Lomentospora prolificans: An Emerging Opportunistic Fungal Pathogen
Abstract
1. Introduction
2. Epidemiology
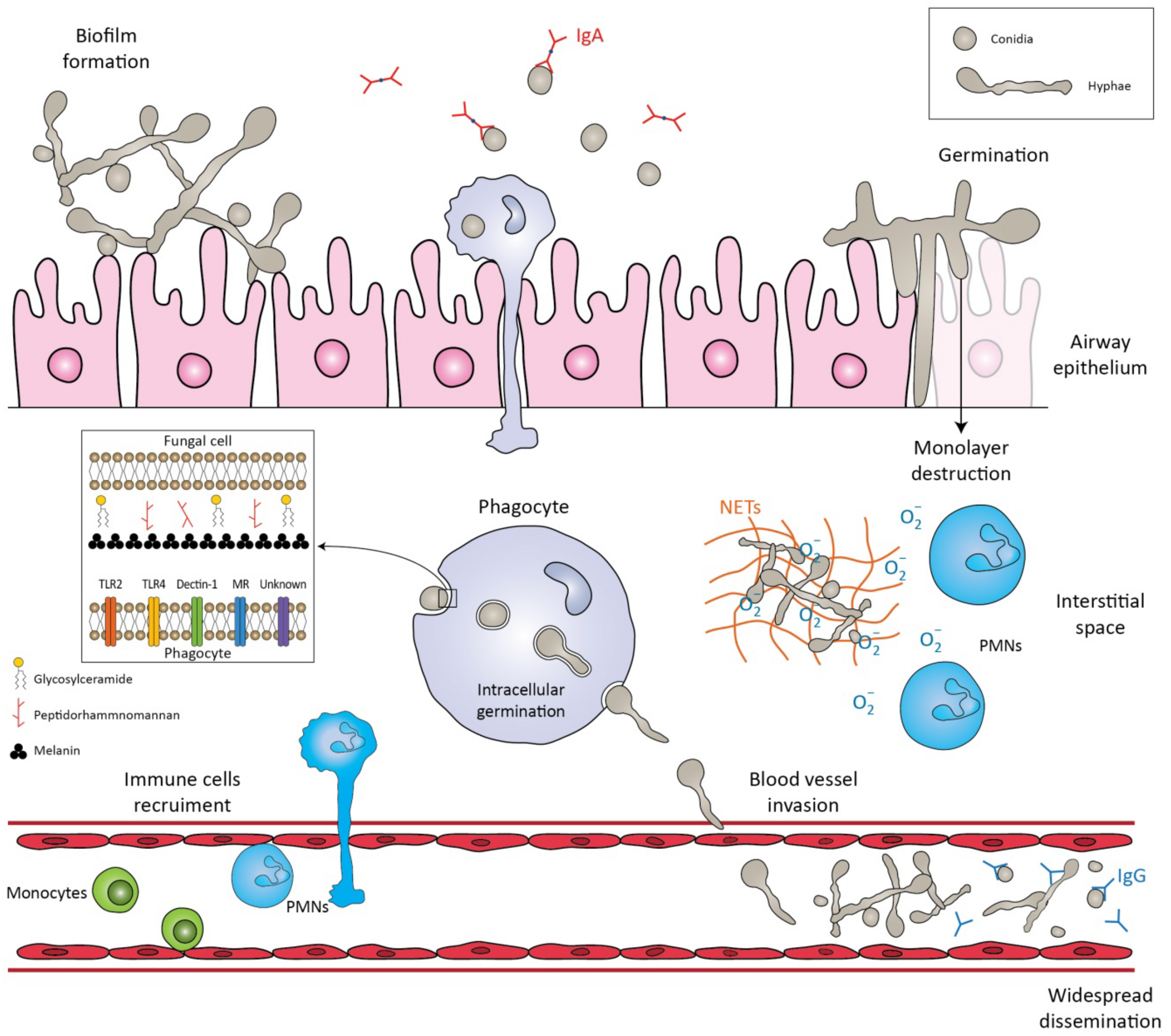
3. Pathogenesis and Host Defense
4. Clinical Presentation
5. Diagnosis
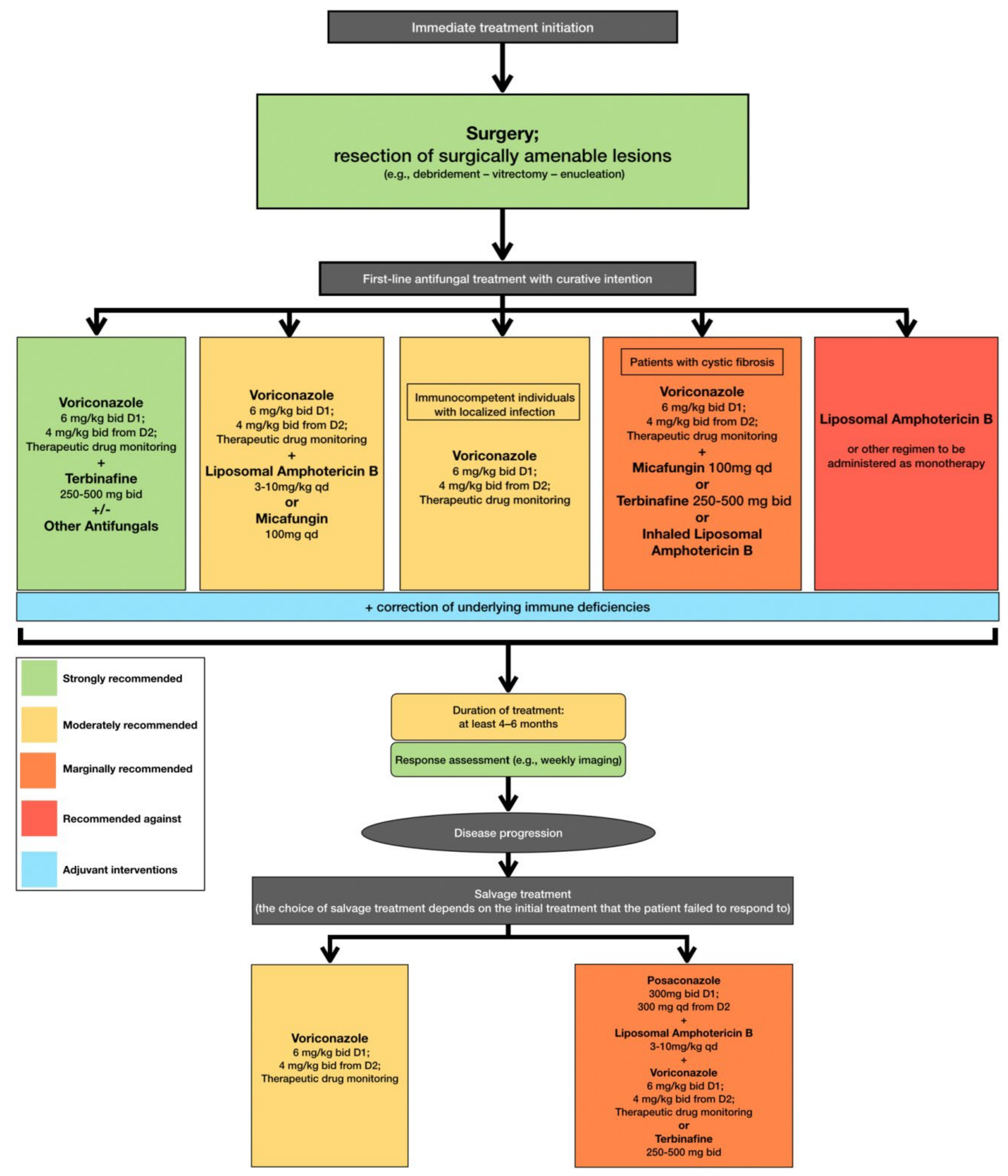
6. Antifungal Therapeutic Strategies
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodriguez-Tudela, J.L.; Berenguer, J.; Guarro, J.; Kantarcioglu, A.S.; Horre, R.; De Hoog, G.S.; Cuenca-Estrella, M. Epidemiology and outcome of Scedosporium prolificans infection, a review of 162 cases. Med. Mycol. 2009, 47, 359–370. [Google Scholar] [CrossRef]
- Hennebert Bj Desai, B.g. Lomentospora prolificans, a new hyphomycete from greenhouse soil. Mycotaxon 1974, 1, 45–50. [Google Scholar]
- Malloch, D.; Salkin, I. A new species of Scedosporium associated with osteomyelitis in humans [Scedosporium inglatum, isolated from a bone biopsy]. Mycotaxon 1984, 21, 247–255. [Google Scholar]
- Gueho, E. Taxonomy of the medical species of Pseudallescheria and Scedosporium. J. Mycol. Med. 1991, 118, 3–9. [Google Scholar]
- Kwon-Chung, K.J.; Bennett, J.E. Pseudallescheriasis and Scedosporium infection. In Medical Mycology; Lea & Febiger: Malvern, PA, USA, 1992; pp. 678–694. [Google Scholar]
- Wilson, C.M.; O’Rourke, E.J.; McGinnis, M.R.; Salkin, I.F. Scedosporium inflatum: Clinical spectrum of a newly recognized pathogen. J. Infect. Dis. 1990, 161, 102–107. [Google Scholar] [CrossRef]
- Marin, J.; Sanz, M.A.; Sanz, G.F.; Guarro, J.; Martinez, M.L.; Prieto, M.; Gueho, E.; Menezo, J.L. Disseminated Scedosporium inflatum infection in a patient with acute myeloblastic leukemia. Eur. J. Clin. Microbiol. Infect. Dis. 1991, 10, 759–761. [Google Scholar] [CrossRef]
- Ramirez-Garcia, A.; Pellon, A.; Rementeria, A.; Buldain, I.; Barreto-Bergter, E.; Rollin-Pinheiro, R.; De Meirelles, J.V.; Xisto, M.I.D.; Ranque, S.; Havlicek, V.; et al. Scedosporium and Lomentospora: An updated overview of underrated opportunists. Med. Mycol. 2018, 56 (Suppl. 1), 102–125. [Google Scholar] [CrossRef]
- Caira, M.; Girmenia, C.; Valentini, C.G.; Sanguinetti, M.; Bonini, A.; Rossi, G.; Fianchi, L.; Leone, G.; Pagano, L. Scedosporiosis in patients with acute leukemia: A retrospective multicenter report. Haematologica 2008, 93, 104–110. [Google Scholar] [CrossRef]
- Idigoras, P.; Pérez-Trallero, E.; Pineiro, L.; Larruskain, J.; López-Lopategui, M.C.; Rodríguez, N.; González, J.M. Disseminated infection and colonization by Scedosporium prolificans: A review of 18 cases, 1990–1999. Clin. Infect. Dis. 2001, 32, E158–E165. [Google Scholar] [CrossRef]
- Lackner, M.; Rezusta, A.; Villuendas, M.C.; Palacian, M.P.; Meis, J.F.; Klaassen, C.H. Infection and colonisation due to Scedosporium in Northern Spain. An in vitro antifungal susceptibility and molecular epidemiology study of 60 isolates. Mycoses 2011, 54 (Suppl. 3), 12–21. [Google Scholar] [CrossRef]
- Delhaes, L.; Harun, A.; Chen, S.C.; Nguyen, Q.; Slavin, M.; Heath, C.H.; Maszewska, K.; Halliday, C.; Robert, V.; Sorrell, T.C.; et al. Molecular typing of Australian Scedosporium isolates showing genetic variability and numerous S. aurantiacum. Emerg. Infect. Dis. 2008, 14, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Husain, S.; Muñoz, P.; Forrest, G.; Alexander, B.D.; Somani, J.; Brennan, K.; Wagener, M.M.; Singh, N. Infections due to Scedosporium apiospermum and Scedosporium prolificans in transplant recipients: Clinical characteristics and impact of antifungal agent therapy on outcome. Clin. Infect. Dis. 2005, 40, 89–99. [Google Scholar] [CrossRef]
- Cooley, L.; Spelman, D.; Thursky, K.; Slavin, M. Infection with Scedosporium apiospermum and S. prolificans, Australia. Emerg. Infect. Dis. 2007, 13, 1170–1177. [Google Scholar] [CrossRef]
- Tortorano, A.M.; Richardson, M.; Roilides, E.; Van Diepeningen, A.; Caira, M.; Munoz, P.; Johnson, E.; Meletiadis, J.; Pana, Z.D.; Lackner, M.; et al. ESCMID and ECMM joint guidelines on diagnosis and management of hyalohyphomycosis: Fusarium spp.; Scedosporium spp. and others. Clin. Microbiol. Infect. 2014, 20 (Suppl. 3), 27–46. [Google Scholar] [CrossRef] [PubMed]
- Meletiadis, J.; Meis, J.F.; Mouton, J.W.; Rodriquez-Tudela, J.L.; Donnelly, J.P.; Verweij, P.E.; Eurofung Network. In vitro activities of new and conventional antifungal agents against clinical Scedosporium isolates. Antimicrob. Agents Chemother. 2002, 46, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Enoch, D.A.; Yang, H.; Aliyu, S.H.; Micallef, C. The Changing Epidemiology of Invasive Fungal Infections. Methods Mol. Biol. 2017, 1508, 17–65. [Google Scholar] [PubMed]
