Biosynthesis of Novel Tellurium Nanorods by Gayadomonas sp. TNPM15 Isolated from Mangrove Sediments and Assessment of Their Impact on Spore Germination and Ultrastructure of Phytopathogenic Fungi
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Isolation of TeNPs-Producing Bacteria
2.3. Bacterial Identification
2.4. Biosynthesis of Tellurium Nanoparticles
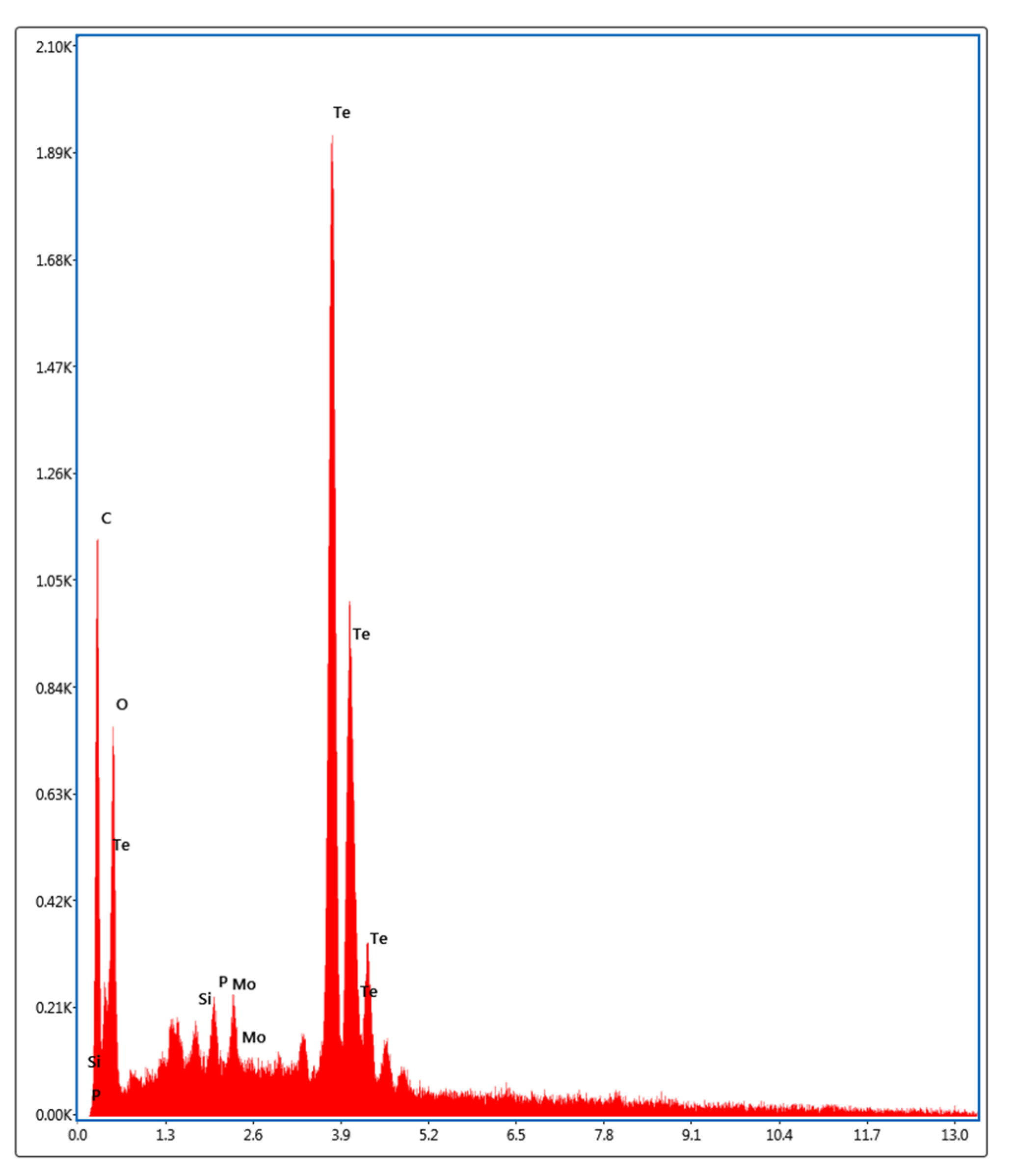
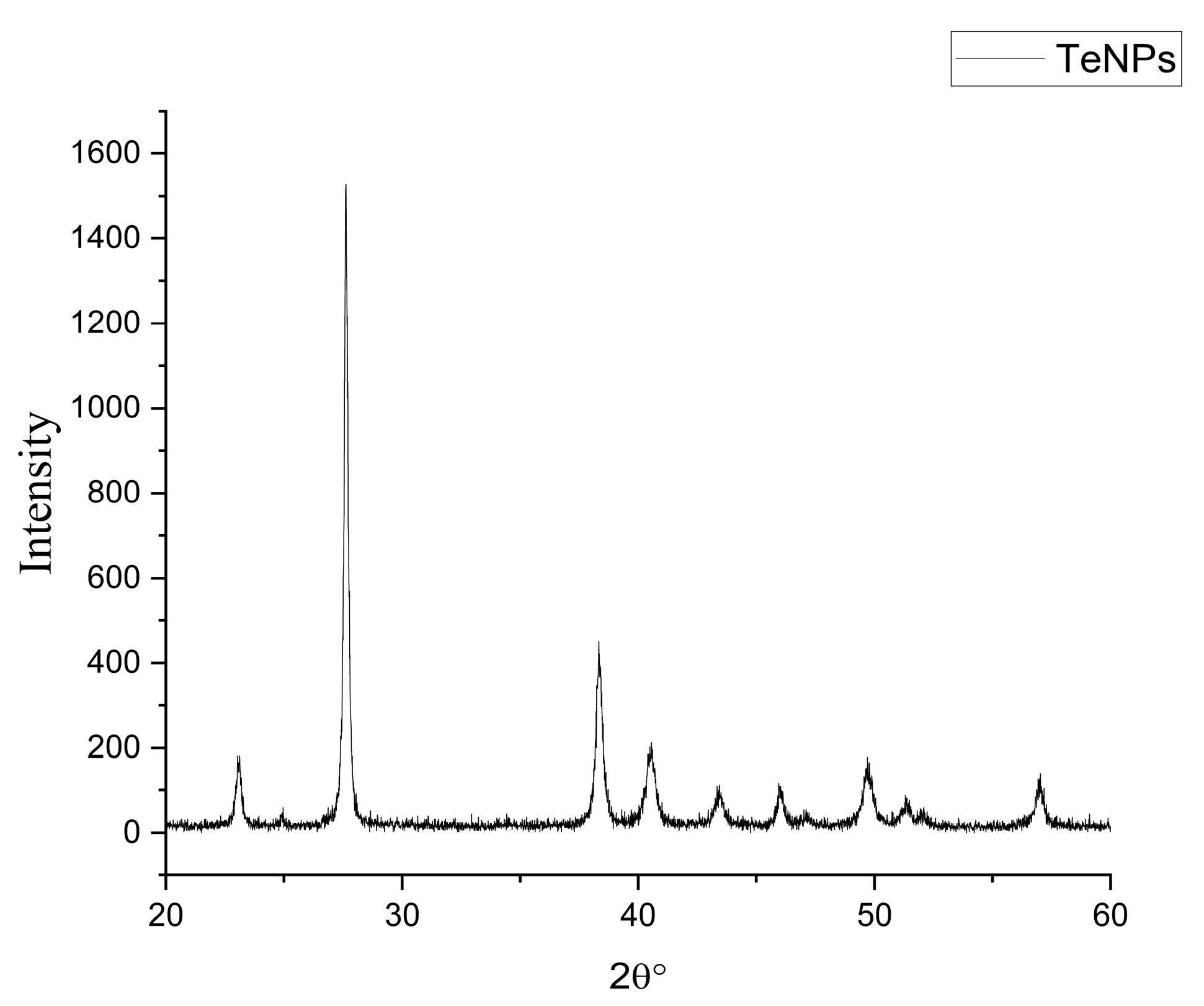
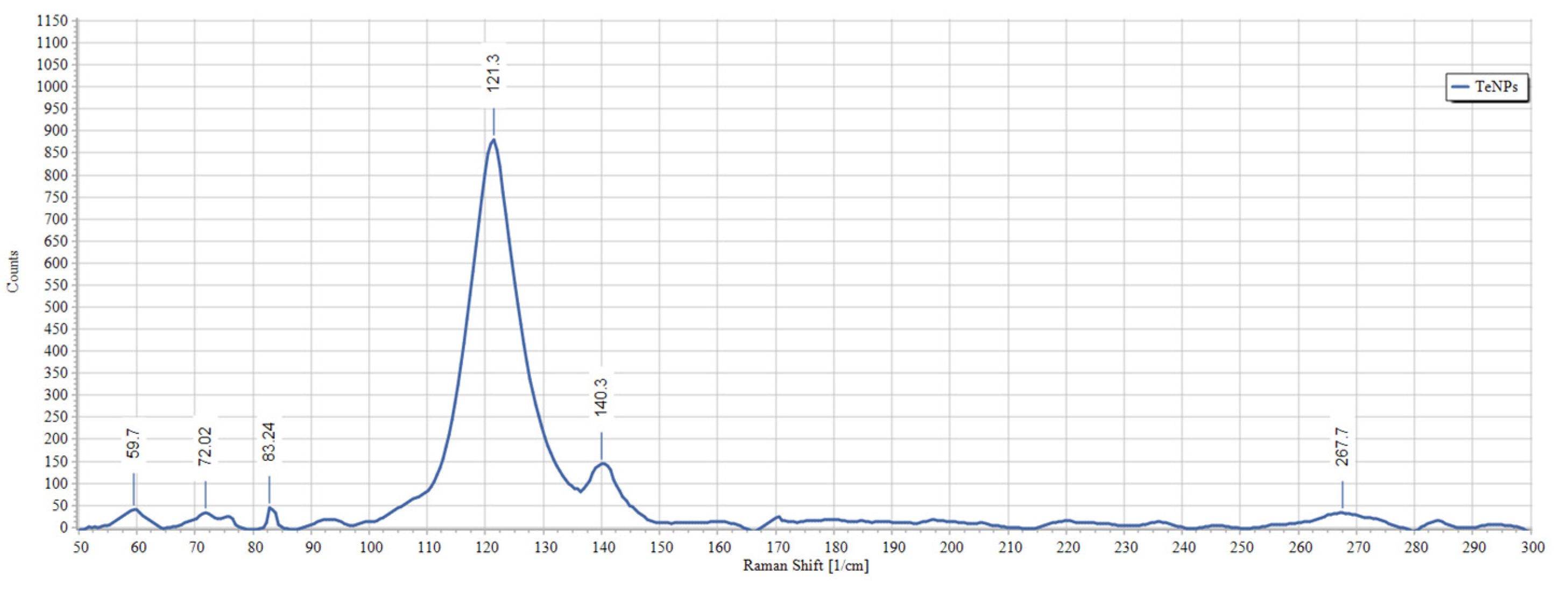
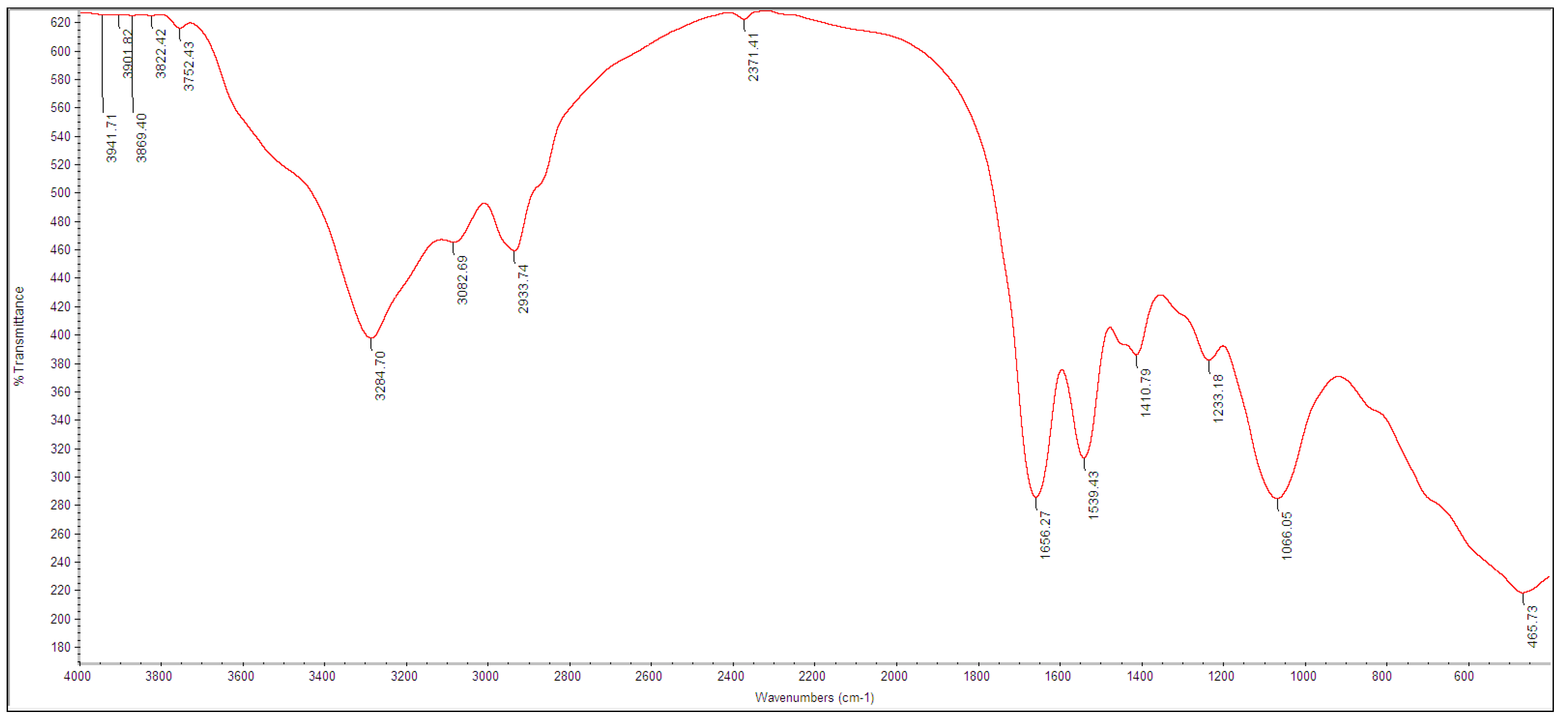
2.5. Characterization of Tellurium Nanoparticles
2.6. Fungal Phytopathogens
2.7. Germination Test
2.8. Spore Morphology and Ultrastructure Analysis
2.9. Leakage of Proteins and DNA
2.10. Impact of TeNPs on Plant Cell Wall-Degrading Enzymes (CWDEs)
2.11. Cytotoxicity Assay
2.12. Statistical Analysis
3. Results
3.1. Isolation of TeNPs-Producing Bacteria
3.2. Biosynthesis and Characterization of TeNPs
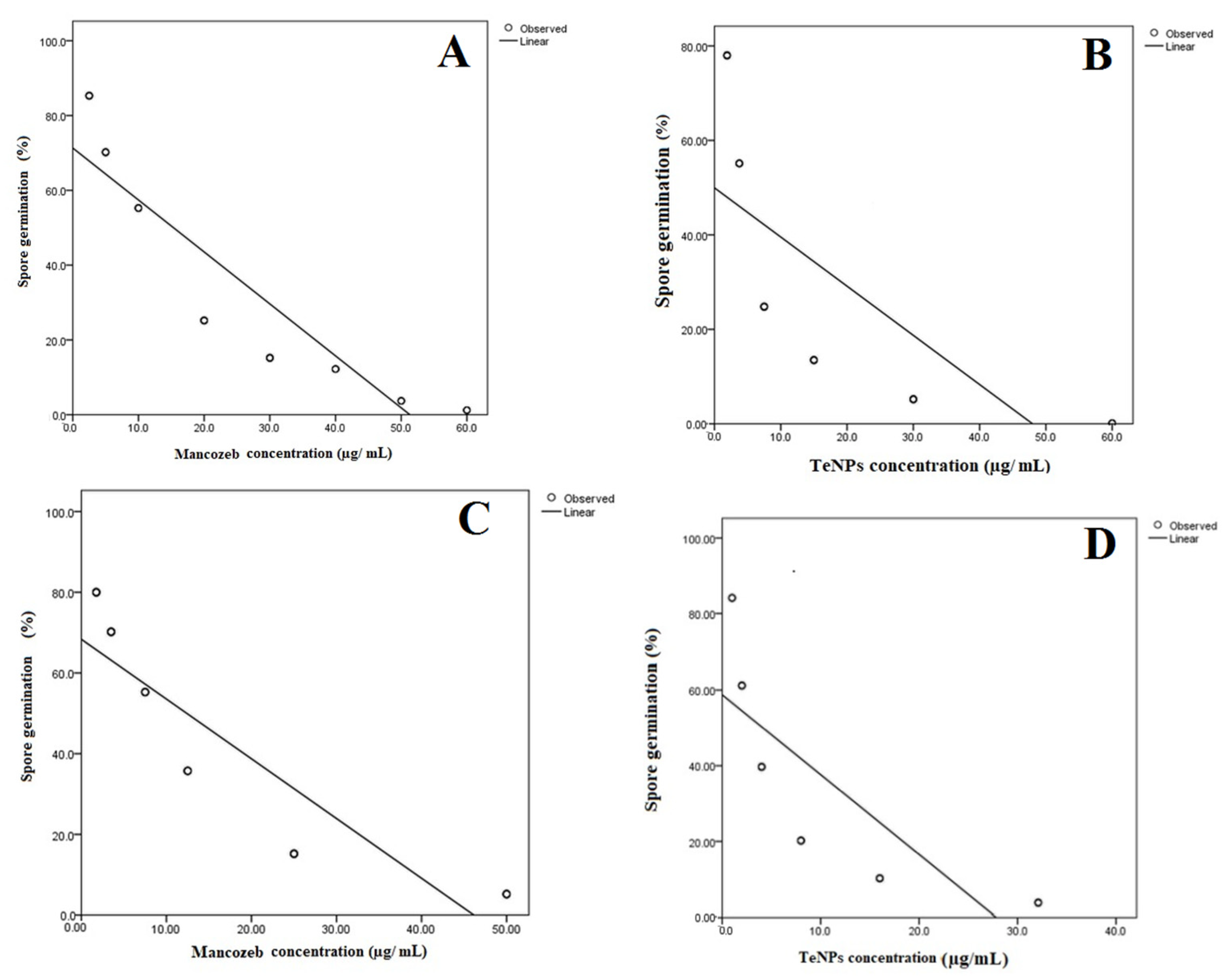
3.3. Germination Test
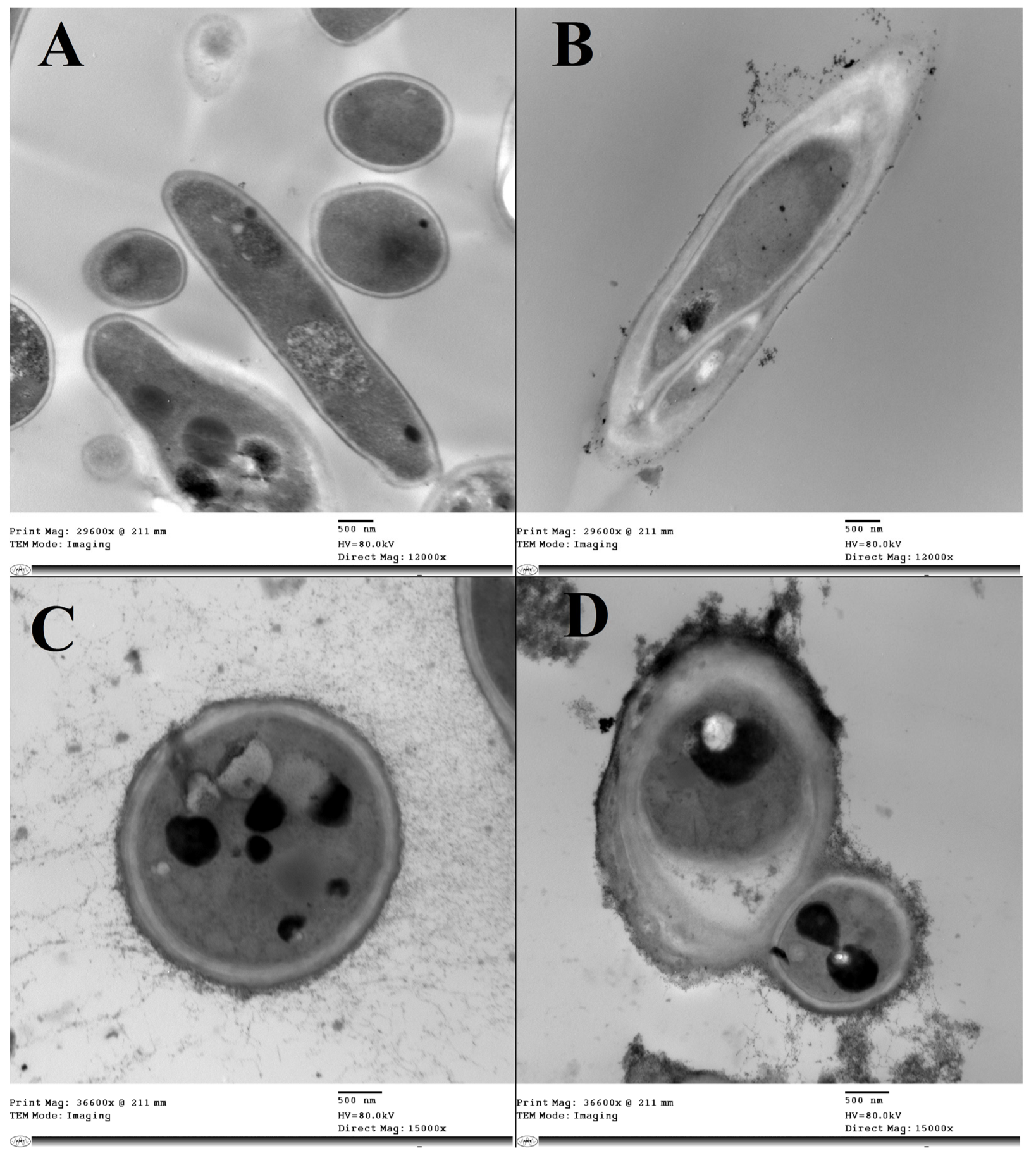
3.4. Spore Morphology and Ultrastructure Analysis
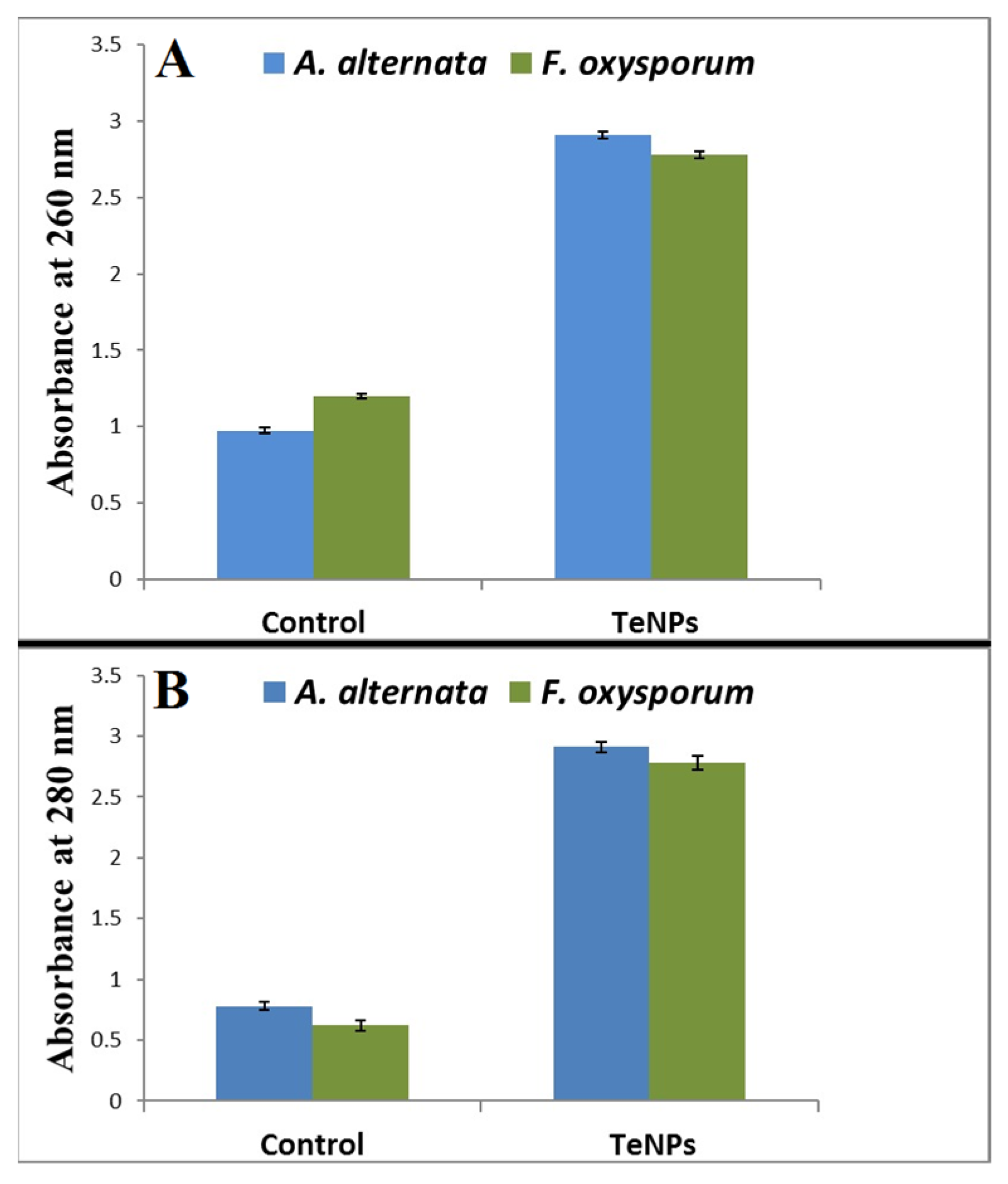
3.5. Leakage of Proteins and DNA
3.6. Impact of TeNPs on Plant Cell Wall-Degrading Enzymes
3.7. Cytotoxicity Assay
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paras; Yadav, K.; Kumar, P.; Teja, D.R.; Chakraborty, S.; Chakraborty, M.; Mohapatra, S.S.; Sahoo, A.; Chou, M.M.C.; Liang, C.-T.; et al. A Review on Low-Dimensional Nanomaterials: Nanofabrication, Characterization and Applications. Nanomatererials 2022, 13, 160. [Google Scholar] [CrossRef]
- Tang, A.; Ren, Q.; Wu, Y.; Wu, C.; Cheng, Y. Investigation into the Antibacterial Mechanism of Biogenic Tellurium Nanoparticles and Precursor Tellurite. Int. J. Mol. Sci. 2022, 23, 11697. [Google Scholar] [CrossRef]
- Domínguez-Álvarez, E.; Rácz, B.; Marć, M.A.; Nasim, M.J.; Szemerédi, N.; Viktorová, J.; Jacob, C.; Spengler, G. Selenium and Tellurium in the Development of Novel Small Molecules and Nanoparticles as Cancer Multidrug Resistance Reversal Agents. Drug Resist. Updat. 2022, 63, 100844. [Google Scholar] [CrossRef]
- Shahbaz, M.; Fatima, N.; Mashwani, Z.U.R.; Akram, A.; ul Haq, E.; Mehak, A.; Abasi, F.; Ajmal, M.; Yousaf, T.; Raja, N.I.; et al. Effect of Phytosynthesized Selenium and Cerium Oxide Nanoparticles on Wheat (Triticum aestivum L.) against Stripe Rust Disease. Molecules 2022, 27, 8149. [Google Scholar] [CrossRef]
- Salem, M.F.; Abd-Elraoof, W.A.; Tayel, A.A.; Alzuaibr, F.M.; Abonama, O.M. Antifungal Application of Biosynthesized Selenium Nanoparticles with Pomegranate Peels and Nanochitosan as Edible Coatings for Citrus Green Mold Protection. J. Nanobiotechnol. 2022, 20, 182. [Google Scholar] [CrossRef]
- Malandrakis, A.A.; Kavroulakis, N.; Chrysikopoulos, C.V. Zinc Nanoparticles: Mode of Action and Efficacy against Boscalid-Resistant Alternaria alternata Isolates. Sci. Total Environ. 2022, 829, 154638. [Google Scholar] [CrossRef]
- Sahin, F.; Celik, N.; Ceylan, A.; Ruzi, M.; Onses, M.S. One-Step Green Fabrication of Antimicrobial Surfaces via in Situ Growth of Copper Oxide Nanoparticles. ACS Omega 2022, 7, 26504–26513. [Google Scholar] [CrossRef]
- Ibarra-Laclette, E.; Blaz, J.; Pérez-Torres, C.A.; Villafán, E.; Lamelas, A.; Rosas-Saito, G.; Ibarra-Juárez, L.A.; García-ávila, C.D.J.; Martínez-Enriquez, A.I.; Pariona, N. Antifungal Effect of Copper Nanoparticles against Fusarium kuroshium, an Obligate Symbiont of Euwallacea kuroshio Ambrosia Beetle. J. Fungi 2022, 8, 347. [Google Scholar] [CrossRef]
