The Effects of Coriandrum sativum L. and Cucurbita pepo L. against Gastrointestinal Parasites in Swine: An In Vivo Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Biochemical Analyses of Coriandrum sativum and Cucurbita pepo
2.2. Swine Husbandry
2.3. Experimental Design and Sampling Procedures
2.4. Evaluation of Antiparasitic Efficacy
2.5. Ontologies and Ethics Statement
2.6. Statistical Analysis
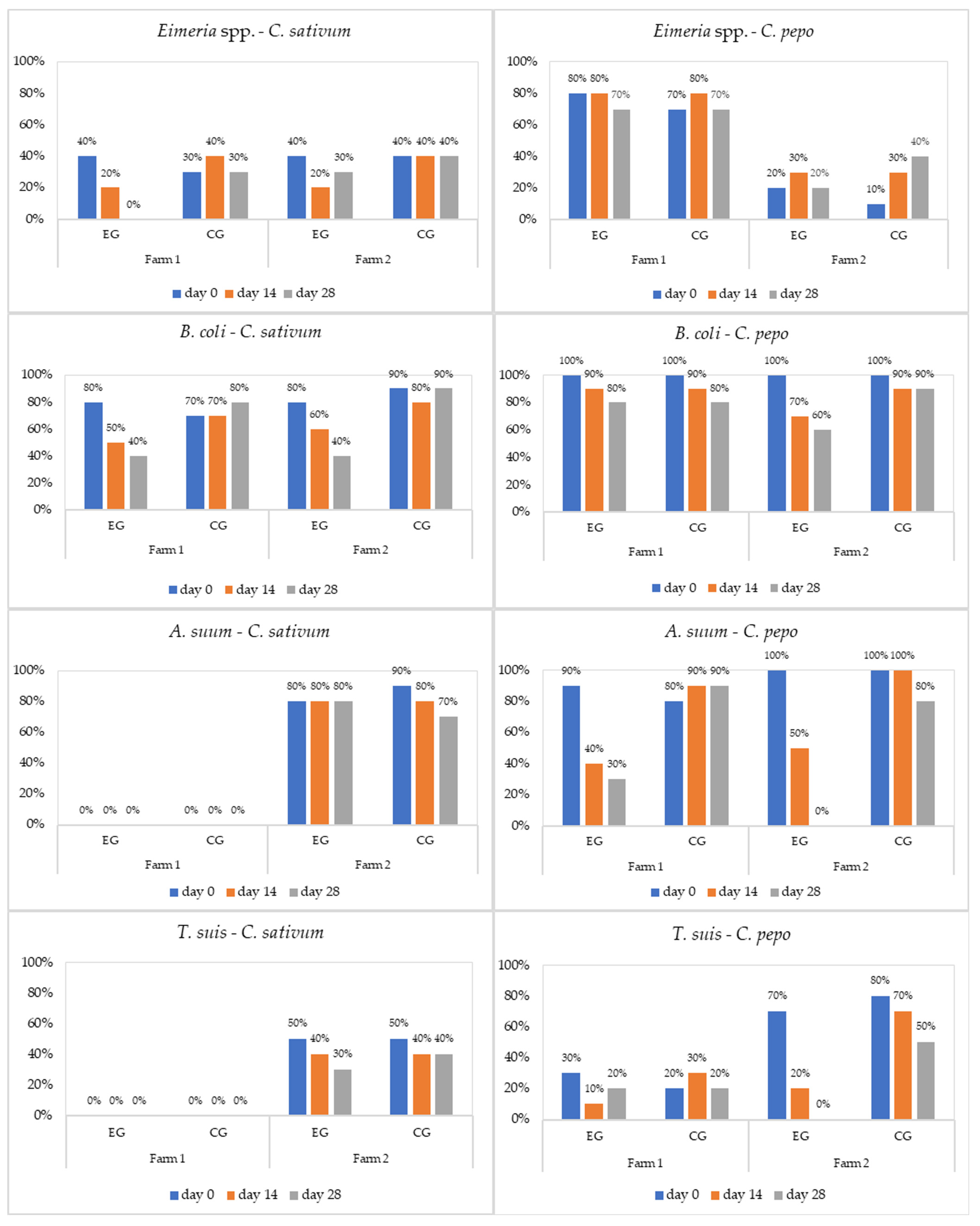
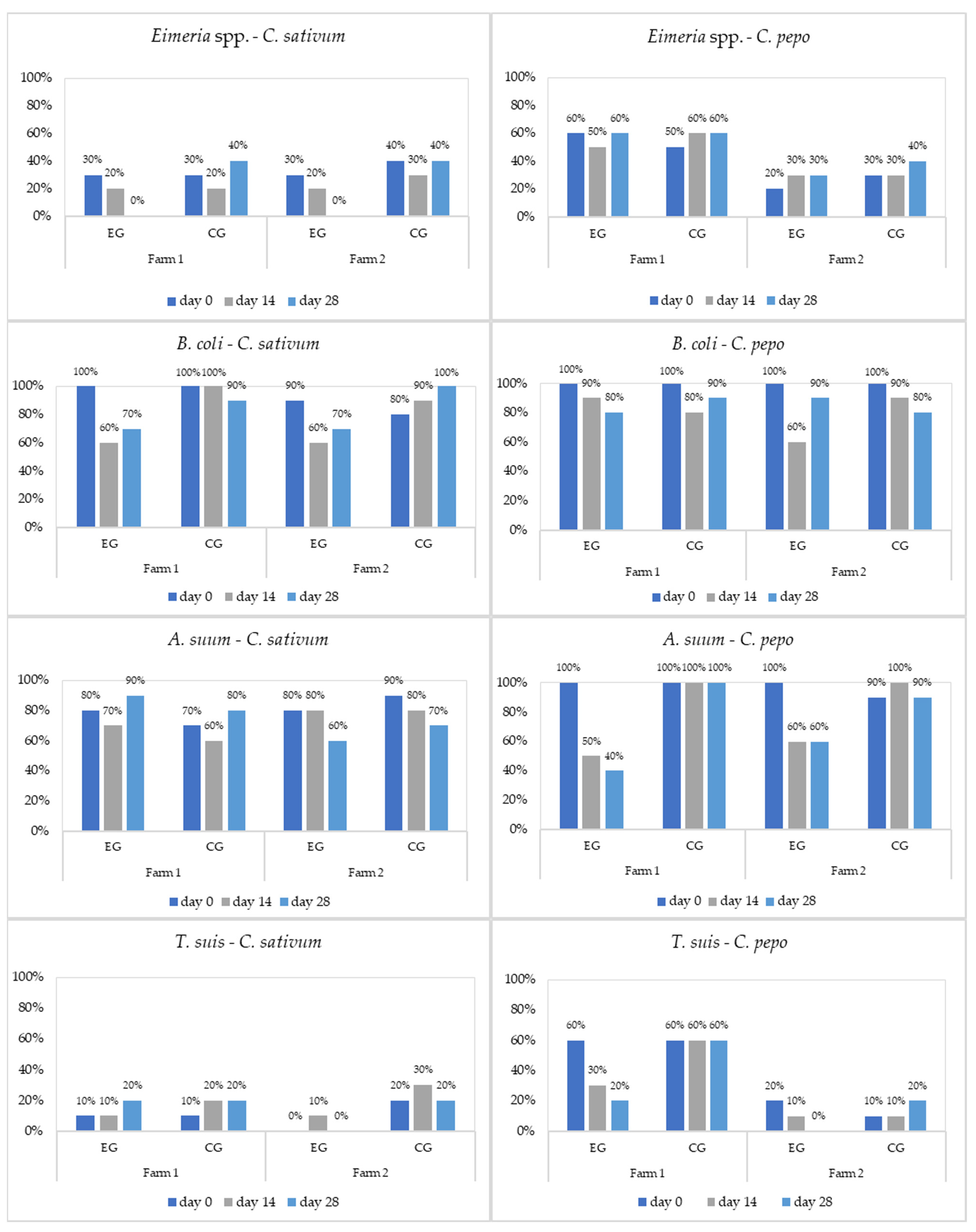
3. Results
3.1. Analysis of Plant Extracts
3.2. Analysis of Antiparasitic Activity of Plants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poux, X. Low input farming systems in Europe: What is at stake. In Low Input Farming Systems: An Opportunity to Develop Sustainable Agriculture, Proceedings of the JRC Summer University Ranco, 2–5 July 2007; Office for Official Publications of the European Communities: Luxembourg, 2008; pp. 1–11. [Google Scholar]
- Botha, M.; Petrescu-Mag, I.V.; Gavriloaie, C. Rustic gene reserves for the future of breed improvement technologies: Old swine (Sus scrofa domesticus) strains and their perspectives. Porc. Res. 2016, 6, 37–56. [Google Scholar]
- Matiuti, M.H.; Hutu, I.; Matiuti, C.L. A possible variant of preserving in vivo the local pigs breeds in Romania. Danub. Anim. Genet. Resour. 2017, 58–61. [Google Scholar]
- Miao, Z.H.; Glatz, P.C.; Ru, Y.J. Review of production, husbandry and sustainability of free-range pig production systems. Asian-Australas. J. Anim. Sci. 2004, 17, 1615–1634. [Google Scholar] [CrossRef]
- Terán Ramírez, V. Efecto del Extracto Hidroalcohólico de Semillas de Coriandrum sativum Sobre la Formación de Larva 2 de Ascaris suum y Trichuris ovis, en Condiciones de Laboratorio. Ph.D. Thesis, Universidad Nacional de Trujillo, Facultad de Ciencias Biológicas Escuela Académico Profesional de Microbiología y Parasitología, Trujillo, Peru, 2015. [Google Scholar]
- Băieş, M.H.; Boros, Z.; Gherman, C.M.; Spînu, M.; Mathe, A.; Pataky, S.; Lefkaditis, M.; Cozma, V. Prevalence of swine gastrointestinal parasites in two free-range farms from nord-west region of Romania. Pathogens 2022, 11, 954. [Google Scholar] [CrossRef]
- Eijck, I.A.J.M.; Borgsteede, F.H.M. A survey of gastrointestinal pig parasites on free-range, organic and conventional pig farms in the Netherlands. Vet. Res. Commun. 2005, 29, 407–414. [Google Scholar] [CrossRef]
- Pettersson, E.; Sjölund, M.; Wallgren, T.; Lind, E.O.; Höglund, J.; Wallgren, P. Management practices related to the control of gastrointestinal parasites on Swedish pig farms. Porc Health Manag. 2021, 7, 12. [Google Scholar] [CrossRef]
- Zajíčková, M.; Nguyen, L.T.; Skálová, L.; Stuchlíková, L.R.; Matoušková, P. Anthelmintics in the future: Current trends in the discovery and development of new drugs against gastrointestinal nematodes. Drug Discov. Today 2020, 25, 430–437. [Google Scholar] [CrossRef]
- Laribi, B.; Kouki, K.; M’Hamdi, M.; Bettaieb, T. Coriander (Coriandrum sativum L.) and its bioactive constituents. Fitoterapia 2015, 103, 9–26. [Google Scholar] [CrossRef]
- Al-Snafi, A.E. Antiparasitic, antiprotozoal, molluscicidal and insecticidal activity of medicinal plants (part 2)–plant based review. Sch. Acad. J. Pharm. 2016, 5, 194–207. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Ghalesefidi, M.J.; Azami, M.; Mohaghegh, M.A.; Hejazi, S.H.; Ghomashlooyan, M. In vitro and in vivo anthelmintic activity of seed extract of Coriandrum sativum compared to Niclosamid against Hymenolepis nana infection. J. Parasit. Dis. 2016, 40, 1307–1310. [Google Scholar] [CrossRef]
