One Health Perspectives on Food Safety in Minimally Processed Vegetables and Fruits: From Farm to Fork
Abstract
1. Introduction
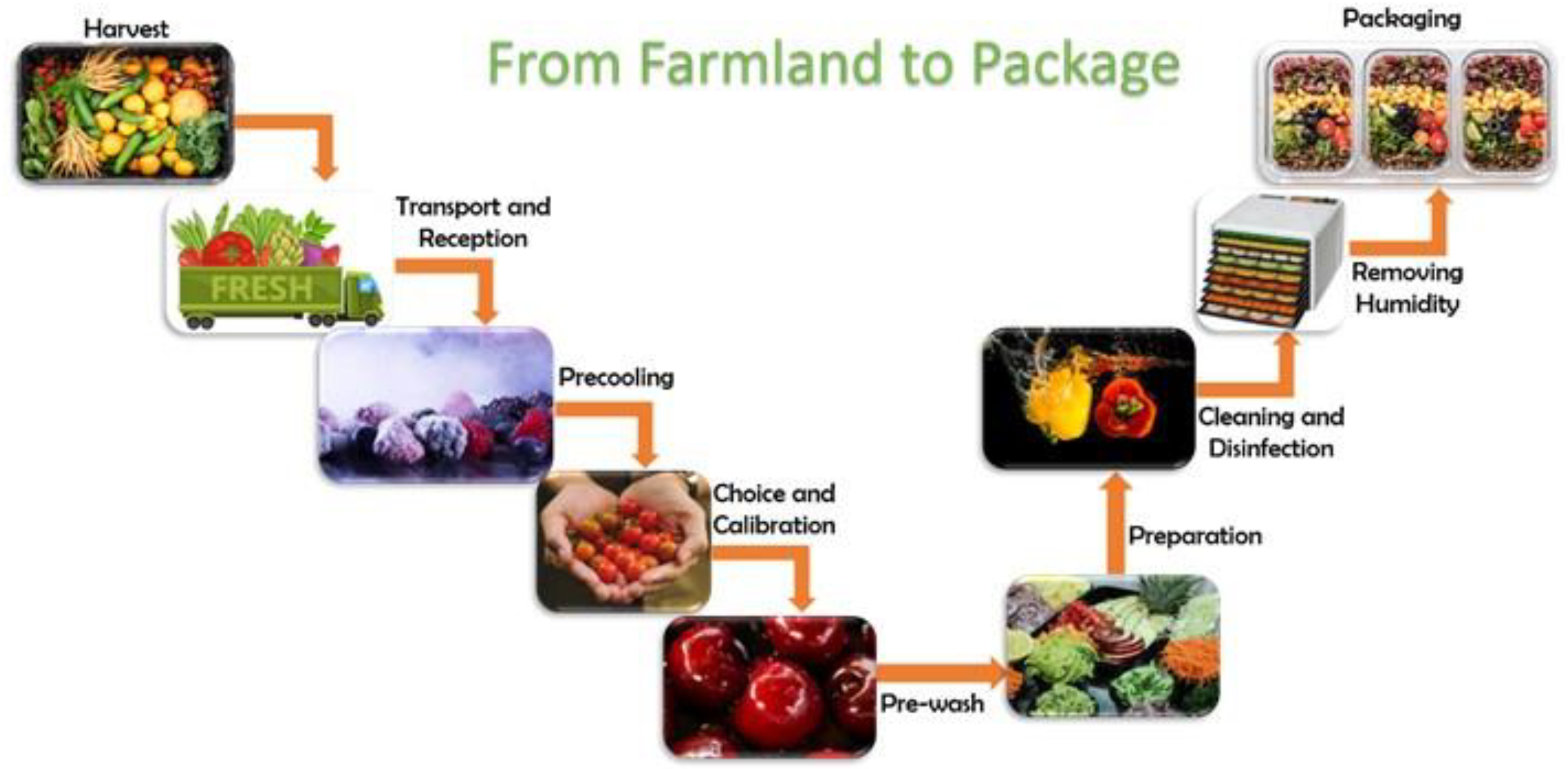
2. Minimally Processed Fruits and Vegetables
2.1. Minimally Processed Fruit and Vegetable Contamination
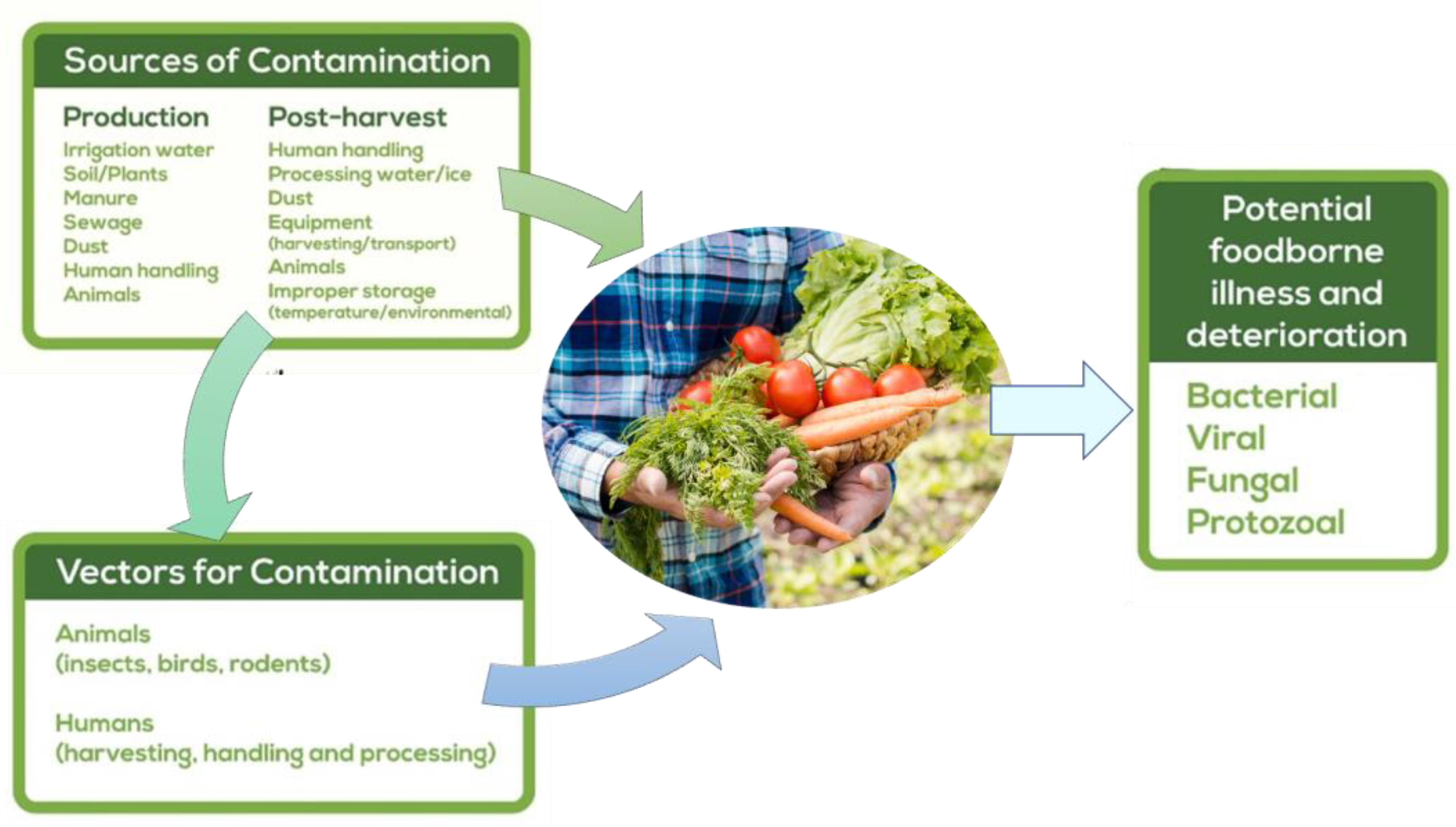
2.2. Main Sources of Microbiological Contamination throughout the Food Chain Production
2.2.1. Soil
2.2.2. Irrigation Water
2.2.3. Insects
2.2.4. Human Manipulation
2.3. Main Types of Contamination
2.3.1. Microbial Quality Indicators
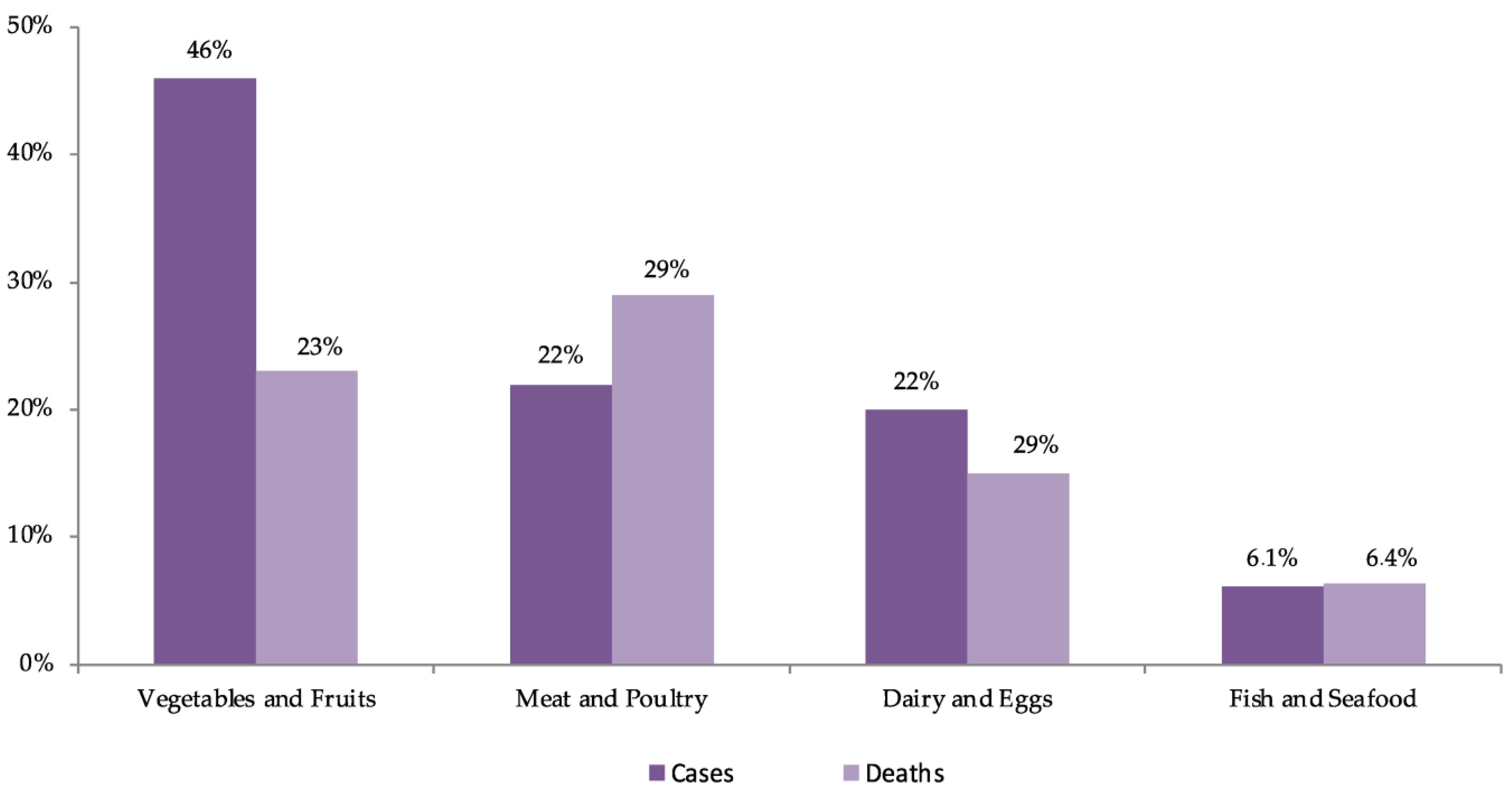
2.3.2. Pathogenic Microorganisms and Foodborne Disease-Related Cases
2.4. Major Decontamination Methodologies: Related Problems and Possible Solutions
2.4.1. Chlorine Disinfection
2.4.2. Other Chemical Methods of Disinfection
2.4.3. Physical Methods of Disinfection
2.5. Possible Future Solutions: The Use of Natural Disinfectants and Smart Packaging as an Alternative for Decontamination of Minimally Processed Fruits and Vegetables
3. One Health Perspectives on Food Safety in Minimally Processed Vegetables and Fruits
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ambikapathi, R.; Schneider, K.R.; Davis, B.; Herrero, M.; Winters, P.; Fanzo, J.C. Global food systems transitions have enabled affordable diets but had less favourable outcomes for nutrition, environmental health, inclusion and equity. Nat. Food 2022, 3, 764–779. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of United Nations. The Future of Food and Agriculture; Trends and Challenges; Food and Agriculture Organization of United Nations: Rome, Italy, 2017; Available online: https://www.fao.org/3/i6583e/i6583e.pdf (accessed on 7 February 2022).
- Vandevijvere, S.; Jaacks, L.M.; Monteiro, C.A.; Moubarac, J.-C.; Girling-Butcher, M.; Lee, A.C.; Pan, A.; Bentham, J.; Swinburn, B. Global trends in ultraprocessed food and drink product sales and their association with adult body mass index trajectories. Obes. Rev. 2019, 20, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.-Z.; Mujumdar, A.S.; Pan, Z.; Vidyarthi, S.K.; Xu, J.; Zielinska, M.; Xiao, H.-W. Emerging chemical and physical disinfection technologies of fruits and vegetables: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2020, 60, 2481–2508. [Google Scholar] [CrossRef]
- High Level Panel of Experts. Food Security and Nutrition: Building a Global Narrative towards 2030. A Report by the High-Level Panel of Experts on Food Security and Nutrition of the Committee on World Food Security, Rome. 2020. Available online: https://www.fao.org/3/ca9731en/ca9731en.pdf (accessed on 16 May 2022).
- Pinela, J.; Ferreira, I.C.F.R. Nonthermal physical technologies to decontaminate and extend the shelf-life of fruits and vegetables: Trends aiming at quality and safety. Crit. Rev. Food Sci. Nutr. 2017, 57, 2095–2111. [Google Scholar] [CrossRef] [PubMed]
- De Corato, U. Improving the shelf-life and quality of fresh and minimally-processed fruits and vegetables for a modern food industry: A comprehensive critical review from the traditional technologies into the most promising advancements. Crit. Rev. Food Sci. Nutr. 2020, 60, 940–975. [Google Scholar] [CrossRef] [PubMed]