- Kamel, S.M.; Elgobashy, S.F.; Omara, R.I.; Derbalah, A.S.; Abdelfatah, M.; El-Shaer, A.; Al-Askar, A.A.; Abdelkhalek, A.; Abd-Elsalam, K.A.; Essa, T.; et al. Antifungal Activity of Copper Oxide Nanoparticles against Root Rot Disease in Cucumber. J. Fungi 2022, 8, 911. [Google Scholar] [CrossRef]
- Da Silva, M.A.; García-Martínez, J.C.; Páez, P.L.; Paraje, M.G. Fungicidal and Antibiofilm Activities of Gold Nanoparticles on Candida tropicalis. Nanomedicine 2022, 17, 1663–1676. [Google Scholar] [CrossRef]
- Soltani Nejad, M.; Samandari Najafabadi, N.; Aghighi, S.; Pakina, E.; Zargar, M. Evaluation of Phoma Sp. Biomass as an Endophytic Fungus for Synthesis of Extracellular Gold Nanoparticles with Antibacterial and Antifungal Properties. Molecules 2022, 27, 1181. [Google Scholar] [CrossRef]
- Zambonino, M.C.; Quizhpe, E.M.; Jaramillo, F.E.; Rahman, A.; Vispo, N.S.; Jeffryes, C.; Dahoumane, S.A. Green Synthesis of Selenium and Tellurium Nanoparticles: Current Trends, Biological Properties and Biomedical Applications. Int. J. Mol. Sci. 2021, 22, 989. [Google Scholar] [CrossRef]
- Ashraf, M.W.; Haider, S.I.; Solangi, A.R.; Memon, A.F. 6 Toxicity of tellurium and its compounds. In Tellurium Chemistry; Banik, B.K., Bajpai, S., Eds.; De Gruyter: Berlin, Germany; Boston, MA, USA, 2022. [Google Scholar] [CrossRef]
- Espinosa-Ortiz, E.J.; Rene, E.R.; Guyot, F.; van Hullebusch, E.D.; Lens, P.N.L. Biomineralization of Tellurium and Selenium-Tellurium Nanoparticles by the White-Rot Fungus Phanerochaete chrysosporium. Int. Biodeterior. Biodegrad. 2017, 124, 258–266. [Google Scholar] [CrossRef]
- Zwar, I.P.; Trotta, C.D.V.; Ziotti, A.B.S.; Neto, M.L.; Araújo, W.L.; de Melo, I.S.; Ottoni, C.A.; de Souza, A.O. Biosynthesis of Silver Nanoparticles Using Actinomycetes, Phytotoxicity on Rice Seeds, and Potential Application in the Biocontrol of Phytopathogens. J. Basic Microbiol. 2023, 63, 64–74. [Google Scholar] [CrossRef]
- Nguyen, D.T.C.; Van Tran, T.; Nguyen, T.T.T.; Nguyen, D.H.; Alhassan, M.; Lee, T. New Frontiers of Invasive Plants for Biosynthesis of Nanoparticles towards Biomedical Applications: A Review. Sci. Total Environ. 2023, 857, 159278. [Google Scholar] [CrossRef]
- Sivakumar, S.R.; Manimaran, K.; Govindasamy, M.; Alzahrani, F.M.; Alsaiari, N.S. Green Synthesis and Characterization of CuO Nanoparticles Using Halymenia dilatata Extract and Its Evaluation of Antimicrobial, Anticancer Activity. Biomass Convers. Biorefinery 2023, 1, 1–10. [Google Scholar] [CrossRef]
- Liang, X.; Perez, M.A.M.J.; Nwoko, K.C.; Egbers, P.; Feldmann, J.; Csetenyi, L.; Gadd, G.M. Fungal Formation of Selenium and Tellurium Nanoparticles. Appl. Microbiol. Biotechnol. 2019, 103, 7241–7259. [Google Scholar] [CrossRef]
- Abo Elsoud, M.M.; Al-Hagar, O.E.A.; Abdelkhalek, E.S.; Sidkey, N.M. Synthesis and Investigations on Tellurium Myconanoparticles. Biotechnol. Rep. 2018, 18, e00247. [Google Scholar] [CrossRef]
- Ao, B.; He, F.; Lv, J.; Tu, J.; Tan, Z.; Jiang, H.; Shi, X.; Li, J.; Hou, J.; Hu, Y.; et al. Green Synthesis of Biogenetic Te(0) Nanoparticles by High Tellurite Tolerance Fungus Mortierella Sp. AB1 with Antibacterial Activity. Front. Microbiol. 2022, 13, 3973. [Google Scholar] [CrossRef]
- Lin, Z.H.; Lee, C.H.; Chang, H.Y.; Chang, H.T. Antibacterial Activities of Tellurium Nanomaterials. Chem. Asian J. 2012, 7, 930–934. [Google Scholar] [CrossRef]