- Kalkal, H.; Kumar, P.; Vohra, S. Advances in control strategies and vaccine development of coccidiosis in poultry. Pharma. Innov. 2021, 10, 1084–1090. [Google Scholar]
- Momin, A.H.; Acharya, S.S.; Gajjar, A.V. Coriandrum sativum-review of advances in phytopharmacology. Int. J. Pharm. Sci. 2012, 3, 1233. [Google Scholar]
- Al-Snafi, A.E. A review on chemical constituents and pharmacological activities of Coriandrum sativum. IOSR J. Pharm. 2016, 6, 17–42. [Google Scholar] [CrossRef]
- Paarakh, P.M. Coriandrum sativum Linn. Review. Pharmacologyonline 2009, 3, 561–573. [Google Scholar]
- Pathak Nimish, L.; Kasture Sanjay, B.; Bhatt Nayna, M.; Rathod Jaimik, D. Phytopharmacological properties of Coriander sativum as a potential medicinal tree: An overview. J. Appl. Pharm. Sci. 2011, 1, 20–25. [Google Scholar]
- Mandal, S.; Mandal, M. Coriander (Coriandrum sativum L.) essential oil: Chemistry and biological activity. Asian Pac. J. Trop. Biomed. 2015, 5, 421–428. [Google Scholar] [CrossRef]
- Kumar, G.P.; Subrahmanyam, S.N. Phytochemical analysis, in-vitro screening for antimicrobial and anthelmintic activity of combined hydroalcoholic seed extracts of four selected folklore indian medicinal plants. Der. Pharm. Lett. 2013, 5, 168–176. [Google Scholar]
- Hussein, S.N.; Shukur, M.S. In-vitro anthelmentic efficacy of pumpkin seed oil (Cucurbita pepo) on toxocariosis (Toxocara cati). Explor. Anim. Med. Res. 2020, 10, 154–161. [Google Scholar]
- Syed, Q.A.; Akram, M.; Shukat, R. Nutritional and therapeutic importance of the pumpkin seeds. Seed 2019, 21, 15798–15803. [Google Scholar] [CrossRef]
- Adnan, M.; Gul, S.; Batool, S.; Fatima, B.; Rehman, A.; Yaqoob, S.; Aziz, M.A. A review on the ethnobotany, phytochemistry, pharmacology and nutritional composition of Cucurbita pepo L. J. Phytopharm. 2017, 6, 133–139. [Google Scholar] [CrossRef]
- Karanja, J.; Mugendi, B.J.; Khamis, F.; Muchugi, A. Nutritional composition of the pumpkin (Cucurbita spp.) seed cultivated from selected regions in Kenya. J. Hortic. Lett. 2013, 3, 17–22. [Google Scholar]
- Vidhya, C.; Loganathan, M.; Bhuvana, S.; Wadje, P.; Rajamani, M. A study on the evaluation of proximate, fatty acid and amino acid profile of two species of pumpkin using advanced techniques. Uttar Pradesh J. Zool. 2022, 43, 74–83. [Google Scholar]
- Dowidar, M.; Ahmed, A.; Mohamed, H. The Critical Nutraceutical Role of Pumpkin Seeds in Human and Animal Health: An Updated Review. Zagazig Vet. J. 2020, 48, 199–212. [Google Scholar] [CrossRef]
- Kinyua, C.M. Modelling and Application of Response Surface Methodology for Optimization of Weight Gain of Eight Weeks Old Kenbro Served with Pumpkin (Cucurbita Pepo L.) Seeds Extract. Ph.D. Dissertation, Chuka University, Chuka, Kenya, 2019. [Google Scholar]
- Achilonu, M.C.; Nwafor, I.C.; Umesiobi, D.O.; Sedibe, M.M. Biochemical proximates of pumpkin (Cucurbitaeae spp.) and their beneficial effects on the general well-being of poultry species. J. Anim. Physiol. Anim. Nutr. 2018, 102, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Fruhwirth, G.O.; Hermetter, A. Seeds and oil of the Styrian oil pumpkin: Components and biological activities. Eur. J. Lipid Sci. Technol. 2007, 109, 1128–1140. [Google Scholar] [CrossRef]
- Etewa, S.E.; Abaza, S.M. Herbal medicine and parasitic diseases. Parasitol. United J. 2011, 4, 3–14. [Google Scholar]
- Bǎieş, M.H.; Gherman, C.; Boros, Z.; Olah, D.; Vlase, A.M.; Cozma-Petrut, A.; Györke, A.; Miere, D.; Vlase, L.; Crişan, G.; et al. The Effects of Allium sativum L., Artemisia absinthium L., Cucurbita pepo L., Coriandrum sativum L., Satureja hortensis L. and Calendula officinalis L. on the Embryogenesis of Ascaris suum Eggs during an In Vitro Experimental Study. Pathogens 2022, 11, 1065. [Google Scholar] [CrossRef]
