Pathogens Manipulating Tick Behavior—Through a Glass, Darkly
Abstract
1. Introduction
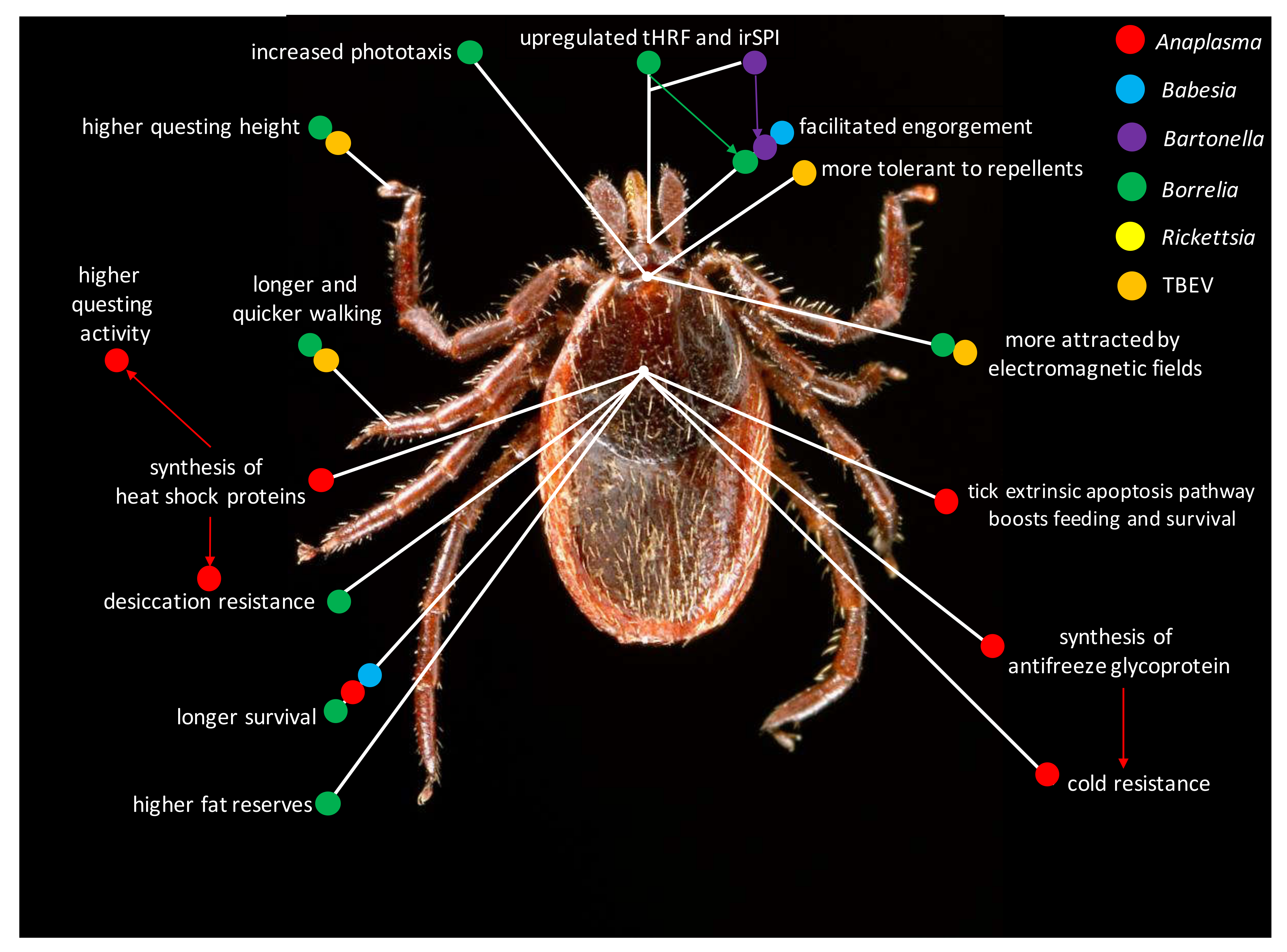
2. Pathogens Infecting Ticks Lead to Major Behavioral Changes
2.1. Behavioral Changes in Borrelia-Infected Ticks
2.1.1. Laboratory Studies
2.1.2. Into the Woods—Field Studies
2.1.3. Borrelia Manipulation of the Host Odors
2.1.4. Ixodes Behavior Meets Physiology
2.2. Behavioral Changes in Anaplasma-Infected Ticks
2.3. Behavioral Changes in Babesia-Infected Ticks
2.4. Behavioral Changes in Bartonella-Infected Ticks
2.5. Behavioral Changes in Rickettsia-Infected Ticks
2.6. Behavioral Changes in TBEV-Infected Ticks
3. Concluding Remarks and Outstanding Challenges for a Research Agenda
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Vector Borne Diseases. Available online: https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases (accessed on 22 June 2020).