- Borghese, R.; Baccolini, C.; Francia, F.; Sabatino, P.; Turner, R.J.; Zannoni, D. Reduction of Chalcogen Oxyanions and Generation of Nanoprecipitates by the Photosynthetic Bacterium Rhodobacter capsulatus. J. Hazard. Mater. 2014, 269, 24–30. [Google Scholar] [CrossRef]
- Borghese, R.; Brucale, M.; Fortunato, G.; Lanzi, M.; Mezzi, A.; Valle, F.; Cavallini, M.; Zannoni, D. Extracellular Production of Tellurium Nanoparticles by the Photosynthetic Bacterium Rhodobacter capsulatus. J. Hazard. Mater. 2016, 309, 202–209. [Google Scholar] [CrossRef]
- Beleneva, I.A.; Kharchenko, U.V.; Kukhlevsky, A.D.; Boroda, A.V.; Izotov, N.V.; Gnedenkov, A.S.; Egorkin, V.S. Biogenic Synthesis of Selenium and Tellurium Nanoparticles by Marine Bacteria and Their Biological Activity. World J. Microbiol. Biotechnol. 2022, 38, 188. [Google Scholar] [CrossRef]
- Presentato, A.; Piacenza, E.; Darbandi, A.; Anikovskiy, M.; Cappelletti, M.; Zannoni, D.; Turner, R.J. Assembly, Growth and Conductive Properties of Tellurium Nanorods Produced by Rhodococcus aetherivorans BCP1. Sci. Rep. 2018, 8, 3923. [Google Scholar] [CrossRef]
- Mirjani, R.; Faramarzi, M.A.; Sharifzadeh, M.; Setayesh, N.; Khoshayand, M.R.; Shahverdi, A.R. Biosynthesis of Tellurium Nanoparticles by Lactobacillus plantarum and the Effect of Nanoparticle-Enriched Probiotics on the Lipid Profiles of Mice. IET Nanobiotechnol. 2015, 9, 300–305. [Google Scholar] [CrossRef]
- Ghosh, S.; Sarkar, B.; Kaushik, A.; Mostafavi, E. Nanobiotechnological Prospects of Probiotic Microflora: Synthesis, Mechanism, and Applications. Sci. Total Environ. 2022, 838, 156212. [Google Scholar] [CrossRef]
- Ramos-Ruiz, A.; Field, J.A.; Wilkening, J.V.; Sierra-Alvarez, R. Recovery of Elemental Tellurium Nanoparticles by the Reduction of Tellurium Oxyanions in a Methanogenic Microbial Consortium. Environ. Sci. Technol. 2016, 50, 1492–1500. [Google Scholar] [CrossRef]
- Ancheeva, E.; Daletos, G.; Proksch, P. Lead Compounds from Mangrove-Associated Microorganisms. Mar. Drugs 2018, 16, 319. [Google Scholar] [CrossRef]
- Debasmita, D.; Sourav, G.; Srimoyee, B. A Review of Metal Resistance Mechanisms by Mangrove Bacteria. Res. J. Biotechnol. 2022, 17, 209–215. [Google Scholar] [CrossRef]
- Alongi, D.M.; Pfitzner, J.; Trott, L.A.; Tirendi, F.; Dixon, P.; Klumpp, D.W. Rapid Sediment Accumulation and Microbial Mineralization in Forests of the Mangrove Kandelia candel in the Jiulongjiang Estuary, China. Estuar. Coast. Shelf Sci. 2005, 63, 605–618. [Google Scholar] [CrossRef]
- Holguin, G.; Vazquez, P.; Bashan, Y. The Role of Sediment Microorganisms in the Productivity, Conservation, and Rehabilitation of Mangrove Ecosystems: An Overview. Biol. Fertil. Soils 2001, 33, 265–278. [Google Scholar] [CrossRef]
- Zhu, D.H.; Song, Q.L.; Nie, F.H.; Wei, W.; Chen, M.M.; Zhang, M.; Lin, H.Y.; Kang, D.J.; Chen, Z.B.; Hay, A.G.; et al. Effects of Environmental and Spatial Variables on Bacteria in Zhanjiang Mangrove Sediments. Curr. Microbiol. 2022, 79, 97. [Google Scholar] [CrossRef]
- Li, K.; Chen, S.; Pang, X.; Cai, J.; Zhang, X.; Liu, Y.; Zhu, Y.; Zhou, X. Natural Products from Mangrove Sediments-Derived Microbes: Structural Diversity, Bioactivities, Biosynthesis, and Total Synthesis. Eur. J. Med. Chem. 2022, 230, 114117. [Google Scholar] [CrossRef]
- Gonzalez-Acosta, B.; Bashan, Y.; Hernandez-Saavedra, N.Y.; Ascencio, F.; De La Cruz-Agüero, G. Seasonal Seawater Temperature as the Major Determinant for Populations of Culturable Bacteria in the Sediments of an Intact Mangrove in an Arid Region. FEMS Microbiol. Ecol. 2006, 55, 311–321. [Google Scholar] [CrossRef]
