Soil Microbes and Plant-Associated Microbes in Response to Radioactive Pollution May Indirectly Affect Plants and Insect Herbivores: Evidence for Indirect Field Effects from Chernobyl and Fukushima
Abstract
1. Introduction
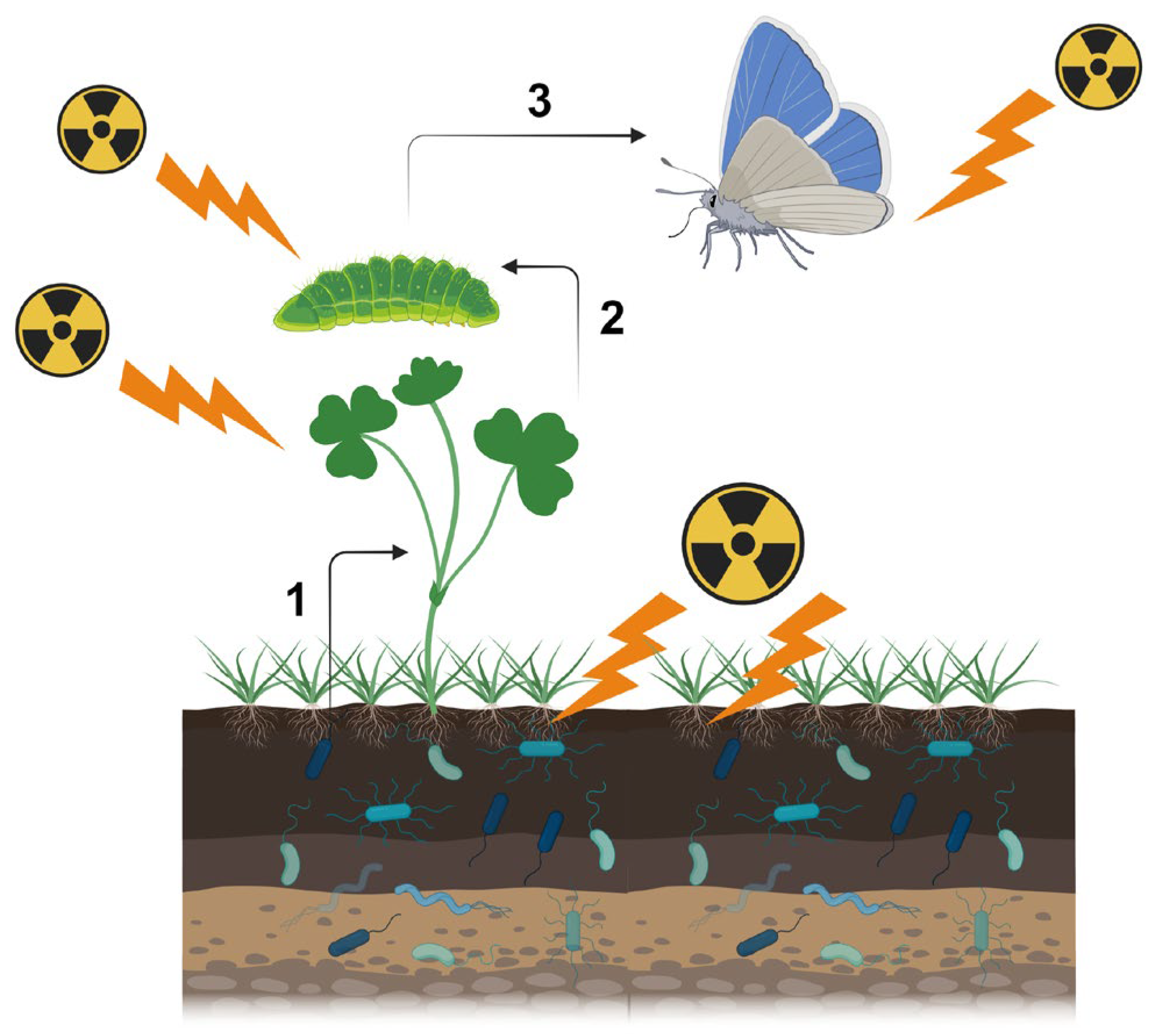
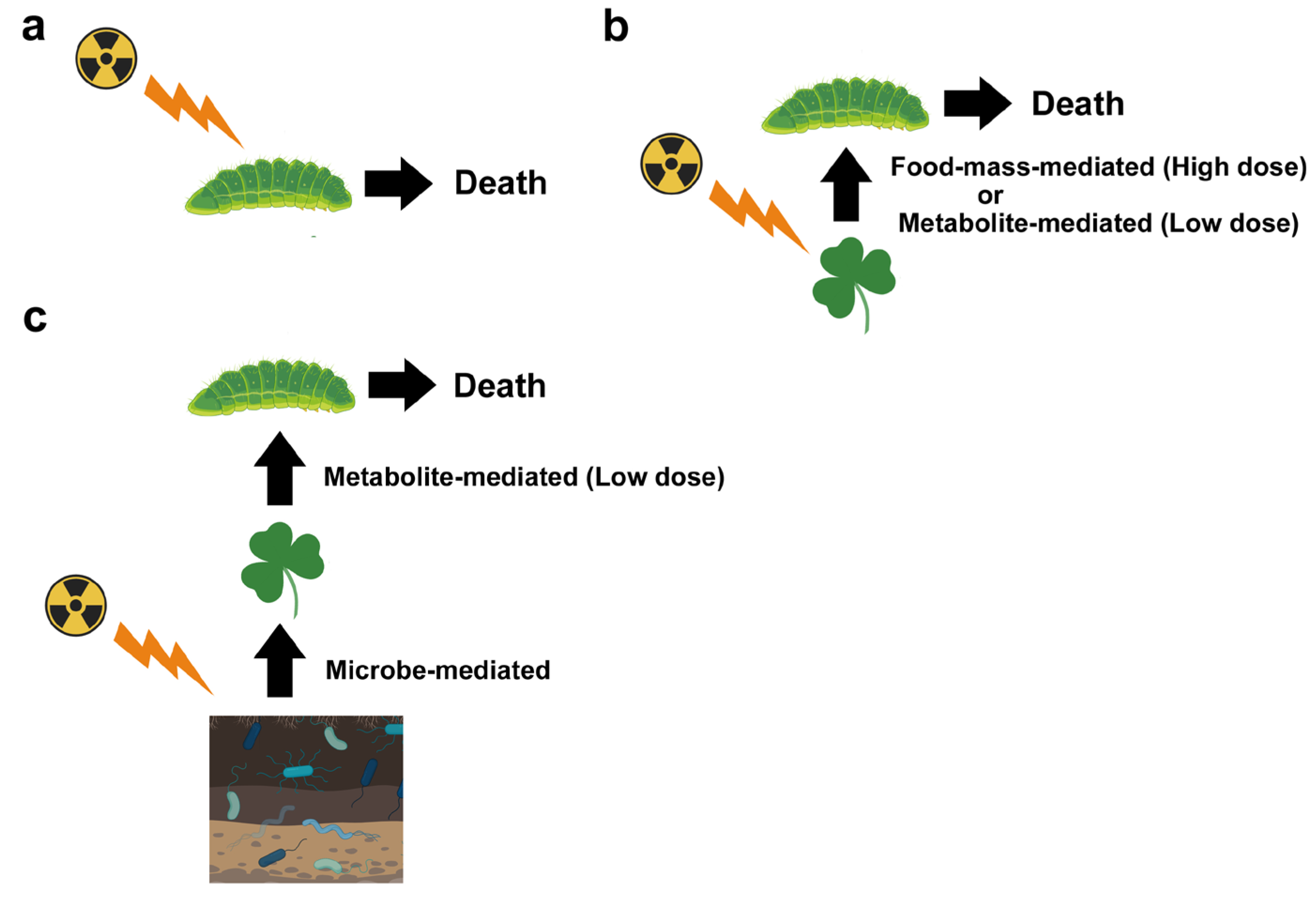
2. Multiple Pathways for Biological Effects
3. Soil Microbes and Soil Invertebrates
3.1. Chernobyl Studies
3.2. Fukushima Studies
3.3. Commonalities between Chernobyl and Fukushima
4. Plant-Associated Microbes
4.1. Chernobyl Studies
4.2. Fukushima Studies
4.3. Commonalities between Chernobyl and Fukushima
5. Plants and Insect Herbivores
5.1. Food-Mass-Mediated Indirect Effects
5.2. Pollen-Mediated Indirect Effects
5.3. Metabolite-Mediated Indirect Effect
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wagner, H.N., Jr. Atoms for peace (and health). J. Nucl. Med. 2004, 45, 24N. [Google Scholar] [PubMed]
- Babic, R.R.; Babic, G.S.; Babic, S.R.; Babic, N.R. 120 years since the discovery of x-rays. Med. Pregl. 2016, 69, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Nüsslin, F. Wilhelm Conrad Röntgen: The scientist and his discovery. Phys. Med. 2020, 79, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, A.; Kathren, R.L.; Willis, C.A. History of the medical uses of radiation: Regulatory and voluntary standards of protection. Health Phys. 1995, 69, 783–823. [Google Scholar] [CrossRef] [PubMed]
- Reed, A.B. History of radiation use in medicine. J. Vasc. Surg. 2011, 53 (Suppl. S1), 3S–5S. [Google Scholar] [CrossRef] [PubMed]
- Scatliff, J.H.; Morris, P.J. From Röntgen to magnetic resonance imaging: The history of medical imaging. N. C. Med. J. 2014, 75, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Kong, F.; Sun, K.; Wang, T.; Guo, T. From classical radiation to modern radiation: Past, present, and future of radiation mutation breeding. Front. Public Health 2021, 9, 768071. [Google Scholar] [CrossRef]
- Riviello-Flores, M.d.l.L.; Cadena-Iñiguez, J.; Ruiz-Posadas, L.d.M.; Arévalo-Galarza, M.d.L.; Castillo-Juárez, I.; Hernández, M.S.; Castillo-Martínez, C.R. Use of gamma radiation for the genetic improvement of underutilized plant varieties. Plants 2022, 11, 1161. [Google Scholar] [CrossRef]
- Whitmore, G.F. One hundred years of X rays in biological research. Radiat. Res. 1995, 144, 148–159. [Google Scholar] [CrossRef]
- D’Mello, J.P.F. Preface. In A Handbook of Environmental Toxicology: Human Disorders and Ecotoxicology; D’Mello, J.P.F., Ed.; CAB International: Wallingford, UK, 2020; pp. xxv–xxxvi. [Google Scholar]
- Geras’kin, S.A.; Fesenko, S.V.; Alexakhin, R.M. Effects of non-human species irradiation after the Chernobyl NPP accident. Environ. Int. 2008, 34, 880–897. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Kanda, J. Geochemistry. Tracking the Fukushima radionuclides. Science 2012, 336, 1115–1116. [Google Scholar] [CrossRef]
- International Atomic Energy Agency (IAEA). Environmental Consequences of the Chernobyl Accident and their Remediation: Twenty Years of Experience; Radiological Assessment Reports Series No. 8; IAEA: Vienna, Austria, 2006. [Google Scholar]
- Aliyu, A.S.; Evangeliou, N.; Mousseau, T.A.; Wu, J.; Ramli, A.T. An overview of current knowledge concerning the health and environmental consequences of the Fukushima Daiichi Nuclear Power Plant (FDNPP) accident. Environ. Int. 2015, 85, 213–228. [Google Scholar] [CrossRef]
- Ministry of the Environment (Government of Japan). Chapter 4. Concept of radiological protection. In BOOKLET to Provide Basic Information Regarding Health Effects of Radiation, 3rd ed.; Ministry of the Environment (Government of Japan): Tokyo, Japan, 2016; Available online: https://www.env.go.jp/en/chemi/rhm/basic-info/1st/04.html (accessed on 13 January 2024).
- International Atomic Energy Agency (IAEA). Effects of Ionizing Radiation on Plants and Animals at Levels Implied by Current Radiation Protection Standards; Technical Reports Series No. 332; IAEA: Vienna, Austria, 1992. [Google Scholar]
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). Sources and Effects of Ionizing Radiation UNSCEAR 1996 Report to the General Assembly, with Scientific Annex; United Nations: New York, NY, USA, 1996. [Google Scholar]
- European Commission. Protection of the Environment; European Union: Luxembourg, 2014; Available online: https://energy.ec.europa.eu/system/files/2014-11/177_0.pdf (accessed on 13 January 2024).
- Møller, A.P.; Mousseau, T.A. Biological consequences of Chernobyl: 20 years on. Trends Ecol. Evol. 2006, 21, 200–207. [Google Scholar] [CrossRef]
- Cannon, G.; Kiang, J.G. A review of the impact on the ecosystem after ionizing irradiation: Wildlife population. Int. J. Radiat. Biol. 2022, 98, 1054–1062. [Google Scholar] [CrossRef]
- Hiyama, A.; Nohara, C.; Taira, W.; Kinjo, S.; Iwata, M.; Otaki, J.M. The Fukushima nuclear accident and the pale grass blue butterfly: Evaluating biological effects of long-term low-dose exposures. BMC Evol. Biol. 2013, 13, 168. [Google Scholar] [CrossRef]
- Garnier-Laplace, J.; Geras’kin, S.; Della-Vedova, C.; Beaugelin-Seiller, K.; Hinton, T.G.; Real, A.; Oudalova, A. Are radiosensitivity data derived from natural field conditions consistent with data from controlled exposures? A case study of Chernobyl wildlife chronically exposed to low dose rates. J. Environ. Radioact. 2013, 121, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Otaki, J.M. Understanding low-dose exposure and field effects to resolve the field-laboratory paradox: Multifaceted biological effects from the Fukushima nuclear accident. In New Trends in Nuclear Science; Awwad, N.S., AlFaify, S.A., Eds.; InTechOpen: London, UK, 2018; pp. 49–71. [Google Scholar] [CrossRef]
