Genomic Characterization of Three Novel Bartonella Strains in a Rodent and Two Bat Species from Mexico
Abstract
1. Introduction
2. Methods
2.1. Sampling and Isolation
2.2. Genomic and Library Methods
2.3. Genome Analysis
2.4. Virulence Factors Characterization
2.5. Pangenome Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Chomel, B.B.; Boulouis, H.-J.; Breitschwerdt, E.B.; Kasten, R.W.; Vayssier-Taussat, M.; Birtles, R.J.; Koehler, J.E.; Dehio, C. Ecological fitness and strategies of adaptation of Bartonella species to their hosts and vectors. Vet. Res. 2009, 40, 22–29. [Google Scholar] [CrossRef]
- Okaro, U.; Addisu, A.; Casanas, B.; Anderson, B. Bartonella Species, an Emerging Cause of Blood-Culture-Negative Endocarditis. Clin. Microbiol. Rev. 2017, 30, 709–746. [Google Scholar] [CrossRef]
- Cheslock, M.A.; Embers, M.E. Human Bartonellosis: An Underappreciated Public Health Problem? Trop. Med. Infect. Dis. 2019, 4, 69. [Google Scholar] [CrossRef]
- Gutiérrez, R.; Krasnov, B.; Morick, D.; Gottlieb, Y.; Khokhlova, I.S.; Harrus, S. Bartonella Infection in Rodents and Their Flea Ectoparasites: An Overview. Vector-Borne Zoonotic Dis. 2015, 15, 27–39. [Google Scholar] [CrossRef]
- Stuckey, M.J.; Chomel, B.B.; de Fleurieu, E.C.; Aguilar-Setién, A.; Boulouis, H.-J.; Chang, C.-C. Bartonella, bats and bugs: A review. Comp. Immunol. Microbiol. Infect. Dis. 2017, 55, 20–29. [Google Scholar] [CrossRef]
- Han, H.; Li, Z.; Li, X.; Liu, J.; Peng, Q.; Wang, R.; Gu, X.; Jiang, Y.; Zhou, C.; Li, D.; et al. Bats and their ectoparasites (Nycteribiidae and Spinturnicidae) carry diverse novel Bartonella genotypes, China. Transbound. Emerg. Dis. 2021, 69, e845–e858. [Google Scholar] [CrossRef]
- Krügel, M.; Król, N.; Kempf, V.A.J.; Pfeffer, M.; Obiegala, A. Emerging rodent-associated Bartonella: A threat for human health? Parasites Vectors 2022, 15, 113. [Google Scholar] [CrossRef]
- McKee, C.D.; Bai, Y.; Webb, C.T.; Kosoy, M.Y. Bats are key hosts in the radiation of mammal-associated Bartonella bacteria. Infect. Genet. Evol. 2021, 89, 104719. [Google Scholar] [CrossRef]
- André, M.R.; Gutiérrez, R.; Ikeda, P.; Amaral, R.B.D.; de Sousa, K.C.M.; Nachum-Biala, Y.; Lima, L.; Teixeira, M.M.G.; Machado, R.Z.; Harrus, S. Genetic diversity of Bartonella spp. in vampire bats from Brazil. Transbound. Emerg. Dis. 2019, 66, 2329–2341. [Google Scholar] [CrossRef]
- Müller, A.; Gutiérrez, R.; Seguel, M.; Monti, G.; Otth, C.; Bittencourt, P.; Sepúlveda, P.; Alabí, A.; Nachum-Biala, Y.; Harrus, S. Molecular survey of Bartonella spp. in rodents and fleas from Chile. Acta Trop. 2020, 212, 105672. [Google Scholar] [CrossRef]
- Ikeda, P.; Seki, M.C.; Carrasco, A.O.T.; Rudiak, L.V.; Miranda, J.M.D.; Gonçalves, S.M.M.; Hoppe, E.G.L.; Albuquerque, A.C.A.; Teixeira, M.M.G.; Passos, C.E.; et al. Evidence and molecular characterization of Bartonella spp. and hemoplasmas in neotropical bats in Brazil. Epidemiol. Infect. 2017, 145, 2038–2052. [Google Scholar] [CrossRef]