- Mishra, R.R.; Prajapati, S.; Das, J.; Dangar, T.K.; Das, N.; Thatoi, H. Reduction of Selenite to Red Elemental Selenium by Moderately Halotolerant Bacillus megaterium Strains Isolated from Bhitarkanika Mangrove Soil and Characterization of Reduced Product. Chemosphere 2011, 84, 1231–1237. [Google Scholar] [CrossRef]
- Farias, P.; Francisco, R.; Morais, P.V. Potential of Tellurite Resistance in Heterotrophic Bacteria from Mining Environments. iScience 2022, 25, 104566. [Google Scholar] [CrossRef]
- Wang, Z.; Bu, Y.; Zhao, Y.; Zhang, Z.; Liu, L.; Zhou, H. Morphology-Tunable Tellurium Nanomaterials Produced by the Tellurite-Reducing Bacterium Lysinibacillus Sp. ZYM-1. Environ. Sci. Pollut. Res. 2018, 25, 20756–20768. [Google Scholar] [CrossRef]
- Horiike, T.; Otsuka, O.; Tanaka, Y.; Terahara, T.; Imada, C.; Yamashita, M. Diversity of Salt-Tolerant Tellurate-Reducing Bacteria in a Marine Environment. J. Gen. Appl. Microbiol. 2019, 65, 246–253. [Google Scholar] [CrossRef]
- Wang, Z.; Yi, X.; Liu, Y.; Zhou, H. Complete Genome Sequence of a Tellurate Reducing Bacteria Sporosarcina Sp. Te-1 Isolated from Bohai Sea. Mar. Genom. 2021, 60, 100888. [Google Scholar] [CrossRef]
- Soda, S.; Ma, W.; Kuroda, M.; Nishikawa, H.; Zhang, Y.; Ike, M. Characterization of Moderately Halotolerant Selenate- and Tellurite-Reducing Bacteria Isolated from Brackish Areas in Osaka. Biosci. Biotechnol. Biochem. 2018, 82, 173–181. [Google Scholar] [CrossRef]
- Davies, C.R.; Wohlgemuth, F.; Young, T.; Violet, J.; Dickinson, M.; Sanders, J.W.; Vallieres, C.; Avery, S.V. Evolving Challenges and Strategies for Fungal Control in the Food Supply Chain. Fungal Biol. Rev. 2021, 36, 15–26. [Google Scholar] [CrossRef]
- Husaini, A.M.; Sakina, A.; Cambay, S.R. Host-Pathogen Interaction in Fusarium oxysporum Infections: Where Do We Stand? Mol. Plant-Microbe Interact. 2018, 31, 889–898. [Google Scholar] [CrossRef]
- Mallik, S.K.; Shahi, N.; Joshi, N.; Pant, K.; Kala, K.; Chandra, S.; Sarma, D. The Emergence of Zoonotic Fusarium oxysporum Infection in Captive-Reared Fingerlings of Golden Mahseer, Tor putitora (Hamilton, 1822) from the Central Himalayan Region of India. Transbound. Emerg. Dis. 2020, 67, 555–563. [Google Scholar] [CrossRef]
- Arasaki, R.; Tanaka, S.; Okawa, K.; Tanaka, Y.; Inoue, T.; Kobayashi, S.; Ito, A.; Maruyama-Inoue, M.; Yamaguchi, T.; Muraosa, Y.; et al. Endophthalmitis Outbreak Caused by Fusarium oxysporum after Cataract Surgery. Am. J. Ophthalmol. Case Rep. 2022, 26, 101397. [Google Scholar] [CrossRef]
- Badali, H.; Cañete-Gibas, C.; Patterson, H.; Sanders, C.; Mermella, B.; Garcia, V.; Mele, J.; Fan, H.; Wiederhold, N.P. In Vitro Activity of Olorofim against Clinical Isolates of the Fusarium oxysporum and Fusarium solani Species Complexes. Mycoses 2021, 64, 748–752. [Google Scholar] [CrossRef]
- Martinko, K.; Ivanković, S.; Lazarević, B.; Ðermić, E.; Ðermić, D. Control of Early Blight Fungus (Alternaria alternata) in Tomato by Boric and Phenylboronic Acid. Antibiotics 2022, 11, 320. [Google Scholar] [CrossRef]
- Kgatle, M.G.; Flett, B.; Truter, M.; Aveling, T.A.S. Control of Alternaria Leaf Blight Caused by Alternaria alternata on Sunflower Using Fungicides and Bacillus amyloliquefaciens. Crop Prot. 2020, 132, 105146. [Google Scholar] [CrossRef]
