Impacts of Temperature and Water Activity Interactions on Growth, Aflatoxin B1 Production and Expression of Major Biosynthetic Genes of AFB1 in Aspergillus flavus Isolates
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Isolates
2.2. Fungal Isolates and Growth Conditions
2.3. Growth Assessment
2.4. Quantification of AFB1 by HPLC
2.5. Extraction of RNA, cDNA Synthesis and the RT-PCR Reaction
2.6. Statistical Analysis
3. Results
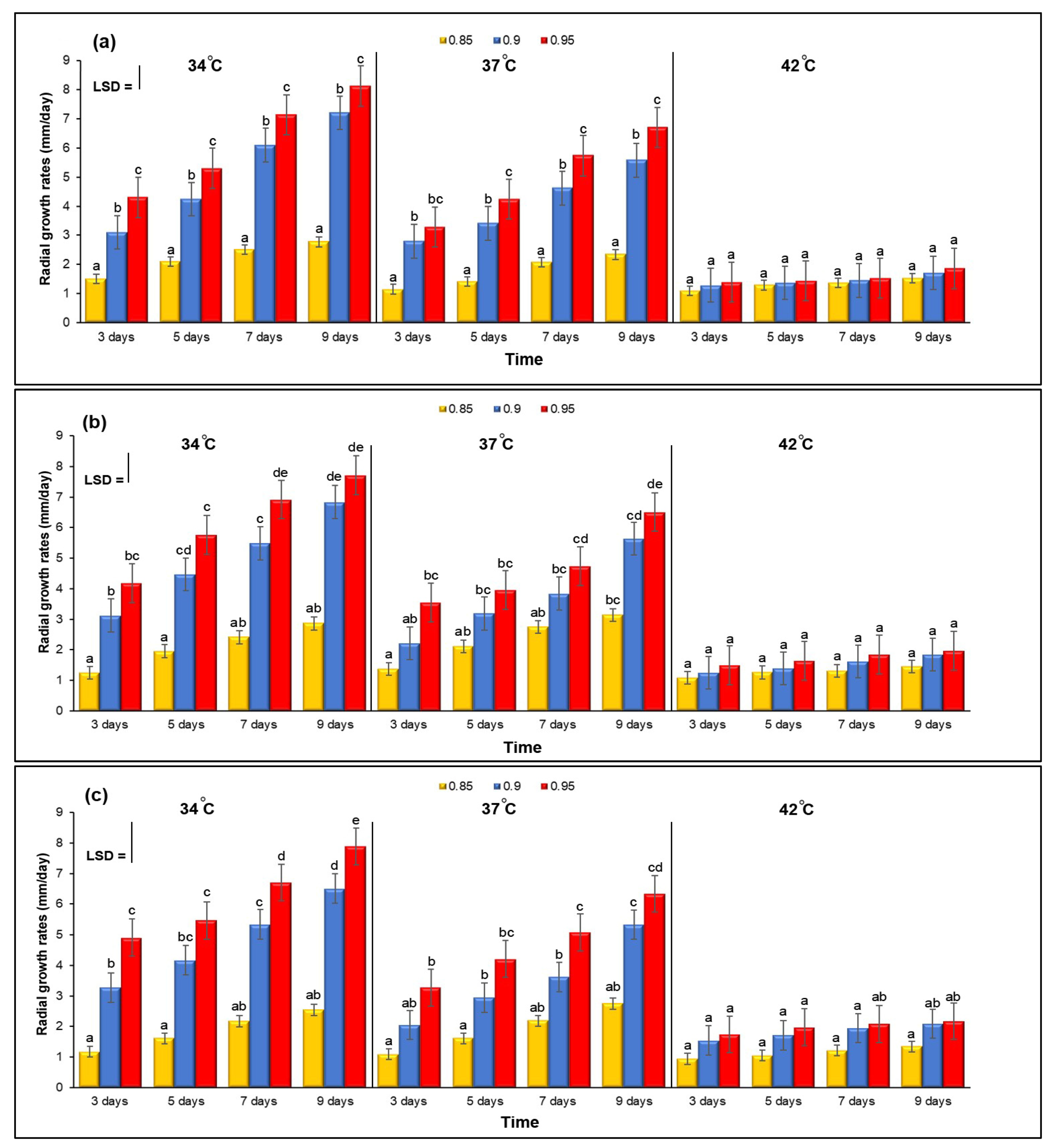
3.1. Effect of aw × Temperature Interactions on Three Isolates of A. flavus Grown on YES Agar Media
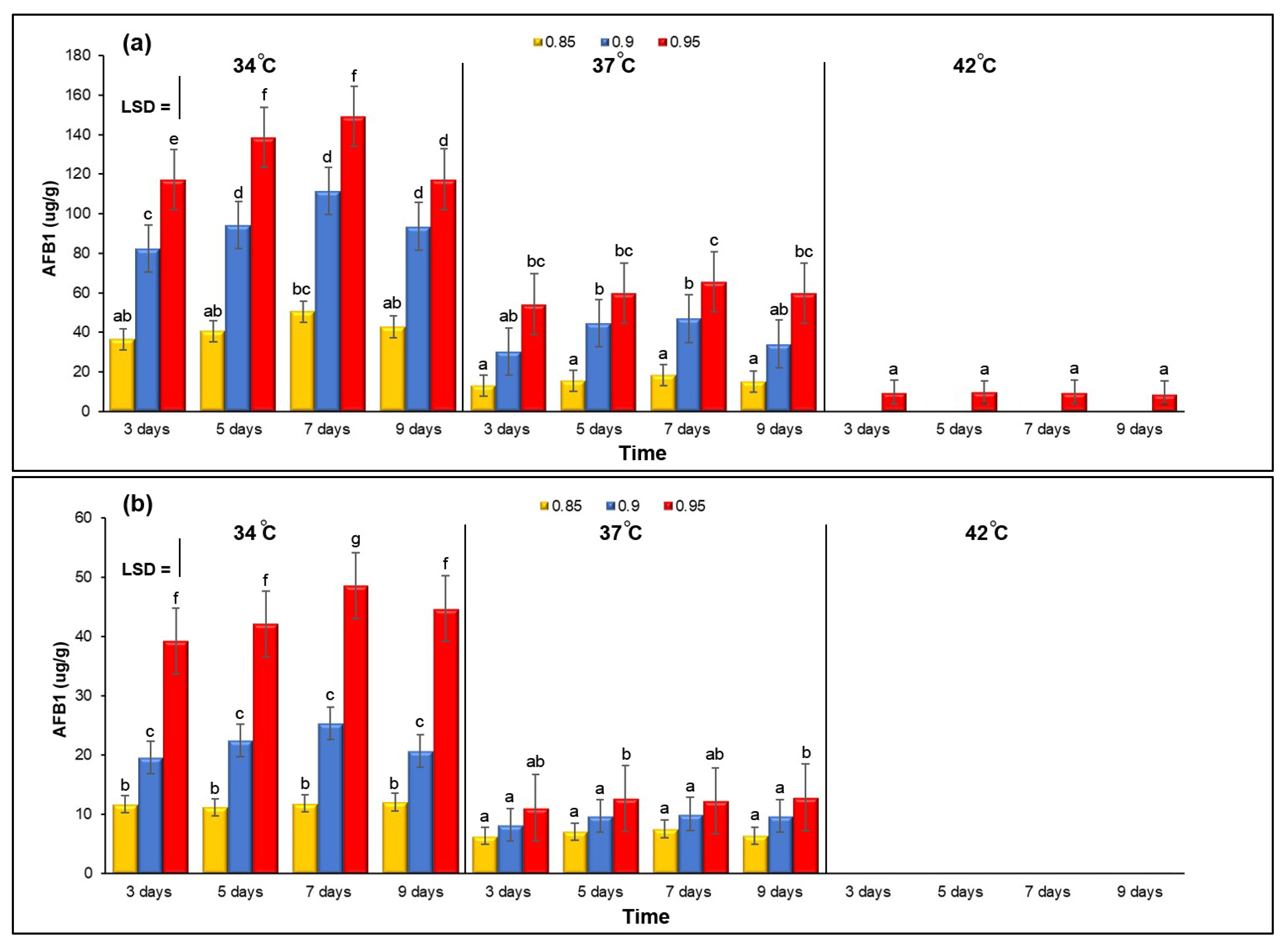
3.2. Effect of aw × Temperature Interactions on AB1 Production by Three A. flavus Isolates on YES Agar Media
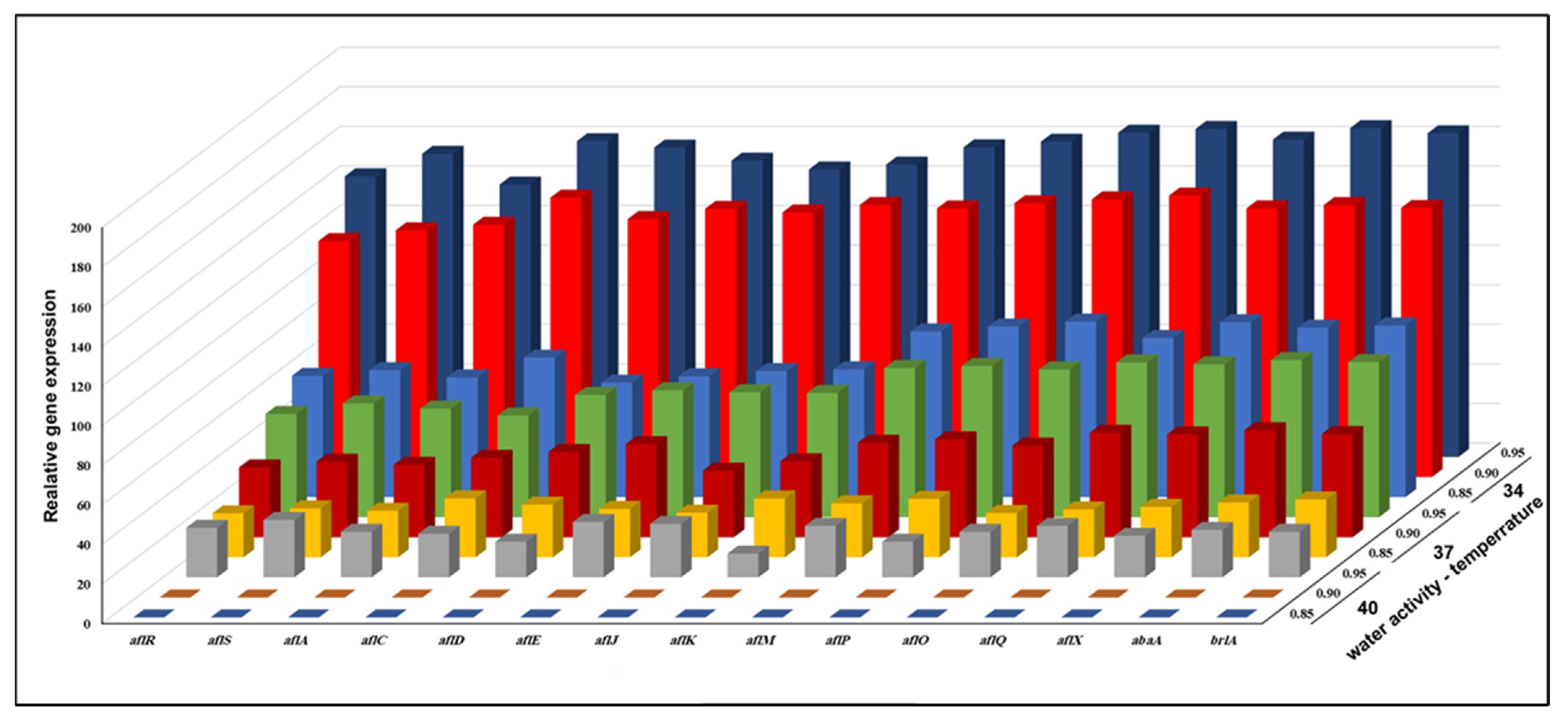
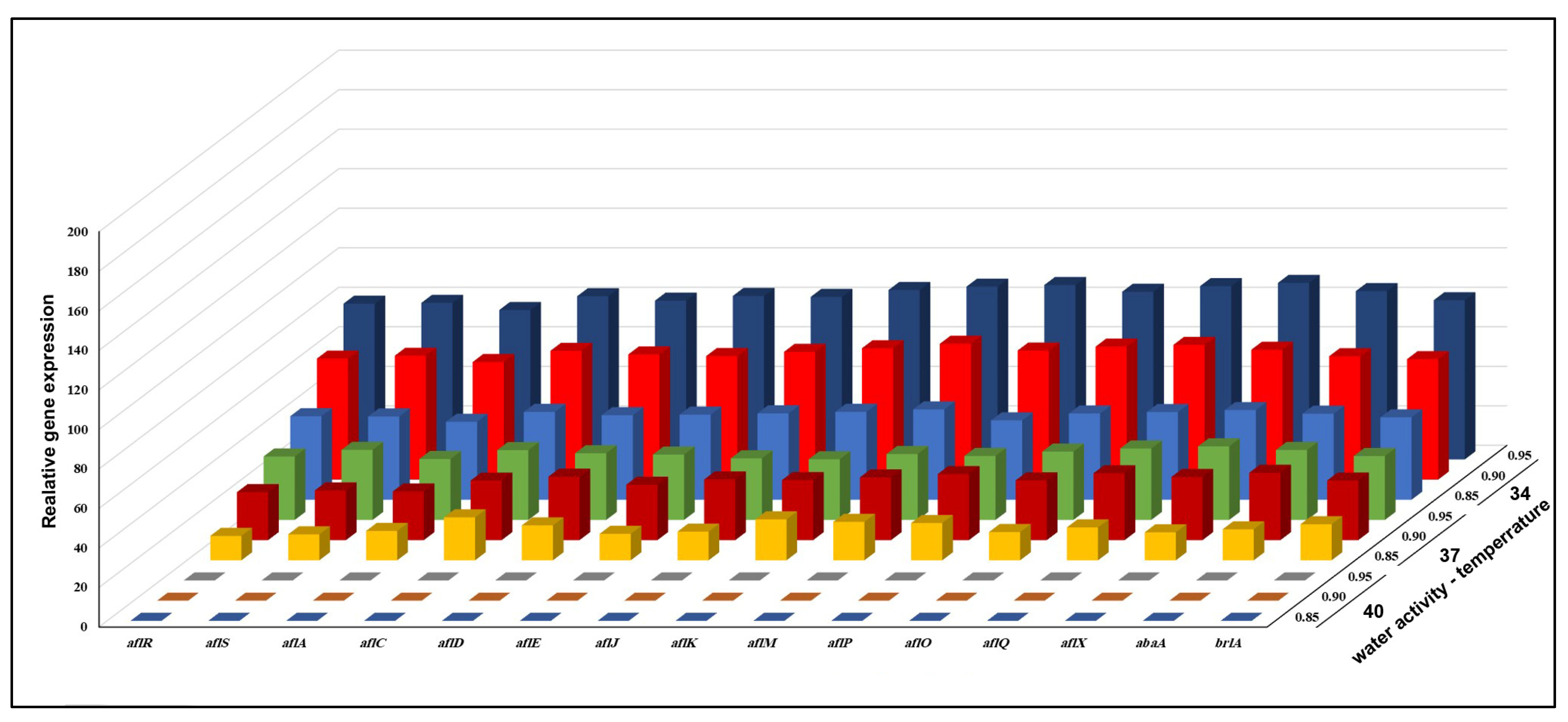
3.3. Effect of aw × Temperature Interactions on the Expression of 15 Genes Involved in the Biosynthesis of AFB1 Production
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tengey, T.K.; Kankam, F.; Ndela, D.N.; Frempong, D.; Appaw, W.O. Growth and Toxigenicity of A. flavus on Resistant and Susceptible Peanut Genotypes. Toxins 2022, 14, 536. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Zhong, H.; Han, X.; Guo, Z.; Yang, W.; Liu, Y.; Yang, K.; Zhuang, Z.; Wang, S. Proteomic profile of Aspergillus flavus in response to water activity. Fungal Biol. 2015, 119, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Wu, F. Perspective: Time to face the fungal threat. Nature 2014, 516, S7. [Google Scholar] [CrossRef] [PubMed]