- Benelli, G.; Duggan, M.F. Management of arthropod vector data–Social and ecological dynamics facing the One Health perspective. Acta Trop. 2018, 182, 80–91. [Google Scholar] [CrossRef]
- Rosenberg, R.; Lindsey, N.P.; Fischer, M.; Gregory, C.J.; Hinckley, A.F.; Mead, P.S.; Paz-Bailey, G.; Waterman, S.H.; Drexler, N.A.; Kersh, G.J. Vital signs: Trends in reported vectorborne disease cases—United States and Territories, 2004–2016. Morb. Mortal. Wkly. Rep. 2018, 67, 496. [Google Scholar] [CrossRef] [PubMed]
- Wilke, A.B.; Beier, J.C.; Benelli, G. Complexity of the relationship between global warming and urbanization–An obscure future for predicting increases in vector-borne infectious diseases. Curr. Opin. Insect Sci. 2019, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mehlhorn, H. Host manipulations by parasites and viruses. In Parasitology Research Monographs; Springer: Berlin, Germany, 2015; Volume 7, ISBN 3319229362. [Google Scholar]
- Lefèvre, T.; Thomas, F. Behind the scene, something else is pulling the strings: Emphasizing parasitic manipulation in vector-borne diseases. Infect. Genet. Evol. 2008, 8, 504–519. [Google Scholar] [CrossRef] [PubMed]
- Poulin, R. Parasite manipulation of host behavior: An update and frequently asked questions. In Advances in the Study of Behavior; Academic Press: Cambridge, MA, USA, 2010; Volume 41, pp. 151–186. [Google Scholar]
- Poulin, R.; Maure, F. Host manipulation by parasites: A look back before moving forward. Trends Parasitol. 2015, 31, 563–570. [Google Scholar] [CrossRef]
- Lacroix, R.; Mukabana, W.R.; Gouagna, L.C.; Koella, J.C. Malaria infection increases attractiveness of humans to mosquitoes. PLoS Biol. 2005, 3, 1590–1593. [Google Scholar] [CrossRef]
- Cator, L.J.; Lynch, P.A.; Read, A.F.; Thomas, M.B. Do malaria parasites manipulate mosquitoes? Trends Parasitol. 2012, 28, 466–470. [Google Scholar] [CrossRef]
- De Moraes, C.M.; Stanczyk, N.M.; Betz, H.S.; Pulido, H.; Sim, D.G.; Read, A.F.; Mescher, M.C. Malaria-induced changes in host odors enhance mosquito attraction. Proc. Natl. Acad. Sci. USA 2014, 111, 11079–11084. [Google Scholar] [CrossRef]
- Cator, L.J.; George, J.; Blanford, S.; Murdock, C.C.; Baker, T.C.; Read, A.F.; Thomas, M.B. “Manipulation” without the parasite: Altered feeding behaviour of mosquitoes is not dependent on infection with malaria parasites. Proc. R. Soc. B Biol. Sci. 2013, 280. [Google Scholar] [CrossRef]
- O’shea, B.; Rebollar-Tellez, E.; Ward, R.D.; Hamilton, J.G.C.; El Naiem, D.; Polwart, A. Enhanced sandfly attraction to Leishmania-infected hosts. Trans. R. Soc. Trop. Med. Hyg. 2002, 96, 117–118. [Google Scholar] [CrossRef]
- Nevatte, T.M.; Ward, R.D.; Sedda, L.; Hamilton, J.G.C. After infection with Leishmania infantum, Golden Hamsters (Mesocricetus auratus) become more attractive to female sand flies (Lutzomyia longipalpis). Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, L.V.; Kirk Hillier, N.; Smith, T.G. Influence of Hepatozoon parasites on host-seeking and host-choice behaviour of the mosquitoes Culex territans and Culex pipiens. Int. J. Parasitol. Parasites Wildl. 2013, 2, 69–76. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hurd, H. Manipulation of Medically Important Insect Vectors by Their Parasites. Annu. Rev. Entomol. 2003, 48, 141–161. [Google Scholar] [CrossRef] [PubMed]
