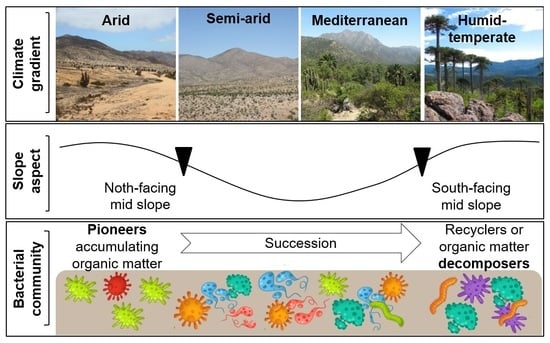
Impact of Climate and Slope Aspects on the Composition of Soil Bacterial Communities Involved in Pedogenetic Processes along the Chilean Coastal Cordillera
Abstract
:1. Introduction
2. Materials and Methods
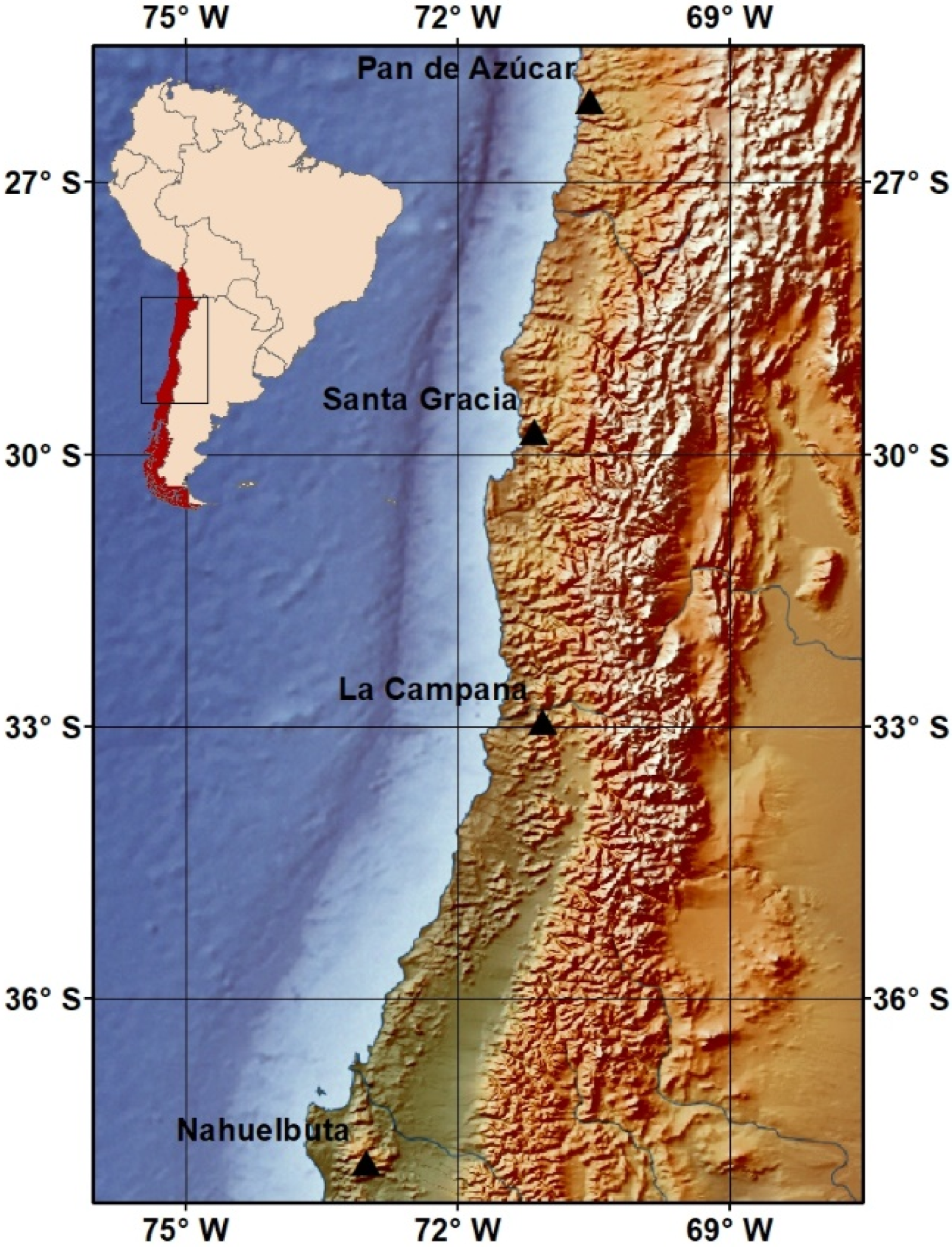
2.1. Study Sites and Soil Sampling
2.2. Soil Chemical Analysis
2.3. DNA Extraction and Sequencing
2.4. Data Analysis
3. Results
3.1. Soil Physical Properties and Soil Chemical Properties
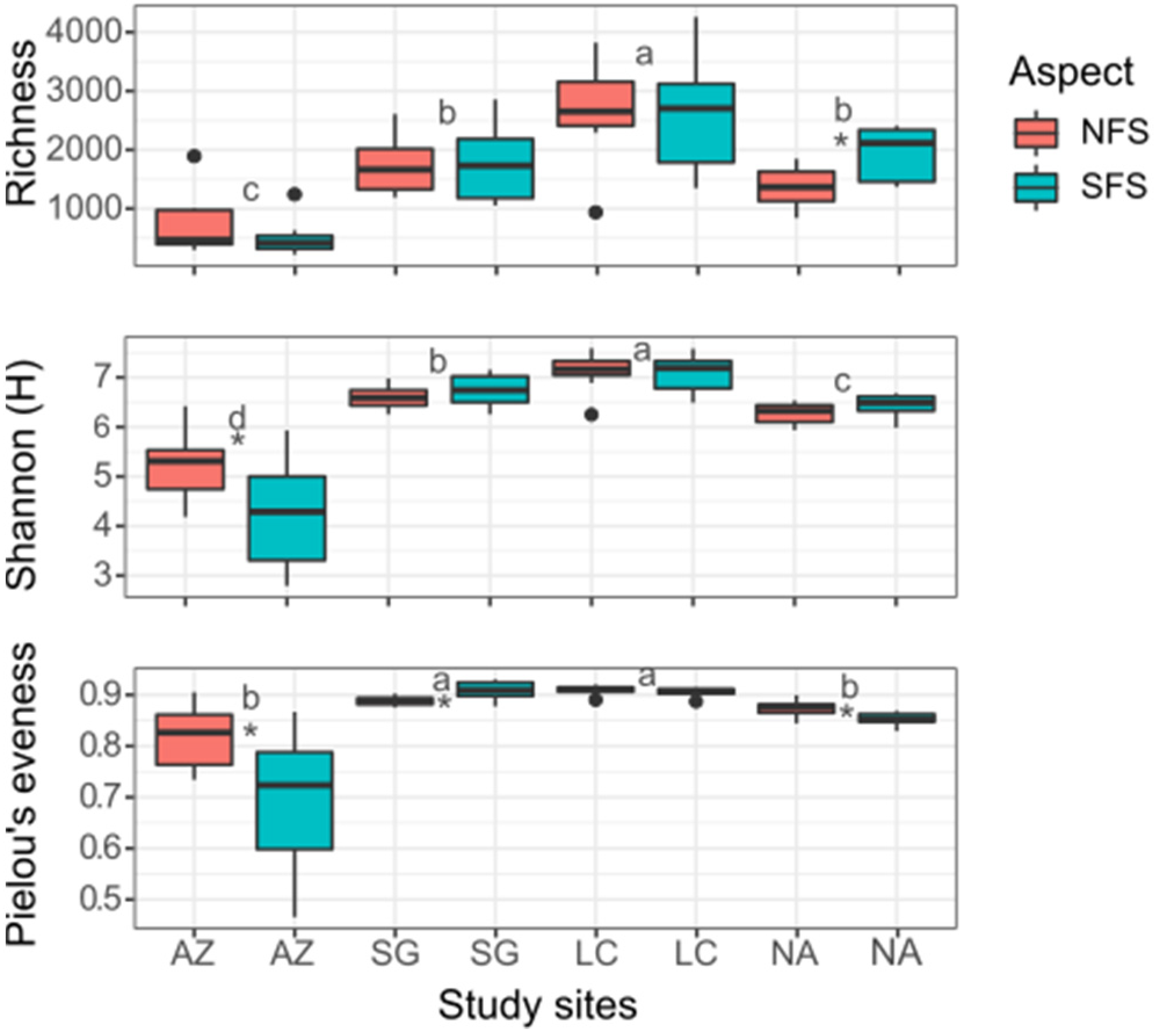
3.2. Diversity Analysis
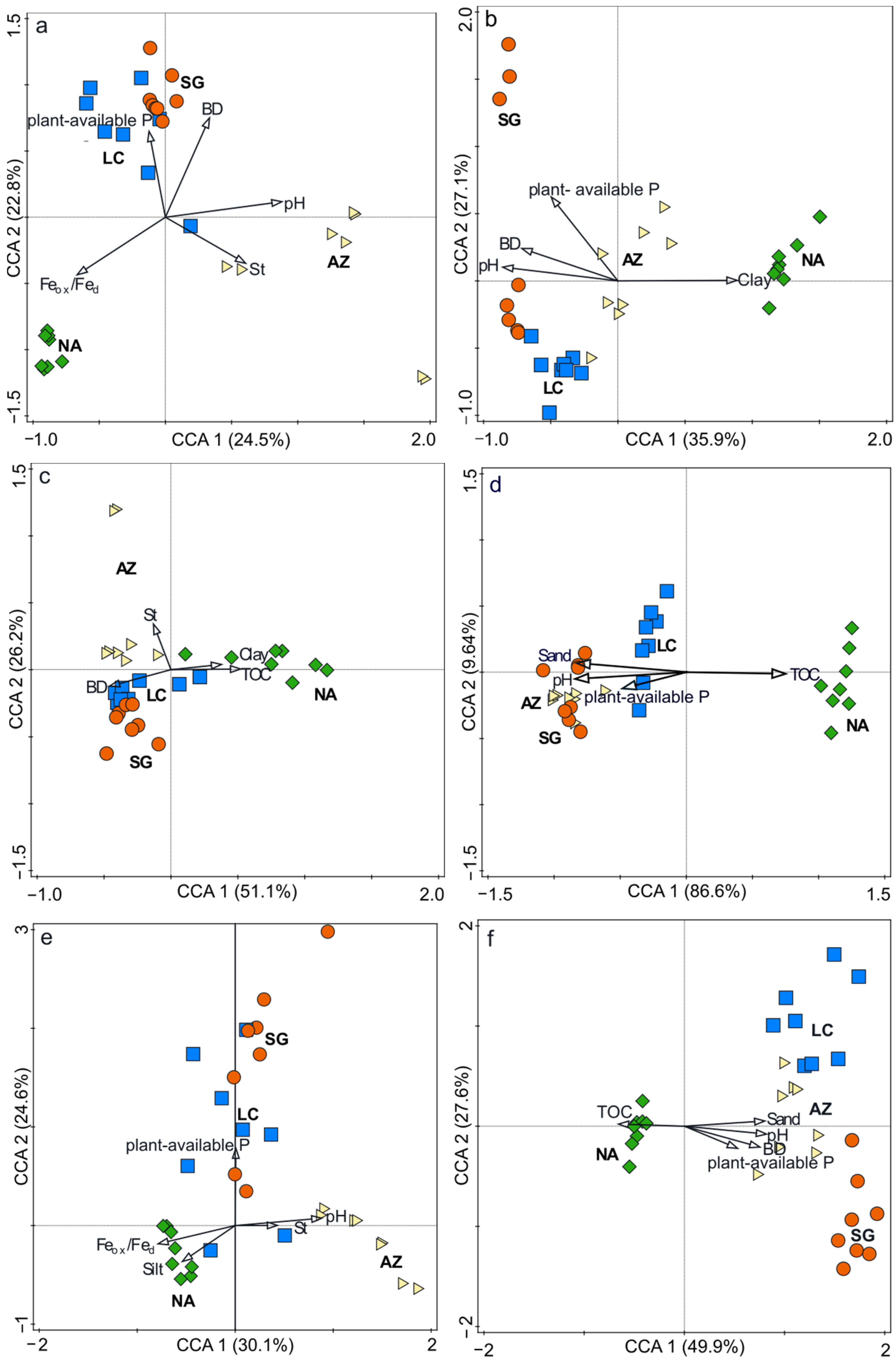
3.3. Community-Composition Analysis
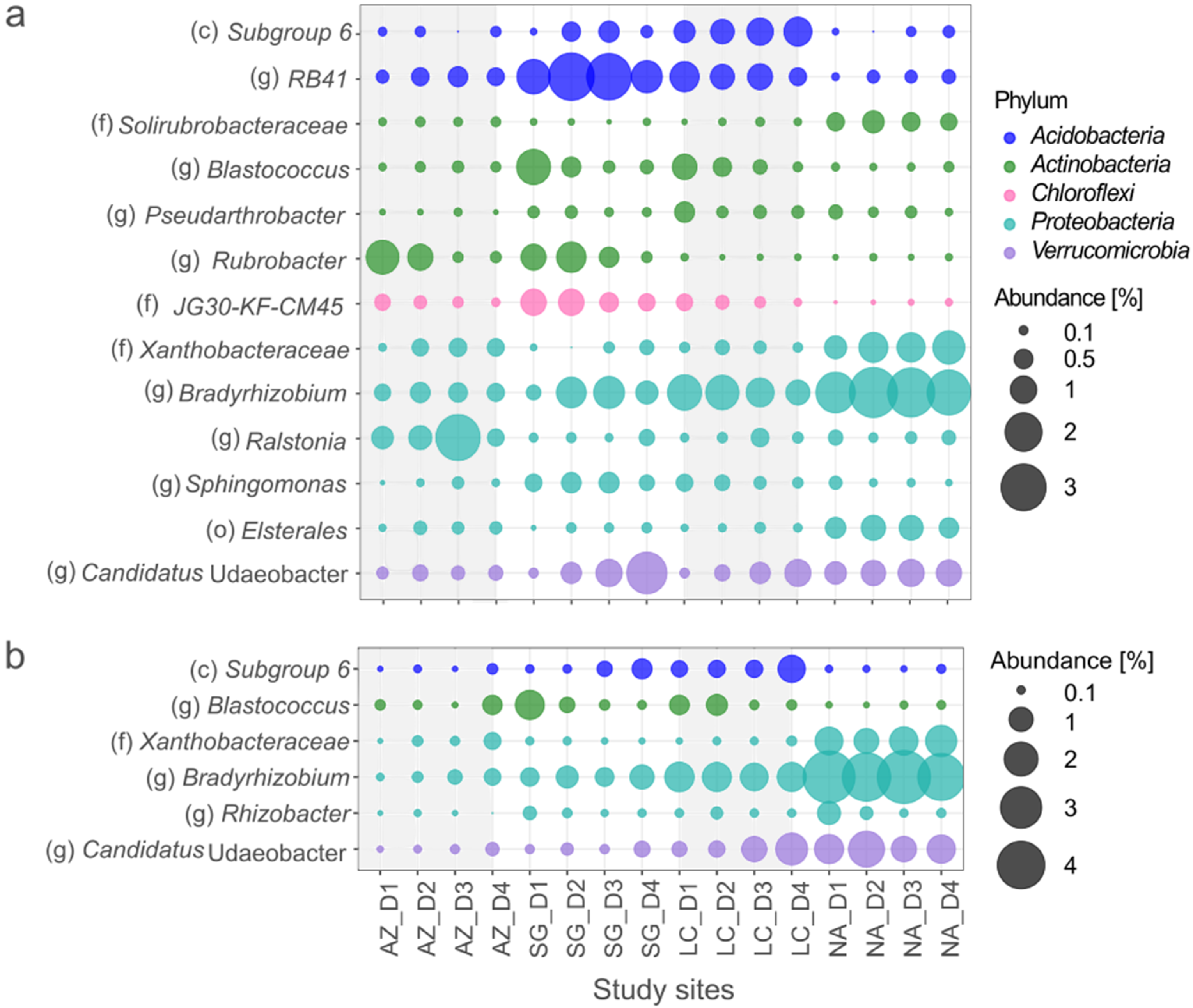
3.4. Bacterial Generalists
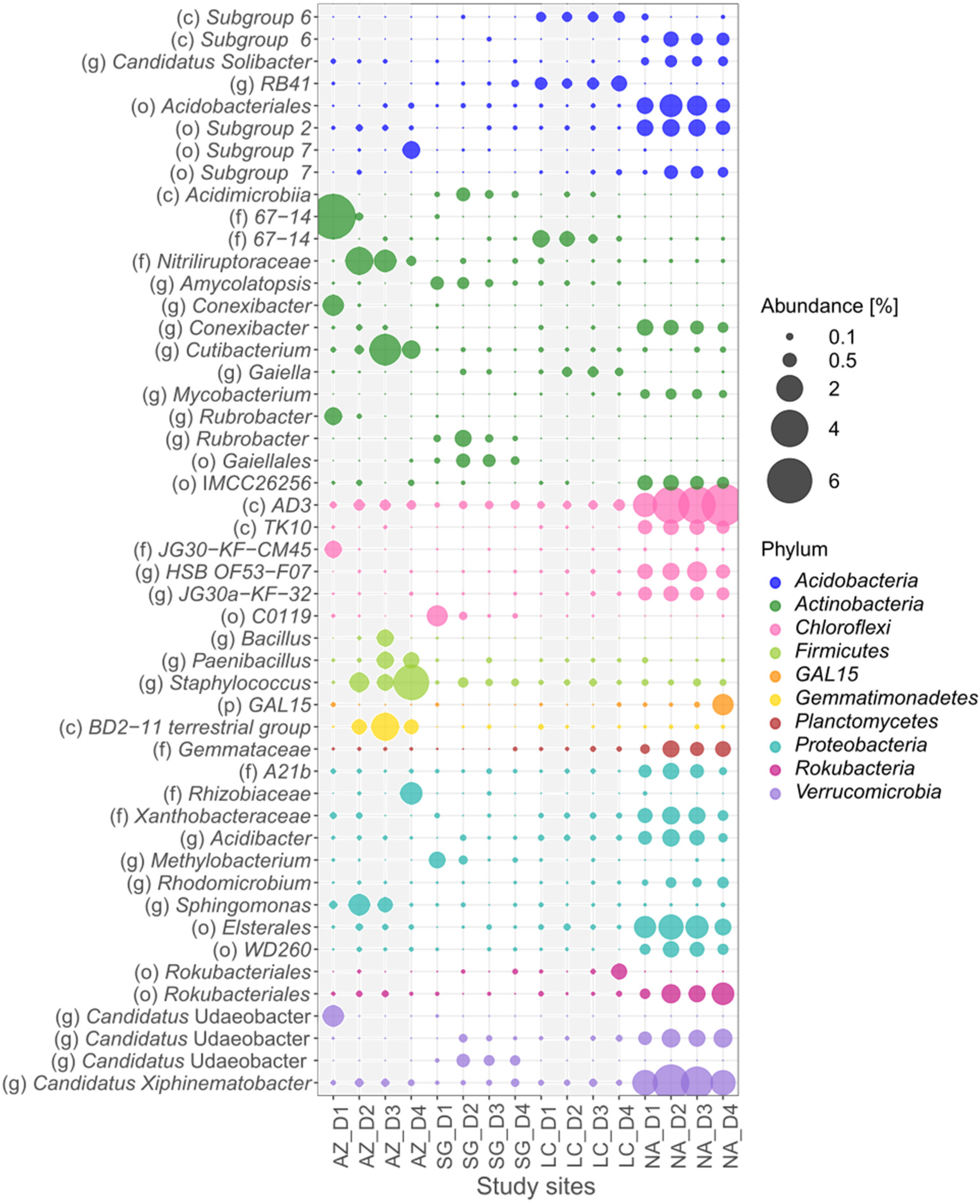
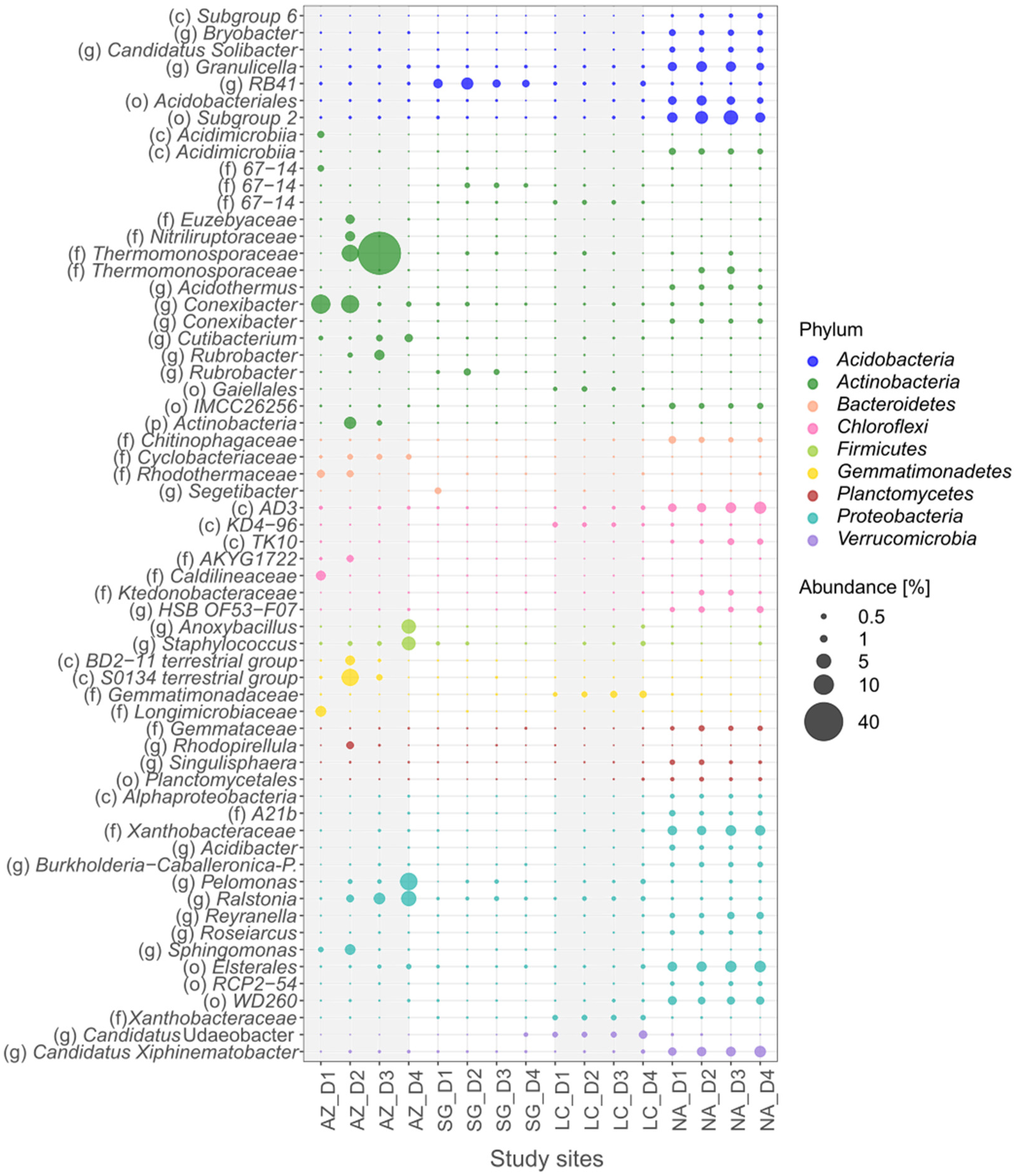
3.5. Bacteria Specialists
4. Discussion
4.1. Climate Gradient
4.2. Slope Aspect
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaviya, N.; Upadhayay, V.K.; Singh, J.; Khan, A.; Panwar, M.; Singh, A.V. Role of Microorganisms in Soil Genesis and Functions. In Mycorrhizosphere and Pedogenesis; Springer: Singapore, 2019; pp. 25–52. [Google Scholar]
- Fatichi, S.; Or, D.; Walko, R.; Vereecken, H.; Young, M.H.; Ghezzehei, T.A.; Hengl, T.; Kollet, S.; Agam, N.; Avissar, R. Soil structure is an important omission in Earth System Models. Nat. Commun. 2020, 11, 522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenny, H. Factors of Soil Formation: A Sytem of Quantitative Pedology; Dover Publications, Inc.: New York, NY, USA, 1941. [Google Scholar]
- Schulz, S.; Brankatschk, R.; Dümig, A.; Kögel-Knabner, I.; Schloter, M.; Zeyer, J. The role of microorganisms at different stages of ecosystem development for soil formation. Biogeosciences 2013, 10, 3983–3996. [Google Scholar] [CrossRef] [Green Version]
- Jacoby, R.; Peukert, M.; Succurro, A.; Koprivova, A.; Kopriva, S. The Role of Soil Microorganisms in Plant Mineral Nutrition-Current Knowledge and Future Directions. Front. Plant Sci. 2017, 8, 1617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahram, M.; Hildebrand, F.; Forslund, S.K.; Anderson, J.L.; Soudzilovskaia, N.A.; Bodegom, P.M.; Bengtsson-Palme, J.; Anslan, S.; Coelho, L.P.; Harend, H.; et al. Structure and function of the global topsoil microbiome. Nature 2018, 560, 233–237. [Google Scholar] [CrossRef]
- Delgado-Baquerizo, M.; Bardgett, R.D.; Vitousek, P.M.; Maestre, F.T.; Williams, M.A.; Eldridge, D.J.; Lambers, H.; Neuhauser, S.; Gallardo, A.; Garcia-Velazquez, L.; et al. Changes in belowground biodiversity during ecosystem development. Proc. Natl. Acad. Sci. USA 2019, 116, 6891–6896. [Google Scholar] [CrossRef] [Green Version]
- Jansson, J.K.; Hofmockel, K.S. Soil microbiomes and climate change. Nat. Rev. Microbiol. 2020, 18, 35–46. [Google Scholar] [CrossRef]
- Bajerski, F.; Wagner, D. Bacterial succession in Antarctic soils of two glacier forefields on Larsemann Hills, East Antarctica. FEMS Microbiol. Ecol. 2013, 85, 128–142. [Google Scholar] [CrossRef]
- Sun, H.; Wu, Y.; Zhou, J.; Bing, H. Variations of bacterial and fungal communities along a primary successional chronosequence in the Hailuogou glacier retreat area (Gongga Mountain, SW China). J. Mt. Sci. 2016, 13, 1621–1631. [Google Scholar] [CrossRef]