- Pandey, B.; Reba, M.; Joshi, P.K.; Seto, K.C. Urbanization and food consumption in India. Sci. Rep. 2020, 10, 17241. [Google Scholar] [CrossRef] [PubMed]
- de Rezende, L.P.; Barbosa, J.; Teixeira, P. Analysis of Alternative Shelf Life-Extending Protocols and Their Effect on the Preservation of Seafood Products. Foods 2022, 11, 1100. [Google Scholar] [CrossRef]
- Gomes, B.A.F.; Alexandre, A.C.S.; de Andrade, G.A.V.; Zanzini, A.P.; de Barros, H.E.A.; Silva, L.M.d.S.F.e.; Costa, P.A.; Boas, E.V.d.B.V. Recent advances in processing and preservation of minimally processed fruits and vegetables: A review—Part 2: Physical methods and global market outlook. Food Chem. Adv. 2023, 2, 100304. [Google Scholar] [CrossRef]
- Sethi, S.; Nayak, S.L.; Joshi, A.; Sharma, R.R. Sanitizers for fresh-cut fruits and vegetables. In Fresh-Cut Fruits and Vegetables, Technologies, and Mechanisms for Safety Control; Siddiqui, M.W., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 99–119. [Google Scholar]
- Tavares, J.; Martins, A.; Fidalgo, L.G.; Lima, V.; Amaral, R.A.; Pinto, C.A.; Silva, A.M.; Saraiva, J.A. Fresh Fish Degradation and Advances in Preservation Using Physical Emerging Technologies. Foods 2021, 10, 780. [Google Scholar] [CrossRef]
- Ben Farhat, L.; Romeo, F.V.; Foti, P.; Russo, N.; Randazzo, C.L.; Caggia, C.; Abidi, F. Multi-Functional Potential of Lactic Acid Bacteria Strains and Antimicrobial Effects in Minimally Processed Pomegranate (Punica granatum L. cv Jolly Red) Arils. Microorganisms 2022, 10, 1876. [Google Scholar] [CrossRef]
- Finger, J.A.F.F.; Santos, I.M.; Silva, G.A.; Bernardino, M.C.; Pinto, U.M.; Maffei, D.F. Minimally Processed Vegetables in Brazil: An Overview of Marketing, Processing, and Microbiological Aspects. Foods 2023, 12, 2259. [Google Scholar] [CrossRef]
- Food and Drug Administration. Guide to Minimize Food Safety Hazards of Fresh-Cut Produce: Draft Guidance for Industry. 2018. Available online: https://www.fda.gov/media/117526/download (accessed on 18 June 2022).
- Teshome, E.; Forsido, S.F.; Rupasinghe, H.P.V.; Keyata, E.O. Potentials of Natural Preservatives to Enhance Food Safety and Shelf Life: A Review. Sci. World J. 2022, 2022, 9901018. [Google Scholar] [CrossRef] [PubMed]
- Amit, S.K.; Uddin, M.; Rahman, R.; Islam, S.M.R.; Khan, M.S. A review on mechanisms and commercial aspects of food preservation and processing. Agric. Food Secur. 2017, 6, 51. [Google Scholar] [CrossRef]
- Testa, R.; Schifani, G.; Migliore, G. Understanding Consumers’ Convenience Orientation. An Exploratory Study of Fresh-Cut Fruit in Italy. Sustainability 2021, 13, 1027. [Google Scholar] [CrossRef]
- Li, X.; Zhang, R.; Hassan, M.M.; Cheng, Z.; Mills, J.; Hou, C.; Realini, C.E.; Chen, L.; Day, L.; Zheng, X.; et al. Active Packaging for the Extended Shelf-Life of Meat: Perspectives from Consumption Habits, Market Requirements and Packaging Practices in China and New Zealand. Foods 2022, 11, 2903. [Google Scholar] [CrossRef] [PubMed]
- Uyttendaele, M.; Franz, E.; Schlüter, O. Food safety, a global challenge. Int. J. Environ. Res. Public Health 2016, 13, 67. [Google Scholar] [CrossRef]
- World Health Organization. Food Safety Fact Sheet. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/food-safety (accessed on 29 June 2023).
