Mycoplasma bovis Infections—Occurrence, Diagnosis and Control
Abstract
1. Introduction
2. Mycoplasma bovis: Key Facts
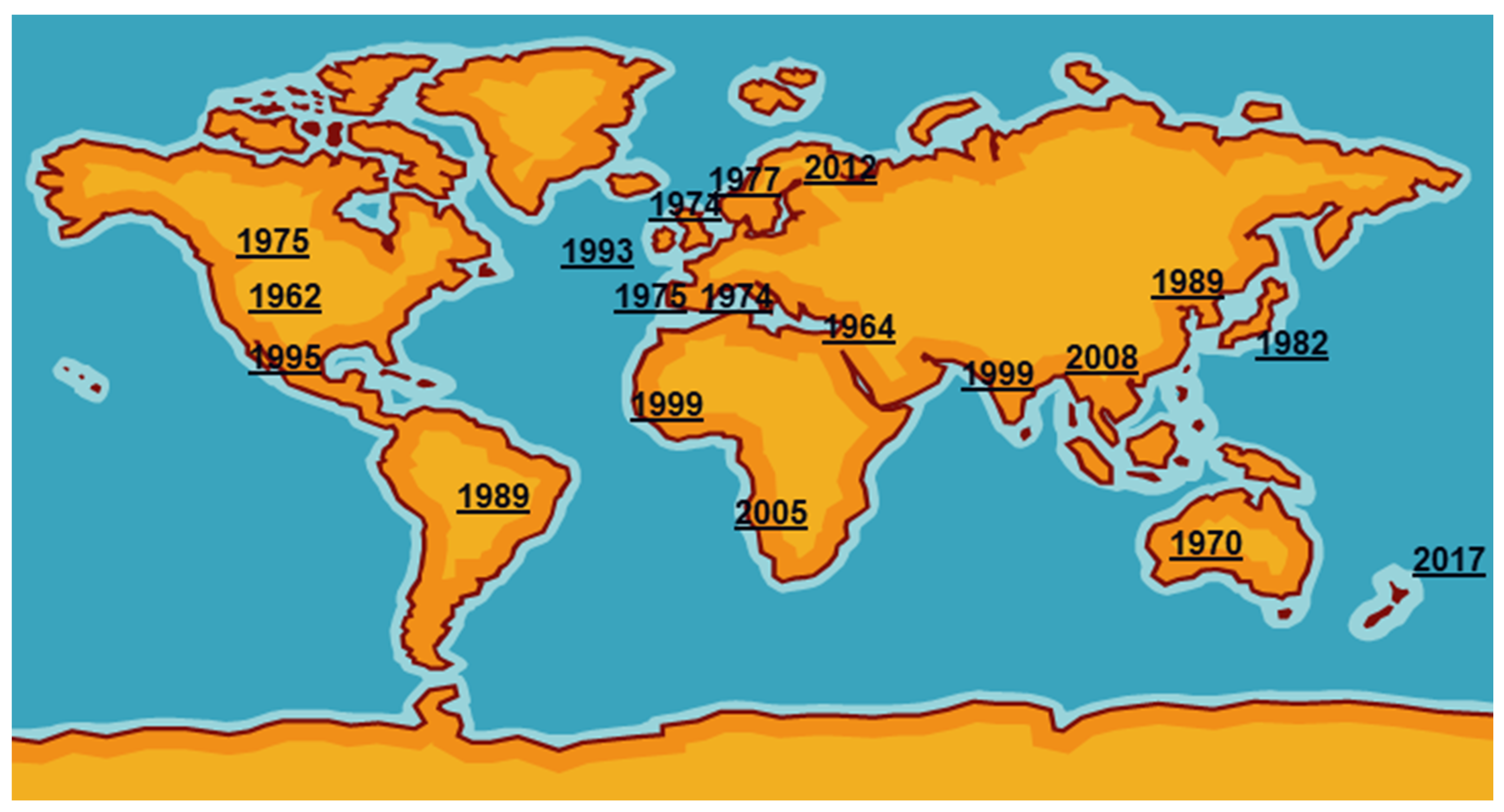
3. Current Reports on the Epizootic Situation of M. bovis
4. Disease Course and Source of M. bovis Infection
5. Currently Used Diagnostic Methods
5.1. Real-Time PCR Assays for M. bovis Detection
5.2. Fast and Cost-Effective Assays for M. bovis Detection
5.3. Immunohistochemistry and In-Situ Hybridization
5.4. A Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry for M. bovis Detection
5.5. Molecular Typing
5.6. Serological Approaches
5.7. Interlaboratory Trials of Diagnostic Tests
5.8. Mixed Infections
6. Control—Recommendations for M. bovis Control Programs
Author Contributions
Funding
Conflicts of Interest
References
- Ministry for Primary Industries. First Case of Mycoplasma Bovis Found in Bay of Plenty. 2017. Available online: https://www.tvnz.co.nz/one-news/new-zealand/first-case-mycoplasma-bovis-found-in-bay-plenty (accessed on 20 June 2020).