- Cortez, K.J.; Roilides, E.; Quiroz-Telles, F.; Meletiadis, J.; Antachopoulos, C.; Knudsen, T.; Buchanan, W.; Milanovich, J.; Sutton, D.A.; Fothergill, A.; et al. Infections caused by Scedosporium spp. Clin. Microbiol. Rev. 2008, 21, 157–197. [Google Scholar] [CrossRef]
- Rougeron, A.; Giraud, S.; Alastruey-Izquierdo, A.; Cano-Lira, J.; Rainer, J.; Mouhajir, A.; Le Gal, S.; Nevez, G.; Meyer, W.; Bouchara, J.P. Ecology of Scedosporium Species: Present Knowledge and Future Research. Mycopathologia 2018, 183, 185–200. [Google Scholar] [CrossRef]
- Mello, T.P.; Aor, A.C.; Oliveira, S.S.; Branquinha, M.H.; Santos, A.L. Conidial germination in Scedosporium apiospermum, S. aurantiacum, S. minutisporum and Lomentospora prolificans: Influence of growth conditions and antifungal susceptibility profiles. Mem. Inst. Oswaldo Cruz 2016, 111, 484–494. [Google Scholar] [CrossRef]
- Mello, T.P.; Aor, A.C.; Goncalves, D.S.; Seabra, S.H.; Branquinha, M.H.; Santos, A.L. Assessment of biofilm formation by Scedosporium apiospermum, S. aurantiacum, S. minutisporum and Lomentospora prolificans. Biofouling 2016, 32, 737–749. [Google Scholar] [CrossRef]
- Kauffman, C.A. Fungal infections. Proc. Am. Thorac. Soc. 2006, 3, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Buldain, I.; Martin-Souto, L.; Antoran, A.; Areitio, M.; Aparicio-Fernandez, L.; Rementeria, A.; Hernando, F.L.; Ramirez-Garcia, A. The Host Immune Response to Scedosporium/Lomentospora. J. Fungi 2021, 7, 75. [Google Scholar] [CrossRef] [PubMed]
- Lamaris, G.A.; Chamilos, G.; Lewis, R.E.; Kontoyiannis, D.P. Virulence studies of Scedosporium and Fusarium species in Drosophila melanogaster. J. Infect. Dis. 2007, 196, 1860–1864. [Google Scholar] [CrossRef] [PubMed]
- Pellon, A.; Ramirez-Garcia, A.; Guruceaga, X.; Zabala, A.; Buldain, I.; Antoran, A.; Anguita, J.; Rementeria, A.; Matute, C.; Hernando, F.L. Microglial immune response is impaired against the neurotropic fungus Lomentospora prolificans. Cell. Microbiol. 2018, 20, e12847. [Google Scholar] [CrossRef] [PubMed]
- Rollin-Pinheiro, R.; da Silva Xisto, M.I.D.; Rochetti, V.P.; Barreto-Bergter, E. Scedosporium Cell Wall: From Carbohydrate-Containing Structures to Host-Pathogen Interactions. Mycopathologia 2020, 185, 931–946. [Google Scholar] [CrossRef]
- Xisto, M.I.; Bittencourt, V.C.; Liporagi-Lopes, L.C.; Haido, R.M.; Mendonca, M.S.; Sassaki, G.; Figueiredo, R.T.; Romanos, M.T.V.; Barreto-Bergter, E. O-glycosylation in cell wall proteins in Scedosporium prolificans is critical for phagocytosis and inflammatory cytokines production by macrophages. PLoS ONE 2015, 10, e0123189. [Google Scholar]
- Xisto, M.; Henao, J.E.M.; Dias, L.D.S.; Santos, G.M.P.; Calixto, R.O.R.; Bernardino, M.C.; Taborda, C.P.; Barreto-Bergter, E. Glucosylceramides From Lomentospora prolificans Induce a Differential Production of Cytokines and Increases the Microbicidal Activity of Macrophages. Front. Microbiol. 2019, 10, 554. [Google Scholar] [CrossRef]
- Al-Laaeiby, A.; Kershaw, M.J.; Penn, T.J.; Thornton, C.R. Targeted Disruption of Melanin Biosynthesis Genes in the Human Pathogenic Fungus Lomentospora prolificans and Its Consequences for Pathogen Survival. Int. J. Mol. Sci. 2016, 17, 444. [Google Scholar] [CrossRef]
- Gil-Lamaignere, C.; Maloukou, A.; Rodriguez-Tudela, J.L.; Roilides, E. Human phagocytic cell responses to Scedosporium prolificans. Med. Mycol. 2001, 39, 169–175. [Google Scholar] [CrossRef]
- Brakhage, A.A.; Bruns, S.; Thywissen, A.; Zipfel, P.F.; Behnsen, J. Interaction of phagocytes with filamentous fungi. Curr. Opin. Microbiol. 2010, 13, 409–415. [Google Scholar] [CrossRef]
- Bronnimann, D.; Garcia-Hermoso, D.; Dromer, F.; Lanternier, F.; The French Mycoses Study Group; Characterization of the Isolates at the NRCMA. Scedosporiosis/lomentosporiosis observational study (SOS): Clinical significance of Scedosporium species identification. Med. Mycol. 2021, 59, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Warris, A.; Netea, M.G.; Verweij, P.E.; Gaustad, P.; Kullberg, B.J.; Weemaes, C.M.; Abrahamsen, T.G. Cytokine responses and regulation of interferon-gamma release by human mononuclear cells to Aspergillus fumigatus and other filamentous fungi. Med. Mycol. 2005, 43, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Bouza, E.; Munoz, P.; Vega, L.; Rodriguez-Creixems, M.; Berenguer, J.; Escudero, A. Clinical resolution of Scedosporium prolificans fungemia associated with reversal of neutropenia following administration of granulocyte colony-stimulating factor. Clin. Infect. Dis. 1996, 23, 192–193. [Google Scholar] [CrossRef]
