Structural and Metabolic Profiling of Lycopersicon esculentum Rhizosphere Microbiota Artificially Exposed at Commonly Used Non-Steroidal Anti-Inflammatory Drugs
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Set-Up
2.2. Rhizosphere Microbiota Analysis through PLFA Approach
2.3. Rhizosphere Microbiota Responses Evaluation
2.3.1. Community-Level Physiological Profile (CLPP)
2.3.2. Emitted VOCs
2.4. Statistical Interpretation of Data
3. Results and Discussions
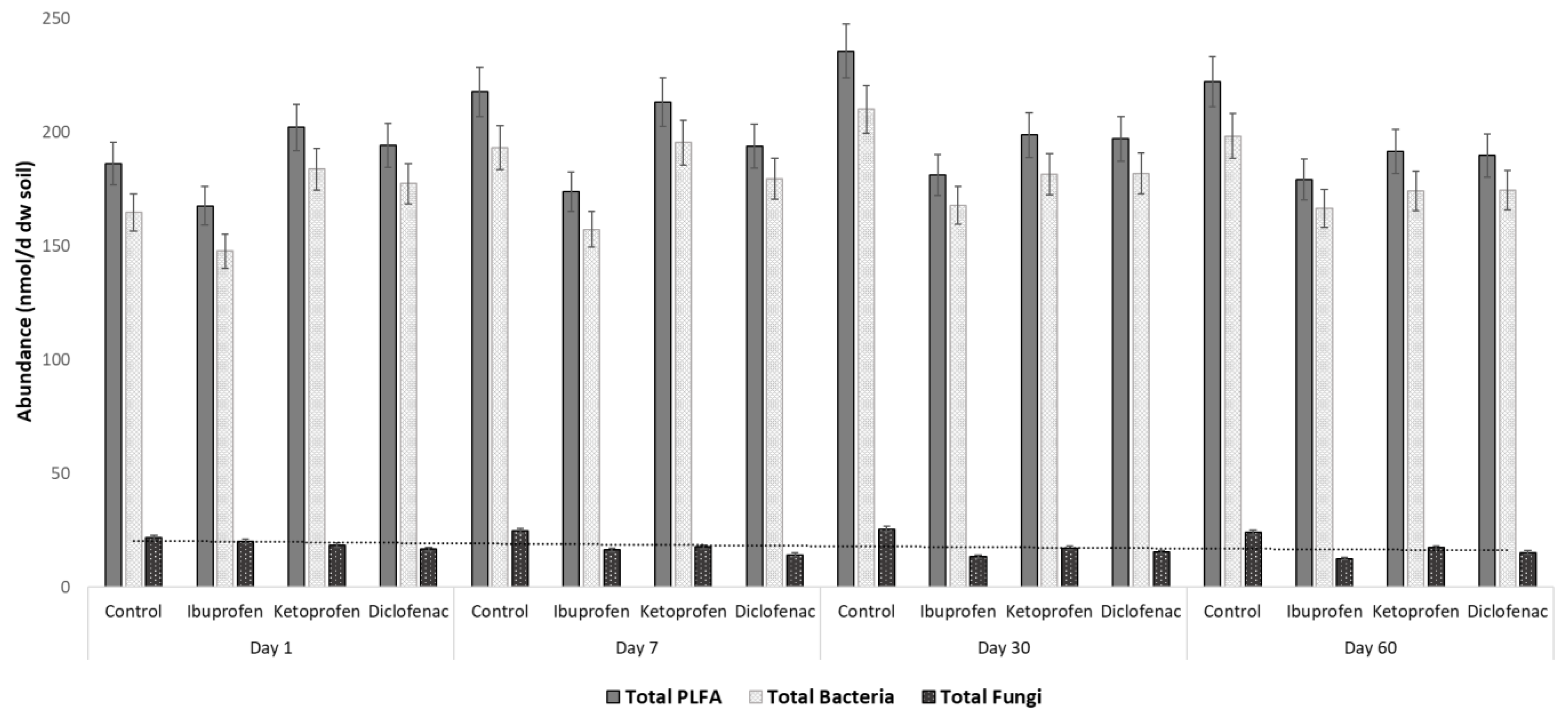
3.1. Rhizosphere Microbiota Abundance Changes with Contamination of NSAIDs
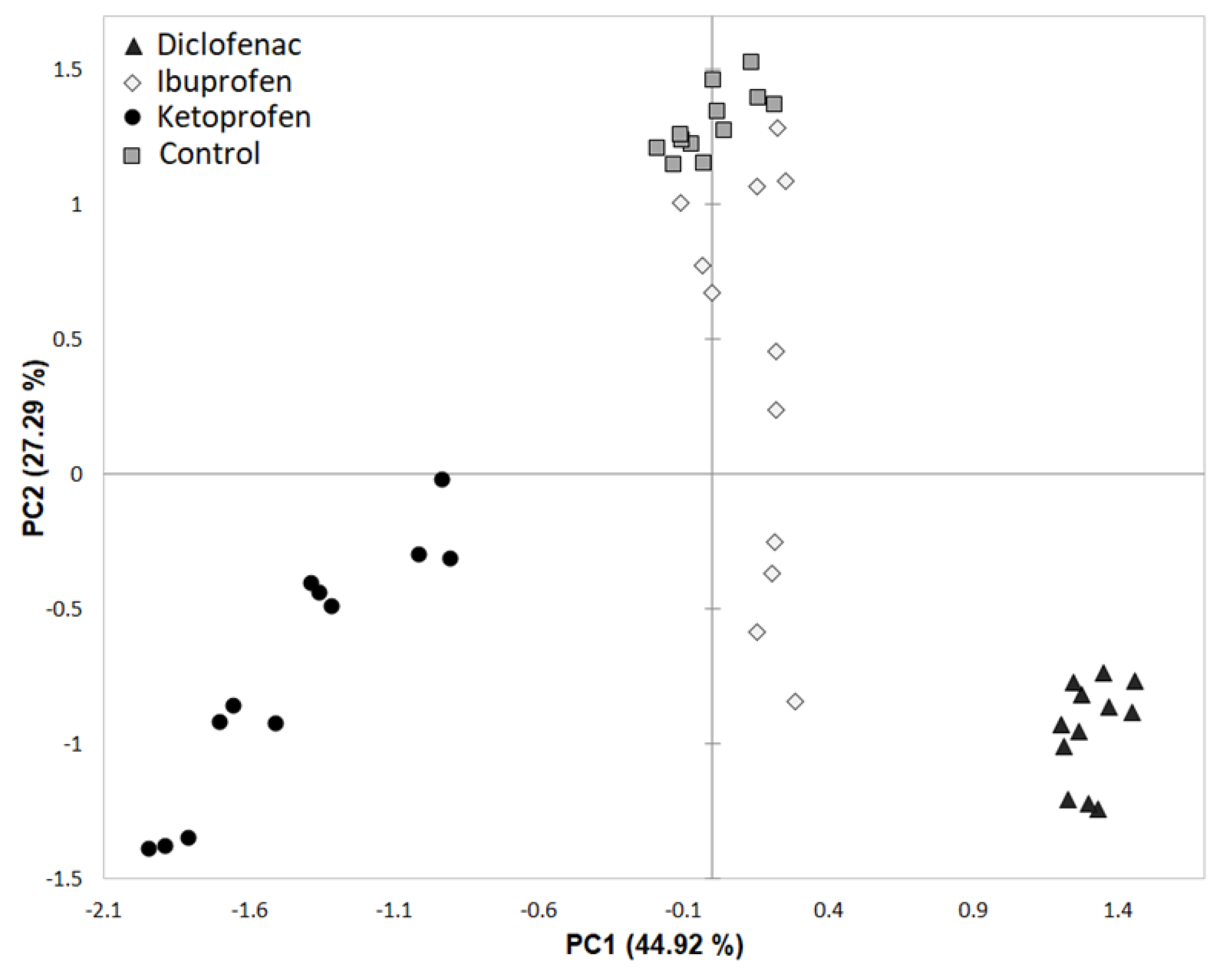
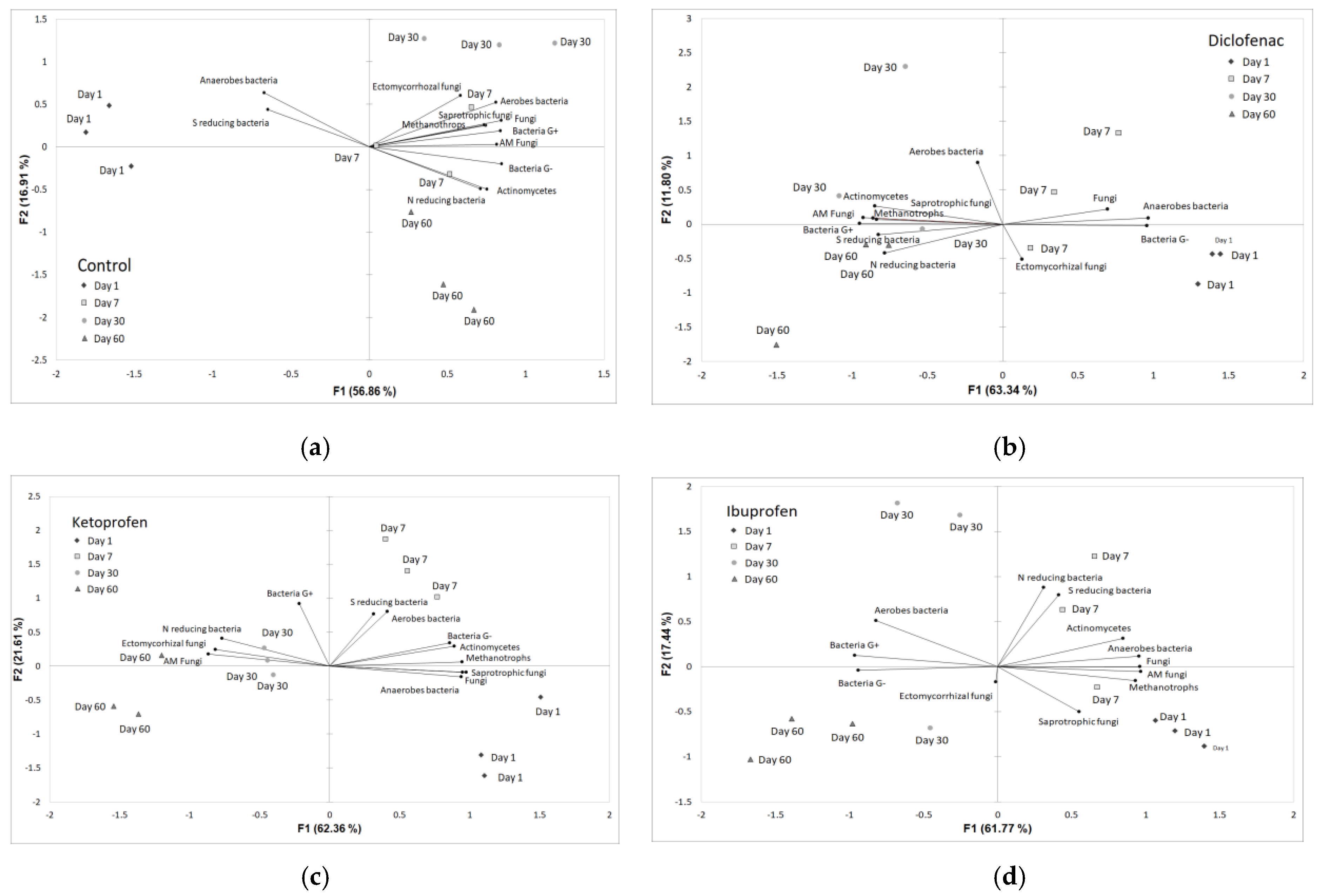
3.2. Rhizosphere Microbiota Community Structure Changes in Time with Contamination of NSAIDs
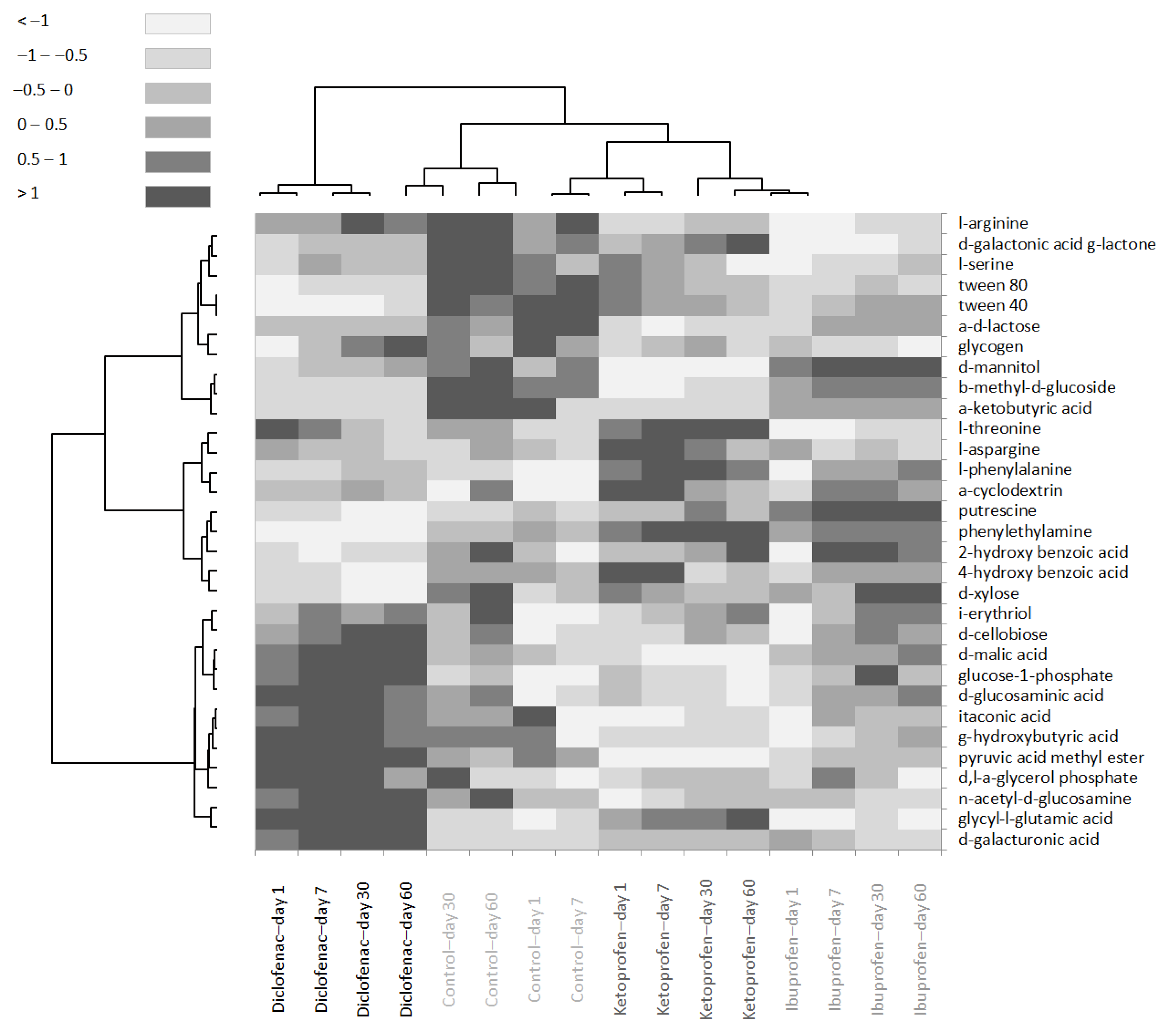
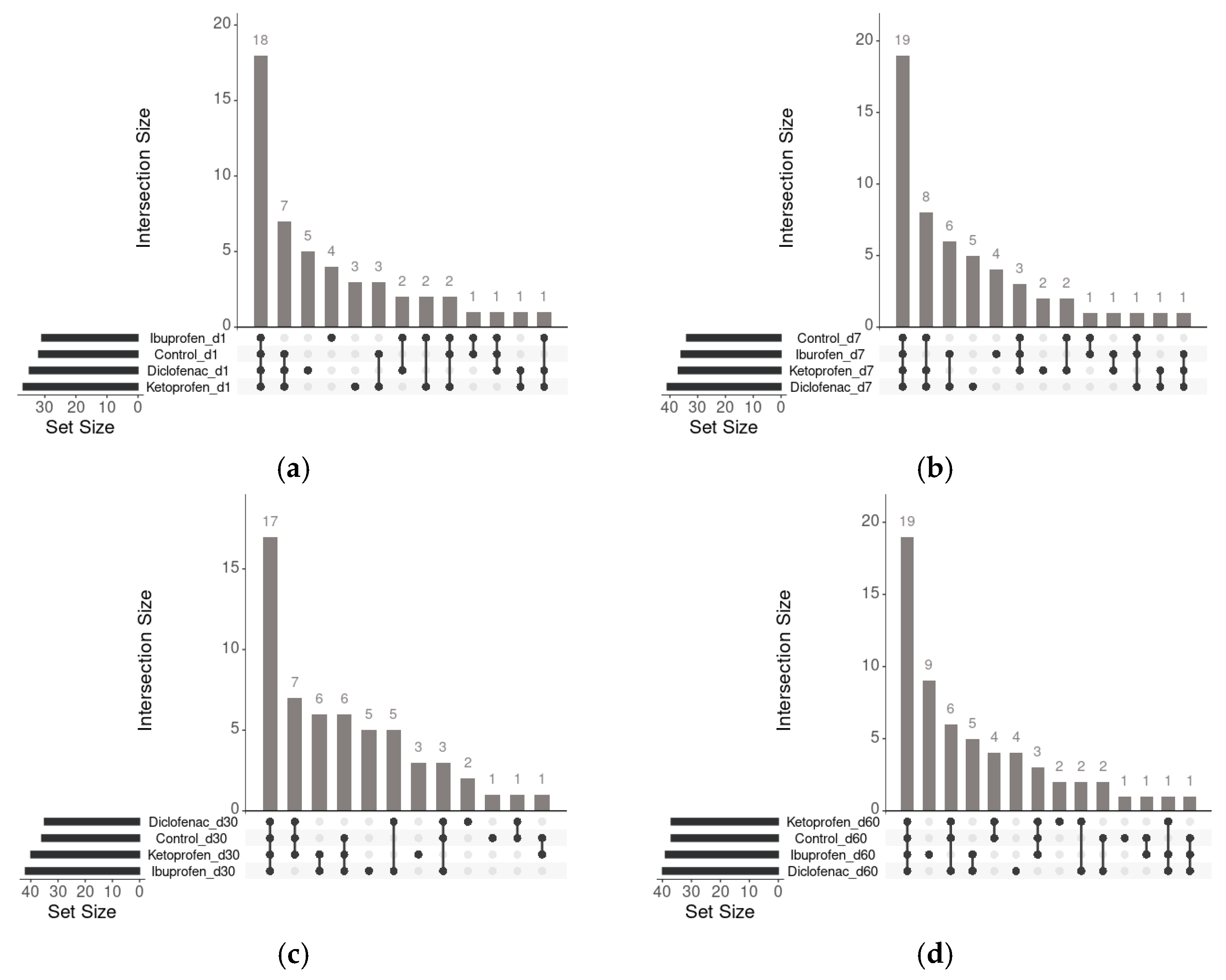
3.3. Community-Level Physiological Profile Changes in Time with Contamination of Nsaids
3.4. Microbiota Emitted Volatile Organic Compounds Changes in Time with Contamination of Nsaids
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kamat, S.; Marathe, P.; Tripathi, R.; Raut, S.; Khatri, N. Over-the-counter medicines: Global perspective and Indian scenario. J. Postgrad. Med. 2020, 66, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, P.; Shukla, P.; Giri, B.S.; Chowdhary, P.; Chandra, R.; Gupta, P.; Pandey, A. Prevalence and hazardous impact of pharmaceutical and personal care products and antibiotics in environment: A review on emerging contaminants. Environ. Res. 2021, 194, 110664. [Google Scholar] [CrossRef] [PubMed]
- Praveenkumarreddy, Y.; Vimalkumar, K.; Ramaswamy, B.R.; Kumar, V.; Singhai, R.K.; Basu, H.; Gopal, C.M.; Vandana, K.E.; Bhat, K.; Udayashankar, H.K.; et al. Assessment of non-steroidal anti-inflammatory drugs from selected wastewater treatment plants of Southwestern India. Emerg. Contam. 2021, 7, 43–51. [Google Scholar] [CrossRef]
