Effects of Mustelid gammaherpesvirus 1 (MusGHV-1) Reactivation in European Badger (Meles meles) Genital Tracts on Reproductive Fitness
Abstract
:1. Introduction
- Patterns of MusGHV-1 reactivation in badger genital tracts by comparing prevalence among females culled at the start of the primary mating season with those culled at the end (where many females are pregnant at the beginning, but all will have given birth by the end);
- Differences in MusGHV-1 reactivation between males and females, and between different age groups;
- Effects of MusGHV-1 reactivation on female reproductive success, male testes weight, and sperm abundance;
- The potential for vertical transmission of MusGHV-1 to the foetus via the placenta during gestation, or via the maternal vaginal tract during parturition; and
- The potential for horizontal transmission through semen.
2. Results
2.1. Risk Factors of MusGHV-1 Reactivation in Badgers
2.2. Interactions of Genital MusGHV-1 Reactivation with Body Weight
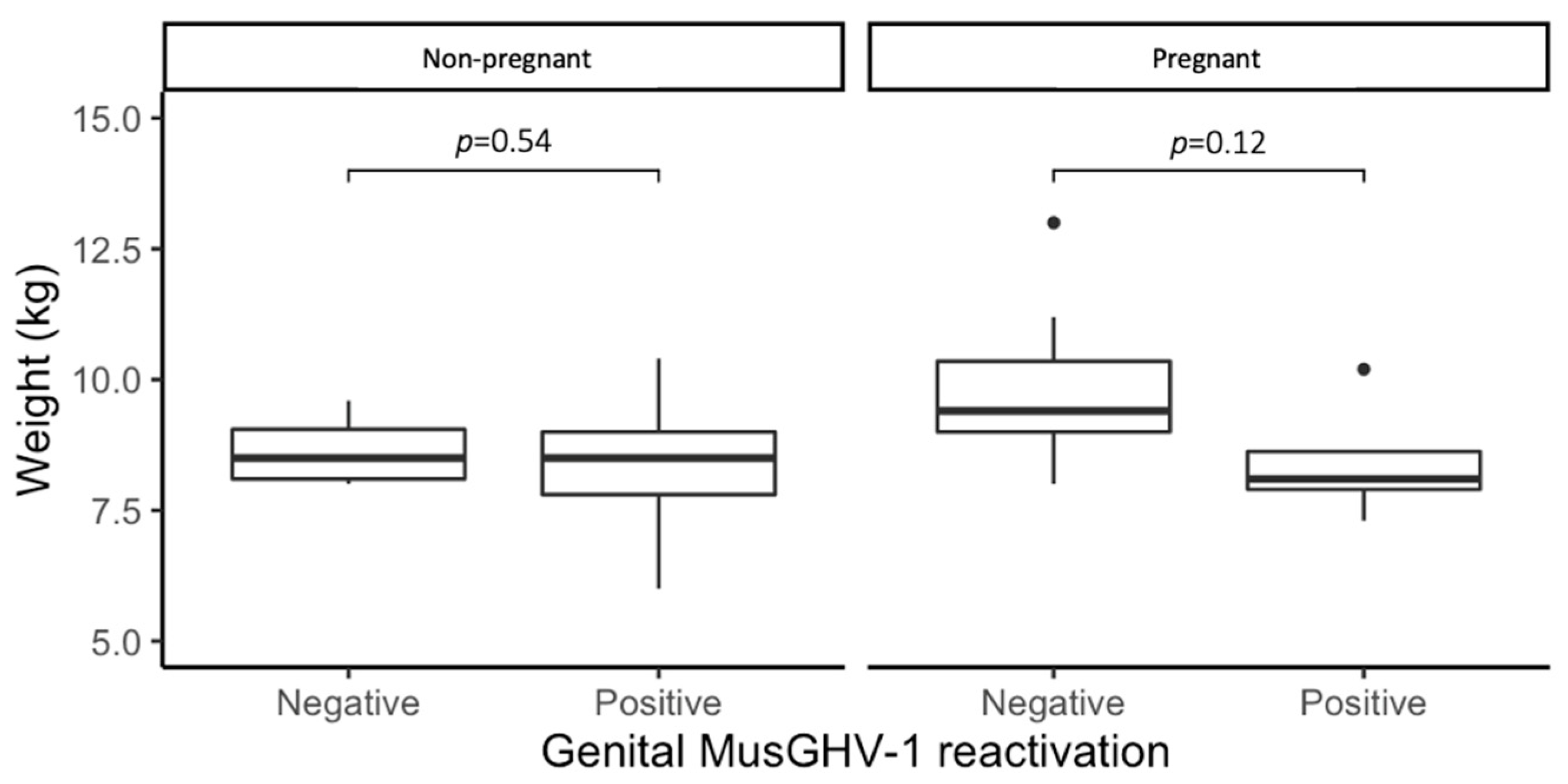
2.3. Effects of Genital MusGHV-1 Reactivation on Reproductive Fitness
2.4. Potential for Horizontal and Vertical Transmission
2.5. Abnormalities in Female Reproductive Tracts Associated with Genital MusGHV
3. Discussion
3.1. Sex-Specific Seasonal Patterns in Genital MusGHV-1 Reactivation
3.2. Effects of Genital MusGHV-1 Reactivation on Reproductive Fitness
3.3. Genital Lesions
3.4. Vertical Transmission of MusGHV-1
3.5. The Significance of Gammaherpesviruses as Drivers of Wildlife Population Dynamics
4. Materials and Methods
4.1. Source of Post-Mortem Badgers
4.2. Assessment of Reproductive Fitness
4.3. Assessing Potential for Vertical and Horizontal Transmission of MusGHV-1
4.4. Histopathological Examination
4.5. MusGHV-1 DNA Detection in Genital Tracts, Semen, and Tissues
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davison, A.J. Herpesvirus systematics. Vet. Microbiol. 2010, 143, 52–69. [Google Scholar] [CrossRef] [Green Version]
- Pellett, P.E. Trunkloads of Viruses. J. Virol. 2014, 88, 13520–13522. [Google Scholar] [CrossRef] [Green Version]
- Azab, W.; Dayaram, A.; Greenwood, A.D.; Osterrieder, N. How Host Specific Are Herpesviruses? Lessons from Herpesviruses Infecting Wild and Endangered Mammals. Annu. Rev. Virol. 2018, 5, 53–68. [Google Scholar] [CrossRef] [PubMed]
- Barton, E.; Mandal, P.; Speck, S.H. Pathogenesis and Host Control of Gammaherpesviruses: Lessons from the Mouse. Annu. Rev. Immunol. 2011, 29, 351–397. [Google Scholar] [CrossRef] [PubMed]
- Grinde, B. Herpesviruses: Latency and reactivation – viral strategies and host response. J. Oral Microbiol. 2013, 5, 22766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chew, T.; Taylor, K.E.; Mossman, K.L. Innate and adaptive immune responses to herpes simplex virus. Viruses 2009, 1, 979–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glaser, R.; Pearl, D.K.; Kiecolt-Glaser, J.K.; Malarkey, W.B. Plasma cortisol levels and reactivation of latent Epstein-Barr virus in response to examination stress. Psychoneuroendocrinology 1994, 19, 765–772. [Google Scholar] [CrossRef]
- Stowe, R.P.; Pierson, D.L.; Barrett, A.D.T. Elevated stress hormone levels relate to Epstein-Barr virus reactivation in astronauts. Psychosom. Med. 2001, 63, 891–895. [Google Scholar] [CrossRef] [Green Version]
- Wilson, A.C.; Mohr, I. A cultured affair: HSV latency and reactivation in neurons. Trends Microbiol. 2012, 20, 604–611. [Google Scholar] [CrossRef] [Green Version]