- Ortoneda, M.; Capilla, J.; Pujol, I.; Pastor, F.J.; Mayayo, E.; Fernandez-Ballart, J.; Guarro, J. Liposomal amphotericin B and granulocyte colony-stimulating factor therapy in a murine model of invasive infection by Scedosporium prolificans. J. Antimicrob. Chemother. 2002, 49, 525–529. [Google Scholar] [CrossRef]
- Antachopoulos, C.; Roilides, E. Cytokines and fungal infections. Br. J. Haematol. 2005, 129, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.O. Emerging applications of recombinant human granulocyte-macrophage colony-stimulating factor. Blood 1998, 92, 4491–4508. [Google Scholar] [PubMed]
- Kurt-Jones, E.A.; Mandell, L.; Whitney, C.; Padgett, A.; Gosselin, K.; Newburger, P.E.; Finberg, R.W. Role of toll-like receptor 2 (TLR2) in neutrophil activation: GM-CSF enhances TLR2 expression and TLR2-mediated interleukin 8 responses in neutrophils. Blood 2002, 100, 1860–1868. [Google Scholar] [CrossRef] [PubMed]
- Willment, J.A.; Lin, H.H.; Reid, D.M.; Taylor, P.R.; Williams, D.L.; Wong, S.Y.; Gordon, S.; Brown, G.D. Dectin-1 expression and function are enhanced on alternatively activated and GM-CSF-treated macrophages and are negatively regulated by IL-10, dexamethasone, and lipopolysaccharide. J. Immunol. 2003, 171, 4569–4573. [Google Scholar] [CrossRef] [PubMed]
- Gil-Lamaignere, C.; Winn, R.M.; Simitsopoulou, M.; Maloukou, A.; Walsh, T.J.; Roilides, E. Inteferon gamma and granulocyte-macrophage colony-stimulating factor augment the antifungal activity of human polymorphonuclear leukocytes against Scedosporium spp.: Comparison with Aspergillus spp. Med. Mycol. 2005, 43, 253–260. [Google Scholar] [CrossRef]
- Winn, R.M.; Gil-Lamaignere, C.; Roilides, E.; Simitsopoulou, M.; Lyman, C.A.; Maloukou, A.; Walsh, T.J. Effects of interleukin-15 on antifungal responses of human polymorphonuclear leukocytes against Fusarium spp. and Scedosporium spp. Cytokine 2005, 31, 1–8. [Google Scholar] [CrossRef]
- Pellon, A.; Ramirez-Garcia, A.; Buldain, I.; Antoran, A.; Rementeria, A.; Hernando, F.L. Immunoproteomics-Based Analysis of the Immunocompetent Serological Response to Lomentospora prolificans. J. Proteome Res. 2016, 15, 595–607. [Google Scholar] [CrossRef]
- Pellon, A.; Ramirez-Garcia, A.; Antoran, A.; Fernandez-Molina, J.V.; Abad-Diaz-de-Cerio, A.; Montanez, D.; Sevilla, M.J.; Rementeria, A.; Hernando, F.L. Scedosporium prolificans immunomes against human salivary immunoglobulin A. Fungal Biol. 2014, 118, 94–105. [Google Scholar] [CrossRef]
- Seidel, D.; Hassler, A.; Salmanton-Garcia, J.; Koehler, P.; Mellinghoff, S.C.; Carlesse, F.; Cheng, M.P.; Falces-Romero, I.; Herbrecht, R.; Jover Saenz, A.; et al. Invasive Scedosporium spp. and Lomentospora prolificans infections in pediatric patients: Analysis of 55 cases from FungiScope(R) and the literature. Int. J. Infect. Dis. 2020, 92, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Jenks, J.D.; Seidel, D.; Cornely, O.A.; Chen, S.; van Hal, S.; Kauffman, C.; Miceli, M.H.; Heinemann, M.; Christner, M.; Jover Saenz, A.; et al. Clinical characteristics and outcomes of invasive Lomentospora prolificans infections: Analysis of patients in the FungiScope((R)) registry. Mycoses 2020, 63, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Berenguer, J.; Rodriguez-Tudela, J.L.; Richard, C.; Alvarez, M.; Sanz, M.A.; Gaztelurrutia, L.; Ayats, J.; Martinez-Suarez, J.V.; Scedosporium Prolificans Spanish Study Group. Deep infections caused by Scedosporium prolificans. A report on 16 cases in Spain and a review of the literature. Medicine 1997, 76, 256–265. [Google Scholar] [CrossRef]
- Maertens, J.; Lagrou, K.; Deweerdt, H.; Surmont, I.; Verhoef, G.E.; Verhaegen, J.; Boogaerts, M.A. Disseminated infection by Scedosporium prolificans: An emerging fatality among haematology patients. Case report and review. Ann. Hematol. 2000, 79, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Vazirani, J.; Westall, G.P.; Snell, G.I.; Morrissey, C.O. Scedosporium apiospermum and Lomentospora prolificans in lung transplant patients—A single center experience over 24 years. Transpl. Infect. Dis. 2021, 23, e13546. [Google Scholar] [CrossRef]