- Manser, M.M.; Saez, A.C.; Chiodini, P.L. Faecal Parasitology: Concentration Methodology Needs to be Better Standardised. PLoS Negl. Trop. Dis. 2016, 10, e0004579. [Google Scholar] [CrossRef]
- Mircean, V.; Cozma, V.; Gyorke, A. Diagnostic Coproscopic in Bolile Parazitare la Animale, (Coproparasitological Diagnostic in Parasitic Diseases in Animals); Risoprint: Cluj-Napoca, Romania, 2011; pp. 23–35. [Google Scholar]
- Dobson, R.J.; Hosking, B.C.; Jacobson, C.L.; Cotter, J.L.; Besier, R.B.; Stein, P.A.; Reid, S.A. Preserving new anthelmintics: A simple method for estimating faecal egg count reduction test (FECRT) confidence limits when efficacy and/or nematode aggregation is high. Vet. Parasitol. 2012, 186, 79–92. [Google Scholar] [CrossRef]
- McKenna, P.B. Further comparison of faecal egg count reduction test procedures: Sensitivity and specificity. N. Z. Vet. J. 2006, 54, 365–366. [Google Scholar] [CrossRef]
- Grzybek, M.; Kukula-Koch, W.; Strachecka, A.; Jaworska, A.; Phiri, A.M.; Paleolog, J.; Tomczuk, K. Evaluation of anthelmintic activity and composition of pumpkin (Cucurbita pepo L.) seed extracts—In vitro and in vivo studies. Int. J. Mol. Sci. 2016, 17, 1456. [Google Scholar] [CrossRef] [PubMed]
- Boros, Z.; Baies, M.H.; Gherman, C.; Cozma, V. The effects of Artemisia absinthium (wormwood), Allium sativum (garlic), Cucurbita pepo (pumpkin), and Coriandrum sativum (coriander) on Trichinella spiralis and Trichinella britovi larvae, in vitro study. Sci. Parasitol. 2021, 22, 70–78. [Google Scholar]
- Singletary, K. Coriander: Overview of potential health benefits. Nutr. Today 2016, 51, 151–161. [Google Scholar] [CrossRef]
- Jabeen, Q.; Bashir, S.; Lyoussi, B.; Gilani, A.H. Coriander fruit exhibits gut modulatory, blood pressure lowering and diuretic activities. J. Ethnopharmacol. 2009, 122, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Burdock, G.A.; Carabin, I.G. Safety assessment of coriander (Coriandrum sativum L.) essential oil as a food ingredient. Food Chem. Toxicol. 2009, 47, 22–34. [Google Scholar] [CrossRef]
- Al-Mofleh, I.A.; Alhaider, A.A.; Mossa, J.S.; Al-Sohaibani, M.O.; Rafatullah, S.; Qureshi, S. Protection of gastric mucosal damage by Coriandrum sativum L. pretreatment in Wistar albino rats. Environ. Toxicol. Pharmacol. 2006, 22, 64–69. [Google Scholar] [CrossRef]
- Khazdair, M.R.; Anaeigoudari, A.; Hashemzehi, M.; Mohebbati, R. Neuroprotective potency of some spice herbs, a literature review. J. Tradit. Complement. Med. 2019, 9, 98–105. [Google Scholar] [CrossRef]
- World Health Organization. Endocrinotoxicity induced by Coriandrum sativa: A case report. WHO Drug Info. 2002, 16, 15.
- Cândido, F.G.; De Oliveira, F.C.; Lima, M.F.C.; Pinto, C.A.; Da Silva, L.L.; Martino, H.S.; Rita de Cássia, G.A. Addition of pooled pumpkin seed to mixed meals reduced postprandial glycemia: A randomized placebo-controlled clinical trial. Nutr. Res. 2018, 56, 90–97. [Google Scholar] [CrossRef]
- Chen, L.; Long, R.; Huang, G.; Huang, H. Extraction and antioxidant activities in vivo of pumpkin polysaccharide. Ind. Crops Prod. 2020, 146, 112199. [Google Scholar] [CrossRef]
- Iwo, M.I.; Insanu, M.; Dass, C.A.S. Development of immunonutrient from pumpkin (Cucurbita moschata Duchense Ex. Lamk.) seed. Procedia Chem. 2014, 13, 105–111. [Google Scholar] [CrossRef]
- Asgary, S.; Moshtaghian, S.J.; Setorki, M.; Kazemi, S.; Rafieian-Kopaei, M.; Adelnia, A.; Shamsi, F. Hypoglycaemic and hypolipidemic effects of pumpkin (Cucurbita pepo L.) on alloxan-induced diabetic rats. Afr. J. Pharm. Pharmacol. 2011, 5, 2620–2626. [Google Scholar]