- Ghosh, R.; Barman, S.; Khatun, J.; Mandal, N.C. Biological Control of Alternaria alternata Causing Leaf Spot Disease of Aloe Vera Using Two Strains of Rhizobacteria. Biol. Control 2016, 97, 102–108. [Google Scholar] [CrossRef]
- De Mers, M. Alternaria alternata as Endophyte and Pathogen. Microbiology 2022, 168, 001153. [Google Scholar] [CrossRef]
- Alastruey-izquierdo, A.; Cuesta, I.; Ros, L.; Mellado, E.; Rodriguez-tudela, J.L. Antifungal Susceptibility Profile of Clinical Alternaria spp. Identified by Molecular Methods. J. Antimicrob. Chemother. 2011, 66, 2585–2587. [Google Scholar] [CrossRef]
- Kim, G.; Yoo, S.J.; Yoo, J.R.; Seo, K.B. The First Case Report of Thorn-Induced Alternaria alternata Infection of the Hand in an Immunocompetent Host. BMC Infect. Dis. 2022, 22, 304. [Google Scholar] [CrossRef]
- Hattab, Z.; Ben Lasfar, N.; Abid, M.; Bellazreg, F.; Fathallah, A.; Hachfi, W.; Letaief, A. Alternaria alternata Infection Causing Rhinosinusitis and Orbital Involvement in an Immunocompetent Patient. New Microbes New Infect. 2019, 32, 100561. [Google Scholar] [CrossRef]
- Medina Cruz, D.; Tien-Street, W.; Zhang, B.; Huang, X.; Vernet Crua, A.; Nieto-Argüello, A.; Cholula-Díaz, J.L.; Martínez, L.; Huttel, Y.; González, M.U.; et al. Citric Juice-Mediated Synthesis of Tellurium Nanoparticles with Antimicrobial and Anticancer Properties. Green Chem. 2019, 21, 1982–1998. [Google Scholar] [CrossRef]
- Shah, V.; Medina-Cruz, D.; Vernet-Crua, A.; Truong, L.B.; Sotelo, E.; Mostafavi, E.; González, M.U.; García-Martín, J.M.; Cholula-Díaz, J.L.; Webster, T.J. Pepper-Mediated Green Synthesis of Selenium and Tellurium Nanoparticles with Antibacterial and Anticancer Potential. J. Funct. Biomater. 2022, 14, 24. [Google Scholar] [CrossRef]
- Morena, A.G.; Bassegoda, A.; Hoyo, J.; Tzanov, T. Hybrid Tellurium-Lignin Nanoparticles with Enhanced Antibacterial Properties. ACS Appl. Mater. Interfaces 2021, 13, 14885–14893. [Google Scholar] [CrossRef]
- Zonaro, E.; Lampis, S.; Turner, R.J.; Junaid, S.; Vallini, G. Biogenic Selenium and Tellurium Nanoparticles Synthesized by Environmental Microbial Isolates Efficaciously Inhibit Bacterial Planktonic Cultures and Biofilms. Front. Microbiol. 2015, 6, 584. [Google Scholar] [CrossRef]
- Zare, B.; Sepehrizadeh, Z.; Faramarzi, M.A.; Soltany-Rezaee-Rad, M.; Rezaie, S.; Shahverdi, A.R. Antifungal Activity of Biogenic Tellurium Nanoparticles against Candida albicans and Its Effects on Squalene monooxygenase Gene Expression. Biotechnol. Appl. Biochem. 2014, 61, 395–400. [Google Scholar] [CrossRef]
- Yoon, S.H.; Ha, S.M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A Taxonomically United Database of 16S RRNA Gene Sequences and Whole-Genome Assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [CrossRef]
- Khalil, N.M.; Abd El-Ghany, M.N.; Rodríguez-Couto, S. Antifungal and Anti-Mycotoxin Efficacy of Biogenic Silver Nanoparticles Produced by Fusarium chlamydosporum and Penicillium chrysogenum at Non-Cytotoxic Doses. Chemosphere 2019, 218, 477–486. [Google Scholar] [CrossRef]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Liang, J.-B.; Chen, Y.-Q.; Lan, C.-Y.; Tam, N.F.Y.; Zan, Q.-J.; Huang, L.-N. Recovery of Novel Bacterial Diversity from Mangrove Sediment. Mar. Biol. 2007, 150, 739–747. [Google Scholar] [CrossRef]
- Farahat, M.G. Enhancement of β-Cyclodextrin Production and Fabrication of Edible Antimicrobial Films Incorporated with Clove Essential Oil/β-Cyclodextrin Inclusion Complex. Microbiol. Biotechnol. Lett 2020, 48, 12–23. [Google Scholar] [CrossRef]