- Davis, D.A.; Rinderknecht, A.S.; Paul Zoeteweij, J.; Aoki, Y.; Read-Connole, E.L.; Tosato, G.; Blauvelt, A.; Yarchoan, R. Hypoxia induces lytic replication of Kaposi sarcoma-associated herpesvirus. Blood 2001, 97, 3244–3250. [Google Scholar] [CrossRef] [Green Version]
- Odumade, O.A.; Hogquist, K.A.; Balfour, H.H. Progress and problems in understanding and managing primary epstein-barr virus infections. Clin. Microbiol. Rev. 2011, 24, 193–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haeri, S.; Baker, A.M.; Boggess, K.A. Prevalence of Epstein-Barr virus reactivation in pregnancy. Am. J. Perinatol. 2010, 27, 715–719. [Google Scholar] [CrossRef] [PubMed]
- François, S.; Vidick, S.; Sarlet, M.; Desmecht, D.; Drion, P.; Stevenson, P.G.; Vanderplasschen, A.; Gillet, L. Illumination of Murine Gammaherpesvirus-68 Cycle Reveals a Sexual Transmission Route from Females to Males in Laboratory Mice. PLoS Pathog. 2013, 9, e1003292. [Google Scholar] [CrossRef] [PubMed]
- Stowe, R.P.; Kozlova, E.V.; Yetman, D.L.; Walling, D.M.; Goodwin, J.S.; Glaser, R. Chronic herpesvirus reactivation occurs in aging. Exp. Gerontol. 2007, 42, 563–570. [Google Scholar] [CrossRef] [Green Version]
- Smith, K.C. Herpesviral abortion in domestic animals. Vet. J. 1997, 153, 253–268. [Google Scholar] [CrossRef]
- Avgil, M.; Ornoy, A. Herpes simplex virus and Epstein-Barr virus infections in pregnancy: Consequences of neonatal or intrauterine infection. Reprod. Toxicol. 2006, 21, 436–445. [Google Scholar] [CrossRef]
- Evermann, J.F.; Ledbetter, E.C.; Maes, R.K. Canine Reproductive, Respiratory, and Ocular Diseases due to Canine Herpesvirus. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 1097–1120. [Google Scholar] [CrossRef]
- Crook, T.; Benavides, J.; Russell, G.; Gilray, J.; Maley, M.; Willoughby, K. Bovine herpesvirus 1 abortion: Current prevalence in the United Kingdom and evidence of hematogenous spread within the fetus in natural cases. J. Vet. Diagn. Investig. 2012, 24, 662–670. [Google Scholar] [CrossRef] [Green Version]
- Patel, J.R.; Heldens, J. Equine herpesviruses 1 (EHV-1) and 4 (EHV-4)—Epidemiology, disease and immunoprophylaxis: A brief review. Vet. J. 2005, 170, 14–23. [Google Scholar] [CrossRef]
- das Neves, C.G.; Mørk, T.; Thiry, J.; Godfroid, J.; Rimstad, E.; Thiry, E.; Tryland, M. Cervid herpesvirus 2 experimentally reactivated in reindeer can produce generalized viremia and abortion. Virus Res. 2009, 145, 321–328. [Google Scholar] [CrossRef]
- Racicot, K.; Cardenas, I.; Wünsche, V.; Aldo, P.; Guller, S.; Means, R.E.; Romero, R.; Mor, G. Viral Infection of the Pregnant Cervix Predisposes to Ascending Bacterial Infection. J. Immunol. 2013, 191, 934–941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mor, G.; Aldo, P.; Alvero, A.B. The unique immunological and microbial aspects of pregnancy. Nat. Rev. Immunol. 2017, 17, 469–482. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, I.; Means, R.E.; Aldo, P.; Koga, K.; Lang, S.M.; Booth, C.; Manzur, A.; Oyarzun, E.; Romero, R.; Mor, G. Viral Infection of the Placenta Leads to Fetal Inflammation and Sensitization to Bacterial Products Predisposing to Preterm Labor. J. Immunol. 2010, 185, 1248–1257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chastant-Maillard, S. Impact of bovine herpesvirus 4 (BoHV-4) on reproduction. Transbound. Emerg. Dis. 2015, 62, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Marenzoni, M.L.; Sforna, M.; Stefanetti, V.; Casagrande Proietti, P.; Brignone, L.; Del Sero, A.; Falcioni, F.; Orvieto, S.; Tamantini, C.; Tiburzi, A.; et al. Detection of Equid herpesvirus type 2 and 5 DNA in uterine flushings of mares with reproductive disorders. Vet. Microbiol. 2014, 174, 570–576. [Google Scholar] [CrossRef]
