/opendata/world-bank-country-classifications-by-income-level-for-2024-2025 (accessed on 22 November 2025).
- World Health Organization. Sexually Transmitted Infections (STIs). Available online: https://www.who.int/news-room/fact-sheets/detail/sexually-transmitted-infections-(stis) (accessed on 20 November 2025).
- Gravett, C.A.; Gravett, M.G.; Martin, E.T.; Bernson, J.D.; Khan, S.; Boyle, D.S.; Lannon, S.M.; Patterson, J.; Rubens, C.E.; Steele, M.S. Serious and life-threatening pregnancy-related infections: Opportunities to reduce the global burden. PLoS Med. 2012, 9, e1001324. [Google Scholar] [CrossRef] [PubMed]
- Hsu, A.; Sarowa, B.K.; Abdessalam, S.F. Ruptured Appendicitis Leading to Development of a Tubo-Ovarian Abscess in a Non-sexually Active Adolescent Patient. Cureus 2023, 15, e41226. [Google Scholar] [CrossRef] [PubMed]
- Center of Disease Control and Prevention. Sexually Transmitted Infections Surveillance. 2021. Available online: https://www.cdc.gov/sti-statistics/media/pdfs/2024/07/2021-STD-Surveillance-Report-PDF_ARCHIVED-2-16-24.pdf (accessed on 22 August 2025).
- Reekie, J.; Donovan, B.; Guy, R.; Hocking, J.S.; Kaldor, J.M.; Mak, D.B.; Pearson, S.; Preen, D.; Stewart, L.; Ward, J.; et al. Risk of Pelvic Inflammatory Disease in Relation to Chlamydia and Gonorrhea Testing, Repeat Testing, and Positivity: A Population-Based Cohort Study. Clin. Infect. Dis. 2018, 66, 437–443. [Google Scholar] [CrossRef]
- Mohllajee, A.P.; Curtis, K.M.; Peterson, H.B. Does insertion and use of an intrauterine device increase the risk of pelvic inflammatory disease among women with sexually transmitted infection? A systematic review. Contraception 2006, 73, 145–153. [Google Scholar] [CrossRef]
- Curtis, K.M.; Jatlaoui, T.C.; Tepper, N.K.; Zapata, L.B.; Horton, L.G.; Jamieson, D.J.; Whiteman, M.K. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. MMWR Recomm. Rep. 2016, 65, 1–66. [Google Scholar] [CrossRef]
- Hubacher, D. Intrauterine devices & infection: Review of the literature. Indian J. Med. Res. 2014, 140 (Suppl. 1), S53–S57. [Google Scholar]
- Pittaway, D.E.; Winfield, A.C.; Maxson, W.; Daniell, J.; Herbert, C.; Wentz, A.C. Prevention of acute pelvic inflammatory disease after hysterosalpingography: Efficacy of doxycycline prophylaxis. Am. J. Obstet. Gynecol. 1983, 147, 623–626. [Google Scholar] [CrossRef]
- Møller, B.R.; Allen, J.; Toft, B.; Hansen, K.B.; Taylor-Robinson, D. Pelvic inflammatory disease after hysterosalpingography associated with Chlamydia trachomatis and Mycoplasma hominis. Br. J. Obstet. Gynaecol. 1984, 91, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- ACOG Practice Bulletin No. 195: Prevention of Infection After Gynecologic Procedures. Obstet. Gynecol. 2018, 131, e172–e189. [CrossRef]
- Mitra, A.; MacIntyre, D.A.; Lee, Y.S.; Smith, A.; Marchesi, J.R.; Lehne, B.; Bhatia, R.; Lyons, D.; Paraskevaidis, E.; Li, J.V.; et al. Cervical intraepithelial neoplasia disease progression is associated with increased vaginal microbiome diversity. Sci. Rep. 2015, 5, 16865. [Google Scholar] [CrossRef]
- Haggerty, C.L.; Totten, P.A.; Tang, G.; Astete, S.G.; Ferris, M.J.; Norori, J.; Bass, D.C.; Martin, D.H.; Taylor, B.D.; Ness, R.B. Identification of novel microbes associated with pelvic inflammatory disease and infertility. Sex. Transm. Infect. 2016, 92, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Hebb, J.K.; Cohen, C.R.; Astete, S.G.; Bukusi, E.A.; Totten, P.A. Detection of novel organisms associated with salpingitis, by use of 16S rDNA polymerase chain reaction. J. Infect. Dis. 2004, 190, 2109–2120. [Google Scholar] [CrossRef]
- Schindlbeck, C.; Dziura, D.; Mylonas, I. Diagnosis of pelvic inflammatory disease (PID): Intra-operative findings and comparison of vaginal and intra-abdominal cultures. Arch. Gynecol. Obstet. 2014, 289, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Greydanus, D.; Bacopoulou, F. Acute pelvic inflammatory disease. Pediatr. Med. 2019, 2, 36. [Google Scholar] [CrossRef]
- Hunter, R.L. Pathology of post primary tuberculosis of the lung: An illustrated critical review. Tuberculosis 2011, 91, 497–509. [Google Scholar] [CrossRef]
- Desai, R.M.; Kumar, S.; Brindini, U. Female genital tuberculosis: A clinicopathological study. Int. J. Reprod. Contracept. Obstet. Gynecol. 2017, 5, 2780–2783. [Google Scholar] [CrossRef][Green Version]
- Namavar Jahromi, B.; Parsanezhad, M.E.; Ghane-Shirazi, R. Female genital tuberculosis and infertility. Int. J. Gynaecol. Obstet. 2001, 75, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, H.; Mohammad, S.; Pathak, P. Tuberculosis in the Female Genital Tract. Cureus 2022, 14, e28708. [Google Scholar] [CrossRef]
- Pradhan, S.; Kyrillos, A.; Hayes, K.; Gromet, E.; Vash-Margita, A.; Pennesi, C.; O’Flynn O’Brien, K.; Gomez-lobo, V. 9. Pelvic Inflammatory Disease in Non-sexually active Pediatric and Adolescent Patients. J. Pediatr. Adolesc. Gynecol. 2020, 33, 242. [Google Scholar] [CrossRef]
- Asemota, O.A.; Girda, E.; Dueñas, O.; Neal-Perry, G.; Pollack, S.E. Actinomycosis pelvic abscess after in vitro fertilization. Fertil. Steril. 2013, 100, 408–411. [Google Scholar] [CrossRef]
- Sawtelle, A.L.; Chappell, N.P.; Miller, C.R. Actinomyces-Related Tubo-Ovarian Abscess in a Poorly Controlled Type II Diabetic With a Copper Intrauterine Device. Mil. Med. 2017, 182, e1874–e1876. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nwanguma, A.; Arora, K. Pseudoactinomycotic Radiate Granules in the Gynecological Tract: A Case Report. Cureus 2024, 16, e69062. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, H.; Zemni, I.; Souissi, M.; Henchiri, H.; Boukhris, S.; Ayadi, M.A.; Achouri, L. Pseudo tumor pelvic actinomycosis revealed by colonic obstruction with hydronephrosis: Can extensive surgery be avoided? A case report. Womens Health 2023, 19, 17455057231181009. [Google Scholar] [CrossRef] [PubMed]
- Saramago, S.M.; Cominho, J.C.; Proença, S.S.M.; Conde, P.J.C.; Nunes, F.M.P. Pelvic Actinomycosis Mimicking Pelvic Malignancy. Rev. Bras. Ginecol. Obstet. 2019, 41, 463–466. [Google Scholar] [CrossRef]
- Bartoš, V.; Doboszová, J.; Sudek, M. Actinomycotic Endomyometritis Associated with a Long-Term Use of Intrauterine Device Lasting for 42 Years. Acta Medica 2019, 62, 35–38. [Google Scholar] [CrossRef]
- González-García, S.M.; Pastrana-Arroyo, M.J.; Medina-Parrilla, E.; González, A.; Martín, J. A Rare Case of Non-IUD-Related Chronic Endometritis caused by Actinomyces Bacteria in a Postmenopausal Woman: A Case Report. P. R. Health Sci. J. 2022, 41, 165–167. [Google Scholar]
- Shunmugam, D.; Shanmugasundaram, S.; Gandhi, A. Rare cause of ovarian mass. BMJ Case Rep. 2018, 2018, bcr-2018-225564. [Google Scholar] [CrossRef]
- Dhillon, A.K.; Fairlie, N.; Finch, G. Pelvic Actinomyces israelii abscess: A differential diagnosis of a pelvic mass. BMJ Case Rep. 2015, 2015, bcr-2015-211595. [Google Scholar] [CrossRef]
- Desteli, G.A.; Gürsu, T.; Bircan, H.Y.; Kızılkılıç, E.; Demiralay, E.; Timurkaynak, F. Thrombocytosis and small bowel perforation: Unusual presentation of abdominopelvic actinomycosis. J. Infect. Dev. Ctries. 2013, 7, 1012–1015. [Google Scholar] [CrossRef][Green Version]
- Lyttle, B.; Johnson, J.V. Chronic Actinomyces Infection Caused by Retained Cervical Cerclage: A Case Report. J. Reprod. Med. 2016, 61, 179–181. [Google Scholar]
- Flynn, A.N.; Lyndon, C.A.; Church, D.L. Identification by 16S rRNA gene sequencing of an Actinomyces hongkongensis isolate recovered from a patient with pelvic actinomycosis. J. Clin. Microbiol. 2013, 51, 2721–2723. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yilmaz, M.; Akbulut, S.; Samdanci, E.T.; Yilmaz, S. Abdominopelvic actinomycosis associated with an intrauterine device and presenting with a rectal mass and hydronephrosis: A troublesome condition for the clinician. Int. Surg. 2012, 97, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.H.; Hong, J.H.; Lee, J.K. Ovarian and vesical actinomycosis: A case report and literature review. Arch. Gynecol. Obstet. 2009, 279, 591–593. [Google Scholar] [CrossRef]
- Pusiol, T.; Morichetti, D.; Pedrazzani, C.; Ricci, F. Abdominal-pelvic actinomycosis mimicking malignant neoplasm. Infect. Dis. Obstet. Gynecol. 2011, 2011, 747059. [Google Scholar] [CrossRef] [PubMed]
- Maxová, K.; Menzlová, E.; Kolařík, D.; Dundr, P.; Halaška, M. Case report: Pelvic actinomycosis. Prague Med. Rep. 2012, 113, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Devendra, K.; Chen, C.M. Pelvic actinomycosis masquerading as an acute abdomen from a small bowel perforation. Singap. Med. J. 2008, 49, 158–159. [Google Scholar]
- Dunn, T.S.; Cothren, C.; Klein, L.; Krammer, T. Pelvic actinomycosis: A case report. J. Reprod. Med. 2006, 51, 435–437. [Google Scholar]
- Iwasaki, M.; Nishikawa, A.; Akutagawa, N.; Fujimoto, T.; Teramoto, M.; Kudo, R. A case of ovarian actinomycosis. Infect. Dis. Obstet. Gynecol. 2003, 11, 171–173. [Google Scholar] [CrossRef]
- Jonas, L.; Baguhl, F.; Wilken, H.P.; Haas, H.J.; Nizze, H. Copper accumulation in actinomyces druses during endometritis after long-term use of an intrauterine contraceptive device. Ultrastruct. Pathol. 2002, 26, 323–329. [Google Scholar] [CrossRef]
- Cobellis, L.; Messalli, E.M.; Pierno, G. Pelvic actinomycosis in menopause: A case report. Maturitas 2001, 39, 79–81. [Google Scholar] [CrossRef]
- Hawnaur, J.M.; Reynolds, K.; McGettigan, C. Magnetic resonance imaging of actinomycosis presenting as pelvic malignancy. Br. J. Radiol. 1999, 72, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, D.; Kustrup, J.F., Jr. Large bowel obstruction due to intrauterine device: Associated pelvic inflammatory disease. Am. Surg. 1999, 65, 1165–1166. [Google Scholar] [CrossRef]
