Synergistic Effects of Combined Flavourzyme and Floating Electrode–Dielectric Barrier Discharge Plasma on Reduction of Escherichia coli Biofilms in Squid (Todarodes pacificus)
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strain, Culture and Growth Condition
2.2. Flavourzyme Preparation and Determination of Minimum Inhibitory Concentration (MIC)
2.3. Sample Preparation and Biofilm Formation
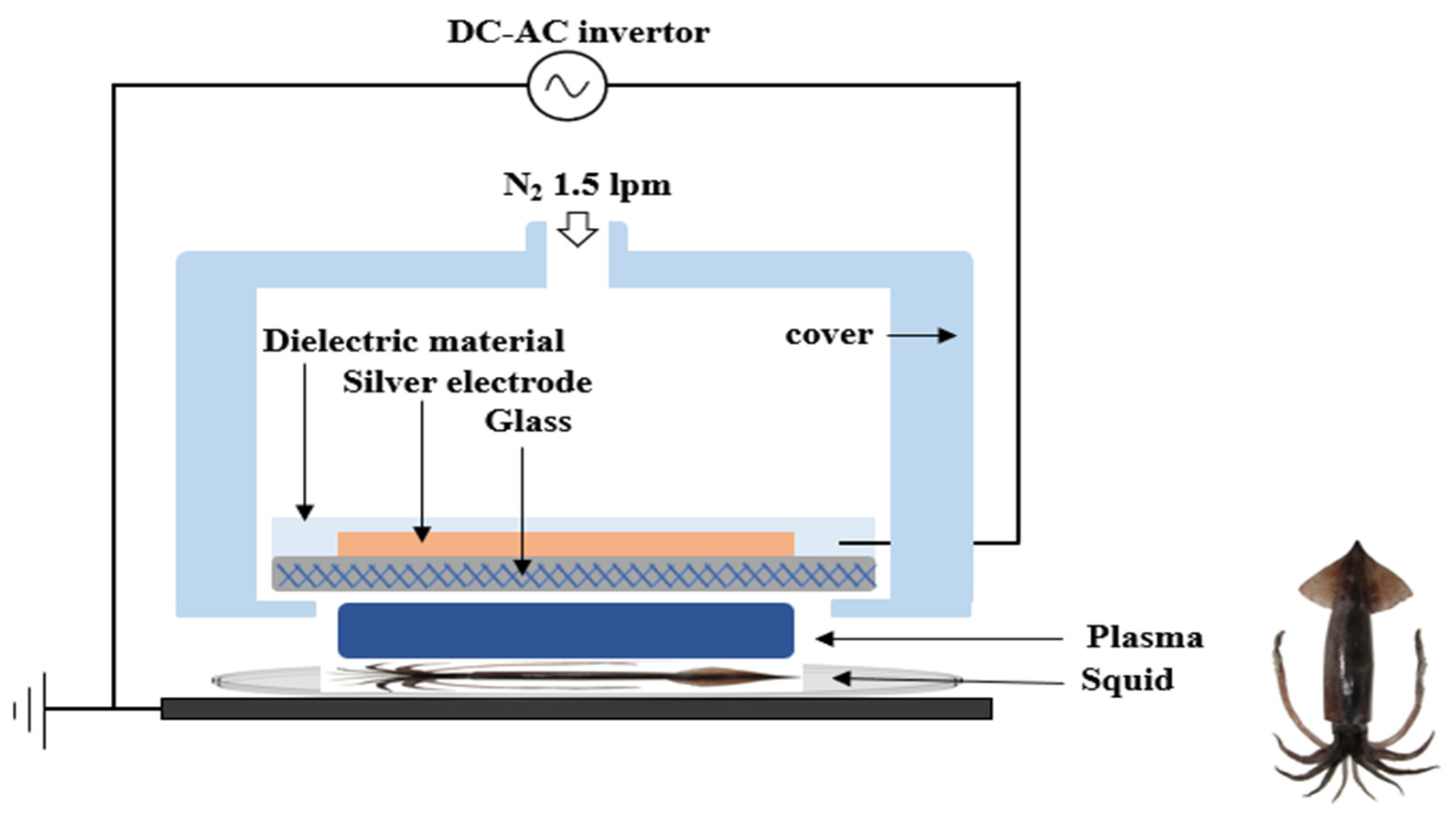
2.4. FE-DBD Plasma Treatment
2.5. Combination Treatment and the Synergistic Reduction Effect Analysis
2.6. Biofilm Detachment Process
2.7. pH Value Measurement
2.8. Statistical Analysis with a Method to Check Normal Distribution
3. Results
3.1. Synergistic Reduction of E. coli Biofilm with Combined Flavourzyme and FE-DBD Plasma Treatment in Squid
3.2. pH Value of Squid Treated with Flavourzyme and FE-DBD Plasma
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FDA (Food and Drug Administration). Foodborne Pathogens. Available online: https://www.fda.gov/food/outbreaks-foodborne-illness/foodborne-pathogens (accessed on 12 September 2023).