- Gagnon, C.A.; Tremblay, J.; Larochelle, D.; Music, N.; Tremblay, D. Identification of a novel herpesvirus associated with cutaneous ulcers in a fisher (Martes pennanti). J. Vet. Diagn. Investig. 2011, 23, 986–990. [Google Scholar] [CrossRef] [Green Version]
- Tseng, M.; Fleetwood, M.; Reed, A.; Gill, V.A.; Harris, R.K.; Moeller, R.B.; Lipscomb, T.P.; Mazet, J.A.K.; Goldstein, T. Mustelid Herpesvirus-2, a Novel Herpes Infection in Northern Sea Otters (Enhydra Lutris Kenyoni). J. Wildl. Dis. 2013, 48, 181–185. [Google Scholar] [CrossRef] [Green Version]
- Lozano, C.C.; Sweanor, L.L.; Wilson-Henjum, G.; Kays, R.W.; Moreno, R.; VandeWoude, S.; Troyer, R.M. Identification of novel gammaherpesviruses in ocelots (Leopardus pardalis) and Bobcats (Lynx rufus) in Panama and Colorado, USA. J. Wildl. Dis. 2015, 51, 911–915. [Google Scholar] [CrossRef]
- Hendrikse, L.D.; Kambli, A.; Kayko, C.; Canuti, M.; Rodrigues, B.; Stevens, B.; Vashon, J.; Lang, A.S.; Needle, D.B.; Troyer, R.M. Identification of a novel gammaherpesvirus in Canada lynx (Lynx canadensis). Viruses 2019, 11, 363. [Google Scholar] [CrossRef] [Green Version]
- Subudhi, S.; Rapin, N.; Dorville, N.; Hill, J.E.; Town, J.; Willis, C.K.R.; Bollinger, T.K.; Misra, V. Isolation, characterization and prevalence of a novel Gammaherpesvirus in Eptesicus fuscus, the North American big brown bat. Virology 2018, 516, 227–238. [Google Scholar] [CrossRef]
- Cabello, J.; Esperón, F.; Napolitano, C.; Hidalgo, E.; Dávila, J.A.; Millán, J. Molecular identification of a novel gammaherpesvirus in the endangered Darwin’s fox (Lycalopex fulvipes). J. Gen. Virol. 2013, 94, 2745–2749. [Google Scholar] [CrossRef]
- Black, W.; Troyer, R.M.; Coutu, J.; Wong, K.; Wolff, P.; Gilbert, M.; Yuan, J.; Wise, A.G.; Wang, S.; Xu, D.; et al. Identification of gammaherpesvirus infection in free-ranging black bears (Ursus americanus). Virus Res. 2019, 259, 46–53. [Google Scholar] [CrossRef]
- Makundi, I.; Koshida, Y.; Endo, Y.; Nishigaki, K. Identification of felis catus gammaherpesvirus 1 in Tsushima leopard cats (Prionailurus bengalensis euptilurus) on Tsushima Island, Japan. Viruses 2018, 10, 378. [Google Scholar] [CrossRef] [Green Version]
- Dugdale, H.L.; Macdonald, D.W.; Pope, L.C.; Burke, T. Polygynandry, extra-group paternity and multiple-paternity litters in European badger (Meles meles) social groups. Mol. Ecol. 2007, 16, 5294–5306. [Google Scholar] [CrossRef] [PubMed]
- Kokko, H.; Ranta, E.; Ruxton, G.; Lundberg, P. Sexually transmitted disease and the evolution of host mating systems. Evolution 2002, 56, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.D.P.; Jetz, W.; Macdonald, D.W. Environmental correlates of badger social spacing across Europe. J. Biogeogr. 2002, 29, 411–425. [Google Scholar] [CrossRef] [Green Version]
- Corner, L.A.L.; Stuart, L.J.; Kelly, D.J.; Marples, N.M. Reproductive biology including evidence for superfetation in the European badger Meles meles (Carnivora: Mustelidae). PLoS ONE 2015, 10, e0138093. [Google Scholar] [CrossRef]
- Annavi, G.; Newman, C.; Dugdale, H.L.; Buesching, C.D.; Sin, Y.W.; Burke, T.; Macdonald, D.W. Neighbouring-group composition and within-group relatedness drive extra-group paternity rate in the European badger (Meles meles). J. Evol. Biol. 2014, 27, 2191–2203. [Google Scholar] [CrossRef] [Green Version]