- Todd, E. Food-Borne Disease Prevention and Risk Assessment. Int. J. Environ. Res. Public Health 2020, 17, 5129. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Centre for Disease Prevention and Control. The European Union One Health 2021 Zoonoses Report. EFSA J. 2022, 20, e07666. [Google Scholar] [CrossRef]
- Garcia, S.N.; Osburn, B.I.; Jay-Russell, M.T. One Health for Food Safety, Food Security, and Sustainable Food Pro-duction. Front. Sustain. Food Syst. 2020, 4, 1. [Google Scholar] [CrossRef]
- World Health Organization. Quadripartite Memorandum of Understanding (MoU) Signed for a New Era of One Health Collaboration. 2022. Available online: https://www.fao.org/3/cb9403en/cb9403en.pdf (accessed on 29 June 2023).
- Aiyedun, S.O.; Onarinde, B.A.; Swainson, M.; Dixon, R.A. Foodborne outbreaks of microbial infection from fresh produce in Europe and North America: A systematic review of data from this millennium. Int. J. Food Sci. Technol. 2021, 56, 2215–2223. [Google Scholar] [CrossRef]
- McDaniel, C.; Jadeja, R. A review of fresh produce outbreaks, current interventions, food safety concerns and potential benefits of novel antimicrobial sodium acid sulfate. MOJ Food Process. Technol. 2019, 7, 59–67. [Google Scholar] [CrossRef]
- Miller, F.A.; Brandão, T.R.S.; Silva, C.L.M. New Approaches for Improving the Quality of Processed Fruits and Vegetables and Their By-Products. Foods 2023, 12, 1353. [Google Scholar] [CrossRef]
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Mullany, E.C.; Abate, K.H.; Abbafati, C.; Abebe, Z.; et al. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- Direção Geral de Saúde. Promoção da Alimentação Saudável. 2022. Available online: https://alimentacaosaudavel.dgs.pt/roda-dos-alimentos/ (accessed on 2 October 2022).
- Public Health England. A Quick Guide to the Government’s Healthy Eating Recommendations. 2018. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/742746/A_quick_guide_to_govt_healthy_eating_update.pdf (accessed on 5 October 2022).
- World Health Organization. Fruit and vegetables for health: Report of the Joint FAO/WHO Workshop on Fruit and Vegetables for Health, 1–3 September 2004, Kobe, Japan. 2005. Available online: https://apps.who.int/iris/handle/10665/43143 (accessed on 4 November 2022).
- World Health Organization. Healthy Diet Fact Sheet, 29 April 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/healthy-diet (accessed on 29 June 2023).
- Freshfel. 2021 Consumption Monitor. (European Fresh Produce Association). Fresh Fruit and Vegetable Production, Trade, Supply, Consumption and Monitor in EU-27. 2023. Available online: https://freshfel.org/what-we-do/consumption-monitor/ (accessed on 3 July 2023).
- Food and Agriculture Organization of United Nations. Fruit and Vegetables—Your Dietary Essentials. The International Year of Fruits and Vegetables 2021. Background Paper. Rome. 2020. Available online: https://www.fao.org/3/cb2395en/online/cb2395en.html (accessed on 14 September 2022).
- Amin, S.; Ganai, S.; Wani, S.; Mir, S.; Ahmad, M. Minimal Processing of Fruits-Principles & Practice, 1st ed.; Photon eBooks (Chapter 1); 2017; Available online: https://www.researchgate.net/publication/348304778_Minimal_Processing_of_Fruits-Principles_Practices_All_Rights_Reserved_With_Photon_Edition_1_Impact_Index_457 (accessed on 28 June 2023).
- Parrish, A. What Is a Processed Food? Michigan State University Extension. 2014. Available online: www.canr.msu.edu/news/what_is_a_processed_food (accessed on 9 November 2022).
- Global Market Insights. Global Processed Fruits and Vegetables Market Size by Type (Fruits, Vegetables), by Product (Fresh, Fresh-Cut, Canned, Frozen, Drying & Dehydration), by Processing Equipment (Preprocessing, Processing, Washing, Filling, Seasoning, Packaging), Industry Analysis Report, Regional Outlook, Growth Potential, Price Trends, Competitive Market Share & Forecast, 2021–2027. 2021. Available online: https://www.gminsights.com/industry-analysis/processed-fruits-and-vegetables-market (accessed on 26 November 2022).
- De Corato, U.; Cancellara, F.A. Measures, technologies, and incentives for cleaning the minimally processed fruits and vegetables supply chain in the Italian food industry. J. Clean. Prod. 2019, 237, 117735. [Google Scholar] [CrossRef]
- Gil, M.I.; Selma, M.V.; Suslow, T.; Jacxsens, L.; Uyttendaele, M.; Allende, A. Pre- and Postharvest Preventive Measures and Intervention Strategies to Control Microbial Food Safety Hazards of Fresh Leafy Vegetables. Crit. Rev. Food Sci. Nutr. 2014, 55, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Muthukkannan, N.; Kalidas, K. Minimally processed fresh-cut-vegetables for healthy consumption. Adv. Res. J. Crop Improv. 2019, 10, 9–14. [Google Scholar] [CrossRef]
- Perera, C.O. Minimal Processing of Fruit and Vegetables. In Handbook of Food Preservation; Rahman, M.S., Ed.; CRC Press: Boca Raton, FL, USA, 2020; pp. 191–205. [Google Scholar]
- Raffo, A.; Paoletti, F. Fresh-Cut Vegetables Processing: Environmental Sustainability and Food Safety Issues in a Comprehensive Perspective. Front. Sustain. Food Syst. 2022, 5, 681459. [Google Scholar] [CrossRef]
- Goodburn, C.; Wallace, C.A. The microbiological efficacy of decontamination methodologies for fresh produce: A review. Food Control 2013, 32, 418–427. [Google Scholar] [CrossRef]
- Carstens, C.K.; Salazar, J.K.; Darkoh, C. Multistate Outbreaks of Foodborne Illness in the United States Associated with Fresh Produce From 2010 to 2017. Front. Microbiol. 2019, 10, 2667. [Google Scholar] [CrossRef]
- Balali, G.I.; Yar, D.D.; Dela, V.G.A.; Adjei-Kusi, P. Microbial Contamination, an Increasing Threat to the Consumption of Fresh Fruits and Vegetables in Today’s World. Int. J. Microbiol. 2020, 2020, 3029295. [Google Scholar] [CrossRef]
- Banach, J.; Van Bokhorst-van de Veen, H.; van Overbeek, L.; van der Zouwen, P.; van der Fels-Klerx, H.; Groot, M.N. The efficacy of chemical sanitizers on the reduction of Salmonella Typhimurium and Escherichia coli affected by bacterial cell history and water quality. Food Control 2017, 81, 137–146. [Google Scholar] [CrossRef]
- Painter, J.A.; Hoekstra, R.M.; Ayers, T.; Tauxe, R.V.; Braden, C.R.; Angulo, F.J.; Griffin, P.M. Attribution of Foodborne Illnesses, Hospitalizations, and Deaths to Food Commodities by using Outbreak Data, United States, 1998–2008. Emerg. Infect. Dis. 2013, 19, 407–415. [Google Scholar] [CrossRef]
- Elias, S.d.O.; Decol, L.T.; Tondo, E.C. Foodborne outbreaks in Brazil associated with fruits and vegetables: 2008 through 2014. Food Qual. Saf. 2018, 2, 173–181. [Google Scholar] [CrossRef]
- Johnson, R. Foodborne Illnesses and Outbreaks from Fresh Produce. Congressional Research Service, In Foco, IF11092. 2019. Available online: https://sgp.fas.org/crs/misc/IF11092.pdf (accessed on 14 November 2022).