- Nguyen, V.K.; Choi, W.; Ha, Y.; Gu, Y.; Lee, C.; Park, J.; Jang, G.; Shin, C.; Cho, S. Microbial Tellurite Reduction and Production of Elemental Tellurium Nanoparticles by Novel Bacteria Isolated from Wastewater. J. Ind. Eng. Chem. 2019, 78, 246–256. [Google Scholar] [CrossRef]
- Baesman, S.M.; Stolz, J.F.; Kulp, T.R.; Oremland, R.S. Enrichment and Isolation of Bacillus beveridgei sp. Nov., a Facultative Anaerobic Haloalkaliphile from Mono Lake, California, That Respires Oxyanions of Tellurium, Selenium, and Arsenic. Extremophiles 2009, 13, 695–705. [Google Scholar] [CrossRef]
- Lloyd-Jones, G.; Williamson, W.M.; Slootweg, T. The Te-Assay: A Black and White Method for Environmental Sample Pre-Screening Exploiting Tellurite Reduction. J. Microbiol. Methods 2006, 67, 549–556. [Google Scholar] [CrossRef]
- Chien, C.C.; Jiang, M.H.; Tsai, M.R.; Chien, C.C. Isolation and Characterization of an Environmental Cadmium- and Tellurite-Resistant Pseudomonas Strain. Environ. Toxicol. Chem. 2011, 30, 2202–2207. [Google Scholar] [CrossRef]
- Amoozegar, M.A.; Ashengroph, M.; Malekzadeh, F.; Reza Razavi, M.; Naddaf, S.; Kabiri, M. Isolation and Initial Characterization of the Tellurite Reducing Moderately Halophilic Bacterium, Salinicoccus Sp. Strain QW6. Microbiol. Res. 2008, 163, 456–465. [Google Scholar] [CrossRef]
- Gutierrez-Monsalve, J.A.; Mosquera, S.; González-Jaramillo, L.M.; Mira, J.J.; Villegas-Escobar, V. Effective Control of Black Sigatoka Disease Using a Microbial Fungicide Based on Bacillus subtilis EA-CB0015 Culture. Biol. Control 2015, 87, 39–46. [Google Scholar] [CrossRef]
- Araya-Castro, K.; Chao, T.C.; Durán-Vinet, B.; Cisternas, C.; Ciudad, G.; Rubilar, O. Green Synthesis of Copper Oxide Nanoparticles Using Protein Fractions from an Aqueous Extract of Brown Algae Macrocystis pyrifera. Processes 2020, 9, 78. [Google Scholar] [CrossRef]
- Labeeb, M.; Badr, A.; Haroun, S.A.; Mattar, M.Z.; El-kholy, A.S. Ultrastructural and Molecular Implications of Ecofriendly Made Silver Nanoparticles Treatments in Pea (Pisum sativum L.). J. Genet. Eng. Biotechnol. 2022, 20, 5. [Google Scholar] [CrossRef]
- Rizwana, H.; Alwhibi, M.S.; Al-Judaie, R.A.; Aldehaish, H.A.; Alsaggabi, N.S. Sunlight-Mediated Green Synthesis of Silver Nanoparticles Using the Berries of Ribes rubrum (Red Currants): Characterisation and Evaluation of Their Antifungal and Antibacterial Activities. Molecules 2022, 27, 2186. [Google Scholar] [CrossRef]
- Barabadi, H.; Kobarfard, F.; Vahidi, H. Biosynthesis and Characterization of Biogenic Tellurium Nanoparticles by Using Penicillium chrysogenum PTCC 5031: A Novel Approach in Gold Biotechnology. Iran. J. Pharm. Res. IJPR 2018, 17, 87. [Google Scholar]
- Singh, D.; Rathod, V.; Ninganagouda, S.; Hiremath, J.; Singh, A.K.; Mathew, J. Optimization and Characterization of Silver Nanoparticle by Endophytic Fungi Penicillium sp. Isolated from Curcuma longa (Turmeric) and Application Studies against MDR E. coli and S. aureus. Bioinorg. Chem. Appl. 2014, 2014, 408021. [Google Scholar] [CrossRef]
- Zonaro, E.; Piacenza, E.; Presentato, A.; Monti, F.; Dell’Anna, R.; Lampis, S.; Vallini, G. Ochrobactrum sp. MPV1 from a Dump of Roasted Pyrites Can Be Exploited as Bacterial Catalyst for the Biogenesis of Selenium and Tellurium Nanoparticles. Microb. Cell Fact. 2017, 16, 215. [Google Scholar] [CrossRef]
