Prevalence of Small Intestinal Bacterial Overgrowth Syndrome in Patients with Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis: A Cross-Sectional Study
Abstract
1. Introduction
2. Patients and Methods
2.1. Study Design
2.2. Study Population
2.2.1. Non-Alcoholic Fatty Liver Disease Group
2.2.2. Control Group
2.3. Interventions
2.4. Laboratory Investigation
2.5. Study Endpoints
2.6. Statistical Analysis
2.7. Sample Size Estimation
2.8. Ethical Approval
3. Results
3.1. Primary Endpoint
3.2. Secondary Endpoints
- (i)
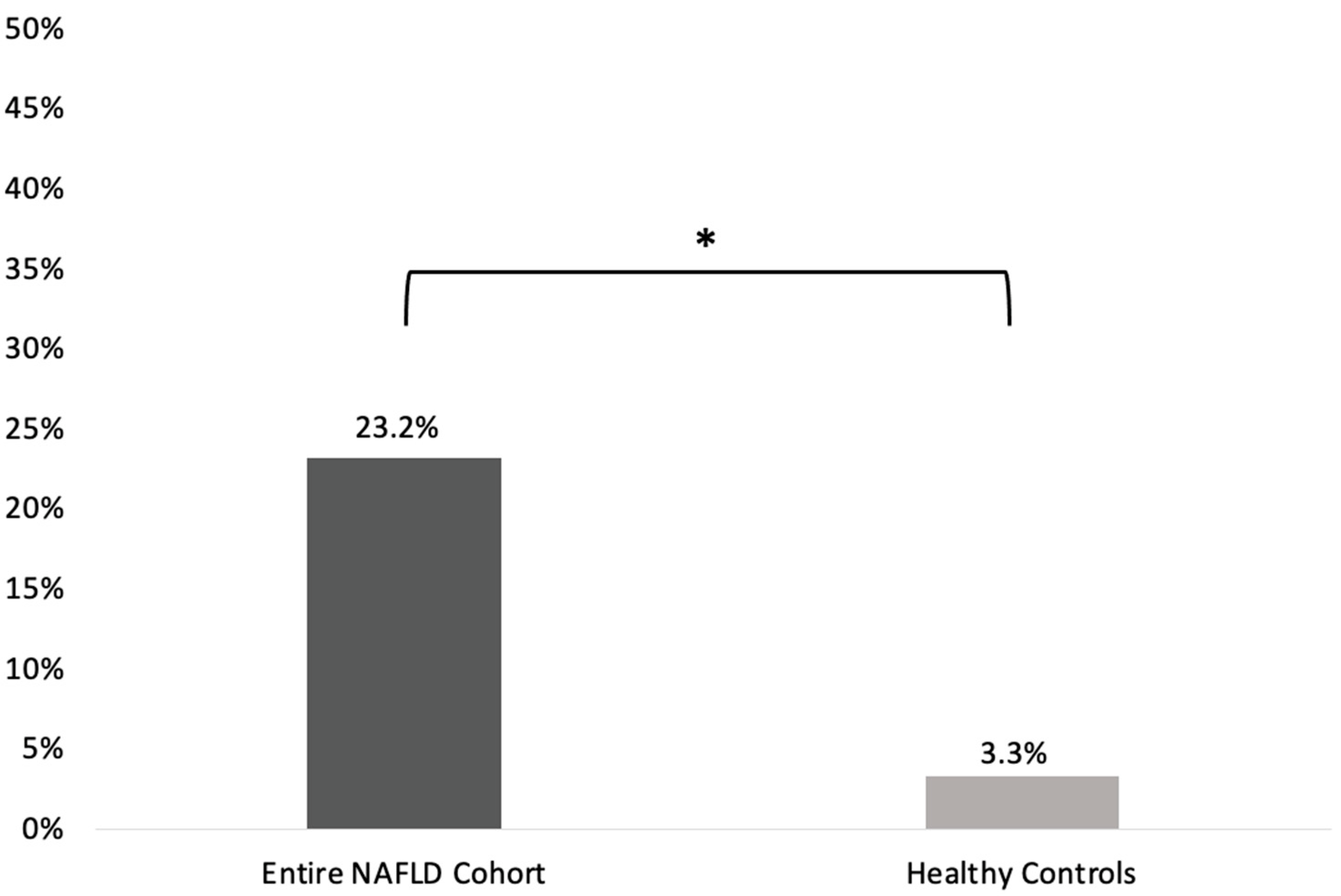
- SIBO prevalence was statistically significantly higher in the entire NAFLD cohort compared to the healthy controls group [22/95 (23.2%) vs. 1/30 (3.3%); p = 0.014; odds ratio (OR) (95%CI): 8.77 (1.12–66.7); Figure 1].
