Characterization of Three New Outer Membrane Adhesion Proteins in Fusobacterium necrophorum
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Cultures and Conditions
2.2. Bovine Endothelial Cell Line
2.3. OMP Extraction
2.4. Binding Assays
2.5. Pull-Down Assays
2.6. Bioinformatics
2.7. Cloning, Expression, and Purification of Recombinant OMPs
2.8. Enzyme-Linked Immunosorbent Assays (ELISAs)
2.9. Adhesion Inhibition Assays
2.10. Statistical Analysis
3. Results
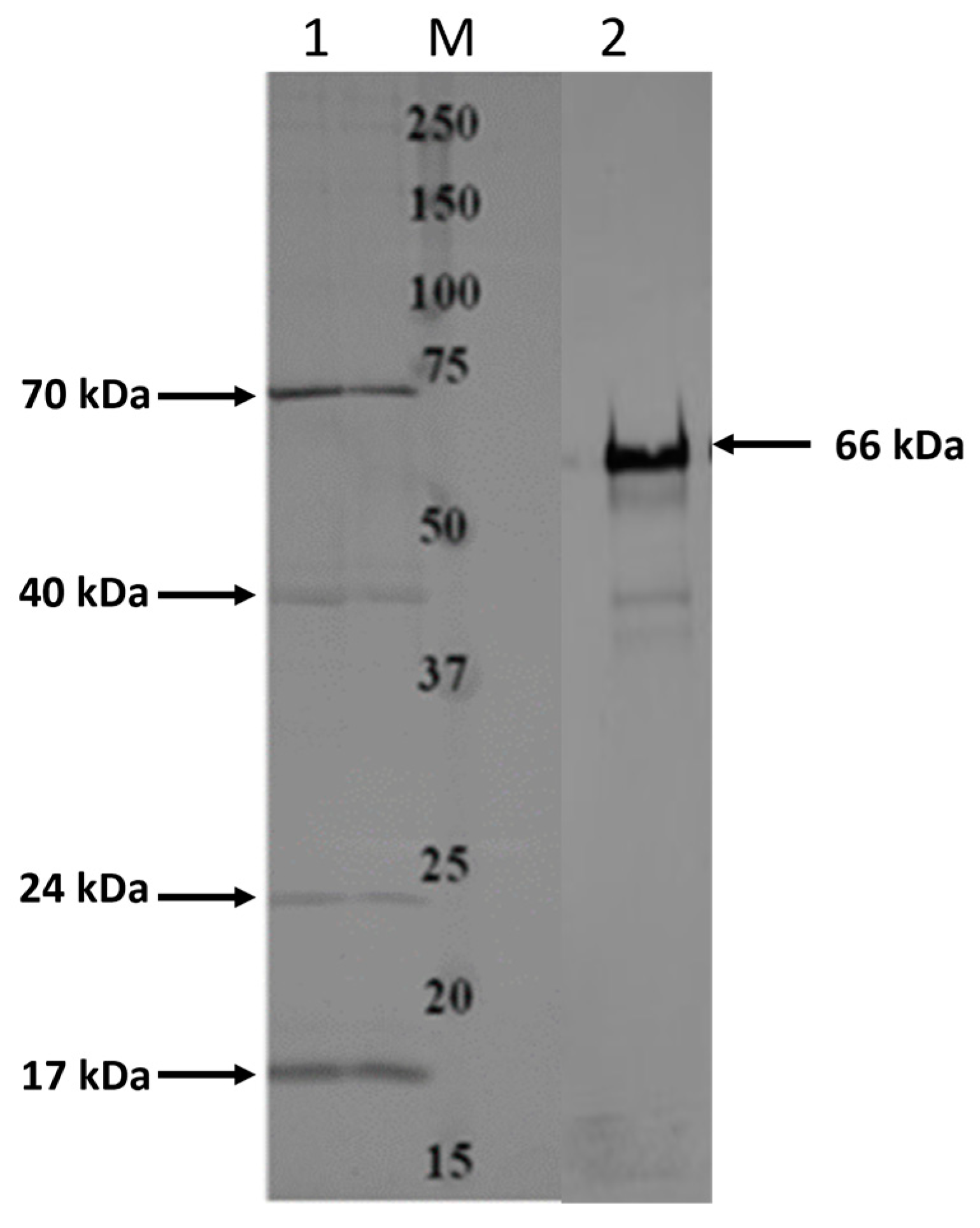