- Arab, F.; Mousavi-Kamazani, M.; Salavati-Niasari, M. Facile Sonochemical Synthesis of Tellurium and Tellurium Dioxide Nanoparticles: Reducing Te(IV) to Te via Ultrasonic Irradiation in Methanol. Ultrason. Sonochem. 2017, 37, 335–343. [Google Scholar] [CrossRef]
- Song, J.M.; Lin, Y.Z.; Zhan, Y.J.; Tian, Y.C.; Liu, G.; Yu, S.H. Superlong High-Quality Tellurium Nanotubes: Synthesis, Characterization, and Optical Property. Cryst. Growth Des. 2008, 8, 1902–1908. [Google Scholar] [CrossRef]
- Zare, B.; Faramarzi, M.A.; Sepehrizadeh, Z.; Shakibaie, M.; Rezaie, S.; Shahverdi, A.R. Biosynthesis and Recovery of Rod-Shaped Tellurium Nanoparticles and Their Bactericidal Activities. Mater. Res. Bull. 2012, 47, 3719–3725. [Google Scholar] [CrossRef]
- Liu, J.W.; Chen, F.; Zhang, M.; Qi, H.; Zhang, C.L.; Yu, S.H. Rapid Microwave-Assisted Synthesis of Uniform Ultralong Te Nanowires, Optical Property, and Chemical Stability. Langmuir 2010, 26, 11372–11377. [Google Scholar] [CrossRef]
- Manikandan, M.; Dhanuskodi, S.; Maheswari, N.; Muralidharan, G.; Revathi, C.; Rajendra Kumar, R.T.; Mohan Rao, G. High Performance Supercapacitor and Non-Enzymatic Hydrogen Peroxide Sensor Based on Tellurium Nanoparticles. Sens. Bio-Sensing Res. 2017, 13, 40–48. [Google Scholar] [CrossRef]
- Najimi, S.; Shakibaie, M.; Jafari, E.; Ameri, A.; Rahimi, N.; Forootanfar, H.; Yazdanpanah, M.; Rahimi, H.R. Acute and Subacute Toxicities of Biogenic Tellurium Nanorods in Mice. Regul. Toxicol. Pharmacol. 2017, 90, 222–230. [Google Scholar] [CrossRef]
- Alonso-Fernandes, E.; Fernández-Llamosas, H.; Cano, I.; Serrano-Pelejero, C.; Castro, L.; Díaz, E.; Carmona, M. Enhancing Tellurite and Selenite Bioconversions by Overexpressing a Methyltransferase from Aromatoleum sp. CIB. Microb. Biotechnol. 2022, 1–16. [Google Scholar] [CrossRef]
- Gomez-Gomez, B.; Corte-Rodríguez, M.; Perez-Corona, M.T.; Bettmer, J.; Montes-Bayón, M.; Madrid, Y. Combined Single Cell and Single Particle ICP-TQ-MS Analysis to Quantitatively Evaluate the Uptake and Biotransformation of Tellurium Nanoparticles in Bacteria. Anal. Chim. Acta 2020, 1128, 116–128. [Google Scholar] [CrossRef]
- Srivastava, P.; Nikhil, E.V.R.; Bragança, J.M.; Kowshik, M. Anti-Bacterial TeNPs Biosynthesized by Haloarcheaon Halococcus salifodinae BK3. Extremophiles 2015, 19, 875–884. [Google Scholar] [CrossRef]
- Mohanty, A.; Kathawala, M.H.; Zhang, J.; Chen, W.N.; Loo, J.S.C.; Kjelleberg, S.; Yang, L.; Cao, B. Biogenic Tellurium Nanorods as a Novel Antivirulence Agent Inhibiting Pyoverdine Production in Pseudomonas aeruginosa. Biotechnol. Bioeng. 2014, 111, 858–865. [Google Scholar] [CrossRef]
- Elakraa, A.A.; Salem, S.S.; El-Sayyad, G.S.; Attia, M.S. Cefotaxime Incorporated Bimetallic Silver-Selenium Nanoparticles: Promising Antimicrobial Synergism, Antibiofilm Activity, and Bacterial Membrane Leakage Reaction Mechanism. RSC Adv. 2022, 12, 26603–26619. [Google Scholar] [CrossRef]
- Parveen, A.; Yalagatti, M.S.; Abbaraju, V.; Deshpande, R. Emphasized Mechanistic Antimicrobial Study of Biofunctionalized Silver Nanoparticles on Model Proteus mirabilis. J. Drug Deliv. 2018, 2018, 3850139. [Google Scholar] [CrossRef]
- Balakumaran, M.D.; Ramachandran, R.; Kalaichelvan, P.T. Exploitation of Endophytic Fungus, Guignardia mangiferae for Extracellular Synthesis of Silver Nanoparticles and Their in Vitro Biological Activities. Microbiol. Res. 2015, 178, 9–17. [Google Scholar] [CrossRef]