- Damiano, R.; Cai, T.; Fornara, P.; Franzese, C.A.; Leonardi, R.; Mirone, V. The role of Cucurbita pepo in the management of patients affected by lower urinary tract symptoms due to benign prostatic hyperplasia: A narrative review. Arch. Ital. Urol. Androl. 2016, 88, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Chari, K.Y.; Polu, P.R.; Shenoy, R.R. An appraisal of pumpkin seed extract in 1, 2-dimethylhydrazine induced colon cancer in wistar rats. J. Toxicol. 2018, 2018, 6086490. [Google Scholar] [CrossRef]
- Magi, E.; Talvik, H.; Jarvis, T. In vivo studies of the effect of medicinal herbs on the pig nodular worm (Oesophagostomum spp.). Helminthologia 2005, 42, 67–69. [Google Scholar]
- De Queiroz-Neto, A.; Mataqueiro, M.I.; Santana, A.E.; Alessi, A.C. Toxicologic evaluation of acute and subacute oral administration of Cucurbita maxima seed extracts to rats and swine. J. Ethnopharmacol. 1994, 43, 45–51. [Google Scholar] [CrossRef]
- Karpagam, T.; Varalakshmi, B.; Bai, J.S.; Gomathi, S. Effect of different doses of Cucurbita pepo linn extract as an anti-inflammatory and analgesic nutraceautical agent on inflamed rats. Int. J. Pharm. Res. Dev. 2011, 3, 184–192. [Google Scholar]
- Krimer-Malešević, V.; Mađarev-Popović, S.; Vaštag, Ž.; Radulović, L.; Peričin, D. Chapter 109. Phenolic acids in pumpkin (Cucurbita pepo L.) seeds. In Nuts and Seeds in Health and Disease Prevention; Preedy, V.R., Watson, R.R., Patel, V.B., Eds.; Academic Press: Serbia, 2011; pp. 925–932. [Google Scholar]
- Njoku, R.C.C.; Abarikwu, S.O.; Uwakwe, A.A.; Mgbudom-Okah, C.J.; Ezirim, C.Y. Dietary fluted pumpkin seeds induce reversible oligospermia and androgen insufficiency in adult rats. Syst. Biol. Reprod. Med. 2019, 65, 437–450. [Google Scholar] [CrossRef]
- Daugschies, A.; Imarom, S.; Ganter, M.; Bollwahn, W. Prevalence of Eimeria spp. in sows at piglet-producing farms in Germany. J. Vet. Med. 2004, 51, 135–139. [Google Scholar] [CrossRef]
- Pop, L.M.; Varga, E.; Coroian, M.; Nedișan, M.E.; Mircean, V.; Dumitrache, M.O.; Györke, A. Efficacy of a commercial herbal formula in chicken experimental coccidiosis. Parasites Vectors 2019, 12, 343. [Google Scholar] [CrossRef]
- Junkuszew, A.; Milerski, M.; Bojar, W.; Szczepaniak, K.; Le Scouarnec, J.; Tomczuk, K.; Bracik, K. Effect of various antiparasitic treatments on lamb growth and mortality. Small Rumin. Res. 2015, 123, 306–313. [Google Scholar] [CrossRef]
- Bǎieş, M.H.; Györke, A.; Cotuţiu, V.D.; Boros, Z.; Cozma-Petruţ, A.; Filip, L.; Vlase, L.; Vlase, A.-M.; Crişan, G.; Spînu, M.; et al. The In Vitro Anticoccidial Activity of Some Herbal Extracts against Eimeria spp. Oocysts Isolated from Piglets. Pathogens 2023, 12, 258. [Google Scholar] [PubMed]
- Aćimović, M.G.; Kostadinović, L.M.; Puvača, N.M.; Popović, S.J.; Urošević, M.I. Phytochemical constituents of selected plants from Apiaceae family and their biological effects in poultry. Food Feed Res. 2016, 43, 35–41. [Google Scholar] [CrossRef]
- Seidavi, A.; Tavakoli, M.; Slozhenkina, M.; Gorlov, I.; Hashem, N.M.; Asroosh, F.; Swelum, A.A. The use of some plant-derived products as effective alternatives to antibiotic growth promoters in organic poultry production: A review. Environ. Sci. Pollut. Res. 2021, 28, 47856–47868. [Google Scholar] [CrossRef] [PubMed]
- Obiad, H.M.; Al-Alousi, T.I.; Al-Jboori, A.H. An epidemiologic study on Cryptosporidium spp. in Kirkuk city with some trials for in vitro treating the parasite. In Second Scientific Conference; Science College, Tikrit University: Tikrit, Iraq, 2012. [Google Scholar]
- Obiad, H.M.; Al-Alousi, T.I.; Al-Jboori, A.H. The in vivo effect of some medicinal plant extracts on Cryptosporidium parasite. J. Univ. Anbar. Pure Sci. 2012, 6, 3. [Google Scholar]