- Lo, T.S.; Chen, F.P.; Chu, K.K.; Soong, Y.K. Advanced actinomycosis involving urogenital organs simulating malignancy: A case report. Chang. Yi Xue Za Zhi 1997, 20, 313–317. [Google Scholar]
- Hinnie, J.; Jaques, B.C.; Bell, E.; Hansell, D.T.; Milroy, R. Actinomycosis presenting as carcinoma. Postgrad. Med. J. 1995, 71, 749–750. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Woo, P.C.; Fung, A.M.; Lau, S.K.; Teng, J.L.; Wong, B.H.; Wong, M.K.; Hon, E.; Tang, G.W.; Yuen, K.Y. Actinomyces hongkongensis sp. nov. a novel Actinomyces species isolated from a patient with pelvic actinomycosis. Syst. Appl. Microbiol. 2003, 26, 518–522. [Google Scholar] [CrossRef]
- Elsayed, S.; George, A.; Zhang, K. Intrauterine contraceptive device-associated pelvic actinomycosis caused by Actinomyces urogenitalis. Anaerobe 2006, 12, 67–70. [Google Scholar] [CrossRef]
- Ferjaoui, M.A.; Arfaoui, R.; Khedhri, S.; Hannechi, M.A.; Abdessamia, K.; Samaali, K.; Fezai, W.; Salhi, M.; Malek, M.; Neji, K. Pelvic actinomycosis: A confusing diagnosis. Int. J. Surg. Case Rep. 2021, 86, 106387. [Google Scholar] [CrossRef]
- Laranjo, M.; Varejão, A.M.; Costa, P.; Peixinho, C. Pelvic actinomycosis: Abdominal mass caused by a forgotten IUD. BMJ Case Rep. 2022, 15, bcr-2022-251392. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Cao, Y.; Zhang, Y.; Niu, L.; Wang, S.; Sang, C. A Case Report of Pelvic Actinomycosis and a Literature Review. Am. J. Case Rep. 2020, 21, e922601. [Google Scholar] [CrossRef] [PubMed]
- Restaino, S.; Gomba, B.; Zero, C.; Stabile, G.; Ronsini, C.; Della Corte, L.; Cianci, S.; Perelli, F.; Piacenti, I.; Driul, L.; et al. Pelvic Actinomycosis and Diagnostic Complexity: Case Report with Literature Review. Healthcare 2025, 13, 485. [Google Scholar] [CrossRef]
- Ayoub, F.; Asour, A.; Miah, A. Case Series of Abdominal Actinomycosis: An Old Diagnostic Conundrum. Cureus 2024, 16, e69763. [Google Scholar] [CrossRef] [PubMed]
- Müller-Holzner, E.; Ruth, N.R.; Abfalter, E.; Schröcksnadel, H.; Dapunt, O.; Martin-Sances, L.; Nogales, F.F. IUD-associated pelvic actinomycosis: A report of five cases. Int. J. Gynecol. Pathol. 1995, 14, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Akhan, S.E.; Dogan, Y.; Akhan, S.; Iyibozkurt, A.C.; Topuz, S.; Yalcin, O. Pelvic actinomycosis mimicking ovarian malignancy: Three cases. Eur. J. Gynaecol. Oncol. 2008, 29, 294–297. [Google Scholar] [PubMed]
- Kayikcioglu, F.; Akif Akgul, M.; Haberal, A.; Faruk Demir, O. Actinomyces infection in female genital tract. Eur. J. Obstet. Gynecol. Reprod. Biol. 2005, 118, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Chudacoff, R.M.; Taylor, R.R. Streptococcus pneumoniae pelvic inflammatory disease. A case report. J. Reprod. Med. 1995, 40, 649–651. [Google Scholar] [PubMed]
- Pasticci, M.B.; Donnini, A.; Mencacci, A.; Lapalorcia, L.M.; Cavazzoni, E.; Baldelli, F. A diagnosis of pneumococcal peritonitis secondary to pyo-salpinx in a young healthy female by culturing peritoneal pus. New Microbiol. 2008, 31, 295–298. [Google Scholar] [PubMed]
- Mack, E.; Wee, H.Y. Pelvic inflammatory disease caused by Streptococcus pneumoniae in a heavy smoker after laparoscopic surgery. Ann. Acad. Med. Singap. 2012, 41, 309–310. [Google Scholar] [CrossRef]
- Sirotnak, A.P.; Eppes, S.C.; Klein, J.D. Tuboovarian abscess and peritonitis caused by Streptococcus pneumoniae serotype 1 in young girls. Clin. Infect. Dis. 1996, 22, 993–996. [Google Scholar] [CrossRef]
- Bucher, A.; Müller, F. Spectrum of abdominal and pelvic infections caused by pneumococci in previously healthy adult women. Eur. J. Clin. Microbiol. Infect. Dis. 2002, 21, 474–477. [Google Scholar] [CrossRef]
- Makhijani, N.; Sondheim, S.E.; Saul, T.; Yetter, E. Loculated Fluid Visualized in Hepatorenal Space with Point-of-care Ultrasound in Patient with Pelvic Inflammatory Disease Caused by Group A Streptococcus: Case Report. Clin. Pract. Cases Emerg. Med. 2024, 8, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Dharia, S.; Shah, S.; Kissinger, M.; Sanders, A.; Singh, G. Group A Streptococcal Endometritis and Toxic Shock causing Septic Pelvic Thrombophlebitis and Septic Pulmonary Emboli. BMJ Case Rep. 2023, 16, bcr-2023-255455. [Google Scholar] [CrossRef] [PubMed]
- Wolfenden, E.; Mittal, M.; Sussman, R. Complex clinical management of group A Streptococcal pelvic inflammatory disease after bilateral tubal ligation in a small community hospital. BMJ Case Rep. 2020, 13, bcr-2020-236326. [Google Scholar] [CrossRef]
- Lusby, H.; Brooks, A.; Hamayoun, E.; Finley, A. Uncommon cause of pelvic inflammatory disease leading to toxic shock syndrome. BMJ Case Rep. 2018, 2018, bcr-2018-224955. [Google Scholar] [CrossRef]
- Lamb, E.K.; Anasti, J.N.; Leonetti, H.B. Group A Streptococcus causing PID from an initial pharyngeal infection. A case report. J. Reprod. Med. 1999, 44, 639–641. [Google Scholar]
- Horii, T.; Izumida, S.; Takeuchi, K.; Tada, T.; Ishikawa, J.; Tsuboi, K. Acute peritonitis and salpingitis associated with streptococcal toxic shock syndrome caused by Lancefield group G alpha-haemolytic Streptococcus dysgalactiae subsp. equisimilis. J. Med. Microbiol. 2006, 55, 953–956. [Google Scholar] [CrossRef] [PubMed]
- Reaves, S.; Mehta, V.; Baxter, J.K.; Ross, R. Postpartum Group A strep sepsis after third trimester uterine prolapse: Case report and literature review. Arch. Gynecol. Obstet. 2022, 306, 1949–1952. [Google Scholar] [CrossRef] [PubMed]
- Riad, M.; Thottacherry, E.; Crawley, C.; Phillip-Abraham, N.; Ibrahim, F. Invasive Group A streptococcal postpartum endometritis associated with multi-organ infarctions: An uncommon case presentation and literature review. Postgrad. Med. 2020, 132, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Blot, M.; de Curraize, C.; Salmon-Rousseau, A.; Gehin, S.; Bador, J.; Chavanet, P.; Neuwirth, C.; Piroth, L.; Amoureux, L. Streptococcus pyogenes: An unusual cause of salpingitis. Case report and review of the literature. Infection 2017, 45, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Solt, I.; Ioffe, Y.; Elmore, R.G.; Solnik, M.J. Group A streptococcal peritonitis and ruptured tubo-ovarian abscess three years after Essure® insertion: A case report. J Womens Health 2011, 20, 781–783. [Google Scholar] [CrossRef] [PubMed]
- Kouijzer, I.J.; Polderman, F.N.; Bekers, E.M.; Bloks, P.H.; Schneeberger, P.M.; de Jager, C.P. Initially unrecognised group A streptococcal pelvic inflammatory disease in a postmenopausal woman. Neth. J. Med. 2014, 72, 494–496. [Google Scholar]
- Snyder, A.; Schmalzle, S.A. Spontaneous Streptococcus pyogenes pelvic inflammatory disease; Case report and review of the literature. IDCases 2020, 20, e00785. [Google Scholar] [CrossRef]
- Gendron, N.; Joubrel, C.; Nedellec, S.; Campagna, J.; Agostini, A.; Doucet-Populaire, F.; Casetta, A.; Raymond, J.; Poyart, C.; Kernéis, S. Group A Streptococcus endometritis following medical abortion. J. Clin. Microbiol. 2014, 52, 2733–2735. [Google Scholar] [CrossRef] [PubMed]
- Garvey, P.; Ledger, W.J. Group a streptococcus in the gynecologic patient. Infect. Dis. Obstet. Gynecol. 1997, 5, 391–394. [Google Scholar] [CrossRef]
- Simpson-Camp, L.; Richardson, E.J.; Alaish, S.M. Streptococcus viridans tubo-ovarian abscess in an adolescent virgin. Pediatr. Int. 2012, 54, 706–709. [Google Scholar] [CrossRef]
- Alenazi, H. Ruptured Appendiceal Diverticulum Leading to Tubo-Ovarian Abscess in a Non-Sexually Active Woman: A Case Study. Am. J. Case Rep. 2024, 25, e945366. [Google Scholar] [CrossRef] [PubMed]
- Mills, D.; Sharon, B.; Schneider, K. Streptococcus constellatus Tubo-ovarian Abscess in a Non-Sexually Active Adolescent Female. Pediatr. Emerg. Care 2018, 34, e100–e101. [Google Scholar] [CrossRef]
- Inglot, M.; Szymczak, A.; Fleischer-Stepniewska, K.; Fleischer, M.; Staszek-Zurowska, B.; Gladysz, A. Tubo-ovarian abscess during therapy of chronic hepatitis C with pegylated interferon and ribavirin. Neuro Endocrinol. Lett. 2011, 32, 1–3. [Google Scholar] [PubMed]
- Algren, S.D.; Strickland, J.L. Beta hemolytic streptococcus group f causing pelvic inflammatory disease in a 14-year-old girl. J. Pediatr. Adolesc. Gynecol. 2005, 18, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Murata, T.; Endo, Y.; Furukawa, S.; Ono, A.; Kiko, Y.; Soeda, S.; Watanabe, T.; Takahashi, T.; Fujimori, K. Successful laparoscopic resection of ovarian abscess caused by Staphylococcus aureus in a 13-year-old girl: A case report and review of literature. BMC Womens Health 2021, 21, 198. [Google Scholar] [CrossRef] [PubMed]
- Punia, R.S.; Aggarwal, R.; Amanjit; Mohan, H. Xanthogranulomatous oophoritis and salpingitis: Late sequelae of inadequately treated staphylococcal PID. Indian. J. Pathol. Microbiol. 2003, 46, 80–81. [Google Scholar] [PubMed]
- Buitrago, M.I.; Crompton, J.A.; Bertolami, S.; North, D.S.; Nathan, R.A. Extremely low excretion of daptomycin into breast milk of a nursing mother with methicillin-resistant Staphylococcus aureus pelvic inflammatory disease. Pharmacotherapy 2009, 29, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Bello, C.; Eskandar, M.; El, G.R.; Sobande, A.; Nour, H.; Shafiq, H. Staphylococcus lugdunensis endometritis: A case report. West. Afr. J. Med. 2007, 26, 243–245. [Google Scholar]
- Ntioudi, M.; Vasiliadou, K.; Charalampidou-Keremidou, P. Idiopathic pyometra and tubo-ovarian abscess in a postmenopausal patient treated conservatively. Ger. Med. Sci. 2022, 20, Doc09. [Google Scholar] [CrossRef]
- Bravender, T.; Matson, S.C. Adolescents, IUDs, PID, and Enterococcus: A report of two cases. J. Pediatr. Adolesc. Gynecol. 2012, 25, e73–e74. [Google Scholar] [CrossRef]