- Haapala, V.; Pohjanvirta, T.; Vähänikkilä, N.; Halkilahti, J.; Simonen, H.; Pelkonen, S.; Soveri, T.; Simojoki, H.; Autio, T. Semen as a source of Mycoplasma bovis mastitis in dairy herds. Vet. Microbiol. 2018, 216, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, R.; Ayling, R.; McAuliffe, L. Mycoplasma Diseases of Ruminants, 1st ed.; CABI Publishing: Oxford, UK, 2008. [Google Scholar]
- Nicholas, R.A.J.; Fox, L.K.; Lysnyansky, I. Mycoplasma mastitis in cattle: To cull or not to cull. Vet. J. 2016, 216, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Klein, U.; de Jong, A.; Youala, M.; El Garch, F.; Stevenin, C.; Moyaert, H.; Rose, M.; Catania, S.; Gyuranecz, M.; Pridmore, A.; et al. New antimicrobial susceptibility data from monitoring of Mycoplasma bovis isolated in Europe. Vet. Microbiol. 2019, 238, 108432. [Google Scholar] [CrossRef] [PubMed]
- Houlihan, M.G.; Veenstra, B.; Christian, M.K.; Nicholas, R.; Ayling, R. Mastitis and arthritis in two dairy herds caused by Mycoplasma bovis. Vet. Rec. 2007, 160, 126–127. [Google Scholar] [CrossRef]
- Maeda, T.; Shibahara, T.; Kimura, K.; Wada, Y.; Sato, K.; Imada, Y.; Ishikawa, Y.; Kadota, K. Mycoplasma bovis-associated suppurative otitis media and pneumonia in bull calves. J. Comp. Pathol. 2003, 129, 100–110. [Google Scholar] [CrossRef]
- Alberti, A.; Filippa Addis, M.; Chessa, B.; Cubeddu, T.; Profiti, M.; Rosati, S.; Ruiu, A.; Pittau, M. Molecular and antigenic characterization of a Mycoplasma bovis strain causing an outbreak of infectious keratoconjunctivitis. J. Vet. Diagn. Invest. 2006, 18, 41–51. [Google Scholar] [CrossRef]
- Kinde, H.; Daft, B.M.; Walker, R.L.; Charlton, B.R.; Petty, R. Mycoplasma bovis associated with decubital abscesses in Holstein calves. J. Vet. Diagn. Invest. 1993, 5, 194–197. [Google Scholar] [CrossRef]
- Kanda, T.; Tanaka, S.; Suwanruengsri, M.; Sukmawinata, E.; Uemura, R.; Yamaguchi, R.; Sueyoshi, M. Bovine Endocarditis Associated with Mycoplasma bovis. J. Comp. Pathol. 2019, 171, 53–58. [Google Scholar] [CrossRef]
- Pardon, B.; Callens, J.; Maris, J.; Allais, L.; Van Praet, W.; Deprez, P.; Ribbens, S. Pathogen-specific risk factors in acute outbreaks of respiratory disease in calves. J. Dairy Sci. 2020, 103, 2556–2566. [Google Scholar] [CrossRef]
- Oliveira, T.E.S.; Pelaquim, I.F.; Flores, E.F.; Massi, R.P.; Valdiviezo, M.J.J.; Pretto-Giordano, L.G.; Alfieri, A.A.; Saut, J.P.E.; Headley, S.A. Mycoplasma bovis and viral agents associated with the development of bovine respiratory disease in adult dairy cows. Transbound. Emerg. Dis. 2019, 67, 82–93. [Google Scholar] [CrossRef]
- Hazelton, M.S.; Morton, J.M.; Parker, A.M.; Sheehy, P.A.; Bosward, K.L.; Malmo, J.; House, J.K. Whole dairy herd sampling to detect subclinical intramammary Mycoplasma bovis infection after clinical mastitis outbreaks. Vet. Microbiol. 2020, 244, 108662. [Google Scholar] [CrossRef] [PubMed]
- Punyapornwithaya, V.; Fox, L.K.; Hancock, D.D.; Gay, J.M.; Alldredge, J.R. Association between an outbreak strain causing mycoplasma bovis mastitis and its asymptomatic carriage in the herd: A case study from Idaho, USA. Prev. Vet. Med. 2010, 93, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Ayling, R.D.; Rosales, R.S.; Barden, G.; Gosney, F.L. Changes in antimicrobial susceptibility of Mycoplasma bovis isolates from Great Britain. Vet. Rec. 2014, 175, 486. [Google Scholar] [CrossRef] [PubMed]
- Gautier-Bouchardon, A.V.; Ferré, S.; Le Grand, D.; Paoli, A.; Gay, E.; Poumarat, F. Overall decrease in the susceptibility of Mycoplasma bovis to antimicrobials over the past 30 years in France. PLoS ONE 2014, 9, e87672. [Google Scholar] [CrossRef]
- Sulyok, K.M.; Kreizinger, Z.; Fekete, L.; Hrivnák, V.; Magyar, T.; Jánosi, S.; Schweitzer, N.; Turcsányi, I.; Makrai, L.; Erdélyi, K.; et al. Antibiotic susceptibility profiles of Mycoplasma bovis strains isolated from cattle in Hungary, Central Europe. BMC Vet. Res. 2014, 10, 256. [Google Scholar] [CrossRef]