- (ii)
- Regarding the different stages of the disease, SIBO prevalence was higher among patients with NASH-associated cirrhosis (8/17; 47.1% [23.3–70.8]) compared to patients with NAFL (8/51; 15.7% [5.7–25.7]; p = 0.02, OR (95%CI): 4.8 [1.4–16.1]) and patients with NASH (6/27; 22.2% [6.5–37.9]). However, the difference in SIBO preavalence between NASH-associated cirrhosis and NASH patients did not reach statistical significance (p = 0.11; OR (95%CI): 3.1 [0.8–11.6]). No statistically significant difference regarding SIBO prevalence was noted between patients with NASH and NAFL (p = 0.54; OR (95%CI): 1.5 [0.5–4.9]; Figure 2).
- (iii)
- SIBO prevalence was significantly higher in NASH and NASH-associated cirrhosis patients compared to healthy controls (p = 0.04; OR (95%CI): 8.3 [1.01–74.1] and p < 0.001; OR (95%CI): 25.8 [2.8–234.8], respectively). Although numerically higher in NAFL, the difference in SIBO prevalence between NAFL patients and healthy controls did not reach significance (p = 0.14; OR (95%CI): 5.4 [0.6–45.4]; Figure 2).
- (iv)
- Cytokine concentrations in the duodenal aspirates (mean pg/mL ± SEM) in NAFLD patients and healthy controls are presented in Table 3. TNFα concentrations in the duodenal aspirates did not differ between NAFLD subjects and the healthy control group (8.79 ± 0.86 vs. 7.80 ± 0.10, p = 0.533). NAFLD patients also had similar levels of IL-1β and IL-6 compared to healthy controls (7.63 ± 1.74 vs. 6.26 ± 2.76, p = 0.701 and 3.17 ± 0.04 vs. 3.14 ± 0.10, p = 0.648, respectively; Table 3). There were no differences in duodenal aspirate cytokine levels among the different stages of NAFLD (p ≤ 0.524).

4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bugianesi, E.; McCullough, A.J.; Marchesini, G. Insulin resistance: A metabolic pathway to chronic liver disease. Hepatology 2005, 42, 987–1000. [Google Scholar] [CrossRef] [PubMed]
- Bhala, N.; Younes, R.; Bugianesi, E. Epidemiology and natural history of patients with NAFLD. Curr. Pharm. Des. 2013, 19, 5169–5176. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; LaVine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The diagnosis and management of non-alcoholic fatty liver disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012, 55, 2005–2023. [Google Scholar] [CrossRef] [PubMed]
- Han, M.A.T.; Saouaf, R.; Ayoub, W.; Todo, T.; Mena, E.; Noureddin, M. Magnetic resonance imaging and transient elastography in the management of Nonalcoholic Fatty Liver Disease (NAFLD). Expert Rev. Clin. Pharmacol. 2017, 10, 379–390. [Google Scholar] [CrossRef]
- Duseja, A.; Chawla, Y.K. Obesity and NAFLD: The role of bacteria and microbiota. Clin. Liver Dis. 2014, 18, 59–71. [Google Scholar] [CrossRef]
- Bedogni, G.; Miglioli, L.; Masutti, F.; Tiribelli, C.; Marchesini, G.; Bellentani, S. Prevalence of and risk factors for nonalcoholic fatty liver disease: The Dionysos nutrition and liver study. Hepatology 2005, 42, 44–52. [Google Scholar] [CrossRef]
- Méndez-Sánchez, N.; Bugianesi, E.; Gish, R.G.; Lammert, F.; Tilg, H.; Nguyen, M.H.; Sarin, S.K.; Fabrellas, N.; Zelber-Sagi, S.; Fan, J.-G.; et al. Global multi-stakeholder endorsement of the MAFLD definition. Lancet Gastroenterol. Hepatol. 2022, 7, 388–390. [Google Scholar] [CrossRef]