- Covin, B.D.; Chapa, H.; Pham, N. Clostridium perfringens of unclear origin causing pelvic inflammatory disease and toxic shock syndrome in a previously healthy young woman. BMJ Case Rep. 2021, 14, bcr-2021-242492. [Google Scholar] [CrossRef]
- Yavuzcan, A.; Cağlar, M.; Dilbaz, S.; Kumru, S.; Avcioğlu, F.; Ustün, Y. Identification of Clostridium septicum in a tubo-ovarian abscess: A rare case and review of the literature. Vojnosanit. Pregl. 2014, 71, 884–888. [Google Scholar] [CrossRef]
- Fischer, M.; Bhatnagar, J.; Guarner, J.; Reagan, S.; Hacker, J.K.; Van Meter, S.H.; Poukens, V.; Whiteman, D.B.; Iton, A.; Cheung, M.; et al. Fatal toxic shock syndrome associated with Clostridium sordellii after medical abortion. N. Engl. J. Med. 2005, 353, 2352–2360. [Google Scholar] [CrossRef]
- Priputnevich, T.; Lyubasovskaya, L.; Muravieva, V.; Kondrakhin, A.; Ignateva, A.; Gordeev, A.; Shmakov, R.; Sukhikh, G.; Yarotskaya, E. Postpartum endometritis and obstetrical sepsis associated with Eggerthella lenta. Case report and review of the literature. J. Matern. Fetal Neonatal Med. 2021, 34, 313–317. [Google Scholar] [CrossRef]
- Veale, R.; Hughes, C.; Woolley, I. A novel case of bilateral tubo-ovarian abscesses attributed to Ruminococcus gnavus without gastrointestinal involvement. Anaerobe 2021, 67, 102312. [Google Scholar] [CrossRef] [PubMed]
- Gensheimer, W.G.; Reddy, S.Y.; Mulconry, M.; Greves, C. Abiotrophia/Granulicatella tubo-ovarian abscess in an adolescent virginal female. J. Pediatr. Adolesc. Gynecol. 2010, 23, e9–e12. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Muraoka, Y.; Toda, Y.; Kiyomoto, C.; Okubo, Y.; Nagashima, T.; Furukawa, S.; Fujiwara, M.; Mochizuki, M.; Kobayashi, Y.; et al. Finegoldia magna myometritis with uterine necrosis after uterine artery embolisation. J. Obstet. Gynaecol. 2017, 37, 688–689. [Google Scholar] [CrossRef]
- Yamagishi, Y.; Mikamo, H.; Tanaka, K.; Watanabe, K. A case of uterine endometritis caused by Atopobium vaginae. J. Infect. Chemother. 2011, 17, 119–121. [Google Scholar] [CrossRef]
- Carrillo-Ávila, J.A.; Bonilla-García, L.; Navarro-Marí, J.M.; Gutiérrez-Fernández, J. The first reported case of pelvic inflammatory disease caused by Actinobaculum massiliense. Anaerobe 2019, 55, 93–95. [Google Scholar] [CrossRef] [PubMed]
- Naha, K.; Dasari, S.; Vivek, G.; Prabhu, M. Primary abdominal nocardiosis masquerading as tubercular pelvic inflammatory disease in an immunocompetent individual. BMJ Case Rep. 2013, 2013, bcr-2012-008076. [Google Scholar] [CrossRef] [PubMed]
- Bonifaz, A.; Espinosa-Díaz, S.; Argáez, J.; Hernández-Castro, R.; Xicohtencatl-Cortes, J.; Tirado-Sánchez, A. Actinomycetoma due to Nocardia brasiliensis with extension to the ovaries. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 211, 224–225. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goodwin, K.; Fleming, N.; Dumont, T. Tubo-ovarian abscess in virginal adolescent females: A case report and review of the literature. J. Pediatr. Adolesc. Gynecol. 2013, 26, e99–e102. [Google Scholar] [CrossRef]
- Verta, S.; Brambs, C.E.; Christmann, C. Large Douglas Abscess with Distinctive Bilateral Salpingitis in a Young Virginal Woman 6 Months Following Small Bowel Perforation at the Level of the Jejunojejunostomy After Roux-en-Y Gastric Bypass: A Case Report. Int. J. Womens Health 2024, 16, 2343–2354. [Google Scholar] [CrossRef]
- Linck, J.; Torres, W.; Dayal, S. Post-hysteroscopy Ruptured Tubo-Ovarian Abscess With Atypical Bacteremia: A Case Report. Cureus 2023, 15, e45618. [Google Scholar] [CrossRef]
- Chen, K.Y.; Tseng, J.Y.; Yang, C.Y. Tubo-ovarian abscess with sepsis in a nonagenarian woman: A case report and literature review. BMC Womens Health 2019, 19, 81. [Google Scholar] [CrossRef]
- Bernick, J.; Beliavsky, A.; Bogoch, I.I. Endometritis and Bacteremia With a New Delhi Metallo-Beta-Lactamase 1 (NDM-1)-containing Organism in a Remote Traveler. J. Obstet. Gynaecol. Can. 2019, 41, 753–754. [Google Scholar] [CrossRef] [PubMed]
- He, X.F.; Du, X.P.; Qiao, C.F. Successful laparoscopic resection of fallopian tube abscess caused by Escherichia coli in a 12-year-old adolescent virgin:a case report and review of the literature. BMC Pediatr. 2023, 23, 282. [Google Scholar] [CrossRef]
- Boleken, M.E.; Günendi, T.; Yol, C.; Kaya, V.; Kocaman, O.H.; Dörterler, M.E. Xanthogranulomatous Salpingitis Presenting as Pyosalpinx in a Non-Sexually Active Adolescent Girl. J. Pediatr. Adolesc. Gynecol. 2023, 36, 324–327. [Google Scholar] [CrossRef]
- Sabzanov, S.; Ganz, M.; Adout, B.; Farahmandpour, N.; Colarusso, J.; Yusupov, D.; Riznyk, N.; Mishail, B.; Miller, D. Recurrent Tubo-Ovarian Abscesses in a Non-sexually Active Adolescent: A Case Report and Review of Atypical Risk Factors. Cureus 2025, 17, e84671. [Google Scholar] [CrossRef]
- Hornemann, A.; Koschitzky, H.; Bohlmann, M.; Hornung, D.; Diedrich, K.; Tafazzoli, K. Isolated pyosalpinx in a 13-year-old virgin. Fertil. Steril. 2009, 91, e2732.e9–e2732.e10. [Google Scholar] [CrossRef]
- Liberty, G.; Hyman, J.H.; Margalioth, E.J. Peri-implantation pelvic inflammatory disease with normal pregnancy outcome. Fertil. Steril. 2007, 88, 969.e1–969.e2. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Hsieh, Y.Y.; Tsai, H.D.; Lin, C.C. Tubo-ovarian abscess presenting as pneumoperitoneum. J. Assist. Reprod. Genet. 2002, 19, 42–43. [Google Scholar] [CrossRef] [PubMed]
- Gray, Y.; Libbey, N.P. Xanthogranulomatous salpingitis and oophoritis: A case report and review of the literature. Arch. Pathol. Lab. Med. 2001, 125, 260–263. [Google Scholar] [CrossRef] [PubMed]
- Raj, J.A.; Jagadeesha, M.; Naveen, S.; Ramachandra, U. Xanthogranulomatous oophoritis: Pathologic findings with clinical correlation. J. Indian. Med. Assoc. 2012, 110, 653–654. [Google Scholar] [PubMed]
- Martin, D.; Dbouk, R.H.; Deleon-Carnes, M.; del Rio, C.; Guarner, J. Haemophilus influenzae acute endometritis with bacteremia: Case report and literature review. Diagn. Microbiol. Infect. Dis. 2013, 76, 235–236. [Google Scholar] [CrossRef]
- Nishimura, Y.; Hagiya, H.; Kawano, K.; Yokota, Y.; Oka, K.; Iio, K.; Hasegawa, K.; Obika, M.; Haruma, T.; Ono, S.; et al. Invasive non-typeable Haemophilus influenzae infection due to endometritis associated with adenomyosis. BMC Infect. Dis. 2020, 20, 521. [Google Scholar] [CrossRef]
- Valayatham, V. Salmonella: The pelvic masquerader. Int. J. Infect. Dis. 2009, 13, e53–e55. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.H.; Jeng, C.J.; Su, S.C.; Wang, K.G. Pelvic abscess caused by Salmonella: A case report. Zhonghua Yi Xue Za Zhi (Taipei) 1996, 57, 457–459. [Google Scholar]
- Sharma, P.; Bhuju, A.; Tuladhar, R.; Parry, C.M.; Basnyat, B. Tubo-ovarian abscess infected by Salmonella typhi. BMJ Case Rep. 2017, 2017, bcr-2017-221213. [Google Scholar] [CrossRef]
- Kudesia, R.; Gupta, D. Pelvic Salmonella infection masquerading as gynecologic malignancy. Obstet. Gynecol. 2011, 118, 475–477. [Google Scholar] [CrossRef]
- Anandathirtha, K.; Shabnam, Z.; Malempati, L.; Ramesh, N. Xanthogranulomatous endometritis with unilateral salpingo-oophoritis in a postmenopausal woman masquerading as a malignancy. BMJ Case Rep. 2023, 16, e247341. [Google Scholar] [CrossRef]
- King, J.A.; Olsen, T.G.; Lim, R.; Nycum, L.R. Pseudomonas aeruginosa-infected IUD associated with pelvic inflammatory disease. A case report. J. Reprod. Med. 2002, 47, 1035–1037. [Google Scholar]
- Alajeel, A.A.; Garland, S.M. An unusual cause of pelvic inflammatory disease due to Neisseria meningitidis. Sex. Health 2004, 1, 157–160. [Google Scholar] [CrossRef]
- AlHabil, Y.; Owda, A.N.; Zaid, B.J.; Hameedi, S.; Saadeddin, L.; Awad, M.A.A. Concurrent acute cystitis, pancolitis, and tubo-ovarian abscess following laparoscopic ovarian cystectomy: A case report. BMC Womens Health 2024, 24, 489. [Google Scholar] [CrossRef] [PubMed]
- Nernsai, P.; Sophonsritsuk, A.; Lertvikool, S.; Jinawath, A.; Chitasombat, M.N. A case report of Tubo-ovarian abscess caused by Burkholderia pseudomallei. BMC Infect. Dis. 2018, 18, 73. [Google Scholar] [CrossRef] [PubMed]
- Shittu, S.; Athar, S.; Khyatt, O.; Chaponda, M.; Thodi, V.; Al-Maslamani, K.; Alansari, L. Maternal sepsis due to Bacteroides fragilis: A case report and review of the literature. J. Med. Case Rep. 2025, 19, 347. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, A.; Black, A.Y.; Lortie, K.; Fleming, N.A. A case of adolescent pelvic inflammatory disease caused by a rare bacterium: Fusobacterium nucleatum. J. Pediatr. Adolesc. Gynecol. 2013, 26, e113–e115. [Google Scholar] [CrossRef]
- Chayachinda, C.; Leelaporn, A.; Ruangvutilert, P.; Thamkhantho, M. Post-partum, post-sterilization tubo-ovarian abscess caused by Fusobacterium necrophorum: A case report. J. Med. Case Rep. 2012, 6, 330. [Google Scholar] [CrossRef]
- Koshy, K.M.; Malik, W.; Roberts, S.C. Myometritis with pelvic septic vein thrombophlebitis secondary to Fusobacterium necrophorum sepsis. BMJ Case Rep. 2022, 15, bcr-2022-250097. [Google Scholar] [CrossRef]
- Tamura, S.; Jwa, S.C.; Tarumoto, N.; Ishihara, O. Septic Shock Caused by Fusobacterium Necrophorum after Sexual Intercourse during Recovery from Infectious Mononucleosis in an Adolescent: A Case Report. J. Pediatr. Adolesc. Gynecol. 2020, 33, 566–569. [Google Scholar] [CrossRef] [PubMed]
- Tai, C.H.; Kuo, S.F.; Lee, C.H. Concurrency of splenomegaly and numerous enlarged mesenteric and retroperitoneal lymph nodes in a patient with pelvic inflammatory disease caused by Edwardsiella tarda: Mimicking lymphoma. Kaohsiung J. Med. Sci. 2019, 35, 446–447. [Google Scholar] [CrossRef] [PubMed]