3.1. Identification and Characterization of OMPs from F. necrophorum subsp. necrophorum with the Ability to Bind a Host Endothelial Cell Line
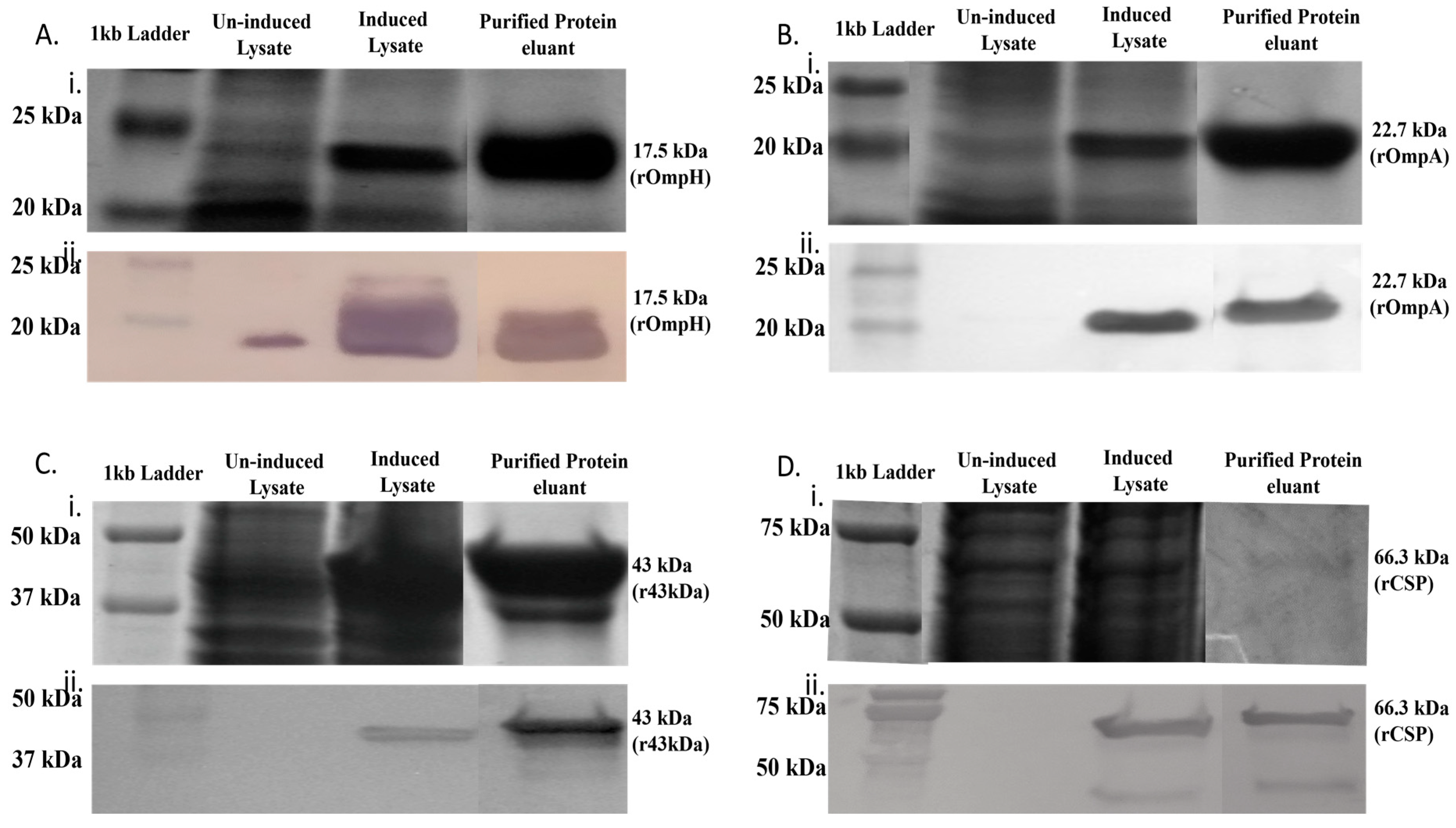
3.2. Expression and Purification of Recombinant OMPs from F. necrophorum
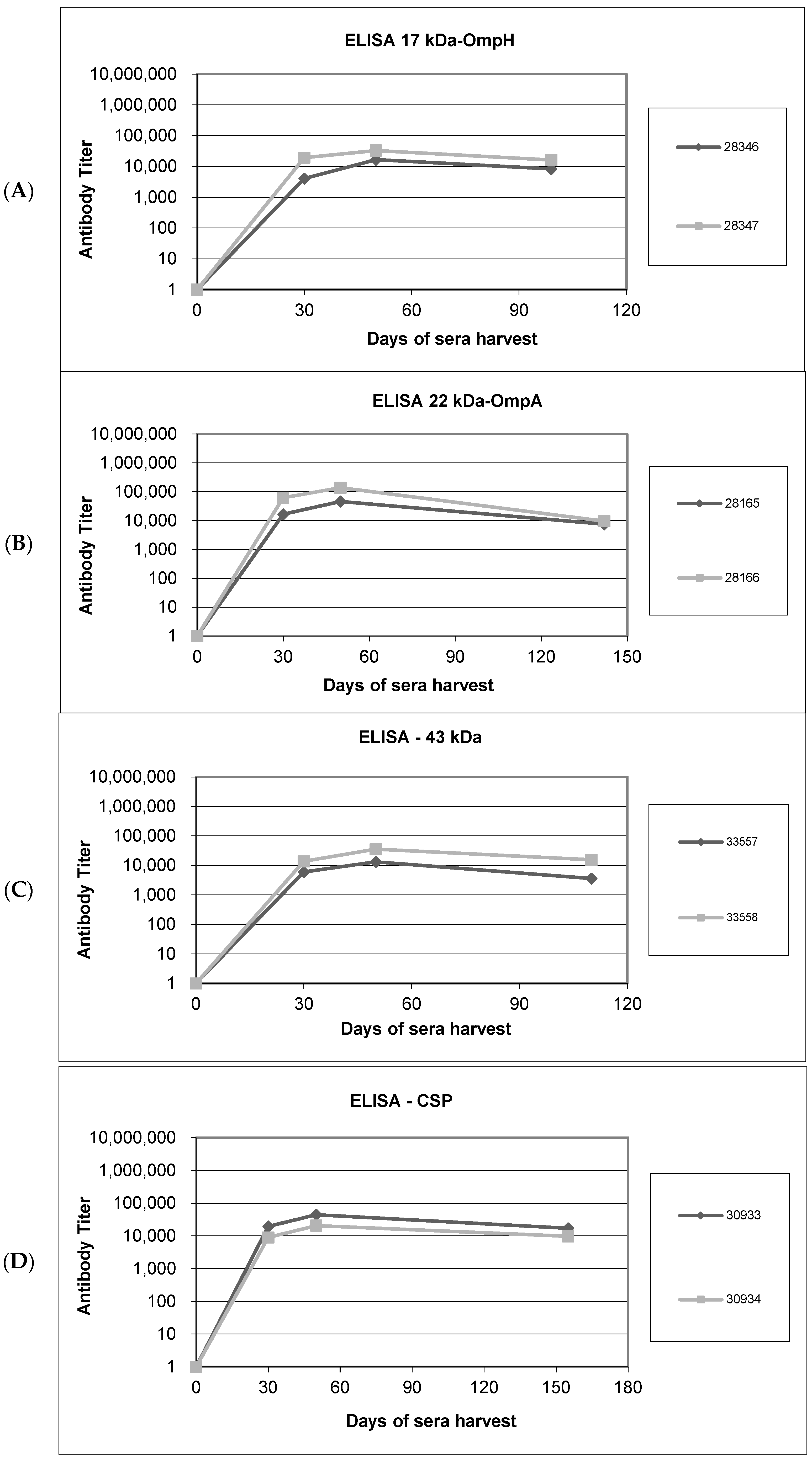
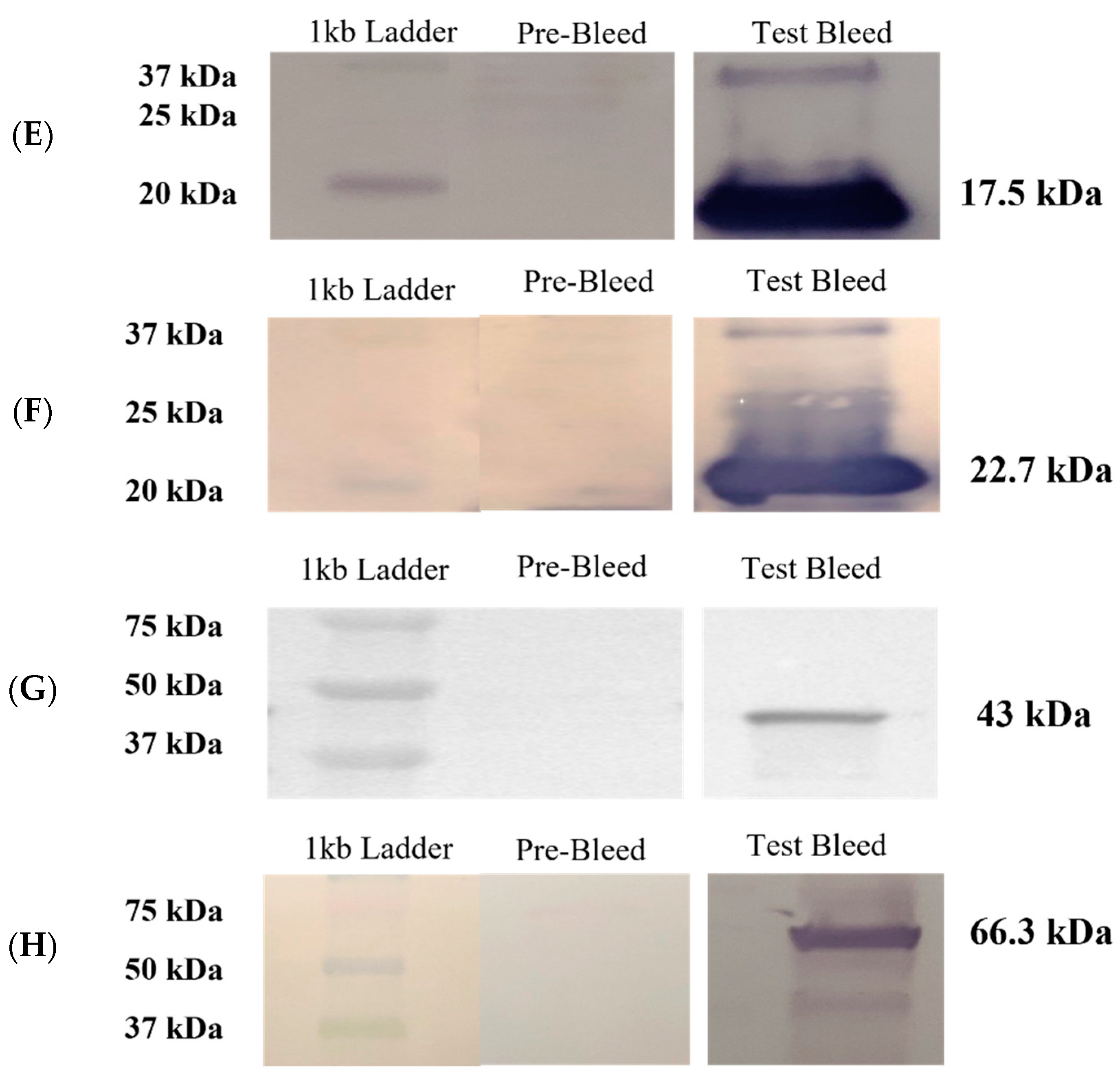
3.3. Humoral Responses to Purified Recombinant Proteins in Immunized Rabbits and Immunoblotting
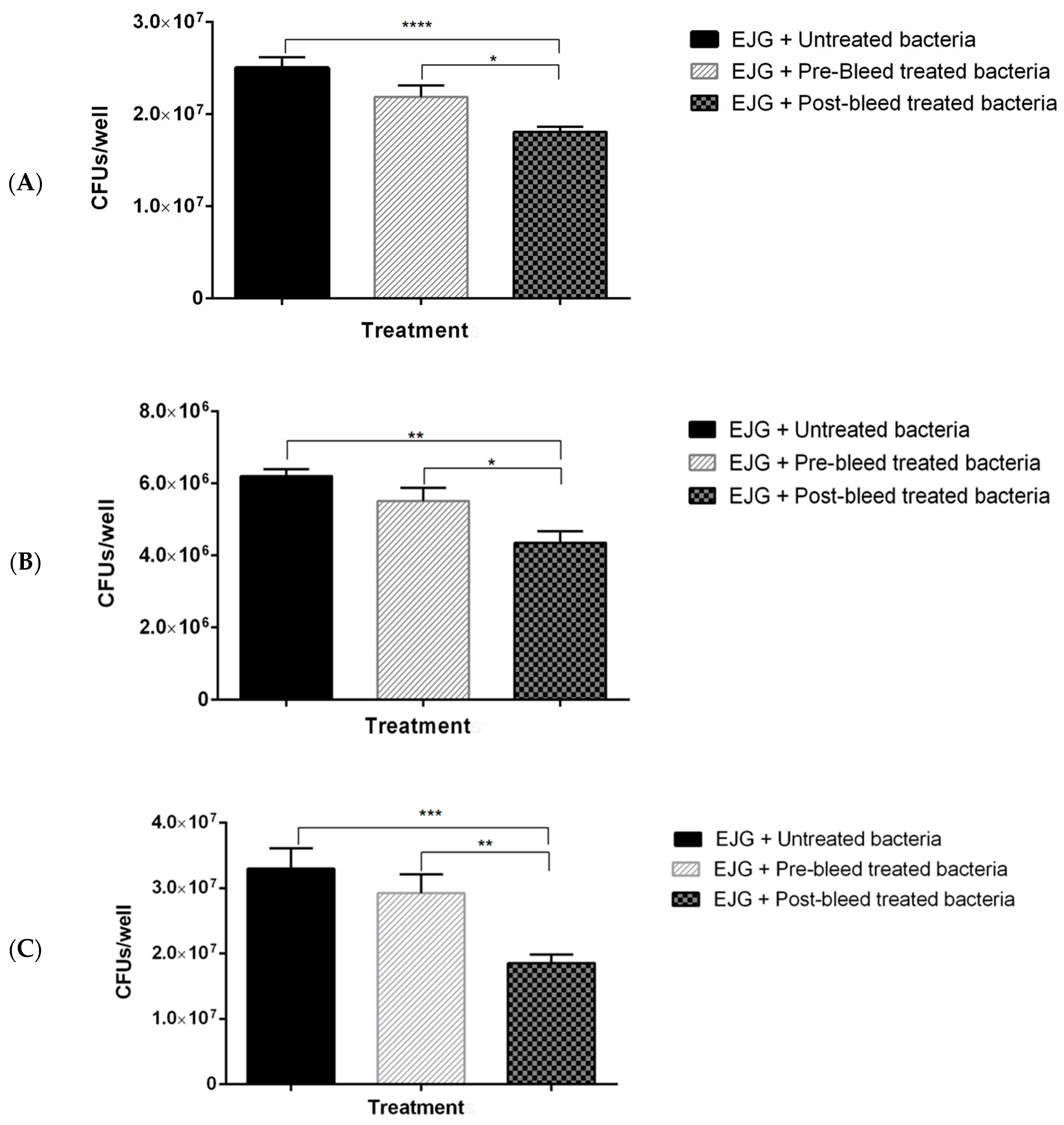
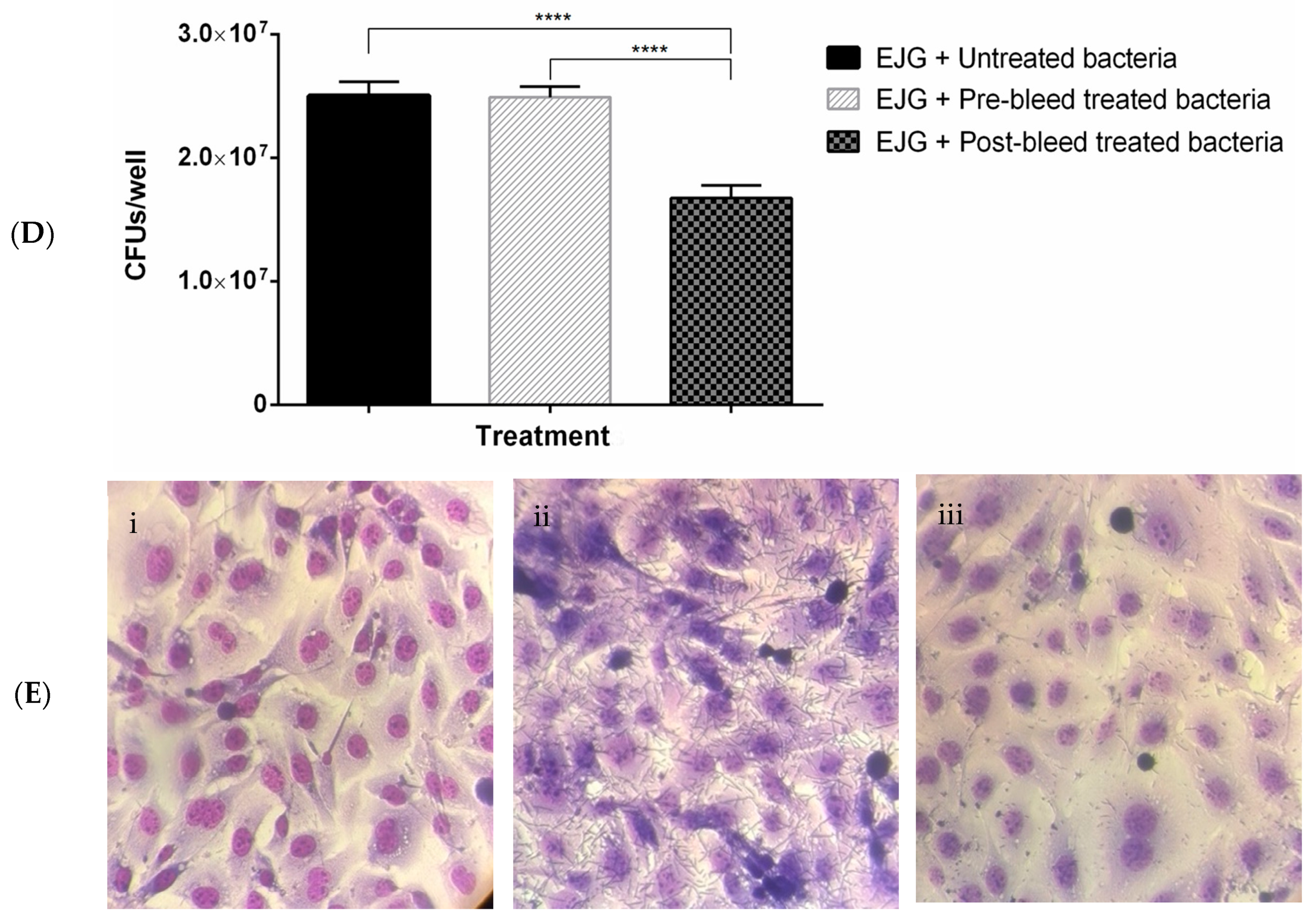
3.4. In Vitro Adhesion Inhibition Assay