- Otaki, J.M.; Sakauchi, K.; Taira, W. The second decade of the blue butterfly in Fukushima: Untangling the ecological field effects after the Fukushima nuclear accident. Integr. Environ. Assess. Manag. 2022, 18, 1539–1550. [Google Scholar] [CrossRef] [PubMed]
- Beaugelin-Seiller, K.; Della-Vedova, C.; Garnier-Laplace, J. Is non-human species radiosensitivity in the lab a good indicator of that in the field? Making the comparison more robust. J. Environ. Radioact. 2020, 211, 105870. [Google Scholar] [CrossRef] [PubMed]
- Hiyama, A.; Nohara, C.; Kinjo, S.; Taira, W.; Gima, S.; Tanahara, A.; Otaki, J.M. The biological impacts of the Fukushima nuclear accident on the pale grass blue butterfly. Sci. Rep. 2012, 2, 570. [Google Scholar] [CrossRef] [PubMed]
- Nohara, C.; Hiyama, A.; Taira, W.; Tanahara, A.; Otaki, J.M. The biological impacts of ingested radioactive materials on the pale grass blue butterfly. Sci. Rep. 2014, 4, 4946. [Google Scholar] [CrossRef] [PubMed]
- Gurung, R.D.; Taira, W.; Sakauchi, K.; Iwata, M.; Hiyama, A.; Otaki, J.M. Tolerance of high oral doses of nonradioactive and radioactive caesium chloride in the pale grass blue butterfly Zizeeria maha. Insects 2019, 10, 290. [Google Scholar] [CrossRef]
- Hancock, S.; Vo, N.T.K.; Omar-Nazir, L.; Batlle, J.V.I.; Otaki, J.M.; Hiyama, A.; Byun, S.H.; Seymour, C.B.; Mothersill, C. Transgenerational effects of historic radiation dose in pale grass blue butterflies around Fukushima following the Fukushima Dai-ichi Nuclear Power Plant meltdown accident. Environ. Res. 2019, 168, 230–240. [Google Scholar] [CrossRef]
- Sakauchi, K.; Taira, W.; Hiyama, A.; Imanaka, T.; Otaki, J.M. The pale grass blue butterfly in ex-evacuation zones 5.5 years after the Fukushima nuclear accident: Contributions of initial high-dose exposure to transgenerational effects. J. Asia-Pac. Entomol. 2020, 23, 242–252. [Google Scholar] [CrossRef]
- Nohara, C.; Taira, W.; Hiyama, A.; Tanahara, A.; Takatsuji, T.; Otaki, J.M. Ingestion of radioactively contaminated diets for two generations in the pale grass blue butterfly. BMC Evol. Biol. 2014, 14, 193. [Google Scholar] [CrossRef]
- Taira, W.; Hiyama, A.; Nohara, C.; Sakauchi, K.; Otaki, J.M. Ingestional and transgenerational effects of the Fukushima nuclear accident on the pale grass blue butterfly. J. Radiat. Res. 2015, 56 (Suppl. S1), i2–i18. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Kinouchi, T.; Fujii, T.; Imanaka, T.; Takahashi, T.; Fukutani, S.; Maki, D.; Nohtomi, A.; Takahashi, S. Observation of morphological abnormalities in silkworm pupae after feeding 137CsCl-supplemented diet to evaluate the effects of low dose-rate exposure. Sci. Rep. 2020, 10, 16055. [Google Scholar] [CrossRef] [PubMed]
- Hesse-Honegger, C.; Wallimann, P. Malformation of true bug (Heteroptera): A phenotype field study on the possible influence of artificial low-level radioactivity. Chem. Biodivers. 2008, 5, 499–539. [Google Scholar] [CrossRef] [PubMed]
- Körblein, A.; Hesse-Honegger, C. Morphological abnormalities in true bugs (Heteroptera) near Swiss nuclear power stations. Chem. Biodivers. 2018, 15, e1800099. [Google Scholar] [CrossRef] [PubMed]
- Akimoto, S. Morphological abnormalities in gall-forming aphids in a radiation-contaminated area near Fukushima Daiichi: Selective impact of fallout? Ecol. Evol. 2014, 4, 355–369. [Google Scholar] [CrossRef]
- Akimoto, S.I.; Li, Y.; Imanaka, T.; Sato, H.; Ishida, K. Effects of radiation from contaminated soil and moss in Fukushima on embryogenesis and egg hatching of the aphid Prociphilus oriens. J. Hered. 2018, 109, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Beresford, N.A.; Scott, E.M.; Copplestone, D. Field effects studies in the Chernobyl Exclusion Zone: Lessons to be learnt. J. Environ. Radioact. 2020, 211, 105893. [Google Scholar] [CrossRef]
- Woodwell, G.M.; Sparrow, A.H. Predicted and observed effects of chronic gamma radiation on a near-climax forest ecosystem. Radiat. Botany 1963, 3, 231–237. [Google Scholar] [CrossRef]
- Caffrey, E.; Leonard, M.; Napier, J.; Neville, D.; Higley, K. Radioecology: Why Bother? J. Environ. Prot. 2014, 5, 181–192. [Google Scholar] [CrossRef]
- Bradshaw, C.; Kapustka, L.; Barnthouse, L.; Brown, J.; Ciffroy, P.; Forbes, V.; Geras’kin, S.; Kautsky, U.; Bréchignac., F. Using an Ecosystem Approach to complement protection schemes based on organism-level endpoints. J. Environ. Radioact. 2014, 136, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Brèchignac, F.; Doi, M. Challenging the current strategy of radiological protection of the environment: Arguments for an ecosystem approach. J. Environ. Radioact. 2009, 100, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Woodwell, G.M. Ecological Effects of Nuclear War; Brookhaven National Laboratory: Upton, NY, USA, 1963. [Google Scholar]
- Bréchignac, F.; Alexakhin, R.; Godoy, J.M.; Oughton, D.; Sheppard, S.; Strand, P. Integrating environment protection, a new challenge: Strategy of the International Union of Radioecology. Radioprotection 2008, 43, 339–356. [Google Scholar] [CrossRef]