- Mora-Palma, J.C.; Rodríguez-Oliver, A.J.; Navarro-Marí, J.M.; Gutiérrez-Fernández, J. Emergent genital infection by Leptotrichia trevisanii. Infect. 2019, 47, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Masschaele, T.; Steyaert, S.; Goethals, R. Leptrotrichia Amnionii, an emerging pathogen of postpartum endometritis. Acta Clin. Belg. 2018, 73, 368–371. [Google Scholar] [CrossRef]
- Tanabe, S.; Uehara, E.; Ichida, K.; Kan, T.; Morishima, S. A case of periodic abdominal pain and fever due to left ovarian abscess. Radiol. Case Rep. 2024, 19, 1361–1365. [Google Scholar] [CrossRef] [PubMed]
- Loïez, C.; Wallet, F.; Husson, M.O.; Courcol, R.J. Pasteurella multocida and intrauterine device: A woman and her pets. Scand. J. Infect. Dis. 2002, 34, 473. [Google Scholar] [CrossRef]
- Roth, T.; Hentsch, C.; Erard, P.; Tschantz, P. Pyosalpinx: Not always a sexual transmitted disease? Pyosalpinx caused by Plesiomonas shigelloides in an immunocompetent host. Clin. Microbiol. Infect. 2002, 8, 803–805. [Google Scholar] [CrossRef][Green Version]
- Kitamura, S.; Matsumura, N.; Ohtake, N.; Kita, M.; Konishi, I. Tubo-ovarian abscess with endometrial cyst probably infected by Campylobacter fetus: Two cases. J. Obstet. Gynaecol. Res. 2016, 42, 1052–1057. [Google Scholar] [CrossRef]
- Walder, G.; Meusburger, H.; Hotzel, H.; Oehme, A.; Neunteufel, W.; Dierich, M.P.; Würzner, R. Chlamydophila abortus pelvic inflammatory disease. Emerg. Infect. Dis. 2003, 9, 1642–1644. [Google Scholar] [CrossRef]
- Nitta, Y.; Shibata, T.; Kato, H.; Nakago, S. Pelvic inflammatory disease associated with cytomegalovirus infection in an immunocompetent adult: Case report and literature review. Clin. Case Rep. 2024, 12, e9323. [Google Scholar] [CrossRef]
- Takagi, I.; Akiyama, H.; Matsuba, H.; Rikitake, J.; Kozuki, Y.; Miyata, Y.; Nakanishi, M.; Inaba, M.; Iwata, N.; Kakiuchi, S. Cytomegalovirus Oophoritis Mimicking Burkitt’s Lymphoma Recurrence: A Case Report and Literature Review. Intern. Med. 2023, 62, 1861–1866. [Google Scholar] [CrossRef] [PubMed]
- Fpathfu, W.A.; Nausheen, Y.; Fpathkfu, R.A.; Khudairi, A.A.; Nemenqani, D. Histopathological features of an incidental case of cytomegalovirus salpingitis in a patient with inflammatory bowel disease. J. Pak. Med. Assoc. 2013, 63, 780–783. [Google Scholar]
- Manfredi, R.; Alampi, G.; Talò, S.; Calza, L.; Tadolini, M.; Martinelli, G.N.; Chiodo, F. Silent oophoritis due to cytomegalovirus in a patient with advanced HIV disease. Int. J. STD AIDS 2000, 11, 410–412. [Google Scholar] [CrossRef] [PubMed]
- Nieto, Y.; Ross, M.; Gianani, R.; Shpall, E.J.; Cagnoni, P.J.; Bearman, S.I.; Jones, R.B. Post-mortem incidental finding of cytomegalovirus oophoritis after an allogeneic stem cell transplant. Bone Marrow Transplant. 1999, 23, 1323–1324. [Google Scholar] [CrossRef][Green Version]
- Hsu, W.C.; Lee, Y.H.; Chang, D.Y. Tuboovarian abscess caused by Candida in a woman with an intrauterine device. Gynecol. Obstet. Invest. 2007, 64, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Calore, E.E.; Calore, N.M.; Cavaliere, M.J. Salpingitis due to Entamoeba histolytica. Braz. J. Infect. Dis. 2002, 6, 97–99. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wadhwa, N.; Raoot, A. An unusual case of adult filarial oophoritis. Int. J. Gynecol. Pathol. 2011, 30, 549–552. [Google Scholar] [CrossRef]
- Chan, R.M.; Lee, P.; Wroblewski, J. Deep-seated trichosporonosis in an immunocompetent patient: A case report of uterine trichosporonosis. Clin. Infect. Dis. 2000, 31, 621. [Google Scholar] [CrossRef]
- Mentessidou, A.; Theocharides, C.; Patoulias, I.; Panteli, C. Enterobius vermicularis-Associated Pelvic Inflammatory Disease in a Child. J. Pediatr. Adolesc. Gynecol. 2016, 29, e25–e27. [Google Scholar] [CrossRef]
- Saleem, F.; Malik, F.; Fatima, S. Enterobius vermicularis in tubo-ovarian abscess: A rare and interesting incidental finding—A case Report. J. Pak. Med. Assoc. 2017, 67, 630–633. [Google Scholar] [PubMed]
- Ngui, R.; Ravindran, S.; Ong, D.B.; Chow, T.K.; Low, K.P.; Nureena, Z.S.; Rajoo, Y.; Chin, Y.T.; Amir, A.; Ahmad, A.F.; et al. Enterobius vermicularis salpingitis seen in the setting of ectopic pregnancy in a Malaysian patient. J. Clin. Microbiol. 2014, 52, 3468–3470. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tandan, T.; Pollard, A.J.; Money, D.M.; Scheifele, D.W. Pelvic inflammatory disease associated with Enterobius vermicularis. Arch. Dis. Child. 2002, 86, 439–440. [Google Scholar] [CrossRef]
- Das, D.K.; Pathan, S.K.; Hira, P.R.; Madda, J.P.; Hasaniah, W.F.; Juma, T.H. Pelvic abscess from enterobius vermicularis. Report of a case with cytologic detection of eggs and worms. Acta Cytol. 2001, 45, 425–429. [Google Scholar] [CrossRef]
- Erhan, Y.; Zekioğlu, O.; Ozdemir, N.; Sen, S. Unilateral salpingitis due to enterobius vermicularis. Int. J. Gynecol. Pathol. 2000, 19, 188–189. [Google Scholar] [CrossRef] [PubMed]
- de Otazu, R.D.; García-Nieto, L.; Izaguirre-Gondra, E.; Mayayo, E.; Ciani, S.; Nogales, F.F. Endometrial coccidiosis. J. Clin. Pathol. 2004, 57, 1104–1105. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, J.H.; Kephart, G.M.; Frankson, J.L. Eosinophilic oophoritis: Association with positive Strongyloides stercoralis serology and clinical response to ivermectin. J. Pediatr. Adolesc. Gynecol. 2006, 19, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Lechner, A.; Bogner, G.; Hasenöhrl, G. Postpartal endomyometritis in a case of unknown tertian malaria. Infection 1997, 25, 185–186. [Google Scholar] [CrossRef]
- Egbe, T.O.; Kobenge, F.M.; Arlette, M.M.J.; Belley-Priso, E. Pyosalpinges after hysterosalpingography in a patient with lower genital tract infection and managed by laparoscopic surgery in a resource low tertiary hospital case report and literature review. Fertil. Res. Pract. 2018, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Andrews, T.J.; Hicar, M.; Islam, S. The Role of a Spinning Top Urethra in the Development of Pyosalpinx in a Precoital Female. Cureus 2022, 14, e27099. [Google Scholar] [CrossRef]
- Colussi, M.; Horwood, G.; McCall, J.; Gale, J.; Singh, S. Septic shock after a saline infusion hysterosalpingosonogram in a woman with stage IV endometriosis and infertility: A case report. Case Rep. Womens Health 2024, 44, e00663. [Google Scholar] [CrossRef]
- Maraqa, T.; Mohamed, M.; Coffey, D.; Sachwani-Daswani, G.R.; Alvarez, C.; Mercer, L. Bilateral recurrent pyosalpinx in a sexually inactive 12-year-old girl secondary to rare variant of Mullerian duct anomaly. BMJ Case Rep. 2017, 2017, bcr-2016-218924. [Google Scholar] [CrossRef]
- King, A.L.; Stamatopoulos, N. Concurrent Escherichia coli tubo-ovarian abscess and Campylobacter jejuni gastroenteritis: A case report. Case Rep. Womens Health 2020, 26, e00192. [Google Scholar] [CrossRef] [PubMed]
- Floyd, R.; Anglim, B. Tubo-ovarian abscess after vaginal delivery: A case report and review of current literature. Case Rep. Womens Health 2023, 39, e00526. [Google Scholar] [CrossRef]
- Noack, F.; Briese, J.; Stellmacher, F.; Hornung, D.; Horny, H.P. Lethal outcome in xanthogranulomatous endometritis. Apmis 2006, 114, 386–388. [Google Scholar] [CrossRef]
- Batool, R.; Abdul Wahab, N.A.; Selvamani, S.; Hennessy, G. Recurrent pyometra coupled with xanthogranulomatous endometritis mimicking pyelonephritis and malignancy: Unravelling clinical complexities. BMJ Case Rep. 2025, 18, bcr-2023-259441. [Google Scholar] [CrossRef]
- Giraldo-Isaza, M.A.; Jaspan, D.; Cohen, A.W. Postpartum endometritis caused by herpes and cytomegaloviruses. Obstet. Gynecol. 2011, 117, 466–467. [Google Scholar] [CrossRef]
- Wu, C.M.; Noska, A. Intrauterine device infection causing concomitant streptococcal toxic shock syndrome and pelvic abscess with Actinomyces odontolyticus bacteraemia. BMJ Case Rep. 2016, 2016, bcr-2015-213236. [Google Scholar] [CrossRef]
- Yu, T. Minimally invasive treatment of uterine necrosis with favorable outcomes: An uncommon case presentation and literature review. BMC Women’s Health 2024, 24, 267. [Google Scholar] [CrossRef] [PubMed]
- Perniola, G.; Di Tucci, C.; Derme, M.; Muzii, L.; Lecce, F.; Benedetti Panici, P. Tuberculous endometritis in woman with abnormal uterine bleeding: A case report and literature review. J. Obstet. Gynaecol. 2021, 41, 671–672. [Google Scholar] [CrossRef] [PubMed]
- Gazos, E.; Tsonis, O.; Gkrozou, F.; Paschopoulos, M. “Silent” post-menopausal genital tuberculosis with lethal outcome. Indian. J. Tuberc. 2020, 67, 357–359. [Google Scholar] [CrossRef]
- Ingec, M.; Erdogan, F.; Kumtepe, Y.; Isaoglu, U.; Gundogdu, C.; Kadanali, S. Management of bilateral fallopian tube carcinoma coexistent with tuberculous salpingitis. J. Obstet. Gynaecol. Res. 2005, 31, 65–67. [Google Scholar] [CrossRef]
- Onuigbo, W.; Esimai, B.; Nwaekpe, C.; Chijioke, G. Tubercular endometritis detected through Pap smear campaign in Enugu, Nigeria. Pan Afr. Med. J. 2012, 11, 47. [Google Scholar]
- Semfke, A.; Wackernagel, C.; Vier, H.; Schütz, A.; Wiechmann, V.; Gillissen, A. Histologically proven isoniazid hepatoxicity in complicated tuberculous salpingitis. Ther. Adv. Respir. Dis. 2009, 3, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Dadhwal, V.; Gupta, N.; Bahadur, A.; Mittal, S. Flare-up of genital tuberculosis following endometrial aspiration in a patient of generalized miliary tuberculosis. Arch. Gynecol. Obstet. 2009, 280, 503–504. [Google Scholar] [CrossRef]
- Cicinelli, E.; Tinelli, R.; Colafiglio, G.; Saliani, N.; Pastore, A. Tubercular endometritis: A rare condition reliably detectable with fluid hysteroscopy. J. Minim. Invasive Gynecol. 2008, 15, 752–754. [Google Scholar] [CrossRef]