- Cleveland, T.E.; Yu, J.; Fedorova, N.; Bhatnagar, D.; Payne, G.A.; Nierman, W.C. Potential of Aspergillus flavus genomics for applications in biotechnology. Trends Biotechnol. 2009, 27, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, X.; Ma, X.; Yu, Q.; Yu, X.; Liu, Y.; Nie, C.; Zhang, Y.; Xing, F. The regulatory mechanism of water activities on aflatoxins biosynthesis and conidia development, and transcription factor AtfB is involved in this regulation. Toxins 2021, 13, 431. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guan, X.; Xing, F.; Lv, C.; Dai, X.; Liu, Y. Effect of water activity and temperature on the growth of Aspergillus flavus, the expression of aflatoxin biosynthetic genes and aflatoxin production in shelled peanuts. Food Control 2017, 82, 325–332. [Google Scholar] [CrossRef]
- Medina, A.; Gilbert, M.K.; Mack, B.M.; Obrian, G.R.; Rodríguez, A.; Bhatnagar, D.; Payne, G.; Magan, N. Interactions between water activity and temperature on the Aspergillus flavus transcriptome and aflatoxin B (1) production. Int. J. Food Microbiol. 2017, 256, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Heydt, M.; Rüfer, C.E.; Abdel-Hadi, A.; Magan, N.; Geisen, R. The production of aflatoxin B1 or G1 by Aspergillus parasiticus at various combinations of temperature and water activity is related to the ratio of aflS to aflR expression. Mycotoxin 2010, 26, 241–246. [Google Scholar] [CrossRef]
- Lv, C.; Jin, J.; Wang, P.; Dai, X.; Liu, Y.; Zheng, M.; Xing, F. Interaction of water activity and temperature on the growth, gene expression and aflatoxin production by Aspergillus flavus on paddy and polished rice. Food Chem. 2019, 293, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Medina, A.; Rodríguez, A.; Sultan, Y.; Magan, N. Climate change factors and A. flavus: Effects on gene expression, growth and aflatoxin production. World Mycotoxin J. 2015, 8, 171–179. [Google Scholar] [CrossRef]