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nagaraja, T.G.; Narayanan, S.K.; Stewart, G.C.; Chengappa, M.M. Fusobacterium Necrophorum Infections in Animals: Pathogenesis and Pathogenic Mechanisms. Anaerobe 2005, 11, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Tadepalli, S.; Narayanan, S.K.; Stewart, G.C.; Chengappa, M.M.; Nagaraja, T.G. Fusobacterium Necrophorum: A Ruminal Bacterium That Invades Liver to Cause Abscesses in Cattle. Anaerobe 2009, 15, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Clifton, R.; Giebel, K.; Liu, N.L.B.H.; Purdy, K.J.; Green, L.E. Sites of Persistence of Fusobacterium Necrophorum and Dichelobacter Nodosus: A Paradigm Shift in Understanding the Epidemiology of Footrot in Sheep. Sci. Rep. 2019, 9, 14429. [Google Scholar] [CrossRef] [PubMed]
- Nyack, B.; Craig, I.; Padmore, C.; Bernard, N. Necrotic Laryngitis in a Calf. Mod. Vet. Pr. 1981, 62, 937–938. [Google Scholar]
- McCoy, E.J.; O’Quinn, T.G.; Schwandt, E.F.; Reinhardt, C.D.; Thomson, D.U. Liver Abscess Severity at Slaughter Does Not Affect Meat Tenderness and Sensory Attributes in Commercially Finished Beef Cattle Fed Without Tylosin Phosphate. Kans. Agric. Exp. Stn. Res. Rep. 2017, 3, 33. [Google Scholar] [CrossRef]
- Harris, M.K.; Eastwood, L.C.; Boykin, C.A.; Arnold, A.N.; Gehring, K.B.; Hale, D.S.; Kerth, C.R.; Griffin, D.B.; Savell, J.W.; Belk, K.E.; et al. National Beef Quality Audit-2016: Assessment of Cattle Hide Characteristics, Offal Condemnations, and Carcass Traits to Determine the Quality Status of the Market Cow and Bull Beef Industry. Transl. Anim. Sci. 2018, 2, 37–49. [Google Scholar] [CrossRef]
- Beef Quality Assurance—BQA—National Beef Quality Audits. Available online: https://www.bqa.org/resources/national-beef-quality-audits (accessed on 27 October 2023).