- Güngördük, K.; Ulker, V.; Sahbaz, A.; Ark, C.; Tekirdag, A.I. Postmenopausal tuberculosis endometritis. Infect. Dis. Obstet. Gynecol. 2007, 2007, 27028. [Google Scholar] [CrossRef]
- Narayan, R.L.; Patel, A.; McDonald, R.J. Tuberculous peritonitis and tuberculous endometritis diagnosed in the same patient by high clinical suspicion and a minimally invasive approach. Mt. Sinai J. Med. 2006, 73, 1112–1114. [Google Scholar] [PubMed]
- Gascón, J.; Acién, P. Large bilateral tubercular pyosalpinx in a young woman with genitourinary malformation: A case report. J. Med. Case Rep. 2014, 8, 176. [Google Scholar] [CrossRef]
- Dwivedi, K.; Prasad, M. Tuberculous endometritis—An unusual cause of postmenopausal bleeding in the United Kingdom. J. Obstet. Gynaecol. 2016, 36, 124–125. [Google Scholar] [CrossRef]
- Perdhana, R.; Sutrisno, S.; Sugiri, Y.J.; Baktiyani, S.C.; Wiyasa, A. Patients with secondary amenorrhea due to tuberculosis endometritis towards the induced anti-tuberculosis drug category 1. Pan Afr. Med. J. 2016, 24, 121. [Google Scholar] [CrossRef]
- Nayar, M.; Chandra, M.; Chitraratha, K.; Kumari Das, S.; Rai Chowdhary, G. Incidence of actinomycetes infection in women using intrauterine contraceptive devices. Acta Cytol. 1985, 29, 111–116. [Google Scholar]
- Westhoff, C. IUDs and colonization or infection with Actinomyces. Contraception 2007, 75, S48–S50. [Google Scholar] [CrossRef]
- Tzelios, C.; Neuhausser, W.M.; Ryley, D.; Vo, N.; Hurtado, R.M.; Nathavitharana, R.R. Female Genital Tuberculosis. Open Forum Infect. Dis. 2022, 9, ofac543. [Google Scholar] [CrossRef]
- Cohen, C.R.; Mugo, N.R.; Astete, S.G.; Odondo, R.; Manhart, L.E.; Kiehlbauch, J.A.; Stamm, W.E.; Waiyaki, P.G.; Totten, P.A. Detection of Mycoplasma genitalium in women with laparoscopically diagnosed acute salpingitis. Sex. Transm. Infect. 2005, 81, 463–466. [Google Scholar] [CrossRef]
- Lis, R.; Rowhani-Rahbar, A.; Manhart, L.E. Mycoplasma genitalium infection and female reproductive tract disease: A meta-analysis. Clin. Infect. Dis. 2015, 61, 418–426. [Google Scholar] [CrossRef]
- Short, V.L.; Totten, P.A.; Ness, R.B.; Astete, S.G.; Kelsey, S.F.; Haggerty, C.L. Clinical presentation of Mycoplasma genitalium Infection versus Neisseria gonorrhoeae infection among women with pelvic inflammatory disease. Clin. Infect. Dis. 2009, 48, 41–47. [Google Scholar] [CrossRef][Green Version]
- Haggerty, C.L.; Ness, R.B.; Totten, P.A.; Farooq, F.; Tang, G.; Ko, D.; Hou, X.; Fiedler, T.L.; Srinivasan, S.; Astete, S.G.; et al. Presence and Concentrations of Select Bacterial Vaginosis-Associated Bacteria Are Associated With Increased Risk of Pelvic Inflammatory Disease. Sex. Transm. Dis. 2020, 47, 344–346. [Google Scholar] [CrossRef]
- Srinivasan, S.; Hoffman, N.G.; Morgan, M.T.; Matsen, F.A.; Fiedler, T.L.; Hall, R.W.; Ross, F.J.; McCoy, C.O.; Bumgarner, R.; Marrazzo, J.M.; et al. Bacterial communities in women with bacterial vaginosis: High resolution phylogenetic analyses reveal relationships of microbiota to clinical criteria. PLoS ONE 2012, 7, e37818. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, I.M.; Rycroft, A.N.; Dogan, B.; Craven, M.; Bromfield, J.J.; Chandler, A.; Roberts, M.H.; Price, S.B.; Gilbert, R.O.; Simpson, K.W. Specific strains of Escherichia coli are pathogenic for the endometrium of cattle and cause pelvic inflammatory disease in cattle and mice. PLoS ONE 2010, 5, e9192. [Google Scholar] [CrossRef] [PubMed]
- Wiesenfeld, H.C.; Hillier, S.L.; Meyn, L.A.; Amortegui, A.J.; Sweet, R.L. Subclinical pelvic inflammatory disease and infertility. Obstet. Gynecol. 2012, 120, 37–43. [Google Scholar] [CrossRef]
- Nishida, N.; Shono, T.; Shono, K.; Hashimoto, Y.; Kawakami, K. Late Occurrence of the Tubo-Ovarian Abscess after Appendectomy for Perforated Appendicitis in a Virginal Adolescent Girl. J. Pediatr. Adolesc. Gynecol. 2022, 35, 509–511. [Google Scholar] [CrossRef]
- Margaux Becker, V.; Silver, S.; Seufert, R.; Muensterer, O.J. The Association of Appendectomy, Adhesions, Tubal Pathology, and Female Infertility. J. Soc. Laparoendosc. Surg. 2019, 23, e2018.00099. [Google Scholar] [CrossRef]
- Meng, Q.; Gao, Q.; Mehrazarin, S.; Tangwanichgapong, K.; Wang, Y.; Huang, Y.; Pan, Y.; Robinson, S.; Liu, Z.; Zangiabadi, A.; et al. Fusobacterium nucleatum secretes amyloid-like FadA to enhance pathogenicity. EMBO Rep. 2021, 22, e52891. [Google Scholar] [CrossRef]
- Park, J.Y.; Lee, T.S.; Noh, E.J.; Jang, A.R.; Ahn, J.H.; Kim, D.Y.; Jung, D.H.; Song, E.J.; Lee, Y.J.; Lee, Y.J.; et al. Receptor-interacting protein kinase 2 contributes to host innate immune responses against Fusobacterium nucleatum in macrophages and decidual stromal cells. Am. J. Reprod. Immunol. 2021, 86, e13403. [Google Scholar] [CrossRef]
- Sehnal, B.; Beneš, J.; Kolářová, Z.; Mojhová, M.; Zikán, M. Pelvic actinomycosis and IUD. Ceska Gynekol. 2018, 83, 386–390. [Google Scholar] [PubMed]
- Wong, V.K.; Turmezei, T.D.; Weston, V.C. Actinomycosis. BMJ 2011, 343, d6099. [Google Scholar] [CrossRef]
- Akbulut, S.; Arikanoglu, Z.; Basbug, M. Tubercular tubo-ovarian cystic mass mimicking acute appendicitis: A case report. J. Med. Case Rep. 2011, 5, 363. [Google Scholar] [CrossRef][Green Version]
- Hillier, S.L.; Bernstein, K.T.; Aral, S. A Review of the Challenges and Complexities in the Diagnosis, Etiology, Epidemiology, and Pathogenesis of Pelvic Inflammatory Disease. J. Infect. Dis. 2021, 224, S23–S28. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.; Guaschino, S.; Cusini, M.; Jensen, J. 2017 European guideline for the management of pelvic inflammatory disease. Int. J. STD AIDS 2018, 29, 108–114. [Google Scholar] [CrossRef]
- Ross, J.; Cole, M.; Evans, C.; Lyons, D.; Dean, G.; Cousins, D.; PPI Representative; British Association for Sexual Health and HIV. United Kingdom National Guideline for the Management of Pelvic Inflammatory Disease (2019 Interim Update). Available online: https://www.bashh.org/_userfiles/pages/files/resources/pidupdate2019.pdf (accessed on 22 August 2025).
- Korn, A.P. Pelvic inflammatory disease in women infected with HIV. AIDS Patient Care STDS 1998, 12, 431–434. [Google Scholar] [CrossRef]
- Peipert, J.F.; Ness, R.B.; Blume, J.; Soper, D.E.; Holley, R.; Randall, H.; Sweet, R.L.; Sondheimer, S.J.; Hendrix, S.L.; Amortegui, A.; et al. Clinical predictors of endometritis in women with symptoms and signs of pelvic inflammatory disease. Am. J. Obstet. Gynecol. 2001, 184, 856–864. [Google Scholar] [CrossRef]
- Ness, R.B.; Soper, D.E.; Richter, H.E.; Randall, H.; Peipert, J.F.; Nelson, D.B.; Schubeck, D.; McNeeley, S.G.; Trout, W.; Bass, D.C.; et al. Chlamydia antibodies, chlamydia heat shock protein, and adverse sequelae after pelvic inflammatory disease: The PID Evaluation and Clinical Health (PEACH) Study. Sex. Transm. Dis. 2008, 35, 129–135. [Google Scholar] [CrossRef]
- Jenkins, S.M.; Vadakekut, E.S. Pelvic Inflammatory Disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Curry, A.; Williams, T.; Penny, M.L. Pelvic Inflammatory Disease: Diagnosis, Management, and Prevention. Am. Fam. Physician 2019, 100, 357–364. [Google Scholar]
- Liu, Y. Actinomycosis-induced adnexal and uterine masses mimicking malignancy on FDG PET/CT. Am. J. Obstet. Gynecol. 2019, 220, 281. [Google Scholar] [CrossRef] [PubMed]
- Vincent, S.; Lowe, S.; Sumida, M.; Dietrich, J. 89. Pelvic Inflammatory Disease in the Sexually Active and Non-sexually Active Pediatric and Adolescent Population. J. Pediatr. Adolesc. Gynecol. 2025, 38, 273–274. [Google Scholar] [CrossRef]
- Cazanave, C.; de Barbeyrac, B. Pelvic inflammatory diseases: Microbiologic diagnosis—CNGOF and SPILF Pelvic Inflammatory Diseases Guidelines. Gynecol. Obstet. Fertil. Senol. 2019, 47, 409–417. [Google Scholar] [CrossRef]
- Amin-Hanjani, S.; Chatwani, A. Endometrial Cultures in Acute Pelvic Inflammatory Disease. Infect. Dis. Obstet. Gynecol. 1995, 3, 56–59. [Google Scholar] [CrossRef]
- Moore, E.; Soper, D.E. Clinical utility of CA125 levels in predicting laparoscopically confirmed salpingitis in patients with clinically diagnosed pelvic inflammatory disease. Infect. Dis. Obstet. Gynecol. 1998, 6, 182–185. [Google Scholar] [CrossRef]
- Duk, J.M.; Kauer, F.M.; Fleuren, G.J.; de Bruijn, H.W. Serum CA 125 levels in patients with a provisional diagnosis of pelvic inflammatory disease. Clinical and theoretical implications. Acta Obstet. Gynecol. Scand. 1989, 68, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.D.; Young, S.W.; Dahiya, N. Ultrasound of Pelvic Pain in the Nonpregnant Woman. Radiol. Clin. N. Am. 2019, 57, 601–616. [Google Scholar] [CrossRef]
- Dick, E.A.; Blanco, A.; De La Hoz Polo, M.; Basilico, R. ESR Essentials: Gynaecological causes of acute pelvic pain in women: A primer for emergent evaluation-practice recommendations by the European Society of Emergency Radiology. Eur. Radiol. 2025, 35, 6682–6695. [Google Scholar] [CrossRef]
- Franco, P.N.; García-Baizán, A.; Aymerich, M.; Maino, C.; Frade-Santos, S.; Ippolito, D.; Otero-García, M. Gynaecological Causes of Acute Pelvic Pain: Common and Not-So-Common Imaging Findings. Life 2023, 13, 2025. [Google Scholar] [CrossRef]
- Revzin, M.V.; Mathur, M.; Dave, H.B.; Macer, M.L.; Spektor, M. Pelvic Inflammatory Disease: Multimodality Imaging Approach with Clinical-Pathologic Correlation. Radio Graphics 2016, 36, 1579–1596. [Google Scholar] [CrossRef] [PubMed]
- Singla, V.; Dua, A.; Singh, T.; Jain, V. Multimodality imaging of acute gynecological emergencies—A pictorial essay. Abdom. Radiol. 2024, 49, 4042–4056. [Google Scholar] [CrossRef]