- Dalkvist, T.; Topping, C.J.; Forbes, V.E. Population-level impacts of pesticide-induced chronic effects on individuals depend more on ecology than toxicology. Ecotoxicol. Environ. Saf. 2009, 72, 1663–1672. [Google Scholar] [CrossRef] [PubMed]
- Clements, W.H.; Rohr, J.R. Community responses to contaminants: Using basic ecological principles to predict ecotoxicological effects. Environ. Toxicol. Chem. 2009, 28, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Mothersill, C.; Seymour, C. Communication of ionising radiation signals—A tale of two fish. Int. J. Radiat. Biol. 2009, 85, 909–919. [Google Scholar] [CrossRef]
- Otaki, J.M. Fukushima’s lessons from the blue butterfly: A risk assessment of the human living environment in the post-Fukushima era. Integr. Environ. Assess. Manag. 2016, 12, 667–672. [Google Scholar] [CrossRef]
- Otaki, J.M. Fukushima Nuclear Accident: Potential health effects inferred from butterfly and human cases. In A Handbook of Environmental Toxicology: Human Disorders and Ecotoxicology; D’Mello, J.P.F., Ed.; CAB International: Wallingford, UK, 2019; pp. 497–514. [Google Scholar]
- Brisbin, I.L., Jr. Avian radioecology. In Current Ornithology, Volume 8; Power, D.M., Ed.; Springer: New York, NY, USA, 1991; pp. 69–140. [Google Scholar]
- Matsui, S. Radiation ecology: Dynamics of radioactive materials in ecosystems and effects on birds. Anim. Husb. 2013, 67, 11–16. (In Japanese) [Google Scholar]
- Jonsson, B.; Forseth, T.; Ugedal, O. Chernobyl radioactivity persists in fish. Nature 1999, 400, 417. [Google Scholar] [CrossRef]
- Otaki, J.M.; Taira, W. Current status of the blue butterfly in Fukushima research. J. Hered. 2018, 109, 178–187. [Google Scholar] [CrossRef]
- Møller, A.P.; Nishiumi, I.; Suzuki, H.; Ueda, K.; Mousseau, T.A. Differences in effects of radiation on abundance of animals in Fukushima and Chernobyl. Ecol. Indic. 2013, 24, 75–81. [Google Scholar] [CrossRef]
- Møller, A.P.; Mousseau, T.A. Reduced abundance of insects and spiders linked to radiation at Chernobyl 20 years after the accident. Biol. Lett. 2009, 5, 356–359. [Google Scholar] [CrossRef] [PubMed]
- Møller, A.P.; Mousseau, T.A. Efficiency of bio-indicators for low-level radiation under field conditions. Ecol. Indic. 2011, 11, 424–430. [Google Scholar] [CrossRef]
- Turner, F.B. Effects of continuous irradiation on animal populations. Adv. Radiat. Biol. 1975, 5, 83–144. [Google Scholar] [CrossRef]
- Lecomte-Pradines, C.; Bonzom, J.M.; Della-Vedova, C.; Beaugelin-Seiller, K.; Villenave, C.; Gaschak, S.; Coppin, F.; Dubourg, N.; Maksimenko, A.; Adam-Guillermin, C.; et al. Soil nematode assemblages as bioindicators of radiation impact in the Chernobyl Exclusion Zone. Sci. Total Environ. 2014, 490, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Møller, A.P.; Mousseau, T.A. Species richness and abundance of forest birds in relation to radiation at Chernobyl. Biol. Lett. 2007, 3, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Zhdanova, N.N.; Vasilevskaia, A.I.; Artyshkova, L.V.; Gavriliuk, V.I.; Lashko, T.N.; Sadovnikov, I.S. Kompleksy pochvennykh mikrokitsetov v zone vliianiia Chernobyl’skoĭ AES [Complexes of soil micromycetes in the area of the influence of the Chernobyl Atomic Electric Power Station]. Mikrobiol. Zh. 1991, 53, 3–9. (In Russian) [Google Scholar]
- Yablokov, A.V.; Nesterenko, V.B.; Nesterenko, A.V. Consequences of the Chernobyl catastrophe for public health and the environment 23 years later. Ann. N. Y. Acad. Sci. 2009, 1181, 318–326. [Google Scholar] [CrossRef]
- Cordero, R.J.; Casadevall, A. Functions of fungal melanin beyond virulence. Fungal Biol. Rev. 2017, 31, 99–112. [Google Scholar] [CrossRef]
- Dadachova, E.; Bryan, R.A.; Huang, X.; Moadel, T.; Schweitzer, A.D.; Aisen, P.; Nosanchuk, J.D.; Casadevall, A. Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi. PLoS ONE 2007, 2, e457. [Google Scholar] [CrossRef]
- Dadachova, E.; Casadevall, A. Ionizing radiation: How fungi cope, adapt, and exploit with the help of melanin. Curr. Opin. Microbiol. 2008, 11, 525–531. [Google Scholar] [CrossRef]
- Romanovskaia, V.A.; Stoliar, S.M.; Malashenko, I.R.; Shatokhina, E.S. Vliianie radiatsii dlitel’nogo deĭstviia na raznoobrazie geterotrofnykh bakteriĭ v pochvakh 10-kilometrovoĭ zony Chernobyl’skoĭ atomnoĭ élektrostantsii [The effect of long-acting radiation on the diversity of heterotrophic bacteria in the soils of a 10-kilometer area around the Chernobyl Atomic Electric Power Station]. Mikrobiol. Z. 1996, 58, 3–12. (In Russian) [Google Scholar]
- Romanovskaia, V.A.; Sokolov, I.G.; Rokitko, P.V.; Chernaia, N.A. Ekologicheskie posledstviia radioaktivnogo zagriazneniia dlia pochvennykh bakteriĭ v 10-km zone ChAES [Ecological consequences of radioactive pollution for soil bacteria within the 10-km region around the Chernobyl Atomic Energy Station]. Mikrobiologiia 1998, 67, 274–280. (In Russian) [Google Scholar]
- Krivolutzkii, D.A.; Pokarzhevskii, A.D. Effects of radioactive fallout on soil animal populations in the 30 km zone of the Chernobyl atomic power station. Sci. Total Environ. 1992, 112, 69–77. [Google Scholar] [CrossRef]