- Day, C.P.; James, O.F. Steatohepatitis: A tale of two “hits”? Gastroenterology 1998, 114, 842–845. [Google Scholar] [CrossRef]
- Dowman, J.K.; Tomlinson, J.W.; Newsome, P.N. Pathogenesis of non-alcoholic fatty liver disease. Qjm: Int. J. Med. 2010, 103, 71–83. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology 2010, 52, 1836–1846. [Google Scholar] [CrossRef]
- Papaefthymiou, A.; Potamianos, S.; Goulas, A.; Doulberis, M.; Kountouras, J.; Polyzos, S.A. Inflammatory Bowel Disease-associated Fatty Liver Disease: The Potential Effect of Biologic Agents. J. Crohn’s Colitis 2022, 16, 852–862. [Google Scholar] [CrossRef]
- Speliotes, E.K.; Butler, J.L.; Palmer, C.D.; Voight, B.F. PNPLA3 variants specifically confer increased risk for histologic nonalcoholic fatty liver disease but not metabolic disease. Hepatology 2010, 52, 904–912. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The human microbiome project. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef]
- Schwiertz, A.; Taras, D.; Schäfer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef]
- Novljan, U.; Pintar, T. Small Intestinal Bacterial Overgrowth in Patients with Roux-en-Y Gastric Bypass and One-Anastomosis Gastric Bypass. Obes. Surg. 2022, 32, 4102–4109. [Google Scholar] [CrossRef]
- Ghoshal, U.C.; Goel, A.; Quigley, E.M.M. Gut microbiota abnormalities, small intestinal bacterial overgrowth, and non-alcoholic fatty liver disease: An emerging paradigm. Indian J. Gastroenterol. 2020, 39, 9–21. [Google Scholar] [CrossRef]
- Gkolfakis, P.; Dimitriadis, G.; Triantafyllou, K. Gut microbiota and non-alcoholic fatty liver disease. Hepatobiliary Pancreat. Dis. Int. 2015, 14, 572–581. [Google Scholar] [CrossRef]
- Zhu, L.; Baker, S.S.; Gill, C.; Liu, W.; Alkhouri, R.; Baker, R.D.; Gill, S.R. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: A connection between endogenous alcohol and NASH. Hepatology 2013, 57, 601–609. [Google Scholar] [CrossRef]
- Mouzaki, M.; Comelli, E.M.; Arendt, B.M.; Bonengel, J.; Fung, S.K.; Fischer, S.E.; McGilvray, I.D.; Allard, J.P. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology 2013, 58, 120–127. [Google Scholar] [CrossRef]
- Wong, V.W.-S.; Tse, C.-H.; Lam, T.-T.Y.; Wong, G.L.-H.; Chim, A.M.-L.; Chu, W.C.-W.; Yeung, D.K.-W.; Law, P.T.-W.; Kwan, H.S.; Yu, J.; et al. Molecular characterization of the fecal microbiota in patients with nonalcoholic steatohepatitis—A longitudinal study. PLoS ONE 2013, 8, e62885. [Google Scholar] [CrossRef] [PubMed]
- Raman, M.; Ahmed, I.; Gillevet, P.M.; Probert, C.S.; Ratcliffe, N.M.; Smith, S.; Greenwood, R.; Sikaroodi, M.; Lam, V.; Crotty, P.; et al. Fecal microbiome and volatile organic compound metabolome in obese humans with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2013, 11, 868–875.e3. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, M.; Saad, R.J.; Long, M.D.; Rao, S.S.C. ACG Clinical Guideline: Small Intestinal Bacterial Overgrowth. Am. J. Gastroenterol. 2020, 115, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Sugano, K.; Tack, J.; Kuipers, E.J.; Graham, D.Y.; El-Omar, E.M.; Miura, S.; Haruma, K.; Asaka, M.; Uemura, N.; Malfertheiner, P. Kyoto global consensus report on Helicobacter pylori gastritis. Gut 2015, 64, 1353–1367. [Google Scholar] [CrossRef]