- Soper, D.E.; Wiesenfeld, H.C. The Continued Challenges in the Diagnosis of Acute Pelvic Inflammatory Disease: Focus on Clinically Mild Disease. J. Infect. Dis. 2021, 224, S75–S79. [Google Scholar] [CrossRef]
- Schindlbeck, C.; Mylonas, I. Pelvic inflammatory disease: Indication for laparoscopy? Gynakol. Prax. 2012, 36, 663–675. [Google Scholar]
- Bălăceanu, L.A.; Grigore, C.; Dina, I.; Gurău, C.D.; Mihai, M.M.; Bălăceanu-Gurău, B. CA125 as a Potential Biomarker in Non-Malignant Serous Effusions: Diagnostic and Prognostic Considerations. J. Clin. Med. 2025, 14, 4152. [Google Scholar] [CrossRef] [PubMed]
- Bourika, V.; Rekoumi, E.A.; Giamarellos-Bourboulis, E.J. Biomarkers to guide sepsis management. Ann. Intensive Care 2025, 15, 103. [Google Scholar] [CrossRef]
- Genç, S.; Toplu, M.I.; Salman, T.; Halk, E.; Özalp, M.; Çaltek, N.; Mihmanlı, V. Procalcitonin and inflammatory biomarkers in tubo-ovarian abscess: Predicting surgical intervention. Ulus. Travma Acil Cerrahi Derg. 2025, 31, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Aytan, P.; Gökulu, S.G.; Durukan, H.; Bozkurt-Babus, S.; Tasin, C.; Aslan, E.S.; Aytan, H. A New Marker for the Diagnosis of Acute Pelvic Inflammatory Disease: Immature Granulocyte. Clin. Lab. 2024, 70. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, N.; Wong, T. Canadian guidelines on sexually transmitted infections, 2006. Cmaj 2007, 176, 175–176. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ong, J.; Bourne, C.; Dean, J.; Ryder, N.; Cornelisse, V.; Murray, S.; Kenchington, P.; Moten, A.; Gibbs, C.; Maunsell, S.; et al. Australian sexually transmitted infection (STI) management guidelines for use in primary care 2022 update. Sex. Health 2022, 20, 1–8. [Google Scholar] [CrossRef]
- Yusuf, H.; Trent, M. Management of Pelvic Inflammatory Disease in Clinical Practice. Ther. Clin. Risk Manag. 2023, 19, 183–192. [Google Scholar] [CrossRef]
- Ferrero, S.; Leone Roberti Maggiore, U.; Paudice, M.; Vellone, V.G.; Perrone, U.; Barra, F. Safety and efficacy of pharmacotherapies for pelvic inflammatory disease and endometriosis. Expert. Opin. Drug Saf. 2025, 24, 273–286. [Google Scholar] [CrossRef]
- Verdon, R. Treatment of uncomplicated pelvic inflammatory disease: CNGOF and SPILF Pelvic Inflammatory Diseases Guidelines. Gynecol. Obstet. Fertil. Senol. 2019, 47, 418–430. [Google Scholar] [CrossRef]
- Dalby, J.; Stoner, B.P. Sexually Transmitted Infections: Updates from the 2021 CDC Guidelines. Am. Fam. Physician 2022, 105, 514–520. [Google Scholar]
- Zhou, T.; Yuan, M.; Cui, P.; Li, J.; Jia, F.; Wang, S.; Liu, R. Effectiveness and safety of morinidazole in the treatment of pelvic inflammatory disease: A multicenter, prospective, open-label phase IV trial. Front Med. 2022, 9, 888186. [Google Scholar] [CrossRef]
- Mohammed, H.; Bokhary, R.; Nassif, M.; Mosli, M. Ovarian Crohn’s Disease: A Case Report and Review of the Literature. Case Rep. Gastrointest. Med. 2020, 2020, 1826469. [Google Scholar] [CrossRef] [PubMed]
- Canas, A.M.; Holloran-Schwartz, B.; Myles, T. Tuboovarian abscess 12 years after total abdominal hysterectomy. Obstet. Gynecol. 2004, 104, 1039–1041. [Google Scholar] [CrossRef]
- Landers, D.V.; Sweet, R.L. Tubo-ovarian abscess: Contemporary approach to management. Rev. Infect. Dis. 1983, 5, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Itani, K.M.; Wilson, S.E.; Awad, S.S.; Jensen, E.H.; Finn, T.S.; Abramson, M.A. Ertapenem versus cefotetan prophylaxis in elective colorectal surgery. N. Engl. J. Med. 2006, 355, 2640–2651. [Google Scholar] [CrossRef] [PubMed]
- Pelak, B.A.; Citron, D.M.; Motyl, M.; Goldstein, E.J.; Woods, G.L.; Teppler, H. Comparative in vitro activities of ertapenem against bacterial pathogens from patients with acute pelvic infection. J. Antimicrob. Chemother. 2002, 50, 735–741. [Google Scholar] [CrossRef][Green Version]
- Sartelli, M.; Chichom-Mefire, A.; Labricciosa, F.M.; Hardcastle, T.; Abu-Zidan, F.M.; Adesunkanmi, A.K.; Ansaloni, L.; Bala, M.; Balogh, Z.J.; Beltrán, M.A.; et al. The management of intra-abdominal infections from a global perspective: 2017 WSES guidelines for management of intra-abdominal infections. World J. Emerg. Surg. 2017, 12, 29. [Google Scholar] [CrossRef]
- Solomkin, J.S.; Mazuski, J.E.; Bradley, J.S.; Rodvold, K.A.; Goldstein, E.J.C.; Baron, E.J.; O’Neill, P.J.; Chow, A.W.; Dellinger, E.P.; Eachempati, S.R.; et al. Diagnosis and Management of Complicated Intra-abdominal Infection in Adults and Children: Guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin. Infect. Dis. 2010, 50, 133–164. [Google Scholar] [CrossRef]
- Tamma, P.D.; Heil, E.L.; Justo, J.A.; Mathers, A.J.; Satlin, M.J.; Bonomo, R.A. Infectious Diseases Society of America 2024 Guidance on the Treatment of Antimicrobial-Resistant Gram-Negative Infections. Clin. Infect. Dis. 2024, ciae403. [Google Scholar] [CrossRef]
- Al-Hasan, M.N.; Eckel-Passow, J.E.; Baddour, L.M. Impact of healthcare-associated acquisition on community-onset Gram-negative bloodstream infection: A population-based study: Healthcare-associated Gram-negative BSI. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1163–1171. [Google Scholar] [CrossRef]
- Schechner, V.; Nobre, V.; Kaye, K.S.; Leshno, M.; Giladi, M.; Rohner, P.; Harbarth, S.; Anderson, D.J.; Karchmer, A.W.; Schwaber, M.J.; et al. Gram-negative bacteremia upon hospital admission: When should Pseudomonas aeruginosa be suspected? Clin. Infect. Dis. 2009, 48, 580–586. [Google Scholar] [CrossRef]
- Micek, S.T.; Welch, E.C.; Khan, J.; Pervez, M.; Doherty, J.A.; Reichley, R.M.; Kollef, M.H. Empiric combination antibiotic therapy is associated with improved outcome against sepsis due to Gram-negative bacteria: A retrospective analysis. Antimicrob. Agents Chemother. 2010, 54, 1742–1748. [Google Scholar] [CrossRef] [PubMed]
- Noor, A.; Khetarpal, S. Anaerobic Infections. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Valour, F.; Sénéchal, A.; Dupieux, C.; Karsenty, J.; Lustig, S.; Breton, P.; Gleizal, A.; Boussel, L.; Laurent, F.; Braun, E.; et al. Actinomycosis: Etiology, clinical features, diagnosis, treatment, and management. Infect. Drug Resist. 2014, 7, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Skoutelis, A.; Petrochilos, J.; Bassaris, H. Successful treatment of thoracic actinomycosis with ceftriaxone. Clin. Infect. Dis. 1994, 19, 161–162. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Hall, V.; Thakker, B.; Gemmell, C.G. Antimicrobial susceptibility testing of Actinomyces species with 12 antimicrobial agents. J. Antimicrob. Chemother. 2005, 56, 407–409. [Google Scholar] [CrossRef] [PubMed]
- Stone, B.; Patel, T.G.; De Silva, T.I.; Green, S.T.; Chapman, A.L.N. Successful treatment of actinomyces brain abscesses using once daily parenteral ceftriaxone in an outpatient setting: A case series. J. Infect. 2008, 57, 426–427. [Google Scholar] [CrossRef]
- Fuchs, P.C.; Barry, A.L.; Brown, S.D. In vitro activities of ertapenem (MK-0826) against clinical bacterial isolates from 11 North American medical centers. Antimicrob. Agents Chemother. 2001, 45, 1915–1918. [Google Scholar] [CrossRef][Green Version]
- Martin, M.V. The use of oral amoxycillin for the treatment of actinomycosis. A clinical and in vitro study. Br. Dent. J. 1984, 156, 252–254. [Google Scholar] [CrossRef]
- Bonifaz, A.; Tirado-Sánchez, A.; Calderón, L.; Montes de Oca, G.; Torres-Camacho, P.; Ponce, R.M. Treatment of cutaneous actinomycosis with amoxicillin/clavulanic acid. J. Dermatol. Treat. 2017, 28, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, K.; Nord, C.E.; Dornbusch, K. Antimicrobial in vitro susceptibility of Actinomyces israelii and arachnia propionica. Scand. J. Infect. Dis. 1977, 9, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Lerner, P.I. Susceptibility of pathogenic actinomycetes to antimicrobial compounds. Antimicrob. Agents Chemother. 1974, 5, 302–309. [Google Scholar] [CrossRef][Green Version]
- Choi, J.; Koh, W.J.; Kim, T.S.; Lee, K.S.; Han, J.; Kim, H.; Kwon, O.J. Optimal duration of IV and oral antibiotics in the treatment of thoracic actinomycosis. Chest 2005, 128, 2211–2217. [Google Scholar] [CrossRef]
- Hamid, D.; Baldauf, J.J.; Cuenin, C.; Ritter, J. Treatment strategy for pelvic actinomycosis: Case report and review of the literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2000, 89, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Sudhakar, S.S.; Ross, J.J. Short-term treatment of actinomycosis: Two cases and a review. Clin. Infect. Dis. 2004, 38, 444–447. [Google Scholar] [CrossRef]
- Trutnovsky, G.; Tamussino, K.; Reich, O. Short-term antibiotic treatment of pelvic actinomycosis. Int. J. Gynaecol. Obstet. 2008, 101, 203–204. [Google Scholar] [CrossRef]
- Hirshberg, A.; Tsesis, I.; Metzger, Z.; Kaplan, I. Periapical actinomycosis: A clinicopathologic study. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2003, 95, 614–620. [Google Scholar] [CrossRef]
- Shree, K.K.; Somashekar, S.; Loganathan, E. Cytomegalovirus induced genital ulcer in human immunodeficiency virus positive patient. Indian. J. Sex. Transm. Dis. AIDS 2022, 43, 198–200. [Google Scholar] [CrossRef] [PubMed]
- Kotton, C.N.; Kumar, D.; Manuel, O.; Chou, S.; Hayden, R.T.; Danziger-Isakov, L.; Asberg, A.; Tedesco-Silva, H.; Humar, A.; on behalf of The Transplantation Society International CMV Consensus Group. The Fourth International Consensus Guidelines on the Management of Cytomegalovirus in Solid Organ Transplantation. Transplantation 2025, 109, 1066–1110. [Google Scholar] [CrossRef] [PubMed]