- de Oliveira, J.G.; Rozental, T.; Guterres, A.; Teixeira, B.R.; Andrade-Silva, B.E.; da Costa-Neto, S.F.; Furtado, M.C.; Moratelli, R.; D’Andrea, P.S.; Lemos, E.R.S. Investigation of Bartonella spp. in brazilian mammals with emphasis on rodents and bats from the Atlantic Forest. Int. J. Parasitol. Parasites Wildl. 2020, 13, 80–89. [Google Scholar] [CrossRef]
- Gonçalves, L.R.; Favacho, A.R.D.M.; Roque, A.L.R.; Mendes, N.S.; Fidelis, O.L., Jr.; Benevenute, J.L.; Herrera, H.M.; D’Andrea, P.S.; de Lemos, E.R.S.; Machado, R.Z.; et al. Association of Bartonella Species with Wild and Synanthropic Rodents in Different Brazilian Biomes. Appl. Environ. Microbiol. 2016, 82, 7154–7164. [Google Scholar] [CrossRef]
- Stuckey, M.J.; Chomel, B.B.; Obregón-Morales, C.; Leyva, J.I.O.; Arechiga-Ceballos, N.; Moreno-Sandoval, H.; Aguilar-Setién, A.; Salas-Rojas, M.; Galvez-Romero, G. Bartonella Infection in Hematophagous, Insectivorous, and Phytophagous Bat Populations of Central Mexico and the Yucatan Peninsula. Am. J. Trop. Med. Hyg. 2017, 97, 413–422. [Google Scholar] [CrossRef]
- Fischedick, F.B.S.; Stuckey, M.J.; Aguilar-Setién, A.; Moreno-Sandoval, H.; Galvez-Romero, G.; Salas-Rojas, M.; Arechiga-Ceballos, N.; Overgaauw, P.A.M.; Kasten, R.W.; Chomel, B.B. Identification of Bartonella Species Isolated from Rodents from Yucatan, Mexico, and Isolation of Bartonella vinsonii subsp. yucatanensis subsp. nov. Vector-Borne Zoonotic Dis. 2016, 16, 636–642. [Google Scholar] [CrossRef]
- Weis, A.M.; Storey, D.B.; Taff, C.C.; Townsend, A.K.; Huang, B.C.; Kong, N.T.; Clothier, K.A.; Spinner, A.; Byrne, B.A.; Weimer, B.C. Genomic Comparison of Campylobacter spp. and Their Potential for Zoonotic Transmission between Birds, Primates, and Livestock. Appl. Environ. Microbiol. 2016, 82, 7165–7175. [Google Scholar] [CrossRef]
- Weimer, B.C. 100K Pathogen Genome Project. Genome Announc. 2017, 5, e00594-17. [Google Scholar] [CrossRef]
- Hernández-Juárez, L.E.; Camorlinga, M.; Méndez-Tenorio, A.; Calderón, J.F.; Huang, B.C.; Bandoy, D.D.R.; Weimer, B.C.; Torres, J. Analyses of publicly available Hungatella hathewayi genomes revealed genetic distances indicating they belong to more than one species. Virulence 2021, 12, 1950–1964. [Google Scholar] [CrossRef]
- Flores-Valdez, M.; Ares, M.A.; Rosales-Reyes, R.; Torres, J.; Girón, J.A.; Weimer, B.C.; Mendez-Tenorio, A.; De la Cruz, M.A. Whole Genome Sequencing of Pediatric Klebsiella pneumoniae Strains Reveals Important Insights Into Their Virulence-Associated Traits. Front. Microbiol. 2021, 12, 711577. [Google Scholar] [CrossRef]
- Chen, P.; Reiter, T.; Huang, B.; Kong, N.; Weimer, B.C. Prebiotic Oligosaccharides Potentiate Host Protective Responses against L. Monocytogenes Infection. Pathogens 2017, 6, 68. [Google Scholar] [CrossRef]
- Aguilar-Zamora, E.; Weimer, B.C.; Torres, R.C.; Gómez-Delgado, A.; Ortiz-Olvera, N.; Aparicio-Ozores, G.; Barbero-Becerra, V.J.; Torres, J.; Camorlinga-Ponce, M. Molecular Epidemiology and Antimicrobial Resistance of Clostridioides difficile in Hospitalized Patients From Mexico. Front. Microbiol. 2022, 12, 787451. [Google Scholar] [CrossRef]
- Trim Galore. Available online: https://github.com/FelixKrueger/TrimGalore (accessed on 18 March 2022).