- Mahmoud, M.A. Detection of Aspergillus flavus in stored peanuts using real-time PCR and the expression of aflatoxin genes in toxigenic and atoxigenic A. flavus isolates. Foodborne Pathog. Dis. 2015, 12, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Heydt, M.; Abdel-Hadi, A.; Magan, N.; Geisen, R. Complex regulation of the aflatoxin biosynthesis gene cluster of Aspergillus flavus in relation to various combinations of water activity and temperature. Int. J. Food Microbiol. 2009, 135, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Christian, G. HPLC Tips and Tricks; Great Britain at the Iden Press: Oxford, UK, 1990; p. 608. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.S.T.; Sarreal, S.B.L.; Chang, P.-K.; Yu, J. Transcriptional regulation of aflatoxin biosynthesis and conidiation in Aspergillus flavus by Wickerhamomyces anomalus WRL-076 for reduction of aflatoxin contamination. Toxins 2019, 11, 81. [Google Scholar] [CrossRef]
- Priesterjahn, E.-M.; Geisen, R.; Schmidt-Heydt, M. Influence of light and water activity on growth and mycotoxin formation of selected isolates of Aspergillus flavus and Aspergillus parasiticus. Microorganisms 2020, 8, 2000. [Google Scholar] [CrossRef]
- Zingales, V.; Taroncher, M.; Martino, P.A.; Ruiz, M.-J.; Caloni, F. Climate Change and Effects on Molds and Mycotoxins. Toxins 2022, 14, 445. [Google Scholar] [CrossRef]
- Donato, C.J.R.; Cendoya, E.; Demonte, L.D.; Repetti, M.R.; Chulze, S.N.; Ramirez, M.L. Influence of abiotic factors (water activity and temperature) on growth and aflatoxin production by Aspergillus flavus in a chickpea-based medium. Int. J. Food Microbiol. 2022, 379, 109841. [Google Scholar] [CrossRef]