- Bacot, A.W.; Martin, C.J. LXVII. Observations on the mechanism of the transmission of plague by fleas. J. Hyg. 1914, 13, 423. [Google Scholar] [PubMed]
- Grimstad, P.R.; Ross, Q.E.; Craig, G.B., Jr. Aedes triseriatus (Diptera: Culicidae) and La Crosse Virus: II. Modification of mosquito feeding behavior by virus infection. J. Med. Entomol. 1980, 17, 1–7. [Google Scholar] [CrossRef]
- Jackson, B.T.; Brewster, C.C.; Paulson, S.L. La Crosse virus infection alters blood feeding behavior in Aedes triseriatus and Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2012, 49, 1424–1429. [Google Scholar] [CrossRef]
- Botto-Mahan, C.; Cattan, P.E.; Medel, R. Chagas disease parasite induces behavioural changes in the kissing bug Mepraia spinolai. Acta Trop. 2006, 98, 219–223. [Google Scholar] [CrossRef]
- Killick-Kendrick, R.; Leaney, A.J.; Ready, P.D.; Molyneux, D.H. Leishmania in phlebotomid sandflies-IV. The transmission of Leishmania mexicana amazonensis to hamsters by the bite of experimentally infected Lutzomyia longipalpis. Proc. R. Soc. London. Ser. B. Biol. Sci. 1977, 196, 105–115. [Google Scholar]
- Platt, K.B.; Linthicum, K.J.; Myint, K.S.A.; Innis, B.L.; Lerdthusnee, K.; Vaughn, D.W. Impact of dengue virus infection on feeding behavior of Aedes aegypti. Am. J. Trop. Med. Hyg. 1997, 57, 119–125. [Google Scholar] [CrossRef]
- Jenni, L.; Molyneux, D.H.; Livesey, J.L.; Galun, R. Feeding behaviour of tsetse flies infected with salivarian trypanosomes. Nature 1980, 283, 383–385. [Google Scholar] [CrossRef]
- Roberts, L.W. Probing by Glossina morsitans morsitans and transmission of Trypanosoma (Nannomonas) congolense. Am. J. Trop. Med. Hyg. 1981, 30, 948–951. [Google Scholar] [CrossRef] [PubMed]
- D’alessandro, A.; Mandel, S. Natural infections and behavior of Trypanosoma rangeli and Trypanosoma cruzi in the vector Rhodnius prolixus in Colombia. J. Parasitol. 1969, 55, 846–852. [Google Scholar] [CrossRef]
- Beach, R.; Kiilu, G.; Leeuwenburg, J. Modification of sand fly biting behavior by Leishmania leads to increased parasite transmission. Am. J. Trop. Med. Hyg. 1985, 34, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-González, M.G.; Flores-Villegas, A.L.; Salazar-Schettino, P.M.; Gutiérrez-Cabrera, A.E.; Rojas-Ortega, E.; Córdoba-Aguilar, A. Zombie bugs? Manipulation of kissing bug behavior by the parasite Trypanosoma cruzi. Acta Trop. 2019, 200, 105177. [Google Scholar] [CrossRef] [PubMed]
- Lima-Camara, T.N.; Bruno, R.V.; Luz, P.M.; Castro, M.G.; Lourenço-de-Oliveira, R.; Sorgine, M.H.F.; Peixoto, A.A. Dengue infection increases the locomotor activity of Aedes aegypti females. PLoS ONE 2011, 6, e17690. [Google Scholar] [CrossRef]
- Reese, S.M.; Beaty, M.K.; Gabitzsch, E.S.; Blair, C.D.; Beaty, B.J. Aedes triseriatus females transovarially infected with La Crosse virus mate more efficiently than uninfected mosquitoes. J. Med. Entomol. 2009, 46, 1152–1158. [Google Scholar] [CrossRef][Green Version]
- Wekesa, J.W.; Copeland, R.S.; Mwangi, R.W. Effect of Plasmodium falciparum on blood feeding behavior of naturally infected Anopheles mosquitoes in western Kenya. Am. J. Trop. Med. Hyg. 1992, 47, 484–488. [Google Scholar] [CrossRef]