- Boelaert, F.; Rizzi, V.; Stoicescu, A.V. Manual for reporting on zoonoses and zoonotic agents, within the framework of Directive 2003/99/EC, and on some other pathogenic microbiological agents for information derived from the year 2018. EFSA Supp. Publ. 2019, 16, 1560E. [Google Scholar] [CrossRef]
- Nahar, S.; Ha, A.J.; Byun, K.H.; Hossain, A.I.; Mizan, M.F.R.; Ha, S.D. Efficacy of flavourzyme against Salmonella Typhimurium, Escherichia coli, and Pseudomonas aeruginosa biofilms on food-contact surfaces. Int. J. Food Microbiol. 2021, 336, 108897. [Google Scholar] [CrossRef] [PubMed]
- Sharm, G.; Sharma, S.; Sharma, P.; Chandola, D.; Dang, S.; Gupta, S.; Gabrani, R. Escherichia coli biofilm: Development and therapeutic strategies. J. Appl. Microbiol. 2016, 121, 309–319. [Google Scholar] [CrossRef]
- Zhou, F.; Wang, D.; Hu, J.; Zhang, Y.; Tan, B.K.; Lin, S. Control measurements of Escherichia coli biofilm: A review. Foods 2022, 11, 2469. [Google Scholar] [CrossRef] [PubMed]
- Corzo-Ariyama, H.A.; García-Heredia, A.; Heredia, N.; García, S.; León, J.; Jaykus, L.; Solís-Soto, L. Phylogroups, pathotypes, biofilm formation and antimicrobial resistance of Escherichia coli isolates in farms and packing facilities of tomato, jalapeño pepper and cantaloupe from Northern Mexico. Int. J. Food Microbiol. 2019, 290, 96–104. [Google Scholar] [CrossRef]
- Liu, C.; Zhao, Y.; Su, W.; Chai, J.; Xu, L.; Cao, J.; Liu, Y. Encapsulated DNase improving the killing efficiency of antibiotics in staphylococcal biofilms. J. Mater. Chem. B 2020, 8, 4395–4401. [Google Scholar] [CrossRef] [PubMed]
- Adator, E.H.; Cheng, M.; Holley, R.; McAllister, T.; Narvaez-Bravo, C. Ability of Shiga toxigenic Escherichia coli to survive within dry-surface biofilms and transfer to fresh lettuce. Int. J. Food Microbiol. 2018, 269, 52–59. [Google Scholar] [CrossRef]
- Fouladkhah, A.; Geornaras, I.; Sofos, J.N. Biofilm formation of O157 and non-O157 shiga toxin-producing Escherichia coli and multidrug-resistant and susceptible Salmonella Typhimurium and Newport and their inactivation by sanitizers. J. Food Sci. 2013, 78, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.K.; Park, S.H.; Song, M.G.; Park, S.Y. Antimicrobial efficacy of quercetin against Vibrio parahaemolyticus biofilm on food surfaces and downregulation of virulence genes. Polymers 2022, 14, 3847. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.K.; Ha, A.J.W.; Nahar, S.; Hossain, M.I.; Ashrafudoulla, M.; Toushik, S.H.; Mizan, M.F.R.; Kang, I.; Ha, S.D. Inhibitory effects of vorinostat (SAHA) against food-borne pathogen Salmonella enterica serotype Kentucky mixed culture biofilm with virulence and quorum-sensing relative expression. Biofouling 2023, 39, 617–628. [Google Scholar] [CrossRef]