- Nilles-Bije, M.; Rivera, W.L. Ultrastructural and molecular characterization of Balantidium coli isolated in the Philippines. Parasitol. Res. 2010, 106, 387–394. [Google Scholar] [CrossRef]
- Hindsbo, O.; Nielsen, C.V.; Andreassen, J.; Willingham, A.L.; Bendixen, M.; Nielsen, M.A.; Nielsen, N.O. Age-dependent occurrence of the intestinal ciliate Balantidium coli in pigs at a Danish research farm. Acta Vet. Scand. 2000, 41, 79–83. [Google Scholar] [CrossRef]
- Thamsborg, S.M.; Nejsum, P.; Mejer, H. Chapter 14. Impact of Ascaris suum in livestock. In Ascaris: The Neglected Parasite; Holland, C., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 363–381. [Google Scholar]
- Ganestya, S.; Djumarga, S.; Utari, C.S. Anthelmintic effects of pumpkin (Cucurbita moschata) seed extract on Ascaris suum in vitro. Asian J. Nat. Prod. Biochem. 2012, 10, 1–6. [Google Scholar] [CrossRef]
- Urban, J.; Kokoska, L.; Langrova, I.; Matejkova, J. In vitro anthelmintic effects of medicinal plants used in Czech Republic. Pharm. Biol. 2008, 46, 808–813. [Google Scholar] [CrossRef]
- Bazh, E.K.; El-Bahy, N.M. In vitro and in vivo screening of anthelmintic activity of ginger and curcumin on Ascaridia galli. Parasitol. Res. 2013, 112, 3679–3686. [Google Scholar] [CrossRef]
- Aziz, A.R.A.; AbouLaila, M.R.; Aziz, M.; Omar, M.A.; Sultan, K. In vitro and in vivo anthelmintic activity of pumpkin seeds and pomegranate peels extracts against Ascaridia galli. Beni-Suef Univ. J. Basic Appl. Sci. 2018, 7, 231–234. [Google Scholar]
- Hellawi, H.; Ibrahim, O.M. Evaluation of anthelmintic activity of N-hexane extract of Cucurbita Maxima and Azadirachta Indica pulp seed against Ascaridia Galli in vitro. Biochem. Cell. Arch. 2021, 21, 211–217. [Google Scholar]
- Summers, R.W.; Elliott, D.E.; Urban, J.F.; Thompson, R.; Weinstock, J. Trichuris suis therapy in Crohn’s disease. Gut 2005, 54, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Jex, A.R.; Nejsum, P.; Schwarz, E.M.; Hu, L.; Young, N.D.; Hall, R.S.; Gasser, R.B. Genome and transcriptome of the porcine whipworm Trichuris suis. Nat. Genet. 2014, 46, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.Q.; Ai, L.; Zou, F.C.; Verweij, J.J.; Jiang, Q.; Li, M.W.; Zhu, X.Q. A multiplex PCR tool for the specific identification of Oesophagostomum spp. from pigs. Parasitol. Res. 2008, 103, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Eguale, T.; Tilahun, G.; Debella, A.; Feleke, A.; Makonnen, E. In vitro and in vivo anthelmintic activity of crude extracts of Coriandrum sativum against Haemonchus contortus. J. Ethnopharmacol. 2007, 110, 428–433. [Google Scholar] [CrossRef]
- Macedo, I.T.F.; Oliveira, L.M.B.D.; Camurça-Vasconcelos, A.L.F.; Ribeiro, W.L.C.; Santos, J.M.L.D.; Morais, S.M.D.; Bevilaqua, C.M.L. In vitro effects of Coriandrum sativum, Tagetes minuta, Alpinia zerumbet and Lantana camara essential oils on Haemonchus contortus. Rev. Bras. Parasitol. Vet. 2013, 22, 463–469. [Google Scholar] [CrossRef]
- Helal, M.A.; Abdel-Gawad, A.M.; Kandil, O.M.; Khalifa, M.M.; Cave, G.W.; Morrison, A.A.; Elsheikha, H.M. Nematocidal effects of a coriander essential oil and five pure principles on the infective larvae of major ovine gastrointestinal nematodes in vitro. Pathogens 2020, 9, 740. [Google Scholar] [CrossRef]
- Strickland, V.J.; Krebs, G.L.; Potts, W. Pumpkin kernel and garlic as alternative treatments for the control of Haemonchus contortus in sheep. Anim. Prod. Sci. 2009, 49, 139–144. [Google Scholar] [CrossRef]
- Matthews, K.K.; O’Brien, D.J.; Whitley, N.C.; Burke, J.M.; Miller, J.E.; Barczewski, R.A. Investigation of possible pumpkin seeds and ginger effects on gastrointestinal nematode infection indicators in meat goat kids and lambs. Small Rumin. Res. 2016, 136, 1–6. [Google Scholar] [CrossRef]