- Rosen, L.E.; Fogarty, U.; O’Keeffe, J.J.; Olea-Popelka, F.J. Monitoring European badger (Meles meles) reproduction under evolving bovine tuberculosis management in Ireland. Eur. J. Wildl. Res. 2019, 65, 97. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Dugdale, H.L.; Macdonald, D.W. Female Receptivity, Embryonic Diapause, and Superfetation in the European Badger (Meles meles): Implications for the Reproductive Tactics of Males and Females Review by: Nobuyuki Yamaguchi, Hannah L Dugdale, and David W Macdonald The Quarterly Revi. Q. Rev. Biol. 2006, 81, 33–48. [Google Scholar] [CrossRef] [Green Version]
- Page, R.J.C.; Ross, J.; Langton, S.D. Seasonality of reproduction in the European badger Meles meles in south-west England. J. Zool. 1994, 233, 69–91. [Google Scholar] [CrossRef]
- Roper, T.J. Badger (Collins New Naturalist Library, Book 114); HarperCollins UK: London, UK, 2010. [Google Scholar]
- Carpenter, P.J.; Pope, L.C.; Greig, C.; Dawson, D.A.; Rogers, L.M.; Erven, K.; Wilson, G.J.; Delahay, R.J.; Cheeseman, C.L.; Burke, T. Mating system of the Eurasian badger, Meles meles, in a high density population. Mol. Ecol. 2005, 14, 273–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bright Ross, J.G.; Newman, C.; Buesching, C.D.; Macdonald, D.W. What lies beneath? Population dynamics conceal pace-of-life and sex ratio variation, with implications for resilience to environmental change. Glob. Chang. Biol. 2020, 26, 3307–3324. [Google Scholar] [CrossRef]
- Newman, C.; Macdonald, D.W.; Anwar, M.A. Coccidiosis in the European badger, Meles meles in Wytham Woods: Infection and consequences for growth and survival. Parasitology 2001, 123, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Apari, P.; de Sousa, J.D.; Müller, V. Why Sexually Transmitted Infections Tend to Cause Infertility: An Evolutionary Hypothesis. PLoS Pathog. 2014, 10, e1004111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kent, A.; Ehlers, B.; Mendum, T.; Newman, C.; Macdonald, D.W.; Chambers, M.; Buesching, C.D. Genital tract screening finds widespread infection with mustelid gammaherpesvirus 1 in the European badger (Meles meles). J. Wildl. Dis. 2017, 54, 133–137. [Google Scholar] [CrossRef] [Green Version]
- Banks, M.; King, D.P.; Daniells, C.; Stagg, D.A.; Gavier-Widen, D. Partial characterization of a novel gammaherpesvirus isolated from a European badger (Meles meles). J. Gen. Virol. 2002, 83, 1325–1330. [Google Scholar] [CrossRef]
- Sin, Y.W.; Annavi, G.; Dugdale, H.L.; Newman, C.; Burke, T.; MacDonald, D.W. Pathogen burden, co-infection and major histocompatibility complex variability in the European badger (Meles meles). Mol. Ecol. 2014, 23, 5072–5088. [Google Scholar] [CrossRef]
- King, D.P.; Mutukwa, N.; Lesellier, S.; Cheeseman, C.; Chambers, M.A.; Banks, M. Detection of Mustelid Herpesvirus-1 Infected European Badgers (Meles meles) in the British Isles. J. Wildl. Dis. 2004, 40, 99–102. [Google Scholar] [CrossRef]
- Young, L.S.; Yap, L.F.; Murray, P.G. Epstein-Barr virus: More than 50 years old and still providing surprises. Nat. Rev. Cancer 2016, 16, 789–802. [Google Scholar] [CrossRef]
- Van Cleemput, J.; Poelaert, K.C.K.; Laval, K.; Nauwynck, H.J. Unravelling the first key steps in equine herpesvirus type 5 (EHV5) pathogenesis using ex vivo and in vitro equine models. Vet. Res. 2019, 50, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, B.; Xie, J.; Van Cleemput, J.; Wei, R.; Opsomer, G.; Nauwynck, H.J. Gammaherpesvirus BoHV-4 infects bovine respiratory epithelial cells mainly at the basolateral side. Vet. Res. 2019, 50, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hjalgrim, H.; Friborg, J.; Melbye, M. The Epidemiology of EBV and its Association with Malignant Disease. In Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis; Cambridge University Press: Cambridge, MA, USA, 2007; pp. 929–959. [Google Scholar]