- Deonalli, D.; Sharma, R.; Jangid, K. Microbial Community Dynamics during Soil Ecosystem Development. In Mining of Microbial Wealth and MetaGenomics; Springer: Singapore, 2017; pp. 57–82. [Google Scholar] [CrossRef]
- Genderjahn, S.; Alawi, M.; Mangelsdorf, K.; Horn, F.; Wagner, D. Desiccation- and Saline-Tolerant Bacteria and Archaea in Kalahari Pan Sediments. Front. Microbiol. 2018, 9, 2082. [Google Scholar] [CrossRef]
- Meier, L.A.; Krauze, P.; Prater, I.; Horn, F.; Schaefer, C.E.G.R.; Scholten, T.; Wagner, D.; Mueller, C.W.; Kühn, P. Pedogenic and microbial interrelation in initial soils under semiarid climate on James Ross Island, Antarctic Peninsula region. Biogeosciences 2019, 16, 2481–2499. [Google Scholar] [CrossRef] [Green Version]
- Garrido-Benavent, I.; Pérez-Ortega, S.; Durán, J.; Ascaso, C.; Pointing, S.B.; Rodríguez-Cielos, R.; Navarro, F.; de Los Ríos, A. Differential colonization and succession of microbial communities in rock and soil substrates on a maritime antarctic glacier forefield. Front. Microbiol. 2020, 11, 126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krauze, P.; Wagner, D.; Yang, S.; Spinola, D.; Kühn, P. Influence of prokaryotic microorganisms on initial soil formation along a glacier forefield on King George Island, maritime Antarctica. Sci. Rep. 2021, 11, 13135. [Google Scholar] [CrossRef] [PubMed]
- Jangid, K.; Whitman, W.B.; Condron, L.M.; Turner, B.L.; Williams, M.A. Progressive and retrogressive ecosystem development coincide with soil bacterial community change in a dune system under lowland temperate rainforest in New Zealand. Plant Soil 2013, 367, 235–247. [Google Scholar] [CrossRef]
- Turner, B.L.; Zemunik, G.; Laliberté, E.; Drake, J.J.; Jones, F.A.; Saltonstall, K.; Chang, C. Contrasting patterns of plant and microbial diversity during long-term ecosystem development. J. Ecol. 2019, 107, 606–621. [Google Scholar] [CrossRef]
- Bardelli, T.; Gomez-Brandon, M.; Ascher-Jenull, J.; Fornasier, F.; Arfaioli, P.; Francioli, D.; Egli, M.; Sartori, G.; Insam, H.; Pietramellara, G. Effects of slope exposure on soil physico-chemical and microbiological properties along an altitudinal climosequence in the Italian Alps. Sci. Total Environ. 2017, 575, 1041–1055. [Google Scholar] [CrossRef]
- Bernhard, N.; Moskwa, L.-M.; Schmidt, K.; Oeser, R.A.; Aburto, F.; Bader, M.Y.; Baumann, K.; von Blanckenburg, F.; Boy, J.; van den Brink, L.; et al. Pedogenic and microbial interrelations to regional climate and local topography: New insights from a climate gradient (arid to humid) along the Coastal Cordillera of Chile. Catena 2018, 170, 335–355. [Google Scholar] [CrossRef]
- Portes, R.d.C.; Spinola, D.N.; Reis, J.S.; Ker, J.C.; Costa, L.M.d.; Fernandes Filho, E.I.; Kühn, P.; Schaefer, C.E.G.R. Pedogenesis across a climatic gradient in tropical high mountains, Cordillera Blanca—Peruvian Andes. Catena 2016, 147, 441–452. [Google Scholar] [CrossRef]
- Boix-Fayos, C.; Calvo-Cases, A.; Imeson, A.; Soriano-Soto, M.; Tiemessen, I. Spatial and short-term temporal variations in runoff, soil aggregation and other soil properties along a Mediterranean climatological gradient. Catena 1998, 33, 123–138. [Google Scholar] [CrossRef]