- Nahar, S.; Jeong, H.L.; Kim, Y.; Ha, A.J.W.; Roy, P.K.; Park, S.H.; Ashrafudoulla, M.; Mizan, M.F.R.; Ha, S.D. Inhibitory effects of Flavourzyme on biofilm formation, quorum sensing, and virulence genes of foodborne pathogens Salmonella Typhimurium and Escherichia coli. Food Res. Int. 2021, 147, 110461. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.; Nag, M.; Sarkar, T.; Dutta, B.; Ray, R.R. Antibiofilm activity of α-amylase from Bacillus subtilis and prediction of the optimized conditions for biofilm removal by response surface methodology (RSM) and artificial neural network (ANN). Appl. Biochem. Biotechnol. 2021, 193, 1853–1872. [Google Scholar] [CrossRef] [PubMed]
- Merz, M.; Eisele, T.; Berends, P.; Appel, D.; Rabe, S.; Blank, I.; Stressler, T.; Fischer, L. Flavourzyme, an enzyme preparation with industrial relevance: Automated nine-step purification and partial characterization of eight enzymes. J. Agric. Food Chem. 2015, 63, 5682–5693. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, L.; Venkitasamy, C.; Guo, S.; Pan, Z.; Ke, H.; Tang, H.; Huang, W.; Zhao, L. Improving the flavor of microbone meal with flavourzyme by response surface method. J. Food Process Eng. 2019, 42, 13040. [Google Scholar] [CrossRef]
- Yang, J.; Jiang, C.; Bao, R.; Liu, M.; Yang, Z.; Xu, W.; Liang, H.; Ji, C.; Li, S.; Zhang, S.; et al. Effects of flavourzyme addition on physicochemical properties, volatile compound components and microbial community succession of Suanzhayu. Int. J. Food Microbiol. 2020, 334, 108839. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yang, J.; Suo, H.; Tan, J.; Zhang, Y.; Song, J. Identification and molecular mechanism of action of antibacterial peptides from flavourzyme-hydrolyzed yak casein against Staphylococcus aureus. J. Dairy Sci. 2023, 106, 3779–3790. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Liu, D.; Xiang, Q.; Ahn, J.; Chen, S.; Ye, X.; Ding, T. Inactivation mechanisms of non-thermal plasma on microbes: A review. Food Control 2017, 75, 83–91. [Google Scholar] [CrossRef]
- Jeon, E.B.; Choi, M.-S.; Kim, J.Y.; Choi, E.H.; Lim, J.S.; Choi, J.; Ha, K.S.; Kwon, J.Y.; Jeong, S.H.; Park, S.Y. Assessment of potential infectivity of human norovirus in the traditional Korean salted clam product “Jogaejeotgal” by floating electrode-dielectric barrier discharge plasma. Food Res. Int. 2021, 141, 110107. [Google Scholar] [CrossRef] [PubMed]
- Svarnas, P.; Giannakopoulos, E.; Kalavrouziotis, I.; Krontiras, C.; Georga, S.; Pasolari, R.S.; Papadopoulos, P.K.; Apostolou, I.; Chrysochoou, D. Sanitary effect of FE-DBD cold plasma in ambient air on sewage biosolids. Sci. Total Environ. 2020, 705, 135940. [Google Scholar] [CrossRef]
- Žigon, J.; Zaplotnik, R.; Ayata, Ü.; Petrič, M.; Dahle, S. The influence of artificial weathering and treatment with FE–DBD plasma in atmospheric conditions on wettability of wood surfaces. Niedersächsisches Symp. Mater. Clausthal. 2019, 14, 559. [Google Scholar]
- Adil, B.H.; Al-Shammari, A.M.; Murbat, H.H. Breast cancer treatment using cold atmospheric plasma generated by the FE-DBD scheme. Clin. Plasma Med. 2020, 19, 100103. [Google Scholar] [CrossRef]