- Machado-Moreira, B.; Richards, K.; Abram, F.; Brennan, F.; Gaffney, M.; Burgess, C.M. Survival of Escherichia coli and Listeria innocua on Lettuce after Irrigation with Contaminated Water in a Temperate Climate. Foods 2021, 10, 2072. [Google Scholar] [CrossRef] [PubMed]
- Brandl, M.T. Fitness of human enteric pathogens on plants and implications for food safety. Annu. Rev. Phytopathol. 2006, 44, 367–392. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific Opinion on the risk posed by pathogens in food of non-animal origin. Part 1 (outbreak data analysis and risk ranking of food/pathogen combinations). EFSA J. 2013, 11, 3025. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific Opinion on the risk posed by pathogens in food of non-animal origin. Part 2 (Salmonella and Norovirus in leafy greens eaten raw as salads). EFSA J. 2014, 12, 3600. [Google Scholar] [CrossRef]
- Macieira, A.; Barbosa, J.; Teixeira, P. Food Safety in Local Farming of Fruits and Vegetables. Int. J. Environ. Res. Public Health 2021, 18, 9733. [Google Scholar] [CrossRef]
- Tauxe, R.; Kruse, H.; Hedberg, C.; Potter, M.; Madden, J.; Wachsmuth, K. Microbial hazards and emerging issues associated with produce a preliminary report to the national advisory committee on microbiologic criteria for foods. J. Food Prot. 1997, 60, 1400–1408. [Google Scholar] [CrossRef]
- Jung, Y.; Jang, H.; Matthews, K.R. Effect of the food production chain from farm practices to vegetable processing on outbreak incidence. Microb. Biotechnol. 2014, 7, 517–527. [Google Scholar] [CrossRef]
- Bintsis, T. Microbial pollution and food safety. AIMS Microbiol. 2018, 4, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Agriopoulou, S.; Stamatelopoulou, E.; Sachadyn-Król, M.; Varzakas, T. Lactic Acid Bacteria as Antibacterial Agents to Extend the Shelf Life of Fresh and Minimally Processed Fruits and Vegetables: Quality and Safety Aspects. Microorganisms 2020, 8, 952. [Google Scholar] [CrossRef] [PubMed]
- Barrera, M.J.; Blenkinsop, R.; Warriner, K. The effect of different processing parameters on the efficacy of commercial post-harvest washing of minimally processed spinach and shredded lettuce. Food Control 2013, 25, 745–751. [Google Scholar] [CrossRef]
- Fongaro, G.; García-González, M.C.; Hernández, M.; Kunz, A.; Barardi, C.R.M.; Rodríguez-Lázaro, D. Different Behavior of Enteric Bacteria and Viruses in Clay and Sandy Soils after Biofertilization with Swine Digestate. Front. Microbiol. 2017, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Hu, H.; Meng, H.; Liu, L.; Xu, X.; Zhao, T. Vertical distribution and affecting factors of Escherichia coli over a 0–400 cm soil profile irrigated with sewage effluents in northern China. Ecotoxicol. Environ. Saf. 2020, 205, 111357. [Google Scholar] [CrossRef] [PubMed]
- Black, Z.; Balta, I.; Black, L.; Naughton, P.J.; Dooley, J.S.G.; Corcionivoschi, N. The Fate of Foodborne Pathogens in Manure Treated Soil. Front. Microbiol. 2021, 12, 781357. [Google Scholar] [CrossRef]
- Alegbeleye, O.O.; Sant’ana, A.S. Manure-borne pathogens as an important source of water contamination: An update on the dynamics of pathogen survival/transport as well as practical risk mitigation strategies. Int. J. Hyg. Environ. Health 2020, 227, 113524. [Google Scholar] [CrossRef]
- Millner, P.D. Manure management. In The Produce Contamination Problem Causes and Solutions, 2nd ed.; Matthews, K.R., Sapers, G.M., Gerba, C.P., Eds.; Academic Press: Cambridge, MA, USA, 2014; pp. 85–106. [Google Scholar]
- Alegbeleye, O.O.; Singleton, I.; Sant’ana, A.S. Sources and contamination routes of microbial pathogens to fresh produce during field cultivation: A review. Food Microbiol. 2018, 73, 177–208. [Google Scholar] [CrossRef]
- Mügler, C.; Ribolzi, O.; Viguier, M.; Janeau, J.-L.; Jardé, E.; Latsachack, K.; Henry-Des-Tureaux, T.; Thammahacksa, C.; Valentin, C.; Sengtaheuanghoung, O.; et al. Experimental and modelling evidence of splash effects on manure borne Escherichia coli washoff. Environ. Sci. Pollut. Res. 2021, 28, 33009–33020. [Google Scholar] [CrossRef]
- Ramos, T.D.M.; Jay-Russell, M.T.; Millner, P.D.; Baron, J.N.; Stover, J.; Pagliari, P.; Hutchinson, M.; Lilley, J.; Rowley, N.; Haghani, V.; et al. Survival and Persistence of Foodborne Pathogens in Manure-Amended Soils and Prevalence on Fresh Produce in Certified Organic Farms: A Multi-Regional Baseline Analysis. Front. Sustain. Food Syst. 2021, 5, 674767. [Google Scholar] [CrossRef]
- Blaiotta, G.; Di Cerbo, A.; Murru, N.; Coppola, R.; Aponte, M. Persistence of bacterial indicators and zoonotic pathogens in contaminated cattle wastes. BMC Microbiol. 2016, 16, 87. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.P.; Patel, J.; Pradhan, A.K. Application of alternative sources of water in agricultural food production—Current trends and future prospects. Curr. Opin. Food Sci. 2022, 47, 100877. [Google Scholar] [CrossRef]
- Liu, H.; Whitehouse, C.A.; Li, B. Presence and Persistence of Salmonella in Water: The Impact on Microbial Quality of Water and Food Safety. Front. Public Health 2018, 6, 159. [Google Scholar] [CrossRef] [PubMed]
- Toro, M.; Weller, D.; Ramos, R.; Diaz, L.; Alvarez, F.P.; Reyes-Jara, A.; Moreno-Switt, A.I.; Meng, J.; Adell, A.D. Environmental and anthropogenic factors associated with the likelihood of detecting Salmonella in agricultural watersheds. Environ. Pollut. 2022, 306, 119298. [Google Scholar] [CrossRef] [PubMed]
- Dobhal, S.; Zhang, G.; Royer, T.; Damicone, J.; Ma, L.M. Survival and growth of foodborne pathogens in pesticide solutions routinely used in leafy green vegetables and tomato production. J. Sci. Food Agric. 2014, 94, 2958–2964. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Luo, Y.; Yang, Y.; Vinyard, B.; Schneider, K.; Meng, J. Effects of Tomato Variety, Temperature Differential, and Post–Stem Removal Time on Internalization of Salmonella enterica Serovar Thompson in Tomatoes. J. Food Prot. 2012, 75, 297–303. [Google Scholar] [CrossRef]
- Rodrigues, C.; da Silva, A.L.B.R.; Dunn, L.L. Factors Impacting the Prevalence of Foodborne Pathogens in Agricultural Water Sources in the Southeastern United States. Water 2019, 12, 51. [Google Scholar] [CrossRef]
- Coleman, S.M.; Bisha, B.; Newman, S.E.; Bunning, M.; Goodridge, L.D. Transmission and Persistence of Salmonella enterica in Nutrient Solution of Hydroponic Greenhouse Grown Tomatoes. HortScience 2017, 52, 713–718. [Google Scholar] [CrossRef]
- Andrade, L.; O’Dwyer, J.; O’Neill, E.; Hynds, P. Surface water flooding, groundwater contamination, and enteric disease in developed countries: A scoping review of connections and consequences. Environ. Pollut. 2018, 236, 540–549. [Google Scholar] [CrossRef]
- Food and Agricultural Organization of the United Nations; World Health Organization. Microbiological Hazards in Fresh Leafy Vegetables and Herbs: Meeting Report. Microbiological Risk Assessment Series, No. 14. Rome, Italy. 2008. Available online: https://apps.who.int/iris/handle/10665/44031 (accessed on 14 November 2022).