- Anderson, R.A.; Koella, J.C.; Hurd, H. The effect of Plasmodium yoelii nigeriensis infection on the feeding persistence of Anopheles stephensi Liston throughout the sporogonic cycle. Proc. R. Soc. B Biol. Sci. 1999, 266, 1729–1733. [Google Scholar] [CrossRef]
- Koella, J.C.; Packer, M.J. Malaria parasites enhance blood-feeding of their naturally infected vector Anopheles punctulatus. Parasitology 1996, 113, 105–109. [Google Scholar] [CrossRef]
- Koella, J.C.; Rieu, L.; Paul, R.E.L. Stage-specific manipulation of a mosquito’s host-seeking behavior by the malaria parasite Plasmodium gallinaceum. Behav. Ecol. 2002, 13, 816–820. [Google Scholar] [CrossRef]
- Schwartz, A.; Koella, J.C. Trade-offs, conflicts of interest and manipulation in Plasmodium-mosquito interactions. Trends Parasitol. 2001, 17, 189–194. [Google Scholar] [CrossRef]
- Duke, A.L. On the effect on the longevity of G. palpalis of trypanosome infections. Ann. Trop. Med. Parasitol. 1928, 22, 25–32. [Google Scholar] [CrossRef]
- Baker, J.R.; Robertson, D.H.H. An experiment on the infectivity to Glossina morsitans of a strain of Trypanosoma rhodesiense and of a strain of T. brucei, with some observations on the longevity of infected flies. Ann. Trop. Med. Parasitol. 1957, 51, 121–135. [Google Scholar]
- Maudlin, I.; Welburn, S.C.; Milligan, P.J.M. Trypanosome infections and survival in tsetse. Parasitology 1998, 116, 23–28. [Google Scholar] [CrossRef] [PubMed]
- El Sawaf, B.M.; El Sattar, S.A.; Shehata, M.G.; Lane, R.P.; Morsy, T.A. Reduced longevity and fecundity in Leishmania-infected sand flies. Am. J. Trop. Med. Hyg. 1994, 51, 767–770. [Google Scholar] [CrossRef]
- Turell, M.J.; Gargan, T.P.; Bailey, C.L. Culex pipiens (Diptera: Culicidae) morbidity and mortality associated with Rift Valley fever virus infection. J. Med. Entomol. 1985, 22, 332–337. [Google Scholar] [CrossRef]
- Fellet, M.R.; Lorenzo, M.G.; Elliot, S.L.; Carrasco, D.; Guarneri, A.A. Effects of infection by Trypanosoma cruzi and Trypanosoma rangeli on the reproductive performance of the vector Rhodnius prolixus. PLoS ONE 2014, 9, 26–32. [Google Scholar] [CrossRef]
- Peterson, J.K.; Graham, A.L.; Elliott, R.J.; Dobson, A.P.; Chávez, O.T. Trypanosoma cruzi–Trypanosoma rangeli co-infection ameliorates negative effects of single trypanosome infections in experimentally infected Rhodnius prolixus. Parasitology 2016, 143, 1157–1167. [Google Scholar] [CrossRef]
- Cordero-Montoya, G.; Flores-Villegas, A.L.; Salazar-Schettino, P.M.; Vences-Blanco, M.O.; Rocha-Ortega, M.; Gutiérrez-Cabrera, A.E.; Rojas-Ortega, E.; Córdoba-Aguilar, A. The cost of being a killer’s accomplice: Trypanosoma cruzi impairs the fitness of kissing bugs. Parasitol. Res. 2019, 118, 2523–2529. [Google Scholar] [CrossRef]
- Cézilly, F.; Thomas, F.; Médoc, V.; Perrot-Minnot, M.J. Host-manipulation by parasites with complex life cycles: Adaptive or not? Trends Parasitol. 2010, 26, 311–317. [Google Scholar] [CrossRef]
- Schmid Hempel, P. Evolutionary Parasitologythe Integrated Study of Infections, Immunology, Ecology, and Genetics; OUP Oxford: Oxford, UK, 2011; ISBN 0199229481. [Google Scholar]
- Gassner, F.; Hartemink, N. Tick–Borrelia interactions: Burden or benefit? In Ecology of Parasite-Vector Interactions; Wageningen Academic Publishers: Wageningen, The Netherlands, 2013; pp. 141–154. [Google Scholar]