- Suzuki, J.; Egami, N. Mortality of the earthworms, Eisenia foetida, after γ-irradiation at different stages of their life history. J. Radiat. Res. 1983, 24, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Hertel-Aas, T.; Oughton, D.H.; Jaworska, A.; Bjerke, H.; Salbu, B.; Brunborg, G. Effects of chronic gamma irradiation on reproduction in the earthworm Eisenia fetida (Oligochaeta). Radiat. Res. 2007, 168, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Handelsman, J. Metagenomics: Application of genomics to uncultured microorganisms. Microbiol. Mol. Biol. Rev. 2004, 68, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Volkova, P.Y.; Geras’kin, S.A. ‘Omic’ technologies as a helpful tool in radioecological research. J. Environ. Radioact. 2018, 189, 156–167. [Google Scholar] [CrossRef]
- Beresford, N.A.; Wood, M.D.; Gashchak, S.; Barnett, C.L. Current ionising radiation doses in the Chernobyl Exclusion Zone do not directly impact on soil biological activity. PLoS ONE 2022, 17, e0263600. [Google Scholar] [CrossRef]
- Mousseau, T.A.; Milinevsky, G.; Kenney-Hunt, J.; Møller, A.P. Highly reduced mass loss rates and increased litter layer in radioactively contaminated areas. Oecologia 2014, 175, 429–437. [Google Scholar] [CrossRef]
- Bonzom, J.M.; Hättenschwiler, S.; Lecomte-Pradines, C.; Chauvet, E.; Gaschak, S.; Beaugelin-Seiller, K.; Della-Vedova, C.; Dubourg, N.; Maksimenko, A.; Garnier-Laplace, J.; et al. Effects of radionuclide contamination on leaf litter decomposition in the Chernobyl exclusion zone. Sci. Total Environ. 2016, 562, 596–603. [Google Scholar] [CrossRef]
- Videvall, E.; Burraco, P.; Orizaola, G. Impact of ionizing radiation on the environmental microbiomes of Chornobyl wetlands. Environ. Pollut. 2023, 330, 121774. [Google Scholar] [CrossRef] [PubMed]
- Chapon, V.; Piette, L.; Vesvres, M.H.; Coppin, F.; Marrec, C.L.; Christen, R.; Theodorakopoulos, N.; Février, L.; Levchuk, S.; Martin-Garin, A.; et al. Microbial diversity in contaminated soils along the T22 trench of the Chernobyl experimental platform. Appl. Geochem. 2012, 27, 1375–1383. [Google Scholar] [CrossRef]
- Bugai, D.; Kashparov, V.; Dewiére, L.; Khomutinin, Y.; Levchuk, S.; Yoschenko, V. Characterization of subsurface geometry and radioactivity distribution in the trench containing Chernobyl clean-up wastes. Environ. Geol. 2005, 47, 869–881. [Google Scholar] [CrossRef]
- Ruban, Y.V.; Shavanova, K.E.; Samofalova, D.O.; Nikonov, S.B.; Pareniuk, O.Y. Comparison of microbiomes of two different ecotypes of the Chernobyl exclusion zone: Points of temporary localization of radioactive waste (PTLRW) and contaminated ecosystems. Hyg. Ecol. 2020, 3, 83–88. [Google Scholar] [CrossRef]
- Shuryak, I.; Dadachova, E. Quantitative modeling of microbial population responses to chronic irradiation combined with other stressors. PLoS ONE 2016, 11, e0147696. [Google Scholar] [CrossRef] [PubMed]
- Zhdanova, N.N.; Tugay, T.; Dighton, J.; Zheltonozhsky, V.; McDermott, P. Ionizing radiation attracts soil fungi. Mycol. Res. 2004, 108, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Karpenko, Y.V.; Redchitz, T.I.; Zheltonozhsky, V.A.; Dighton, J.; Zhdanova, N.N. Comparative responses of microscopic fungi to ionizing radiation and light. Folia Microbiol. 2006, 51, 45–49. [Google Scholar] [CrossRef]
- Zavilgelsky, G.B.; Abilev, S.K.; Sukhodolets, V.V.; Ahmad, S.I. Isolation and analysis of UV and radio-resistant bacteria from Chernobyl. J. Photochem. Photobiol. B 1998, 43, 152–157. [Google Scholar] [CrossRef]
- Narumi, I. Unlocking radiation resistance mechanisms: Still a long way to go. Trends Microbiol. 2003, 11, 422–425. [Google Scholar] [CrossRef]
- Zhdanova, N.N.; Zakharchenko, V.A.; Vember, V.V.; Nakonechnaya, L.T. Fungi from Chernobyl: Mycobiota of the inner regions of the containment structures of the damaged nuclear reactor. Mycol. Res. 2000, 104, 1421–1426. [Google Scholar] [CrossRef]
- Averesch, N.J.H.; Shunk, G.K.; Kern, C. Cultivation of the dematiaceous fungus Cladosporium sphaerospermum aboard the International Space Station and effects of ionizing radiation. Front. Microbiol. 2022, 13, 877625. [Google Scholar] [CrossRef]
- Byrne, A.R. Radioactivity in fungi in Slovenia, Yugoslavia, following the Chernobyl accident. J. Environ. Radioact. 1988, 6, 177–183. [Google Scholar] [CrossRef]
- Haselwandter, K.; Berreck, M.; Brunner, P. Fungi as bioindicators of radiocaesium contamination: Pre- and post-Chernobyl activities. Trans. British Mycol. Soc. 1988, 90, 171–174. [Google Scholar] [CrossRef]
- Steine, M.; Linkov, I.; Yoshida, S. The role of fungi in the transfer and cycling of radionuclides in forest ecosystems. J. Environ. Radioact. 2002, 58, 217–241. [Google Scholar] [CrossRef] [PubMed]
- Fukuyama, T.; Takenaka, C. Upward mobilization of 137Cs in surface soils of Chamaecyparis obtusa Sieb. et Zucc. (hinoki) plantation in Japan. Sci. Total Environ. 2004, 318, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Rafferty, B.; Dawson, D.E.; Kliashtorin, A.L. Decomposition in two pine forests: The mobilisation of 137Cs and K from forest litter. Soil Biol. Biochem. 1997, 29, 1673–1681. [Google Scholar] [CrossRef]
- Wada, T. Radiocesium contamination of marine and freshwater fish after the Fukushima Dai-ichi Nuclear Power Plant accident. Geochemistry 2021, 55, 159–175. [Google Scholar] [CrossRef]
- Ihara, H.; Kumagai, A.; Hori, T.; Nanba, K.; Aoyagi, T.; Takasaki, M.; Katayama, Y. Direct comparison of bacterial communities in soils contaminated with different levels of radioactive cesium from the first Fukushima nuclear power plant accident. Sci. Total Environ. 2021, 756, 143844. [Google Scholar] [CrossRef]