- Pyleris, E.; Giamarellos-Bourboulis, E.J.; Tzivras, D.; Koussoulas, V.; Barbatzas, C.; Pimentel, M. The prevalence of overgrowth by aerobic bacteria in the small intestine by small bowel culture: Relationship with irritable bowel syndrome. Dig. Dis. Sci. 2012, 57, 1321–1329. [Google Scholar] [CrossRef]
- Giamarellos-Bourboulis, E.J.; Pyleris, E.; Barbatzas, C.; Pistiki, A.; Pimentel, M. Small intestinal bacterial overgrowth is associated with irritable bowel syndrome and is independent of proton pump inhibitor usage. BMC Gastroenterol. 2016, 16, 67. [Google Scholar] [CrossRef]
- Giamarellos-Bourboulis, E.; Tang, J.; Pyleris, E.; Pistiki, A.; Barbatzas, C.; Brown, J.; Lee, C.C.; Harkins, T.T.; Kim, G.; Weitsman, S.; et al. Molecular assessment of differences in the duodenal microbiome in subjects with irritable bowel syndrome. Scand. J. Gastroenterol. 2015, 50, 1076–1087. [Google Scholar] [CrossRef]
- Leventogiannis, K.; Gkolfakis, P.; Spithakis, G.; Tsatali, A.; Pistiki, A.; Sioulas, A.; Giamarellos-Bourboulis, E.J.; Triantafyllou, K. Effect of a Preparation of Four Probiotics on Symptoms of Patients with Irritable Bowel Syndrome: Association with Intestinal Bacterial Overgrowth. Probiotics Antimicrob. Proteins 2019, 11, 627–634. [Google Scholar] [CrossRef]
- Wijarnpreecha, K.; Lou, S.; Watthanasuntorn, K.; Kroner, P.T.; Cheungpasitporn, W.; Lukens, F.J.; Pungpapong, S.; Keaveny, A.P.; Ungprasert, P. Small intestinal bacterial overgrowth and nonalcoholic fatty liver disease: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2020, 32, 601–608. [Google Scholar] [CrossRef]
- Augustyn, M.; Grys, I.; Kukla, M. Small intestinal bacterial overgrowth and nonalcoholic fatty liver disease. Clin. Exp. Hepatol. 2019, 5, 1–10. [Google Scholar] [CrossRef]
- GuimarÃes, V.M.; Santos, V.N.; Borges PS de, A.; DEFarias, J.L.R.; Grillo, P.; Parise, E.R. Peripheral blood endotoxin levels are not associated with small intestinal bacterial overgrowth in nonalcoholic fatty liver disease without cirrhosis. Arq. Gastroenterol. 2020, 57, 471–476. [Google Scholar] [CrossRef]
- Mikolasevic, I.; Delija, B.; Mijic, A.; Stevanovic, T.; Skenderevic, N.; Sosa, I.; Krznaric-Zrnic, I.; Abram, M.; Krznaric, Z.; Domislovic, V.; et al. Small intestinal bacterial overgrowth and non-alcoholic fatty liver disease diagnosed by transient elastography and liver biopsy. Int. J. Clin. Pract. 2021, 75, e13947. [Google Scholar] [CrossRef]
- Kapil, S.; Duseja, A.; Sharma, B.K.; Singla, B.; Chakraborti, A.; Das, A.; Ray, P.; Dhiman, R.K.; Chawla, Y. Small intestinal bacterial overgrowth and toll-like receptor signaling in patients with non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 2016, 31, 213–221. [Google Scholar] [CrossRef]
- Shi, H.; Mao, L.; Wang, L.; Quan, X.; Xu, X.; Cheng, Y.; Zhu, S.; Dai, F. Small intestinal bacterial overgrowth and orocecal transit time in patients of nonalcoholic fatty liver disease. Eur. J. Gastroenterol. Hepatol. 2021, 33 (Suppl. S1), e535–e539. [Google Scholar] [CrossRef]