- Jarmer, T.; Shoshany, M. Relationships between soil spectral and chemical properties along a climatic gradient in the Judean desert. Arid. Land Res. Manag. 2016, 30, 123–137. [Google Scholar] [CrossRef]
- Laliberte, E.; Kardol, P.; Didham, R.K.; Teste, F.P.; Turner, B.L.; Wardle, D.A. Soil fertility shapes belowground food webs across a regional climate gradient. Ecol. Lett. 2017, 20, 1273–1284. [Google Scholar] [CrossRef]
- Rasmussen, C.; Dahlgren, R.A.; Southard, R.J. Basalt weathering and pedogenesis across an environmental gradient in the southern Cascade Range, California, USA. Geoderma 2010, 154, 473–485. [Google Scholar] [CrossRef]
- Carletti, P.; Vendramin, E.; Pizzeghello, D.; Concheri, G.; Zanella, A.; Nardi, S.; Squartini, A. Soil humic compounds and microbial communities in six spruce forests as function of parent material, slope aspect and stand age. Plant Soil 2008, 315, 47–65. [Google Scholar] [CrossRef]
- Bennie, J.; Huntley, B.; Wiltshire, A.; Hill, M.O.; Baxter, R. Slope, aspect and climate: Spatially explicit and implicit models of topographic microclimate in chalk grassland. Ecol. Model. 2008, 216, 47–59. [Google Scholar] [CrossRef]
- Huang, Y.-M.; Liu, D.; An, S.-S. Effects of slope aspect on soil nitrogen and microbial properties in the Chinese Loess region. Catena 2015, 125, 135–145. [Google Scholar] [CrossRef]
- Chu, H.; Xiang, X.; Yang, J.; Adams, J.M.; Zhang, K.; Li, Y.; Shi, Y. Effects of Slope Aspects on Soil Bacterial and Arbuscular Fungal Communities in a Boreal Forest in China. Pedosphere 2016, 26, 226–234. [Google Scholar] [CrossRef]
- Scholten, T.; Goebes, P.; Kühn, P.; Seitz, S.; Assmann, T.; Bauhus, J.; Bruelheide, H.; Buscot, F.; Erfmeier, A.; Fischer, M. On the combined effect of soil fertility and topography on tree growth in subtropical forest ecosystems—A study from SE China. J. Plant Ecol. 2017, 10, 111–127. [Google Scholar] [CrossRef]
- Oeser, R.A.; Stroncik, N.; Moskwa, L.-M.; Bernhard, N.; Schaller, M.; Canessa, R.; van den Brink, L.; Köster, M.; Brucker, E.; Stock, S.; et al. Chemistry and microbiology of the Critical Zone along a steep climate and vegetation gradient in the Chilean Coastal Cordillera. Catena 2018, 170, 183–203. [Google Scholar] [CrossRef] [Green Version]
- Pen-Mouratov, S.; Berg, N.; Genzer, N.; Ukabi, S.; Shargil, D.; Steinberger, Y. Do slope orientation and sampling location determine soil biota composition? Front. Biol. China 2009, 4, 364–375. [Google Scholar] [CrossRef]
- Crits-Christoph, A.; Robinson, C.K.; Barnum, T.; Fricke, W.F.; Davila, A.F.; Jedynak, B.; McKay, C.P.; DiRuggiero, J. Colonization patterns of soil microbial communities in the Atacama Desert. Microbiome 2013, 1, 28. [Google Scholar] [CrossRef] [Green Version]
- Neilson, J.W.; Califf, K.; Cardona, C.; Copeland, A.; Van Treuren, W.; Josephson, K.L.; Knight, R.; Gilbert, J.A.; Quade, J.; Caporaso, J.G. Significant impacts of increasing aridity on the arid soil microbiome. MSystems 2017, 2, e00195-16. [Google Scholar] [CrossRef] [Green Version]