- Tamm, M.; Malouf, M.; Glanville, A. Pulmonary scedosporium infection following lung transplantation. Transpl. Infect. Dis. 2001, 3, 189–194. [Google Scholar] [CrossRef]
- Pianalto, K.M.; Alspaugh, J.A. New Horizons in Antifungal Therapy. J. Fungi 2016, 2, 26. [Google Scholar] [CrossRef]
- Rammaert, B.; Puyade, M.; Cornely, O.A.; Seidel, D.; Grossi, P.; Husain, S.; Picard, C.; Lass-Florl, C.; Manuel, O.; Le Pavec, J.; et al. Perspectives on Scedosporium species and Lomentospora prolificans in lung transplantation: Results of an international practice survey from ESCMID fungal infection study group and study group for infections in compromised hosts, and European Confederation of Medical Mycology. Transpl. Infect. Dis. 2019, 21, e13141. [Google Scholar]
- Cobo, F.; Lara-Oya, A.; Rodriguez-Granger, J.; Sampedro, A.; Aliaga-Martinez, L.; Navarro-Mari, J.M. Infections caused by Scedosporium/Lomentospora species: Clinical and microbiological findings in 21 cases. Med. Mycol. 2018, 56, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.; Stevens, R.; Konecny, P. Lomentospora prolificans endocarditis—Case report and literature review. BMC Infect. Dis. 2016, 16, 36. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, Y.; Okugawa, S.; Tatsuno, K.; Ikeda, M.; Misawa, Y.; Koyano, S.; Tsuji, E.; Yanagimoto, S.; Hatakeyama, S.; Moriya, K.; et al. Scedosporium prolificans Endocarditis: Case Report and Literature Review. Intern. Med. 2016, 55, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Tascini, C.; Bongiorni, M.G.; Leonildi, A.; Giannola, G.; Soldati, E.; Arena, G.; Doria, R.; Germenia, C.; Menichetti, F. Pacemaker endocarditis with pulmonary cavitary lesion due to Scedosporium prolificans. J. Chemother. 2006, 18, 667–669. [Google Scholar] [CrossRef] [PubMed]
- Ochi, Y.; Hiramoto, N.; Takegawa, H.; Yonetani, N.; Doi, A.; Ichikawa, C.; Imai, Y.; Ishikawa, T. Infective endocarditis caused by Scedosporium prolificans infection in a patient with acute myeloid leukemia undergoing induction chemotherapy. Int. J. Hematol. 2015, 101, 620–625. [Google Scholar] [CrossRef]
- Fernandez Guerrero, M.L.; Askari, E.; Prieto, E.; Gadea, I.; Roman, A. Emerging infectious endocarditis due to Scedosporium prolificans: A model of therapeutic complexity. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 1321–1324. [Google Scholar] [CrossRef]
- Carreter de Granda, M.E.; Richard, C.; Conde, E.; Iriondo, A.; Marco de Lucas, F.; Salesa, R.; Zubizarreta, A. Endocarditis caused by Scedosporium prolificans after autologous peripheral blood stem cell transplantation. Eur. J. Clin. Microbiol. Infect. Dis. 2001, 20, 215–217. [Google Scholar] [CrossRef]
- Tamaki, M.; Nozaki, K.; Onishi, M.; Yamamoto, K.; Ujiie, H.; Sugahara, H. Fungal meningitis caused by Lomentospora prolificans after allogeneic hematopoietic stem cell transplantation. Transpl. Infect. Dis. 2016, 18, 601–605. [Google Scholar] [CrossRef]
- Madrigal, V.; Alonso, J.; Bureo, E.; Figols, F.J.; Salesa, R. Fatal meningoencephalitis caused by Scedosporium inflatum (Scedosporium prolificans) in a child with lymphoblastic leukemia. Eur. J. Clin. Microbiol. Infect. Dis. 1995, 14, 601–603. [Google Scholar] [CrossRef]
- Nenoff, P.; Gutz, U.; Tintelnot, K.; Bosse-Henck, A.; Mierzwa, M.; Hofmann, J.; Horn, L.C.; Haustein, U.F. Disseminated mycosis due to Scedosporium prolificans in an AIDS patient with Burkitt lymphoma. Mycoses 1996, 39, 461–465. [Google Scholar] [CrossRef]
- Gow-Lee, V.J.; Moyers, J.T.; Rogstad, D.K. Fatal recurrent disseminated Lomentospora prolificans infection during autologous hematopoietic stem cell transplantation: A case report and review, and discussion on the importance of prolonged neutropenia. Transpl. Infect. Dis. 2021, 23, e13701. [Google Scholar] [CrossRef]
- Salesa, R.; Burgos, A.; Ondiviela, R.; Richard, C.; Quindos, G.; Ponton, J. Fatal disseminated infection by Scedosporium inflatum after bone marrow transplantation. Scand. J. Infect. Dis. 1993, 25, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Uno, K.; Kasahara, K.; Kutsuna, S.; Katanami, Y.; Yamamoto, Y.; Maeda, K.; Konishi, M.; Ogawa, T.; Yoneda, T.; Yoshida, K.; et al. Infective endocarditis and meningitis due to Scedosporium prolificans in a renal transplant recipient. J. Infect. Chemother. 2014, 20, 131–133. [Google Scholar] [CrossRef] [PubMed]