- Elhadi, I.M.; Koko, W.S.; Dahab, M.M.; El Imam, Y.M.; El Mageed, M.A.E.A. Antigiardial activity of some Cucurbita species and Lagenaria siceraria. J. For. Prod. Ind. 2013, 2, 43–47. [Google Scholar]
- Barratt, J.; Ellis, J.; Harkness, J.; Marriott, D.; Stark, D. Evaluation of the in vitro antiprotozoal activity of various dry plant extracts against Dientamoeba fragilis. J. Infect. Dis. Ther. 2013, 1, 1000111. [Google Scholar]
- Grabensteiner, E.; Liebhart, D.; Arshad, N.; Hess, M. Antiprotozoal activities determined in vitro and in vivo of certain plant extracts against Histomonas meleagridis, Tetratrichomonas gallinarum and Blastocystis sp. Parasitol. Res. 2008, 103, 1257–1264. [Google Scholar] [CrossRef] [PubMed]
- Kabbashi, A.S.; Koko, W.S.; Mohammed, S.E.A.; Musa, N.; Osman, E.E.; Dahab, M.M.; Mohammed, A.K. In vitro amoebicidal, antimicrobial and antioxidant activities of the plants Adansonia digitata and Cucurbita maxima. Adv. Med. Plant Res. 2014, 2, 50–57. [Google Scholar]
- Ezeani, C.; Ezenyi, I.; Erhunse, N.; Sahal, D.; Akunne, T.; Okoli, C. Assessment of antimalarial medicinal plants used in Nigerian ethnomedicine reveals antimalarial potential of Cucurbita pepo leaf extract. Heliyon 2022, 8, e09916. [Google Scholar] [CrossRef]
- Salman, S.S.; Ardalan, N.M. Evaluation of Amygdalin (B17) and Cucurbita pepo (Pumpkin seed) activity against Blastocystis from diarrheic patients in Baghdad, Iraq: In Vitro Study. Baghdad Sci. J. 2022, 19, 0016. [Google Scholar] [CrossRef]
- Li, T.; Ito, A.; Chen, X.; Long, C.; Okamoto, M.; Raoul, F.; Craig, P.S. Usefulness of pumpkin seeds combined with areca nut extract in community-based treatment of human taeniasis in northwest Sichuan Province, China. Acta Trop. 2012, 124, 152–157. [Google Scholar] [CrossRef]
- Acorda, J.A.; Mangubat, I.Y.E.C.; Divina, B.P. Evaluation of the in vivo efficacy of pumpkin (Cucurbita pepo) seeds against gastrointestinal helminths of chickens. Turk. J. Vet. Anim. Sci. 2019, 43, 206–211. [Google Scholar] [CrossRef]
- Ayaz, E.; Gökbulut, C.; Coşkun, H.; Uçar Türker, A.; Özsoy, Ş.; Ceylan, K. Evaluation of the anthelmintic activity of pumpkin seeds (Cucurbita maxima) in mice naturally infected with Aspiculuris tetraptera. J. Pharmacogn. Phytother. 2015, 7, 189–193. [Google Scholar]
- Alhawiti, A.O.; Toulah, F.H.; Wakid, M.H. Anthelmintic potential of Cucurbita pepo seeds on Hymenolepis nana. Acta Parasitol. 2019, 64, 276–281. [Google Scholar] [CrossRef]
- Monzote, L.; Herrera, I.; Satyal, P.; Setzer, W.N. In-vitro evaluation of 52 commercially-available essential oils against Leishmania amazonensis. Molecules 2019, 24, 1248. [Google Scholar] [CrossRef] [PubMed]
- Rondon, F.C.M.; Bevilaqua, C.M.L.; Accioly, M.P.; Morais, S.M.D.; Andrade-Júnior, H.F.D.; Carvalho, C.A.D.; Magalhães, H.C.R. In vitro efficacy of Coriandrum sativum, Lippia sidoides and Copaifera reticulata against Leishmania chagasi. Rev. Bras. Parasitol. Vet. 2012, 21, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Rondon, F.C.; Bevilaqua, C.M.; Accioly, M.P.; Morais, S.M.; Andrade-Junior, H.F.; Machado, L.K.; Rodrigues, A.C.M. In vitro effect of Aloe vera, Coriandrum sativum and Ricinus communis fractions on Leishmania infantum and on murine monocytic cells. Vet. Parasitol. 2011, 178, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Dawwas, A. Investigation of biochemical effect of phenols extract isolated from Coriandrum sativum seeds against Echinococcus granulosus parasite in vitro. J. Thi-Qar Sci. 2008, 1, 2–9. [Google Scholar]