- Higo, M.; Kang, D.J.; Isobe, K. First report of community dynamics of arbuscular mycorrhizal fungi in radiocesium degradation lands after the Fukushima-Daiichi Nuclear disaster in Japan. Sci. Rep. 2019, 9, 8240. [Google Scholar] [CrossRef] [PubMed]
- Bever, J.D.; Richardson, S.C.; Lawrence, B.M.; Holmes, J.; Watson, M. Preferential allocation to beneficial symbiont with spatial structure maintains mycorrhizal mutualism. Ecol. Lett. 2009, 12, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Barto, E.K.; Hilker, M.; Müller, F.; Mohney, B.K.; Weidenhamer, J.D.; Rillig, M.C. The fungal fast lane: Common mycorrhizal networks extend bioactive zones of allelochemicals in soils. PLoS ONE 2011, 6, e27195. [Google Scholar] [CrossRef]
- Huang, Y.; Kaneko, Y.; Nakamori, T.; Miura, T.; Tanaka, Y.; Nonaka, M.; Takenaka, C. Radiocesium immobilization to leaf litter by fungi during first-year decomposition in a deciduous forest in Fukushima. J. Environ. Radioact. 2016, 152, 28–34. [Google Scholar] [CrossRef]
- Garnier-Laplace, J.; Beaugelin-Seiller, K.; Hinton, T.G. Fukushima wildlife dose reconstruction signals ecological consequences. Environ. Sci. Technol. 2011, 45, 5077–5078. [Google Scholar] [CrossRef]
- Saito, M.; Yamada, A.; Matsuda, Y.; Yamato, M. Absorption and accumulation of radioactive cesium by fungi. Chem. Biol. 2013, 5, 748–751. (In Japanese) [Google Scholar]
- Yamada, T.; Murakawa, I.; Saito, T.; Omura, K.; Takatoku, K.; Saiki, M.; Iguchi, K.; Inoue, M.; Saito, H.; Tsuji, K.; et al. Radiocaesium accumulation in wild mushrooms from low-level contaminated area due to the Fukushima Daiichi Nuclear Power Plant Accident-A case study in the University of Tokyo forests. Radioisotopes 2013, 62, 141–147. [Google Scholar] [CrossRef][Green Version]
- Arrange, A.A.; Phelps, T.J.; Benoit, R.E.; Palumbo, A.V.; White, D.C. Bacterial sensitivity to UV light as a model for ionizing radiation resistance. J. Microbiol. Methods 1993, 18, 127–136. [Google Scholar] [CrossRef]
- Kawaguchi, Y.; Shibuya, M.; Kinoshita, I.; Yatabe, J.; Narumi, I.; Shibata, H.; Hayashi, R.; Fujiwara, D.; Murano, Y.; Hashimoto, H.; et al. DNA damage and survival time course of deinococcal cell pellets during 3 years of exposure to outer space. Front. Microbiol. 2020, 11, 2050. [Google Scholar] [CrossRef] [PubMed]
- Musilova, M.; Wright, G.; Ward, J.M.; Dartnell, L.R. Isolation of radiation-resistant bacteria from Mars analog Antarctic Dry Valleys by preselection, and the correlation between radiation and desiccation resistance. Astrobiology 2015, 15, 1076–1090. [Google Scholar] [CrossRef] [PubMed]
- Barescut, J.; Lariviere, D.; Stocki, T.; Alexakhin, R.; Geras’kin, S. 25 years after the accident at the Chernobyl nuclear power plant: Radioecological lessons. Radioprotection 2011, 46, S595–S600. [Google Scholar] [CrossRef]
- Cheng, F.; Huang, X.; Qin, Q.; Chen, Z.; Li, F.; Song, W. The effect of aboveground long-term low-dose ionizing radiation on soil microbial diversity and structure. Front. Ecol. Evol. 2023, 11, 1184582. [Google Scholar] [CrossRef]
- Geras’kin, S.A.; Dikarev, V.G.; Zyablitskaya, Y.Y.; Oudalova, A.A.; Spirin, Y.V. Cytogenetic effects of radiation on agricultural plants observed in the Chernobyl region during the first years after the accident. KURRI-KR 2002, 79, 287–296. [Google Scholar]
- Boratyński, Z.; Arias, J.; Garcia, C.; Mappes, T.; Mousseau, T.A.; Møller, A.P.; Pajares, A.J.M.; Piwczyński, M.; Tukalenko, E. Ionizing radiation from Chernobyl affects development of wild carrot plants. Sci. Rep. 2016, 6, 39282. [Google Scholar] [CrossRef] [PubMed]
- van der Heijden, M.G.; Bardgett, R.D.; van Straalen, N.M. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar] [CrossRef]
- Zhu, J.; Sun, X.; Zhang, Z.D.; Tang, Q.Y.; Gu, M.Y.; Zhang, L.J.; Hou, M.; Sharon, A.; Yuan, H.L. Effect of ionizing radiation on the bacterial and fungal endophytes of the halophytic plant Kalidium schrenkianum. Microorganisms 2021, 9, 1050. [Google Scholar] [CrossRef]
- Vurukonda, S.S.K.P.; Giovanardi, D.; Stefani, E. Plant growth promoting and biocontrol activity of Streptomyces spp. as endophytes. Int. J. Mol. Sci. 2018, 19, 952. [Google Scholar] [CrossRef]
- Parray, J.A.; Jan, S.; Kamili, A.N.; Qadri, R.A.; Egamberdieva, D.; Ahmad, P. Current perspectives on plant growth-promoting Rhizobacteria. J. Plant Growth Regul. 2016, 35, 877–902. [Google Scholar] [CrossRef]
- Dias, M.P.; Bastos, M.S.; Xavier, V.B.; Cassel, E.; Astarita, L.V.; Santarém, E.R. Plant growth and resistance promoted by Streptomyces spp. in tomato. Plant Physiol. Biochem. 2017, 118, 479–493. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Kim, Y.C.; Leveau, J.; McSpadden Gardener, B.B.; Pierson, E.A.; Pierson, L.S., III; Ryu, C.M. The multifactorial basis for plant health promotion by plant-associated bacteria. Appl. Environ. Microbiol. 2011, 77, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, A.; Guscha, N.; Dyachenko, A. Effects of low dose radiation on plant-pathogen interactions in Chernobyl Zone. Radioprotection 2008, 43, 148. [Google Scholar] [CrossRef]
- Hinton, T.G.; Alexakhin, R.; Balonov, M.; Gentner, N.; Hendry, J.; Prister, B.; Strand, P.; Woodhead, D. Radiation-induced effects on plants and animals: Findings of the United Nations Chernobyl Forum. Health Phys. 2007, 93, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Adam-Guillermin, C.; Hertal-Aas, T.; Oughton, D.; Blanchard, L.; Alonzo, F.; Armant, O.; Horemans, N. Radiosensitivity and transgenerational effects in non-human species. Ann. ICRP 2018, 47, 327–341. [Google Scholar] [CrossRef]