- Faour-Klingbeil, D.; Murtada, M.; Kuri, V.; Todd, E.C. Understanding the routes of contamination of ready-to-eat vegetables in the Middle East. Food Control 2016, 62, 125–133. [Google Scholar] [CrossRef]
- Berry, E.D.; Wells, J.E.; Durso, L.M.; Friesen, K.M.; Bono, J.L.; Suslow, T.V. Occurrence of Escherichia coli O157:H7 in Pest Flies Captured in Leafy Greens Plots Grown Near a Beef Cattle Feedlot. J. Food Prot. 2019, 82, 1300–1307. [Google Scholar] [CrossRef]
- Sela, S.; Nestel, D.; Pinto, R.; Nemny-Lavy, E.; Bar-Joseph, M. Mediterranean fruit fly as a potential vector of bacterial pathogens. Appl. Environ. Microbiol. 2005, 71, 4052–4056. [Google Scholar] [CrossRef]
- Gil, M.I.; Allende, A. Water and wastewater use in the fresh produce industry: Food safety and environmental im-plications. In Quantitative Methods for Food Safety and Quality in the Vegetable Industry; Pérez-Rodríguez, F., Skandamis, P., Valdramidis, V., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; pp. 59–76. [Google Scholar] [CrossRef]
- European Food Safety Authority. Urgent advice on the public health risk of Shiga-toxin producing Escherichia coli in fresh vegetables. EFSA J. 2011, 9, 2274. [Google Scholar] [CrossRef]
- Ali, A.; Yeoh, W.K.; Forney, C.; Siddiqui, M.W. Advances in postharvest technologies to extend the storage life of minimally processed fruits and vegetables. Crit. Rev. Food Sci. Nutr. 2017, 58, 2632–2649. [Google Scholar] [CrossRef] [PubMed]
- Perumal, A.B.; Huang, L.; Nambiar, R.B.; He, Y.; Li, X.; Sellamuthu, P.S. Application of essential oils in packaging films for the preservation of fruits and vegetables: A review. Food Chem. 2022, 375, 131810. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Teplitski, M. Microbiological food safety considerations in shelf-life extension of fresh fruits and vegetables. Curr. Opin. Biotechnol. 2023, 80, 102895. [Google Scholar] [CrossRef] [PubMed]
- Zaho, Y. Pathogens in fruit. In Improving the Safety of Fresh Fruit and Vegetables, 1st ed.; Jongen, W., Ed.; Woodhead Publishing Limited: Sawston, UK, 2005; pp. 44–88. [Google Scholar]
- Iturralde-García, R.D.; Cinco-Moroyoqui, F.J.; Martínez-Cruz, O.; Ruiz-Cruz, S.; Wong-Corral, F.J.; Borboa-Flores, J.; Cornejo-Ramírez, Y.I.; Bernal-Mercado, A.T.; Del-Toro-Sánchez, C.L. Emerging Technologies for Prolonging Fresh-Cut Fruits’ Quality and Safety during Storage. Horticulturae 2022, 8, 731. [Google Scholar] [CrossRef]
- Siroli, L.; Patrignani, F.; Serrazanetti, D.I.; Tabanelli, G.; Montanari, C.; Gardini, F.; Lanciotti, R. Lactic acid bacteria and natural antimicrobials to improve the safety and shelf-life of minimally processed sliced apples and lamb’s lettuce. Food Microbiol. 2015, 47, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Jeddi, M.Z.; Yunesian, M.; Gorji, M.E.; Noori, N.; Pourmand, M.R.; Khaniki, G.J. Microbial evaluation of fresh, minimally-processed vegetables and bagged sprouts from chain supermarkets. J. Health Popul. Nutr. 2014, 32, 391–399. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4221445/ (accessed on 14 November 2023). [PubMed]
- Cerna-Cortes, J.F.; Leon-Montes, N.; Cortes-Cueto, A.L.; Salas-Rangel, L.P.; Helguera-Repetto, A.C.; Lopez-Hernandez, D.; Rivera-Gutierrez, S.; Fernandez-Rendon, E.; Gonzalez-Y-Merchand, J.A. Microbiological quality of ready-to-eat vegetables collected in Mexico City: Occurrence of aerobic-mesophilic bacteria, fecal coliforms, and potentially pathogenic nontuberculous mycobacteria. BioMed Res. Int. 2015, 2015, 789508. [Google Scholar] [CrossRef]
- Hualpa, D.; Toledo, Z.; Meneses, M.; Feng, P. Microbiological quality of minimally, processed, ready-to-eat, vegetables in Loja, Ecuador. Rev. Politécnica 2018, 41, 45–50. Available online: http://scielo.senescyt.gob.ec/scielo.php?script=sci_arttext&pid=S1390-01292018000200045 (accessed on 14 November 2023).
- Abaza, A. Bacteriological assessment of some vegetables and ready-to-eat salads in Alexandria, Egypt. J. Egypt. Public Health Assoc. 2017, 92, 177–187. [Google Scholar] [CrossRef]
- Bencardino, D.; Vitali, L.A.; Petrelli, D. Microbiological evaluation of ready-to-eat iceberg lettuce during shelf-life and effectiveness of household washing methods. Ital. J. Food Saf. 2018, 7, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Cvetkovic, D.; Pavlovic, D.; Ranitovic, A.; Markov, S. Microbiological quality of fresh vegetable salads from the Novi Sad market. Acta Period. Technol. 2018, 49, 21–30. [Google Scholar] [CrossRef]
- Łepecka, A.; Zielińska, D.; Szymański, P.; Buras, I.; Kołożyn-Krajewska, D. Assessment of the microbiological quality of ready-to-eat salads—Are there any reasons for concern about public health? Int. J. Environ. Res. Public Health 2022, 19, 1582. [Google Scholar] [CrossRef]
- Liao, C.; Wang, L. The microbial quality of commercial chopped romaine lettuce before and after the “use by” date. Front. Microbiol. 2022, 13, 850720. [Google Scholar] [CrossRef] [PubMed]
- Anthony, A.A.; Divine-Anthony, O. Bacteria Associated with the Spoilage of Salad, their resistotyping and Potential Public Health Implications. Int. J. Eng. Res. Sci. 2015, 1, 55–59. [Google Scholar]
- Poubol, J.; Izumi, H. Shelf life and microbial quality of fresh-cut mango cubes stored in high CO2 atmospheres. J. Food Sci. 2005, 70, M69–M74. [Google Scholar] [CrossRef]
- Santos, M.; Cavaco, A.; Gouveia, J.; Novais, M.; Nogueira, P.; Pedroso, L.; Ferreira, M.A.S.S. Evaluation of minimally processed salads commercialized in Portugal. Food Control 2012, 23, 275–281. [Google Scholar] [CrossRef]
- Odonkor, S.T.; Ampofo, J.K. Escherichia coli as an indicator of bacteriological quality of water: An overview. Microbiol. Res. 2013, 4, 2. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Hamilton, M.J.; Wang, P.; Staley, C.; Matteson, S.; Birr, A.; Sadowsky, M.J. Geographic isolation of Escherichia coli genotypes in sediments and water of the Seven Mile Creek—A constructed riverine watershed. Sci. Total Environ. 2015, 538, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Patel, N.; Gadedjisso-Tossou, A.; Patra, S.; Singh, N.; Singh, P.K. Incidence of Escherichia coli in Vegetable Crops and Soil Profile Drip Irrigated with Primarily Treated Municipal Wastewater in a Semi-Arid Peri Urban Area. Agriculture 2020, 10, 291. [Google Scholar] [CrossRef]
- Coniglio, M.A.; Faro, G.; Marranzano, M. The importance of the microbiological quality of ready-to-eat salads from a public health perspective. J. Food Process. Technol. 2016, 7, 577. [Google Scholar] [CrossRef]
- Veterinærinstituttet. Escherichia coli og Salmonella i Friske Spiseklare Vegetabiler 2021. Report 29/2022. 2022. Available online: https://www.mattilsynet.no/mat_og_vann/smitte_fra_mat_og_drikke/bakterier_i_mat_og_drikke/rapport_smittestoffer_i_sukker-erter_og_vasket_salat_2021.48288/binary/Rapport:%20Smittestoffer%20i%20sukkererter%20og%20vasket%20salat%202021 (accessed on 18 November 2022).
- European Commission. Regulation (EC) No. 2073/2005 of 15 November 2005 on microbiological criteria for foodstuffs. Off. J. Eur. Union—Legis. 2005, 338, 1–26. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32005R2073&qid=1672315192734 (accessed on 12 November 2022).
- Gurtler, J.B.; E Gibson, K. Irrigation water and contamination of fresh produce with bacterial foodborne pathogens. Curr. Opin. Food Sci. 2022, 47, 100889. [Google Scholar] [CrossRef]
- Esmael, A.; Al-Hindi, R.R.; Albiheyri, R.S.; Alharbi, M.G.; Filimban, A.A.R.; Alseghayer, M.S.; Almaneea, A.M.; Alhadlaq, M.A.; Ayubu, J.; Teklemariam, A.D. Fresh Produce as a Potential Vector and Reservoir for Human Bacterial Pathogens: Revealing the Ambiguity of Interaction and Transmission. Microorganisms 2023, 11, 753. [Google Scholar] [CrossRef] [PubMed]
- Ramos, B.; Miller, F.A.; Brandão, T.R.S.; Teixeira, P.; Silva, C.L.M. Fresh fruits and vegetables—An overview on applied methodologies to improve its quality and safety. Innov. Food Sci. Emerg. Technol. 2013, 20, 1–15. [Google Scholar] [CrossRef]
- Enkirch, T.; Eriksson, R.; Persson, S.; Schmid, D.; Aberle, S.W.; Löf, E.; Wittesjö, B.; Holmgren, B.; Johnzon, C.; Gustafsson, E.X.; et al. Hepatitis A outbreak linked to imported frozen strawberries by sequencing, Sweden and Austria, June to September 2018. Eurosurveillance 2018, 23, 1800528. [Google Scholar] [CrossRef]
- Ruscher, C.; Faber, M.; Werber, D.; Stark, K.; Bitzegeio, J.; Michaelis, K.; Sagebiel, D.; Wenzel, J.J.; Enkelmann, J. Resurgence of an international hepatitis A outbreak linked to imported frozen strawberries, Germany, 2018 to 2020. Eurosurveillance 2020, 25, 1900670. [Google Scholar] [CrossRef]
- Gossner, C.M.; de Jong, B.; Hoebe, C.J.; Kornschober, C.; Schmid, D.; Quoilin, S.; Parmakova, K.; Petrov, P.; Koliou, M.; Kralova, R.; et al. Event-based surveillance of food- and waterborne diseases in Europe: Urgent inquiries (outbreak alerts) during 2008 to 2013. EuroSurveillance 2015, 20, 21166. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=21166 (accessed on 14 November 2023). [CrossRef]
- King, L.A.; Nogareda, F.; Weill, F.-X.; Mariani-Kurkdjian, P.; Loukiadis, E.; Gault, G.; Jourdan-DaSilva, N.; Bingen, E.; Macé, M.; Thevenot, D.; et al. Outbreak of shiga toxin-producing Escherichia coli O104:H4 associated with organic fenugreek sprouts, France, June 2011. Clin. Infect. Dis. 2012, 54, 1588–1594. [Google Scholar] [CrossRef]
- World Health Organization. Public Health Review of the Enterohaemorrhagic Escherichia coli Outbreak in Germany. 2011. Available online: https://apps.who.int/iris/bitstream/handle/10665/349978/WHO-EURO-2011-4324-44087-62183-eng.pdf?sequence=1&isAllowed=y (accessed on 29 June 2023).