- Joshi, S.G.; Cooper, M.; Yost, A.; Paff, M.; Ercan, U.K.; Fridman, G.; Friedman, G.; Fridman, A.; Brooks, A.D. Nonthermal dielectric-barrier discharge plasma-induced inactivation involves oxidative DNA damage and membrane lipid peroxidation in Escherichia coli. Antimicrob. Agents Chemother. 2011, 55, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Song, M.G.; Roy, P.K.; Jeon, E.B.; Kim, S.H.; Heu, M.S.; Lee, J.S.; Choi, J.S.; Kim, J.S.; Park, S.Y. Effect of Dielectric Barrier discharge plasma against Listeria monocytogenes mixed-culture biofilms on food-contact surfaces. Antibiotics 2023, 12, 609. [Google Scholar] [CrossRef] [PubMed]
- Yong, H.I.; Lee, S.H.; Kim, S.Y.; Park, S.; Park, J.; Choe, W.; Jo, C. Color development, physiochemical properties, and microbiological safety of pork jerky processed with atmospheric pressure plasma. Innov. Food Sci. Emerg. Technol. 2019, 53, 78–84. [Google Scholar] [CrossRef]
- Koban, I.; Geisel, M.H.; Holtfreter, B.; Jablonowski, L.; Hübner, N.O.; Matthes, R.; Masur, K.; Weltmann, K.D.; Kramer, A.; Kocher, T. Synergistic effects of nonthermal plasma and disinfecting agents against dental biofilms in vitro. Int. Sch. Res. Not. 2013, 2013, 573262. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Li, W.; Li, C.; Lin, L. Synergistic effect between Helichrysum italicum essential oil and cold nitrogen plasma against Staphylococcus aureus biofilms on different food-contact surfaces. Food Sci. Technol. 2016, 51, 2493–2501. [Google Scholar] [CrossRef]
- Roy, K.P.; Song, M.G.; Park, S.Y. Impact of quercetin against Salmonella Typhimurium biofilm formation on food-contact surfaces and molecular mechanism pattern. Foods 2022, 11, 977. [Google Scholar] [CrossRef] [PubMed]
- Toushik, S.H.; Park, J.-H.; Kim, K.; Ashrafudoulla; Ulrich, M.S.I.; Mizan, F.R.; Roy, P.K.; Shim, W.-B.; Kim, Y.-M.; Park, S.H.; et al. Antibiofilm efficacy of Leuconostoc mesenteroides J. 27-derived postbiotic and food-grade essential oils against Vibrio parahaemolyticus, Pseudomonas aeruginosa, and Escherichia coli alone and in combination, and their application as a green preservative in the seafood industry. Food Res. Int. 2022, 156, 111163. [Google Scholar] [CrossRef] [PubMed]
- Koivunen, J.; Heinonen-Tanski, H. Inactivation of enteric microorganisms with chemical disinfectants, UV irradiation and combined chemical/UV treatments. Water Res. 2005, 39, 1519–1526. [Google Scholar] [CrossRef]
- Serra, D.O.; Hengge, R. Bacterial multicellularity: The biology of Escherichia coli building large-scale biofilm communities. Annu. Rev. Microbiol. 2021, 75, 269–290. [Google Scholar] [CrossRef]
- Mukherjee, S.; Lekshmi, M.; Ammini, P.; Nayak, B.B.; Kumar, S.H. Survival of the blaNDM-harbouring Escherichia coli in tropical seawater and conjugative transfer of resistance markers. Arch. Microbiol. 2021, 203, 4273–4279. [Google Scholar] [CrossRef] [PubMed]
- Prakasan, S.; Lekshmi, M.; Ammini, P.; Balange, A.K.; Nayak, B.B.; Kumar, S.H. Occurrence, pathogroup distribution and virulence genotypes of Escherichia coli from fresh seafood. Food Control 2022, 133, 108669. [Google Scholar] [CrossRef]