- Li, L.; Pan, H.; Deng, L.; Qian, G.; Wang, Z.; Li, W.; Zhong, C. The Antifungal Activity and Mechanism of Silver Nanoparticles against Four Pathogens Causing Kiwifruit Post-Harvest Rot. Front. Microbiol. 2022, 13, 3418. [Google Scholar] [CrossRef]
- Zhou, L.; Zhao, X.; Li, M.; Lu, Y.; Ai, C.; Jiang, C.; Liu, Y.; Pan, Z.; Shi, J. Antifungal Activity of Silver Nanoparticles Synthesized by Iturin against Candida albicans in Vitro and in Vivo. Appl. Microbiol. Biotechnol. 2021, 105, 3759–3770. [Google Scholar] [CrossRef]
- Balakumaran, M.D.; Ramachandran, R.; Balashanmugam, P.; Mukeshkumar, D.J.; Kalaichelvan, P.T. Mycosynthesis of Silver and Gold Nanoparticles: Optimization, Characterization and Antimicrobial Activity against Human Pathogens. Microbiol. Res. 2016, 182, 8–20. [Google Scholar] [CrossRef]
- Gullino, M.L.; Tinivella, F.; Garibaldi, A.; Kemmitt, G.M.; Bacci, L.; Sheppard, B. Mancozeb: Past, Present, and Future. Plant Dis. 2010, 94, 1076–1087. [Google Scholar] [CrossRef]
- Dania, V.O.; Olaleye, E.A. Chemical Characterization of Essential Oil Constituents of Three Selected Botanicals and Their Antimicrobial Activity against Postharvest Rot Pathogens of Tomato (Solanum lycopersicum L.). Arch. Phytopathol. Plant Prot. 2022, 55, 564–582. [Google Scholar] [CrossRef]
- Sun, Z.; Yang, L.M.; Han, M.; Han, Z.M.; Yang, L.; Cheng, L.; Yang, X.; Lv, Z. liang Biological Control Ginseng Grey Mold and Plant Colonization by Antagonistic Bacteria Isolated from Rhizospheric Soil of Panax ginseng Meyer. Biol. Control 2019, 138, 104048. [Google Scholar] [CrossRef]
- Lorrai, R.; Ferrari, S. Host Cell Wall Damage during Pathogen Infection: Mechanisms of Perception and Role in Plant-Pathogen Interactions. Plants 2021, 10, 399. [Google Scholar] [CrossRef]




| Phytopathogen | Relative Enzyme Activity (%) | ||
|---|---|---|---|
| Cellulase | Pectinase | Xylanase | |
| F. oxysporum AUMC 10313 | 21.8 ± 2.4 | 48.7 ± 1.6 | 72.1 ± 1.1 |
| A. alternata AUMC 3882 | 15.1 ± 2.7 | 40.7 ± 1.9 | 70.7 ± 1.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd El-Ghany, M.N.; Hamdi, S.A.; Korany, S.M.; Elbaz, R.M.; Farahat, M.G. Biosynthesis of Novel Tellurium Nanorods by Gayadomonas sp. TNPM15 Isolated from Mangrove Sediments and Assessment of Their Impact on Spore Germination and Ultrastructure of Phytopathogenic Fungi. Microorganisms 2023, 11, 558. https://doi.org/10.3390/microorganisms11030558
Abd El-Ghany MN, Hamdi SA, Korany SM, Elbaz RM, Farahat MG. Biosynthesis of Novel Tellurium Nanorods by Gayadomonas sp. TNPM15 Isolated from Mangrove Sediments and Assessment of Their Impact on Spore Germination and Ultrastructure of Phytopathogenic Fungi. Microorganisms. 2023; 11(3):558. https://doi.org/10.3390/microorganisms11030558
Chicago/Turabian StyleAbd El-Ghany, Mohamed N., Salwa A. Hamdi, Shereen M. Korany, Reham M. Elbaz, and Mohamed G. Farahat. 2023. "Biosynthesis of Novel Tellurium Nanorods by Gayadomonas sp. TNPM15 Isolated from Mangrove Sediments and Assessment of Their Impact on Spore Germination and Ultrastructure of Phytopathogenic Fungi" Microorganisms 11, no. 3: 558. https://doi.org/10.3390/microorganisms11030558
APA StyleAbd El-Ghany, M. N., Hamdi, S. A., Korany, S. M., Elbaz, R. M., & Farahat, M. G. (2023). Biosynthesis of Novel Tellurium Nanorods by Gayadomonas sp. TNPM15 Isolated from Mangrove Sediments and Assessment of Their Impact on Spore Germination and Ultrastructure of Phytopathogenic Fungi. Microorganisms, 11(3), 558. https://doi.org/10.3390/microorganisms11030558