- Taj-Aldeen, S.J.; Rammaert, B.; Gamaletsou, M.; Sipsas, N.V.; Zeller, V.; Roilides, E.; Kontoyiannis, D.P.; Miller, A.O.; Petraitis, V.; Walsh, T.J.; et al. Osteoarticular Infections Caused by Non-Aspergillus Filamentous Fungi in Adult and Pediatric Patients: A Systematic Review. Medicine 2015, 94, e2078. [Google Scholar] [CrossRef] [PubMed]
- Bhagavatula, S.; Vale, L.; Evans, J.; Carpenter, C.; Barnes, R.A. Scedosporium prolificans osteomyelitis following penetrating injury: A case report. Med. Mycol. Case Rep. 2014, 4, 26–29. [Google Scholar] [CrossRef]
- Dalton, P.A.; Munckhof, W.J.; Walters, D.W. Scedosporium prolificans: An uncommon cause of septic arthritis. ANZ J. Surg. 2006, 76, 661–663. [Google Scholar] [CrossRef] [PubMed]
- Kesson, A.M.; Bellemore, M.C.; O’mara, T.J.; Ellis, D.H.; Sorrell, T.C. Scedosporium prolificans osteomyelitis in an immunocompetent child treated with a novel agent, hexadecylphospocholine (miltefosine), in combination with terbinafine and voriconazole: A case report. Clin. Infect. Dis. 2009, 48, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Steinbach, W.J.; Schell, W.A.; Miller, J.L.; Perfect, J.R. Scedosporium prolificans osteomyelitis in an immunocompetent child treated with voriconazole and caspofungin, as well as locally applied polyhexamethylene biguanide. J. Clin. Microbiol. 2003, 41, 3981–3985. [Google Scholar] [CrossRef]
- Lee, J.; Wilson, M.; Casey, N. Eradication of Lomentospora prolificans Osteomyelitis of the Wrist with Combination Antifungal Therapy, Voriconazole Bone Cement, and Surgical Debridement. Case Rep Orthop. 2020, 2020, 8271471. [Google Scholar] [CrossRef]
- Garcia-Vidal, C.; Cabellos, C.; Ayats, J.; Font, F.; Ferran, E.; Fernandez-Viladrich, P. Fungal postoperative spondylodiscitis due to Scedosporium prolificans. Spine J. 2009, 9, e1–e7. [Google Scholar] [CrossRef]
- Quaesaet, L.; Stindel, E.; Lanternier, F.; Williams, T.; Jaffuel, S.; Moalic, E.; Garcia Hermoso, D.; Lortholary, O.; Ansart, S. Miltefosine-based regimen as salvage therapy in Lomentospora prolificans bone and joint infection. Med. Mal. Infect. 2018, 48, 63–65. [Google Scholar] [CrossRef] [PubMed]
- Daniele, L.; Le, M.; Parr, A.F.; Brown, L.M. Scedosporium prolificans Septic Arthritis and Osteomyelitis of the Hip Joints in an Immunocompetent Patient: A Case Report and Literature Review. Case Rep. Orthop. 2017, 2017, 3809732. [Google Scholar] [PubMed]
- Wangchinda, W.; Chongtrakool, P.; Tanboon, J.; Jitmuang, A. Lomentospora prolificans vertebral osteomyelitis with spinal epidural abscess in an immunocompetent woman: Case report and literature review. Med. Mycol. Case Rep. 2018, 21, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Pace, C.S.; Frankenhoff, J.A.; Isaacs, J.E. Scedosporium prolificans Septic Arthritis. J. Hand Microsurg. 2017, 9, 37–38. [Google Scholar]
- Mcguire, T.W.; Bullock, J.D.; Bullock, J.D., Jr.; Elder, B.L.; Funkhouser, J.W. Fungal endophthalmitis. An experimental study with a review of 17 human ocular cases. Arch. Ophthalmol. 1991, 109, 1289–1296. [Google Scholar] [CrossRef]
- Wood, G.M.; McCormack, J.G.; Muir, D.B.; Ellis, D.H.; Ridley, M.F.; Pritchard, R.; Harrison, M. Clinical features of human infection with Scedosporium inflatum. Clin. Infect. Dis. 1992, 14, 1027–1033. [Google Scholar] [CrossRef]
- Vagefi, M.R.; Kim, E.T.; Alvarado, R.G.; Duncan, J.L.; Howes, E.L.; Crawford, J.B. Bilateral endogenous Scedosporium prolificans endophthalmitis after lung transplantation. Am. J. Ophthalmol. 2005, 139, 370–373. [Google Scholar] [CrossRef]
- Mckelvie, P.A.; Wong, E.Y.; Chow, L.P.; Hall, A.J. Scedosporium endophthalmitis: Two fatal disseminated cases of Scedosporium infection presenting with endophthalmitis. Clin. Exp. Ophthalmol. 2001, 29, 330–334. [Google Scholar] [CrossRef]
- O’hearn, T.M.; Geiseler, P.J.; Bhatti, R.A.; Eliott, D. Control of disseminated scedosporium prolificans infection and endophthalmitis. Retin Cases Brief Rep. 2010, 4, 18–19. [Google Scholar] [CrossRef]
- Chiam, N.; Rose, L.V.; Waters, K.D.; Elder, J.E. Scedosporium prolificans endogenous endophthalmitis. J. AAPOS 2013, 17, 627–629. [Google Scholar] [CrossRef]