- Smolyakov, R.; Talalay, B.; Yanai-Inbar, I.; Pak, I.; Alkan, M. Enterobius vermicularis infection of female genital tract: A report of three cases and review of literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 107, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Rawla, P.; Sharma, S. Enterobius Vermicularis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Chou, A.; Austin, R.L. Entamoeba histolytica Infection. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Brun, J.-L.; Castan, B.; de Barbeyrac, B.; Cazanave, C.; Charvériat, A.; Faure, K.; Mignot, S.; Verdon, R.; Fritel, X.; Graesslin, O. Pelvic inflammatory diseases: Updated French guidelines. J. Gynecol. Obstet. Human. Reprod. 2020, 49, 101714. [Google Scholar] [CrossRef]
- Demirtas, O.; Akman, L.; Demirtas, G.S.; Hursitoglu, B.S.; Yilmaz, H. The role of the serum inflammatory markers for predicting the tubo-ovarian abscess in acute pelvic inflammatory disease: A single-center 5-year experience. Arch. Gynecol. Obstet. 2013, 287, 519–523. [Google Scholar] [CrossRef]
- Ribak, R.; Schonman, R.; Sharvit, M.; Schreiber, H.; Raviv, O.; Klein, Z. Can the Need for Invasive Intervention in Tubo-ovarian Abscess Be Predicted? The Implication of C-reactive Protein Measurements. J. Minim. Invasive Gynecol. 2020, 27, 541–547. [Google Scholar] [CrossRef]
- Hwang, J.H.; Kim, B.W.; Kim, S.R.; Kim, J.H. The prediction of surgical intervention in patients with tubo-ovarian abscess. J. Obstet. Gynaecol. 2022, 42, 97–102. [Google Scholar] [CrossRef]
- Marshall, A.; Wimsett, J.; Handforth, C.; Unsworth, L.; Wilson, J.; Van Der Merwe, A.M.; Oyston, C. The Tubo-ovarian abscess study (TOAST): A single-center retrospective review of predictors of failed medical management. Int. J. Gynaecol. Obstet. 2025, 170, 927–935. [Google Scholar] [CrossRef]
- Shigemi, D.; Matsui, H.; Fushimi, K.; Yasunaga, H. Laparoscopic Compared With Open Surgery for Severe Pelvic Inflammatory Disease and Tubo-Ovarian Abscess. Obstet. Gynecol. 2019, 133, 1224–1230. [Google Scholar] [CrossRef]
- Witkin, S.S.; Minis, E.; Athanasiou, A.; Leizer, J.; Linhares, I.M. Chlamydia trachomatis: The Persistent Pathogen. Clin. Vaccine Immunol. 2017, 24, 10. [Google Scholar] [CrossRef] [PubMed]
- Colombel, J.F.; Shin, A.; Gibson, P.R. AGA Clinical Practice Update on Functional Gastrointestinal Symptoms in Patients With Inflammatory Bowel Disease: Expert Review. Clin. Gastroenterol. Hepatol. 2019, 17, 380–390.e1. [Google Scholar] [CrossRef] [PubMed]
- Hakim, J.; Childress, K.J.; Hernandez, A.M.; Bercaw-Pratt, J.L. Tubo-Ovarian Abscesses in Nonsexually Active Adolescent Females: A Large Case Series. J. Adolesc. Health 2019, 65, 303–305. [Google Scholar] [CrossRef] [PubMed]
- Kairys, N.; Roepke, C. Tubo-Ovarian Abscess. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Lareau, S.M.; Beigi, R.H. Pelvic inflammatory disease and tubo-ovarian abscess. Infect. Dis. Clin. N. Am. 2008, 22, 693–708. [Google Scholar] [CrossRef]
- Zhu, M.; Huang, F.; Xu, J.; Chen, W.; Ding, B.; Shen, Y. Risk factors and nomogram construction for predicting women with chronic pelvic pain:a cross-sectional population study. Heliyon 2024, 10, e34534. [Google Scholar] [CrossRef]
- Howard, F.M. Chronic Pelvic Pain, Pelvic Inflammatory Disease and Adhesions. In Encyclopedia of Pain; Gebhart, G.F., Schmidt, R.F., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 652–655. [Google Scholar]
- Haggerty, C.L.; Schulz, R.; Ness, R.B. Lower quality of life among women with chronic pelvic pain after pelvic inflammatory disease. Obstet. Gynecol. 2003, 102, 934–939. [Google Scholar] [CrossRef]
- Weström, L. Pelvic inflammatory disease. JAMA 1991, 266, 2612. [Google Scholar] [CrossRef]
- Møller, B.R.; Kristiansen, F.V.; Thorsen, P.; Frost, L.; Mogensen, S.C. Sterility of the uterine cavity. Acta Obstet. Gynecol. Scand. 1995, 74, 216–219. [Google Scholar] [CrossRef]
- Ness, R.B.; Soper, D.E.; Holley, R.L.; Peipert, J.; Randall, H.; Sweet, R.L.; Sondheimer, S.J.; Hendrix, S.L.; Amortegui, A.; Trucco, G.; et al. Effectiveness of inpatient and outpatient treatment strategies for women with pelvic inflammatory disease: Results from the Pelvic Inflammatory Disease Evaluation and Clinical Health (PEACH) Randomized Trial. Am. J. Obstet. Gynecol. 2002, 186, 929–937. [Google Scholar] [CrossRef]
- Kawwass, J.F.; Crawford, S.; Kissin, D.M.; Session, D.R.; Boulet, S.; Jamieson, D.J. Tubal factor infertility and perinatal risk after assisted reproductive technology. Obstet. Gynecol. 2013, 121, 1263–1271. [Google Scholar] [CrossRef]
- Hunt, S.; Vollenhoven, B. Pelvic inflammatory disease and infertility. Aust. J. General. Pract. 2023, 52, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tan, X.; Lv, A. Association between pelvic inflammatory disease and infertility in American women: Results from the National Health and Nutrition Survey 2013 to 2018. BMC Women’s Health 2025, 25, 578. [Google Scholar]
- Haggerty, C.L.; Ness, R.B. Diagnosis and treatment of pelvic inflammatory disease. Womens Health 2008, 4, 383–397. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Huang, C.C.; Lin, S.Y.; Chang, C.Y.; Lin, W.C.; Chung, C.H.; Lin, F.H.; Tsao, C.H.; Lo, C.M.; Chien, W.C. Association of pelvic inflammatory disease (PID) with ectopic pregnancy and preterm labor in Taiwan: A nationwide population-based retrospective cohort study. PLoS ONE 2019, 14, e0219351. [Google Scholar] [CrossRef] [PubMed]
- Brim, A.C.S.; Barretto, V.R.D.; Reis-Oliveira, J.G.; da Silveira de Araújo, R.B.; Romeo, A. Risk factors for ectopic pregnancy occurrence: Systematic review and meta-analysis. Int. J. Gynaecol. Obstet. 2025, 168, 919–932. [Google Scholar] [CrossRef]
- Quintar, A.A.; Mukdsi, J.H.; del Valle Bonaterra, M.; Aoki, A.; Maldonado, C.A.; Pérez Alzaa, J. Increased expression of uteroglobin associated with tubal inflammation and ectopic pregnancy. Fertil. Steril. 2008, 89, 1613–1617. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhao, W.H.; Zhu, Q.; Cao, S.J.; Ping, H.; Xi, X.; Qin, G.J.; Yan, M.X.; Zhang, D.; Qiu, J.; et al. Risk factors for ectopic pregnancy: A multi-center case-control study. BMC Pregnancy Childbirth 2015, 15, 187. [Google Scholar] [CrossRef]
- Hufstetler, K.; Llata, E.; Miele, K.; Quilter, L.A.S. Clinical Updates in Sexually Transmitted Infections, 2024. J. Womens Health 2024, 33, 827–837. [Google Scholar] [CrossRef]
- Lehtoranta, L.; Ala-Jaakkola, R.; Laitila, A.; Maukonen, J. Healthy Vaginal Microbiota and Influence of Probiotics Across the Female Life Span. Front. Microbiol. 2022, 13, 819958. [Google Scholar] [CrossRef]
- Jonsson, S.; Jonsson, H.; Lundin, E.; Häggström, C.; Idahl, A. Pelvic inflammatory disease and risk of epithelial ovarian cancer: A national population-based case-control study in Sweden. Am. J. Obstet. Gynecol. 2024, 230, e71–e75. [Google Scholar] [CrossRef]
- Stewart, L.M.; Stewart, C.J.R.; Spilsbury, K.; Cohen, P.A.; Jordan, S. Association between pelvic inflammatory disease, infertility, ectopic pregnancy and the development of ovarian serous borderline tumor, mucinous borderline tumor and low-grade serous carcinoma. Gynecol. Oncol. 2020, 156, 611–615. [Google Scholar] [CrossRef]
- Towns, J.M.; Williamson, D.A.; Bradshaw, C.S. Case of Mycoplasma genitalium pelvic inflammatory disease with perihepatitis. Sex. Transm. Infect. 2021, 97, 628. [Google Scholar] [CrossRef]
- Loehr, S.; Bitter, C. Fitz Hugh Curtis Case Report. J. Educ. Teach. Emerg. Med. 2020, 5, V19–V21. [Google Scholar] [CrossRef] [PubMed]
- Ronghe, V.; Modak, A.; Gomase, K.; Mahakalkar, M.G. From Prevention to Management: Understanding Postoperative Infections in Gynaecology. Cureus 2023, 15, e46319. [Google Scholar] [CrossRef] [PubMed]
- Low, N.; Mueller, M.; Van Vliet, H.A.; Kapp, N. Perioperative antibiotics to prevent infection after first-trimester abortion. Cochrane Database Syst. Rev. 2012, 2012, Cd005217. [Google Scholar] [CrossRef]
- Petousis, S.; Angelou, P.; Almperis, A.; Laganà, A.S.; Titilas, G.; Margioula-Siarkou, C.; Dinas, K. Prophylactic Antibiotics before Gynecologic Surgery: A Comprehensive Review of Guidelines. J. Pers. Med. 2024, 14, 327. [Google Scholar] [CrossRef]
- Marcinkowski, K.A.; Mehta, V.; Mercier, R.; Berghella, V. Pelvic inflammatory disease in pregnancy: A systematic review focusing on perinatal outcomes. Am. J. Obstet. Gynecol. MFM 2022, 4, 100643. [Google Scholar] [CrossRef]
- Center of Disease Control and Prevention. Sexually Transmitted Infections Surveillance. 2023. Available online: https://www.cdc.gov/sti-statistics/annual/index.html (accessed on 22 August 2025).
- Li, T.; Liu, Z.H.; Li, K.; Bai, H.H. Evaluation of the vaginal microbiome in clinical diagnosis and management of vaginal infectious diseases. Chin. Med. J. 2019, 132, 1100–1103. [Google Scholar] [CrossRef]
- Graspeuntner, S.; Bohlmann, M.K.; Gillmann, K.; Speer, R.; Kuenzel, S.; Mark, H.; Hoellen, F.; Lettau, R.; Griesinger, G.; König, I.R.; et al. Microbiota-based analysis reveals specific bacterial traits and a novel strategy for the diagnosis of infectious infertility. PLoS ONE 2018, 13, e0191047. [Google Scholar] [CrossRef]
- Rana, S.; Panjwal, P.; Malik, N.; Bhatia, V.; Kushawaha, S.K.; Ashawat, M.S. The silent threat: Pelvic inflammatory disease and long-term health outcomes with management. J. Endometr. Pelvic Pain. Disord. 2025, 17, 22840265251350207. [Google Scholar] [CrossRef]
- Hasan, Z.; Begum, N.; Ahmed, S.; Yasmin, M. Association of opportunistic bacterial pathogens with female infertility: A case-control study. J. Obstet. Gynaecol. Res. 2025, 51, e16243. [Google Scholar] [CrossRef]