- Craig, M.I.; Barrandeguy, M.E.; Fernández, F.M. Equine herpesvirus 2 (EHV-2) infection in thoroughbred horses in Argentina. BMC Vet. Res. 2005, 1, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.K.; Lee, I. The Molecular Detection of Equine Herpesviruses 2 and 5 in Genital Swabs from Clinically Normal Thoroughbred Mares in South Korea. J. Equine Vet. Sci. 2019, 79, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Pascottini, O.B.; Xie, J.; Desmarets, L.; Cui, T.; Opsomer, G.; Nauwynck, H.J. Presence of gammaherpesvirus BoHV-4 in endometrial cytology samples is not associated with subclinical endometritis diagnosed at artificial insemination in dairy cows. Vet. Microbiol. 2019, 229, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, D.P.; Damania, B. Kaposi sarcoma associated herpesvirus pathogenesis (KSHV)—An update. Curr. Opin. Virol. 2013, 3, 238–244. [Google Scholar] [CrossRef] [Green Version]
- Kangro, H.O.; Osman, H.K.; Lau, Y.L.; Heath, R.B.; Yeung, C.Y.; Ng, M.H. Seroprevalence of antibodies to human herpesviruses in England and Hong Kong. J. Med. Virol. 1994, 43, 91–96. [Google Scholar] [CrossRef]
- Longnecker, R.M.; Kieff, E.; Cohen, J.I. Epstein-Barr Virus. In Fields Virology: Sixth Edition; Wolters Kluwer Health Adis (ESP): Alphen aan den Rijn, The Netherlands, 2013; pp. 1898–1959. [Google Scholar]
- Bandyopadhyay, S.; Das, S.; Baruah, K.K.; Chakravarty, P.; Chakrabarty, D.; Sarkar, T.; Pal, B.; De, S.; Pan, D.; Bera, A.K.; et al. Detection of bovine herpesvirus 1 sequences in yaks (Bos grunniens) with keratoconjunctivitis, using a highly sensitive nested polymerase chain reaction. OIE Rev. Sci. Tech. 2010, 29, 695–703. [Google Scholar] [CrossRef] [Green Version]
- Cavignac, Y.; Esclatine, A. Herpesviruses and autophagy: Catch me if you can! Viruses 2010, 2, 314–333. [Google Scholar] [CrossRef]
- Lockhart, A.B.; Thrall, P.H.; Antonovics, J. Sexually transmitted diseases in animals: Ecological and evolutionary implications. Biol. Rev. 1996, 71, 415–471. [Google Scholar] [CrossRef]
- Uchii, K.; Minamoto, T.; Honjo, M.N.; Kawabata, Z. Seasonal reactivation enables Cyprinid herpesvirus 3 to persist in a wild host population. FEMS Microbiol. Ecol. 2014, 87, 536–542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sugianto, N.A.; Heistermann, M.; Newman, C.; Macdonald, D.W.; Buesching, C.D. Density-dependent reproductive strategies provide a flexible mechanism for assuring mating success in the European badger (Meles meles): Evidence from hormonal measures. PLoS ONE 2019, 14, e0203910. [Google Scholar] [CrossRef] [Green Version]
- Charlton, B.D.; Newman, C.; Macdonald, D.W.; Buesching, C.D. Male European badger churrs: Insights into call function and motivational basis. Mamm. Biol. 2020, 100, 429–438. [Google Scholar] [CrossRef]
- Sugianto, N.A.; Buesching, C.D.; Heistermann, M.; Newman, C.; Macdonald, D.W. Linking plasma sex steroid hormone levels to the condition of external genitalia in European badgers (Meles meles): A critical evaluation of traditional field methodology. Mamm. Biol. 2018, 93, 97–108. [Google Scholar] [CrossRef]