- Ghoshal, U.C.; Baba, C.S.; Ghoshal, U.; Alexander, G.; Misra, A.; Saraswat, V.A.; Choudhuri, G. Low-grade small intestinal bacterial overgrowth is common in patients with non-alcoholic steatohepatitis on quantitative jejunal aspirate culture. Indian J. Gastroenterol. 2017, 36, 390–399. [Google Scholar] [CrossRef]
- De Oliveira, J.M.; Pace, F.L.; Ghetti, F.D.F.; Barbosa, K.V.B.D.; Cesar, D.E.; Chebli, J.M.F.; Ferreira, L.E.V.V.D.C. Non-alcoholic Steatohepatitis: Comparison of Intestinal Microbiota between Different Metabolic Profiles. A Pilot Study. J. Gastrointest. Liver Dis. 2020, 29, 369–376. [Google Scholar] [CrossRef]
- Scarpellini, E.; Abenavoli, L.; Cassano, V.; Rinninella, E.; Sorge, M.; Capretti, F.; Rasetti, C.; Baroni, G.S.; Luzza, F.; Santori, P.; et al. The Apparent Asymmetrical Relationship Between Small Bowel Bacterial Overgrowth, Endotoxemia, and Liver Steatosis and Fibrosis in Cirrhotic and Non-Cirrhotic Patients: A Single-Center Pilot Study. Front. Med. 2022, 9, 872428. [Google Scholar] [CrossRef]
- Tziatzios, G.; Gkolfakis, P.; Papanikolaou, I.S.; Mathur, R.; Pimentel, M.; Damoraki, G.; Giamarellos-Bourboulis, E.J.; Dimitriadis, G.; Triantafyllou, K. High Prevalence of Small Intestinal Bacterial Overgrowth among Functional Dyspepsia Patients. Dig. Dis. 2021, 39, 382–390. [Google Scholar] [CrossRef]
- Miele, L.; Valenza, V.; La Torre, G.; Montalto, M.; Cammarota, G.; Ricci, R.; Mascianà, R.; Forgione, A.; Gabrieli, M.L.; Perotti, G.; et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology 2009, 49, 1877–1887. [Google Scholar] [CrossRef]
- Sabaté, J.-M.; Jouët, P.; Harnois, F.; Mechler, C.; Msika, S.; Grossin, M.; Coffin, B. High prevalence of small intestinal bacterial overgrowth in patients with morbid obesity: A contributor to severe hepatic steatosis. Obes. Surg. 2008, 18, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Fitriakusumah, Y.; Lesmana, C.R.A.; Bastian, W.P.; Jasirwan, C.O.M.; Hasan, I.; Simadibrata, M.; Kurniawan, J.; Sulaiman, A.S.; Gani, R.A. The role of Small Intestinal Bacterial Overgrowth (SIBO) in Non-alcoholic Fatty Liver Disease (NAFLD) patients evaluated using Controlled Attenuation Parameter (CAP) Transient Elastography (TE): A tertiary referral center experience. BMC Gastroenterol. 2019, 19, 43. [Google Scholar] [CrossRef] [PubMed]
- Juanola, O.; Ferrusquía-Acosta, J.; García-Villalba, R.; Zapater, P.; Magaz, M.; Marín, A.; Olivas, P.; Baiges, A.; Bellot, P.; Turon, F.; et al. Circulating levels of butyrate are inversely related to portal hypertension, endotoxemia, and systemic inflammation in patients with cirrhosis. FASEB J. 2019, 33, 11595–11605. [Google Scholar] [CrossRef] [PubMed]
- Su, T.; Lai, S.; Lee, A.; He, X.; Chen, S. Meta-analysis: Proton pump inhibitors moderately increase the risk of small intestinal bacterial overgrowth. J. Gastroenterol. 2018, 53, 27–36. [Google Scholar] [CrossRef]
- Cole, H.L.; Pennycook, S.; Hayes, P.C. The impact of proton pump inhibitor therapy on patients with liver disease. Aliment. Pharmacol. Ther. 2016, 44, 1213–1223. [Google Scholar] [CrossRef]