- Duarte, G.T.; Volkova, P.Y.; Perez, F.F.; Horemans, N. Chronic ionizing radiation of plants: An evolutionary factor from direct damage to non-target effects. Plants 2023, 12, 1178. [Google Scholar] [CrossRef] [PubMed]
- Volkova, P.Y.; Geras’kin, S.A.; Kazakova, E.A. Radiation exposure in the remote period after the Chernobyl accident caused oxidative stress and genetic effects in Scots pine populations. Sci. Rep. 2017, 7, 43009. [Google Scholar] [CrossRef]
- Volkova, P.Y.; Bondarenko, E.V.; Kazakova, E.A. Radiation hormesis in plants. Curr. Opin. Toxicol. 2022, 30, 100334. [Google Scholar] [CrossRef]
- Gudkov, I.N.; Gaychenko, V.A.; Pareniuk, O.Y.; Grodzinsky, D.M. Changes in biocenoses in the Chernobyl NPP accident zone. Nucl. Phys. Atomic Energy 2011, 12, 362–374. [Google Scholar]
- Branco, S.; Schauster, A.; Liao, H.L.; Ruytinx, J. Mechanisms of stress tolerance and their effects on the ecology and evolution of mycorrhizal fungi. New Phytol. 2022, 235, 2158–2175. [Google Scholar] [CrossRef] [PubMed]
- Kothamasi, D.; Wannijn, J.; van Hees, M.; Nauts, R.; van Gompel, A.; Vanhoudt, N.; Vandenhove, H. Exposure to ionizing radiation affects the growth of ectomycorrhizal fungi and induces increased melanin production and increased capacities of reactive oxygen species scavenging enzymes. J. Environ. Radioact. 2019, 197, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Dixit, S.K.; Senthil-Kumar, M. Drought stress predominantly endures Arabidopsis thaliana to Pseudomonas syringae infection. Front. Plant Sci. 2016, 7, 808. [Google Scholar] [CrossRef] [PubMed]
- Sakauchi, K.; Taira, W.; Otaki, J.M. Metabolomic profiles of the creeping wood sorrel Oxalis corniculata in radioactively contaminated fields in Fukushima: Dose-dependent changes in key metabolites. Life 2022, 12, 115. [Google Scholar] [CrossRef] [PubMed]
- Peng, A.; Liu, J.; Gao, Y.; Chen, Z. Distribution of endophytic bacteria in Alopecurus aequalis Sobol and Oxalis corniculata L. from soils contaminated by polycyclic aromatic hydrocarbons. PLoS ONE 2013, 8, e83054. [Google Scholar] [CrossRef] [PubMed]
- Mufti, R.; AmnaRafique, M.; Haq, F.; Munis, M.F.H.; Masood, S.; Mumtaz, A.S.; Chaudhary, H.J. Genetic diversity and metal resistance assessment of endophytes isolated from Oxalis corniculata. Soil Environ. 2015, 34, 89–99. [Google Scholar]
- Sakauchi, K.; Taira, W.; Otaki, J.M. Metabolomic response of the creeping wood sorrel Oxalis corniculata to low-dose radiation exposure from Fukushima’s contaminated soil. Life 2021, 11, 990. [Google Scholar] [CrossRef]
- Aung, H.P.; Djedidi, S.; Oo, A.Z.; Aye, Y.S.; Yokoyama, T.; Suzuki, S.; Sekimoto, H.; Bellingrath-Kimura, S.D. Growth and 137Cs uptake of four Brassica species influenced by inoculation with a plant growth-promoting rhizobacterium Bacillus pumilus in three contaminated farmlands in Fukushima prefecture, Japan. Sci. Total Environ. 2015, 521–522, 261–269. [Google Scholar] [CrossRef]
- Aqvist, J.; Luzhkov, V. Ion permeation mechanism of the potassium channel. Nature 2000, 404, 881–884. [Google Scholar] [CrossRef]
- Hayashi, G.; Shibato, J.; Imanaka, T.; Cho, K.; Kubo, A.; Kikuchi, S.; Satoh, K.; Kimura, S.; Ozawa, S.; Fukutani, S.; et al. Unraveling low-level gamma radiation-responsive changes in expression of early and late genes in leaves of rice seedlings at Iitate Village, Fukushima. J. Hered. 2014, 105, 723–738. [Google Scholar] [CrossRef]
- Rakwal, R.; Hayashi, G.; Shibato, J.; Deepak, S.A.; Gundimeda, S.; Simha, U.; Padmanaban, A.; Gupta, R.; Han, S.I.; Kim, S.T.; et al. Progress toward rice seed OMICS in low-level gamma radiation environment in Iitate Village, Fukushima. J. Hered. 2018, 109, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Ichikawa, S.; Kubota, M.; Hoshino, J.; Kubota, Y.; Maruyama, K.; Fuma, S.; Kawaguchi, I.; Yoschenko, V.I.; Yoshida, S. Morphological defects in native Japanese fir trees around the Fukushima Daiichi Nuclear Power Plant. Sci. Rep. 2015, 5, 13232. [Google Scholar] [CrossRef] [PubMed]
- Yoschenko, V.; Nanba, K.; Yoshida, S.; Watanabe, Y.; Takase, T.; Sato, N.; Keitoku, K. Morphological abnormalities in Japanese red pine (Pinus densiflora) at the territories contaminated as a result of the accident at Fukushima Dai-Ichi Nuclear Power Plant. J. Environ. Radioact. 2016, 165, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, M. Endophytic actinomycetes: Biocontrol agents and growth promoters. In Bacteria in Agrobiology: Pant Growth Responses; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 201–220. [Google Scholar] [CrossRef]
- Martino, E.; Perotto, S. Mineral Transformations by Mycorrhizal Fungi. Geomicrobiol. J. 2010, 27, 609–623. [Google Scholar] [CrossRef]
- Clasen, B.E.; Silveira, A.D.O.; Baldoni, D.B.; Montagner, D.F.; Jacques, R.J.S.; Antoniolli, Z.I. Characterization of Ectomycorrhizal species through molecular biology tools and morphotyping. Sci. Agric. 2018, 75, 246–254. [Google Scholar] [CrossRef]
- Li, F.; Deng, X.; Chen, H.; Lu, H.; Guo, W.; Song, W.; Ge, L. Long-term gamma radiation effect on functional traits of Tradescantia Flumnensis L. Pol. J. Environ. Stud. 2022, 31, 1153–1160. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; White, J.F., Jr.; Arnold, A.E.; Redman, R.S. Fungal endophytes: Diversity and functional roles. New Phytol. 2009, 182, 314–330. [Google Scholar] [CrossRef] [PubMed]