- Wick, R.R.; Judd, L.M.; Gorrie, C.L.; Holt, K.E. Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput. Biol. 2017, 13, e1005595. [Google Scholar] [CrossRef]
- PATRIC. Pathosystems Resource Integration Center. Available online: https://www.patricbrc.org/ (accessed on 18 May 2022).
- MiGa. Microbial Genome Atlas Online. Available online: http://microbial-genomes.org/ (accessed on 7 October 2022).
- Schwengers, O.; Jelonek, L.; Dieckmann, M.A.; Beyvers, S.; Blom, J.; Goesmann, A. Bakta: Rapid and standardized annotation of bacterial genomes via alignment-free sequence identification. Microb. Genom. 2021, 7, 000685. [Google Scholar] [CrossRef]
- Seemann, T. Prokka: Rapid Prokaryotic Genome Annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Brettin, T.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Olsen, G.J.; Olson, R.; Overbeek, R.; Parrello, B.; Pusch, G.D.; et al. RASTtk: A modular and extensible implementation of the RAST algorithm for building custom annotation pipelines and annotating batches of genomes. Sci. Rep. 2015, 5, 8365. [Google Scholar] [CrossRef]
- Tatusova, T.; DiCuccio, M.; Badretdin, A.; Chetvernin, V.; Nawrocki, E.P.; Zaslavsky, L.; Lomsadze, A.; Pruitt, K.D.; Borodovsky, M.; Ostell, J. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 2016, 44, 6614–6624. [Google Scholar] [CrossRef]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree 2—Approximately Maximum-Likelihood Trees for Large Alignments. PLoS ONE 2010, 5, e9490. [Google Scholar] [CrossRef]
- Wattam, A.R.; Davis, J.J.; Assaf, R.; Boisvert, S.; Brettin, T.; Bun, C.; Conrad, N.; Dietrich, E.M.; Disz, T.; Gabbard, J.L.; et al. Improvements to PATRIC, the all-bacterial Bioinformatics Database and Analysis Resource Center. Nucleic Acids Res. 2017, 45, D535–D542. [Google Scholar] [CrossRef]
- Lee, I.; Kim, Y.O.; Park, S.-C.; Chun, J. OrthoANI: An improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol. 2016, 66, 1100–1103. [Google Scholar] [CrossRef]
- Goris, J.; Konstantinidis, K.T.; Klappenbach, J.A.; Coenye, T.; Vandamme, P.; Tiedje, J.M. DNA–DNA hybridization values and their relationship to whole-genome sequence similarities. Int. J. Syst. Evol. Microbiol. 2007, 57, 81–91. [Google Scholar] [CrossRef]
- Ciufo, S.; Kannan, S.; Sharma, S.; Badretdin, A.; Clark, K.; Turner, S.; Brover, S.; Schoch, C.L.; Kimchi, A.; DiCuccio, M. Using average nucleotide identity to improve taxonomic assignments in prokaryotic genomes at the NCBI. Int. J. Syst. Evol. Microbiol. 2018, 68, 2386–2392. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.; Dehio, C. Role of distinct type-IV-secretion systems and secreted effector sets in host adaptation by pathogenic Bartonella species. Cell. Microbiol. 2019, 21, e13004. [Google Scholar] [CrossRef] [PubMed]
- Parks, D.H.; Imelfort, M.; Skennerton, C.T.; Hugenholtz, P.; Tyson, G.W. CheckM: Assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 2015, 25, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Roary: Rapid Large-Scale Prokaryote Pan Genome Analysis. Available online: https://github.com/sanger-pathogens/Roary (accessed on 12 November 2022).
- Page, A.J.; Cummins, C.A.; Hunt, M.; Wong, V.K.; Reuter, S.; Holden, M.T.G.; Fookes, M.; Falush, D.; Keane, J.A.; Parkhill, J. Roary: Rapid large-scale prokaryote pan genome analysis. Bioinformatics 2015, 31, 3691–3693. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.T.; Irber, L. sourmash: A library for MinHash sketching of DNA. J. Open Source Softw. 2016, 1, 27. [Google Scholar] [CrossRef]
- Howe, K.; Bateman, A.; Durbin, R. QuickTree: Building huge Neighbour-Joining trees of protein sequences. Bioinformatics 2002, 18, 1546–1547. [Google Scholar] [CrossRef]
- Roary_plots: Gene Presence and Absence, Frequency Plot and Piechart of Pangenome. Available online: https://github.com/sanger-pathogens/Roary/tree/master/contrib/roary_plots (accessed on 12 November 2022).