- Bakhshimoghaddam, F.; Alizadeh, M. Contribution of gut microbiota to nonalcoholic fatty liver disease: Pathways of mechanisms. Clin. Nutr. ESPEN 2021, 44, 61–68. [Google Scholar] [CrossRef]
- Pierantonelli, I.; Rychlicki, C.; Agostinelli, L.; Giordano, D.M.; Gaggini, M.; Fraumene, C.; Saponaro, C.; Manghina, V.; Sartini, L.; Mingarelli, E.; et al. Author Correction: Lack of NLRP3-inflammasome leads to gut-liver axis derangement, gut dysbiosis and a worsened phenotype in a mouse model of NAFLD. Sci. Rep. 2017, 7, 17568. [Google Scholar] [CrossRef]
- Wood, N.J. Dysbiosis driven by inflammasome deficiency exacerbates hepatic steatosis and governs rate of NAFLD progression. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 123. [Google Scholar] [CrossRef]
- Henao-Mejia, J.; Elinav, E.; Jin, C.; Hao, L.; Mehal, W.Z.; Strowig, T.; Thaiss, C.A.; Kau, A.L.; Eisenbarth, S.C.; Jurczak, M.J.; et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 2012, 482, 179–185. [Google Scholar] [CrossRef]
- Abu-Shanab, A.; Quigley, E.M.M. The role of the gut microbiota in nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 691–701. [Google Scholar] [CrossRef]
- Lin, E.A.; Barlow, G.M.; Mathur, R. The microbiome in non-alcoholic fatty liver disease: Associations and implications. Ann. Gastroenterol. 2014, 27, 181–183. [Google Scholar]
- Leite, G.; Morales, W.; Weitsman, S.; Celly, S.; Parodi, G.; Mathur, R.; Barlow, G.M.; Sedighi, R.; Millan, M.J.V.; Rezaie, A.; et al. The duodenal microbiome is altered in small intestinal bacterial overgrowth. PLoS ONE 2020, 15, e0234906. [Google Scholar] [CrossRef]
- Rehan, M.; Al-Bahadly, I.; Thomas, D.G.; Avci, E. Capsule robot for gut microbiota sampling using shape memory alloy spring. Int. J. Med. Robot. 2020, 16, 1–14. [Google Scholar] [CrossRef]
- Waimin, J.F.; Nejati, S.; Jiang, H.; Qiu, J.; Wang, J.; Verma, M.S.; Rahimi, R. Smart capsule for non-invasive sampling and studying of the gastrointestinal microbiome. RSC Adv. 2020, 10, 16313–16322. [Google Scholar] [CrossRef]
- Rezaie, A.; Buresi, M.; Lembo, A.; Lin, H.; McCallum, R.; Rao, S.; Schmulson, M.; Valdovinos, M.; Zakko, S.; Pimentel, M. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. Am. J. Gastroenterol. 2017, 112, 775–784. [Google Scholar] [CrossRef]
- Long, M.T.; Noureddin, M.; Lim, J.K. AGA Clinical Practice Update: Diagnosis and Management of Nonalcoholic Fatty Liver Disease in Lean Individuals: Expert Review. Gastroenterology 2022, 163, 764–774.e1. [Google Scholar] [CrossRef]
| Entire NAFLD Cohort (n = 95) | NAFL (n = 51) | NASH (n = 27) | NASH Cirrhosis (n = 17) | Healthy Controls (n = 30) | * p-Value | ** p-Value | |
|---|---|---|---|---|---|---|---|
| Age (years), mean (±SD) | 55 (10.9) | 54.7 (9.2) | 49.8 (11.8) | 63.9 (8.5) | 50.9 (14.6) | 0.103 | <0.001 |
| Gender, n (%) | 0.209 | 0.110 | |||||
| Female | 43 (45.3) | 27 (52.9) | 12 (44.4) | 4 (23.5) | 18 (60) | ||
| Male | 52 (54.7) | 24 (47.1) | 15 (55.6) | 13 (76.5) | 12 (40) | ||
| Weight (kg), mean (±SD) | 90.7 (19.1) | 89.2 (16.6) | 91.1 (21.1) | 94.4 (23.2) | 80.8 (19.5) | 0.016 | 0.630 |
| Height (cm), mean (±SD) | 176 (6.3) | 180 (8.6) | 171 (7.4) | 171 (11.5) | 171 (6.4) | 0.644 | 0.786 |