- Ryu, S.H.; Park, S.G.; Choi, S.M.; Hwang, Y.O.; Ham, H.J.; Kim, S.U.; Lee, Y.K.; Kim, M.S.; Park, G.Y.; Kim, K.S.; et al. Antimicrobial resistance and resistance genes in Escherichia coli strains isolated from commercial fish and seafood. Int. J. Food Microbiol. 2012, 152, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Sofos, J.N.; Geornaras, I. Overview of current meat hygiene and safety risks and summary of recent studies on biofilms, and control of Escherichia coli O157:H7 in nonintact, and Listeria monocytogenes in ready-to-eat, meat products. Meat Ence. 2010, 86, 2–14. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, C.G.C. Assessing manufacturers recommended concentrations of commercial sanitizers on inactivation of Listeria monocytogenes. Food Control 2012, 26, 194–199. [Google Scholar]
- Elchinger, P.H.; Delattre, C.; Faure, S.; Roy, O.; Badel, S.; Bernardi, T.; Taillefumier, C.; Michaud, P. Effect of protease against biofilms of Staphylococcus aureus and Staphylococcus epidermidis. Lett. Appl. Microbiol. 2014, 59, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Dong, R.Y.; Ma, L.; Qian, Y.L.; Liu, Z.Y. Combined anti-biofilm enzymes strengthen the eradicate effect of Vibrio parahaemolyticus biofilm: Mechanism on cpsA-J expression and application on different carriers. Foods 2022, 11, 1305. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Xu, R.; Gou, L.; Liu, Z.; Zhao, Y.; Liu, D.; Zhang, L.; Chen, H.; Kong, M.G. Mechanism of virus inactivation by cold atmospheric-pressure plasma and plasma-activated water. Appl. Environ. Microbiol. 2018, 84, e00726-18. [Google Scholar] [CrossRef] [PubMed]
- Niemira, B.A.; Boyd, G.; Sites, J. Cold plasma rapid decontamination of food contact surfaces contaminated with Salmonella biofilms. J. Food Sci. 2014, 79, M917–M922. [Google Scholar] [CrossRef]
- Modic, M.; McLeod, N.P.; Sutton, J.M.; Walsh, J.L. Cold atmospheric pressure plasma elimination of clinically important single-and mixed-species biofilms. Int. J. Antimicrob. Agents 2017, 49, 375–378. [Google Scholar] [CrossRef]
- Khosravi, S.; Jafari, S.; Zamani, H.; Nilkar, M. Inactivation of Staphylococcus aureus and Escherichia coli biofilms by air-based atmospheric-pressure DBD plasma. Appl. Biochem. Biotechnol. 2021, 193, 3641–3650. [Google Scholar] [CrossRef] [PubMed]
- Czapka, T.; Maliszewska, I.; Olesiak-Bańska, J. Influence of atmospheric pressure non-thermal plasma on inactivation of biofilm cells. Plasma Chem. Plasma Process. 2018, 38, 1181–1197. [Google Scholar] [CrossRef]
- Roy, P.K.; Jeon, E.B.; Park, S.Y. Effects of nonthermal dielectric barrier discharge plasma against Listeria monocytogenes and quality of smoked salmon fillets. J. Food Saf. 2022, 42, 13012. [Google Scholar] [CrossRef]
- Jeon, E.B.; Choi, M.S.; Kim, J.Y.; Park, S.Y. Synergistic effects of mild heating and dielectric barrier discharge plasma on the reduction of Bacillus cereus in red pepper powder. Foods 2020, 9, 171. [Google Scholar] [CrossRef] [PubMed]