- Centers for Disease Control. Estimates of Foodborne Illnessin the United States. 2014. Available online: http://www.cdc.gov/foodborneburden/attribution/ (accessed on 28 September 2022).
- Centers for Disease Control. List of Selected Multistate Foodborne Outbreak Investigation. 2016. Available online: https://www.cdc.gov/listeria/outbreaks/frozen-vegetables-05-16/index.html (accessed on 14 July 2022).
- Bennett, S.D.; Sodha, S.V.; Ayers, T.L.; Lynch, M.F.; Gould, L.H.; Tauxe, R.V. Produce-associated foodborne disease outbreaks, USA, 1998–2013. Epidemiol. Infect. 2018, 146, 1397–1406. [Google Scholar] [CrossRef] [PubMed]
- Luna-Guevara, J.J.; Arenas-Hernandez, M.M.P.; de la Peña, C.M.; Silva, J.L.; Luna-Guevara, M.L. The Role of Pathogenic E. coli in Fresh Vegetables: Behavior, Contamination Factors, and Preventive Measures. Int. J. Microbiol. 2019, 2019, 2894328. [Google Scholar] [CrossRef] [PubMed]
- Interagency Food Safety Analytics Collaboration. Foodborne Illness Source Attribution Estimates for 2020 for Salmonella, Escherichia coli O157, and Listeria monocytogenes Using Multi-Year Outbreak Surveillance Data, United States. GA and D.C.: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Food and Drug Administration, U.S. Department of Agriculture’s Food Safety and Inspection Service. 2022. Available online: https://www.cdc.gov/foodsafety/ifsac/pdf/p19-2020-report-triagency-508.pdf (accessed on 2 July 2023).
- Centers for Disease Control and Prevention. List of Multistate Foodborne Outbreak Notices. 2023. Available online: https://www.cdc.gov/foodsafety/outbreaks/lists/outbreaks-list.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Ffoodsafety%2Foutbreaks%2Fmultistate-outbreaks%2Foutbreaks-list.html (accessed on 6 July 2023).
- Diseases, T.L.I. C difficile—A rose by any other name. Lancet Infect. Dis. 2019, 19, 449. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, D.S.; Costa, M.C.; Dew WM, V.; Weese, J.S. Clostridium difficile in vegetables, Canada. Lett. Appl. Microbiol. 2010, 51, 600–602. [Google Scholar] [CrossRef] [PubMed]
- Candel-Pérez, C.; Ros-Berruezo, G.; Martínez-Graciá, C. A review of Clostridioides [Clostridium] difficile occurrence through the food chain. Food Microbiol. 2019, 77, 118–129. [Google Scholar] [CrossRef]
- Lim, S.C.; Foster, N.F.; Elliott, B.; Riley, T.V. High prevalence of Clostridium difficile on retail root vegetables, Western Australia. J. Appl. Microbiol. 2017, 124, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Usui, M.; Maruko, A.; Harada, M.; Kawabata, F.; Sudo, T.; Noto, S.; Sato, T.; Shinagawa, M.; Takahashi, S.; Tamura, Y. Prevalence and characterization of Clostridioides difficile isolates from retail food products (vegetables and meats) in Japan. Anaerobe 2019, 61, 102132. [Google Scholar] [CrossRef]
- Barbosa, J.; Campos, A.; Teixeira, P. Methods currently applied to study the prevalence of Clostridioides difficile in foods. AIMS Agric. Food 2020, 5, 102–128. [Google Scholar] [CrossRef]
- Pal, M.; Bulcha, M.R. Clostridium difficile as an emerging foodborne pathogen of public health significance. Acta Sci. Microbiol. 2021, 4, 46–49. [Google Scholar] [CrossRef]
- Eckert, C.; Burghoffer, B.; Barbut, F. Contamination of ready-to-eat raw vegetables with Clostridium difficile in France. J. Med. Microbiol. 2013, 62, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Lund, B.M.; Peck, M.W. A Possible Route for Foodborne Transmission of Clostridium difficile? Foodborne Pathog. Dis. 2015, 12, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Ünlü, G. The Prevalence, Transmission, and Control of C. difficile in Food. Food Technol. Mag. 2020, 74, 72–77. Available online: https://www.ift.org/news-and-publications/food-technology-magazine/issues/2020/february/columns/the-prevalence-transmission--and-control-of-c-difficile-in-food (accessed on 11 December 2022).
- Mirlohi, M.; Yamoudy, M.; Isfahani, B.N.; Jalali, M.; Esfandiari, Z.; Hosseini, N.S. Isolation of toxigenic Clostridium difficile from ready-to-eat salads by multiplex polymerase chain reaction in Isfahan, Iran. Adv. Biomed. Res. 2015, 4, 87. [Google Scholar] [CrossRef] [PubMed]
- Bakri, M.M.; Brown, D.J.; Butcher, J.P.; Sutherland, A.D. Clostridium difficile in Ready-to-Eat Salads, Scotland. Emerg. Infect. Dis. 2009, 15, 817–818. [Google Scholar] [CrossRef]
- Rodriguez-Palacios, A.; Ilic, S.; LeJeune, J.T. Clostridium difficile with Moxifloxacin/Clindamycin Resistance in Vegetables in Ohio, USA, and Prevalence Meta-Analysis. J. Pathog. 2014, 2014, 158601. [Google Scholar] [CrossRef]
- Bhilwadikar, T.; Pounraj, S.; Manivannan, S.; Rastogi, N.K.; Negi, P.S. Decontamination of Microorganisms and Pesticides from Fresh Fruits and Vegetables: A Comprehensive Review from Common Household Processes to Modern Techniques. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1003–1038. [Google Scholar] [CrossRef]
- Botondi, R.; Barone, M.; Grasso, C. A Review into the Effectiveness of Ozone Technology for Improving the Safety and Preserving the Quality of Fresh-Cut Fruits and Vegetables. Foods 2021, 10, 748. [Google Scholar] [CrossRef]
- Finnegan, E.; O’beirne, D. Characterising Deterioration Patterns in Fresh-Cut Fruit Using Principal Component Analysis. II: Effects of Ripeness Stage, Seasonality, Processing and Packaging. Postharvest Biol. Technol. 2015, 100, 91–98. [Google Scholar] [CrossRef]
- Warriner, K.; Namvar, A. Recent advances in fresh produce post-harvest decontamination technologies to enhance microbiological safety. Stewart Postharvest Rev. 2013, 9, 1–8. [Google Scholar] [CrossRef]
- Allende, A.; Ölmez, H. Strategies to Combat Microbial Hazards Associated with Fresh Produce. Cult. Thermoscientific 2015, 35. Available online: https://assets.thermofisher.com/TFS-Assets/MBD/Vector-Information/Culture-35-2-Microbial-Hazards-Fresh-Produce-LT2174A-EN.pdf (accessed on 28 June 2023).