| Feed | C. sativum Group | C. pepo Group | ||||
|---|---|---|---|---|---|---|
| Weaners % | Fatteners % | Sows % | Weaners % | Fatteners % | Sows % | |
| corn | 38.16 | 46.05 | 37.65 | 37.5 | 45.15 | 36 |
| wheat | 20 | 25 | 25 | 20 | 25 | 25 |
| barley | 30 | 12 | 20 | 30 | 12 | 20 |
| peas | 10 | 15 | 15 | 10 | 15 | 15 |
| calcium carbonate | 1.5 | 1.5 | 1.5 | 1.5 | 1.5 | 1.5 |
| coriander fruits | 0.34 | 0.45 | 0.85 | - | - | - |
| pumpkin seeds | - | - | - | 1 | 1.35 | 2.5 |
| Parasite | Farm | Group | p-Values–Friedman Test | |||||
|---|---|---|---|---|---|---|---|---|
| C. sativum | C. pepo | |||||||
| Day 0 | Day 14 | Day 28 | Day 0 | Day 14 | Day 28 | |||
| Eimeria spp. | F1 | EG CG | 0.0498 # 0.5818 # | 0.7640 # 0.6387 # | ||||
| p-value | 0.7337 * | 0.3847 * | 0.2730 * | 0.7723 * | 0.3854 * | 0.4822 * | ||
| F2 | EG CG | 0.9170 # 0.4677 # | 0.3679 # 0.3499 # | |||||
| p-value | >0.9999 * | 0.3257 * | 0.6501 * | 0.7337 * | 0.7624 * | 0.3075 * | ||
| B. coli | F1 | EG CG | 0.0076 # 0.6331 # | 0.4724 # 0.0484 # | ||||
| p-value | 0.8501 * | 0.2123 * | 0.0073 * | 0.5708 * | 0.9699 * | 0.7055 * | ||
| F2 | EG CG | 0.0484 # 0.4520 # | 0.0071 # 0.2231 # | |||||
| p-value | 0.9097 * | 0.0211 * | 0.0013 * | 0.4274 * | 0.0052 * | 0.0233 * | ||
| A. suum | F1 | EG CG | - - | 0.0029 # 0.1561 # | ||||
| p-value | - | - | - | 0.3847 * | 0.0013 * | 0.0012 * | ||
| F2 | EG CG | 0.0125 # 0.0451 # | 0.0001 # 0.4820 # | |||||
| p-value | 0.7624 * | 0.7913 * | 0.2730 * | 0.7913 * | 0.0002 * | 0.0008 * | ||
| T. suis | F1 | EG CG | - - | 0.3679 # 0.7515 # | ||||
| p-value* | - | - | - | 0.7055 * | 0.4057 * | 0.9097 * | ||
| F2 | EG CG | 0.4724 # 0.9556 # | 0.0049 # 0.0626 # | |||||
| p-value | 0.9699 * | 0.7337 * | 0.8798 * | 0.6501 * | 0.0233 * | 0.0640 * | ||
| Cryptosporidium spp. | F1 | EG CG | 3 3 | 3 2 | 2 1 | 4 3 | 2 8 | 2 8 |
| p-value | 0.8142 ** | 0.6517 ** | 0.6053 ** | 0.6749 ** | 0.7910 ** | 0.7910 ** | ||
| F2 | EG CG | 1 1 | 1 1 | 1 2 | 1 1 | 1 2 | 1 1 | |
| p-value | 0.7368 ** | 0.7368 ** | 0.6053 ** | 0.7368 ** | 0.6053 ** | 0.7368 ** | ||
| Parasite | Farm | Group | p-Values–Friedman Test | |||||
|---|---|---|---|---|---|---|---|---|
| C. sativum | C. pepo | |||||||
| Day 0 | Day 14 | Day 28 | Day 0 | Day 14 | Day 28 | |||
| Eimeria spp. | F1 | EG CG | 0.1738 # 0.5488 # | 0.5308 # 0.5079 # | ||||
| p-value | 0.9397 * | 0.9699 * | 0.1405 * | 0.7337 * | 0.6231 * | 0.6776 * | ||
| F2 | EG CG | 0.1738 # 0.6873 # | 0.6918 # 0.7470 # | |||||
| p-value | 0.7913 * | 0.7055 * | 0.1405 * | 0.7337 * | 0.9397 * | 0.7913 * | ||
| B. coli | F1 | EG CG | 0.0164 # 0.3469 # | 0.0032 # 0.7165 # | ||||
| p-value | >0.9999 * | 0.0082 * | 0.0257 * | >0.9999 * | 0.3447 * | 0.0284 * | ||
| F2 | EG CG | 0.0178 # 0.8187 # | 0.0062 # 0.9131 # | |||||
| p-value | 0.5205 * | 0.0028 * | 0.0041 * | 0.9699 * | 0.0211 * | 0.5205 * | ||
| A. suum | F1 | EG CG | 0.0618 # 0.3796 # | 0.0002 # 0.8975 # | ||||
| p-value | 0.7624 * | 0.7913 * | 0.6501 * | 0.4057 * | 0.0002 * | 0.0002 * | ||
| F2 | EG CG | 0.2545 # 0.0283 # | 0.0003 # 0.4516 # | |||||
| p-value | 0.0821 * | 0.1306 * | 0.1509 * | 0.3847 * | 0.0002 * | 0.0017 * | ||
| T. suis | F1 | Case Control | 0.3679 # 0.3679 # | 0.1117 # 0.7640 # | ||||
| p-value | >0.999 * | 0.7055 | >0.999 * | 0.7624 * | 0.1212 * | 0.0757 * | ||
| F2 | EG CG | 0.3679 # n.a. | 0.3679 # n.a. | |||||
| p-value * | 0.4727 * | 0.4497 * | 0.4727 * | 0.7337 * | >0.9999 * | 0.4727 * | ||
| Parasite | Farm | Group | p-Values—Friedman Test | |||||
|---|---|---|---|---|---|---|---|---|
| C. sativum | C. pepo | |||||||
| Day 0 | Day 14 | Day 28 | Day 0 | Day 14 | Day 28 | |||