- Baik, K.Y.; Jo, H.; Ki, S.H.; Kwon, G.C.; Cho, G. Synergistic effect of hydrogen peroxide and cold atmospheric pressure plasma-jet for microbial disinfection. Appl. Sci. 2023, 13, 3324. [Google Scholar] [CrossRef]
- Pankaj, S.K.; Misra, N.N.; Cullen, P.J. Kinetics of tomato peroxidase inactivation by atmospheric pressure cold plasma based on dielectric barrier discharge. Innov. Food Sci. Emerg. Technol. 2013, 19, 153–157. [Google Scholar] [CrossRef]
| Microorganisms | Flavourzyme Concentration (MIC) | FE-DBD Plasma Treatment (min) | ||||
|---|---|---|---|---|---|---|
| 0 | 5 | 10 | 30 | 60 | ||
| E. coli | 0 | - | 0.19 ± 0.12 | 0.68 ± 0.11 | 0.72 ± 0.07 | 1.03 ± 0.09 |
| 1/8 | 0.26 ± 0.10 | 0.56 ± 0.08 | 0.77 ± 0.08 | 1.00 ± 0.01 j | 1.44 ± 0.14 i | |
| 1/4 | 0.54 ± 0.18 | 0.79 ± 0.01 | 1.08 ± 0.05 j | 1.11 ± 0.01 j | 1.92 ± 0.02 g | |
| 2/4 | 1.15 ± 0.11 j | 1.97 ± 0.05 g | 2.38 ± 0.04 f | 2.75 ± 0.21 e | 3.07 ± 0.14 d | |
| 3/4 | 1.71 ± 0.13 h | 3.01 ± 0.19 d | 3.38 ± 0.15 c | 3.90 ± 0.05 b | 4.29 ± 0.19 a | |
| Microorganisms | Flavourzyme Concentration (MIC) | FE-DBD Plasma Treatment (min) | |||
|---|---|---|---|---|---|
| 5 | 10 | 30 | 60 | ||
| E. coli | 1/8 | 0.11 ± 0.03 | 0.02 ± 0.08 | 0.02 ± 0.04 | −0.59 ± 0.06 |
| 1/4 | 0.06 ± 0.13 | 0.05 ± 0.18 | −0.15 ± 0.17 | 0.35 ± 0.14 | |
| 2/4 | 0.63 ± 0.10 | 0.74 ± 0.12 | 0.88 ± 0.03 | 0.89 ± 0.14 | |
| 3/4 | 1.11 ± 0.11 b | 1.18 ± 0.20 b | 1.47 ± 0.03 a | 1.55 ± 0.03 a | |
| Flavourzyme Concentration (MIC) | FE-DBD Plasma Treatment (min) | |||||
|---|---|---|---|---|---|---|
| 0 | 5 | 10 | 30 | 60 | ||
| pH | 3/4 | 6.42 ± 0.19 NS | 6.42 ± 0.05 | 6.38 ± 0.03 | 6.36 ± 0.02 | 6.36 ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.H.; Roy, P.K.; Park, S.Y. Synergistic Effects of Combined Flavourzyme and Floating Electrode–Dielectric Barrier Discharge Plasma on Reduction of Escherichia coli Biofilms in Squid (Todarodes pacificus). Microorganisms 2024, 12, 1188. https://doi.org/10.3390/microorganisms12061188
Kim SH, Roy PK, Park SY. Synergistic Effects of Combined Flavourzyme and Floating Electrode–Dielectric Barrier Discharge Plasma on Reduction of Escherichia coli Biofilms in Squid (Todarodes pacificus). Microorganisms. 2024; 12(6):1188. https://doi.org/10.3390/microorganisms12061188
Chicago/Turabian StyleKim, So Hee, Pantu Kumar Roy, and Shin Young Park. 2024. "Synergistic Effects of Combined Flavourzyme and Floating Electrode–Dielectric Barrier Discharge Plasma on Reduction of Escherichia coli Biofilms in Squid (Todarodes pacificus)" Microorganisms 12, no. 6: 1188. https://doi.org/10.3390/microorganisms12061188
APA StyleKim, S. H., Roy, P. K., & Park, S. Y. (2024). Synergistic Effects of Combined Flavourzyme and Floating Electrode–Dielectric Barrier Discharge Plasma on Reduction of Escherichia coli Biofilms in Squid (Todarodes pacificus). Microorganisms, 12(6), 1188. https://doi.org/10.3390/microorganisms12061188