- Trumble, B.C.; Blackwell, A.D.; Stieglitz, J.; Thompson, M.E.; Suarez, I.M.; Kaplan, H.; Gurven, M. Associations between male testosterone and immune function in a pathogenically stressed forager-horticultural population. Am. J. Phys. Anthropol. 2016, 161, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Troyer, R.M.; Beatty, J.A.; Stutzman-Rodriguez, K.R.; Carver, S.; Lozano, C.C.; Lee, J.S.; Lappin, M.R.; Riley, S.P.D.; Serieys, L.E.K.; Logan, K.A.; et al. Novel Gammaherpesviruses in North American Domestic Cats, Bobcats, and Pumas: Identification, Prevalence, and Risk Factors. J. Virol. 2014, 88, 3914–3924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLuckie, A.; Tasker, S.; Dhand, N.K.; Spencer, S.; Beatty, J.A. High prevalence of Felis catus gammaherpesvirus 1 infection in haemoplasma-infected cats supports co-transmission. Vet. J. 2016, 214, 117–121. [Google Scholar] [CrossRef] [Green Version]
- Woodroffe, R.; Macdonald, D.W. Costs of breeding status in the European badger, Meles meles. J. Zool. 1995, 235, 237–245. [Google Scholar] [CrossRef]
- Macdonald, D.W.; Harmsen, B.J.; Johnson, P.J.; Newman, C. Increasing frequency of bite wounds with increasing population density in Eurasian badgers, Meles meles. Anim. Behav. 2004, 67, 745–751. [Google Scholar] [CrossRef]
- Barrandeguy, M.; Vissani, A.; Olguin, C.; Barbara, G.; Valenzuela, H.; Becerra, L.; Tordoya, M.; Miño, S.; Thiry, E. Experimental infection with equid herpesvirus 3 in seronegative and seropositive mares. Vet. Microbiol. 2012, 160, 319–326. [Google Scholar] [CrossRef]
- Kaspersen, M.D.; Höllsberg, P. Seminal shedding of human herpesviruses. Virol. J. 2013, 10, 226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carr, B.R.; Parker, C.R.; Madden, J.D.; MacDonald, P.C.; Porter, J.C. Maternal plasma adrenocorticotropin and cortisol relationships throughout human pregnancy. Am. J. Obstet. Gynecol. 1981, 139, 416–422. [Google Scholar] [CrossRef]
- Egyed, L.; Sassi, G.; Tibold, J.; Mádl, I.; Szenci, O. Symptomless intrauterine transmission of bovine herpesvirus 4 to bovine fetuses. Microb. Pathog. 2011, 50, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Becher, N.; Waldorf, K.A.; Hein, M.; Uldbjerg, N. The cervical mucus plug: Structured review of the literature. Acta Obstet. Gynecol. Scand. 2009, 88, 502–513. [Google Scholar] [CrossRef] [PubMed]
- Loux, S.C.; Scoggin, K.E.; Troedsson, M.H.T.; Squires, E.L.; Ball, B.A. Characterization of the cervical mucus plug in mares. Reproduction 2017, 153, 197–210. [Google Scholar] [CrossRef] [PubMed]
- Racicot, K.; Mor, G. Risks associated with viral infections during pregnancy. J. Clin. Investig. 2017, 127, 1591–1599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Decaro, N.; Martella, V.; Buonavoglia, C. Canine Adenoviruses and Herpesvirus. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 799–814. [Google Scholar] [CrossRef]
- Bryans, J.T. Herpesviral diseases affecting reproduction in the horse. Vet. Clin. N. Am. Large Anim. Pract. 1980, 2, 303–312. [Google Scholar] [CrossRef]
- Blanchard, T.L.; Kenney, R.M.; Timoney, P.J. Venereal disease. Vet. Clin. N. Am. Equine Pract. 1992, 8, 191–203. [Google Scholar] [CrossRef]
- Josey, W.E.; Zaki, S.A. Relation of Cytohistopathology of Genital Herpesvirus Infection to Cervical Anaplasia. Cancer Res. 1973, 33, 1452–1463. [Google Scholar]
- Beatty, J.A.; Sharp, C.R.; Duprex, W.P.; Munday, J.S. Novel feline viruses: Emerging significance of gammaherpesvirus and morbillivirus infections. J. Feline Med. Surg. 2019, 21, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Hachkuga, T. Detection of Epstein-Barr Virus DNA from a Lymphoma-like Lesion of the Uterine Cervix. Gynecol. Oncol. 1992, 73, 69–73. [Google Scholar] [CrossRef]