| Eimeria spp. | F1 | EG CG | 0.3679 # 0.7788 # | 0.4668 # 0.7548 # | ||||
| p-value | 0.7337 * | 0.9699 * | 0.4497 * | 0.7055 * | 0.5454 * | 0.3075 * | ||
| F2 | EG CG | 0.0957 # 0.8521 # | 0.5522 # 0.3679 # | |||||
| p-value | 0.4497 * | 0.5708 * | 0.5967 * | 0.6501 * | 0.9699 * | 0.4497 * | ||
| B. coli | F1 | EG CG | 0.0297 # 0.8789 # | 0.1040 # 0.5811 # | ||||
| p-value | 0.6232 * | 0.1736 * | 0.0257 * | 0.4963 * | 0.9699 * | 0.4727 * | ||
| F2 | Case Control | 0.0344 # 0.1822 # | 0.2609 # 0.7733 # | |||||
| p-value | 0.6776 * | 0.0015 * | 0.0032 * | 0.5708 * | 0.1736 * | 0.6776 * | ||
| Oesophagostomum spp. | F1 | EG CG | - - | 0.1353 # n.a. | ||||
| p-value | - | - | 0.4727 * | 0.4727 * | 0.9698 * | 0.9698 * | ||
| F2 | EG CG | - - | - - | |||||
| p-value | - | - | 0.0757 * | - | - | - | ||
| A. suum | F1 | EG CG | 0.3679 # 0.3679 # | - - | ||||
| p-value | 0.7337 * | 0.4727 * | 0.9699 * | 0.3258 * | 0.0058 * | 0.0073 * | ||
| F2 | EG CG | 0.0845 # n.a. | 0.0051 # 0.9726 # | |||||
| p-value | 0.0073 * | 0.0588 * | 0.5454 * | 0.5454 * | 0.0452 * | 0.0073 * | ||
| Cryptosporidium spp. | F1 | EG | 1 | 0 | 0 | 2 | 1 | 0 |
| CG | 1 | 1 | 1 | 1 | 1 | 2 | ||
| F2 | EG | 1 | 1 | 1 | 1 | 1 | 1 | |
| CG | 0 | 0 | 1 | 0 | 1 | 0 | ||
| Parasite | C. sativum (Day 14) | C. sativum (Day 28) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weaners | Fatteners | Sows | Weaners | Fatteners | Sows | |||||||
| F1 | F2 | F1 | F2 | F1 | F2 | F1 | F2 | F1 | F2 | F1 | F2 | |
| Eimeria spp. | 71.4 | 72.1 | 80 | 30.6 | 60 | 41.5 | 100 | 25.4 | 100 | 100 | 50 | 75.7 |
| B. coli | 29.6 | 68.9 | 44.4 | 62.4 | 23.2 | 74.2 | 84.4 | 79.5 | 50.4 | 20.1 | 67.4 | 31.2 |
| A. suum | - | 18.1 | 8.1 | 13.9 | - | 0 | - | 30.3 | 0 | 7.2 | - | 0 |
| T. suis | - | 0 | 0 | 0 | - | - | - | 3.3 | 0 | 0 | - | - |
| Parasite | C. pepo (day 14) | C. pepo (day 28) | ||||||||||
| Weaners | Fatteners | Sows | Weaners | Fatteners | Sows | |||||||
| F1 | F2 | F1 | F2 | F1 | F2 | F1 | F2 | F1 | F2 | F1 | F2 | |
| Eimeria spp. | 11.6 | 96.6 | 13.9 | 33.3 | 45.4 | 0 | 24.9 | 94.7 | 35.9 | 0 | 61.1 | 0 |
| B. coli | 2.3 | 59.5 | 22.9 | 54.9 | 3.0 | 30.1 | 0 | 34.1 | 45.1 | 24.8 | 33.6 | 22.2 |
| A. suum | 77.4 | 80.9 | 83.5 | 79.7 | 87.1 | 70.3 | 79.7 | 100 | 84.5 | 95.9 | 85.9 | 88.9 |
| T. suis | 91.6 | 80.7 | 50.1 | 75.0 | - | - | 91.0 | 100 | 57.7 | 100 | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Băieş, M.-H.; Cotuţiu, V.-D.; Spînu, M.; Mathe, A.; Cozma-Petruț, A.; Miere, D.; Bolboacǎ, S.D.; Cozma, V. The Effects of Coriandrum sativum L. and Cucurbita pepo L. against Gastrointestinal Parasites in Swine: An In Vivo Study. Microorganisms 2023, 11, 1230. https://doi.org/10.3390/microorganisms11051230
Băieş M-H, Cotuţiu V-D, Spînu M, Mathe A, Cozma-Petruț A, Miere D, Bolboacǎ SD, Cozma V. The Effects of Coriandrum sativum L. and Cucurbita pepo L. against Gastrointestinal Parasites in Swine: An In Vivo Study. Microorganisms. 2023; 11(5):1230. https://doi.org/10.3390/microorganisms11051230
Chicago/Turabian StyleBăieş, Mihai-Horia, Vlad-Dan Cotuţiu, Marina Spînu, Attila Mathe, Anamaria Cozma-Petruț, Doina Miere, Sorana D. Bolboacǎ, and Vasile Cozma. 2023. "The Effects of Coriandrum sativum L. and Cucurbita pepo L. against Gastrointestinal Parasites in Swine: An In Vivo Study" Microorganisms 11, no. 5: 1230. https://doi.org/10.3390/microorganisms11051230
APA StyleBăieş, M.-H., Cotuţiu, V.-D., Spînu, M., Mathe, A., Cozma-Petruț, A., Miere, D., Bolboacǎ, S. D., & Cozma, V. (2023). The Effects of Coriandrum sativum L. and Cucurbita pepo L. against Gastrointestinal Parasites in Swine: An In Vivo Study. Microorganisms, 11(5), 1230. https://doi.org/10.3390/microorganisms11051230