- Khan, R.; Ghazali, F.M.; Mahyudin, N.A.; Samsudin, N.I.P. Aflatoxin Biosynthesis, Genetic Regulation, Toxicity, and Control Strategies: A Review. J. Fungi 2021, 7, 606. [Google Scholar] [CrossRef]
- Baazeem, A.; Rodriguez, A.; Medina, A.; Magan, N. Impacts of climate change interacting abiotic factors on growth, aflD and aflR gene expression and aflatoxin B1 production by Aspergillus flavus strains in vitro and on pistachio nuts. Toxins 2021, 13, 385. [Google Scholar] [CrossRef]
- Chananya, C.; Warapa, M.; Thanapoom, M. Comparative study on the effect of temperature and water activity on Aspergillus flavus and Aspergillus carbonarius isolates growth and mycotoxin production on a chili powder medium. Cogent Food Agric. 2020, 6, 1782097. [Google Scholar]
- Norlia, M.; Jinap, S.; Nor-Khaizura, M.A.R.; Radu, S.; John, J.M.; Rahman, M.A.H.; Mshelia, L.P.; Zawiyah, S. Modelling the effect of temperature and water activity on the growth rate of Aspergillus flavus and aflatoxin production in peanut meal extract agar. Int. J. Food Microbiol. 2020, 335, 108836. [Google Scholar] [CrossRef] [PubMed]
- Shabeer, S.; Asad, S.; Jamal, A.; Ali, A. Aflatoxin contamination, its impact and management strategies: An updated review. Toxins 2022, 14, 307. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Payne, G.A.; Nierman, W.C.; Machida, M.; Bennett, J.W.; Campbell, B.C. Aspergillus flavus genomics as a tool for studying the mechanism of aflatoxin formation. Food Addit. Contam. Part A 2008, 25, 1152–1157. [Google Scholar]