- Schulze-Makuch, D.; Wagner, D.; Kounaves, S.P.; Mangelsdorf, K.; Devine, K.G.; de Vera, J.P.; Schmitt-Kopplin, P.; Grossart, H.P.; Parro, V.; Kaupenjohann, M.; et al. Transitory microbial habitat in the hyperarid Atacama Desert. Proc. Natl. Acad. Sci. USA 2018, 115, 2670–2675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knief, C.; Bol, R.; Amelung, W.; Kusch, S.; Frindte, K.; Eckmeier, E.; Jaeschke, A.; Dunai, T.; Fuentes, B.; Mörchen, R.; et al. Tracing elevational changes in microbial life and organic carbon sources in soils of the Atacama Desert. Glob. Planet. Chang. 2020, 184, 103078. [Google Scholar] [CrossRef]
- Schulze-Makuch, D.; Lipus, D.; Arens, F.L.; Baqué, M.; Bornemann, T.L.; de Vere, J.-P.; Flury, M.; Frösler, J.; Heinz, J.; Hwang, Y. Microbial hotspots in lithic microhabitats inferred from DNA fractionation and metagenomics in the Atacama Desert. Microorganisms 2021, 9, 1038. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.F.; Fernández, B.; Varas, E.; Pastén, P.; Gómez, D.; Rengifo, P.; Muñoz, J.; Atenas, M.; Jofré, J.C.; Moreno, T. Chilean water resources. In The Geology of Chile; Geological Society of London: London, UK, 2007; pp. 215–230. [Google Scholar]
- Übernickel, K.; Ehlers, T.A.; Ershadi, M.R.; Paulino, L.; Fuentes Espoz, J.-P.; Maldonado, A.; Oses-Pedraza, R.; von Blanckenburg, F. Time Series of Meteorological Station Data in the EarthShape Study Areas in the Coastal Cordillera, Chile; GFZ: Potsdam, Germany, 2020. [Google Scholar]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108, 4516–4522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data; Babraham Bioinformatics: Cambridgeshire, UK, 2010. [Google Scholar]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [Green Version]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Quince, C.; Mahé, F. VSEARCH: A versatile open source tool for metagenomics. PeerJ 2016, 4, e2584. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Stevens, M.H.H.; Oksanen, M.J.; Suggests, M. The vegan package. Community Ecol. Package 2007, 10, 719. [Google Scholar]
- Roberts, D.W.; Roberts, M.D.W. Package ‘labdsv’. Ordination Multivar. 2016, 775, 21–51. [Google Scholar]
- Wickham, H. ggplot2. Wiley Interdiscip. Rev. Comput. Stat. 2011, 3, 180–185. [Google Scholar] [CrossRef]
- Šmilauer, P.; Lepš, J. Multivariate Analysis of Ecological Data Using CANOCO 5; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Fierer, N.; Jackson, R.B. The diversity and biogeography of soil bacterial communities. Proc. Natl. Acad. Sci. USA 2006, 103, 626–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rousk, J.; Baath, E.; Brookes, P.C.; Lauber, C.L.; Lozupone, C.; Caporaso, J.G.; Knight, R.; Fierer, N. Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J. 2010, 4, 1340–1351. [Google Scholar] [CrossRef]
- Ramírez, P.B.; Fuentes-Alburquenque, S.; Díez, B.; Vargas, I.; Bonilla, C.A. Soil microbial community responses to labile organic carbon fractions in relation to soil type and land use along a climate gradient. Soil Biol. Biochem. 2020, 141, 107692. [Google Scholar] [CrossRef]
- Lauber, C.L.; Hamady, M.; Knight, R.; Fierer, N. Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl. Environ. Microbiol. 2009, 75, 5111–5120. [Google Scholar] [CrossRef] [Green Version]
- Samolov, E.; Baumann, K.; Büdel, B.; Jung, P.; Leinweber, P.; Mikhailyuk, T.; Karsten, U.; Glaser, K. Biodiversity of algae and cyanobacteria in biological soil crusts collected along a climatic gradient in Chile using an integrative approach. Microorganisms 2020, 8, 1047. [Google Scholar] [CrossRef]