- Nagaraja, T.G.; Sun, Y.; Wallace, N.; Kemp, K.E.; Parrott, C.J. Effects of Tylosin on Concentrations of Fusobacterium Necrophorum and Fermentation Products in the Rumen of Cattle Fed a High-Concentrate Diet. Am. J. Vet. Res. 1999, 60, 1061–1065. [Google Scholar]
- DiCostanzo, A. Promising Management Strategies and Feed Additives to Prevent Liver Abscesses and Disease. In Proceedings of the American Association of Bovine Practitioners Conference Proceedings, Salt Lake City, UT, USA, 7–9 October 2021; pp. 44–52. [Google Scholar]
- Amachawadi, R.G.; Bohney, S.; Nagaraja, T.G. Metabolome of Purulent Materials of Liver Abscesses from Crossbred Cattle and Holstein Steers Fed Finishing Diets with or without In-Feed Tylosin. J. Anim. Sci. 2023, 101, skac427. [Google Scholar] [CrossRef]
- Taylor, E.A.; Ossa-Trujillo, C.; Vinasco, J.; Jordan, E.R.; García Buitrago, J.A.; Hagevoort, R.; Norman, K.N.; Lawhon, S.D.; Piñeiro, J.M.; Levent, G.; et al. Use of Critically Important Antimicrobial Classes Early in Life May Adversely Impact Bacterial Resistance Profiles during Adult Years: Potential Co-Selection for Plasmid-Borne Fluoroquinolone and Macrolide Resistance via Extended-Spectrum Beta-Lactam Use in Dairy Cattle. Lett. Appl. Microbiol. 2021, 72, 220–224. [Google Scholar] [CrossRef]
- Foreman, J.H. Merck Veterinary Manual; Merck: New York, NY, USA, 2014. [Google Scholar]
- Solan, R.; Pereira, J.; Lupas, A.N.; Kolodny, R.; Ben-Tal, N. Gram-Negative Outer-Membrane Proteins with Multiple β-Barrel Domains. Proc. Natl. Acad. Sci. USA 2021, 118, e2104059118. [Google Scholar] [CrossRef]
- Koebnik, R.; Locher, K.P.; Van Gelder, P. Structure and Function of Bacterial Outer Membrane Proteins: Barrels in a Nutshell. Mol. Microbiol. 2000, 37, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Rollauer, S.E.; Sooreshjani, M.A.; Noinaj, N.; Buchanan, S.K. Outer Membrane Protein Biogenesis in Gram-Negative Bacteria. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20150023. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, J.D.; Kleinekathöfer, U.; Winterhalter, M. How to Enter a Bacterium: Bacterial Porins and the Permeation of Antibiotics. Chem. Rev. 2021, 121, 5158–5192. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Gart, E.; Nagaraja, T.G.; Narayanan, S. Adhesion of Fusobacterium Necrophorum to Bovine Endothelial Cells Is Mediated by Outer Membrane Proteins. Vet. Microbiol. 2013, 162, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Coppenhagen-Glazer, S.; Sol, A.; Abed, J.; Naor, R.; Zhang, X.; Han, Y.W.; Bachrach, G. Fap2 of Fusobacterium Nucleatum Is a Galactose-Inhibitable Adhesin Involved in Coaggregation, Cell Adhesion, and Preterm Birth. Infect. Immun. 2015, 83, 1104–1113. [Google Scholar] [CrossRef] [PubMed]
- Gur, C.; Ibrahim, Y.; Isaacson, B.; Yamin, R.; Abed, J.; Gamliel, M.; Enk, J.; Bar-On, Y.; Stanietsky-Kaynan, N.; Coppenhagen-Glazer, S.; et al. Binding of the Fap2 Protein of Fusobacterium Nucleatum to Human Inhibitory Receptor TIGIT Protects Tumors from Immune Cell Attack. Immunity 2015, 42, 344–355. [Google Scholar] [CrossRef] [PubMed]
- Thibau, A.; Dichter, A.A.; Vaca, D.J.; Linke, D.; Goldman, A.; Kempf, V.A.J. Immunogenicity of Trimeric Autotransporter Adhesins and Their Potential as Vaccine Targets. Med. Microbiol. Immunol. 2019, 209, 243–263. [Google Scholar] [CrossRef]
- Keshri, A.K.; Kaur, R.; Rawat, S.S.; Arora, N.; Pandey, R.K.; Kumbhar, B.V.; Mishra, A.; Tripathi, S.; Prasad, A. Designing and Development of Multi-Epitope Chimeric Vaccine against Helicobacter Pylori by Exploring Its Entire Immunogenic Epitopes: An Immunoinformatic Approach. BMC Bioinform. 2023, 24, 358. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, M.; Xu, Z.; He, Y.; Zhao, X.; Cheng, H.; Chen, X.; Xu, J.; Ding, Z. The Screening of the Protective Antigens of Aeromonas Hydrophila Using the Reverse Vaccinology Approach: Potential Candidates for Subunit Vaccine Development. Vaccines 2023, 11, 1266. [Google Scholar] [CrossRef]
- Hatfaludi, T.; Al-Hasani, K.; Boyce, J.D.; Adler, B. Outer Membrane Proteins of Pasteurella Multocida. Vet. Microbiol. 2010, 144, 1–17. [Google Scholar] [CrossRef]
- Nie, D.; Hu, Y.; Chen, Z.; Li, M.; Hou, Z.; Luo, X.; Mao, X.; Xue, X. Outer Membrane Protein A (OmpA) as a Potential Therapeutic Target for Acinetobacter Baumannii Infection. J. Biomed. Sci. 2020, 27, 26. [Google Scholar] [CrossRef]
- Narayanan, S.K.; Nagaraja, T.G.; Okwumabua, O.; Staats, J.; Chengappa, M.M.; Oberst, R.D. Ribotyping to Compare Fusobacterium Necrophorum Isolates from Bovine Liver Abscesses, Ruminal Walls, and Ruminal Contents. Appl. Environ. Microbiol. 1997, 63, 4671–4678. [Google Scholar] [CrossRef] [PubMed]