- Hadfield, J.; Croucher, N.J.; Goater, R.J.; AbuDahab, K.; Aanensen, D.M.; Harris, S.R. Phandango: An interactive viewer for bacterial population genomics. Bioinformatics 2018, 34, 292–293. [Google Scholar] [CrossRef]
- La Scola, B.; Zeaiter, Z.; Khamis, A.; Raoult, D. Gene-sequence-based criteria for species definition in bacteriology: The Bartonella paradigm. Trends Microbiol. 2003, 11, 318–321. [Google Scholar] [CrossRef]
- Kosoy, M.; Mckee, C.; Albayrak, L.; Fofanov, Y. Genotyping of Bartonella bacteria and their animal hosts: Current status and perspectives. Parasitology 2018, 145, 543–562. [Google Scholar] [CrossRef]
- Rainey, F.A. How to Describe New Species of Prokaryotes. Methods Microbiol. 2011, 38, 7–14. [Google Scholar] [CrossRef]
- Amaral, R.B.D.; Cardozo, M.V.; Varani, A.D.M.; Furquim, M.E.C.; Dias, C.M.; de Assis, W.O.; da Silva, A.R.; Herrera, H.M.; Machado, R.Z.; André, M.R. First Report of Bartonella spp. in Marsupials from Brazil, with a Description of Bartonella harrusi sp. nov. and a New Proposal for the Taxonomic Reclassification of Species of the Genus Bartonella. Microorganisms 2022, 10, 1609. [Google Scholar] [CrossRef]
- Kordick, D.L.; Swaminathan, B.; Greene, C.E.; Wilson, K.H.; Whitney, A.M.; O’Connor, S.; Hollis, D.G.; Matar, G.M.; Steigerwalt, A.G.; Malcolm, G.B.; et al. Bartonella vinsonii subsp. berkhoffii subsp. nov., Isolated from Dogs; Bartonella vinsonii subsp. vinsonii; and Emended Description of Bartonella vinsonii. Int. J. Syst. Evol. Microbiol. 1996, 46, 704–709. [Google Scholar] [CrossRef]
- Buffet, J.-P.; Kosoy, M.; Vayssier-Taussat, M. Natural history of Bartonella-infecting rodents in light of new knowledge on genomics, diversity and evolution. Futur. Microbiol. 2013, 8, 1117–1128. [Google Scholar] [CrossRef]