- Yu, J.; Fedorova, N.D.; Montalbano, B.G.; Bhatnagar, D.; Cleveland, T.E.; Bennett, J.W.; William, C.N. Tight control of mycotoxin biosynthesis gene expression in Aspergillus flavus by temperature as revealed by RNA-Seq. FEMS Microbiol. Lett. 2011, 322, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Gallo, A.; Solfrizzo, M.; Epifani, F.; Panzarini, G.; Perrone, G. Effect of temperature and water activity on gene expression and aflatoxin biosynthesis in Aspergillus flavus on almond medium. International. Int. J. Food Microbiol. 2016, 217, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Chi, C.; Yu, J.; Shan, S.; Li, Q.; Guan, B.; Nieman, W.C.; Bennett, J.W. The inhibitory effect of Bacillus megaterium on aflatoxin and cyclopiazonic acid biosynthetic pathway gene expression in Aspergillus flavus. Appl. Microbiol. Biotechnol. 2014, 98, 5161–5172. [Google Scholar] [CrossRef] [PubMed]
- Bernáldez, V.; Córdoba, J.J.; Delgado, J.; Bermúdez, E.; Rodríguez, A. Gene expression analysis to predict aflatoxins B1 and G1 contamination in some plant origin foods. LWT 2018, 93, 517–524. [Google Scholar] [CrossRef]
- Natarajan, S.; Balachandar, D.; Senthil, N.; Paranidharan, V. Interaction of water activity and temperature on growth, gene expression, and aflatoxin B1 production in Aspergillus flavus on Indian senna (Cassia angustifolia Vahl.). Int. J. Food Microbiol. 2022, 361, 109457. [Google Scholar] [CrossRef] [PubMed]
- Kebede, H.; Abbas, H.K.; Fisher, D.K.; Bellaloui, N. Relationship between aflatoxin contamination and physiological responses of corn plants under drought and heat stress. Toxins 2012, 4, 1385–1403. [Google Scholar] [CrossRef] [PubMed]
- Segal, L.M.; Wilson, R.A. Reactive oxygen species metabolism and plant-fungal interactions. Fungal Genet Biol. 2018, 110, 1–9. [Google Scholar] [CrossRef]