- Frank, A.C.; Guzmán, J.P.S.; Shay, J.E. Transmission of bacterial endophytes. Microorganisms 2017, 5, 70. [Google Scholar] [CrossRef]
- Lindroth, R.L.; Hofman, R.W.; Campbell, B.D.; McNabb, W.C.; Hunt, D.Y. Population differences in Trifolium repens L. response to ultraviolet-B radiation: Foliar chemistry and consequences for two lepidopteran herbivores. Oecologia 2000, 122, 20–28. [Google Scholar] [CrossRef]
- U’Ren, J.M.; Lutzoni, F.; Miadlikowska, J.; Laetsch, A.D.; Arnold, A.E. Host and geographic structure of endophytic and endolichenic fungi at a continental scale. Am. J. Bot. 2012, 99, 898–914. [Google Scholar] [CrossRef] [PubMed]
- Rillig, M.; Allen, M. What is the role of arbuscular mycorrhizal fungi in plant-to-ecosystem responses to elevated atmospheric CO2? Mycorrhiza 1999, 9, 1–8. [Google Scholar] [CrossRef]
- Sanders, I.R.; Streitwolf-Engel, R.; van der Heijden, M.G.; Boller, T.; Wiemken, A. Increased allocation to external hyphae of arbuscular mycorrhizal fungi under CO2 enrichment. Oecologia 1998, 117, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Michiels, K.; Vanderleyden, J.; Van Gool, A. Azospirillum—Plant root associations: A review. Biol. Fert. Soils 1989, 8, 356–368. [Google Scholar] [CrossRef]
- Taskaev, A.I.; Frolova, N.P.; Popova, O.N.; Shevchenko, V.A. The monitoring of herbaceous seeds in the 30-km zone of the Chernobyl nuclear accident. Sci. Total Environ. 1992, 112, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Suvorova, L.I.; Spirin, D.A.; Martyushov, V.Z.; Smirnov, E.G.; Tarasov, O.V.; Shein, G.P. Assessment of biological and ecological consequences of radioactive contamination of biogeocenoses. In Radiation Aspects of the Chernobyl Accident, Volume 2; Izrael, Y.A., Ed.; Hydrometeoizdat: St. Petersburg, Russia, 1993; pp. 321–325. (In Russian) [Google Scholar]
- Alexakhin, R.M.; Buldakov, L.A.; Gubanov, V.A.; Drozhko, Y.G.; Ilyin, L.A.; Kryshev, I.I.; Linge, I.I.; Romanov, G.N.; Savki, M.N. Large Radiation Accidents: Consequences and Protective Counter-Measures; IzdAT Publisher: Moscow, Russia, 2004. [Google Scholar]
- Grodzinsky, D.M.; Gudkov, I.N. Radiation injury of the plant in the zone of influence of the accident on Chernobyl Nuclear Power Plant. Radiat. Biol. Radioecol. 2006, 46, 189–199. (In Russian) [Google Scholar]
- Møller, A.P.; Shyu, J.C.; Mousseau, T.A. Ionizing radiation from Chernobyl and the fraction of viable pollen. Int. J. Plant Sci. 2016, 177, 727–735. [Google Scholar] [CrossRef]
- Kim, D.R.; Cho, G.; Jeon, C.W.; Weller, D.M.; Thomashow, L.S.; Paulitz, T.C.; Kwak, Y.S. A mutualistic interaction between Streptomyces bacteria, strawberry plants and pollinating bees. Nat. Commun. 2019, 10, 4802. [Google Scholar] [CrossRef]
- Liu, H.; Macdonald, C.A.; Cook, J.; Anderson, I.C.; Singh, B.K. An Ecological Loop: Host Microbiomes across Multitrophic Interactions. Trends Ecol. Evol. 2019, 34, 1118–1130. [Google Scholar] [CrossRef]
- Møller, A.P.; Barnier, F.; Mousseau, T.A. Ecosystems effects 25 years after Chernobyl: Pollinators, fruit set and recruitment. Oecologia 2012, 170, 1155–1165. [Google Scholar] [CrossRef] [PubMed]
- Imanaka, T. Chernobyl and Fukushima: Comparison of accident process and radioactive contamination. Kagaku 2016, 86, 0252–0257. (In Japanese) [Google Scholar]
- Ludovici, G.M.; Chierici, A.; de Souza, S.O.; d’Errico, F.; Iannotti, A.; Malizia, A. Effects of ionizing radiation on flora ten years after the Fukushima Dai-ichi disaster. Plants 2022, 11, 222. [Google Scholar] [CrossRef]
- Yoshioka, A.; Mishima, Y.; Fukasawa, K. Pollinators and other flying insects inside and outside the Fukushima evacuation zone. PLoS ONE 2015, 10, e0140957. [Google Scholar] [CrossRef]
- Hiyama, A.; Taira, W.; Nohara, C.; Iwasaki, M.; Kinjo, S.; Iwata, M.; Otaki, J.M. Spatiotemporal abnormality dynamics of the pale grass blue butterfly: Three years of monitoring (2011–2013) after the Fukushima nuclear accident. BMC Evol. Biol. 2015, 15, 15. [Google Scholar] [CrossRef]
- Møller, A.P.; Nishiumi, I.; Mousseau, T.A. Cumulative effects of radioactivity from Fukushima on the abundance and biodiversity of birds. J. Ornithol. 2015, 156, 297–305. [Google Scholar] [CrossRef]
- Møller, A.P.; Hagiwara, A.; Matsui, S.; Kasahara, S.; Kawatsu, K.; Nishiumi, I.; Suzuki, H.; Ueda, K.; Mousseau, T.A. Abundance of birds in Fukushima as judged from Chernobyl. Environ. Pollut. 2012, 164, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Bonisoli-Alquati, A.; Koyama, K.; Tedeschi, D.J.; Kitamura, W.; Sukuzi, H.; Ostermiller, S.; Arai, E.; Møller, A.P.; Mousseau, T.A. Abundance and genetic damage of barn swallows from Fukushima. Sci. Rep. 2015, 5, 9432. [Google Scholar] [CrossRef] [PubMed]
- Murase, K.; Murase, J.; Horie, R.; Endo, K. Effects of the Fukushima Daiichi nuclear accident on goshawk reproduction. Sci. Rep. 2015, 5, 9405. [Google Scholar] [CrossRef] [PubMed]
- Gange, A.C.; Eschen, R.; Wearn, J.A.; Thawer, A.; Sutton, B.C. Differential effects of foliar endophytic fungi on insect herbivores attacking a herbaceous plant. Oecologia 2012, 168, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, J.P.F. Phytotoxins. In A Handbook of Environmental Toxicology: Human Disorders and Ecotoxicology; D’Mello, J.P.F., Ed.; CAB International: Wallingford, UK, 2020; pp. 3–18. [Google Scholar]