- Alenzi, A.; Hunter, C.; Spencer, J.; Roberts, J.; Craft, J.; Pahl, O.; Escudero, A. Pharmaceuticals effect and removal, at environmentally relevant concentrations, from sewage sludge during anaerobic digestion. Bioresour. Technol. 2021, 319, 124102. [Google Scholar] [CrossRef] [PubMed]
- Mordechaay, E.B.; Mordehay, V.; Tachitzky, J.; Chefetz, B. Pharmaceuticals in edible crops irrigated with reclaimed wastewater: Evidence from large survey in Israel. J. Hazard. Mater. 2021, 416, 126184. [Google Scholar] [CrossRef]
- Grabicova, K.; Grabic, R.; Fedorova, G.; Stanova, A.V.; Blaha, M.; Randak, T.; Brooks, B.W.; Zlabek, V. Water reuse and aquaculture: Pharmaceutical bioaccumulation by fish during tertiary treatment in wastewater stabilization pond. Environ. Pollut. 2020, 267, 115593. [Google Scholar] [CrossRef]
- Mejias, C.; Santos, L.J.; Aparicio, I.; Alonso, E. Occurrence of pharmaceuticals and their metabolites in sewage sludge and soil: A review on their distribution and environmental risk assessment. Trends Environ. Anal. Chem. 2021, 30, e00125. [Google Scholar] [CrossRef]
- Xu, J.; Wu, L.; Chen, W.; Chang, C.A. Leaching potential of nonsteroidal anti-inflammatory drugs in soils. Environ. Toxicol. Chem. 2010, 29, 800–807. [Google Scholar] [CrossRef]
- Duan, Y.P.; Meng, X.Z.; Wen, Z.H.; Chen, L. Acidic pharmaceuticals in domestic wastewater and receiving water from hyper-urbanization city of China (Shanghai): Environmental release and ecological risk. Environ. Sci. Pollut. Res. 2013, 20, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Cycon, M.; Borymski, S.; Zolnierczyk, B.; Piotrowska-Seget, Z. Variable effects of Non-steroidal Anti-inflammatory Drugs (NSAIDs) on selected biochemical processes mediated by soil microorganisms. Front. Microbiol. 2016, 7, 1969. [Google Scholar] [CrossRef]
- Papaioannou, D.; Koukoulakis, P.H.; Papageorgiou, M.; Lambropoulou, D.A.; Kalavrouziotis, I.K. Investigation of pharmaceutical and personal care product interactions of soil and beets (Beta vulgaris L.) under the effect of wastewater reuse. Chemosphere 2020, 236, 124553. [Google Scholar] [CrossRef] [PubMed]