- Ayling, R.D.; Baker, S.E.; Peek, M.L.; Simon, A.J.; Nicholas, R.A.J. Comparison of in vitro activity of danofloxacin, florfenicol, oxytetracycline, spectinomycin and tilmicosin against recent field isolates of Mycoplasma bovis. Vet. Rec. 2000, 146, 745–747. [Google Scholar] [CrossRef]
- Dudek, K.; Bednarek, D.; Ayling, R.D.; Kycko, A.; Reichert, M. Preliminary study on the effects of enrofloxacin, flunixin meglumine and pegbovigrastim on Mycoplasma bovis pneumonia. BMC Vet. Res. 2019, 15, 371. [Google Scholar] [CrossRef]
- Ridley, A.; Hateley, G. Mycoplasma bovis investigations in cattle. Vet. Rec. 2018, 183, 256–258. [Google Scholar]
- Dudek, K.; Bednarek, D.; Ayling, R.D.; Kycko, A.; Szacawa, E.; Karpińska, T.A. An experimental vaccine composed of two adjuvants gives protection against Mycoplasma bovis in calves. Vaccine 2016, 34, 3051–3058. [Google Scholar] [CrossRef]
- Nicholas, R.A.; Ayling, R.D.; Stipkovits, L.P. An experimental vaccine for calf pneumonia caused by Mycoplasma bovis: Clinical, cultural, serological and pathological findings. Vaccine 2002, 20, 3569–3575. [Google Scholar] [CrossRef]
- Vähänikkilä, N.; Pohjanvirta, T.; Haapala, V.; Simojoki, H.; Soveri, T.; Browning, G.F.; Pelkonen, S.; Wawegama, N.K.; Autio, T. Characterisation of the course of Mycoplasma bovis infection in naturally infected dairy herds. Vet. Microbiol. 2019, 231, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, R.A. Bovine mycoplasmosis: Silent and deadly. Vet. Rec. 2011, 168, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Technical Advisory Group (TAG) Report. 2019. Available online: https://www.mpi.govt.nz/dmsdocument/37754-report-of-the-mycoplasma-bovis-technical-advisory-group-tag-in-response-to-the-terms-of-reference-june-2019-18-october-2019 (accessed on 5 August 2020).
- Timonen, A.A.E.; Autio, T.; Pohjanvirta, T.; Häkkinen, L.; Katholm, J.; Petersen, A.; Mõtus, K.; Kalmus, P. Dynamics of the within-herd prevalence of Mycoplasma bovis intramammary infection in endemically infected dairy herds. Vet. Microbiol. 2020, 242, 108608. [Google Scholar] [CrossRef] [PubMed]
- Gille, L.; Evrard, J.; Callens, J.; Supré, K.; Grégoire, F.; Boyen, F.; Haesebrouck, F.; Deprez, P.; Pardon, B. The presence of Mycoplasma bovis in colostrum. Vet. Res. 2020, 51, 54. [Google Scholar] [CrossRef]
- Passchyn, P.; Piepers, S.; De Meulemeester, L.; Boyen, F.; Haesebrouck, F.; De Vliegher, S. Between-herd prevalence of Mycoplasma bovis in bulk milk in Flanders, Belgium. Res. Vet. Sci. 2012, 92, 219–220. [Google Scholar] [CrossRef]
- Stott, G.H.; Wiersma, F.; Menefee, B.E.; Radwanski, F.R. Influence of environment on passive immunity in calves. J. Dairy Sci. 1976, 59, 1306–1311. [Google Scholar] [CrossRef]
- Alabdullah, H.; Schneider, C.; Fox, L. Effect of dexamethasone administration on shedding of Mycoplasma bovis in calves. In Proceedings of the Third International Symposium on Mastitis and Milk Quality, St. Louis, MO, USA, 22–24 September 2011. [Google Scholar]
- Alabdullah, H.A.; Fox, L.K.; Gay, J.M.; Barrington, G.M.; Mealey, R.H. Effects of dexamethasone and Mycoplasma bovis on bovine neutrophil function in vitro. Vet. Immunol. Immunopathol. 2015, 164, 67–73. [Google Scholar] [CrossRef]
- Ayling, R.D.; Bashiruddin, S.E.; Nicholas, R.A.J. Mycoplasma species and related organisms isolated from ruminants in Britain between 1990 and 2000. Vet. Rec. 2004, 155, 413–416. [Google Scholar] [CrossRef]
- Gagea, M.I.; Bateman, K.G.; Shanahan, R.A.; van Dreumel, T.; McEwen, B.J.; Carman, S.; Archambault, M.; Caswell, J.L. Naturally occurring Mycoplasma bovis-associated pneumonia and polyarthritis in feedlot beef calves. J. Vet. Diagn. Invest. 2006, 18, 29–40. [Google Scholar] [CrossRef]
- Ryan, M.J.; Wyand, D.S.; Hill, D.L.; Tourtellotte, M.E.; Yang, T.J. Morphologic changes following intraarticular inoculation of Mycoplasma bovis in calves. Vet. Pathol. 1983, 20, 472–487. [Google Scholar] [CrossRef]
- Foster, A.P.; Naylor, R.D.; Howie, N.M.; Nicholas, R.A.; Ayling, R.D. Mycoplasma bovis and otitis in dairy calves in the United Kingdom. Vet. J. 2009, 179, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Ayling, R.; Nicholas, R.; Hogg, R.; Wessels, J.; Scholes, S.; Byrne, W.; Hill, M.; Moriarty, J.; O’Brien, T. Mycoplasma bovis isolated from brain tissue of calves. Vet. Rec. 2005, 156, 391–392. [Google Scholar] [CrossRef] [PubMed]