- Bartz, J.A.; Yuk, H.-G.; Mahovic, M.J.; Warren, B.R.; Sreedharan, A.; Schneider, K.R. Internalization of Salmonella enterica by tomato fruit. Food Control 2015, 55, 141–150. [Google Scholar] [CrossRef]
- Macarisin, D.; Wooten, A.; De Jesus, A.; Hur, M.; Bae, S.; Patel, J.; Evans, P.; Brown, E.; Hammack, T.; Chen, Y. Internalization of Listeria monocytogenes in cantaloupes during dump tank washing and hydrocooling. Int. J. Food Microbiol. 2017, 257, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Luo, Y.; Bauchan, G.R.; Feng, H.; Stommel, J.R. Visualizing Pathogen Internalization Pathways in Fresh Tomatoes Using MicroCT and Confocal Laser Scanning Microscopy. Food Control 2018, 85, 276–282. [Google Scholar] [CrossRef]
- Gómez-López, V.M.; Marín, A.; Allende, A.; Beuchat, L.R.; Gil, M.I. Postharvest handling conditions affect internalization of salmonella in baby spinach during washing. J. Food Prot. 2013, 76, 1145–1151. [Google Scholar] [CrossRef]
- Riggio, G.; Jones, S.; Gibson, K. Risk of Human Pathogen Internalization in Leafy Vegetables During Lab-Scale Hydroponic Cultivation. Horticulturae 2019, 5, 25. [Google Scholar] [CrossRef]
- Santos, M.I.S.; Marques, C.; Mota, J.; Pedroso, L.; Lima, A. Applications of Essential Oils as Antibacterial Agents in Minimally Processed Fruits and Vegetables—A Review. Microorganisms 2022, 10, 760. [Google Scholar] [CrossRef]
- Yaron, S.; Römling, U. Biofilm formation by enteric pathogens and its role in plant colonization and persistence. Microb. Biotechnol. 2014, 7, 496–516. [Google Scholar] [CrossRef]
- Coroneo, V.; Carraro, V.; Marras, B.; Marrucci, A.; Succa, S.; Meloni, B.; Pinna, A.; Angioni, A.; Sanna, A.; Schintu, M. Presence of Trihalomethanes in ready-to-eat vegetables disinfected with chlorine. Food Addit. Contam. Part A 2017, 34, 2111–2117. [Google Scholar] [CrossRef]
- Matsuzaki, S.; Azuma, K.; Lin, X.; Kuragano, M.; Uwai, K.; Yamanaka, S.; Tokuraku, K. Farm use of calcium hydroxide as an effective barrier against pathogens. Sci. Rep. 2021, 11, 7941. [Google Scholar] [CrossRef] [PubMed]
- Birmpa, A.; Sfika, V.; Vantarakis, A. Ultraviolet light and Ultrasound as non-thermal treatments for the inactivation of microorganisms in fresh ready-to-eat foods. Int. J. Food Microbiol. 2013, 167, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Mahendran, R.; Ramanan, K.R.; Barba, F.J.; Lorenzo, J.M.; López-Fernández, O.; Munekata, P.E.; Roohinejad, S.; Sant’Ana, A.S.; Tiwari, B.K. Recent advances in the application of pulsed light processing for improving food safety and increasing shelf life. Trends Food Sci. Technol. 2019, 88, 67–79. [Google Scholar] [CrossRef]
- Fan, K.; Zhang, M.; Bhandari, B.; Jiang, F. A combination treatment of ultrasound and ε-polylysine to improve microorganisms and storage quality of fresh-cut lettuce. LWT 2019, 113, 108315. [Google Scholar] [CrossRef]
- Carvalho, F.; Coimbra, A.T.; Silva, L.; Duarte, A.P.; Ferreira, S. Melissa officinalis essential oil as an antimicrobial agent against Listeria monocytogenes in watermelon juice. Food Microbiol. 2023, 109, 104105. [Google Scholar] [CrossRef]
- Gyawali, R.; Ibrahim, S.A. Natural products as antimicrobial agents. Food Control 2014, 46, 412–429. [Google Scholar] [CrossRef]
- Hayek, S.A.; Gyawali, R.; Ibrahim, S.A. Antimicrobial natural products. In Microbial Pathogens and Strategies for Combating Them: Science, Technology, and Education; Mendéz-Villas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2013; pp. 910–921. Available online: https://tarjomefa.com/wp-content/uploads/2016/11/5690-English.pdf (accessed on 2 December 2022).
- Santos, M.; Martins, S.; Pedroso, L.; Sousa, I.; Ferreira, M.A.S.S. Potential bio-activity of whey fermented extract as sanitizer of organic grown lettuce. Food Control 2015, 50, 477–481. [Google Scholar] [CrossRef]
- Santos, M.I.; Lima, A.I.; Monteiro, S.A.; Ferreira, R.M.; Pedroso, L.; Sousa, I.; Ferreira, M.A.S.S. Preliminary Study on the Effect of Fermented Cheese Whey on Listeria monocytogenes, Escherichia coli O157:H7, and Salmonella Goldcoast Populations Inoculated onto Fresh Organic Lettuce. Foodborne Pathog. Dis. 2016, 13, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Meireles, A.; Giaouris, E.; Simões, M. Alternative disinfection methods to chlorine for use in the fresh-cut industry. Food Res. Int. 2016, 82, 71–85. [Google Scholar] [CrossRef]
- Santos, M.I.S.; Fradinho, P.; Martins, S.; Lima, A.I.G.; Ferreira, R.M.S.B.; Pedroso, L.; Ferreira, M.A.S.S.; Sousa, I. A Novel Way for Whey: Cheese Whey Fermentation Produces an Effective and Environmentally-Safe Alternative to Chlorine. Appl. Sci. 2019, 9, 2800. [Google Scholar] [CrossRef]
- Madhusudan, P.; Chellukuri, N.; Shivakumar, N. Smart packaging of food for the 21st century—A review with futuristic trends, their feasibility and economics. Mater. Today Proc. 2018, 5, 21018–21022. [Google Scholar] [CrossRef]
- Young, E.; Mirosa, M.; Bremer, P. A Systematic Review of Consumer Perceptions of Smart Packaging Technologies for Food. Front. Sustain. Food Syst. 2020, 4, 63. [Google Scholar] [CrossRef]
- Bánáti, D. European perspectives of food safety. J. Sci. Food Agric. 2014, 94, 1941–1946. [Google Scholar] [CrossRef] [PubMed]
- Gizaw, Z. Public health risks related to food safety issues in the food market: A systematic literature review. Environ. Health Prev. Med. 2019, 24, 68. [Google Scholar] [CrossRef] [PubMed]
- van Schothorst, M. Microbiological Risk Assessment of foods in international trade. Saf. Sci. 2002, 40, 359–382. [Google Scholar] [CrossRef]
- Mathews, K.H., Jr.; Bernstein, J.; Buzby, J.C. International trade of meat/poultry products and food safety issues. In International Trade and Food Safety: Economic Theory and Case Studies; Agricultural Economic Report No 828; Buzby, J.C., Ed.; Economic Research Service: Washington, DC, USA, 2003; pp. 48–73. [Google Scholar]
- Lang, T. The new globalisation, food and health: Is public health receiving its due emphasis? J. Epidemiol. Community Health 1998, 52, 538–539. [Google Scholar] [CrossRef] [PubMed][Green Version]
- King, L.J. One Health and Food Safety. IOM (Institute of Medicine). In Improving Food Safety through a One Health Approach; The National Academies Press: Washington, DC, USA, 2012. [Google Scholar]
- Boqvist, S.; Söderqvist, K.; Vågsholm, I. Food safety challenges and One Health within Europe. Acta Vet. Scand. 2018, 60, 1. [Google Scholar] [CrossRef]

| Changes in Industrial Production | Changes in the Consumption Habits | Others |
|---|---|---|
| Increasingly broad and centralized production | Ongoing trend toward greater consumption of foods not prepared at home | Increased proportion of vulnerable population (elderly, immunocompromised, chronic patients) |
| Distribution of products over large distances | Increase in salad bar popularity | Improved epidemiological surveillance |
| Increasing popularity of minimal processed products | Growing consumption of fresh fruits, vegetables, and natural juices | Enhance diagnostic and pathogens identification and traceability |
| Growth in global trade of fresh produce all over the world | Growing interest for healthier diets | Emerging pathogens with new skills and low infectious doses |
| Fresh Cut Vegetables | Mesophilic Aerobic Microorganisms (log cfu.g−1) | Reference |
|---|---|---|
| Salads | 5.5–7.4 | [90] |
| Vegetables | 5.3–7.5 | [90] |
| Salads | 3.0–6.6 | [91] |
| Lettuce | 4.57–6.78 | [92] |
| Vegetables | 5.47–7.82 | [92] |
| Salads | 3.8–9.4 | [93] |
| Iceberg lettuce | 6.03–8.43 | [94] |
| Salads | 5.8–7.1 | [95] |
| Salads | 2.36–9.30 | [96] |
| Romaine Lettuce | 5.71–7.89 | [97] |
| Fresh Cut Vegetables | Enterobacteriaceae (E) or Coliforms (C) log ufc.g−1 | Escherichia coli % of Positive | References |
|---|---|---|---|
| Salads | 1.9–6.0 (C) | 30.0 | [90] |
| Vegetables | <1.0–> 5.5 (C) | 9.4 | [90] |
| Salads | <0.48–3.1 (C) | - | [91] |
| Salads | - | 10.0 | [104] |
| Lettuce | 2.18–5.66 (C) | 25.7 | [92] |
| Vegetables | 0.48–> 5.04 (C) | 16.0 | [92] |
| Salads | 4.15–6.15 (C) | 4 | [93] |
| Iceberg lettuce | - | 0 | [94] |
| Salads | 4.4–6.9 (E) | 6.7 | [95] |
| Salads | 1.30–7.48 (E) | 50 | [96] |
| RTE * Product | Microorganisms | |||||
|---|---|---|---|---|---|---|
| Salmonella spp. | L. monocytogenes | STEC ** | ||||
| N° of Samples | % + | N° of Samples | % + | N° of Samples | % + | |