- Milholland, M.T.; Castro-Arellano, I.; Suzán, G.; Garcia-Peña, G.E.; Lee, T.E.; Rohde, R.E.; Aguirre, A.A.; Mills, J.N. Global Diversity and Distribution of Hantaviruses and Their Hosts. EcoHealth 2018, 15, 163–208. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Brisson, D.; Oggenfuss, K.; Devine, J.; Levy, M.Z.; Keesing, F. Effects of a zoonotic pathogen, Borrelia burgdorferi, on the behavior of a key reservoir host. Ecol. Evol. 2018, 8, 4074–4083. [Google Scholar] [CrossRef]
- Rubio, A.V.; Ávila-Flores, R.; Osikowicz, L.M.; Bai, Y.; Suzán, G.; Kosoy, M.Y. Prevalence and Genetic Diversity of Bartonella Strains in Rodents from Northwestern Mexico. Vector-Borne Zoonotic Dis. 2014, 14, 838–845. [Google Scholar] [CrossRef]
- Kosoy, M.; Goodrich, I. Comparative Ecology of Bartonella and Brucella Infections in Wild Carnivores. Front. Vet. Sci. 2019, 5, 322. [Google Scholar] [CrossRef]
- Harms, A.; Dehio, C. Intruders below the Radar: Molecular Pathogenesis of Bartonella spp. Clin. Microbiol. Rev. 2012, 25, 42–78. [Google Scholar] [CrossRef]
- Davoust, B.; Marié, J.-L.; Dahmani, M.; Berenger, J.-M.; Bompar, J.-M.; Blanchet, D.; Cheuret, M.; Raoult, D.; Mediannikov, O. Evidence of Bartonella spp. in Blood and Ticks (Ornithodoros hasei) of Bats, in French Guiana. Vector-Borne Zoonotic Dis. 2016, 16, 516–519. [Google Scholar] [CrossRef]
- Moskaluk, A.E.; Stuckey, M.J.; Jaffe, D.A.; Kasten, R.W.; Aguilar-Setién, A.; Olave-Leyva, J.I.; Galvez-Romero, G.; Obregón-Morales, C.; Salas-Rojas, M.; García-Flores, M.M.; et al. Molecular Detection ofBartonellaSpecies in Blood-Feeding Bat Flies from Mexico. Vector-Borne Zoonotic Dis. 2018, 18, 258–265. [Google Scholar] [CrossRef]