- Zagrobelny, M.; de Castro, É.C.P.; Møller, B.L.; Bak, S. Cyanogenesis in arthropods: From chemical warfare to nuptial gifts. Insects 2018, 9, 51. [Google Scholar] [CrossRef] [PubMed]
- Clay, K. Fungal endophytes of grasses: A defensive mutualism between plants and fungi. Ecology 1988, 69, 10–16. [Google Scholar] [CrossRef]
- Morita, A.; Sakauchi, K.; Taira, W.; Otaki, J.M. Ingestional toxicity of radiation-dependent metabolites of the host plant for the pale grass blue butterfly: A mechanism of field effects of radioactive pollution in Fukushima. Life 2022, 12, 615. [Google Scholar] [CrossRef] [PubMed]
- Jomon, K.; Kuroda, Y.; Ajisaka, M.; Sakai, H. A new antibiotic, ikarugamycin. J. Antibiot. 1972, 25, 271–280. [Google Scholar] [CrossRef]
- Lacret, R.; Oves-Costales, D.; Gómez, C.; Díaz, C.; de la Cruz, M.; Pérez-Victoria, I.; Vicente, F.; Genilloud, O.; Reyes, F. New ikarugamycin derivatives with antifungal and antibacterial properties from Streptomyces zhaozhouensis. Mar. Drugs 2014, 13, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Saeed, S.I.; Aklilu, E.; Mohammedsalih, K.M.; Adekola, A.A.; Mergani, A.E.; Mohamad, M.; Kamaruzzaman, N.F. Antibacterial activity of ikarugamycin against intracellular Staphylococcus aureus in bovine mammary epithelial cells in vitro infection model. Biology 2021, 10, 958. [Google Scholar] [CrossRef]
- Krivolutskii, D.A. Problems of sustainable development and ecological indication in radioactively contaminated areas. Russ. J. Ecol. 2000, 31, 233–237. [Google Scholar] [CrossRef]
- Woodwell, G.M.; Brower, J.H. An aphid population explosion induced by chronic gamma irradiation of a forest. Ecology 1967, 48, 680–683. [Google Scholar] [CrossRef]
- Ochiai, K.; Hayama, S.; Nakiri, S.; Nakanishi, S.; Ishii, N.; Uno, T.; Kato, T.; Konno, F.; Kawamoto, Y.; Tsuchida, S.; et al. Low blood cell counts in wild Japanese monkeys after the Fukushima Daiichi nuclear disaster. Sci. Rep. 2014, 4, 5793. [Google Scholar] [CrossRef]
- Urushihara, Y.; Suzuki, T.; Shimizu, Y.; Ohtaki, M.; Kuwahara, Y.; Suzuki, M.; Uno, T.; Fujita, S.; Saito, A.; Yamashiro, H.; et al. Haematological analysis of Japanese macaques (Macaca fuscata) in the area affected by the Fukushima Daiichi Nuclear Power Plant accident. Sci. Rep. 2018, 8, 16748. [Google Scholar] [CrossRef]
- Hayama, S.I.; Tsuchiya, M.; Ochiai, K.; Nakiri, S.; Nakanishi, S.; Ishii, N.; Kato, T.; Tanaka, A.; Konno, F.; Kawamoto, Y.; et al. Small head size and delayed body weight growth in wild Japanese monkey fetuses after the Fukushima Daiichi nuclear disaster. Sci. Rep. 2017, 7, 3528. [Google Scholar] [CrossRef]
- Hayama, S.I.; Nakanishi, S.; Tanaka, A.; Konno, F.; Kawamoto, Y.; Omi, T. Influence of radiation exposure to delayed fetal growth in wild Japanese monkeys after the Fukushima accident. Front. Vet. Sci. 2023, 10, 1151361. [Google Scholar] [CrossRef] [PubMed]
- Nohara, C.; Hiyama, A.; Taira, W.; Otaki, J.M. Robustness and radiation resistance of the pale grass blue butterfly from radioactively contaminated areas: A possible case of adaptive evolution. J. Hered. 2018, 109, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Horemans, N.; Spurgeon, D.J.; Lecomte-Pradines, C.; Saenen, E.; Bradshaw, C.; Oughton, D.; Rasnaca, I.; Kamstra, J.H.; Adam-Guillermin, C. Current evidence for a role of epigenetic mechanisms in response to ionizing radiation in an ecotoxicological context. Environ. Pollut. 2019, 251, 469–483. [Google Scholar] [CrossRef] [PubMed]
- Gombeau, K.; Bonzom, J.M.; Cavalié, I.; Camilleri, V.; Orjollet, D.; Dubourg, N.; Beaugelin-Seiller, K.; Bourdineaud, J.P.; Lengagne, T.; Armant, O.; et al. Dose-dependent genomic DNA hypermethylation and mitochondrial DNA damage in Japanese tree frogs sampled in the Fukushima Daiichi area. J. Environ. Radioact. 2020, 225, 106429. [Google Scholar] [CrossRef]
- Hancock, S.; Vo, N.T.K.; Goncharova, R.I.; Seymour, C.B.; Byun, S.H.; Mothersill, C.E. One-Decade-Spanning transgenerational effects of historic radiation dose in wild populations of bank voles exposed to radioactive contamination following the chernobyl nuclear disaster. Environ. Res. 2020, 80, 108816. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakauchi, K.; Otaki, J.M. Soil Microbes and Plant-Associated Microbes in Response to Radioactive Pollution May Indirectly Affect Plants and Insect Herbivores: Evidence for Indirect Field Effects from Chernobyl and Fukushima. Microorganisms 2024, 12, 364. https://doi.org/10.3390/microorganisms12020364
Sakauchi K, Otaki JM. Soil Microbes and Plant-Associated Microbes in Response to Radioactive Pollution May Indirectly Affect Plants and Insect Herbivores: Evidence for Indirect Field Effects from Chernobyl and Fukushima. Microorganisms. 2024; 12(2):364. https://doi.org/10.3390/microorganisms12020364
Chicago/Turabian StyleSakauchi, Ko, and Joji M. Otaki. 2024. "Soil Microbes and Plant-Associated Microbes in Response to Radioactive Pollution May Indirectly Affect Plants and Insect Herbivores: Evidence for Indirect Field Effects from Chernobyl and Fukushima" Microorganisms 12, no. 2: 364. https://doi.org/10.3390/microorganisms12020364
APA StyleSakauchi, K., & Otaki, J. M. (2024). Soil Microbes and Plant-Associated Microbes in Response to Radioactive Pollution May Indirectly Affect Plants and Insect Herbivores: Evidence for Indirect Field Effects from Chernobyl and Fukushima. Microorganisms, 12(2), 364. https://doi.org/10.3390/microorganisms12020364