- Wolny-Koladka, K.; Lenart-Boron, A. Phenotypic and molecular assessment of drug resistance profile and genetic diversity of waterborne Escherichia coli. Water Air Soil Pollut. 2016, 227, 146. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wolny-Koladka, K.; Zdaniewicz, M. Antibiotic resistance of Escherichia coli isolated from processing of brewery waste with the addition of bulking agents. Sustainability 2021, 13, 10174. [Google Scholar] [CrossRef]
- Gama-Rodrigues, A.C. Soil organic matter, nutrient cycling, and biological dinitrogen-fixation in agroforestry systems. Agrofor. Syst. 2011, 81, 191–193. [Google Scholar] [CrossRef]
- Ramirez-Guizar, S.; Sykes, H.; Perry, J.D.; Schwalbe, E.C.; Stanforth, S.P.; Perez-Perez, C.I.; Dean, J.R. A chromatographic approach to distinguish Gram-positive from Gram-negative bacteria using exogenous volatile organic compound metabolites. J. Chromatogr. A 2017, 1501, 79–88. [Google Scholar] [CrossRef]
- Brown, R.W.; Bull, I.D.; Journeaux, T.; Chadwick, D.R.; Jones, D.L. Volatile organic compounds (VOCs) allow sensitive differentiation of biological soil quality. Soil Biol. Biochem. 2021, 156, 108187. [Google Scholar] [CrossRef]
- Orlandini, V.; Maida, I.; Fondi, M.; Perrin, E.; Papaleo, M.C.; Bosi, E.; Pascale, D.; Tutino, M.L.; Michaud, L.; Giudice, A.L.; et al. Genomic analysis of three sponge associated Arthrobacter Antactic strains, inhibiting the growth of Burkholderia cepacian complex bacteria by synthesizing volatile organic compounds. Microbiol. Res. 2014, 169, 593–601. [Google Scholar] [CrossRef]
- Lopez, S.M.Y.; Pastorino, G.N.; Balatti, P.A. Volatile organic compounds profile synthesized and released by endophytes of tomato (Solanum lycopersici L.) and their antagonistic role. Arch. Microbiol. 2021, 203, 1383–1397. [Google Scholar] [CrossRef]