| Fruit, vegetables, and juices | 6261 | 0.05 | 1383 | 3.0 | 1922 | 0.52 |
| Salads | 2194 | 0.05 | 844 | 0.95 | 301 | 0 |
| Spices and herbs | 1529 | 0.72 | 115 | 0 | 296 | 0.34 |
| Sprouted seeds | 512 | 0 | - | - | 617 | 0.16 |
| Product | Microorganisms | ||
|---|---|---|---|
| Salmonella spp. | E. coli O157 | L. monocytogenes | |
| Fruits | 14.9 | 3.2 | 24.8 |
| Seeded vegetables | 12.0 | 1.2 | - |
| Other produce | 8.6 | 2.6 | 12.3 |
| Vegetable row crops | 4.1 | 58.1 | 14.1 |
| Sprouts | 3.7 | 1.5 | 2.9 |
| Grains—beans | 0.9 | 0.9 | - |
| Total | 44.2 | 67.5 | 54.1 |
| Year | Product | Microorganism | Involved States | No. Cases | No. Hospitaliz. | No. Deaths |
|---|---|---|---|---|---|---|
| 2018 | Frozen Shredded Coconut | Salmonella I 4,[5],12:b:- and Salmonella Newport | 9 | 27 | 6 | 0 |
| Raw Sprouts | Salmonella Montevideo | 3 | 10 | 0 | 0 | |
| Dried Coconut | Salmonella Typhimurium | 8 | 14 | 3 | 0 | |
| Romaine Lettuce | Escherichia coli O157:H7 | 36 | 210 | 96 | 5 | |
| Pre-Cut Melon | Salmonella Adelaide | 9 | 77 | 36 | 0 | |
| Fresh Produce Vegetable Trays | Cyclospora | 4 | 250 | 8 | 0 | |
| Fresh Express Salad Mix | Cyclospora | 16 | 511 | 24 | 0 | |
| Romaine Lettuce | Escherichia coli O157:H7 | 16 | 62 | 25 | 0 | |
| 2019 | Pre-Cut Melons | Salmonella Carrau | 10 | 137 | 38 | 0 |
| Fresh Papayas | Salmonella Uganda | 9 | 81 | 27 | 0 | |
| Fresh Basil | Cyclospora | 11 | 241 | 6 | 0 | |
| Romaine Lettuce | Escherichia coli O157:H7 | 27 | 167 | 85 | 0 | |
| Salad Kits | Escherichia coli O157:H7 | 5 | 10 | 4 | 0 | |
| Cut Fruit | Salmonella Javiana | 14 | 165 | 73 | 0 | |
| 2020 | Clover Sprouts | Escherichia coli O103 | 10 | 51 | 3 | 0 |
| Enoki Mushrooms | Listeria monocytogenes | 17 | 36 | 31 | 4 | |
| Bagged Salad Mix | Cyclospora | 14 | 701 | 38 | 0 | |
| Onions | Salmonella Newport | 48 | 1127 | 167 | 0 | |
| Peaches | Salmonella Enteritidis | 17 | 101 | 28 | 0 | |
| Wood Ear Mushrooms | Salmonella Stanley | 12 | 55 | 6 | 0 | |
| Leafy Greens | Escherichia coli O157:H7 | 19 | 40 | 20 | 0 | |
| 2021 | Packaged Salad Greens | Salmonella Typhimurium | 4 | 31 | 4 | 0 |
| Onions | Salmonella Oranienburg | 39 | 1040 | 260 | 0 | |
| Baby Spinach | Escherichia coli O157:H7 | 10 | 15 | 4 | 0 | |
| Packaged Salads | Listeria monocytogenes | 13 | 18 | 16 | 3 | |
| Packaged Salads | Listeria monocytogenes | 8 | 10 | 10 | 1 | |
| Packaged Salads | Escherichia coli O157:H7 | 4 | 10 | 4 | 1 | |
| 2022 | Strawberries | Hepatitis A Virus | 4 | 19 | 13 | 0 |
| Frozen Falafel | Escherichia coli O121 | 6 | 24 | 6 | 0 | |
| Alfalfa Sprouts | Salmonella Typhimurium | 8 | 63 | 10 | 0 | |
| 2023 | Leafy Greens | Listeria monocytogenes | 16 | 19 | 18 | 0 |
| Frozen Strawberries | Hepatitis A | 4 | 10 | 4 | 0 |
| Country | Product | % Positive | Reference |
|---|---|---|---|
| Scotland | Ready-to-Eat Salads | 7.5% (3/40) | [132] |
| Canada | Vegetables | 4.5% (5/111) | [122] |
| France | Ready-to-Eat Salads and Raw Vegetables | 3.3% (3/104) | [128] |
| USA | Vegetable Products | 2.4% (3/125) | [133] |
| Iran | Ready-to-Eat Salads | 6% (6/106) | [131] |
| Australia | Root Vegetables | 10% (30/300) | [124] |
| Japan | Fresh Vegetables | 3.3% (8/242) | [125] |
| Disinfection Method | Effect | Advantages | Disadvantages | References |
|---|---|---|---|---|
| Chlorine | Antimicrobial efficacy related to oxidation capacity by a short contact time in chilled water | Ease and economic application | High amount of highly polluted wastewater Occurrence of residues of trihalomethanes and chloramines Banned in some European countries | [7,43] |
| Chlorine dioxide | Antimicrobial efficacy related to higher oxidative capacity than chlorine | No reaction with nitrogen-containing compounds to form carcinogenic by-products | Requires long exposure time, which affects the organoleptic properties of the product | [4,7,44] |
| Organic acids | Reduction in internal cellular pH, disruption of membrane transport and permeability Anion accumulation | Washes of lactic, citric, acetic, tartaric, and ascorbic acid rapidly inactivate a broad spectrum of bacteria Maintain the products’ quality Considered GRAS | Low antimicrobial efficacy | [4,7,43] |
| Hydrogen peroxide | Strong oxidizing power Generates other cytotoxic oxidizing species such as hydroxyl radicals | Excellent disinfectant agent mostly employed into post-harvest facilities (spaces and materials) Used in preventing post-harvest losses in table grapes, potatoes, strawberries, and lemons rather than in disinfecting fresh and MPFV produce | Reactive oxygen species being toxic to living cells Causes browning of shredded lettuce | [7,44] |
| Ozonated water | Strong oxidative and microbial agent | Allows extended shelf life High reactivity Spontaneously decomposes to oxygen, leaving no residues on treated produce Active against bacteria, fungi, virus, and bacterial and fungal spores Considered GRAS | Long exposure time is needed Corrosiveness of products Capital cost | [4,7,43,44] |
| Electrolyzed water (EW) | Has a strong bactericidal effect | Low operational expenses Easy operation Safe and eco-friendly No significant alteration of product quality Reduced concentration of chlorine in the wash water | High cost and limited availability of equipment Very short shelf life and requires on-site generation Corrosive hazard of strong acid EW Cl2 production | [4,7,44] |
| Calcium-based solutions | Maintain the vegetable cell wall integrity Inhibit plant tissue senescence Have antibacterial properties Calcium salts have been used as a firming agent for fruit | Allows extended shelf life The final product can significantly increase the calcium content | Limited efficacy as antimicrobial Bitterness and off flavors associated with calcium chloride May be too expensive | [7] |
| Ionizing radiation | Reduces bacteria, yeast, molds, parasites, protozoa, and insects Inactivates genetic material of the living cells | Environmentally friendly and time effective Ionizing irradiation shows beneficial effects in reducing the microbial population Extend the shelf life and maintain the quality of fruits and vegetables. Leaves no residue on the food | A high-dose irradiation is required Quality may be affected Texture alteration | [4,7,40] |
| Ultraviolet | Strong antibacterial agent due to genetic damage Induction of resistance mechanisms in different fruit and vegetables against pathogens | Relatively inexpensive and easy-to-use equipment Broad-spectrum bactericidal effect Allows extended shelf life Environmentally friendly | Can cause damage to the treated tissue and increased stress and respiration rate Induces a lignifications-like process Low penetration of UV light Long treatment times Negative impact on product sensory quality Low efficacy at high organic matter level Complex standardization of application at commercial scale | [4,7,43,149] |
| Modified atmosphere packaging (MAP) | Low levels of O2 and high levels of CO2 reduce the produce respiration rate, with the benefit of delaying senescence | Allows extended shelf life Preserves quality Fresh-cut products are more tolerant to higher CO2 concentrations than intact products | Changes of the gas composition Fermentation and formation of off flavor compounds May allow growth of pathogenic bacteria | [7,40] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, M.I.; Grácio, M.; Silva, M.C.; Pedroso, L.; Lima, A. One Health Perspectives on Food Safety in Minimally Processed Vegetables and Fruits: From Farm to Fork. Microorganisms 2023, 11, 2990. https://doi.org/10.3390/microorganisms11122990
Santos MI, Grácio M, Silva MC, Pedroso L, Lima A. One Health Perspectives on Food Safety in Minimally Processed Vegetables and Fruits: From Farm to Fork. Microorganisms. 2023; 11(12):2990. https://doi.org/10.3390/microorganisms11122990
Chicago/Turabian StyleSantos, Maria Isabel, Madalena Grácio, Mariana Camoesas Silva, Laurentina Pedroso, and Ana Lima. 2023. "One Health Perspectives on Food Safety in Minimally Processed Vegetables and Fruits: From Farm to Fork" Microorganisms 11, no. 12: 2990. https://doi.org/10.3390/microorganisms11122990
APA StyleSantos, M. I., Grácio, M., Silva, M. C., Pedroso, L., & Lima, A. (2023). One Health Perspectives on Food Safety in Minimally Processed Vegetables and Fruits: From Farm to Fork. Microorganisms, 11(12), 2990. https://doi.org/10.3390/microorganisms11122990