- Kanci, A.; Wawegama, N.K.; Marenda, M.S.; Mansell, P.D.; Browning, G.F.; Markham, P.F. Reproduction of respiratory mycoplasmosis in calves by exposure to an aerosolised culture of Mycoplasma bovis. Vet. Microbiol. 2017, 210, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Jasper, D.E.; Al-Aubaidi, J.M.; Fabricant, J. Epidemiologic observations on mycoplasma mastitis. Cornell Vet. 1974, 64, 407–415. [Google Scholar]
- Maunsell, F.P.; Woolums, A.R.; Francoz, D.; Rosenbush, R.F.; Step, D.L.; Wilson, D.J.; Janzen, E.D. Mycoplasma bovis infections in cattle. ACVIM Consensus Statement. J. Vet. Intern. Med. 2011, 25, 772–783. [Google Scholar] [CrossRef]
- Lysnyansky, I.; Freed, M.; Rosales, R.S.; Mikula, I.; Khateb, N.; Gerchman, I.; van Straten, M.; Levisohn, S. An overview of Mycoplasma bovis mastitis in Israel (2004–2014). Vet. J. 2016, 207, 180–183. [Google Scholar] [CrossRef]
- Hermeyer, K.; Peters, M.; Brügmann, M.; Jacobsen, B.; Hewicker-Trautwein, M. Demonstration of Mycoplasma bovis by immunohistochemistry and in situ hybridization in an aborted bovine fetus and neonatal calf. J. Vet. Diagn. Invest. 2012, 24, 364–369. [Google Scholar] [CrossRef]
- Pfützner, H.; Sachse, K. Mycoplasma bovis as an agent of mastitis, pneumonia, arthritis and genital disorders in cattle. Rev. Sci. Tech. Off. Int. Epiz. 1996, 15, 1477–1494. [Google Scholar] [CrossRef]
- Poumarat, F.; Longchambon, D.; Martel, J.L. Application of dot immunobinding on membrane filtration (MF dot) to the study of relationships within “M. mycoides cluster” and within “glucose and arginine-negative cluster” of ruminant mycoplasmas. Vet. Microbiol. 1992, 32, 375–390. [Google Scholar] [CrossRef]
- Nicholas, R.A.J.; Baker, S.E. Recovery of mycoplasmas from animals. In Mycoplasma Protocols; Miles, R.J., Nicholas, R.A.J., Eds.; Humana Press: Totowa, NJ, USA, 1998; pp. 37–44. [Google Scholar]
- Behera, S.; Rana, R.; Gupta, P.K.; Kumar, D.; Sonal; Rekha, V.; Arun, T.R.; Jena, D. Development of real-time PCR assay for the detection of Mycoplasma bovis. Trop. Anim. Health Prod. 2018, 50, 875–882. [Google Scholar] [CrossRef]
- Andrés-Lasheras, S.; Zaheer, R.; Ha, R.; Lee, C.; Jelinski, M.; McAllister, T.A. A direct qPCR screening approach to improve the efficiency of Mycoplasma bovis isolation in the frame of a broad surveillance study. J. Microbiol. Methods 2020, 169, 105805. [Google Scholar] [CrossRef] [PubMed]
- Appelt, S.; Aly, S.S.; Tonooka, K.; Glenn, K.; Xue, Z.; Lehenbauer, T.W.; Marco, M.L. Development and comparison of loop-mediated isothermal amplification and quantitative polymerase chain reaction assays for the detection of Mycoplasma bovis in milk. J. Dairy Sci. 2019, 102, 1985–1996. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.M.; House, J.K.; Hazelton, M.S.; Bosward, K.L.; Sheehy, P.A. Comparison of culture and a multiplex probe PCR for identifying Mycoplasma species in bovine milk, semen and swab samples. PLoS ONE 2017, 12, e0173422. [Google Scholar] [CrossRef] [PubMed]
- Pansri, P.; Katholm, J.; Krogh, K.M.; Aagaard, A.K.; Schmidt, L.M.B.; Kudirkiene, E.; Larsen, L.E.; Olsen, J.E. Evaluation of novel multiplex qPCR assays for diagnosis of pathogens associated with the bovine respiratory disease complex. Vet. J. 2020, 256, 105425. [Google Scholar] [CrossRef] [PubMed]
- Conrad, C.C.; Daher, R.K.; Stanford, K.; Amoako, K.K.; Boissinot, M.; Bergeron, M.G.; Alexander, T.; Cook, S.; Ralston, B.; Zaheer, R.; et al. A Sensitive and Accurate Recombinase Polymerase Amplification Assay for Detection of the Primary Bacterial Pathogens Causing Bovine Respiratory Disease. Front. Vet. Sci. 2020, 7, 208. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, A.; Imran, M.; Yaqub, T.; Tayyab, M.; Shehzad, W.; Mingala, C.N.; Chang, Y.-F. Development and validation of a loop-mediated isothermal amplification assay for the detection of Mycoplasma bovis in mastitic milk. Folia Microb. 2018, 63, 373–380. [Google Scholar] [CrossRef]