- Taylor, A.; Wiffen, S.J.; Kennedy, C.J. Post-traumatic Scedosporium inflatum endophthalmitis. Clin. Exp. Ophthalmol. 2002, 30, 47–48. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, L.J.; Snibson, G.; Joseph, C.; Taylor, H.R. Scedosporium prolificans sclerokeratitis. Aust. N. Z. J. Ophthalmol. 1994, 22, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Crawford, G.J.; Morlet, G.C. Scedosporium prolificans corneoscleritis: A successful outcome. Aust. N. Z. J. Ophthalmol. 1997, 25, 169–171. [Google Scholar] [CrossRef]
- Arthur, S.; Steed, L.L.; Apple, D.J.; Peng, Q.; Howard, G.; Escobar-Gomez, M. Scedosporium prolificans keratouveitis in association with a contact lens retained intraocularly over a long term. J. Clin. Microbiol. 2001, 39, 4579–4582. [Google Scholar] [CrossRef] [PubMed]
- Sayah, D.M.; Schwartz, B.S.; Kukreja, J.; Singer, J.P.; Golden, J.A.; Leard, L.E. Scedosporium prolificans pericarditis and mycotic aortic aneurysm in a lung transplant recipient receiving voriconazole prophylaxis. Transpl. Infect. Dis. 2013, 15, E70–E74. [Google Scholar] [CrossRef]
- Guadalajara MC, V.; Hernandez Gonzalez, A.; Carrasco Garcia De Leon, S.; Rojo, M.G.; Del Real Francia, M.A. Mycotic Cerebral Aneurysms Secondary to Scedosporium prolificans Infection in a Patient with Multiple Sclerosis. J. Clin. Neurol. 2018, 14, 601–603. [Google Scholar] [CrossRef]
- Wen, S.C.; Phillips, D.; Gwee, A. Scedosporium prolificans Otitis Externa. Pediatr. Infect. Dis. J. 2016, 35, 1277. [Google Scholar] [CrossRef]
- Ference, E.H.; Kubak, B.M.; Zhang, P.; Suh, J.D. Successful Treatment of Scedosporium Sinusitis in Two Lung Transplant Recipients: Review of the Literature and Recommendations for Management. Allergy Rhinol. 2019, 10, 1–8. [Google Scholar] [CrossRef]
- Jeong, J.C.; Lee, H.; Lee, S.W.; Park, J.Y.; Ahn, S.Y.; Park, H.; Kim, E.C.; Park, S.S.; Ahn, C.; Oh, K.H. Fungal peritonitis due to Scedosporium prolificans. Perit. Dial. Int. 2011, 31, 213–215. [Google Scholar]
- Chen, S.C.; Halliday, C.L.; Hoenigl, M.; Cornely, O.A.; Meyer, W. Scedosporium and Lomentospora Infections: Contemporary Microbiological Tools for the Diagnosis of Invasive Disease. J. Fungi 2021, 7, 23. [Google Scholar] [CrossRef]
- Donnelly, J.P.; Chen, S.C.; Kauffman, C.A.; Steinbach, W.J.; Baddley, J.W.; Verweij, P.E.; Clancy, C.J.; Wingard, J.R.; Lockhart, S.R.; Groll, A.H.; et al. Revision and Update of the Consensus Definitions of Invasive Fungal Disease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin. Infect. Dis. 2020, 71, 1367–1376. [Google Scholar] [CrossRef]
- Kimura, M.; Maenishi, O.; Ito, H.; Ohkusu, K. Unique histological characteristics of Scedosporium that could aid in its identification. Pathol. Int. 2010, 60, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Guarner, J.; Brandt, M.E. Histopathologic diagnosis of fungal infections in the 21st century. Clin. Microbiol. Rev. 2011, 24, 247–280. [Google Scholar] [CrossRef] [PubMed]
- Hoenigl, M.; Salmanton-García, J.; Walsh, T.J.; Nucci, M.; Neoh, C.F.; Jenks, J.D.; Lackner, M.; Sprute, R.; Al-Hatmi, A.M.; Bassetti, M.; et al. Global guideline for the diagnosis and management of rare mould infections: An initiative of the European Confederation of Medical Mycology in cooperation with the International Society for Human and Animal Mycology and the American Society for Microbiology. Lancet Infect. Dis. 2021, 21, e246–e257. [Google Scholar]
- Perfect, J.R.; Schell, W.A. The new fungal opportunists are coming. Clin. Infect. Dis. 1996, 22 (Suppl. 2), S112–S118. [Google Scholar] [CrossRef] [PubMed]
- Cuenca-Estrella, M.; Bassetti, M.; Lass-Florl, C.; Racil, Z.; Richardson, M.; Rogers, T.R. Detection and investigation of invasive mould disease. J. Antimicrob. Chemother. 2011, 66 (Suppl. 1), i15–i24. [Google Scholar] [CrossRef] [PubMed]
- Lau, A.; Chen, S.; Sorrell, T.; Carter, D.; Malik, R.; Martin, P.; Halliday, C. Development and clinical application of a panfungal PCR assay to detect and identify fungal DNA in tissue specimens. J. Clin. Microbiol. 2007, 45, 380–385. [Google Scholar] [CrossRef]
- Buitrago, M.J.; Bernal-Martinez, L.; Castelli, M.V.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. Performance of panfungal- and specific-PCR-based procedures for etiological diagnosis of invasive fungal diseases on tissue biopsy specimens with proven infection: A 7-year retrospective analysis from a reference laboratory. J. Clin. Microbiol. 2014, 52, 1737–1740. [Google Scholar] [CrossRef]