- Levi, T.; Kilpatrick, A.M.; Mangel, M.; Wilmers, C.C. Deer, predators, and the emergence of Lyme disease. Proc. Natl. Acad. Sci. USA 2012, 109, 10942–10947. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Maggi, F.; Canale, A.; Mehlhorn, H. Lyme disease is on the rise—How about tick repellents? A global view. Entomol. Gen. 2019, 39, 61–72. [Google Scholar] [CrossRef]
- Lefcort, H.; Durden, L.A. The effect of infection with Lyme disease spirochetes (Borrelia burgdorferi) on the phototaxis, activity, and questing height of the tick vector Ixodes scapularis. Parasitology 1996, 113, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Narasimhan, S.; Zhang, L.; Liu, L.; Wang, P.; Fikrig, E. Tick histamine release factor is critical for Ixodes scapularis engorgement and transmission of the Lyme disease agent. PLoS Pathog. 2010, 6. [Google Scholar] [CrossRef] [PubMed]
- Romashchenko, A.V.; Ratushnyak, A.S.; Zapara, T.A.; Tkachev, S.E.; Moshkin, M.P. The correlation between tick (Ixodes persulcatus Sch.) questing behaviour and synganglion neuronal responses to odours. J. Insect Physiol. 2012, 58, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Alekseev, A.N.; Jensen, P.M.; Dubinina, H.V.; Smirnova, L.A.; Makrouchina, N.A.; Zharkov, S.D. Peculiarities of behaviour of taiga (Ixodes persulcatus) and sheep (Ixodes ricinus) ticks (Acarina: Ixodidae) determined by different methods. Folia Parasitol. (Praha). 2000, 47, 147–153. [Google Scholar] [CrossRef]
- Alekseev, A.N.; Dubinina, H.V. Symbiotical relationships in the complicated vector-pathogen system. Dokl. Akad. Nauk. 1994, 338, 259–261. [Google Scholar]
- Alekseev, A.N. Tick pathogen interactions: Behaviour of infected and uninfected ticks (Ixodidae). In Acarology IX; Mitchell, R., Horn, D.J., Needham, G.R., Welbourn, W.C., Eds.; Ohio Biological Survey: Columbus, OH, USA, 1996; Volume 1, pp. 113–115. [Google Scholar]
- Perret, J.L. Computer-assisted Laboratory Observations and Field Studies of the Host-finding Behaviour of the Tick ’Ixodes ricinus’ (Acarina: Ixodidae): Ecological Implications of Climate and Light. Ph.D. Thesis, University of Neuchátel, Neuchátel, Switzerland, 2003. [Google Scholar]
- Gassner, F. Tick Tactics: Interactions Between Habitat Characteristics, Hosts and Microorganisms in Relation to the Biology of the Sheep Tick Ixodes Ricinus. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2010. [Google Scholar]
- Alekseev, A.N.; Dubinina, H.V. Abiotic parameters and diel and seasonal activity of Borrelia-infected and uninfected Ixodes persulcatus (Acarina: Ixodidae). J. Med. Entomol. 2000, 37, 9–15. [Google Scholar] [CrossRef]
- Lane, R.S.; Mun, J.; Peribáñez, M.A.; Stubbs, H.A. Host-seeking behavior of Ixodes pacificus (Acari: Ixodidae) nymphs in relation to environmental parameters in dense-woodland and woodland-grass habitats. J. Vector Ecol. 2007, 32, 342–357. [Google Scholar] [CrossRef]
- Faulde, M.K.; Robbins, R.G. Tick infestation risk and Borrelia burgdorferi s.l. infection-induced increase in host-finding efficacy of female Ixodes ricinus under natural conditions. Exp. Appl. Acarol. 2008, 44, 137. [Google Scholar] [CrossRef]