- Itoh, M.; Hirano, Y.; Yamakawa, K.; Yasutomi, I.; Kuramoto, K.; Furuok, M.; Yamada, K. Combination of procedure for ultra rapid extraction (PURE) and loop-mediated isothermal amplification (LAMP) for rapid detection of Mycoplasma bovis in milk. J. Vet. Med. Sci. 2020, 19–0695. [Google Scholar] [CrossRef]
- Zhao, G.; Hou, P.; Huan, Y.; He, C.; Wang, H.; He, H. Development of a recombinase polymerase amplification combined with a lateral flow dipstick assay for rapid detection of the Mycoplasma bovis. BMC Vet. Res. 2018, 14, 412. [Google Scholar] [CrossRef]
- Bokma, J.; Van Driessche, L.; Deprez, P.; Haesebrouck, F.; Vahl, M.; Weesendorp, E.; Deurenberg, R.H.; Pardon, P.; Boyen, P. Rapid identification of 1 Mycoplasma bovis from bovine bronchoalveolar lavage fluid with MALDI-TOF MS after enrichment procedure. J. Clin. Microbiol. 2020, 58, e00004-20. [Google Scholar] [CrossRef]
- Thurmond, M.C.; Tyler, J.W.; Luiz, D.M.; Holmberg, C.A.; Picanso, J.P. The effect of pre-enrichment on recovery of Streptococcus agalactiae, Staphylococcus aureus and mycoplasma from bovine milk. Epidemiol. Infect. 1989, 103, 465–474. [Google Scholar] [CrossRef]
- Punyapornwithaya, V.; Fox, L.K.; Gay, G.M.; Hancock, D.D.; Alldredge, J.R. Short communication: The effect of centrifugation and resuspension on the recovery of Mycoplasma species from milk. J. Dairy Sci. 2009, 92, 4444–4447. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fan, P.; Zhou, S.; Zhang, L. Loop-mediated isothermal amplification (LAMP): A novel rapid detection platform for pathogens. Microb. Pathog. 2017, 107, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Nunoya, T.; Omori, T.; Tomioka, H.; Umeda, F.; Suzuki, T.; Uetsuka, K. Intracellular Localization of Mycoplasma bovis in the Bronchiolar Epithelium of Experimentally Infected Calves. J. Comp. Path. 2020, 176, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Kleinschmidt, S.; Spergser, J.; Rosengarten, R.; Hewicker-Trautwein, M. Long-term survival of Mycoplasma bovis in necrotic lesions and in phagocytic cells as demonstrated by transmission and immunogold electron microscopy in lung tissue from experimentally infected calves. Vet. Microbiol. 2013, 162, 949–953. [Google Scholar] [CrossRef] [PubMed]
- Bell-Rogers, P.; Parker, L.; Cai, H.Y. Multi-locus sequence types of Mycoplasma bovis isolated from Ontario, Canada in the past three decades have a temporal distribution. J. Vet. Diagn. Invest. 2018, 30, 130–135. [Google Scholar] [CrossRef]
- Register, K.B.; Lysnyansky, I.; Jelinski, M.D.; Boatwright, W.D.; Waldner, M.; Bayles, D.O.; Pilo, P.; Alt, D.P. Comparison of two multilocus sequence typing schemes for Mycoplasma bovis and revision of the PubMLST reference method. J. Clin. Microbiol. 2020, 58, e00283-20. [Google Scholar] [CrossRef]
- Yair, Y.; Borovok, I.; Mikula, I.; Falk, R.; Fox, L.K.; Gophna, U.; Lysnyansky, I. Genomics-based epidemiology of bovine Mycoplasma bovis strains in Israel. BMC Genom. 2020, 21, 70. [Google Scholar] [CrossRef]
- Parker, A.M.; Shukla, A.; House, J.K.; Hazelton, M.S.; Bosward, K.L.; Kokotovic, B.; Sheehy, P.A. Genetic characterization of Australian Mycoplasma bovis isolates through whole genome sequencing analysis. Vet. Microbiol. 2016, 196I, 118–125. [Google Scholar] [CrossRef]
- Rasheed, M.A.; Qi, J.; Zhu, X.; Chenfei, H.; Menghwar, H.; Khan, F.A.; Guo, A. Comparative genomics of Mycoplasma bovis strains reveals that decreased virulence with increasing passages might correlate with potential virulence-related factors. Front. Cell. Infect. Microbiol. 2017, 7, 177. [Google Scholar] [CrossRef]
- Sachse, K.; Pfützner, H.; Hötzel, H.; Demuth, B.; Heller, M.; Berthold, E. Comparison of various diagnostic methods for the detection of Mycoplasma bovis. Rev. Sci. Tech.-Off. Int. Epiz. 1993, 12, 571–580. [Google Scholar] [CrossRef]