- Johnson, N.; Aréchiga-Ceballos, N.; Aguilar-Setién, A. Vampire Bat Rabies: Ecology, Epidemiology and Control. Viruses 2014, 6, 1911–1928. [Google Scholar] [CrossRef] [PubMed]
- Benavides, J.A.; Valderrama, W.; Recuenco, S.; Uieda, W.; Suzán, G.; Avila-Flores, R.; Velasco-Villa, A.; Almeida, M.; De Andrade, F.A.; Molina-Flores, B.; et al. Defining New Pathways to Manage the Ongoing Emergence of Bat Rabies in Latin America. Viruses 2020, 12, 1002. [Google Scholar] [CrossRef]
- Wray, A.K.; Olival, K.J.; Morán, D.; Lopez, M.R.; Alvarez, D.; Navarrete-Macias, I.; Liang, E.; Simmons, N.B.; Lipkin, W.I.; Daszak, P.; et al. Viral Diversity, Prey Preference, and Bartonella Prevalence in Desmodus rotundus in Guatemala. EcoHealth 2016, 13, 761–774. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Kosoy, M.; Recuenco, S.; Alvarez, D.; Moran, D.; Turmelle, A.; Ellison, J.; Garcia, D.L.; Estevez, A.; Lindblade, K.; et al. Bartonella spp. in Bats, Guatemala. Emerg. Infect. Dis. 2011, 17, 1269–1272. [Google Scholar] [CrossRef] [PubMed]
- Becker, D.J.; Bergner, L.; Bentz, A.; Orton, R.J.; Altizer, S.; Streicker, D.G. Genetic diversity, infection prevalence, and possible transmission routes of Bartonella spp. in vampire bats. PLoS Neglected Trop. Dis. 2018, 12, e0006786. [Google Scholar] [CrossRef]
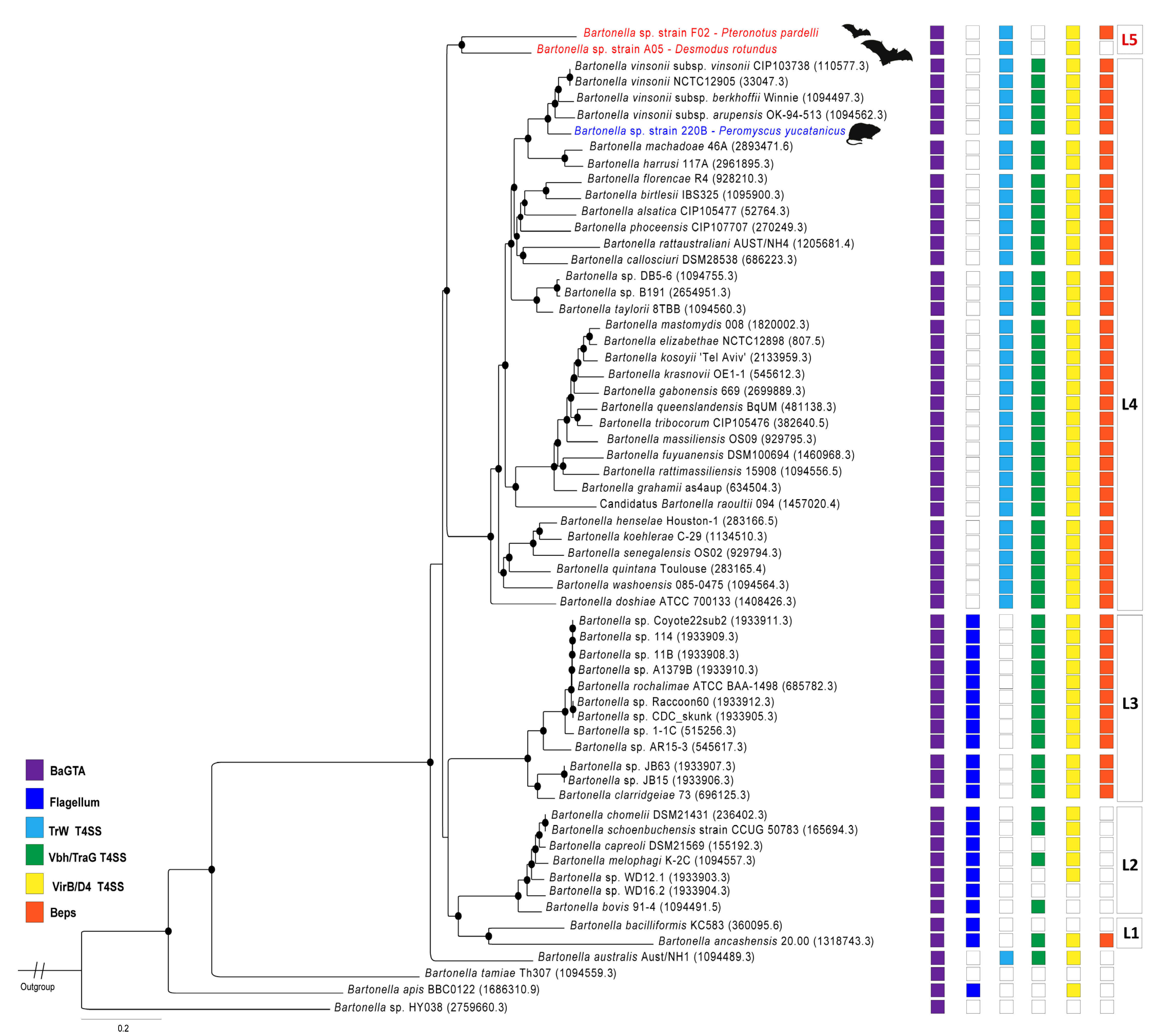
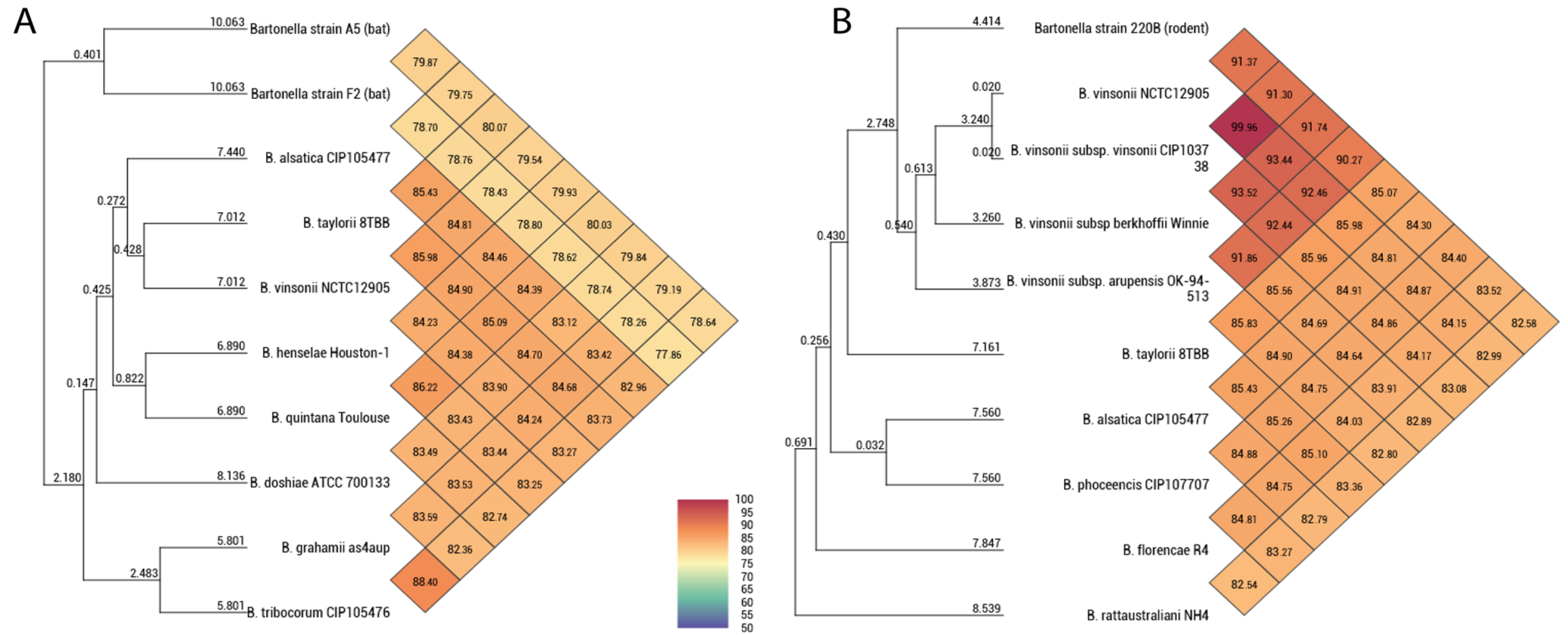
| Bartonella Strain 220B | Bartonella Strain F02 | Bartonella Strain A05 | |
|---|---|---|---|
| Host | Peromyscus yucatanicus (mouse) | Pteronotus parnellii (insectivorous bat) | Desmodus rotundus (vampire bat) |
| Contigs | 67 | 20 | 19 |
| Total length (bp) | 1,929,784 | 1,448,328 | 1,559,146 |
| DNA GC content (mol%) | 38.46 | 36.20 | 38.49 |
| Essential genes (106) | 105 | 106 | 106 |