- Robinson, A.; Busula, A.O.; Voets, M.A.; Beshir, K.B.; Caulfield, J.C.; Powers, S.J.; Verhulst, N.O.; Winskill, P.; Muwanguzi, J.; Birkett, M.A.; et al. Plasmodium-associated changes in human odor attract mosquitoes. Proc. Natl. Acad. Sci. USA 2018, 115, E4209–E4218. [Google Scholar] [CrossRef] [PubMed]
- Van Duijvendijk, G.; Van Andel, W.; Fonville, M.; Gort, G.; Hovius, J.W.; Sprong, H.; Takken, W. A Borrelia afzelii infection increases larval tick burden on Myodes glareolus (Rodentia: Cricetidae) and Nymphal Body Weight of Ixodes ricinus (Acari: Ixodidae). J. Med. Entomol. 2018, 54, 422–428. [Google Scholar] [CrossRef]
- Berret, J.; Voordouw, M.J. Lyme disease bacterium does not affect attraction to rodent odour in the tick vector. Parasites Vectors 2015, 8, 1–9. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ramamoorthi, N.; Narasimhan, S.; Pal, U.; Bao, F.; Yang, X.F.; Fish, D.; Anguita, J.; Norgard, M.V.; Kantor, F.S.; Anderson, J.F.; et al. The Lyme disease agent exploits a tick protein to infect the mammalian host. Nature 2005, 436, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, C.; Gern, L. Search for blood or water is influenced by Borrelia burgdorferi in Ixodes ricinus. Parasites Vectors 2015, 8, 4–11. [Google Scholar] [CrossRef]
- Herrmann, C.; Gern, L. Survival of Ixodes ricinus (Acari: Ixodidae) under challenging conditions of temperature and humidity is influenced by Borrelia burgdorferi sensu lato infection. J. Med. Entomol. 2010, 47, 1196–1204. [Google Scholar] [CrossRef]
- Herrmann, C.; Voordouw, M.J.; Gern, L. Ixodes ricinus ticks infected with the causative agent of Lyme disease, Borrelia burgdorferi sensu lato, have higher energy reserves. Int. J. Parasitol. 2013, 43, 477–483. [Google Scholar] [CrossRef]
- Herrmann, C.; Gern, L. Do the level of energy reserves, hydration status and Borrelia infection influence walking by Ixodes ricinus (Acari: Ixodidae) ticks? Parasitology 2012, 139, 330–337. [Google Scholar] [CrossRef]
- Randolph, S.E.; Storey, K. Impact of microclimate on immature tick-rodent host interactions (Acari: Ixodidae): Implications for parasite transmission. J. Med. Entomol. 1999, 36, 741–748. [Google Scholar] [CrossRef]
- Cabezas-Cruz, A.; Alberdi, P.; Ayllón, N.; Valdés, J.J.; Pierce, R.; Villar, M.; de la Fuente, J. Anaplasma phagocytophilum increases the levels of histone modifying enzymes to inhibit cell apoptosis and facilitate pathogen infection in the tick vector Ixodes scapularis. Epigenetics 2016, 11, 303–319. [Google Scholar] [CrossRef]
- de la Fuente, J.; Villar, M.; Cabezas-Cruz, A.; Estrada-Peña, A.; Ayllón, N.; Alberdi, P. Tick–Host–Pathogen Interactions: Conflict and Cooperation. PLoS Pathog. 2016, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, J.; Antunes, S.; Bonnet, S.; Cabezas-Cruz, A.; Domingos, A.G.; Estrada-Peña, A.; Johnson, N.; Kocan, K.M.; Mansfield, K.L.; Nijhof, A.M.; et al. Tick-Pathogen Interactions and Vector Competence: Identification of Molecular Drivers for Tick-Borne Diseases. Front. Cell. Infect. Microbiol. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Cabezas-Cruz, A.; Estrada-Peña, A.; Rego, R.O.M.; De la Fuente, J. Tick-pathogen Ensembles: Do molecular interactions lead ecological innovation? Front. Cell. Infect. Microbiol. 