- Yang, G.; Wagg, C.; Veresoglou, S.D.; Hempel, S.; Rillig, M.C. How soil biota drive ecosystem stability. Trends Plant Sci. 2018, 23, 1057–1067. [Google Scholar] [CrossRef]
- Maron, P.-A.; Sarr, A.; Kaisermann, A.; Lévêque, J.; Mathieu, O.; Guigue, J.; Karimi, B.; Bernard, L.; Dequiedt, S.; Terrat, S. High microbial diversity promotes soil ecosystem functioning. Appl. Environ. Microbiol. 2018, 84, e02738-17. [Google Scholar] [CrossRef] [Green Version]
- Neilson, J.W.; Quade, J.; Ortiz, M.; Nelson, W.M.; Legatzki, A.; Tian, F.; LaComb, M.; Betancourt, J.L.; Wing, R.A.; Soderlund, C.A.; et al. Life at the hyperarid margin: Novel bacterial diversity in arid soils of the Atacama Desert, Chile. Extrem. Life Under Extrem. Cond. 2012, 16, 553–566. [Google Scholar] [CrossRef]
- Zhang, B.; Wu, X.; Tai, X.; Sun, L.; Wu, M.; Zhang, W.; Chen, X.; Zhang, G.; Chen, T.; Liu, G. Variation in actinobacterial community composition and potential function in different soil ecosystems belonging to the arid Heihe River Basin of Northwest China. Front. Microbiol. 2019, 10, 2209. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Baquerizo, M.; Maestre, F.T.; Reich, P.B.; Trivedi, P.; Osanai, Y.; Liu, Y.R.; Hamonts, K.; Jeffries, T.C.; Singh, B.K. Carbon content and climate variability drive global soil bacterial diversity patterns. Ecol. Monogr. 2016, 86, 373–390. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, M.; Zhang, X.; Sun, Q.; Liu, R.; Lian, B. Shift of the microbial communities from exposed sandstone rocks to forest soils during pedogenesis. Int. Biodeterior. Biodegrad. 2019, 140, 21–28. [Google Scholar] [CrossRef]
- Biesgen, D.; Frindte, K.; Maarastawi, S.; Knief, C. Clay content modulates differences in bacterial community structure in soil aggregates of different size. Geoderma 2020, 376, 114544. [Google Scholar] [CrossRef]
- Xu, Q.; Vandenkoornhuyse, P.; Li, L.; Guo, J.; Zhu, C.; Guo, S.; Ling, N.; Shen, Q. Microbial generalists and specialists differently contribute to the community diversity in farmland soils. J. Adv. Res. 2021; in press. [Google Scholar] [CrossRef]
- Szekely, A.J.; Langenheder, S. The importance of species sorting differs between habitat generalists and specialists in bacterial communities. FEMS Microbiol. Ecol. 2014, 87, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Bell, T.H.; Bell, T. Many roads to bacterial generalism. FEMS Microbiol. Ecol. 2021, 97, fiaa240. [Google Scholar] [CrossRef]
- Mariadassou, M.; Pichon, S.; Ebert, D. Microbial ecosystems are dominated by specialist taxa. Ecol. Lett. 2015, 18, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Willms, I.M.; Rudolph, A.Y.; Göschel, I.; Bolz, S.H.; Schneider, D.; Penone, C.; Poehlein, A.; Schöning, I.; Nacke, H. Globally abundant “Candidatus Udaeobacter” benefits from release of antibiotics in soil and potentially performs trace gas scavenging. Msphere 2020, 5, e00186-20. [Google Scholar] [CrossRef]
- Delgado-Baquerizo, M.; Oliverio, A.M.; Brewer, T.E.; Benavent-González, A.; Eldridge, D.J.; Bardgett, R.D.; Maestre, F.T.; Singh, B.K.; Fierer, N. A global atlas of the dominant bacteria found in soil. Science 2018, 359, 320–325. [Google Scholar] [CrossRef] [Green Version]