- Sanon, A.; Andrianjaka, Z.N.; Prin, Y.; Thioulouse, B.J.; Comte, G.; Duponnois, R. Rhizosphere microbiota interferes with plant-plant interactions. Plant Soil 2009, 321, 259–278. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Lee, Y.H. The bacterial community in the rhizosphere of Kimchi cabbage restructured by volatile compounds emitted from rhizobacterium Proteus vulgaris JBLS202. Appl. Soil Ecol. 2016, 105, 48–56. [Google Scholar] [CrossRef]
- Fincheira, P.; Quiroz, A.; Tortella, G.; Diez, M.C.; Rubilar, O. Current advances in plant-microbe communication via volatile organic compounds as an innovative strategy to improve plant growth. Microbiol. Res. 2021, 247, 126726. [Google Scholar] [CrossRef] [PubMed]
- Pino-Otin, R.M.; Muniz, S.; Val, J.; Navarro, E. Effects of 18 pharmaceuticals on the physiological diversity of edaphic microorganisms. Sci. Total Environ. 2017, 595, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, W.; Wang, J.; Zhu, L.; Wang, J.; Yan, S.; Ahmad, Z. Toxicity of enrofloxacin and cadmium alone and in combination to enzymatic activities and microbial community structure in soil. Environ. Geochem. Health 2019, 41, 2593–2606. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wu, J.; Ying, G.G.; Luo, Z.; Feng, H. Changes in functional diversity of soil microbial community with addition of antibiotics sulfamethoxazole and chlortetracycline. Appl. Microbiol. Biotechnol. 2012, 95, 1615–1623. [Google Scholar] [CrossRef] [PubMed]
- Ramires, P.F.; Tavella, A.R.; Escarrone, L.M.; Volcao, L.M.; Honscha, L.C.; Brum, R.L.; Silva, A.B.; Silva, F.M.R., Jr. Ecotoxicity of triclosan in soil: An approach using different species. Environ. Sci. Pollut. Res. 2021, 28, 41233–41241. [Google Scholar] [CrossRef]
- Chessa, L.; Pusino, A.; Garau, G.; Mangia, N.P.; Pinna, M.V. Soil microbial response to tetracycline in two different soils amended with cow manure. Environ. Sci. Pollut. Res. 2016, 23, 5807–5817. [Google Scholar] [CrossRef]
- Kovacs, E.D.; Silaghi-Dumitrescu, L.; Kovacs, M.H.; Roman, C. Determination of the uptake of ibuprofen, ketoprofen, and diclofenac by tomatoes, radishes, and lettuce by gas chromatography-mass spectrometry (GC-MS). Anal. Lett. 2021, 54, 314–330. [Google Scholar] [CrossRef]