- Nielsen, P.K.; Petersen, M.B.; Nielsen, L.R.; Halasa, T.; Toft, N. Latent class analysis of bulk tank milk PCR and ELISA testing for herd level diagnosis of Mycoplasma bovis. Prev. Vet. Med. 2015, 121, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Gille, L.; Callens, J.; Supré, K.; Boyen, F.; Haesebrouck, F.; Van Driessche, L.; van Leenen, K.; Deprez, P.; Pardon, B. Use of breeding bull and absence of a calving pen as a risk factors for the presence of Mycoplasma bovis in dairy herds. J. Dairy Sci. 2018, 101, 8284–8290. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.-M.; Aspán, A.; Wisselink, H.J.; Smid, B.; Ridley, A.; Pelkonen, S.; Autio, T.; Lauritsen, K.T.; Kensø, J.; Gaurivaud, P.; et al. A European inter-laboratory trial to evaluate the performance of three serological methods for diagnosis of Mycoplasma bovis infection in cattle using latent class analysis. BMC Vet. Res. 2019, 15, 369. [Google Scholar] [CrossRef] [PubMed]
- Wawegama, N.K.; Browning, G.F.; Kanci, A.; Marenda, M.S.; Markham, F.F. Development of a Recombinant Protein-Based Enzyme-Linked Immunosorbent Assay for Diagnosis of Mycoplasma bovis Infection in Cattle. Clin. Vaccine Immunol. 2014, 21, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.B.; Wawegama, N.K.; Denwood, M.; Markham, P.F.; Browning, G.F.; Nielsen, L.R. Mycoplasma bovis antibody dynamics in naturally exposed dairy calves according to two diagnostic tests. BMC Vet. Res. 2018, 14, 258. [Google Scholar] [CrossRef] [PubMed]
- Wisselink, H.J.; Smid, B.; Plater, J.; Ridley, A.; Andersson, A.-M.; Aspán, A.; Pohjanvirta, T.; Vähänikkilä, N.; Larsen, H.; Høgberg, J.; et al. A European interlaboratory trial to evaluate the performance of different PCR methods for Mycoplasma bovis diagnosis. BMC Vet. Res. 2019, 15, 86. [Google Scholar] [CrossRef]
- Chazel, M.; Tardy, F.; Le Grand, D.; Calavas, D.; Poumarat, F. Mycoplasmoses of ruminants in France: Recent data from the national surveillance network. BMC Vet. Res. 2010, 6, 32. [Google Scholar] [CrossRef]
- Mackie, D.P.; Finley, D.; Brice, N.; Ball, H.J. Mixed mycoplasma mastitis outbreak in a dairy herd. Vet. Rec. 2000, 147, 335–336. [Google Scholar] [CrossRef]
- Jasper, D.E. Prevalence of mycoplasmal mastitis in the western states. Calif. Vet. 1980, 43, 24–26. [Google Scholar]
- McAuliffe, L.; Ellis, R.J.; Lawes, J.R.; Ayling, R.D.; Nicholas, R.A.J. 16S rDNA PCR and denaturing gradient gel electrophoresis; a single generic test for detecting and differentiating Mycoplasma species. J. Med. Microbiol. 2005, 54, 731–739. [Google Scholar] [CrossRef]
- Loy, J.D.; Leger, L.; Workman, A.M.; Clawson, M.L.; Bulut, E.; Wang, B. Development of a multiplex real-time PCR assay using two thermocycling platforms for detection of major bacterial pathogens associated with bovine respiratory disease complex from clinical samples. J. Vet. Diagn. Invest. 2018, 30, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Pereyre, S.; Tardy, F.; Renaudin, H.; Cauvin, E.; Del Prá Netto Machado, L.; Tricot, A.; Benoit, F.; Treilles, M.; Bébéar, C. Identification and Subtyping of Clinically Relevant Human and Ruminant Mycoplasmas by Use of Matrix-Assisted Laser Desorption Ionization–Time of Flight Mass Spectrometry J. Clin. Microbiol. 2013, 51, 3314–3323. [Google Scholar] [CrossRef] [PubMed]
- O’Farrell, K.; Dillon, P.; Mee, j.; Crosse, S.; Nnolan, M.; Byrne, N.; Reidy, M.; Flynn, F.; Condon, T. Strategy for restocking Moorepark after depopulation following BSE. Ir. Vet. J. 2001, 54, 70–75. [Google Scholar]
- Lysnyansky, I.; Mikula, I.; Ozeri, R.; Bellaiche, M.; Nicholas, R.A.J.; van Straten, M. Mycoplasma bovis Seroprevalence in Israeli Dairy Herds, Feedlots and Imported Cattle. Isr. J. Vet. Med. 2017, 72, 13–16. [Google Scholar]
- Stabel, J.R.; Hurd, S.; Calvente, L.; Rosenbusch, R.F. Destruction of Mycobacterium paratuberculosis, Salmonella spp., and Mycoplasma spp. in raw milk by a commercial on-farm high-temperature, short-time pasteurizer. J. Dairy Sci. 2004, 87, 2177–2183. [Google Scholar] [CrossRef]
- Gelsinger, S.L.; Heinrichs, A.J. Comparison of immune responses in calves fed heat-treated or unheated colostrum. J. Dairy Sci. 2017, 100, 4090–4101. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, H.; Gondaira, S.; Iwano, H.; Hirose, K.; Nakajima, K.; Kawai, K.; Hagiwara, K.; Tamura, Y.; Nagahata, H. Mycoplasma species isolated from intramammary infection of Japanese dairy cows. Vet. Rec. 2013, 172, 557. [Google Scholar] [CrossRef]