- Favet, J.; Lapanje, A.; Giongo, A.; Kennedy, S.; Aung, Y.Y.; Cattaneo, A.; Davis-Richardson, A.G.; Brown, C.T.; Kort, R.; Brumsack, H.J.; et al. Microbial hitchhikers on intercontinental dust: Catching a lift in Chad. ISME J. 2013, 7, 850–867. [Google Scholar] [CrossRef] [PubMed]
- Llado, S.; Lopez-Mondejar, R.; Baldrian, P. Forest Soil Bacteria: Diversity, Involvement in Ecosystem Processes, and Response to Global Change. Microbiol. Mol. Biol. Rev. 2017, 81, e00063-16. [Google Scholar] [CrossRef] [Green Version]
- Adamczyk, M.; Hagedorn, F.; Wipf, S.; Donhauser, J.; Vittoz, P.; Rixen, C.; Frossard, A.; Theurillat, J.-P.; Frey, B. The soil microbiome of Gloria Mountain summits in the Swiss Alps. Front. Microbiol. 2019, 10, 1080. [Google Scholar] [CrossRef]
- Richmond, C.E.; Breitburg, D.L.; Rose, K.A. The role of environmental generalist species in ecosystem function. Ecol. Model. 2005, 188, 279–295. [Google Scholar] [CrossRef]
- Sanchez-Maranon, M.; Miralles, I.; Aguirre-Garrido, J.F.; Anguita-Maeso, M.; Millan, V.; Ortega, R.; Garcia-Salcedo, J.A.; Martinez-Abarca, F.; Soriano, M. Changes in the soil bacterial community along a pedogenic gradient. Sci. Rep. 2017, 7, 14593. [Google Scholar] [CrossRef]
- Aanderud, Z.T.; Saurey, S.; Ball, B.A.; Wall, D.H.; Barrett, J.E.; Muscarella, M.E.; Griffin, N.A.; Virginia, R.A.; Adams, B.J. Stoichiometric Shifts in Soil C:N:P Promote Bacterial Taxa Dominance, Maintain Biodiversity, and Deconstruct Community Assemblages. Front. Microbiol. 2018, 9, 1401. [Google Scholar] [CrossRef]
- O’Brien, F.J.M.; Almaraz, M.; Foster, M.A.; Hill, A.F.; Huber, D.P.; King, E.K.; Langford, H.; Lowe, M.A.; Mickan, B.S.; Miller, V.S.; et al. Soil Salinity and pH Drive Soil Bacterial Community Composition and Diversity Along a Lateritic Slope in the Avon River Critical Zone Observatory, Western Australia. Front. Microbiol. 2019, 10, 1486. [Google Scholar] [CrossRef]
- Jung, P.; Baumann, K.; Lehnert, L.W.; Samolov, E.; Achilles, S.; Schermer, M.; Wraase, L.M.; Eckhardt, K.U.; Bader, M.Y.; Leinweber, P. Desert breath—How fog promotes a novel type of soil biocenosis, forming the coastal Atacama Desert’s living skin. Geobiology 2020, 18, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Jung, P.; Baumann, K.; Emrich, D.; Springer, A.; Felde, V.J.; Dultz, S.; Baum, C.; Frank, M.; Büdel, B.; Leinweber, P. Lichens Bite the Dust–A Bioweathering Scenario in the Atacama Desert. Iscience 2020, 23, 101647. [Google Scholar] [CrossRef] [PubMed]
- Oeser, R.A.; von Blanckenburg, F. Do degree and rate of silicate weathering depend on plant productivity? Biogeosciences 2020, 17, 4883–4917. [Google Scholar] [CrossRef]
- Koester, M.; Stock, S.C.; Nájera, F.; Abdallah, K.; Gorbushina, A.; Prietzel, J.; Matus, F.; Klysubun, W.; Boy, J.; Kuzyakov, Y. From rock eating to vegetarian ecosystems—Disentangling processes of phosphorus acquisition across biomes. Geoderma 2021, 388, 114827. [Google Scholar] [CrossRef]
- Yan, Y.; Klinkhamer, P.G.L.; van Veen, J.A.; Kuramae, E.E. Environmental filtering: A case of bacterial community assembly in soil. Soil Biol. Biochem. 2019, 136, 107531. [Google Scholar] [CrossRef]