- Gao, X.; Kolomiets, M.V. Host-derived lipids and oxylipins are crucial signals in modulating mycotoxin production by fungi. Toxin Rev. 2009, 28, 79–88. [Google Scholar] [CrossRef]
- Roze, L.V.; Chanda, A.; Linz, J.E. Compartmentalization and molecular traffic in secondary metabolism: A new understanding of established cellular processes. Fungal Genet. Biol. 2011, 48, 35–48. [Google Scholar] [CrossRef]
- Perrone, G.; Massimo, F.; Angel, M.; Michelangelo, P.; Magan, N. Toxigenic fungi and mycotoxins in a climate change scenario: Ecology, genomics, distribution, prediction and prevention of the risk. Microorganisms 2020, 8, 1496. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.K.; Ji, I.K.; Vivek, K.B.; Kwanwoo, K.; Hoomin, L.; Sonam, S.; Jesus, S.-G.; Jianbo, X.; Sajad, A.; Yun, S.H.; et al. Mycotoxins in food and feed: Toxicity, preventive challenges, and advanced detection techniques for associated diseases. Crit. Rev. Food Sci. Nutr. 2022, 21, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Sang, Y.L.; So, Y.W.; Hwa, Y.C.; Seongeun, H.; Gyoungju, N.; Hyang, S.C. Transcriptomic responses of Aspergillus flavus to temperature and oxidative stresses during aflatoxin production. Sci. Rep. 2021, 11, 2803. [Google Scholar] [CrossRef] [PubMed]
| Genes | Length (bp) | Primer Sequence |
|---|---|---|
| aflR | 88 | F: CCTTTCTCACTACTCGGGTTT R: GCAGGTAATCAATAATGTCGG |
| aflS | 109 | F: CTCGATGCGGCAGTGTATCT R: ACACCTCCACATGAGCCTTG |
| aflA | 111 | F: CATGCTGTTAACCCCCGACT R: AATTGGGCTAGGAAACCGGG |
| aflC | 61 | F: TGCATGGCGATGTGGTAGTT R: GTAAGGCCGCGGAAGAAAG |
| aflD | 106 | F: ATGCTCCCGTCCTACTGTTT R: ATGTTGGTGATGGTGCTGAT |
| aflE | 134 | F: GTGTGGAGGAAGTGATGCGA R: CGGGGTAAGTCCGTTAGCTC |
| aflJ | 146 | F: GCGTGATCAGTCGTCAATGC R: CAGGATGAGCGGTTGGTTCT |