| Diabetes mellitus type 2, (%) | 0.295 | 0.391 | |||||
| Yes | 21 (22.1) | 14 (27.5) | 4 (14.8) | 3 (17.6) | 4 (13.3) | ||
| No | 74 (77.9) | 37 (72.5) | 23 (85.2) | 14 (82.4) | 26 (86.7) | ||
| PPI and/or H2RA intake, n (%) | 0.007 | 0.398 | |||||
| Yes | 19 (20) | 12 (23.5) | 3 (11.1) | 4 (23.5) | 0 (0) | ||
| No | 76 (80) | 39 (76.5) | 24 (88.9) | 13 (76.5) | 30 (100) |
| Bacterial Species, n (%) | Entire NAFLD Cohort, n (%) | Healthy Controls, n (%) |
|---|---|---|
| Escherichia coli | 10 (45.5) | - |
| Staphylococcus aureus | 4 (18.2) | - |
| Pseudomonas aeruginosa | 3 (13.6) | - |
| Enterococus faecalis | 2 (9.1) | - |
| Klebsiella pneumoniae | 1 (4.5) | - |
| Providencia alcalifaciens | 1 (4.5) | - |
| Acinetobacter spp. | 1 (4.5) | - |
| Enterobacter cloacae | - | 1 (100) |
| Total, n/N (%) | 22/95 (23.2%) | 1/30 (3.3%) |
| Concentration in Duodenal Fluid * | Entire NAFLD Cohort (n = 95) | NAFL (n = 51) | NASH (n = 27) | NASH-Cirrhosis (n = 17) | Healthy Controls (n = 30) | p-Value ** | p-Value *** |
|---|---|---|---|---|---|---|---|
| TNFα | 8.79 ± 0.86 | 7.80 ± 0.10 | 10.69 ± 2.89 | 8.57 ± 0.72 | 7.80 ± 0.10 | 0.533 | 0.345 |
| IL-1β | 7.63 ± 1.74 | 8.51 ± 2.92 | 8.69 ± 2.75 | 3.45 ± 1.11 | 6.26 ± 2.76 | 0.701 | 0.524 |
| IL-6 | 3.17 ± 0.04 | 3.22 ± 0.07 | 3.12 ± 0.04 | 3.12 ± 0.04 | 3.14 ± 0.10 | 0.648 | 0.417 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gkolfakis, P.; Tziatzios, G.; Leite, G.; Papanikolaou, I.S.; Xirouchakis, E.; Panayiotides, I.G.; Karageorgos, A.; Millan, M.J.; Mathur, R.; Weitsman, S.; et al. Prevalence of Small Intestinal Bacterial Overgrowth Syndrome in Patients with Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis: A Cross-Sectional Study. Microorganisms 2023, 11, 723. https://doi.org/10.3390/microorganisms11030723
Gkolfakis P, Tziatzios G, Leite G, Papanikolaou IS, Xirouchakis E, Panayiotides IG, Karageorgos A, Millan MJ, Mathur R, Weitsman S, et al. Prevalence of Small Intestinal Bacterial Overgrowth Syndrome in Patients with Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis: A Cross-Sectional Study. Microorganisms. 2023; 11(3):723. https://doi.org/10.3390/microorganisms11030723
Chicago/Turabian StyleGkolfakis, Paraskevas, Georgios Tziatzios, Gabriela Leite, Ioannis S. Papanikolaou, Elias Xirouchakis, Ioannis G. Panayiotides, Athanasios Karageorgos, Maria Jesus Millan, Ruchi Mathur, Stacy Weitsman, and et al. 2023. "Prevalence of Small Intestinal Bacterial Overgrowth Syndrome in Patients with Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis: A Cross-Sectional Study" Microorganisms 11, no. 3: 723. https://doi.org/10.3390/microorganisms11030723
APA StyleGkolfakis, P., Tziatzios, G., Leite, G., Papanikolaou, I. S., Xirouchakis, E., Panayiotides, I. G., Karageorgos, A., Millan, M. J., Mathur, R., Weitsman, S., Dimitriadis, G. D., Giamarellos-Bourboulis, E. J., Pimentel, M., & Triantafyllou, K. (2023). Prevalence of Small Intestinal Bacterial Overgrowth Syndrome in Patients with Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis: A Cross-Sectional Study. Microorganisms, 11(3), 723. https://doi.org/10.3390/microorganisms11030723