- Fierer, N.; Leff, J.W.; Adams, B.J.; Nielsen, U.N.; Bates, S.T.; Lauber, C.L.; Owens, S.; Gilbert, J.A.; Wall, D.H.; Caporaso, J.G. Cross-biome metagenomic analyses of soil microbial communities and their functional attributes. Proc. Natl. Acad. Sci. USA 2012, 109, 21390–21395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berkelmann, D.; Schneider, D.; Engelhaupt, M.; Heinemann, M.; Christel, S.; Wijayanti, M.; Meryandini, A.; Daniel, R. How Rainforest Conversion to Agricultural Systems in Sumatra (Indonesia) Affects Active Soil Bacterial Communities. Front. Microbiol. 2018, 9, 2381. [Google Scholar] [CrossRef] [PubMed]
- Dukunde, A.; Schneider, D.; Schmidt, M.; Veldkamp, E.; Daniel, R. Tree Species Shape Soil Bacterial Community Structure and Function in Temperate Deciduous Forests. Front. Microbiol. 2019, 10, 1519. [Google Scholar] [CrossRef]
- Ai, C.; Zhang, S.; Zhang, X.; Guo, D.; Zhou, W.; Huang, S. Distinct responses of soil bacterial and fungal communities to changes in fertilization regime and crop rotation. Geoderma 2018, 319, 156–166. [Google Scholar] [CrossRef]
- Jiang, Y.; Lei, Y.; Yang, Y.; Korpelainen, H.; Niinemets, Ü.; Li, C. Divergent assemblage patterns and driving forces for bacterial and fungal communities along a glacier forefield chronosequence. Soil Biol. Biochem. 2018, 118, 207–216. [Google Scholar] [CrossRef] [Green Version]
- Wiegand, S.; Jogler, M.; Jogler, C. On the maverick Planctomycetes. FEMS Microbiol. Rev. 2018, 42, 739–760. [Google Scholar] [CrossRef] [Green Version]
- Muller, E.E. Determining microbial niche breadth in the environment for better ecosystem fate predictions. MSystems 2019, 4, e00080-19. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-J.; Leung, P.M.; Wood, J.L.; Bay, S.K.; Hugenholtz, P.; Kessler, A.J.; Shelley, G.; Waite, D.W.; Franks, A.E.; Cook, P.L. Metabolic flexibility allows bacterial habitat generalists to become dominant in a frequently disturbed ecosystem. ISME J. 2021, 15, 2986–3004. [Google Scholar] [CrossRef]
- Hartley, A.J.; Chong, G.; Houston, J.; Mather, A.E. 150 million years of climatic stability: Evidence from the Atacama Desert, northern Chile. J. Geol. Soc. 2005, 162, 421–424. [Google Scholar] [CrossRef]
- Mucina, L.; Wardell-Johnson, G.W. Landscape age and soil fertility, climatic stability, and fire regime predictability: Beyond the OCBIL framework. Plant Soil 2011, 341, 1–23. [Google Scholar] [CrossRef]
- Arroyo, M.T.K.; Zedler, P.H.; Fox, M.D. Ecology and Biogeography of Mediterranean Ecosystems in Chile, California, and Australia; Springer Science & Business Media: New York, NY, USA, 2012; Volume 108. [Google Scholar]
- Cowling, R.M.; Potts, A.J.; Bradshaw, P.L.; Colville, J.; Arianoutsou, M.; Ferrier, S.; Forest, F.; Fyllas, N.M.; Hopper, S.D.; Ojeda, F. Variation in plant diversity in mediterranean-climate ecosystems: The role of climatic and topographical stability. J. Biogeogr. 2015, 42, 552–564. [Google Scholar] [CrossRef]
- Xue, R.; Yang, Q.; Miao, F.; Wang, X.; Shen, Y. Slope aspect influences plant biomass, soil properties and microbial composition in alpine meadow on the Qinghai-Tibetan plateau. J. Soil Sci. Plant Nutr. 2018, 18, 1–12. [Google Scholar] [CrossRef]