- Blight, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Frostegard, A.; Tunlid, A.; Baath, E. Use and misuse of PLFA measurements in soils. Soil Biol. Biochem. 2011, 43, 1621–1625. [Google Scholar] [CrossRef]
- Joergensen, R.G.; Wichern, F. Quantitative assessment of the fungal contribution to microbial tissue in soil. Soil Biol. Biochem. 2008, 40, 2977–2991. [Google Scholar] [CrossRef]
- Veuger, B.; Pitcher, A.; Schouten, S.; Sinninghe Damste, J.S.; Middelburg, J.J. Nitrification and growth of autotrophic nitrifying bacteria and Thaumarchaeota in the costal North Sea. Biogeoscience 2013, 10, 1775–1785. [Google Scholar] [CrossRef]
- Moulineux, C.J.; Gange, A.C.; Connop, S.P.; Newport, D.J. Are microbial communities in green roof substrates comparable to those in post-industrial sites?—A preliminary study. Urban Ecosyst. 2015, 18, 1245–1260. [Google Scholar] [CrossRef]
- Frostegard, A.; Baath, E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol. Fertil. Soils 1996, 22, 59–65. [Google Scholar] [CrossRef]
- Lindahl, V.; Bakken, L.R. Evaluation of methods for extraction of bacteria from soil. FEMS Microbiol. Ecol. 1995, 16, 135–142. [Google Scholar] [CrossRef]
- Sofo, A.; Ricciuti, P. A standardized method for estimating the functional diversity of soil bacterial community by Biolog® EcoPlatesTM assay—The case study of a sustainable olive orchard. Appl. Sci. 2019, 9, 4035. [Google Scholar] [CrossRef]
- Khan, A.; Mathelier, A. Intervene: A tool for intersection and visualization of multiple gene genomic region sets. BMC Bioinform. 2017, 18, 287. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Romero, I.; Romero, E.; Wittich, R.M.; Dillewijn, P. Bacterial ecotoxicity and shifts in bacterial communities associated with the removal of ibuprofen, diclofenac and triclosan in biopurification systems. Sci. Total Environ. 2020, 741, 140461. [Google Scholar] [CrossRef]