- Osborn, M.J.; Munson, R. Separation of the Inner (Cytoplasmic) and Outer Membranes of Gram-Negative Bacteria. Methods Enzymol. 1972, 3973, 642–653. [Google Scholar]
- Kumar, A.; Menon, S.; Nagaraja, T.G.; Narayanan, S. Identification of an Outer Membrane Protein of Fusobacterium Necrophorum Subsp. Necrophorum That Binds with High Affinity to Bovine Endothelial Cells. Vet. Microbiol. 2015, 176, 196–201. [Google Scholar] [CrossRef]
- Novinrooz, A.; Salehi, T.Z.; Firouzi, R.; Arabshahi, S.; Derakhshandeh, A. In-Silico Design, Expression, and Purification of Novel Chimeric Escherichia Coli O157:H7 OmpA Fused to LTB Protein in Escherichia Coli. PLoS ONE 2017, 12, e0173761. [Google Scholar] [CrossRef]
- Jones, G.; Jayappa, H.; Hunsaker, B.; Sweeney, D.; Rapp-Gabrielson, V.; Wasmoen, T.; Nagaraja, T.G.; Swingle, S.; Branine, M. Efficacy of an Arcanobacterium Pyogenes Fusobacterium Necrophorum Bacterin-Toxoid as an Aid in the Prevention of Liver Abscesses in Feedlot Cattle. Bov. Pract. 2004, 38, 36–44. [Google Scholar] [CrossRef]
- Saginala, S.; Nagaraja, T.G.; Tan, Z.L.; Lechtenberg, K.F.; Chengappa, M.M.; Hine, P.M. The Serum Neutralizing Antibody Response in Cattle to Fusobacterium Necrophorum Leukotoxoid and Possible Protection against Experimentally Induced Hepatic Abscesses. Vet. Res. Commun. 1996, 20, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Saginala, S.; Nagaraja, T.G.; Lechtenberg, K.F.; Chengappa, M.M.; Kemp, K.E.; Hine, P.M. Effect of Fusobacterium Necrophorum Leukotoxoid Vaccine on Susceptibility to Experimentally Induced Liver Abscesses in Cattle. J. Anim. Sci. 1997, 75, 1160–1166. [Google Scholar] [CrossRef]
- Haque, S.; Sengupta, S.; Khan, A.; Mukhopadhyay, A.K.; Bhan, M.K.; Kumar, R.; Jailkhani, B. Immune Response of S. Typhi-Derived Vi Polysaccharide and Outer Membrane Protein a Conjugate in Mice. Pediatr. Neonatol. 2023, 64, 518–527. [Google Scholar] [CrossRef]
- Zhang, B.-Z.; Hu, D.; Dou, Y.; Xiong, L.; Wang, X.; Hu, J.; Xing, S.-Z.; Li, W.; Cai, J.-P.; Jin, M.; et al. Identification and Evaluation of Recombinant Outer Membrane Proteins as Vaccine Candidates Against Klebsiella Pneumoniae. Front. Immunol. 2021, 12, 730116. [Google Scholar] [CrossRef]
- Vasfi Marandi, M.; Mittal, K.R. Role of Outer Membrane Protein H (OmpH)- and OmpA-Specific Monoclonal Antibodies from Hybridoma Tumors in Protection of Mice against Pasteurella Multocida. Infect. Immun. 1997, 65, 4502–4508. [Google Scholar] [CrossRef]
- Jaydari, A.; Forouharmehr, A.; Nazifi, N. Determination of Immunodominant Scaffolds of Com1 and OmpH Antigens of Coxiella Burnetii. Microb. Pathog. 2019, 126, 298–309. [Google Scholar] [CrossRef]
- Finco, O.; Bonci, A.; Agnusdei, M.; Scarselli, M.; Petracca, R.; Norais, N.; Ferrari, G.; Garaguso, I.; Donati, M.; Sambri, V.; et al. Identification of New Potential Vaccine Candidates against Chlamydia Pneumoniae by Multiple Screenings. Vaccine 2005, 23, 1178–1188. [Google Scholar] [CrossRef]
- Bista, P.K.; Pillai, D.; Roy, C.; Scaria, J.; Narayanan, S.K. Comparative Genomic Analysis of Fusobacterium Necrophorum Provides Insights into Conserved Virulence Genes. Microbiol. Spectr. 2022, 10, e00297-22. [Google Scholar] [CrossRef]
- Sthitmatee, N.; Yano, T.; Lampang, K.N.; Suphavilai, C.; Kataoka, Y.; Sawada, T. A 39-kDa Capsular Protein Is a Major Cross-Protection Factor as Demonstrated by Protection of Chickens with a Live Attenuated Pasteurella multocida Strain of P-1059. J. Vet. Med. Sci. 2013, 75, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Muenthaisong, A.; Nambooppha, B.; Rittipornlertrak, A.; Tankaew, P.; Varinrak, T.; Muangthai, K.; Atthikanyaphak, K.; Sawada, T.; Sthitmatee, N. An Intranasal Vaccination with a Recombinant Outer Membrane Protein H against Haemorrhagic Septicemia in Swamp Buffaloes. Vet. Med. Int. 2020, 2020, 3548973. [Google Scholar] [CrossRef] [PubMed]
- Sogasu, D.; Girija, A.S.S.; Gunasekaran, S.; Priyadharsini, J.V. Molecular Characterization and Epitope-Based Vaccine Predictions for ompA Gene Associated with Biofilm Formation in Multidrug-Resistant Strains of A.Baumannii. Silico Pharmacol. 2021, 9, 15. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Liao, Y.; Zhang, J.; Wang, Y.; Liu, Z.; Cheng, P.; Wang, X.; Zou, Q.; Gu, J. Rational Design and Evaluation of an Artificial Escherichia Coli K1 Protein Vaccine Candidate Based on the Structure of OmpA. Front. Cell. Infect. Microbiol. 2018, 8, 172. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Fu, F.; Li, X.; Chen, X.; Wang, W.; Lang, Y.; Cong, F.; Liu, C.; Tong, G.; Li, X. Identification of the Immunogenic Outer Membrane Protein A Antigen of Haemophilus Parasuis by a Proteomics Approach and Passive Immunization with Monoclonal Antibodies in Mice. Clin. Vaccine Immunol. CVI 2011, 18, 1695–1701. [Google Scholar] [CrossRef]