- Lehtinen, M.; Koskela, P.; Ögmundsdottir, H.M.; Bloigu, A.; Dillner, J.; Gudnadottir, M.; Hakulinen, T.; Kjartansdottir, A.; Kvarnung, M.; Pukkala, E.; et al. Maternal herpesvirus infections and risk of acute lymphoblastic leukemia in the offspring. Am. J. Epidemiol. 2003, 158, 207–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abade dos Santos, F.A.; Monteiro, M.; Pinto, A.; Carvalho, C.L.; Peleteiro, M.C.; Carvalho, P.; Mendonça, P.; Carvalho, T.; Duarte, M.D. First description of a herpesvirus infection in genus Lepus. BioRxiv Preprint 2020, 15, e0231795. [Google Scholar] [CrossRef]
- De Castro, F.; Bolker, B. Mechanisms of disease-induced extinction. Ecol. Lett. 2005, 8, 117–126. [Google Scholar] [CrossRef]
- Ryder, J.J.; Miller, M.R.; White, A.; Knell, R.J.; Boots, M. Host-parasite population dynamics under combined frequency- and density-dependent transmission. Oikos 2007, 116, 2017–2026. [Google Scholar] [CrossRef]
- McCallum, H. Tasmanian devil facial tumour disease: Lessons for conservation biology. Trends Ecol. Evol. 2008, 23, 631–637. [Google Scholar] [CrossRef]
- McCallum, H.; Kerlin, D.H.; Ellis, W.; Carrick, F. Assessing the significance of endemic disease in conservation—Koalas, chlamydia, and koala retrovirus as a case study. Conserv. Lett. 2018, 11, e124225. [Google Scholar] [CrossRef]
- O’Keeffe, J.J. Description of a medium term national strategy toward eradication of Tuberculosis in cattle in Ireland. Proc. Vet. Epidemiol. Econ. 2006, 11, 1–7. [Google Scholar]
- Hancox, M. Field age determination in the european badger. Rev. Ecol. 1988, 43, 399–404. [Google Scholar]
- Fournier-Chambrillon, C.; Bifolchi, A.; Mazzola-Rossi, E.; Sourice, S.; Albaret, M.; Bray, Y.; Ceña, J.-C.; Maya, F.U.; Agraffel, T.; Fournier, P. Reliability of stained placental scar counts in farmed American mink and application to free-ranging mustelids. J. Mammal. 2010, 91, 818–826. [Google Scholar] [CrossRef] [Green Version]
- Scott-Wilson, J. Laboratory Manual for the Examination of Human Semen and Semen-Cervical Mucus Interaction. J. R. Soc. Med. 1982, 75, 58. [Google Scholar]
- Wilson, E.B. Probable Inference, the Law of Succession, and Statistical Inference. J. Am. Stat. Assoc. 1927, 22, 209. [Google Scholar] [CrossRef]
- Zweig, M.H.; Campbell, G. Receiver-operating characteristic (ROC) plots: A fundamental evaluation tool in clinical medicine. Clin. Chem. 1993, 39, 561–577. [Google Scholar] [CrossRef]
- Statistics Kingdom Sample Size Calculator. Statistics Kingdom. Available online: http://www.statskingdom.com/sample_size_regression.html (accessed on 15 August 2020).



| Group | Estimate | Standard Error | z Value | Adjusted OR | 95% CI of OR | p Value |
|---|---|---|---|---|---|---|
| (Intercept) | −1.1697 | 0.6466 | −1.809 | 0.31 | 0.09–1.1 | 0.07 |
| Sex (vs. Female) | ||||||
| Male | 1.9023 | 0.6442 | 2.953 | 6.7 | 1.9–23.67 | 0.003 |
| Age group | ||||||
| Young adults | 1.5527 | 0.7243 | 2.144 | 4.72 | 1.14–19.53 | 0.032 |
| Old adults | 1.3507 | 0.7671 | 1.761 | 3.86 | 0.86–17.36 | 0.078 |
| Positive n | Total n | Prevalence | 95% CI | Chi-Square Test | |
|---|---|---|---|---|---|
| Season | |||||
| Winter | 52 | 80 | 65% | 54.1–74.5% | X2 = 0.503, df = 1, p = 0.478 |
| Spring | 37 | 64 | 57.80% | 45.6–69.1% | |
| Sex | |||||
| Male | 53 | 71 | 74.60% | 63.4–83.3% | X2 = 8.741, df = 1, p = 0.003 |