| Completeness (%) | 99.1 | 100 | 100 |
| Contamination (%) | 0.9 | 1.9 | 0.9 |
| Quality (%) | 94.6 | 90.5 | 95.5 |
| Protein coding genes (CDS) | 1978 | 1320 | 1424 |
| tRNA genes | 39 | 39 | 40 |
| rRNA genes | 3 | 3 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves-Oliveira, J.; Gutierrez, R.; Schlesener, C.L.; Jaffe, D.A.; Aguilar-Setién, A.; Boulouis, H.-J.; Nachum-Biala, Y.; Huang, B.C.; Weimer, B.C.; Chomel, B.B.; et al. Genomic Characterization of Three Novel Bartonella Strains in a Rodent and Two Bat Species from Mexico. Microorganisms 2023, 11, 340. https://doi.org/10.3390/microorganisms11020340
Gonçalves-Oliveira J, Gutierrez R, Schlesener CL, Jaffe DA, Aguilar-Setién A, Boulouis H-J, Nachum-Biala Y, Huang BC, Weimer BC, Chomel BB, et al. Genomic Characterization of Three Novel Bartonella Strains in a Rodent and Two Bat Species from Mexico. Microorganisms. 2023; 11(2):340. https://doi.org/10.3390/microorganisms11020340
Chicago/Turabian StyleGonçalves-Oliveira, Jonathan, Ricardo Gutierrez, Cory Lee Schlesener, David A. Jaffe, Alvaro Aguilar-Setién, Henri-Jean Boulouis, Yaarit Nachum-Biala, Bihua C. Huang, Bart C. Weimer, Bruno B. Chomel, and et al. 2023. "Genomic Characterization of Three Novel Bartonella Strains in a Rodent and Two Bat Species from Mexico" Microorganisms 11, no. 2: 340. https://doi.org/10.3390/microorganisms11020340
APA StyleGonçalves-Oliveira, J., Gutierrez, R., Schlesener, C. L., Jaffe, D. A., Aguilar-Setién, A., Boulouis, H.-J., Nachum-Biala, Y., Huang, B. C., Weimer, B. C., Chomel, B. B., & Harrus, S. (2023). Genomic Characterization of Three Novel Bartonella Strains in a Rodent and Two Bat Species from Mexico. Microorganisms, 11(2), 340. https://doi.org/10.3390/microorganisms11020340