- Frkova, Z.; Vystavna, Y.; Koubova, A.; Grabicova, K.; Grabic, R.; Kodesova, R.; Chronakova, A. Microbial response to selected pharmaceuticals in agricultural soils: Microcosm study on the roles of soil, treatment, and time. Soil Biol. Biochem. 2020, 149, 107924. [Google Scholar] [CrossRef]
- Dai, L.; Wu, T.; Xiong, Y.; Ni, H.; Ding, Y.; Zhang, W.; Chu, S.; Ju, S.; Yu, J. Ibuprofen-mediated potential inhibition of biofilm development and quorum sensing in Pseudomonas aeruginosa. Life Sci. 2019, 237, 116947. [Google Scholar] [CrossRef]
- Oliviera, I.M.; Borges, A.; Borges, F.; Simoes, M. Repurposing ibuprofen to control Staphylococcus aureus biofilm. Eur. J. Med. Chem. 2019, 166, 197–205. [Google Scholar] [CrossRef]
- Paje, M.L.F.; Kuhlicke, U.; Winkler, M.; Neu, T.R. Inhibition of lotic biofilms by diclofenac. Appl. Microbiol. Biotechnol. 2002, 59, 488–492. [Google Scholar] [PubMed]
- Aleanizy, F.S.; Alqahtani, F.Y.; Eltayb, E.K.; Alrumikan, N.; Almebki, R.; Alhossan, A.; Almangour, T.A.; AlQahtani, H. Evaluating the effects of antibiotics sub-inhibitory dose on Pseudomonas aeruginosa quorum sensing dependent virulence and its phenotypes. Saudi J. Biol. Sci. 2021, 28, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Dastidar, S.G.; Ganguly, K.; Chaudhuri, K.; Chakrabarty, A.N. The anti-bacterial action of diclofenac shown by inhibition of DNA synthesis. Int. J. Antimicrob. Agents 2000, 14, 249–251. [Google Scholar] [CrossRef]
- Zhou, G.; Li, N.; Rene, R.E.; Liu, Q.; Dai, M.; Kong, Q. Chemical composition of extracellular polymeric substances and evolution of microbial community in activated sludge exposed to ibuprofen. J. Environ. Manag. 2019, 246, 267–274. [Google Scholar] [CrossRef]
| Soil Property | Argic Phaeozem |
|---|---|
| Clay | 27.2 ± 0.94 |
| Sand | 16.1 ± 0.20 |
| Silt | 56.7 ± 1.37 |
| Texture | Silty Clay Loam |
| Moisture (cm3/cm3) | 0.344 ± 0.01 |
| Soil temperature (°C) | 10.4 ± 0.09 |
| Organic carbon (%) | 6.2 ± 0.12 |
| pH | 5.9 ± 0.09 |
| Parameter | Conditions |
|---|---|
| Inlet temperature | 280 °C |
| Split mode | 40:1 |