- Ruiz-Diez, B.; Martin-Diez, F.; Rodriguez-Tudela, J.L.; Alvarez, M.; Martinez-Suarez, J.V. Use of random amplification of polymorphic DNA (RAPD) and PCR-fingerprinting for genotyping a Scedosporium prolificans (inflatum) outbreak in four leukemic patients. Curr. Microbiol. 1997, 35, 186–190. [Google Scholar] [CrossRef]
- Bouchara, J.P.; Hsieh, H.Y.; Croquefer, S.; Barton, R.; Marchais, V.; Pihet, M.; Chang, T.C. Development of an oligonucleotide array for direct detection of fungi in sputum samples from patients with cystic fibrosis. J. Clin. Microbiol. 2009, 47, 142–152. [Google Scholar] [CrossRef]
- Harun, A.; Blyth, C.C.; Gilgado, F.; Middleton, P.; Chen, S.C.; Meyer, W. Development and validation of a multiplex PCR for detection of Scedosporium spp. in respiratory tract specimens from patients with cystic fibrosis. J. Clin. Microbiol. 2011, 49, 1508–1512. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.R.; Bialek, R.; Kibbler, C.C.; Cuenca-Estrella, M.; Jensen, H.E.; Kontoyiannis, D.P. Molecular Techniques for Genus and Species Determination of Fungi From Fresh and Paraffin-Embedded Formalin-Fixed Tissue in the Revised EORTC/MSGERC Definitions of Invasive Fungal Infection. Clin. Infect. Dis. 2021, 72 (Suppl. 2), S109–S113. [Google Scholar] [CrossRef] [PubMed]
- Wilkendorf, L.S.; Bowles, E.; Buil, J.B.; van der Lee, H.A.L.; Posteraro, B.; Sanguinetti, M.; Verweij, P.E. Update on Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry Identification of Filamentous Fungi. J. Clin. Microbiol. 2020, 58, e01263-20. [Google Scholar] [CrossRef] [PubMed]
- Zvezdanova, M.E.; Escribano, P.; Ruiz, A.; Martinez-Jimenez, M.C.; Pelaez, T.; Collazos, A.; Guinea, J.; Bouza, E.; Rodriguez-Sanchez, B. Increased species-assignment of filamentous fungi using MALDI-TOF MS coupled with a simplified sample processing and an in-house library. Med. Mycol. 2019, 57, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Sitterle, E.; Giraud, S.; Leto, J.; Bouchara, J.P.; Rougeron, A.; Morio, F.; Dauphin, B.; Angebault, C.; Quesne, G.; Beretti, J.L.; et al. Matrix-assisted laser desorption ionization-time of flight mass spectrometry for fast and accurate identification of Pseudallescheria/Scedosporium species. Clin. Microbiol. Infect. 2014, 20, 929–935. [Google Scholar] [CrossRef]
- Odabasi, Z.; Paetznick, V.L.; Rodriguez, J.R.; Chen, E.; Mcginnis, M.R.; Ostrosky-Zeichner, L. Differences in beta-glucan levels in culture supernatants of a variety of fungi. Med. Mycol. 2006, 44, 267–272. [Google Scholar] [CrossRef]
- Karageorgopoulos, D.E.; Vouloumanou, E.K.; Ntziora, F.; Michalopoulos, A.; Rafailidis, P.I.; Falagas, M.E. beta-D-glucan assay for the diagnosis of invasive fungal infections: A meta-analysis. Clin. Infect. Dis. 2011, 52, 750–770. [Google Scholar] [CrossRef]
- Nishimori, M.; Takahashi, T.; Suzuki, E.; Kodaka, T.; Hiramoto, N.; Itoh, K.; Tsunemine, H.; Yarita, K.; Kamei, K.; Takegawa, H.; et al. Fatal fungemia with Scedosporium prolificans in a patient with acute myeloid leukemia. Med. Mycol. J. 2014, 55, E63–E70. [Google Scholar] [CrossRef]
- Thornton, C.R.; Ryder, L.S.; Le Cocq, K.; Soanes, D.M. Identifying the emerging human pathogen Scedosporium prolificans by using a species-specific monoclonal antibody that binds to the melanin biosynthetic enzyme tetrahydroxynaphthalene reductase. Environ. Microbiol. 2015, 17, 1023–1038. [Google Scholar] [CrossRef]
- Martin-Souto, L.; Buldain, I.; Areitio, M.; Aparicio-Fernandez, L.; Antoran, A.; Bouchara, J.P.; Martin-Gomez, M.T.; Rementeria, A.; Hernando, F.L.; Ramirez-Garcia, A. ELISA Test for the Serological Detection of Scedosporium/Lomentospora in Cystic Fibrosis Patients. Front. Cell. Infect. Microbiol. 2020, 10, 602089. [Google Scholar] [CrossRef]
- Cuenca-Estrella, M.; Alastruey-Izquierdo, A.; Alcazar-Fuoli, L.; Bernal-Martinez, L.; Gomez-Lopez, A.; Buitrago, M.J.; Mellado, E.; Rodriguez-Tudela, J.L. In vitro activities of 35 double combinations of antifungal agents against Scedosporium apiospermum and Scedosporium prolificans. Antimicrob. Agents Chemother. 2008, 52, 1136–1139. [Google Scholar] [CrossRef] [PubMed]
- Pellon, A.; Ramirez-Garcia, A.; Buldain, I.; Antoran, A.; Martin-Souto, L.; Rementeria, A.; Hernando, F.L. Pathobiology of Lomentospora prolificans: Could this species serve as a model of primary antifungal resistance? Int. J. Antimicrob. Agents 2018, 51, 10–15. [Google Scholar] [CrossRef]