2017, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Cabezas-Cruz, A.; Alberdi, P.; Valdés, J.J.; Villar, M.; de la Fuente, J. Anaplasma phagocytophilum infection subverts carbohydrate metabolic pathways in the tick vector, Ixodes scapularis. Front. Cell. Infect. Microbiol. 2017, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Busby, A.T.; Ayllón, N.; Kocan, K.M.; Blouin, E.F.; de La Fuente, G.; Galindo, R.C.; Villar, M.; de la Fuente, J. Expression of heat shock proteins and subolesin affects stress responses, Anaplasma phagocytophilum infection and questing behaviour in the tick, Ixodes scapularis. Med. Vet. Entomol. 2012, 26, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Ayllón, N.; Naranjo, V.; Hajduek, O.; Villar, M.; Galindo, R.C.; Kocan, K.M.; Alberdi, P.; Šíma, R.; Cabezas-Cruz, A.; Rückert, C.; et al. Nuclease Tudor-SN is involved in tick dsRNAMediated RNA interference and feeding but not in defense against flaviviral or Anaplasma phagocytophilum rickettsial infection. PLoS ONE 2015, 10, 1–18. [Google Scholar] [CrossRef]
- Neelakanta, G.; Sultana, H.; Fish, D.; Anderson, J.F.; Fikrig, E. Anaplasma phagocytophilum induces Ixodes scapularis ticks to express an antifreeze glycoprotein gene that enhances their survival in the cold. J. Clin. Investig. 2010, 120, 3179–3190. [Google Scholar] [CrossRef]
- Ayllón, N.; Villar, M.; Galindo, R.C.; Kocan, K.M.; Šíma, R.; López, J.A.; Vázquez, J.; Alberdi, P.; Cabezas-Cruz, A.; Kopáček, P.; et al. Systems Biology of Tissue-Specific Response to Anaplasma phagocytophilum Reveals Differentiated Apoptosis in the Tick Vector Ixodes scapularis. PLoS Genet. 2015, 11, 1–29. [Google Scholar] [CrossRef]
- Villar, M.; Ayllón, N.; Alberdi, P.; Moreno, A.; Moreno, M.; Tobes, R.; Mateos-Hernández, L.; Weisheit, S.; Bell-Sakyi, L.; de la Fuente, J. Integrated metabolomics, transcriptomics and proteomics identifies metabolic pathways affected by Anaplasma phagocytophilum infection in tick cells. Mol. Cell. Proteomics 2015, 14, 3154–3172. [Google Scholar] [CrossRef]
- Randolph, S.E. The effect of Babesia microti on feeding and survival in its tick vector, Ixodes trianguliceps. Parasitology 1991, 102, 9–16. [Google Scholar] [CrossRef]
- Hu, R.; Hyland, K.E.; Markowski, D. Effects of Babesia microti Infection on Feeding Pattern, Engorged Body Weight, and Molting Rate of Immature Ixodes scapularis (Acari: Ixodidae). J. Med. Entomol. 1997, 34, 559–564. [Google Scholar] [CrossRef]
- Liu, X.Y.; de la Fuente, J.; Cote, M.; Galindo, R.C.; Moutailler, S.; Vayssier-Taussat, M.; Bonnet, S.I. IrSPI, a tick serine protease inhibitor involved in tick feeding and Bartonella henselae infection. PLoS Negl Trop Dis 2014, 8, e2993. [Google Scholar] [CrossRef] [PubMed]
- Frątczak, M.; Vargová, B.; Tryjanowski, P.; Majláth, I.; Jerzak, L.; Kurimský, J.; Cimbala, R.; Jankowiak, Ł.; Conka, Z.; Majláthová, V. Infected Ixodes ricinus ticks are attracted by electromagnetic radiation of 900 MH. Ticks Tick. Borne. Dis. 2020, 11, 101416. [Google Scholar] [CrossRef] [PubMed]
- Süss, J.; Schrader, C.; Falk, U.; Wohanka, N. Tick-borne encephalitis (TBE) in Germany—Epidemiological data, development of risk areas and virus prevalence in field-collected ticks and in ticks removed from humans. Int. J. Med. Microbiol. Suppl. 2004, 293, 69–79. [Google Scholar] [CrossRef]