- Kauf, A.C.; Rosenbusch, R.F.; Paape, M.J.; Bannerman, D.D. Innate immune response to intramammary Mycoplasma bovis infection. J. Dairy Sci. 2007, 90, 3336–3348. [Google Scholar] [CrossRef]
- Punyapornwithaya, V.; Fox, L.K.; Hancock, D.D.; Gay, J.M.; Wenz, J.R.; Alldredge, J.R. Incidence and transmission of Mycoplasma bovis mastitis in Holstein dairy cows in a hospital pen: A case study. Prev. Vet. Med. 2011, 98, 74–78. [Google Scholar] [CrossRef]
- Murai, K.; Lehenbauer, T.W.; Champagne, J.D.; Glenn, K.; Aly, S.S. Cost-effectiveness of diagnostic strategies using quantitative real-time PCR and bacterial culture to identify contagious mastitis cases in large dairy herds. Prev. Vet. Med. 2014, 113, 522–535. [Google Scholar] [CrossRef]
- Mahdizadeh, S.; Sawford, K.; van Andel, M.; Browning, G.F. Efficacy of citric acid and sodium hypochlorite as disinfectants against Mycoplasma bovis. Vet. Microbiol. 2020, 243, 108630. [Google Scholar] [CrossRef] [PubMed]
- Boddie, R.L.; Owens, W.E.; Ray, C.H.; Nickerson, S.C.; Boddie, N.T. Germicidal activities of representatives of five different teat dip classes against three bovine mycoplasma species using a modified excised teat model. J. Dairy Sci. 2002, 85, 1909–1912. [Google Scholar] [CrossRef]
| Course of M. bovis Infection | Type of Research (Experimental/Survey) | Cattle Sector | Main Clinical Signs/Lesions/Subclinical | Methods Used for the Infection Confirmation/Presence | Reference |
|---|---|---|---|---|---|
| pneumonia | survey | beef | caseonecrotic bronchopneumonia; fibrinosuppurative bronchopneumonia | IHC; PCR | [33] |
| experimental | dairy-cross | nasal discharge; coughing; caseonecrotic pneumonia | ELISA for M. bovis antigen detection; IHC; ELISA for specific antibody detection | [19] | |
| mastitis | survey | dairy | clinical mastitis; subclinical mastitis | culture; real-time PCR; two different ELISAs for specific antibody detection (MilA IgG ELISA; BioX ELISA) | [23] |
| survey | clinical mastitis; subclinical mastitis | culture; PCR; ELISA for specific antibody detection | [13] | ||
| arthritis | survey | beef | arthritis; tenosynovitis | culture; passive hemagglutination test | [33] |
| experimental | dairy | joint swelling; lameness/fibrinosuppurative synovitis and tenosynovitis; thrombus presence | culture; indirect hemagglutination test | [34] | |
| otitis | survey | dairy | ear droop; otic exudate | ELISA for specific antibody detection; DGGE | [35] |
| survey | beef | ear droop; exudative otitis media; facial paralysis; occasionally nasal exudate; nystagmus, head tilt, ataxia/suppurative lesions in the middle ear; lung consolidation (most cases); cerebellar meningitis (some cases) | culturing; immune-peroxidase test; PCR; IHC; transmission electron microscopy | [7] | |
| kerato-conjunctivitis | survey | beef | “pink eye” signs | culture; RAPD; PCR-RFLP; DNA sequencing | [8] |
| brain disorders | survey | dairy | head tilt; central nervous system signs/purulent meningitis | ELISA for specific antibody detection | [36] |
| lethargy, blindness; teeth grinding/cerebral hemisphere necrosis | enrichment and capture ELISA | ||||
| endocarditis | survey | beef | no clinical signs; caseated lesions in the heart | culture; uvrC gene PCR; loop-mediated isothermal amplification assay; IHC | [10] |
| Source of Infection/Route of Infection Transmission | Type of Research (Experimental/Survey) | Cattle Sector | Number of Herd/Farms Tested | Methods Used for the Infection Confirmation/Detection | Reference |
|---|---|---|---|---|---|
| colostrum | survey | dairy | 4 | qPCR | [26] |
| survey | dairy, beef and dairy-mixed | 17 | real-time PCR | [27] | |
| milk | survey | dairy | 4 | qPCR; culturing; core-genome multilocus sequence typing (cgMLST) | [26] |
| semen | survey | dairy | 2 | culturing; real-time PCR; WGS; cgMLST analysis | [2] |