| Female | 36 | 73 | 49.30% | 38.2–60.5% | |
| Age group a,b | |||||
| Yearling | 2 | 2 | 100% | 34.2–100% | X2 = 2.559, df = 1, p = 0.11 (Young:Middle aged) |
| Young | 21 | 29 | 72.40% | 54.3–85.3% | X2 = 0.034, df = 1, p = 0.854 (Young:Old) |
| Middle aged | 10 | 20 | 50% | 29.9–70.1% | X2 = 1.667, df = 1, p = 0.197 (Middle aged:Old) |
| Old | 14 | 20 | 70% | 48.1–85.4% | |
| Season:Sex | |||||
| Winter:Male | 33 | 40 | 82.50% | 68.1–91.3% | X2 = 9.2857, df = 1, p = 0.002 |
| Winter:Female | 19 | 40 | 47.50% | 32.9–62.5% | |
| Spring:Male | 20 | 31 | 64.50% | 46.9–78.9% | X2 = 0.6388, df = 1, p = 0.424 |
| Spring:Female | 17 | 33 | 51.50% | 35.2–67.5% | |
| Sex:Age group a,b | |||||
| Male:Yearling | 1 | 1 | 100% | 20.7–100% | p = 0.59 (Young:Middle aged) |
| Male:Young | 11 | 12 | 91.60% | 64.6–98.5% | p = 0.553 (Young:Old) |
| Male:Middle aged | 9 | 12 | 75% | 46.8–91.1% | p = 1 (Middle aged:Old) |
| Male:Old | 7 | 9 | 77.80% | 45.3–93.7% | |
| Female:Yearling | 1 | 1 | 100% | 20.7–100% | p = 0.042 (Young:Middle aged) |
| Female:Young | 10 | 17 | 58.80% | 36–78.4% | p = 1 (Young:Aged) |
| Female:Middle aged | 1 | 8 | 12.50% | 2.2–47.1% | p = 0.059 (Middle age:Old) |
| Female:Old | 7 | 11 | 63.60% | 35.4–84.8% |
| Reproductive Status | Spring | Winter | Young | Middle Aged | Old | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (+) | Total | % | 95% CI | (+) | Total | % | 95% CI | (+) | (-) | (+) | (-) | (+) | (-) | |
| Not pregnant | 11 | 17 | 64.7% | 41.3–82.7% | 8 | 12 | 66.7% | 39.1–86.2% | 6 | 1 | 1 | 3 | - | - |
| Pregnant a | - | - | - | - | 4 | 18 | 22.2% | 9.0–45.2% | 0 | 5 | 0 | 3 | 4 | 3 |
| Recently pregnant | 7 | 16 | 43.8% | 23.1–66.8% | 3 | 6 | 50.0% | 18.8–81.2% | 3 | 1 | 0 | 1 | 0 | 1 |
| Recently pregnant, aborted | - | - | - | - | 4 | 4 | 100.0% | 51.0–100% | 1 | 0 | - | - | 3 | 0 |
| Pregnant n a | Total n b | Rate | 95% CI | Chi-Square Test | |
|---|---|---|---|---|---|
| Season | |||||
| Winter | 24 | 39 | 61.50% | 45.9–75.1% | X2 = 0.3486, df = 1, p = 0.555 |
| Spring | 16 | 31 | 51.60% | 34.8–68% | |
| Genital MusGHV-1 | |||||
| Positive | 14 | 34 | 41.20% | 26.4–57.8% | X2 = 5.672, df = 1, p = 0.017 |
| Negative | 26 | 36 | 72.20% | 56–84.2% | |
| Season:Genital MusGHV-1 | |||||
| Winter:Positive | 7 | 18 | 38.90% | 20.3–61.4% | X2 = 4.0032, df = 1, p = 0.045 |
| Winter:Negative | 17 | 21 | 81% | 60–92.3% | |
| Spring:Positive | 7 | 16 | 43.80% | 23.1–66.8% | X2 = 0.2972, df = 1, p = 0.586 |
| Spring:Negative | 9 | 15 | 60% | 35.7–80.2% | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, M.-s.; Fogarty, U.; Byrne, A.W.; O’Keeffe, J.; Newman, C.; Macdonald, D.W.; Buesching, C.D. Effects of Mustelid gammaherpesvirus 1 (MusGHV-1) Reactivation in European Badger (Meles meles) Genital Tracts on Reproductive Fitness. Pathogens 2020, 9, 769. https://doi.org/10.3390/pathogens9090769
Tsai M-s, Fogarty U, Byrne AW, O’Keeffe J, Newman C, Macdonald DW, Buesching CD. Effects of Mustelid gammaherpesvirus 1 (MusGHV-1) Reactivation in European Badger (Meles meles) Genital Tracts on Reproductive Fitness. Pathogens. 2020; 9(9):769. https://doi.org/10.3390/pathogens9090769
Chicago/Turabian StyleTsai, Ming-shan, Ursula Fogarty, Andrew W. Byrne, James O’Keeffe, Chris Newman, David W. Macdonald, and Christina D. Buesching. 2020. "Effects of Mustelid gammaherpesvirus 1 (MusGHV-1) Reactivation in European Badger (Meles meles) Genital Tracts on Reproductive Fitness" Pathogens 9, no. 9: 769. https://doi.org/10.3390/pathogens9090769