- Šťovíček, A.; Kim, M.; Or, D.; Gillor, O. Microbial community response to hydration-desiccation cycles in desert soil. Sci. Rep. 2017, 7, 45735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cisterna, R.G. Dirección Meteorológica de Chile—Anuarios Climatológicos. Available online: https://climatologia.meteochile.gob.cl/application/publicaciones/documentoPdf/anuario/anuario-2019.pdf (accessed on 20 October 2021).
- Lehnert, L.W.; Thies, B.; Bendix, J. A new high spatial resolution low stratus/fog retrieval for the Atacama Desert. Remote Sens. Environ. 2020, 236, 111445. [Google Scholar] [CrossRef]
- Miralles, I.; Soria, R.; Lucas-Borja, M.; Soriano, M.; Ortega, R. Effect of biocrusts on bacterial community composition at different soil depths in Mediterranean semi-arid ecosystems. Sci. Total Environ. 2020, 733, 138613. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Liu, J.; Zhang, B.; Zhao, H.; Huang, G. Habitat-specific environmental factors regulate spatial variability of soil bacterial communities in biocrusts across northern China’s drylands. Sci. Total Environ. 2020, 719, 137479. [Google Scholar] [CrossRef]
- Maestre, F.T.; Delgado-Baquerizo, M.; Jeffries, T.C.; Eldridge, D.J.; Ochoa, V.; Gozalo, B.; Quero, J.L.; Garcia-Gomez, M.; Gallardo, A.; Ulrich, W. Increasing aridity reduces soil microbial diversity and abundance in global drylands. Proc. Natl. Acad. Sci. USA 2015, 112, 15684–15689. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Wang, S.; Chen, J. Transformations from specialists to generalists cause bacterial communities are more stable than micro-eukaryotic communities under anthropogenic activity disturbance. Sci. Total Environ. 2021, 790, 148141. [Google Scholar] [CrossRef]
- Clavel, J.; Julliard, R.; Devictor, V. Worldwide decline of specialist species: Toward a global functional homogenization? Front. Ecol. Environ. 2011, 9, 222–228. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez, V.; Moskwa, L.-M.; Oses, R.; Kühn, P.; Riveras-Muñoz, N.; Seguel, O.; Scholten, T.; Wagner, D. Impact of Climate and Slope Aspects on the Composition of Soil Bacterial Communities Involved in Pedogenetic Processes along the Chilean Coastal Cordillera. Microorganisms 2022, 10, 847. https://doi.org/10.3390/microorganisms10050847
Rodriguez V, Moskwa L-M, Oses R, Kühn P, Riveras-Muñoz N, Seguel O, Scholten T, Wagner D. Impact of Climate and Slope Aspects on the Composition of Soil Bacterial Communities Involved in Pedogenetic Processes along the Chilean Coastal Cordillera. Microorganisms. 2022; 10(5):847. https://doi.org/10.3390/microorganisms10050847
Chicago/Turabian StyleRodriguez, Victoria, Lisa-Marie Moskwa, Rómulo Oses, Peter Kühn, Nicolás Riveras-Muñoz, Oscar Seguel, Thomas Scholten, and Dirk Wagner. 2022. "Impact of Climate and Slope Aspects on the Composition of Soil Bacterial Communities Involved in Pedogenetic Processes along the Chilean Coastal Cordillera" Microorganisms 10, no. 5: 847. https://doi.org/10.3390/microorganisms10050847
APA StyleRodriguez, V., Moskwa, L.-M., Oses, R., Kühn, P., Riveras-Muñoz, N., Seguel, O., Scholten, T., & Wagner, D. (2022). Impact of Climate and Slope Aspects on the Composition of Soil Bacterial Communities Involved in Pedogenetic Processes along the Chilean Coastal Cordillera. Microorganisms, 10(5), 847. https://doi.org/10.3390/microorganisms10050847