| Oven temperature program | 170 °C, increase with 28 °C·min−1 until 288 °C, followed by a new increase with 60 °C·min−1 until 310 °C. This final temperature was maintained constant for 1.25 min |
| Flow | 1.2 mL·min−1 |
| Detector temperature | 300 °C |
| NSAIDs | Day | Fungi/ Bacteria | Gram (−)/ Gram (+) | Aerobes/ Anaerobes | Ectomycorrhizal/ Saprotrophic |
|---|---|---|---|---|---|
| Control | 1 | 0.131 | 2.585 | 2.157 | 0.695 |
| 7 | 0.128 | 2.422 | 3.286 | 0.496 | |
| 30 | 0.122 | 2.151 | 3.090 | 0.669 | |
| 60 | 0.121 | 2.518 | 3.274 | 0.614 | |
| Ibuprofen | 1 | 0.135 | 4.926 | 2.111 | 0.702 |
| 7 | 0.104 | 3.382 | 2.351 | 0.916 | |
| 30 | 0.080 | 3.165 | 3.211 | 0.849 | |
| 60 | 0.075 | 3.157 | 4.820 | 0.835 | |
| Ketoprofen | 1 | 0.100 | 2.379 | 2.805 | 1.033 |
| 7 | 0.090 | 1.887 | 3.747 | 1.441 | |
| 30 | 0.095 | 1.950 | 3.977 | 1.594 | |
| 60 | 0.099 | 1.946 | 4.386 | 2.360 | |
| Diclofenac | 1 | 0.094 | 2.764 | 4.063 | 0.722 |
| 7 | 0.079 | 2.113 | 5.825 | 0.564 | |
| 30 | 0.085 | 1.604 | 6.740 | 0.555 | |
| 60 | 0.087 | 1.359 | 9.143 | 0.499 |
| NSAIDs | Day | AWCD | S | H | E |
|---|---|---|---|---|---|
| Control | 1 | 0.26 ± 0.011 | 16.67 ± 0.577 | 3.20 ± 0.027 | 1.14 ± 0.006 |
| 7 | 0.24 ± 0.007 | 13.33 ± 1.155 | 3.04 ± 0.016 | 1.17 ± 0.035 | |
| 30 | 0.30 ± 0.002 | 17 ± 0.00 | 3.21 ± 0.006 | 1.13 ± 0.002 | |
| 60 | 0.32 ± 0.003 | 20.33 ± 1.155 | 3.21 ± 0.004 | 1.07 ± 0.022 | |
| Ibuprofen | 1 | 0.19 ± 0.003 | 9 ± 0.00 | 3.03 ± 0.02 | 1.38 ± 0.009 |
| 7 | 0.25 ± 0.006 | 11.33 ± 0.577 | 3.16 ± 0.061 | 1.30 ± 0.011 | |
| 30 | 0.27 ± 0.005 | 13.00 ± 1.732 | 3.22 ± 0.012 | 1.26 ± 0.057 | |
| 60 | 0.26 ± 0.002 | 12.00 ± 0.00 | 3.20 ± 0.002 | 1.29 ± 0.001 | |
| Ketoprofen | 1 | 0.24 ± 0.003 | 14.67 ± 0.577 | 3.15 ± 0.008 | 1.17 ± 0.019 |
| 7 | 0.25 ± 0.004 | 15.0 ± 0.00 | 3.13 ± 0.026 | 1.15 ±0.010 | |
| 30 | 0.24 ± 0.002 | 13.0 ± 0.00 | 3.16 ± 0.003 | 1.23 ± 0.001 | |
| 60 | 0.23 ± 0.001 | 11.33 ± 0.577 | 3.12 ± 0.003 | 1.29 ± 0.025 | |
| Diclofenac | 1 | 0.25 ± 0.009 | 13.0 ± 0.00 | 3.14 ± 0.020 | 1.23 ± 0.072 |