| aflK | 114 | F: GCTGGGCATTCCAGTACGAT R: CCCATCAACTGACTGTGGCT |
| aflM | 100 | F: GAGCCAAAGTCGTGGTGAAC R: GCCTGGATTGCGATAGCGTC |
| aflP | 109 | F: CACGCTTTCAGAGCAGGTAA R: TTCGGTGGAGGAGGGAGTT |
| aflO | 115 | F: GTCCCGTTTCCTGGGTTGAT R: GCTTTCGATTGCTGCCCAAA |
| aflQ | 134 | F: GCACCAACAATTCGGCTCTG R: TGTGGAAGGGTGGAAGATGC |
| aflX | 112 | F: AGTCCTCAACATAGCCGCTG R: TAGTCCCCCAGGTTTGACGA |
| abaA | 120 | F: ACTGGCAAAAGGAGGTCGAG R: ATTCGAACGGTCTGCTGGTT |
| brlA | 131 | F: TCTAGCGGGGATGACCTCAA R: CCGAAGGAAGCCAAAAGTGC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Zaban, M.I. Impacts of Temperature and Water Activity Interactions on Growth, Aflatoxin B1 Production and Expression of Major Biosynthetic Genes of AFB1 in Aspergillus flavus Isolates. Microorganisms 2023, 11, 1199. https://doi.org/10.3390/microorganisms11051199
Al-Zaban MI. Impacts of Temperature and Water Activity Interactions on Growth, Aflatoxin B1 Production and Expression of Major Biosynthetic Genes of AFB1 in Aspergillus flavus Isolates. Microorganisms. 2023; 11(5):1199. https://doi.org/10.3390/microorganisms11051199
Chicago/Turabian StyleAl-Zaban, Mayasar I. 2023. "Impacts of Temperature and Water Activity Interactions on Growth, Aflatoxin B1 Production and Expression of Major Biosynthetic Genes of AFB1 in Aspergillus flavus Isolates" Microorganisms 11, no. 5: 1199. https://doi.org/10.3390/microorganisms11051199
APA StyleAl-Zaban, M. I. (2023). Impacts of Temperature and Water Activity Interactions on Growth, Aflatoxin B1 Production and Expression of Major Biosynthetic Genes of AFB1 in Aspergillus flavus Isolates. Microorganisms, 11(5), 1199. https://doi.org/10.3390/microorganisms11051199