| air-borne | experimental | dairy-cross | not applicable | culturing; polC PCR; MilA IgG ELISA; post mortem examination; histopathological examination | [37] |
| intrauterine | survey | dairy | not described | culturing; IHC; ISH | [41] |
| Assay/Target | Samples | Limit of Detection | Sensitivity | Specificity | Reference |
|---|---|---|---|---|---|
| real-time PCR/uvrC | lung samples (n = 30); milk samples (n = 21) | 100 fg DNA; 40 genome copies/reaction; 250 CFU/mL | 103-fold more sensitive than conventional PCR | 100% (evaluated for 6 Mycoplasma spp. and 6 species of bacteria) | [45] |
| qPCR/uvrC | deep nasopharyngeal swabs (n = 208) | 1.61 × 102 CFU/mL | 100% | 87.27% | [46] |
| qPCR/gltX | milk samples from individual quarters (n = 9); bulk tank milk samples (n = 59) | 10–100 genome equivalents/reaction; 1 × 104–1 × 105 cells/mL | 100% | 94.4% (evaluated for 3 Mycoplasma spp.) | [47] |
| real-time multiplex PCR M. bovis/uvrC M. californicum/rpoB M. bovigenitalium/16S–23S rRNA | swab samples (n = 95); semen samples (n = 44); individual milk samples (n = 114); bulk tank milk samples (n = 221) | 1.3 × 102 –1.3 × 107 CFU/mL | not applicable | 100% (evaluated for 10 Mycoplasma spp. and 11 species of bacteria) | [48] |
| multiplex qPCR Pneumo 4B/M. bovis M. haemolytica P. multocida H. somni | tracheal aspirate samples (n = 176) | 10 genome copies; 1.1–3.3 log10 CFU/0.5 mL | 0.96 | 0.71 (evaluated for 6 Mycoplasma spp. and 66 species of bacteria) | [49] |
| multiplex qPCR Mastit 4/M. bovis Staphylococcus aureus Streptococcus agalactiae Streptococcus uberis | milk samples | - | - | - | [26] |
| real-time multiplex RPA/M. bovis/uvrC M. haemolytica/nmaA | deep nasopharyngeal swabs (n = 100) | 40 genome copies/reaction | - | 98.0 (evaluated for 10 Mycoplasma spp. and 35 species of bacteria) | [50] |
| real-time multiplex PCR PathoProof™ Mastitis Major 4.2/M. bovis Staphylococcus aureus Streptococcus agalactiae Streptococcus uberis | milk samples | - | - | - | [13] |
| real-time PCR VetMAX™ M. bovis | tissue samples, bronchoalveolar lavage fluid samples, synovial fluid, milk samples | 10 genome copies/reaction | 100% | 100% (evaluated for 50 other bacteria species, including M. agalactiae, Streptococcus uberis and Streptococcus dysgalactiae | [27] |
| LAMP/uvrC, gyrB | milk samples from 95 dairy farms | 5 × 101 CFU/mL | 96.8%–100% | 94.7%–100% (evaluated for 2 Mycoplasma spp. and 4 species of bacteria) | [51] |
| LAMP/oppD | milk samples from individual quarters (n = 9); bulk tank milk samples (n = 59) | 10 genome equivalents/reaction; 1 × 104 cells/mL | 87.5% | 82.4% (evaluated for 3 Mycoplasma spp.) | [47] |
| PURE-LAMP not applicable | bulk tank milk samples (n = 12); mature milk samples (n = 73); colostrum/transitional milk samples (n = 74); mastitis milk samples (n = 122) | >102 CFU/mL of milk | 57.0%–97.0% | 100% (evaluated for 5 Mycoplasma spp.) | [52] |
| RPA-LFD/uvrC, oppD-oppF | nasal swab samples (n = 288); fresh lung samples (n = 80); joint fluid samples (n = 32); bulk tank milk samples (n = 42) | 20 genome copies/reaction | 99.0% | 95.61% (evaluated for 10 Mycoplasma spp. and 13 species of bacteria) | [53] |
| MALDI-TOF MS | culture-enriched bronchoalveolar lavage fluid samples (n = 104) | not applicable | 86.6% | 86.4% | [54] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dudek, K.; Nicholas, R.A.J.; Szacawa, E.; Bednarek, D. Mycoplasma bovis Infections—Occurrence, Diagnosis and Control. Pathogens 2020, 9, 640. https://doi.org/10.3390/pathogens9080640
Dudek K, Nicholas RAJ, Szacawa E, Bednarek D. Mycoplasma bovis Infections—Occurrence, Diagnosis and Control. Pathogens. 2020; 9(8):640. https://doi.org/10.3390/pathogens9080640
Chicago/Turabian StyleDudek, Katarzyna, Robin A. J. Nicholas, Ewelina Szacawa, and Dariusz Bednarek. 2020. "Mycoplasma bovis Infections—Occurrence, Diagnosis and Control" Pathogens 9, no. 8: 640. https://doi.org/10.3390/pathogens9080640
APA StyleDudek, K., Nicholas, R. A. J., Szacawa, E., & Bednarek, D. (2020). Mycoplasma bovis Infections—Occurrence, Diagnosis and Control. Pathogens, 9(8), 640. https://doi.org/10.3390/pathogens9080640