- Yustes, C.; Guarro, J. In vitro synergistic interaction between amphotericin B and micafungin against Scedosporium spp. Antimicrob. Agents Chemother. 2005, 49, 3498–3500. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martin-Vicente, A.; Guarro, J.; Capilla, J. Does a triple combination have better activity than double combinations against multiresistant fungi? Experimental in vitro evaluation. Int. J. Antimicrob. Agents 2017, 49, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Schemuth, H.; Dittmer, S.; Lackner, M.; Sedlacek, L.; Hamprecht, A.; Steinmann, E.; Buer, J.; Rath, P.M.; Steinmann, J. In vitro activity of colistin as single agent and in combination with antifungals against filamentous fungi occurring in patients with cystic fibrosis. Mycoses 2013, 56, 297–303. [Google Scholar] [CrossRef]
- Homa, M.; Galgoczy, L.; Toth, E.; Toth, L.; Papp, T.; Chandrasekaran, M.; Kadaikunnan, S.; Alharbi, N.S.; Vagvolgyi, C. In vitro antifungal activity of antipsychotic drugs and their combinations with conventional antifungals against Scedosporium and Pseudallescheria isolates. Med. Mycol. 2015, 53, 890–895. [Google Scholar] [CrossRef]
- Howden, B.P.; Slavin, M.A.; Schwarer, A.P.; Mijch, A.M. Successful control of disseminated Scedosporium prolificans infection with a combination of voriconazole and terbinafine. Eur. J. Clin. Microbiol. Infect. Dis. 2003, 22, 111–113. [Google Scholar] [CrossRef]
- Farina, C.; Marchesi, G.; Passera, M.; Diliberto, C.; Russello, G. Comparative study of the in vitro activity of various antifungal drugs against Scedosporium spp. in aerobic and hyperbaric atmosphere versus normal atmosphere. J. Mycol. Med. 2012, 22, 142–148. [Google Scholar] [CrossRef]
- Castanheira, M.; Duncanson, F.P.; Diekema, D.J.; Guarro, J.; Jones, R.N.; Pfaller, M.A. Activities of E1210 and comparator agents tested by CLSI and EUCAST broth microdilution methods against Fusarium and Scedosporium species identified using molecular methods. Antimicrob. Agents Chemother. 2012, 56, 352–357. [Google Scholar] [CrossRef]
- Miyazaki, M.; Horii, T.; Hata, K.; Watanabe, N.A.; Nakamoto, K.; Tanaka, K.; Shirotori, S.; Murai, N.; Inoue, S.; Matsukura, M.; et al. In vitro activity of E1210, a novel antifungal, against clinically important yeasts and molds. Antimicrob. Agents Chemother. 2011, 55, 4652–4658. [Google Scholar] [CrossRef]
- Wiederhold, N.P. Review of the Novel Investigational Antifungal Olorofim. J. Fungi 2020, 6, 122. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Rai, N.J.; Cunneen, S.; Cornelissen, K.; Rex, J.H.; Heath, C.H.; Harvey, E. A case of Lomentospora prolificans treated with the novel antifungal olorofim. In Proceedings of the 30th European Congress of Clinical Microbiology and Infectious Diseases, Paris, France, 18–21 April 2020. [Google Scholar]
- Tio, S.; Thursky, K.; Ng, G.; Rex, J.; Carney, D.; Slavin, M. Olorofim for a case of severe disseminated Lomentospora prolificans infection. In Proceedings of the 30th European Congress of Clinical Microbiology and Infectious Diseases, Paris, France, 18–21 April 2020. [Google Scholar]
- Pellon, A.; Ramirez-Garcia, A.; Buldain, I.; Antoran, A.; Rementeria, A.; Hernando, F.L. Molecular and cellular responses of the pathogenic fungus Lomentospora prolificans to the antifungal drug voriconazole. PLoS ONE 2017, 12, e0174885. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konsoula, A.; Tsioutis, C.; Markaki, I.; Papadakis, M.; Agouridis, A.P.; Spernovasilis, N. Lomentospora prolificans: An Emerging Opportunistic Fungal Pathogen. Microorganisms 2022, 10, 1317. https://doi.org/10.3390/microorganisms10071317
Konsoula A, Tsioutis C, Markaki I, Papadakis M, Agouridis AP, Spernovasilis N. Lomentospora prolificans: An Emerging Opportunistic Fungal Pathogen. Microorganisms. 2022; 10(7):1317. https://doi.org/10.3390/microorganisms10071317
Chicago/Turabian StyleKonsoula, Afroditi, Constantinos Tsioutis, Ioulia Markaki, Michail Papadakis, Aris P. Agouridis, and Nikolaos Spernovasilis. 2022. "Lomentospora prolificans: An Emerging Opportunistic Fungal Pathogen" Microorganisms 10, no. 7: 1317. https://doi.org/10.3390/microorganisms10071317
APA StyleKonsoula, A., Tsioutis, C., Markaki, I., Papadakis, M., Agouridis, A. P., & Spernovasilis, N. (2022). Lomentospora prolificans: An Emerging Opportunistic Fungal Pathogen. Microorganisms, 10(7), 1317. https://doi.org/10.3390/microorganisms10071317