| 7 | 0.27 ± 0.006 | 13.67 ± 0.577 | 3.04 ± 0.108 | 1.16 ± 0.037 | |
| 30 | 0.28 ± 0.003 | 15.33 ± 1.155 | 3.13 ± 0.010 | 1.15 ± 0.029 | |
| 60 | 0.27 ± 0.006 | 15.67 ± 1.155 | 3.10 ± 0.020 | 1.13 ± 0.025 |
| Volatile Organic Compounds | Control | Ketoprofen | Ibuprofen | Diclofenac |
|---|---|---|---|---|
| Alcohol | 12 | 13 | 18 | 12 |
| Aromatic compounds | 11 | 11 | 15 | 11 |
| Ketone | 13 | 14 | 11 | 12 |
| Terpene | 18 | 18 | 14 | 12 |
| Organic acids | 12 | 12 | 9 | 15 |
| Aldehyde | 6 | 8 | 6 | 8 |
| Sulphur compounds | 2 | 1 | 6 | 5 |
| Ester | 4 | 4 | 5 | 5 |
| Alkane | 12 | 11 | 10 | 11 |
| Nitrogen compounds | 5 | 5 | 4 | 3 |
| Alkene | 4 | 3 | 2 | 6 |
| Furans | 1 | 0 | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovacs, E.D.; Silaghi-Dumitrescu, L.; Roman, C.; Tian, D. Structural and Metabolic Profiling of Lycopersicon esculentum Rhizosphere Microbiota Artificially Exposed at Commonly Used Non-Steroidal Anti-Inflammatory Drugs. Microorganisms 2022, 10, 254. https://doi.org/10.3390/microorganisms10020254
Kovacs ED, Silaghi-Dumitrescu L, Roman C, Tian D. Structural and Metabolic Profiling of Lycopersicon esculentum Rhizosphere Microbiota Artificially Exposed at Commonly Used Non-Steroidal Anti-Inflammatory Drugs. Microorganisms. 2022; 10(2):254. https://doi.org/10.3390/microorganisms10020254
Chicago/Turabian StyleKovacs, Emoke Dalma, Luminita Silaghi-Dumitrescu, Cecilia Roman, and Di Tian. 2022. "Structural and Metabolic Profiling of Lycopersicon esculentum Rhizosphere Microbiota Artificially Exposed at Commonly Used Non-Steroidal Anti-Inflammatory Drugs" Microorganisms 10, no. 2: 254. https://doi.org/10.3390/microorganisms10020254
APA StyleKovacs, E. D., Silaghi-Dumitrescu, L., Roman, C., & Tian, D. (2022). Structural and Metabolic Profiling of Lycopersicon esculentum Rhizosphere Microbiota Artificially Exposed at Commonly Used Non-Steroidal Anti-Inflammatory Drugs. Microorganisms, 10(2), 254. https://doi.org/10.3390/microorganisms10020254