- Ahn, J.; Hong, M.; Yoo, S.; Lee, E.; Won, H.; Yoon, I.; Jung, J.; Lee, H. Soluble Expression of OmpA from Haemophilus Parasuis in Escherichia Coli and Its Protective Effects in the Mouse Model of Infection. J. Microbiol. Biotechnol. 2012, 22, 1307–1309. [Google Scholar] [CrossRef]
- Smith, S.G.J.; Mahon, V.; Lambert, M.A.; Fagan, R.P. A Molecular Swiss Army Knife: OmpA Structure, Function and Expression. FEMS Microbiol. Lett. 2007, 273, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Orme, R.; Douglas, C.W.I.; Rimmer, S.; Webb, M. Proteomic Analysis of Escherichia Coli Biofilms Reveals the Overexpression of the Outer Membrane Protein OmpA. Proteomics 2006, 6, 4269–4277. [Google Scholar] [CrossRef] [PubMed]
- Chalifour, A.; Jeannin, P.; Gauchat, J.-F.; Blaecke, A.; Malissard, M.; N’Guyen, T.; Thieblemont, N.; Delneste, Y. Direct Bacterial Protein PAMP Recognition by Human NK Cells Involves TLRs and Triggers α-Defensin Production. Blood 2004, 104, 1778–1783. [Google Scholar] [CrossRef] [PubMed]
- Lo, R.Y.C.; Sorensen, L.S. The Outer Membrane Protein OmpA of Mannheimia Haemolytica A1 Is Involved in the Binding of Fibronectin. FEMS Microbiol. Lett. 2007, 274, 226–231. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, J.; Hsieh, C.-L.; Gelincik, O.; Devolder, B.; Sei, S.; Zhang, S.; Lipkin, S.M.; Chang, Y.-F. Proteomic Characterization of Outer Membrane Vesicles from Gut Mucosa-Derived Fusobacterium Nucleatum. J. Proteom. 2019, 195, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.-C.; Resman, F.; Hörhold, F.; Riesbeck, K. Comparative Genomic Analysis Reveals Distinct Genotypic Features of the Emerging Pathogen Haemophilus Influenzae Type f. BMC Genom. 2014, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Heise, T.; Dersch, P. Identification of a Domain in Yersinia Virulence Factor YadA That Is Crucial for Extracellular Matrix-Specific Cell Adhesion and Uptake. Proc. Natl. Acad. Sci. USA 2006, 103, 3375–3380. [Google Scholar] [CrossRef]
- Xiao, J.; Jiang, J.; He, X.; Zhang, S.; Wang, Z.; Wang, F.; Wang, L.; Guo, D. Evaluation of Immunoprotective Effects of Fusobacterium Necrophorum Outer Membrane Proteins 43K OMP, Leukotoxin and Hemolysin Multi-Component Recombinant Subunit Vaccine in Mice. Front. Vet. Sci. 2021, 8, 780377. [Google Scholar] [CrossRef]
- Asadi, A.; Razavi, S.; Talebi, M.; Gholami, M. A Review on Anti-Adhesion Therapies of Bacterial Diseases. Infection 2019, 47, 13–23. [Google Scholar] [CrossRef]
- Pen, G.; Yang, N.; Teng, D.; Hao, Y.; Mao, R.; Wang, J. The Outer Membrane Proteins and Their Synergy Triggered the Protective Effects against Pathogenic Escherichia Coli. Microorganisms 2022, 10, 982. [Google Scholar] [CrossRef]
| Gene | Primer | Tm |
|---|---|---|
| 17 kDa F | 5’ TAGGATCCAGATAAAATTGCAGTGGTG 3’ | 56.9 °C |
| 17 kDa R | 5’ CGCTGCAGTTATTTTATTGTTTCCAT 3’ | 54.8 °C |
| 22 kDa F | 5’ TAGGATCCCGAAGAAGGAAATATAAAC 3’ | 54.3 °C |
| 22 kDa R | 5’ CGCTGCAGCTAGTTATATTTTGGAGC 3’ | 58.1 °C |
| 43 kDa F | 5’ TTGGGATCCTAAAGAAGTGATGCCTGCT 3’ | 61.5 °C |
| 43 kDa R | 5’ ACCTGCAGTTAGAAAGTAACTTTCATACC 3’ | 56.8 °C |
| CSP F | 5’ GCCGGATCCAGAAGATCCGGTAATAAAAAGA 3’ | 61.2 °C |
| CSP R | 5’ ACCTGCAGTTATTTGTTTATTAATTCTTC 3’ | 52.9 °C |
| Accession | Description | # AAs | MW (kDa) |
|---|---|---|---|
| WP_035904032.1 | OmpH family protein | 159 | 17.5 |
| WP_005961133.1 | OmpA family protein | 217 | 22.7 |
| JQ740821.1 | 43 kDa outer membrane protein | 377 | 42.9 |
| WP_035916891.1 | Cell surface protein | 638 | 66.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bista, P.K.; Pillai, D.; Narayanan, S.K. Characterization of Three New Outer Membrane Adhesion Proteins in Fusobacterium necrophorum. Microorganisms 2023, 11, 2968. https://doi.org/10.3390/microorganisms11122968
Bista PK, Pillai D, Narayanan SK. Characterization of Three New Outer Membrane Adhesion Proteins in Fusobacterium necrophorum. Microorganisms. 2023; 11(12):2968. https://doi.org/10.3390/microorganisms11122968
Chicago/Turabian StyleBista, Prabha K., Deepti Pillai, and Sanjeev K. Narayanan. 2023. "Characterization of Three New Outer Membrane Adhesion Proteins in Fusobacterium necrophorum" Microorganisms 11, no. 12: 2968. https://doi.org/10.3390/microorganisms11122968
APA StyleBista, P. K., Pillai, D., & Narayanan, S. K. (2023). Characterization of Three New Outer Membrane Adhesion Proteins in Fusobacterium necrophorum. Microorganisms, 11(12), 2968. https://doi.org/10.3390/microorganisms11122968