- Mel’nikova, O.V.; Botvinkin, A.D.; Danchinova, G.A. Comparative data on the tick-borne encephalitis virus infectiousness of hungry and satiated taiga ticks (based on the results of an immunoenzyme analysis). Med. Parazitol. (Mosk). 1997, 1, 44–49. [Google Scholar]
- Romanenko, V.N.; Kondrat’eva, L.M. The infection of ixodid ticks collected from humans with the tick-borne encephalitis virus in Tomsk city and its suburbs. Parazitologiia 2011, 45, 3–10. [Google Scholar]
- Belova, O.A.; Burenkova, L.A.; Karganova, G.G. Different tick-borne encephalitis virus (TBEV) prevalences in unfed versus partially engorged ixodid ticks - Evidence of virus replication and changes in tick behavior. Ticks Tick. Borne Dis. 2012, 3, 240–246. [Google Scholar] [CrossRef]
- Alekseev, A.N.; Burenkova, L.A.; Chunikhin, S.P. Behavioral characteristics of Ixodes persulcatus P. Sch. ticks infected with the tick-borne encephalitis virus. Med. Parazitol. (Mosk). 1988, 2, 71–75. [Google Scholar]
- Poulin, R. Evolution of parasite life history traits: Myths and reality. Parasitol. Today 1995, 11, 342–345. [Google Scholar] [CrossRef]
- Lee, J.H.; Rowley, W.A.; Platt, K.B. Longevity and spontaneous flight activity of Culex tarsalis (Diptera: Culicidae) infected with western equine encephalomyelitis virus. J. Med. Entomol. 2000, 37, 187–193. [Google Scholar] [CrossRef]
- Bennett, K.E.; Hopper, J.E.; Stuart, M.A.; West, M.; Drolet, B.S. Blood-feeding behavior of vesicular stomatitis virus infected Culicoides sonorensis (Diptera: Ceratopogonidae). J. Med. Entomol. 2008, 45, 921–926. [Google Scholar] [CrossRef]
- Guglielmone, A.A.; Robbins, R.G.; Apanaskevich, D.A.; Petney, T.N.; Estrada-Peña, A.; Horak, I.G. Individual Species Accounts. In The Hard Ticks of the World; Springer: Dordrecht, The Netherlands, 2014; pp. 13–218. [Google Scholar]
- Romano, D.; Stefanini, C.; Canale, A.; Benelli, G. Artificial blood feeders for mosquito and ticks—Where from, where to? Acta Trop. 2018, 183, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Romano, D.; Rocchigiani, G.; Caselli, A.; Mancianti, F.; Canale, A.; Stefanini, C. Behavioral asymmetries in ticks–lateralized questing of Ixodes ricinus to a mechatronic apparatus delivering host-borne cues. Acta Trop. 2018, 178, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and tick-borne diseases: A One Health perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef]
- Benelli, G.; Senthil-Nathan, S. Together in the Fight against Arthropod-Borne Diseases: A One Health Perspective. Int. J. Environ. Res. Publ. Health. 2018, 16, 4876. [Google Scholar] [CrossRef]
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benelli, G. Pathogens Manipulating Tick Behavior—Through a Glass, Darkly. Pathogens 2020, 9, 664. https://doi.org/10.3390/pathogens9080664
Benelli G. Pathogens Manipulating Tick Behavior—Through a Glass, Darkly. Pathogens. 2020; 9(8):664. https://doi.org/10.3390/pathogens9080664
Chicago/Turabian StyleBenelli, Giovanni. 2020. "Pathogens Manipulating Tick Behavior—Through a Glass, Darkly" Pathogens 9, no. 8: 664. https://doi.org/10.3390/pathogens9080664
APA StyleBenelli, G. (2020). Pathogens Manipulating Tick Behavior—Through a Glass, Darkly. Pathogens, 9(8), 664. https://doi.org/10.3390/pathogens9080664