- Filardo, S.; Di Pietro, M.; Porpora, M.G.; Recine, N.; Farcomeni, A.; Latino, M.A.; Sessa, R. Diversity of Cervical Microbiota in Asymptomatic Chlamydia trachomatis Genital Infection: A Pilot Study. Front. Cell Infect. Microbiol. 2017, 7, 321. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Niu, X.; Han, N.; Wang, B.; Du, P.; Na, R.; Chen, C.; Liao, Q. Predictive value of the composition of the vaginal microbiota in bacterial vaginosis, a dynamic study to identify recurrence-related flora. Sci. Rep. 2016, 6, 26674. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, E.; Kim, S.; Tomita, K.; Minase, T.; Kayano, M.; Watanabe, H.; Tetsuka, M.; Sasaki, M.; Iwayama, H.; Sanai, H.; et al. Evaluation of Lipopolysaccharide and Interleukin-6 as Useful Screening Tool for Chronic Endometritis. Int. J. Mol. Sci. 2024, 25, 2017. [Google Scholar] [CrossRef] [PubMed]

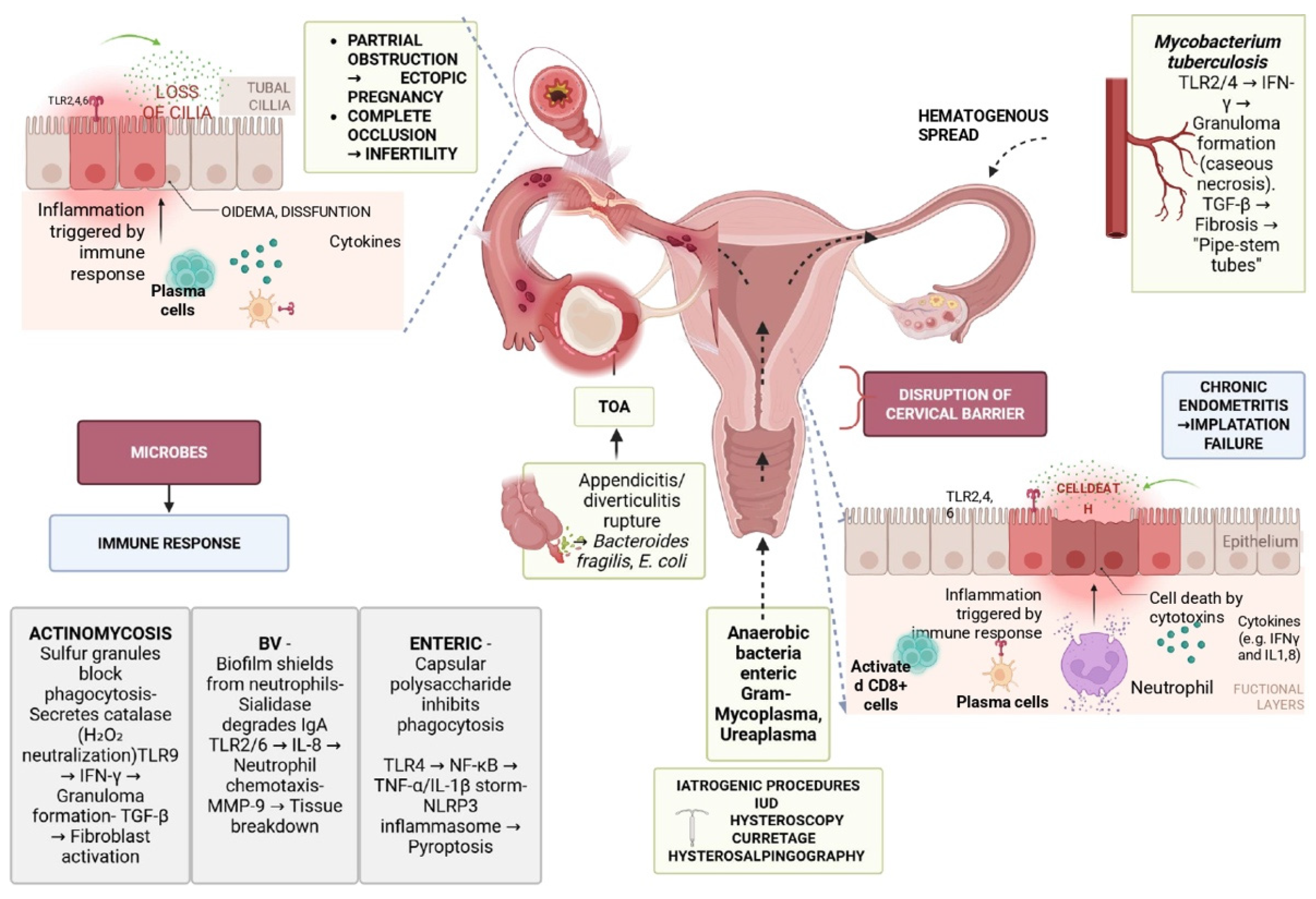
| Microorganism Identified | Number of Cases | References |
|---|---|---|
| Gram positive | ||
| Actinomyces spp. | 35 | [43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77] |
| S. pneumoniae | 5 | [78,79,80,81,82] |
| GAS | 14 | [83,84,85,86,87,88,89,90,91,92,93,94,95,96] |
| S. Viridans | 4 | [97,98,99,100] |
| Beta hemolytic streptococcus group F | 1 | [101] |
| S. aureus | 3 | [102,103,104] |
| S. lugdunensis | 1 | [105] |
| Enterococcus spp. | 2 | [106,107] |
| Clostridium spp. | 3 | [108,109,110] |
| Eggerthella lenta | 1 | [111] |
| Ruminococcus gnavus | 1 | [112] |
| Abiotrophia/Granulicatella spp. | 1 | [113] |
| Finegoldia magna | 1 | [114] |
| Atopobium vaginae | 1 | [115] |
| Actinobaculum massiliense | 1 | [116] |
| Nocardia spp. | 2 | [117,118] |
| Gram negative | ||
| E. coli | 13 | [119,120,121,122,123,124,125,126,127,128,129,130,131] |
| H. influenzae | 2 | [132,133] |
| Salmonella spp. | 4 | [134,135,136,137] |
| K. pneumoniae | 1 | [138] |
| P. aeruginosa | 1 | [139] |
| N. meningitidis | 1 | [140] |
| ESBL-Gram-negative pathogen | 1 | [141] |
| Burkholderia pseudomallei | 1 | [142] |
| B. fragilis | 1 | [143] |
| Fusobacterium spp. | 4 | [144,145,146,147] |
| Edwardsiella tarda | 1 | [148] |
| Leptotrichia spp. | 2 | [149,150] |
| Citrobacter freundii | 1 | [151] |
| Pasteurella multocida | 1 | [152] |
| Porphyromonas asaccharolytica | 1 | [100] |
| Plesiomonas shigelloides | 1 | [153] |
| Cambylobacter fetus | 1 | [154] |
| Chlamydophila abortus | 1 | [155] |
| Viruses | ||
| CMV | 5 | [156,157,158,159,160] |
| Fungi | ||
| Candida spp. | 1 | [161] |
| Trichosporon beigelii | 1 | [162] |
| Parasites | ||
| Filarian worm-pathogen not identified | 1 | [163] |
| Entamoeba histolytica | 1 | [164] |
| Enterobius vermicularis | 6 | [165,166,167,168,169,170] |
| Coccidia spp. | 1 | [171] |
| Strongyloides stercoralis | 1 | [172] |
| Plasmodium vivax | 1 | [173] |
| Polymicrobial | 12 | [24,174,175,176,177,178,179,180,181,182,183,184] |
| Mycobacterium spp. | 15 | [185,186,187,188,189,190,191,192,193,194,195,196] |
| Empiric Treatment (Hospitalized Individuals) | ||
|---|---|---|
| Regimen | Dosage | Notes |
| Ceftriaxone or cefoxitin or cefotetan + Doxycycline + Metronidazole | Ceftriaxone 1 g IV q24h/Cefoxitin 2 g IV q6h/Cefotetan 2 g IV q12h plus Doxycycline 100 mg orally or IV q12h plus Metronidazole 500 mg orally or IV q12h | Oral doxycycline is preferred if tolerated due to equivalent bioavailability. |
| Alternative regimens | ||
| Clindamycin + Gentamicin | Clindamycin 900 mg IV q8h plus Gentamicin 3–5 mg/kg IV daily or 2 mg/kg loading dose then 1.5 mg/kg q8h | |
| Ampicillin–sulbactam + Doxycycline | Ampicillin–sulbactam 3 g IV q6h plus Doxycycline 100 mg q12h | |
| Ampicillin + Clindamycin + Gentamicin | Ampicillin 2 g IV q6h plus Clindamycin 900 mg IV q8h plus Gentamicin 2 mg/kg loading dose then 1.5 mg/kg q8h | |
| Imipenem-cilastatin | Imipenem-cilastatin 500 mg IV q6h | |
| Azithromycin or moxifloxacin or levofloxacin + Metronidazole | Azithromycin 500 mg IV daily for 1–2 days, then 250 mg orally daily for 7 days or moxifloxacin 400 mg orally once daily or levofloxacin 500 mg IV once daily plus Metronidazole 500 mg orally q12h | Alternative if cephalosporin allergy is present. |
| Empiric Treatment (Non-Hospitalized individuals) | ||
| Ceftriaxone or Cefoxitin or Cefotaxime, or Ceftizoxime + Doxycycline or azithromycin + Metronidazole | Ceftriaxone 500 mg IM (<150 kg) or 1 g IM (≥150 kg), Cefoxitin 2 g IM with Probenecid 1 g orally, Cefotaxime 1 g IM, or Ceftizoxime 1 g IM + Doxycycline 100 mg orally q12h or Azithromycin 500 mg daily for 1–2 days, then 250 mg daily (or 1 g once weekly for 2 weeks) plus Metronidazole 500 mg orally q12h | For patients who are intolerant to doxycycline, azithromycin is preferred. |
| Doxycycline + Clindamycin | Doxycycline 100 mg q12h plus Clindamycin 450 mg q6h | For patients who are intolerant to metronidazole. |
| Levofloxacin or Moxifloxacin + Metronidazole | Levofloxacin 500 mg qd or Moxifloxacin 400 mg orally qd plus Metronidazole 500 mg q12h | Alternative if cephalosporin allergy is present. |
| Targeted Therapy (for specific pathogens) | ||
| Enterobacterales and P. aeruginosa | ||
| Ceftriaxone or cefepime or ceftazidime | Ceftriaxone 2 g IV qd or Ceftazidime 2 g IV q8h or Cefepime 2 g IV q8h | Ceftazidime and Cefipime provide coverage for P. aeruginosa. |
| Aztreonam or Ciprofloxacin or Levofloxacin | Aztreonam 2 g every q8h or Ciprofloxacin 400 mg IV q12h or Levofloxacin 750 mg IV qd | For severe b-lactam allergy. |
| Ertapenem or Piperacillin-tazobactam or Meropenem or Imipenem | Ertapenem 1 g IV qd or Piperacillin-tazobactam 4.5 g q6h or Meropenem 2 g q8h or Imipenem 500 mg IV q6h | Alternative regimens. Ertapenem does not provide coverage for P. aeruginosa. |
| Ceftolozane-tazobactam | Ceftolozane-tazobactam 2.5 g q8h | Suitable for ESBL-producing pathogens. |
| Ceftazidime-Avibactam or Imipenem-Cilastatin-Relebactam or Meropenem-Vaborbactam or Cefiderocol | Ceftazidime-Avibactam 2.5 g q8h IV or Imipenem-Cilastatin-Relebactam 1.25 g q6h IV or Meropenem-Vaborbactam 4 g q8h IV or Cefiderocol 2g q8h IV | Reserved for CRE, CRPA or CRAB. |
| Anaerobes | ||
| Metronidazole | Metronidazole 500 mg q12h | First-choice regimen. |
| Clindamycin or Amoxicillin–clavulanate or Τicarcillin–clavulanate or Piperacillin–tazobactam or Ampicillin–sulbactam or Meropenem or Imipenem | Clindamycin 450 mg q6h or Amoxicillin–clavulanate 1 g q8h or Τicarcillin–clavulanate 3.1g q6h or Piperacillin–tazobactam 4.5 g q6h or Ampicillin–sulbactam 3 g q6h or Meropenem 2 g q8h or Imipenem 500 mg IV q6h | Alternative regimen. |
| PID due to Actinomycosis | ||
| Penicillin G IV or Ampicillin IV followed by Penicillin V or Amoxicillin per os | Penicillin G 20–24 million units daily, divided q4–6h or Ampicillin IV 2 g q4–6h followed by Penicillin V or Amoxicillin per os 2–3 g per day, divided in 3–4 doses | |
| 2 g q4–6h | Alternative regimen. | |
| Ceftriaxone IV | 2 g qd | For non-severe or IgE-mediated allergy. |
| Doxycycline IV | 100 mg q12h | Preferred for severe non-IgE-mediated allergy. |
| Carbapenems (e.g., Ertapenem) | 1 g IV every 24 h | For severe allergy. |
| Doxycycline or Tetracycline or Erythromycin or Azithromycin per os | Doxycycline 100 mg q12h or Tetracycline 500 mg q6h or Erythromycin 500 mg q6h or Azithromycin 500 mg qd | For penicillin-allergic patients. |
| PID due to CMV | ||
| Valganciclovir per os | 900 mg q12h | Mild–moderate CMV disease. |
| Ganciclovir IV | 5 mg/kg q12h | Severe/life-threatening CMV disease. |
| Maribavir or foscarnet | Dose individualized | Alternative regimen. |
| PID due to parasitic infection | ||
| Enterobius vermicularis | ||
| Albendazole | 400 mg single dose repeated after 2 weeks | |
| Mebendazole or | Mebendazole 100 mg single dose, repeated after 2 weeks or Pyrantel pamoate 11 mg/kg (max 1 g) single dose, repeated after 2 weeks | Alternative option.. |
| Entamoeba histolytica | ||
| Metronidazole + Luminal Agent (Paromomycin or Diiodohydroxyquin or Diloxanide furoate) | Metronidazole 500–750 mg orally q8h plus Paromomycin: 25–30 mg/kg/day divided q8h Diiodohydroxyquin: 650 mg orally Diloxanide furoate: 500 mg orally q8h | Alternatives to metronidazole include tinidazole, ornidazole, nitazoxanide |
| PID due to Tuberculosis | ||
| Isoniazid + Rifampicin + Pyrazinamide + Ethambutol followed by two drug regimen (Isoniazid+ Rifampicin) next 8 weeks | Isoniazid 300 mg daily plus Rifampicin 10 mg/kg (8–12 mg) once daily plus Pyrazinamide 1000 mg (40–<55 kg), 1500 mg (55–75 kg), 2000 mg (>75 kg) daily plus Ethambutol 15 mg/kg orally once a day for initial treatment or Initial dose: 25 mg/kg orally once a day for 60 days Maintenance dose: 15 mg/kg orally once a day for previous antituberculosis treatment | (First 8 weeks) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polyzou, E.; Ntalaki, E.; Gavatha, M.; Akinosoglou, K. Non-Sexually Transmitted Infection (STI)-Related Pelvic Inflammatory Disease (PID). Microorganisms 2025, 13, 2813. https://doi.org/10.3390/microorganisms13122813
Polyzou E, Ntalaki E, Gavatha M, Akinosoglou K. Non-Sexually Transmitted Infection (STI)-Related Pelvic Inflammatory Disease (PID). Microorganisms. 2025; 13(12):2813. https://doi.org/10.3390/microorganisms13122813
Chicago/Turabian StylePolyzou, Eleni, Evangelia Ntalaki, Maria Gavatha, and Karolina Akinosoglou. 2025. "Non-Sexually Transmitted Infection (STI)-Related Pelvic Inflammatory Disease (PID)" Microorganisms 13, no. 12: 2813. https://doi.org/10.3390/microorganisms13122813
APA StylePolyzou, E., Ntalaki, E., Gavatha, M., & Akinosoglou, K. (2025). Non-Sexually Transmitted Infection (STI)-Related Pelvic Inflammatory Disease (PID). Microorganisms, 13(12), 2813. https://doi.org/10.3390/microorganisms13122813






